Abstract
The fish pathogen Photobacterium damselae subsp. piscicida produces the siderophore piscibactin. A gene cluster that resembles the Yersinia high-pathogenicity island (HPI) encodes piscibactin biosynthesis. Here, we report that this HPI-like cluster is part of a hitherto-uncharacterized 68-kb plasmid dubbed pPHDP70. This plasmid lacks homologs of genes that mediate conjugation, but we found that it could be transferred at low frequencies from P. damselae subsp. piscicida to a mollusk pathogenic Vibrio alginolyticus strain and to other Gram-negative bacteria, likely dependent on the conjugative functions of the coresident plasmid pPHDP60. Following its conjugative transfer, pPHDP70 restored the capacity of a vibrioferrin mutant of V. alginolyticus to grow under low-iron conditions, and piscibactin became detectable in its supernatant. Thus, pPHDP70 appears to harbor all the genes required for piscibactin biosynthesis and transport. P. damselae subsp. piscicida strains cured of pPHDP70 no longer produced piscibactin, had impaired growth under iron-limited conditions, and exhibited markedly decreased virulence in fish. Collectively, our findings highlight the importance of pPHDP70, with its capacity for piscibactin-mediated iron acquisition, in the virulence of P. damselae subsp. piscicida. Horizontal transmission of this plasmid-borne piscibactin synthesis gene cluster in the marine environment may facilitate the emergence of new pathogens.
INTRODUCTION
The emergence of bacterial pathogens in aquaculture systems has been a subject of great interest to microbiologists. Widespread horizontal gene transfer in marine environments and the close interactions among bacterial species in the cramped confines of aquaculture facilities make mobile-element acquisition a driving force in the evolutionary processes promoting the emergence of new virulence strategies (1–3). Photobacterium damselae subsp. piscicida, the causative agent of fish photobacteriosis, is one of the most devastating pathogens in marine aquaculture (4). This bacterium, a member of the family Vibrionaceae, shares a species designation with P. damselae subsp. damselae, a bacterium that causes vibriosis in a variety of marine animals and is also the causative agent of infections in humans (5). The two P. damselae subspecies seem to cause disease through distinct mechanisms, and the differential presence of plasmids has contributed significantly to their divergence in virulence gene content (6, 7).
A major virulence factor in P. damselae subsp. piscicida is the metalloprotease A-B toxin AIP56, which induces apoptosis in fish macrophages and neutrophils. Interestingly, AIP56 is encoded in a 10-kb plasmid dubbed pPHDP10 (6). P. damselae subsp. piscicida spreads systemically following infection, suggesting that the pathogen faces an iron-limiting environment within the host. In this regard, it has been reported that P. damselae subsp. piscicida can obtain iron from human transferrin by a mechanism involving the production of a siderophore (8). Later, a gene cluster (the irp cluster) was found to be required for the biosynthesis of a phenolate-like siderophore in P. damselae subsp. piscicida (9). Initially, it was shown that insertional inactivation of the putative biosynthetic gene irp1 abolished the biosynthesis of the P. damselae subsp. piscicida siderophore. Recently, the chemical structure of this siderophore, which was dubbed piscibactin, was determined and demonstrated to be similar but not identical to that of yersiniabactin (10). Indeed, the irp cluster was found to have a structure similar to that of the Yersinia high-pathogenicity island (HPI), a chromosome-borne high-pathogenicity island that encodes the biosynthesis and transport of the siderophore yersiniabactin in pathogenic Yersinia species (11). First identified in Yersinia spp., the HPI has been subsequently found in several Enterobacteriaceae genera (12, 13).
Mobile genetic elements appear to have played a major role in shaping the genome of P. damselae subsp. piscicida. In addition to pPHDP10, encoding AIP56 toxin, a conjugative plasmid, pPHDP60, which encodes a putative type II secretion system has been recently described (14), and an SXT/R391 family integrating conjugative element dubbed ICEPdaSpa1 was also identified in this subspecies (15). The structural features of the irp cluster and its similarities to the Yersinia HPI initially suggested that the piscibactin biosynthesis genes might constitute a pathogenicity island (PAI) (9). In the present study, we report that the irp cluster is part of a novel mobilizable 68-kb plasmid here dubbed pPHDP70. pPHDP70 appears to be sufficient for piscibactin synthesis, and it proved to be required for P. damselae subsp. piscicida siderophore-mediated iron acquisition and virulence in fish. Notably, pPHDP70 provided piscibactin synthesis and utilization capabilities to a mollusk-pathogenic strain of Vibrio alginolyticus following its conjugative transfer aided by the coresident plasmid pPHDP60.
MATERIALS AND METHODS
Bacteria, plasmids, oligonucleotides, and media.
The bacterial strains and plasmids used in this study are listed in Table 1. Marine bacteria were routinely grown at 25°C in either tryptic soy agar or tryptic soy broth (Difco) supplemented with 1% NaCl (TSA-1 and TSB-1, respectively). Escherichia coli strains were grown at 37°C in Luria-Bertani (LB) broth or LB agar. Antibiotics were used at the following final concentrations: kanamycin (Km) at 25 μg ml−1, ampicillin sodium salt (Ap) at 50 μg ml−1, chloramphenicol (Cm) at 20 μg ml−1, rifampin (Rif) at 50 μg ml−1, and nalidixic acid (Nal) at 40 μg ml−1. All stocks were filter sterilized and stored at −20°C. The iron chelator 2,2′-dipyridyl (Sigma) was prepared at 10 mM in water. The oligonucleotides used in this study are listed in Table 2.
TABLE 1.
Strains and plasmids used in this study
| Strain or plasmid | Description or relevant characteristicsa | Source or reference |
|---|---|---|
| Strains | ||
| P. damselae subsp. piscicida | ||
| DI21 | Highly virulent isolate, harbors pPHDP70, pPHDP60, pPHDP10 | Seabream, Spain |
| DI21-Rif | DI21 derivative, spontaneous Rifr mutant | 9 |
| CS31 | DI21 with pSJR52 inserted in pPHDP70; irp1 disruptant | 9 |
| AR84 | CS31 cured of plasmid pPHDP70 | This study |
| Vibrio alginolyticus | ||
| TA15 | Isolate from carpet shell clams (Ruditapes decussatus) | 31 |
| AR13 | TA15 ΔpvsA, deficient in vibrioferrin siderophore production | This study |
| AR43 | AR13 transconjugant for pPHDP70::Km | This study |
| AR45 | AR13 transconjugant for pPHDP70::pSJR52 | This study |
| Aeromonas salmonicida RSP74.1 | Isolated from turbot (Scophthalmus maximus) | 51 |
| Vibrio anguillarum RV22 | Isolated from turbot (Scophthalmus maximus) | 52 |
| Escherichia coli | ||
| DH5α | Cloning strain | Laboratory stock |
| S17-1 λ pir | RP4-2(Km::Tn7, Tc::Mu-1) pro-82 λpir recA1 endA1 thiE1 hsdR17 creC510 | Laboratory stock |
| XL1-blue MR | Δ(mcrA)183 Δ(mcrCB-hsdSMR-mrr)173 endA1 supE44 thi-1 recA1 gyrA96 relA1 lac | Stratagene |
| MG1655 | E. coli K-12, F− | 53 |
| BI533 | MG1655 Nalr | 54 |
| MC1061 | F− araD139 Δ(ara-leu)7696 galE15 galK16 Δ(lac)X74 rpsL hsdR2(rK− mK+) mcrA mcrB1 | 55 |
| Plasmids | ||
| pKD4 | Template for Kmr gene | 21 |
| pNidKan | Suicide vector, derived from pCVD442, Kmr | 19 |
| pSJR52 | 4,672-bp EcoRI-XbaI fragment including the 3′ end of irp2 and the 5′ end of irp1, cloned into pNidKan | 9 |
| pPHDP70 | 68-kb native plasmid of strain DI21, contains piscibactin gene cluster | This study |
| pPHDP70::Km | pPHDP70 with a Kmr gene from pKD4 inserted between orf19 and orf20 | This study |
| pPHDP70::pSJR52 | pPHDP70 with pSJR52 inserted: disrupts irp1 gene | 9 |
| pPHDP60 | Conjugative 68-kb native plasmid of strain DI21 | 14 |
| pPHDP60::Cm | pPHDP60 with a Cmr gene inserted into orf48 | 14 |
Apr, ampicillin resistance; Kmr, kanamycin resistance; Rifr, rifampin resistance; Cmr, chloramphenicol resistance; Nalr, nalidixic acid resistance.
TABLE 2.
