Abstract
Radiofrequency ablation may prevent or treat atrial and ventricular arrhythmias. Since some of these arrhythmias are associated with sudden cardiac death, it has been hypothesized that ablation may prevent sudden death in certain cases. We performed a literature search to better understand under which circumstances ablation may prevent sudden death and found little randomized data demonstrating the long-term effects of ablation. Current literature shows that ablation clearly prevents symptoms of arrhythmia and may reduce the incidence of sudden cardiac death in select patients, although data does not indicate improved mortality. Ongoing clinical trials are needed to better define the role of ablation in preventing sudden cardiac death.
Keywords: radiofrequency ablation, sudden cardiac death

M. Atoui, M.D.
Introduction
The estimated incidence of sudden cardiac death (SCD) in the United States ranges from 180,000 to more than 450,000 per year,1 and it is responsible for more than 50% of all U.S. deaths related to cardiovascular disease. The principal mechanism of SCD is ventricular fibrillation, and studies suggest that atrial fibrillation may be associated with an increased risk of sudden cardiac death in certain populations.
In 1982, direct current (DC) catheter ablation was first described to perform atrioventricular junction ablation on patients with atrial fibrillation.2 Although DC ablation was effective, it was replaced with a safer method using a radiofrequency (RF) generator.3 Within a few years, RF ablation was used for treatment of multiple arrhythmias4–8 and has been proven to reduce arrhythmia symptoms in many patients. Although it has been hypothesized that RF ablation (RFA) may prevent sudden death in certain cases, there is little literature demonstrating the effects of RFA on mortality. The following review discusses the current theories underlying the mechanisms of certain arrhythmias and reviews literature regarding the role of RFA in prevention of sudden cardiac death.
Ablation of Accessory Pathway to Prevent SCD
Atrioventricular reentry tachycardia (AVRT) can be sustained by an accessory pathway—a reentry circuit in which one end is the accessory pathway and the other is the AV node. Antegrade conduction down the AV node leads to a narrow complex (orthodromic) tachycardia (e.g., atrial fibrillation), whereas antegrade conduction down the accessory pathway, which is less common, leads to a wide complex (antidromic) tachycardia (e.g., ventricular tachycardia). Up to 30% of patients with accessory pathways can have atrial fibrillation (AF), whereas the incidence of AF is only 3% to 4% in the general population.9–12 If AF is rapidly conducted down the accessory pathway, it can lead to ventricular fibrillation (VF). Thus, the speed of conduction in the accessory pathway is critical.
In patients with accessory pathways and symptoms, ablation is generally recommended. This is especially the case if they present with VF,13 as patients with Wolff-Parkinson-White (WPW) syndrome have a post-ablation mortality similar to the general population.14 Although there is little controversy over ablation of accessory pathways associated with symptomatic tachycardia, there is significant debate about ablation of accessory pathways in asymptomatic patients. The incidence of SCD in these patients is low, but in young patients it can be especially devastating. General practice seems to be aggressive ablation in patients under age 35 since their pathways are often faster. Moreover, the decision to ablate may be different in asymptomatic patients with high-risk occupations.
The WPW registry was an 8-year prospective study of either symptomatic or asymptomatic WPW patients referred for evaluation of ablation. Among 2,169 enrolled patients, 1,001 (550 asymptomatic) did not undergo RF ablation (RFA) while 1,168 (206 asymptomatic) did undergo ablation. In the non-RFA group, VF occurred in 1.5% of patients and was associated with a short accessory pathway antegrade refractory period (P < 0.001) and atrioventricular reentrant tachycardia that triggered atrial fibrillation (P < 0.001) without symptoms. Ablation was successful in 98.5% of these patients, and no further malignant arrhythmias or VFs occurred over the 8-year follow-up. Time-dependent receiver-operating characteristic curves for predicting VF pinpointed an optimal anterograde effective refractory period of the accessory pathway cutoff of 240 ms.15 This study was criticized as it mixed the symptomatic and asymptomatic groups. In addition, there was no mortality in the 15 patients who had VF, 13 of them in the asymptomatic group.16 Nevertheless, this prospective registry confirmed the safety and success of performing RF ablation in WPW patients, as there was no further recurrence of malignant arrhythmia causing SCD.
Ventricular Tachycardia in Structural Heart Disease
Patients with structural heart disease and low left ventricular ejection fraction (LVEF) are at increased risk of VT and SCD. Typically these patients receive an implantable cardioverter defibrillator (ICD) for the primary and secondary prevention of SCD.17–19 However, the wide use of ICDs to treat heart failure has increased the number of patients exposed to inappropriate ICD activations, or shocks, which in turn correlates with worse outcomes including pain, anxiety, depression, post-traumatic stress disorder, myocardial injury, and increased mortality.20,21 Antiarrhythmic drugs can be used to treat these patients but are not always effective and have multiple side effects.
