Abstract
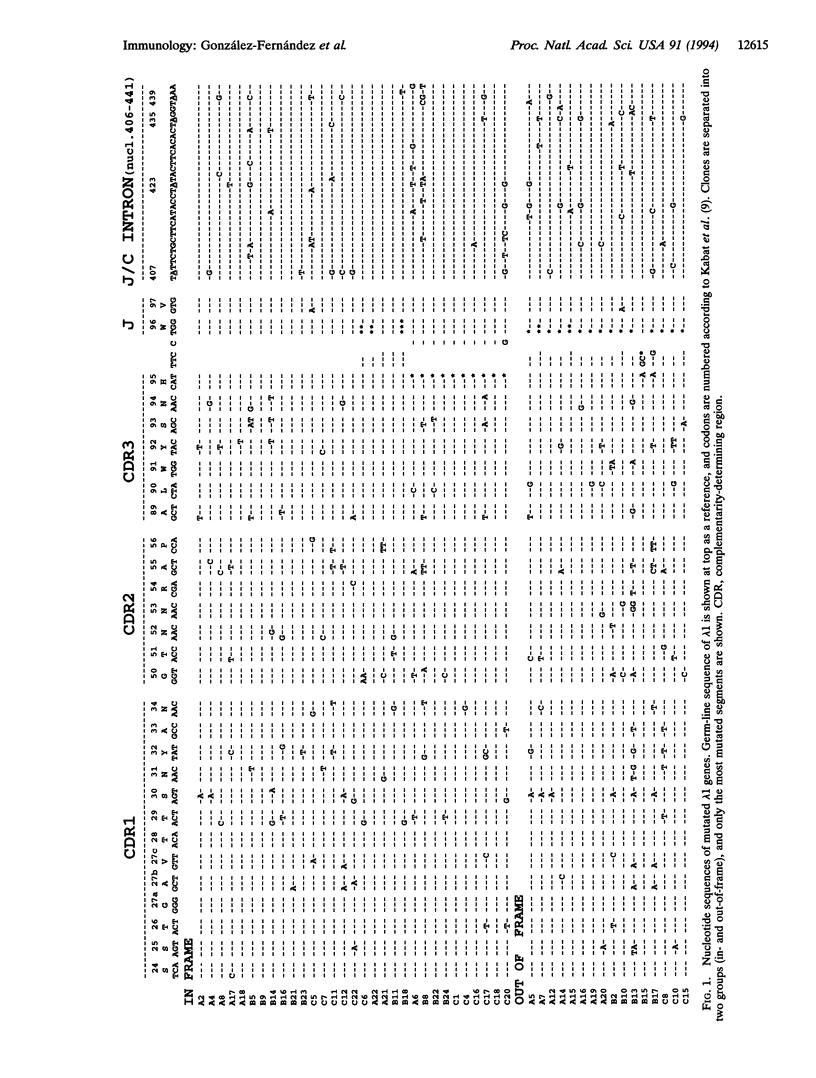
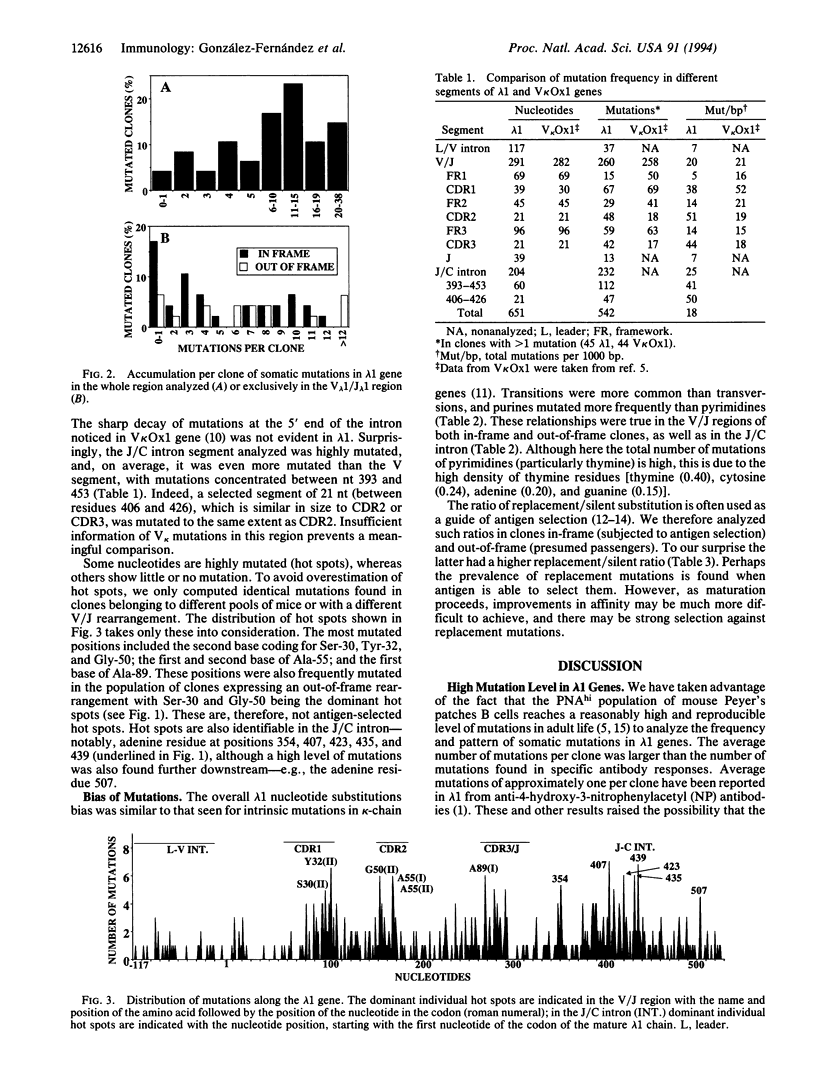
The rate and nature of hypermutation of immunoglobulin genes are of prime importance in the affinity maturation of antibodies. Although a considerable body of information has been gathered for kappa light chains, there is much less data for lambda chains. We have derived a large data base of somatic mutants of mouse lambda 1 light chains from Peyer's patches germinal center B cells. The endogenous lambda 1 genes mutate at a rate comparable to that previously found for a kappa transgene (V kappa ox1). There are intrinsic hot spots of mutation common to both in-frame and out-of-frame rearrangements; these hot spots cluster in hypermutating domains. In contrast to the pattern seen for V kappa Ox1, the hot spot clusters are found not only in complementarity-determining region (CDR)1 but also in CDR2 and CDR3; mutations also cluster in the joining/constant region intron. The differences between the pattern of mutations in V kappa Ox1 and lambda 1 light chains are discussed.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berek C., Berger A., Apel M. Maturation of the immune response in germinal centers. Cell. 1991 Dec 20;67(6):1121–1129. doi: 10.1016/0092-8674(91)90289-b. [DOI] [PubMed] [Google Scholar]
- Berek C., Milstein C. Mutation drift and repertoire shift in the maturation of the immune response. Immunol Rev. 1987 Apr;96:23–41. doi: 10.1111/j.1600-065x.1987.tb00507.x. [DOI] [PubMed] [Google Scholar]
- Betz A. G., Neuberger M. S., Milstein C. Discriminating intrinsic and antigen-selected mutational hotspots in immunoglobulin V genes. Immunol Today. 1993 Aug;14(8):405–411. doi: 10.1016/0167-5699(93)90144-a. [DOI] [PubMed] [Google Scholar]
- Brown M., Stenzel-Poore M., Stevens S., Kondoleon S. K., Ng J., Bächinger H. P., Rittenberg M. B. Immunologic memory to phosphocholine keyhole limpet hemocyanin. Recurrent mutations in the lambda 1 light chain increase affinity for antigen. J Immunol. 1992 Jan 15;148(2):339–346. [PubMed] [Google Scholar]