Oligonucleotides used in this study
| Purpose and oligonucleotide | Sequence | bp amplified |
|---|---|---|
| entD-orf21 | ||
| HPI-103 | ATTGCGACGCATGTTGTATG | 2,134 |
| vibD-3′ | CATGCCTCCAAGGTTATCAC | |
| parAB | ||
| pCW046-5′-2 | ATTGCGACGCATGTTGTATG | 1,359 |
| pCW046-3′-1 | TATTCACGGTGATGACTAAC | |
| orf28 (putative carbonic anhydrase) | ||
| mig5-5′-1 | ACGTTTGTTTTAGCAACAGC | 459 |
| mig5-3′-1 | TGTCACCTAATGATGTGGTG | |
| orf48 (fimbrial usher) | ||
| pCW042-5′4 | CATTATGTCAGCATCTTCCG | 522 |
| usher-1 | TACTCGTGTAGTTATATCCG | |
| repA | ||
| RepA-F | GAAAGCTCAGATGCCATGCC | 2,407 |
| pCW044-5-1 | GAAAGCTCAGATGCCATGCC | |
| Amplification of pPHDP70 iterons | ||
| ITERON_F | GAAAGCTCAGATGCCATGCC | 1,429 |
| ITERON_R | GAAAGCTCAGATGCCATGCC | |
| Kmr marker amplification from pKD4 | ||
| Kan-1 | GCGGATCCGGCTTACATGGCGATAGCTA | 1,115 |
| Kan-2 | GCGGATCCCCCCGCGCTGGAGGATCATC | |
| Flanking DNA between orf19 and orf20 for Kmr marker insertion in pPHDP70 | ||
| HPmut1 | GCTCTAGACTATCTAACGCTTCGGGGAA | 3,314 |
| HPmut2 | GCGGATCCGCATTTCAGGACGATACACC | |
| HPmut3 | GCGGATCCGTTTACACATTGAGGACGAA | 3,314 |
| HPmut4 | GCGAATTCTGGCGCGGCATTTAAGGCCA | |
| Amplification of the Kmr marker and flanking pPHDP70 DNA | ||
| HP-Sac-A | GCGAGCTCCTATCTAACGCTTCGGGGAA | 7,771 |
| HP-Sac-B | GCGAGCTCTGGCGCGGCATTTAAGGCCA | |
| V. alginolyticus pvsA deletion | ||
| TA15-pvsAMut1 | GCGGGCCCCGTTGACGACCACAGTACGA | 1,088 |
| TA15-pvsAMut2 | GCGAATTCATGGGTCAGCGCACGTGCAC | |
| TA15-pvsAMut3 | GCGAATTCCGGATTAAAGTGACTATCAC | 1,015 |
| TA15-pvsAMut4 | GCTCTAGAAATACGCCCGTTGACCTTAT |
Cosmid library construction, DNA sequencing, and annotation.
Genomic DNA of P. damselae subsp. piscicida DI-21 was purified using the genome DNA kit from Q-BIOgene, partially digested with Sau3AI, and ligated into SuperCos 1 cosmid vector (Stratagene). The ligated products were packaged into bacteriophage lambda particles (Gigapack III Gold packaging extract; Stratagene, USA) and introduced into E. coli XL1-Blue MR cells. Nucleotide sequences were determined using the CEQ DTCS-Quick Start kit (Beckman Coulter) and a CEQ 8000 capillary DNA sequencer (Beckman Coulter). Plasmid annotation was carried out with RAST server (16), and a plasmid graphical map was generated with CGView Server (17). G+C content analyses were conducted with the Artemis Comparison Tool (18). Protein domains were searched using the Pfam database (http://pfam.sanger.ac.uk/).
Construction of a V. alginolyticus pvsA mutant.
The pvsA gene of V. alginolyticus TA15, encoding a biosynthetic enzyme of the siderophore vibrioferrin, was deleted by allelic exchange with the Kmr suicide vector pNidkan as previously described (19). A nonpolar deletion was constructed by using PCR amplification of the amino- and carboxy-terminal fragments of pvsA, which, when fused together, would result in an in-frame deletion. The plasmid construction containing the deleted allele was mated from E. coli S17-1λpir into Vibrio alginolyticus TA15, selecting for Ap (TA15 is naturally Apr) and Km resistance for plasmid integration and subsequently for sucrose resistance (15% [wt/vol]) for a second recombination event.
Construction of a kanamycin cassette-marked version of pPHDP70, conjugative assays, and plasmid curing.
We constructed a labeled pPHDP70 by inserting a Kmr cassette through a double crossover between orf19 and orf20 (Fig. 1) by allelic exchange using the Apr suicide vector pKEK229 (20). In brief, ca. 3,300-bp sequences upstream and downstream of the intergenic region that separates orf19 and orf20 were PCR amplified and ligated to a Km resistance gene amplified from plasmid pKD4 (21). The three DNA fragments were ligated into the suicide vector pKEK229. The resulting plasmid, pAJR8, was mobilized from E. coli S17-1λpir into P. damselae subsp. piscicida DI21-Rif. Suicide plasmid cointegration into pPHDP70 was selected on agar plates containing Ap, Km, and Rif. A second recombination event causing suicide plasmid loss and allelic exchange was selected by Km and sucrose (15% [wt/vol]) resistance. The resulting strain, DI21 pPHDP70::Km, was used as a donor in conjugation experiments carried out as previously described (15) using different bacterial species as recipients (Tables 1 and 3). In brief, exponentially growing cells of donor and recipient strains were mixed at a 1:1 ratio, concentrated 10 times by centrifugation, spotted on TSA plates prepared with seawater, and incubated at 25°C for 24 h. After incubation, mating spots were collected and resuspended to make serial dilutions. Transconjugants were selected at 25°C on TSA-1 plates containing kanamycin and the respective antibiotics to select for the recipient strain. The plasmid transfer frequency was expressed as the number of transconjugants per recipient cell. In order to obtain a strain cured of plasmid pPHDP70, we used a pAJR8 first-crossover strain and further selected for Kms and sucrose-resistant colonies that lost the pPHDP70-pAJR8 cointegrate. Plasmid DNA from transconjugants was extracted using the plasmid Midikit from Qiagen following the manufacturer's recommendations and subjected to restriction enzyme digestion with NotI.
FIG 1.
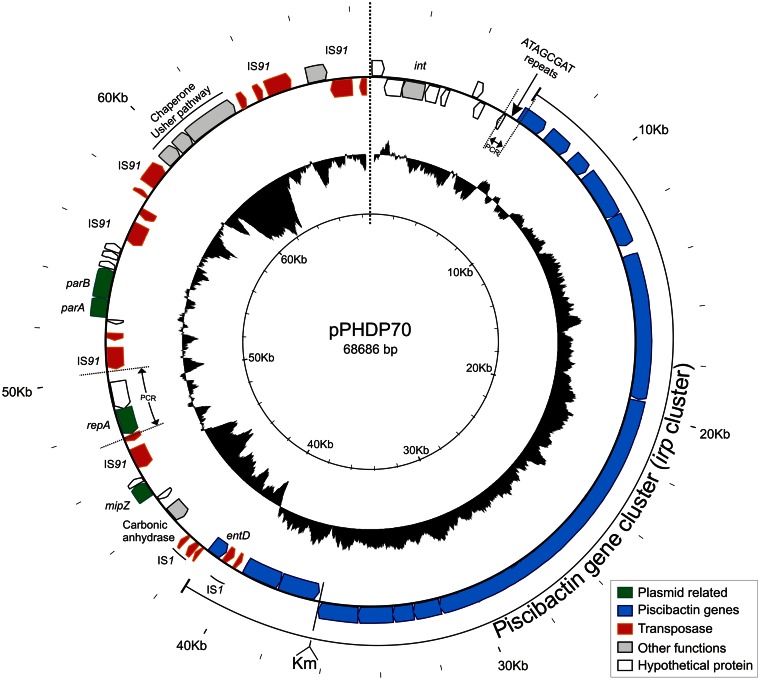
Circular genome of pPHDP70. ORFs are shown as arrows in the outer track. The position of the putative iterons (ATAGCGAT repeats) is indicated. Km denotes the point where a Kmr gene was inserted to mark pPHDP70. Iteron and repA regions are bracketed by discontinuous lines and labeled “PCR” to denote the PCR-amplified fragments that were used to assess potential replication determinants of the plasmid. The deviation of the G+C content from the mean (43.1%), calculated with a window of 500 bp, is indicated in the middle track. The inside track denotes positions in kilobases. Note that IS91 is present in six copies and accounts for a 10% of the total plasmid sequence. The piscibactin siderophore gene cluster is highlighted.
TABLE 3.
Frequency of pPHDP70 transmissibility between strains
| Donor | Recipient | Frequencya |
|---|---|---|
| P. damselae subsp. piscicida DI21(pPHDP70::Km) | E. coli MC1061 | 1.5 × 10−7 (pPHDP70) |
| V. alginolyticus TA15 | 1.4 × 10−7 (pPHDP70) | |
| A. salmonicida RSP74.1 | 1.2 × 10−7 (pPHDP70) | |
| V. anguillarum RV22 | 1.1 × 10−7 (pPHDP70) | |
| E. coli MC1061(pPHDP70::Km) | E. coli BI533b | ND (pPHDP70) |
| E. coli MC1061(pPHDP70::Km, pPHDP60::Cm) | E. coli BI533 | 1.6 × 10−7 (pPHDP70) |
| E. coli BI533 | ND (pPHDP70 and pPHDP60) | |
| E. coli BI533 | 5 × 10−5 (pPHDP60) |
The frequency of conjugal transfer was calculated as the number of transconjugants per recipient cell. The results are the means of at least 3 independent assays; standard errors are ≤20%. The plasmid whose transmissibility was studied is indicated in parentheses (either pPHDP70 alone, pPHDP60 alone, or the simultaneous transmissibility of pPHDP70 and pPHDP60). ND, not detected.