To reduce the incidence of these shocks, ablation may be performed. Two studies suggest that ablation may also be associated with a trend toward reduced mortality. In the first retrospective study, Bunch et al. compared three groups of patients: those with a history of ICD shocks who did not receive VT ablation, those with a history of ICD shocks who did receive VT ablation, and those with ICDs who had no history of ICD shocks. After long-term follow-up (> 5 years), patients who were treated with VT ablation after an ICD shock had lower mortality and heart failure hospitalization compared to medically treated ICD-shocked patients. Moreover, this improvement was independent of the ejection fraction of both ischemic and nonischemic cardiomyopathy.22 This mortality benefit may potentially be explained by (1) the reduction or prevention of additional ICD therapies that contribute to morbidity and mortality, (2) minimizing exposure to long-term antiarrhythmic therapies, and (3) improved efficacy of antiarrhythmic therapies.20,22 More recently, Tung and Shivkumar also showed reduced mortality in patients undergoing successful ablation.23
Currently, there is no randomized data of VT ablation that shows reductions in mortality. However, ablation clearly reduces VT recurrence in multiple trials. In SMASH-VT, a prospective, randomized, unblinded, multicenter trial, VT substrate-based ablation was performed on patients with ischemic heart disease during ICD implantation. The patients receiving ablation and ICD showed reduced ICD shocks at 2-year follow-up and a trend toward reduced mortality compared to those receiving ICD without ablation (9% vs 17%, P = 0.24).24 In the VTACH trial, another prospective, open, randomized, controlled multicenter trial, early ablation during ICD implantation for secondary prevention in patients with ischemic cardiomyopathy and stable VT showed reduced ICD shocks. Estimates for survival free from VT/VF at 2 years were 47% for the group receiving ICD and ablation versus 29% for those receiving ICD only. Although there was no change in morality, the study showed a reduction in hospital admission due to cardiovascular reasons in the ablation group.25 Two current trials, STAR VT and PARTITA, are evaluating the use of early VT ablation in preventing sudden death and could change future management in these patients. In summary, ablation in patients with VT and structural heart disease likely reduces VT burden and ICD shocks, but data does not yet exist to show that ablation prevents sudden death.
Ventricular Fibrillation in Normal Hearts
Ventricular fibrillation is the main mechanism of sudden cardiac death. Idiopathic VF (IVF) in the absence of structural heart disease or surface electrocardiographic abnormalities accounts for 5% to 10% of survivors of out-of hospital cardiac arrest.26–28 This population has a median age of 40 to 45 years. A disease is considered “idiopathic” when present knowledge has yet to identify the underlying cause. Therefore, a diagnosis of IVF is based on the exclusion of currently known structural and primary electrical heart diseases after the patient has undergone a complete noninvasive, invasive, and genetic workup. Furthermore, this diagnosis is to be reiterated with both the patient and any family member who may show an inheritable condition, such as catecholaminergic polymorphic ventricular tachycardia, long-QT syndrome, Brugada syndrome, arrhythmogenic right ventricular cardiomyopathy, hypertrophic cardiomyopathy, and familial hypercholesterolemia.29 In a study by Gray et al., an early repolarization pattern in the inferolateral leads was associated with an increased risk for sudden cardiac death.30
Mapping during VF has shown that fibrillation is perpetuated by reentrant or spiral waves, while other data suggest the role of specific sources triggering the arrhythmia.31,32 In numerous case reports and at least one multicenter study, IVF was triggered by a premature ventricular complex (PVC) originating in the Purkinje fibers.32 Haïssaguerre et al. found that these triggers were involved in IVF induction and were originating from the right or left Purkinje system.31,32 They were able to localize the source by mapping the earliest electrogram relative to the onset of the ectopic QRS complex. An initial sharp potential (10 ms in duration) preceding by 15 ms the larger and slower ventricular electrogram during sinus rhythm represented a peripheral Purkinje component, whereas longer intervals indicated proximal Purkinje fascicle activation. After ablation, and at 24-months average follow-up, 24 patients (89%) had no recurrence of VF and were not taking antiarrhythmic drugs.32
To evaluate the long-term effect, Haïssaguerre et al. studied 38 consecutive patients who underwent ablation of primary IVF initiated by short-coupled ventricular premature beat (VPB).33 Patients were on a median of two antiarrhythmic drugs. All patients had experienced at least one documented VF, and 87% had experienced two VF episodes in the preceding year. During a median postprocedural follow-up of approximately 5 years, 7 (18%) of 38 patients experienced some recurrence of VF at a median of 4 months. Of those seven patients, five underwent repeated ablation without further VF recurrences (Figure 1). Ablation significantly reduced the number of significant events (confirmed VF or aborted sudden death) from 4 before ablation to 0 after ablation (P < 0.01). One finding was the typical and consistent left bundle-branch block pattern with left superior axis for VPBs originating in the right Purkinje system. However, VPBs originating in the left Purkinje system produced more variable 12-lead electrocardiogram patterns, reflecting the more complex and extended Purkinje arborization on the left (Figure 2). Adverse outcome was only associated with the presence of bundle-branch block in the targeted ventricle before ablation as the result of mechanical trauma “bumping,” possibly obscuring ipsilateral Purkinje potentials during sinus rhythm and thus complicating subsequent ablation.33 Although this study did not eliminate the need for ICD implantation, it did decrease the recurrence of VF and further ICD shocks. Further prospective studies would need to be done to determine if this decrease in VT recurrence and ICD shocks translated to a morbidity and mortality benefit in this young population.34
Figure 1.