- Cumano A., Rajewsky K. Clonal recruitment and somatic mutation in the generation of immunological memory to the hapten NP. EMBO J. 1986 Oct;5(10):2459–2468. doi: 10.1002/j.1460-2075.1986.tb04522.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisen H. N., Reilly E. B. Lambda chains and genes in inbred mice. Annu Rev Immunol. 1985;3:337–365. doi: 10.1146/annurev.iy.03.040185.002005. [DOI] [PubMed] [Google Scholar]
- Ford J. E., McHeyzer-Williams M. G., Lieber M. R. Analysis of individual immunoglobulin lambda light chain genes amplified from single cells is inconsistent with variable region gene conversion in germinal-center B cell somatic mutation. Eur J Immunol. 1994 Aug;24(8):1816–1822. doi: 10.1002/eji.1830240814. [DOI] [PubMed] [Google Scholar]
- Ford J. E., McHeyzer-Williams M. G., Lieber M. R. Chimeric molecules created by gene amplification interfere with the analysis of somatic hypermutation of murine immunoglobulin genes. Gene. 1994 May 16;142(2):279–283. doi: 10.1016/0378-1119(94)90275-5. [DOI] [PubMed] [Google Scholar]
- Golding G. B., Gearhart P. J., Glickman B. W. Patterns of somatic mutations in immunoglobulin variable genes. Genetics. 1987 Jan;115(1):169–176. doi: 10.1093/genetics/115.1.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Fernández A., Milstein C. Analysis of somatic hypermutation in mouse Peyer's patches using immunoglobulin kappa light-chain transgenes. Proc Natl Acad Sci U S A. 1993 Nov 1;90(21):9862–9866. doi: 10.1073/pnas.90.21.9862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawkins R. E., Russell S. J., Winter G. Selection of phage antibodies by binding affinity. Mimicking affinity maturation. J Mol Biol. 1992 Aug 5;226(3):889–896. doi: 10.1016/0022-2836(92)90639-2. [DOI] [PubMed] [Google Scholar]
- Jacob J., Kelsoe G., Rajewsky K., Weiss U. Intraclonal generation of antibody mutants in germinal centres. Nature. 1991 Dec 5;354(6352):389–392. doi: 10.1038/354389a0. [DOI] [PubMed] [Google Scholar]
- Kolchanov N. A., Solovyov V. V., Rogozin I. B. Peculiarities of immunoglobulin gene structures as a basis for somatic mutation emergence. FEBS Lett. 1987 Apr 6;214(1):87–91. doi: 10.1016/0014-5793(87)80018-2. [DOI] [PubMed] [Google Scholar]