BI533 is Nalr E. coli MG1655.
Siderophore production assay and growth under iron-limiting conditions.
To evaluate the ability of P. damselae subsp. piscicida and V. alginolyticus strains to grow under iron-restricted conditions, overnight cultures in TSB-1 of the parental and mutant strains were adjusted to an optical density at 600 nm (OD600) of 0.5 and diluted 1:15 in CM9 minimal medium (19) in the presence of the iron chelator 2,2′-dipyridyl (Sigma) at concentrations of 60 μM (for P. damselae subsp. piscicida) and 16 μM (for V. alginolyticus). Growth under iron-sufficient conditions was measured by culturing the same strains in CM9 minimal medium supplemented with 10 μM ferric sulfate. Cultures were incubated at 25°C with shaking at 150 rpm, and growth (OD600) was measured after 16 h. Siderophore production (A630) was measured in 16-h culture supernatants using the chrome azurol-S (CAS) liquid assay (22). Assays were carried out in triplicate, and mean values with standard deviations are depicted.
Analytical detection of piscibactin-Ga(III) complex.
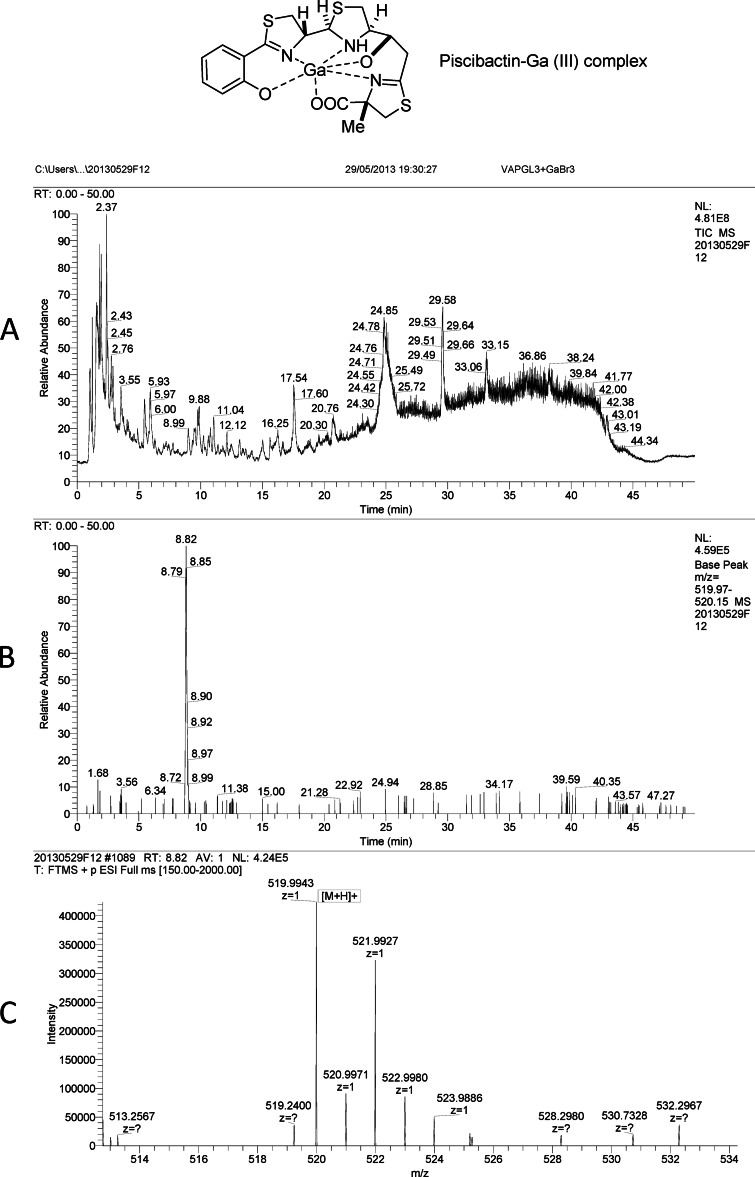
A 1.36-liter batch of the centrifuged cell-free culture broth of Vibrio alginolyticus strain AR43 was concentrated under vacuum to 410 ml. A portion of 75 ml was transferred to a round-bottom flask, and 4.0 mg of GaBr3 was added slowly over 5 min and gently stirred for another 10 min. After incubation at 4°C for 24 h, the solution was passed through one Oasis hydrophilic lipophilic balanced (HLB) cartridge (Waters) (35 cm3, 6 g), which was previously conditioned and equilibrated with 60 ml of acetonitrile (solvent B) and 60 ml of water (solvent A). It was eluted with 30 ml of the following mixtures of solvent A and solvent B: 1:0, 7:3, 1:1, 7:3, and 0:1. The fraction eluted with A-B at 1:1, named VAPGL3 (3.0 mg), was subjected to high-pressure liquid chromatography (HPLC)-high-resolution electrospray ionization mass (HRESIMS) analysis using a Atlantis dC18 column (100 by 4.6 mm, 5 μm) with a 35.0-min gradient from 10 to 100% CH3CN-H2O, then 5 min at 100% CH3CN, and finally a 10.0-min gradient from 100 to 10% H2O-CH3CN, at a flow rate of 1 ml min−1.
Virulence assays.
The pathogenicity of P. damselae subsp. piscicida DI21-Rif (parental), CS31 (insertional mutant with disrupted irp1), and AR84 (pPHDP70-cured) strains for turbot (Scophthalmus maximus) was assayed using 10 fish (average weight, 15 g) per strain tested, as previously described (23). Fish were maintained in 50-liter tanks with aeration and water recirculation and were kept under starvation during the experiment. Fish were inoculated intraperitoneally with 100 μl of a bacterial dose of 1 × 106 cells/fish, and mortalities were recorded daily for 7 days. A group of control fish was inoculated with 100 μl saline solution (0.85% NaCl). The statistical significance of differences in percent survival for P. damselae subsp. piscicida infection experiments was determined using the Kaplan-Meier method and a log rank test.
Nucleotide sequence accession number.
The complete sequence of pPHDP70 has been deposited in the GenBank database with the accession number KP100338.
RESULTS
Identification of pPHDP70 in P. damselae subsp. piscicida DI21.
In previous work, we found that the siderophore produced by P. damselae subsp. piscicida DI21 was encoded by a gene cluster (irp cluster) whose structure resembled that of a pathogenicity island (9). To investigate the genetic context of this cluster, we constructed and screened a cosmid library of DI21. Two overlapping cosmids that contained irp cluster markers were shown to be contiguous, and additional sequencing revealed the complete sequence of a circular structure. Furthermore, during the last stages of this project, we finished sequencing the complete genome of P. damselae subsp. piscicida DI21 (GenBank accession no. GCA_000300355.3) (to be published elsewhere), which also revealed a plasmid molecule whose sequence and structure were identical to those inferred by the cosmid-based sequencing approach. We dubbed this circular molecule pPHDP70, a hitherto-uncharacterized plasmid from P. damselae subsp. piscicida strain DI21, isolated from a diseased seabream (Sparus aurata) on a fish farm in Galicia (northwestern Spain).
Sequence analysis and general features of pPHDP70.
The nucleotide sequence of pPHDP70 is 68,686 bp and contains 53 potential open reading frames (ORFs). The gene organization and the G+C content of this plasmid are summarized in Fig. 1, and the similarities of the pPHDP70 gene products to proteins in P. damselae and other bacteria are listed in Table S1 in the supplemental material. The plasmid's average G+C content is 43.1%, similar to the mean G+C content of the P. damselae subsp. piscicida DI21 genome, which is 41.2%. There is a wide distribution of G+C content along pPHDP70, varying from 21% to 54%, suggesting that the plasmid contains genes from different sources. The lowest G+C content (21%) of the plasmid corresponds to a gene block that includes homologs (ORF47 and ORF48) of the Edwardsiella ictaluri proteins FimD and FimC, two components of a putative chaperone usher pathway (24).
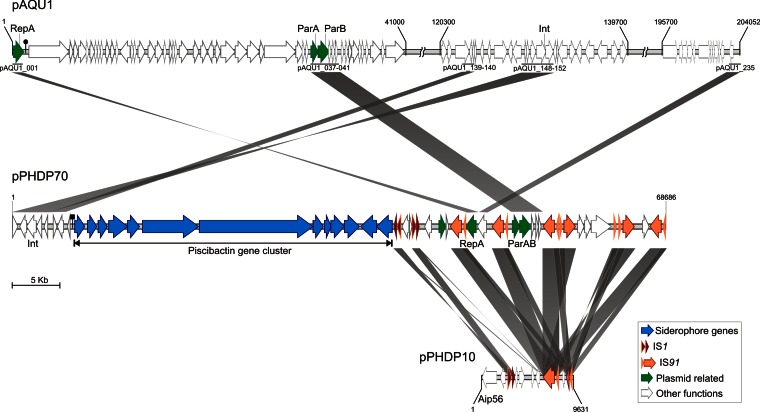
The pPHDP70 backbone has been targeted by three distinct insertion sequence (IS) elements, IS91, IS1, and IS3 (Fig. 1 and 2). IS91 occurs in a multicopy fashion (six copies), accounting for 10% of the total plasmid sequence, whereas IS1 (constituted by two ORFs, tnpA and tnpB) and IS3 are found in double and single copy, respectively. Interestingly, pPHDP70 does not contain pseudogenes generated by insertion of transposases into coding sequences. Instead, multigenic modules are bracketed by IS1 and IS91 copies (Fig. 1). The IS1 and IS91 sequences are identical to those found in the previously described P. damselae subsp. piscicida plasmid pPHDP10 (accession no. DQ069059), which encodes the AIP56 toxin (Fig. 2). IS1 is also found in other Vibrio species on plasmids and in chromosomal locations.