Twelve-lead electrocardiogram during radiofrequency ablation at the right Purkinje system. The start of ablation (arrow) typically resulted in ventricular tachycardia followed by sinus rhythm, as shown in this example. Reprinted from Journal of the American College of Cardiology, 54(6), Knecht S, Sacher F, Wright M, Hocini M, Nogami A, Arentz T, et al., Long term follow up of idiopathic ventricular fibrillation ablation a multicenter study, 522–528, Copyright 2009, with permission from Elsevier.
Figure 2.

Twelve-lead electrocardiograms (left) and their corresponding location in the anteroposterior fluoroscopic view (right, red asterisks). The origin of ventricular premature beat (VPB) triggering ventricular fibrillation (VF) was the left Purkinje either at the posterior (A) or the anterior (B) insertion, the right Purkinje (C), and the right ventricular outflow track (D). Related fluoroscopic views with a decapolar catheter inserted in the left ventricle (E and F), an ablation catheter inserted in the right ventricle (G), and a quadripolar catheter inserted at the His position (E to G). Ventricular premature beat originating in the left Purkinje system (A and B and related anteroposterior fluoroscopic view) produce more variable 12-lead electrocardiogram (ECG) patterns, reflecting the more complex and extended Purkinje arborization on the left. VPBs originating in the right Purkinje system (C and related anteroposterior fluoroscopic view) typically have a left bundle-branch block pattern with left superior axis. Ventricular premature beat originating from the right ventricular outflow track (D) have the classical aspect with a left bundle-branch block pattern and an inferior axis. Reprinted from Journal of the American College of Cardiology, 54(6), Knecht S, Sacher F, Wright M, Hocini M, Nogami A, Arentz T, et al., Long term follow up of idiopathic ventricular fibrillation ablation a multicenter study, 522–528, Copyright 2009, with permission from Elsevier.
Ventricular Fibrillation in Structural Heart Disease
In patients with structural heart disease, the usual presentation of electrical storm, which necessitates ablation, is VT that degenerates into VF.35 Electrical storm in patients with structural heart disease has various etiologies (e.g., coronary artery disease, idiopathic dilated cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy). In ischemic heart disease, VF can potentially occur at an early stage soon after an acute myocardial infarction (AMI) or at a later stage after scar formation. During an AMI, ischemia of the His–Purkinje system may increase susceptibility to VF. Most cases are managed with beta blockers, amiodarone, and revascularization; however, there are reported cases and small series about the efficacy of catheter ablation in treating electrical storms that are caused by PVC's. These PVCs seem to originate with surviving Purkinjee fibers in the scar border, and ablation of these PVCs has been shown to reduce and control VF episodes.36–38
Marrouche et al. reported on 29 patients with ischemic cardiomyopathy and VF triggered by PVCs. Eight had VF refractory to medical management and underwent mapping, and five of them had PVCs during the procedure and underwent ablation. The researchers noted that the PVCs always originated in the scar border and were proceeded by Purkinje-like potentials (PLP). After 10 months follow-up, no patients had VF storm although one patient had VT and one had a single VF episode. While it should be noted that this is a select population of patients with PVCs clearly triggering VF, it is nonetheless a promising therapy to reduce VF burden.39
In another series, 11 consecutive patients with VF storm and nonischemic cardiomyopathy were given antiarrhythmic drugs and/or heart failure management.40 Five of them had recurrent VF and underwent electrophysiology study (EPS) and catheter ablation. Three-dimensional electroanatomical mapping revealed posterior wall scar in four of the five patients. Radiofrequency ablation targeting PLP around the scar borders was successfully performed in four of them. The patient without PLP did not undergo ablation and was the only one with recurrent VF, which required ICD shocks at 1-year follow-up. In patients with VF and dilated cardiomyopathy, left ventricular posterior wall scar near the mitral annulus appears to be a common finding, and ablation targeting PLP along the scar border zone seems to prevent VF recurrence in these patients.40 Other rare cases of successful ablation were reported in VF patients refractory to medical treatment.39
Brugada Syndrome
Brugada syndrome (BrS) attracted a lot of attention since its introduction as a clinical entity in 1992 due to its association with sudden cardiac death in otherwise healthy young adults and, less frequently, infants and children.41,42 It was noted to be triggered by fever in some. It is a familial disease with an autosomal dominance pattern of inheritance, and more than 13 genes were identified, the first involving loss of function of INa (SCN5A).43 The pathophysiology of BrS is controversial, but it is generally felt that heterogeneity—especially in the right ventricular outflow tract (RVOT) epicardium—is responsible for VF. It was also noted that VF episodes were preceded by VPB's mostly originating from the RVOT area, thus making it the target for ablation.44,45
ICD implantation is the effective treatment to prevent SCD in BrS patients. Quinidine was shown to prevent induction of VF in patients with multiple ICD shocks.46,47 However, many challenges exist with ICD implantation (e.g., young population, not affordable in some countries) and quinidine treatment (e.g., difficult for lifetime compliance due to side effects). In addition, BrS patients receive frequent ICD shocks, adding to their complication rates.48
Radiofrequency ablation in an experimental model of BrS suggested higher success in eliminating VT.49 Other small case series reported the efficacy of using radiofrequency ablation targeting RVOT myocardial triggers to treat electrical storm or reduce VF recurrence.50,51 Interestingly, the baseline ECG normalizes in some of these patients.