- Küppers R., Zhao M., Hansmann M. L., Rajewsky K. Tracing B cell development in human germinal centres by molecular analysis of single cells picked from histological sections. EMBO J. 1993 Dec 15;12(13):4955–4967. doi: 10.1002/j.1460-2075.1993.tb06189.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McHeyzer-Williams M. G., Nossal G. J., Lalor P. A. Molecular characterization of single memory B cells. Nature. 1991 Apr 11;350(6318):502–505. doi: 10.1038/350502a0. [DOI] [PubMed] [Google Scholar]
- Milstein C., Even J., Berek C. Molecular events during the onset and maturation of the antibody response. Biochem Soc Symp. 1986;51:173–182. [PubMed] [Google Scholar]
- Motoyama N., Okada H., Azuma T. Somatic mutation in constant regions of mouse lambda 1 light chains. Proc Natl Acad Sci U S A. 1991 Sep 15;88(18):7933–7937. doi: 10.1073/pnas.88.18.7933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rada C., González-Fernández A., Jarvis J. M., Milstein C. The 5' boundary of somatic hypermutation in a V kappa gene is in the leader intron. Eur J Immunol. 1994 Jun;24(6):1453–1457. doi: 10.1002/eji.1830240632. [DOI] [PubMed] [Google Scholar]
- Rada C., Gupta S. K., Gherardi E., Milstein C. Mutation and selection during the secondary response to 2-phenyloxazolone. Proc Natl Acad Sci U S A. 1991 Jul 1;88(13):5508–5512. doi: 10.1073/pnas.88.13.5508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reth M., Hämmerling G. J., Rajewsky K. Analysis of the repertoire of anti-NP antibodies in C57BL/6 mice by cell fusion. I. Characterization of antibody families in the primary and hyperimmune response. Eur J Immunol. 1978 Jun;8(6):393–400. doi: 10.1002/eji.1830080605. [DOI] [PubMed] [Google Scholar]
- Steele E. J., Rothenfluh H. S., Both G. W. Defining the nucleic acid substrate for somatic hypermutation. Immunol Cell Biol. 1992 Apr;70(Pt 2):129–144. doi: 10.1038/icb.1992.18. [DOI] [PubMed] [Google Scholar]
- Tomlinson I. M., Walter G., Marks J. D., Llewelyn M. B., Winter G. The repertoire of human germline VH sequences reveals about fifty groups of VH segments with different hypervariable loops. J Mol Biol. 1992 Oct 5;227(3):776–798. doi: 10.1016/0022-2836(92)90223-7. [DOI] [PubMed] [Google Scholar]
- Yelton D. E., Desaymard C., Scharff M. D. Use of monoclonal anti-mouse immunoglobulin to detect mouse antibodies. Hybridoma. 1981;1(1):5–11. doi: 10.1089/hyb.1.1981.1.5. [DOI] [PubMed] [Google Scholar]