FIG 2.
Linear graphical representation and comparative analysis of the genomes of the two P. damselae subsp. piscicida plasmids pPHDP70 and pPHDP10 and the P. damselae subsp. damselae plasmid pAQU1. ORFs encoding transposases and plasmid-related functions are highlighted. Homologous ORFs shared between plasmids are denoted by diagonal gray bands. Note that pPHDP70 shares plasmid backbone genes with pAQU1 and insertion sequences with pPHDP10.
pPHDP70 shares four discrete sequence blocks with the pAQU1 backbone (Fig. 2), a plasmid recently sequenced from a Japanese isolate of P. damselae subsp. damselae (accession no. NC016983) (25). The shared blocks comprise 14 genes, with average similarity values between the two plasmids of 98% at the nucleotide level and 100% at the amino acid level, and putatively encode functions for replication and plasmid maintenance (repA, parA, parB, and int) as well as several hypothetical proteins. Interestingly, repA and parAB are flanked by copies of IS91 in pPHDP70 (Fig. 2).
ORF33 showed 96% identity to the putative replication protein RepA of pAQU1; however, the putative iteron sequences described in pAQU1 are absent from pPHDP70. There is a region upstream of orf9 (encoding a 3-deoxy-d-arabino-heptulosonate-7-phosphate [DAHP] synthase) containing 16 direct repeats (Fig. 1) of the octameric sequence ATAGCGAT that might constitute the iterons for RepA-mediated replication; however, we did not find sequences homologous to this octameric putative iteron in DNA databases. In an initial attempt to dissect the elements necessary for pPHDP70 replication, we ligated the iteron region to a kanamycin resistance (Kmr) cassette and assessed its ability to undergo replication in E. coli at 25°C and at 37°C. Regardless of the temperature, we were able to obtain Kmr transformants only when iteron sequences coexisted with repA (orf33) cloned into a plasmid. However, these transformants, which were shown by PCR to contain the putative iterons and the Kmr cassette, showed a microcolony phenotype. These results suggest that although the cis- and trans-acting elements tested (iterons and repA) appear to be sufficient to enable low levels of replication, efficient replication of pPHDP70 requires additional, unknown factors.
The piscibactin cluster has similarity to chromosome-encoded HPI-like clusters in other species.
The hallmark of pPHDP70 is the presence of an HPI-like pathogenicity island that includes the 11 previously identified irp (iron-regulated proteins) genes (9). The complete sequence of pPHDP70 in the present study uncovered novel genes potentially involved in siderophore synthesis (entD) and transport (the ABC transporter genes orf20 and orf21) (Fig. 3). Together with the previously reported putative outer membrane receptor gene frpA, orf20 and orf21 are candidates to constitute the dedicated transport system for the piscibactin siderophore.
FIG 3.
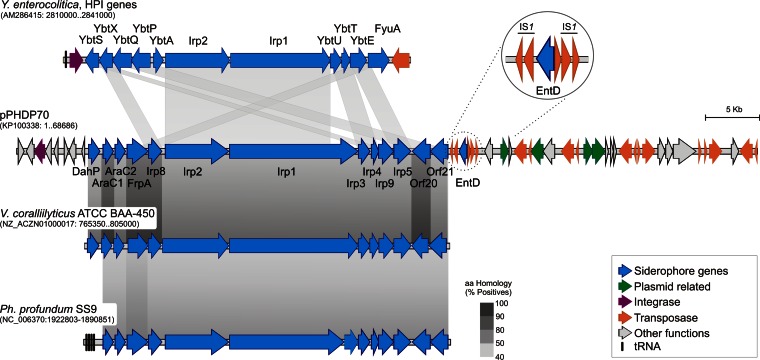
Comparative analysis of P. damselae subsp. piscicida plasmid pPHDP70 with homologous gene clusters in Yersinia enterocolitica (high-pathogenicity island [HPI]), Vibrio coralliilyticus, and Photobacterium profundum, highlighting the region of siderophore-related genes (in blue). Homologous ORFs shared between pairs of sequences are connected by gray bands. The percent similarity refers to the amino acid sequences of the predicted proteins and is represented in similarity intervals in grayscale tones according to the scale shown at the bottom right. In parentheses below each species name, the GenBank accession number and the nucleotide position interval represented for each species are shown. Note that the cluster of pPHDP70 is plasmid borne and the homologous clusters are all chromosomal.
Each siderophore-related pPHDP70 gene has a homologous counterpart in the Yersinia HPI, with the exceptions of dahP, araC1, araC2, entD, orf20, and orf21. The proteins shared between pPHDP70 and HPI showed similarity values at the amino acid level ranging from 44 to 62% (see Table S1 in the supplemental material), and the gene arrangement differs between the PAIs (Fig. 3). Although the two clusters are not exactly syntenic, both encode the determinants for biosynthesis, transport, and regulation of two distinct but similar siderophores, piscibactin (10) and yersiniabactin (26), respectively. Notably, by an in silico search in databases, we found gene clusters homologous to the pPHDP70-borne irp cluster in isolates of several Vibrionaceae species. These include human and marine animal pathogens such as Vibrio cholerae, V. mimicus, V. coralliilyticus, V. ordalii, and V. anguillarum. The closest clusters are found in V. coralliilyticus and Photobacterium profundum and show a high degree of synteny with the irp cluster (Fig. 3).
The pPHDP70 irp cluster includes two biosynthetic genes that are absent from the HPI and from many of the related Vibrionaceae clusters. One gene encodes a 3-deoxy-d-arabino-heptulosonate-7-phosphate (DAHP) synthase (ORF9) involved in chorismate synthesis and is necessary for production of salicilic acid, one of the piscibactin components (10). DAHP synthases specifically involved in providing chorismate for siderophore production have been reported in other Vibrionaceae (27). A second gene, entD, encodes the enzyme phosphopantheteinyl transferase, which is required to activate nonribosomal peptide synthesis (NRPS) domains (28). Homologs of entD have not been found in the other irp-like clusters (Fig. 3). In Yersinia, the phosphopantheteinyl transferase gene ybtD, which is homologous to the P. damselae subsp. piscicida entD, is located outside the HPI (29). It is tempting to speculate that entD may have been recruited by natural selection to be closely associated with the rest of the piscibactin synthesis-related genes. The recruitment hypothesis for P. damselae subsp. piscicida entD is reinforced by the observation that entD is bracketed between tandem copies of tnpAB transposase genes (IS1) (Fig. 3).
pPHDP70 can be conjugally transferred to a variety of species of Gammaproteobacteria, helped by the coresident plasmid pPHDP60.
Although pPHDP70 appears to lack genes encoding a conjugation system, we investigated whether this plasmid could be conjugally transferred to different recipients. We found that pPHDP70::Km was transmissible from DI21-Rif to Escherichia coli, Aeromonas salmonicida, Vibrio anguillarum, and V. alginolyticus at low frequencies (Table 3). PCR assays targeting a number of different pPHDP70 markers and plasmid purification and restriction analyses revealed that the entire pPHDP70 plasmid was being transferred to the transconjugants (data not shown).
However, we were unable to detect transfer of pPHDP70::Km from the E. coli MC1061 (pPHDP70::Km) transconjugant (Table 3). This observation coupled with the apparent lack of pPHDP70-encoded conjugation machinery suggested that pPHDP70 mobilization relies on conjugative functions encoded elsewhere in the genome of P. damselae subsp. piscicida DI21. An in silico search in the genome sequence of this strain revealed a putative type IV secretion system in pPHDP60, a conjugative plasmid recently described (14). We hypothesized that pPHDP60 might enable the mobilization of pPHDP70 in trans. Consistent with this idea, we found that an E. coli MC1061 donor that harbored pPHDP70::Km and a Cm-labeled version of pPHDP60 (pPHDP60::Cm) could transfer pPHDP70::Km to E. coli BI533; both pPHDP70::Km and pPHDP60::Cm were independently mobilized to E. coli BI533 at frequencies of 1.6 × 10−7 (pPHDP70::Km) and 5 × 10−5 (pPHDP60::Cm) (Table 3). Of 40 pPHDP70::Km transconjugants tested from these conjugation assays, none was Cmr, suggesting that transfer of pPHDP70 did not require concomitant transfer of pPHDP60 or cointegrate formation. Additionally, restriction analyses of plasmid DNA from transconjugants demonstrated that the pPHDP70 in the transconjugants did not contain additional DNA in its structure (data not shown). Since the two plasmids coexist in strain DI21, these findings suggest that pPHDP70 might be capable of conjugative transfer in the natural environment.
pPHDP70 promotes piscibactin biosynthesis and utilization in a heterologous host upon conjugative transfer.