Nademanee et al. reported the first series of nine symptomatic patients with type I BrS and multiple VFs requiring ICD shocks. They performed endocardial and epicardial mapping (Figures 3, 4). Delayed depolarization rather than repolarization was thought to be the underlying mechanism, as all nine patients had unique abnormal low voltage, long duration of local activation, and fractionated late potentials that seemed to cluster exclusively in anterior RVOT epicardium. Ablation at these sites rendered VT/VF noninducible in 78% and normalized the ECG in 89%. At 20 months, no recurrence of VF was found, although one patient was on amiodarone.52 Although this study had many limitations, it presented the possibility of finding the arrhythmogenic substrate for catheter ablation and, subsequently, anticipating better outcomes in this young population.
Figure 3.

Left lateral view of the right ventricular outflow tract (RVOT) displays the difference in ventricular electrograms between the endocardial (ENDO) and epicardial (EPI) site of the anterior RVOT of the same patient. The left and right insets display bipolar and unipolar electrograms recorded from the epicardium and endocardium from the same site of the RVOT, respectively. Bi-DIST indicates bipolar distal; Bi-PROX, bipolar proximal; Uni-DIST, unipolar distal; and Uni-PROX, unipolar proximal. Reprinted from Circulation, 123(12), Nademanee K, Veerakul G, Chandanamattha P, Chaothawee L, Ariyachaipanich A, Jirasirirojanakorn K, et al., Prevention of ventricular fibrillation episodes in Brugada syndrome by catheter ablation over the anterior right ventricular outflow tract epicardium, 1270–79, Copyright 2011, with permission from Wolters Kluwer Health Inc. Promotional and commercial use of the material in print, digital or mobile device format is prohibited without the permission from the publisher Wolters Kluwer Health.
Figure 4.
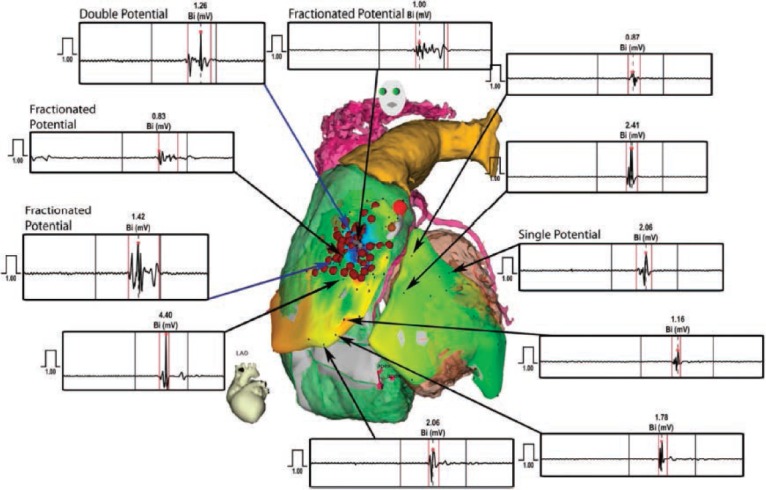
Comparison of ventricular electrograms recorded from different sites in both the left ventricle (LV) and right ventricle (RV). Reprinted from Circulation, 123(12), Nademanee K, Veerakul G, Chandanamattha P, Chaothawee L, Ariyachaipanich A, Jirasirirojanakorn K, et al., Prevention of ventricular fibrillation episodes in Brugada syndrome by catheter ablation over the anterior right ventricular outflow tract epicardium, 1270–79, Copyright 2011, with permission from Wolters Kluwer Health Inc. Promotional and commercial use of the material in print, digital or mobile device format is prohibited without the permission from the publisher Wolters Kluwer Health.