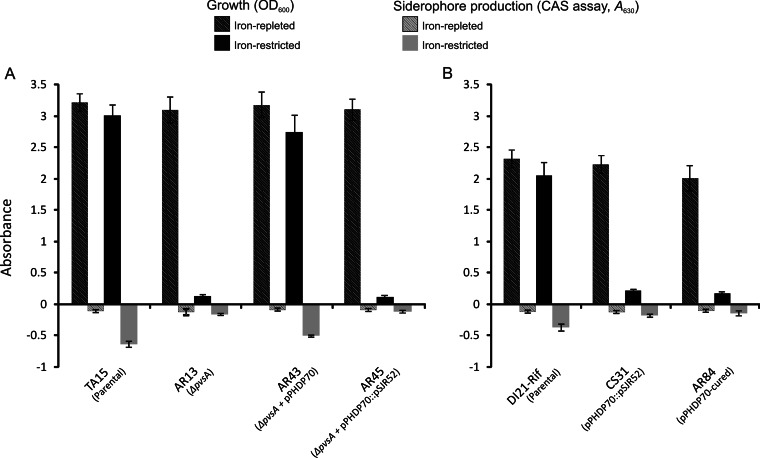
We recently determined the chemical structure of piscibactin and proposed a pathway for its biosynthesis based on an NRPS-mediated mechanism (10). The pPHDP70-borne irp1 gene is necessary for piscibactin biosynthesis (9), but it is not clear whether pPHDP70 encodes all the biological functions required for piscibactin biosynthesis and its efficient utilization as an iron source. To explore this possibility, we used V. alginolyticus(pPHDP70) transconjugants derived as described above. The parental V. alginolyticus TA15 strain was originally isolated in an episode of mortality of carpet shell clam (Ruditapes decussatus) larvae (30) and harbors a gene cluster for production and utilization of the siderophore vibrioferrin. Vibrioferrin is a hydroxamate siderophore synthesized by an NRPS-independent mechanism (31) that does not share genes with the piscibactin NRPS-mediated mechanism. Therefore, we considered this a good model heterologous host to assess the sufficiency of pPHDP70 for piscibactin synthesis. TA15 vibrioferrin synthesis was abolished by inactivation of the biosynthetic gene pvsA. As expected, the pvsA mutant strain AR13 exhibited impaired growth under conditions of iron limitation and had reduced siderophore activity (Fig. 4A). In contrast, when the pvsA mutant V. alginolyticus harbored pPHDP70 (strain AR43), its ability to grow under iron-limiting conditions was restored and it had detectable siderophore activity (Fig. 4A). As a control, we introduced a pPHDP70 with an inactivated irp1 gene (pPHDP70::pSJR52) by conjugation into AR13 (TA15 ΔpvsA). The resulting strain (AR45) exhibited impaired growth under iron limitation and no siderophore activity (Fig. 4A). These findings indicate that the conjugally transmitted pPHDP70 can bestow upon the heterologous V. alginolyticus host the capacity to produce a siderophore.
FIG 4.
Growth (OD600) and siderophore production (CAS assay) (A630) of V. alginolyticus (A) and P. damselae subsp. piscicida (B) parental strains and mutants in CM9 minimal medium after 16 h of cultivation under iron-replete conditions (10 μM ferric sulfate) and under iron-limited conditions (the iron chelator 2,2′-dipyridyl at 60 μM for P. damselae subsp. piscicida, and 16 μM for V. alginolyticus). The CAS assay yields more-negative values as the siderophore activity increases. The assays were carried out in triplicate, and mean values with standard deviations are shown.
We used the methodology based on HLB cartridges and liquid chromatography-mass spectrometry (LC-MS) that we developed for isolation of piscibactin (10) to test whether V. alginolyticus pPHDP70 transconjugants produced piscibactin. Cell-free culture supernatants of strain AR43 were incubated with an excess of GaBr3, fractionated through HLB cartridges, and separated over a reverse-phase column by HPLC. In this way, we detected a compound with retention time (tR) of 8.82 min, which, by HRESIMS analyses, showed an m/z of 519.9943/521.9927 and a distinctive gallium isotopic ratio (3:2) in concordance with those of piscibactin-Ga(III) complex (Fig. 5). Collectively, these observations provide strong evidence that pPHDP70 encodes the necessary biosynthetic, transport, and regulatory functions to enable synthesis and utilization of piscibactin following its conjugative transfer to a different species.
FIG 5.
LC-MS experiments for the detection of the piscibactin-Ga(III) complex. (A) Total ion chromatogram (TIC) of the fraction eluted with H2O-CH3CN (1:1) from the Oasis HLB cartridge (VAPGL3) containing piscibactin-Ga(III) complex; (B) extracted mass chromatogram (m/z 519.97 to 520.15) showing the peak with retention time of 8.82 min; (C) (+)-HRESIMS of the peak at a tR of 8.82 min identified as piscibactin-Ga(III) complex: m/z 519.9943/521.9927 ([M + H]+); calc. for C19H21N3O4S3Ga, 519.9950/521.9941. Chromatographic conditions: Atlantis dC18 column (100 by 4.6 mm, 5 μm) (Waters); 35-min gradient from 10 to 100% of CH3CN in H2O, then 5 min at 100% CH3CN, and finally 10 min from 100 to 10% CH3CN in H2O; flow rate, 1 ml min−1.
Curing pPHDP70 from P. damselae subsp. piscicida impairs growth under iron-limiting conditions, abolishes piscibactin siderophore production, and diminishes virulence for fish.
We cured P. damselae subsp. piscicida DI21-Rif of pPHDP70 by inserting a suicide plasmid in the irp1 gene and selecting for loss of the cointegrate to assess the role of pPHDP70 in iron utilization and virulence. PCR assays for plasmid markers as well as plasmid extraction and restriction analysis demonstrated the absence of pPHDP70 sequences from the cured strain, AR84 (data not shown). DI21-Rif and AR84 had similar growth in CM9 minimal medium supplemented with 10 μM FeSO4 (iron-sufficient conditions) (Fig. 4B). However, when the same strains were grown in CM9 in the presence of the iron chelator 2,2′-dipyridyl, AR84 exhibited markedly impaired growth compared with the parental strain as well as reduced reactivity in the CAS siderophore assay, showing results similar to those for CS31, a DI21 derivative in which irp1 is inactivated (Fig. 4B). In addition, chemical analysis did not detect the presence of piscibactin in the culture supernatants of AR84 (data not shown).
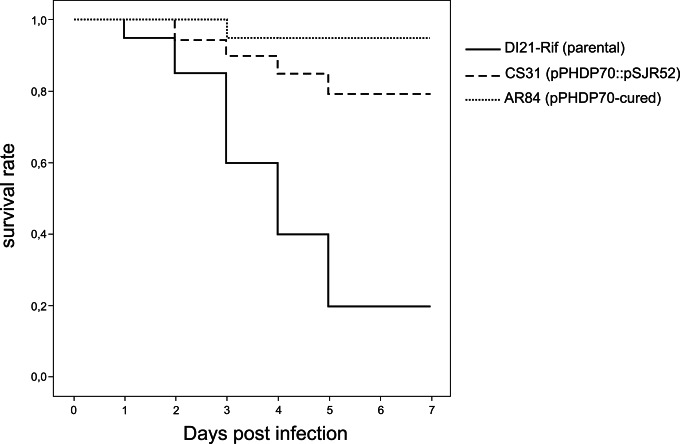
We inoculated turbot fingerlings with DI21-Rif, AR84, and CS31 to assess the contribution of pPHDP70 to the virulence of P. damselae subsp. piscicida for fish. The two non-piscibactin-producing strains (AR84 and CS31) caused much less mortality in the fish than DI21-Rif (P < 0.001 by the log rank test) (Fig. 6). Although the differences in the number of deaths caused by the cured strain versus insertional mutant did not reach statistical significance (P = 0.152), the cured strain caused fewer fish deaths (1 out of 10) than the single irp mutant (4 out of 10). Altogether, our results indicate that production of siderophores by the irp cluster in pPHDP70 contributes significantly to the virulence of P. damselae subsp. piscicida for fish.
FIG 6.
Survival of turbot (Scophthalmus maximus) inoculated with P. damselae subsp. piscicida strains DI21-Rif (parental), CS31 (insertional mutation in irp1 gene), and AR84 (pPHDP70 cured). The virulence of each strain was tested using 10 fish (average weight, 15 g), as previously described (23), inoculating 1 × 106 cells/fish. Mortalities were recorded daily for 7 days. The statistical significance of differences in percent survival for P. damselae subsp. piscicida strains was determined using the Kaplan-Meier method and a log rank test.
pPHDP70 shows geographical specificity within P. damselae subsp. piscicida isolates.
A variety of molecular evidence (32–36) indicates that there are two distinct P. damselae subsp. piscicida clonal lineages: a lineage that comprises all the European isolates and a lineage that includes the Japanese and U.S. isolates. We found that the distribution of the irp cluster in P. damselae subsp. piscicida isolates follows a geographical pattern and is restricted to European isolates (9). We wondered whether the modular piscibactin-synthesizing plasmid pPHDP70 found in strain DI21 is conserved in P. damselae subsp. piscicida isolates. We used PCR assays to test for the presence of genes from different parts of pPHDP70 (entD-orf21, parAB, orf28, and orf48) in 14 P. damselae subsp. piscicida isolates from diverse regions of isolation (Table 4). The results were equivalent for all the genes assayed: all P. damselae subsp. piscicida strains isolated in Europe contained all the genes tested, whereas none of the American and Asian isolates tested contained any of these genes. Moreover, the primer pair combination used for entD allowed us to corroborate that this gene is invariably linked to the irp cluster (orf21) and separated from it by an IS1 element. In addition, we also found that a total of 17 P. damselae subsp. damselae isolates from different origins (Table 4) tested negative in PCR tests for pPHDP70 markers.
TABLE 4.
Results of PCR screening for presence of pPHDP70 gene markers in P. damselae subsp. piscicida and P. damselae subsp. damselae strains
| Species and strain | Source | Presence of gene |
|||
|---|---|---|---|---|---|
| entD-orf21 | parAB | orf28 | orf48 | ||
| P. damselae subsp. piscicida | |||||
| DI21 | Seabream, Spain | + | + | + | + |
| PC554.2 | Sole, Spain | + | + | + | + |
| PC435.1 | Sole, Spain | + | + | + | + |
| B51 | Seabream, Spain | + | + | + | + |
| 666.1 | Seabass, Portugal | + | + | + | + |
| IT-1 | Seabream, Italy | + | + | + | + |
| ATLIT 2 | Morone sp., Israel | + | + | + | + |
| ATCC 17911 | White perch, USA | − | − | − | − |
| ATCC 29690 | Yellowtail, Japan | − | − | − | − |
| EPOY 8803-II | Red grouper, Japan | − | − | − | − |
| MP-7801 | Yellowtail, Japan | − | − | − | − |
| MZS-8001 | Yellowtail, Japan | − | − | − | − |
| P3333 | Yellowtail, Japan | − | − | − | − |
| P3335 | Yellowtail, Japan | − | − | − | − |
| P. damselae subsp. damselae | |||||
| RM-71 | Turbot, Spain | − | − | − | − |
| RG-91 | Turbot, Spain | − | − | − | − |
| RG-153 | Turbot, Spain | − | − | − | − |
| RG-214 | Turbot, Spain | − | − | − | − |
| PC586.1 | Seabream, Spain | − | − | − | − |
| 309 | Mussel, Spain | − | − | − | − |
| LD-07 | Seabream, Spain | − | − | − | − |
| 340 | Seawater, Spain | − | − | − | − |
| 158 | Eel, Belgium | − | − | − | − |
| 162 | Eel, Belgium | − | − | − | − |
| PG-801 | Shrimp, Taiwan | − | − | − | − |
| J3G-801 | Shrimp, Taiwan | − | − | − | − |
| 192 | Dolphin, USA | − | − | − | − |
| ATCC 35083 | Brown shark, USA | − | − | − | − |
| 238 | Dolphin, USA | − | − | − | − |
| CDC-2227-81 | Human, USA | − | − | − | − |
| ATCC 33539 | Damselfish, USA | − | − | − | − |
DISCUSSION
The P. damselae subsp. piscicida irp genes were known to be clustered in a pathogenicity island-like structure related to the Yersinia HPI (9) and were implicated in the synthesis of piscibactin, a phenolate siderophore (10). Here, we discovered that this HPI-like cluster is contained within a transmissible plasmid, pPHDP70. pPHDP70 is not self-transmissible; instead, its transmission from P. damselae subsp. piscicida to other organisms relied on pPHDP60, a coresident plasmid that encodes a type IV secretion system. Conjugal transfer of pPHDP70 to a V. alginolyticus strain deficient in siderophore synthesis enabled the transconjugant to grow under low-iron conditions and to synthesize piscibactin, providing strong evidence that pPHDP70 encodes all the information for synthesis and transport of this siderophore. A strain cured of pPHDP70 showed impaired growth under low-iron conditions, no piscibactin synthesis, and markedly reduced virulence in fish. Thus, pPHDP70 is an unusual virulence plasmid that contains a transmissible form of a pathogenicity island that is ordinarily locked into the chromosome.
In general, the irp cluster has a higher G+C content than the remainder of the plasmid, consistent with the idea that during pPHDP70 evolution, the irp cluster, which was derived from a different source than the rest of the plasmid, fused with an ancestral version of the plasmid. The heterogeneous origin of pPHDP70 genes is also suggested by the diverse taxonomy of the species showing the closest homologs to pPHDP70 genes. Thus, pPHDP70 can be considered a mosaic, whose evolution appears to have depended on recombination of genetic information from a variety of sources, including a likely chromosomal irp cluster.
We have shown that gene clusters homologous to pPHDP70 irp cluster are present in several species of vibrios. The high similarity at the amino acid level among homologs and the fact that they share the same functional domains in the biosynthetic proteins (data not shown) strongly suggest that these different Vibrio species produce piscibactin-related siderophores. Indeed, a recent study revealed that closely related Vibrio species in the marine environment can produce the same or similar siderophore molecules, suggesting that evolutionary forces have promoted the use of the siderophore-mediated mechanism as a “public good” to be used by species of the same microbial community (37).
It is important to note that none of the homologous clusters found in other Vibrionaceae is plasmid borne; they are all chromosomal. Indeed, one of the distinctive features of the P. damselae subsp. piscicida irp cluster is that it is a plasmid-borne PAI-like element. Although PAIs were first identified as chromosomal regions (38), parts of some virulence plasmids are also considered PAIs: the invasion regions of Shigella plasmids (39) and the Yersinia plasmid cluster encoding Yop proteins (40). Pantoea agglomerans, a plant pathogen, is thought to have recently evolved into a host-specific tumorigenic pathogen by acquisition of a plasmid-borne PAI (41). However, in contrast to the pPHDP70 cluster, these other examples of plasmid-borne PAIs do not have chromosomal counterparts. Chromosomal gene clusters similar to the pPHDP70 irp cluster include the Yersinia HPI and homologous clusters described in yersiniabactin-producing strains of Escherichia, Citrobacter, Klebsiella, and Serratia (12, 13, 42). In addition, an HPI was found as part of an integrative and conjugative element (ICE) in E. coli (HPI-ICEEc1) (43), in K. pneumoniae (HPI-ICEKp1) (44), and in Enterobacter hormaechei (HPI-ICEEh1) (45). Recently, an ICE in Streptococcus equi that putatively encodes the synthesis of a yersiniabactin and piscibactin-like siderophore was reported, whose biosynthetic proteins bear similarity to HPI- and irp-encoded proteins (46). Similarly, the mobile element ICEPm1, common to Proteus mirabilis, Providencia stuartii, and Morganella morganii, contains the Yersinia HPI within its structure (47). These observations, together with the similarity of some Vibrionaceae clusters to the Yersinia HPI and the remarkable chemical similarities of piscibactin and yersiniabactin, suggest that HPI/irp-like clusters have evolved as potential horizontally transmissible elements in diverse bacterial species. Thus, piscibactin- and yersiniabactin-synthetic genes appear to have evolved on and likely exchanged between a variety of mobile elements, including plasmids, ICE, and PAIs, which enables their transmission among diverse bacteria in both in terrestrial and aquatic ecosystems.
Horizontal gene transfer is thought to be extensive among the Vibrionaceae (48). The transmissibility of pPHDP70 is another example of the key role that mobile elements have had in underlying the emergence of disease-causing vibrios. Notably, we demonstrated that a virulence plasmid can be transferred from a fish pathogen (P. damselae subsp. piscicida) to a mollusk pathogen (Vibrio alginolyticus) that inhabits the same environment. This suggests that pPHDP70 might also be capable of undergoing horizontal transfer under natural conditions, perhaps at frequencies higher than those we found in our study, which was conducted under laboratory conditions.
P. damselae subsp. piscicida may be an obligate pathogen, as no free-living state has been described to date. This pathogen is predicted to face an iron-limiting environment in fish, and therefore the acquisition and spread of pPHDP70 might have constituted a driving force in the evolution of the European P. damselae subsp. piscicida lineage. The restriction of pPHDP70 to European isolates is consistent with the existence of two lineages and suggests that pPHDP70 might be useful as a genetic marker for epidemiological typing of P. damselae subsp. piscicida strains. Our findings also leave open the question of which siderophore is produced by Asian and American P. damselae subsp. piscicida isolates as well as by P. damselae subsp. damselae strains, which do not harbor pPHDP70.
When P. damselae subsp. piscicida was cured of pPHDP70, the strain's virulence was markedly diminished, indicating that pPHDP70 is a bona fide virulence plasmid. The importance of siderophore production in virulence has been reported for other fish pathogens (49), and in some cases this biological function has been found to be plasmid encoded, as is the case for the anguibactin siderophore system of Vibrio anguillarum encoded in plasmid pJM1 (50). The possibility that pPHDP70 carries additional genes with a role in virulence (for example, the chaperone usher pathway genes) cannot be ruled out, since the cured strain caused fewer fish deaths (1 out of 10) than the single irp mutant (4 out of 10), although these differences did not reach statistical significance (P = 0.152). Additional studies will be needed to ascertain whether pPHDP70 encodes properties in addition to piscibactin-mediated iron uptake that also contribute to P. damselae subsp. piscicida virulence.