Figure 5.

The 12-lead ECGs from the same patient, recorded at the beginning and end of the procedure, show normalization of the Brugada ECG pattern during ablation. Reprinted from Circulation, 123(12), Nademanee K, Veerakul G, Chandanamattha P, Chaothawee L, Ariyachaipanich A, Jirasirirojanakorn K, et al., Prevention of ventricular fibrillation episodes in Brugada syndrome by catheter ablation over the anterior right ventricular outflow tract epicardium, 1270–79, Copyright 2011, with permission from Wolters Kluwer Health Inc. Promotional and commercial use of the material in print, digital or mobile device format is prohibited without the permission from the publisher Wolters Kluwer Health.
Summary
Radiofrequency ablation has been shown to reduce symptoms and improve quality of life in many arrhythmias. In select situations such as rapid-conduction AF down an accessory pathway and VF triggered by PVCs, ablation likely reduces sudden cardiac death. In other situations, ablation clearly reduces symptoms of arrhythmia, but there is little data suggesting that mortality is improved. Future studies may determine the benefit of ablation to prevent sudden death.
Conflict of Interest Disclosure: Dr. Lakkireddy is a consultant to Biosense Webster Inc. and St. Jude Medical, Inc., and Dr. Mahapatra is an employee of St. Jude Medical, Inc.
References
- 1.Kong MH, Fonarow GC, Peterson ED et al. Systematic review of the incidence of sudden cardiac death in the United States. J Am Coll Cardiol. 2011 Feb 15;57(7):794–801. doi: 10.1016/j.jacc.2010.09.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Scheinman MM, Morady F, Hess DS, Gonzalez R. Catheter-induced ablation of the atrioventricular junction to control refractory supraventricular arrhythmias. JAMA. 1982 Aug 20;248(7):851–5. [PubMed] [Google Scholar]
- 3.Huang SK, Bharati S, Graham AR, Lev M, Marcus FI, Odell RC. Closed chest catheter desiccation of the atrioventricular junction using radiofrequency energy--a new method of catheter ablation. J Am Coll Cardiol. 1987 Feb;9(2):349–58. doi: 10.1016/s0735-1097(87)80388-1. [DOI] [PubMed] [Google Scholar]
- 4.Feld GK, Fleck RP, Chen PS et al. Radiofrequency catheter ablation for the treatment of human type 1 atrial flutter. Identification of a critical zone in the reentrant circuit by endocardial mapping techniques. Circulation. 1992 Oct;86(4):1233–40. doi: 10.1161/01.cir.86.4.1233. [DOI] [PubMed] [Google Scholar]
- 5.Haissaguerre M, Gaita F, Fischer B et al. Elimination of atrioventricular nodal reentrant tachycardia using discrete slow potentials to guide application of radiofrequency energy. Circulation. 1992 Jun;85(6):2162–75. doi: 10.1161/01.cir.85.6.2162. [DOI] [PubMed] [Google Scholar]
- 6.Jackman WM, Wang XZ, Friday KJ et al. Catheter ablation of accessory atrioventricular pathways (Wolff-Parkinson-White syndrome) by radiofrequency current. N Engl J Med. 1991 Jun 6;324(23):1605–11. doi: 10.1056/NEJM199106063242301. [DOI] [PubMed] [Google Scholar]
- 7.Calkins H, Sousa J, el-Atassi R et al. Diagnosis and cure of the Wolff-Parkinson-White syndrome or paroxysmal supraventricular tachycardias during a single electrophysiologic test. N Engl J Med. 1991 Jun 6;324(23):1612–8. doi: 10.1056/NEJM199106063242302. [DOI] [PubMed] [Google Scholar]
- 8.Stevenson WG, Khan H, Sager P et al. Identification of reentry circuit sites during catheter mapping and radiofrequency ablation of ventricular tachycardia late after myocardial infarction. Circulation. 1993 Oct;88(4 Pt 1):1647–70. doi: 10.1161/01.cir.88.4.1647. [DOI] [PubMed] [Google Scholar]
- 9.Berkman NL, Lamb LE. The Wolff-Parkinson-White electrocardiogram. A follow-up study of five to twenty-eight years. N Engl J Med. 1968 Feb 29;278(9):492–4. doi: 10.1056/NEJM196802292780906. [DOI] [PubMed] [Google Scholar]
- 10.Haissaguerre M, Fischer B, Labbé T et al. Frequency of recurrent atrial fibrillation after catheter ablation of overt accessory pathways. Am J Cardiol. 1992 Feb 15;69(5):493–7. doi: 10.1016/0002-9149(92)90992-8. [DOI] [PubMed] [Google Scholar]
- 11.Miyasaka Y, Barnes ME, Gersh BJ et al. Secular trends in incidence of atrial fibrillation in Olmsted County, Minnesota, 1980 to 2000, and implications on the projections for future prevalence. Circulation. 2006 Jul 11;114(2):119–25. doi: 10.1161/CIRCULATIONAHA.105.595140. [DOI] [PubMed] [Google Scholar]
- 12.Sharma AD, Klein GJ, Guiraudon GM, Milstein S. Atrial fibrillation in patients with Wolff-Parkinson-White syndrome: incidence after surgical ablation of the accessory pathway. Circulation. 1985 Jul;72(1):161–9. doi: 10.1161/01.cir.72.1.161. [DOI] [PubMed] [Google Scholar]
- 13.Blomström-Lundqvist C, Scheinman MM, Aliot EM. ACC/AHA/ESC guidelines for the management of patients with supraventricular arrhythmias--executive summary. a report of the American college of cardiology/American heart association task force on practice guidelines and the European society of cardiology committee for practice guidelines (writing committee to develop guidelines for the management of patients with supraventricular arrhythmias) developed in collaboration with NASPE-Heart Rhythm Society. J Am Coll Cardiol. 2003 Oct 15;42(8):1493–531. doi: 10.1016/j.jacc.2003.08.013. et al.; European Society of Cardiology Committee, NASPE-Heart Rhythm Society. [DOI] [PubMed] [Google Scholar]
- 14.Borregaard R, Lukac P, Gerdes C et al. Radiofrequency ablation of accessory pathways in patients with the Wolff-Parkinson-White syndrome: the long-term mortality and risk of atrial fibrillation. Europace. 2015 Jan;17(1):117–22. doi: 10.1093/europace/euu176. [DOI] [PubMed] [Google Scholar]
- 15.Pappone C, Vicedomini G, Manguso F et al. Wolff-Parkinson-White syndrome in the era of catheter ablation: insights from a registry study of 2169 patients. Circulation. 2014 Sep 2;130(10):811–9. doi: 10.1161/CIRCULATIONAHA.114.011154. [DOI] [PubMed] [Google Scholar]
- 16.Obeyesekere MN, Klein GJ. The asymptomatic Wolff-Parkinson-White patient: time to be more proactive? Circulation. 2014 Sep 2;130(10):805–7. doi: 10.1161/CIRCULATIONAHA.114.011856. [DOI] [PubMed] [Google Scholar]
- 17.Bardy GH, Lee KL, Mark DB. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005 Jan 20;352(3):225–37. doi: 10.1056/NEJMoa043399. et al.; Sudden Cardiac Death in Heart Failure Trial (SCD-HeFT) Investigators. [DOI] [PubMed] [Google Scholar]
- 18.Buxton AE, Lee KL, Fisher JD, Josephson ME, Prystowsky EN, Hafley G. A randomized study of the prevention of sudden death in patients with coronary artery disease. Multicenter Unsustained Tachycardia Trial Investigators. N Engl J Med. 1999 Dec 16;341(25):1882–90. doi: 10.1056/NEJM199912163412503. [DOI] [PubMed] [Google Scholar]
- 19.Moss AJ, Zareba W, Hall WJ. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002 Mar 21;346(12):877–83. doi: 10.1056/NEJMoa013474. et al.; Multicenter Automatic Defibrillator Implantation Trial II Investigators. [DOI] [PubMed] [Google Scholar]
- 20.Moss AJ, Schuger C, Beck CA. Reduction in inappropriate therapy and mortality through ICD programming. N Engl J Med. 2012 Dec 13;367(24):2275–83. doi: 10.1056/NEJMoa1211107. et al; MADIT-RIT Trial Investigators. [DOI] [PubMed] [Google Scholar]
- 21.Sears SF, Hauf JD, Kirian K, Hazelton G, Conti JB. Posttraumatic stress and the implantable cardioverter-defibrillator patient: what the electrophysiologist needs to know. Circ Arrhythm Electrophysiol. 2011 Apr;4(2):242–50. doi: 10.1161/CIRCEP.110.957670. [DOI] [PubMed] [Google Scholar]
- 22.Bunch TJ, Weiss JP, Crandall BG et al. Patients treated with catheter ablation for ventricular tachycardia after an ICD shock have lower long-term rates of death and heart failure hospitalization than do patients treated with medical management only. Heart Rhythm. 2014 Apr;11(4):533–40. doi: 10.1016/j.hrthm.2013.12.014. [DOI] [PubMed] [Google Scholar]