Our results highlight the emerging similarities between terrestrial and marine animal pathogens, in terms of both the presence of similar siderophore-mediated iron-sequestering systems and their location on mobile elements. The recent discovery of a fish-pathogenic Edwardsiella tarda strain that acquired the locus of enterocyte effacement by horizontal transfer (2) indicates that lateral gene flow can surmount barriers between aquatic and terrestrial ecosystems and has a powerful influence on the generation of new virulence strategies. Our findings represent the first description of an HPI-like cluster borne on a mobilizable plasmid and demonstrate that the piscibactin cluster can be efficiently expressed in a recipient species following conjugative transfer. Transmission of this plasmid among different bacterial species in the marine environment enables recipients to synthesize a novel siderophore and thereby to potentially occupy new niches.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by grants AGL2012-39274-C02-01/02 and AGL2013-48353-R from the Ministry of Economy and Competitiveness (MINECO) of Spain (both cofunded by the FEDER Programme from the European Union) and by grant EM2012/043 from Xunta de Galicia, Spain.
Footnotes
Supplemental material for this article may be found at http://dx.doi.org/10.1128/AEM.01580-15.
REFERENCES
- 1.Sobecky PA, Hazen TH. 2009. Horizontal gene transfer and mobile genetic elements in marine systems. Methods Mol Biol 532:435–453. doi: 10.1007/978-1-60327-853-9_25. [DOI] [PubMed] [Google Scholar]
- 2.Nakamura Y, Takano T, Yasuike M, Sakai T, Matsuyama T, Sano M. 2013. Comparative genomics reveals that a fish pathogenic bacterium Edwardsiella tarda has acquired the locus of enterocyte effacement (LEE) through horizontal gene transfer. BMC Genomics 14:642. doi: 10.1186/1471-2164-14-642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Muziasari WI, Managaki S, Pärnänen K, Karkman A, Lyra C, Tamminen M, Suzuki S, Virta M. 2014. Sulphonamide and trimethoprim resistance genes persist in sediments at Baltic sea aquaculture farms but are not detected in the surrounding environment. PLoS One 9:e92702. doi: 10.1371/journal.pone.0092702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Magariños B, Toranzo AE, Romalde JL. 1996. Phenotypic and pathobiological characteristics of Pasteurella piscicida. Annu Rev Fish Dis 6:41–64. doi: 10.1016/S0959-8030(96)90005-8,10.1016/S0959-8030(96)00004-X. [DOI] [Google Scholar]
- 5.Rivas AJ, Lemos ML, Osorio CR. 2013. Photobacterium damselae subsp. damselae, a bacterium pathogenic for marine animals and humans. Front Microbiol 4:283. doi: 10.3389/fmicb.2013.00283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Do Vale A, Silva MT, dos Santos NMS, Nascimento DS, Reis-Rodrigues P, Costa-Ramos C, Ellis AE, Azevedo JE. 2005. AIP56, a novel plasmid-encoded virulence factor of Photobacterium damselae subsp. piscicida with apoptogenic activity against sea bass macrophages and neutrophils. Mol Microbiol 58:1025–1038. doi: 10.1111/j.1365-2958.2005.04893.x. [DOI] [PubMed] [Google Scholar]
- 7.Rivas AJ, Balado M, Lemos ML, Osorio CR. 2011. The Photobacterium damselae subsp. damselae hemolysins damselysin and HlyA are encoded within a new virulence plasmid. Infect Immun 79:4617–4627. doi: 10.1128/IAI.05436-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Magariños B, Romalde JL, Lemos ML, Barja JL, Toranzo AE. 1994. Iron uptake by Pasteurella piscicida and its role in pathogenicity for fish. Appl Environ Microbiol 60:2990–2998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Osorio CR, Juiz-Rio S, Lemos ML. 2006. A siderophore biosynthesis gene cluster from the fish pathogen Photobacterium damselae subsp. piscicida is structurally and functionally related to the Yersinia high-pathogenicity island. Microbiology 152:3327–3341. doi: 10.1099/mic.0.29190-0. [DOI] [PubMed] [Google Scholar]
- 10.Souto A, Montaos MA, Rivas AJ, Balado M, Osorio CR, Rodríguez J, Lemos ML, Jiménez C. 2012. Structure and biosynthetic assembly of Piscibactin, a new siderophore from Photobacterium damselae subsp. piscicida, predicted from genome analysis. Eur J Org Chem 2012:5693–5700. doi: 10.1002/ejoc.201200818. [DOI] [Google Scholar]
- 11.Carniel E, Guilvout I, Prentice M. 1996. Characterization of a large chromosomal “high-pathogenicity island” in biotype 1B Yersinia enterocolitica. J Bacteriol 178:6743–6751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bach S, de Almeida A, Carniel E. 2000. The Yersinia high-pathogenicity island is present in different members of the family Enterobacteriaceae. FEMS Microbiol Lett 183:289–294. doi: 10.1111/j.1574-6968.2000.tb08973.x. [DOI] [PubMed] [Google Scholar]
- 13.Olsson C, Olofsson T, Ahrné S, Molin G. 2003. The Yersinia HPI is present in Serratia liquefaciens isolated from meat. Lett Appl Microbiol 37:275–280. doi: 10.1046/j.1472-765X.2003.01387.x. [DOI] [PubMed] [Google Scholar]
- 14.Balado M, Lemos ML, Osorio CR. 2013. Genetic characterization of pPHDP60, a novel conjugative plasmid from the marine fish pathogen Photobacterium damselae subsp. piscicida. Plasmid 70:154–159. doi: 10.1016/j.plasmid.2013.02.004. [DOI] [PubMed] [Google Scholar]
- 15.Osorio CR, Marrero J, Wozniak RA, Lemos ML, Burrus V, Waldor MK. 2008. Genomic and functional analysis of ICEPdaSpaI, a fish-pathogen-derived SXT-related integrating conjugative element that can mobilize a virulence plasmid. J Bacteriol 190:3353–3361. doi: 10.1128/JB.00109-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, Edwards RA, Formsma K, Gerdes S, Glass EM, Kubal M, Meyer F, Olsen GJ, Olson R, Osterman AL, Overbeek RA, McNeil LK, Paarmann D, Paczian T, Parrello B, Pusch GD, Reich C, Stevens R, Vassieva O, Vonstein V, Wilke A, Zagnitko O. 2008. The RAST Server: rapid annotation using subsystems technology. BMC Genomics 9:75. doi: 10.1186/1471-2164-9-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Grant JR, Stothard P. 2008. The CGView Server: a comparative genomics tool for circular genomes. Nucleic Acids Res 36:181–184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Carver TJ, Rutherford KM, Berriman M, Rajandream MA, Barrell BG, Parkhill J. 2005. ACT: the Artemis comparison tool. Bioinformatics 21:3422–3423. doi: 10.1093/bioinformatics/bti553. [DOI] [PubMed] [Google Scholar]
- 19.Mouriño S, Osorio CR, Lemos ML. 2004. Characterization of the heme uptake cluster genes in the fish pathogen Vibrio anguillarum. J Bacteriol 186:6159–6167. doi: 10.1128/JB.186.18.6159-6167.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Correa NE, Lauriano CM, McGee R, Klose KE. 2000. Phosphorylation of the flagellar regulatory protein FlrC is necessary for Vibrio cholerae motility and enhanced colonization. Mol Microbiol 35:743–755. doi: 10.1046/j.1365-2958.2000.01745.x. [DOI] [PubMed] [Google Scholar]
- 21.Datsenko KA, Wanner BL. 2000. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci U S A 97:6640–6645. doi: 10.1073/pnas.120163297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schwyn B, Neilands JB. 1987. Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56. doi: 10.1016/0003-2697(87)90612-9. [DOI] [PubMed] [Google Scholar]
- 23.Magariños B, Santos Y, Romalde JL, Rivas C, Barja JL, Toranzo AE. 1992. Pathogenic activities of the live cells and extracellular products of the fish pathogen Pasteurella piscicida. J Gen Microbiol 138:2491–2498. doi: 10.1099/00221287-138-12-2491. [DOI] [PubMed] [Google Scholar]
- 24.Busch A, Waksman G. 2012. Chaperone-usher pathways: diversity and pilus assembly mechanism. Philos Trans R Soc Lond B Biol Sci 367:1112–1122. doi: 10.1098/rstb.2011.0206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nonaka L, Maruyama F, Miyamoto M, Miyakoshi M, Kurokawa K, Masuda M. 2012. Novel conjugative transferable multiple drug resistance plasmid pAQU1 from Photobacterium damselae subsp. damselae isolated from marine aquaculture environment. Microbes Environ 27:263–272. doi: 10.1264/jsme2.ME11338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gehring AM, DeMoll E, Fetherston JD, Mori I, Mayhew GF, Blattner F, Walsh CT, Perry RD. 1998. Iron acquisition in plague: molecular logic in the biosynthesis of yersiniabactin by Yersinia pestis. Chem Biol 5:573–586. doi: 10.1016/S1074-5521(98)90115-6. [DOI] [PubMed] [Google Scholar]