- 23.Tung R, Shivkumar K. Integrated care for management of ventricular arrhythmias: can a specialized unit and catheter ablation improve mortality? Circulation. 2013 Apr 2;127(13):1354–6. doi: 10.1161/CIRCULATIONAHA.113.001775. [DOI] [PubMed] [Google Scholar]
- 24.Reddy VY, Reynolds MR, Neuzil P et al. Prophylactic catheter ablation for the prevention of defibrillator therapy. N Engl J Med. 2007 Dec 27;357(26):2657–65. doi: 10.1056/NEJMoa065457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kuck KH, Schaumann A, Eckardt L et al. Catheter ablation of stable ventricular tachycardia before defibrillator implantation in patients with coronary heart disease (VTACH): a multicentre randomised controlled trial. Lancet. 2010 Jan 2;375(9708):319–40. doi: 10.1016/S0140-6736(09)61755-4. [DOI] [PubMed] [Google Scholar]
- 26.Survivors of out-of-hospital cardiac arrest with apparently normal heart. Need for definition and standardized clinical evaluation. Consensus Statement of the Joint Steering Committees of the Unexplained Cardiac Arrest Registry of Europe and of the Idiopathic Ventricular Fibrillation Registry of the United States. Circulation. 1997 Jan 7;95(1):265–72. doi: 10.1161/01.cir.95.1.265. [DOI] [PubMed] [Google Scholar]
- 27.Belhassen B, Viskin S. Idiopathic ventricular tachycardia and fibrillation. J Cardiovasc Electrophysiol. 1993 Jun;4(3):356–68. doi: 10.1111/j.1540-8167.1993.tb01236.x. [DOI] [PubMed] [Google Scholar]
- 28.Meissner MD, Lehmann MH, Steinman RT et al. Ventricular fibrillation in patients without significant structural heart disease: a multicenter experience with implantable cardioverter-defibrillator therapy. J Am Coll Cardiol. 1993 May;21(6):1406–12. doi: 10.1016/0735-1097(93)90317-t. [DOI] [PubMed] [Google Scholar]
- 29.Tan HL, Hofman N, van Langen IM, van der Wal AC, Wilde AA. Sudden unexplained death: heritability and diagnostic yield of cardiological and genetic examination in surviving relatives. Circulation. 2005 Jul 12;112(2):207–13. doi: 10.1161/CIRCULATIONAHA.104.522581. [DOI] [PubMed] [Google Scholar]
- 30.Gray RA, Jalife J, Panfilov AV et al. Mechanisms of cardiac fibrillation. Science. 1995 Nov 17;270(5239):1222–3. author reply 1224–5. [PubMed] [Google Scholar]
- 31.Haïssaguerre M, Shah DC, Jaïs P et al. Role of Purkinje conducting system in triggering of idiopathic ventricular fibrillation. Lancet. 2002 Feb 23;359(9307):677–8. doi: 10.1016/S0140-6736(02)07807-8. [DOI] [PubMed] [Google Scholar]
- 32.Haïssaguerre M, Shoda M, Jaïs P et al. Mapping and ablation of idiopathic ventricular fibrillation. Circulation. 2002 Aug 20;106(8):962–7. doi: 10.1161/01.cir.0000027564.55739.b1. [DOI] [PubMed] [Google Scholar]
- 33.Knecht S, Sacher F, Wright M et al. Long-term follow-up of idiopathic ventricular fibrillation ablation: a multicenter study. J Am Coll Cardiol. 2009 Aug 4;54(6):522–8. doi: 10.1016/j.jacc.2009.03.065. [DOI] [PubMed] [Google Scholar]
- 34.Verma A, Kilicaslan F, Marrouche NF et al. Prevalence, predictors, and mortality significance of the causative arrhythmia in patients with electrical storm. J Cardiovasc Electrophysiol. 2004 Nov;15(11):1265–70. doi: 10.1046/j.1540-8167.2004.04352.x. [DOI] [PubMed] [Google Scholar]
- 35.Kozeluhova M, Peichl P, Cihak R et al. Catheter ablation of electrical storm in patients with structural heart disease. Europace. 2011 Jan;13(1):109–13. doi: 10.1093/europace/euq364. [DOI] [PubMed] [Google Scholar]
- 36.Bänsch D, Oyang F, Antz M et al. Successful catheter ablation of electrical storm after myocardial infarction. Circulation. 2003 Dec 16;108(24):3011–6. doi: 10.1161/01.CIR.0000103701.30662.5C. [DOI] [PubMed] [Google Scholar]
- 37.Enjoji Y, Mizobuchi M, Muranishi H et al. Catheter ablation of fatal ventricular tachyarrhythmias storm in acute coronary syndrome--role of Purkinje fiber network. J Interv Card Electrophysiol. 2009 Dec;26(3):207–15. doi: 10.1007/s10840-009-9394-7. [DOI] [PubMed] [Google Scholar]
- 38.Tan VH, Yap J, Hsu LF, Liew R. Catheter ablation of ventricular fibrillation triggers and electrical storm. Europace. 2012 Dec;14(12):1687–95. doi: 10.1093/europace/eus050. [DOI] [PubMed] [Google Scholar]
- 39.Marrouche NF, Verma A, Wazni O et al. Mode of initiation and ablation of ventricular fibrillation storms in patients with ischemic cardiomyopathy. J Am Coll Cardiol. 2004 May 5;43(9):1715–20. doi: 10.1016/j.jacc.2004.03.004. [DOI] [PubMed] [Google Scholar]
- 40.Sinha AM, Schmidt M, Marschang H et al. Role of left ventricular scar and Purkinje-like potentials during mapping and ablation of ventricular fibrillation in dilated cardiomyopathy. Pacing Clin Electrophysiol. 2009 Mar;32(3):286–90. doi: 10.1111/j.1540-8159.2008.02233.x. [DOI] [PubMed] [Google Scholar]
- 41.Antzelevitch C, Brugada P, Borggrefe M et al. Brugada syndrome: report of the second consensus conference: endorsed by the Heart Rhythm Society and the European Heart Rhythm Association. Circulation. 2005 Feb 8;111(5):659–70. doi: 10.1161/01.CIR.0000152479.54298.51. [DOI] [PubMed] [Google Scholar]
- 42.Brugada P, Brugada J. Right bundle branch block, persistent ST segment elevation and sudden cardiac death: a distinct clinical and electrocardiographic syndrome. A multicenter report. J Am Coll Cardiol. 1992 Nov 15;20(6):1391–6. doi: 10.1016/0735-1097(92)90253-j. [DOI] [PubMed] [Google Scholar]
- 43.Antzelevitch C, Brugada P, Brugada J, Brugada R, Towbin JA, Nademanee K. Brugada syndrome: 1992–2002: a historical perspective. J Am Coll Cardiol. 2003 May 21;41(10):1665–71. doi: 10.1016/s0735-1097(03)00310-3. [DOI] [PubMed] [Google Scholar]
- 44.Haïssaguerre M, Extramiana F, Hocini M et al. Mapping and ablation of ventricular fibrillation associated with long-QT and Brugada syndromes. Circulation. 2003 Aug 26;108(8):925–8. doi: 10.1161/01.CIR.0000088781.99943.95. [DOI] [PubMed] [Google Scholar]
- 45.Kakishita M, Kurita T, Matsuo K et al. Mode of onset of ventricular fibrillation in patients with Brugada syndrome detected by implantable cardioverter defibrillator therapy. J Am Coll Cardiol. 2000 Nov 1;36(5):1646–53. doi: 10.1016/s0735-1097(00)00932-3. [DOI] [PubMed] [Google Scholar]
- 46.Belhassen B, Glick A, Viskin S. Efficacy of quinidine in high-risk patients with Brugada syndrome. Circulation. 2004 Sep 28;110(13):1731–7. doi: 10.1161/01.CIR.0000143159.30585.90. [DOI] [PubMed] [Google Scholar]
- 47.Schweizer PA, Becker R, Katus HA, Thomas D. Successful acute and long-term management of electrical storm in Brugada syndrome using orciprenaline and quinine/quinidine. Clin Res Cardiol. 2010 Jul;99(7):467–70. doi: 10.1007/s00392-010-0145-7. [DOI] [PubMed] [Google Scholar]
- 48.Sarkozy A, Boussy T, Kourgiannides G et al. Long-term follow-up of primary prophylactic implantable cardioverter-defibrillator therapy in Brugada syndrome. Eur Heart J. 2007 Feb;28(3):334–44. doi: 10.1093/eurheartj/ehl450. [DOI] [PubMed] [Google Scholar]
- 49.Morita H, Zipes DP, Morita ST, Lopshire JC, Wu J. Epicardial ablation eliminates ventricular arrhythmias in an experimental model of Brugada syndrome. Heart Rhythm. 2009 May;6(5):665–71. doi: 10.1016/j.hrthm.2009.01.007. [DOI] [PubMed] [Google Scholar]
- 50.Maury P, Hocini M, Haïssaguerre M. Electrical storms in Brugada syndrome: review of pharmacologic and ablative therapeutic options. Indian Pacing Electrophysiol J. 2005 Jan 1;5(1):25–34. [PMC free article] [PubMed] [Google Scholar]
- 51.Nakagawa E, Takagi M, Tatsumi H, Yoshiyama M. Successful radiofrequency catheter ablation for electrical storm of ventricular fibrillation in a patient with Brugada syndrome. Circ J. 2008 Jun;72(6):1025–9. doi: 10.1253/circj.72.1025. [DOI] [PubMed] [Google Scholar]
- 52.Nademanee K, Veerakul G, Chandanamattha P et al. Prevention of ventricular fibrillation episodes in Brugada syndrome by catheter ablation over the anterior right ventricular outflow tract epicardium. Circulation. 2011 Mar 29;123(12):1270–9. doi: 10.1161/CIRCULATIONAHA.110.972612. [DOI] [PubMed] [Google Scholar]