- 27.Balado M, Osorio CR, Lemos ML. 2008. Biosynthetic and regulatory elements involved in the production of the siderophore vanchrobactin in Vibrio anguillarum. Microbiology 154:1400–1413. doi: 10.1099/mic.0.2008/016618-0. [DOI] [PubMed] [Google Scholar]
- 28.Crosa JH, Walsh CT. 2002. Genetics and assembly line enzymology of siderophore biosynthesis in bacteria. Microbiol Mol Biol Rev 66:223–249. doi: 10.1128/MMBR.66.2.223-249.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bobrov AG, Geoffroy VA, Perry RD. 2002. Yersiniabactin production requires the thioesterase domain of HMWP2 and YbtD, a putative phosphopantetheinylate transferase. Infect Immun 70:4204–4214. doi: 10.1128/IAI.70.8.4204-4214.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gómez-León J, Villamil L, Lemos ML, Novoa B, Figueras A. 2005. Isolation of Vibrio alginolyticus and Vibrio splendidus from aquacultured carpet shell clam (Ruditapes decussatus) larvae associated with mass mortalities. Appl Environ Microbiol 71:98–104. doi: 10.1128/AEM.71.1.98-104.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tanabe T, Funahashi T, Nakao H, Miyoshi S, Shinoda S, Yamamoto S. 2003. Identification and characterization of genes required for biosynthesis and transport of the siderophore vibrioferrin in Vibrio parahaemolyticus. J Bacteriol 185:6938–6949. doi: 10.1128/JB.185.23.6938-6949.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Magariños B, Osorio CR, Toranzo AE, Romalde JL. 1997. Applicability of ribotyping for intraspecific classification and epidemiological studies of Photobacterium damsela subsp. piscicida. Syst Appl Microbiol 20:634–639. doi: 10.1016/S0723-2020(97)80035-5. [DOI] [Google Scholar]
- 33.Magariños B, Toranzo AE, Barja JL, Romalde JL. 2000. Existence of two geographically-linked clonal lineages in the bacterial fish pathogen Photobacterium damselae subsp. piscicida evidenced by random amplified polymorphic DNA analysis. Epidemiol Infect 125:213–219. doi: 10.1017/S0950268899004252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Thyssen A, Van Eygen S, Hauben L, Goris J, Swings J, Ollevier F. 2000. Application of AFLP for taxonomic and epidemiological studies of Photobacterium damselae subsp. piscicida. Int J Syst Bacteriol 50:1013–1019. doi: 10.1099/00207713-50-3-1013. [DOI] [PubMed] [Google Scholar]
- 35.Kvitt H, Ucko M, Colorni A, Batargias C, Zlotkin A, Knibb W. 2002. Photobacterium damselae ssp. piscicida: detection by direct amplification of 16S rRNA gene sequences and genotypic variation as determined by amplified fragment length polymorphism (AFLP). Dis Aquat Organ 48:187–195. doi: 10.3354/dao048187. [DOI] [PubMed] [Google Scholar]
- 36.Hawke JP, Thune RL, Cooper RK, Judice E, Kelly-Smith M. 2003. Molecular and phenotypic characterization of strains of Photobacterium damselae subsp. piscicida isolated from hybrid striped bass cultured in Louisiana, USA. J Aquat Anim Health 15:189–201. doi: 10.1577/H02-043. [DOI] [Google Scholar]
- 37.Cordero OX, Ventouras LA, DeLong EF, Polz MF. 2012. Public good dynamics drive evolution of iron acquisition strategies in natural bacterioplankton populations. Proc Natl Acad Sci U S A 109:20059–20064. doi: 10.1073/pnas.1213344109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hacker J, Bender L, Ott M, Wingender J, Lund B, Marre R, Goebel W. 1990. Deletions of chromosomal regions coding for fimbriae and hemolysins occur in vitro and in vivo in various extraintestinal Escherichia coli isolates. Microb Pathog 8:213–225. doi: 10.1016/0882-4010(90)90048-U. [DOI] [PubMed] [Google Scholar]
- 39.Buchrieser C, Glaser P, Rusniok C, Nedjari H, d' Hauteville H, Kunst F, Sansonetti P, Parsot C. 2000. The virulence plasmid pWR100 and the repertoire of proteins secreted by the type III secretion apparatus of Shigella flexneri. Mol Microbiol 38:760–771. doi: 10.1046/j.1365-2958.2000.02179.x. [DOI] [PubMed] [Google Scholar]
- 40.Cornelis GR, Boland A, Boyd AP, Geuijen C, Iriarte M, Neyt C, Sory MP, Stainier I. 1998. The virulence plasmid of Yersinia, an antihost genome. Microbiol Mol Biol Rev 62:1315–1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Barash I, Manulis-Sasson S. 2009. Recent evolution of bacterial pathogens: the gall-forming Pantoea agglomerans case. Annu Rev Phytopathol 47:133–152. doi: 10.1146/annurev-phyto-080508-081803. [DOI] [PubMed] [Google Scholar]
- 42.Schubert S, Rakin A, Karch H, Carniel E, Heesemann J. 1998. Prevalence of the “high-pathogenicity island” of Yersinia species among Escherichia coli strains that are pathogenic to humans. Infect Immun 66:480–485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Schubert S, Dufke S, Sorsa J, Heesemann J. 2004. A novel integrative and conjugative element (ICE) of Escherichia coli: the putative progenitor of the Yersinia high-pathogenicity island. Mol Microbiol 51:837–848. [DOI] [PubMed] [Google Scholar]
- 44.Lin TL, Lee CZ, Hsieh PF, Tsai SF, Wang JT. 2008. Characterization of integrative and conjugative element ICEKp1-associated genomic heterogeneity in a Klebsiella pneumoniae strain isolated from a primary liver abscess. J Bacteriol 190:515–526. doi: 10.1128/JB.01219-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Paauw A, Leverstein-van Hall MA, Verhoef J, Fluit AC. 2010. Evolution in quantum leaps: multiple combinatorial transfers of HPI and other genetic modules in Enterobacteriaceae. PLoS One 5:e8662. doi: 10.1371/journal.pone.0008662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Heather Z, Holden MT, Steward KF, Parkhill J, Song L, Challis GL, Robinson C, Davis-Poynter N, Waller AS. 2008. A novel streptococcal integrative conjugative element involved in iron acquisition. Mol Microbiol 70:1274–1292. doi: 10.1111/j.1365-2958.2008.06481.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Flannery EL, Mody L, Mobley HLT. 2009. Identification of a modular pathogenicity island that is widespread among urease-producing uropathogens and shares features with a diverse group of mobile elements. Infect Immun 77:4887–4894. doi: 10.1128/IAI.00705-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hazen TH, Pan L, Gu JD, Sobecky PA. 2010. The contribution of mobile genetic elements to the evolution and ecology of Vibrios. FEMS Microbiol Ecol 74:485–499. doi: 10.1111/j.1574-6941.2010.00937.x. [DOI] [PubMed] [Google Scholar]
- 49.Fernández L, Márquez I, Guijarro JA. 2004. Identification of specific in vivo-induced (ivi) genes in Yersinia ruckery and analysis of ruckerbactin, a catecholate siderophore iron acquisition system. Appl Environ Microbiol 70:5199–5207. doi: 10.1128/AEM.70.9.5199-5207.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Di Lorenzo M, Stork M, Tolmasky ME, Actis LA, Farrell D, Welch TJ, Crosa LM, Wertheimer AM, Chen Q, Salinas P, Waldbeser L, Crosa JH. 2003. Complete sequence of virulence plasmid pJM1 from the marine fish pathogen Vibrio anguillarum strain 775. J Bacteriol 185:5822–5830. doi: 10.1128/JB.185.19.5822-5830.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Najimi M, Lemos ML, Osorio CR. 2008. Distribution of small plasmids in Aeromonas salmonicida subsp. salmonicida strains isolated from NW Spain and Portugal: evidence of clonality in strain isolated from turbot, Psetta maxima (L). J Fish Dis 31:469–472. doi: 10.1111/j.1365-2761.2007.00896.x. [DOI] [PubMed] [Google Scholar]
- 52.Lemos ML, Salinas P, Toranzo AE, Barja JL, Crosa JH. 1988. Chromosome-mediated iron uptake system in pathogenic strains of Vibrio anguillarum. J Bacteriol 170:1920–1925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Guyer MS, Reed RE, Steitz T, Low KB. 1981. Identification of a sex-factor-affinity site in E. coli as gamma delta. Cold Spring Harb Symp Quant Biol 45:135–140. doi: 10.1101/SQB.1981.045.01.022. [DOI] [PubMed] [Google Scholar]
- 54.Hochhut B, Marrero J, Waldor MK. 2000. Mobilization of plasmids and chromosomal DNA mediated by the SXT element, a constin found in Vibrio cholerae O139. J Bacteriol 182:2043–2047. doi: 10.1128/JB.182.7.2043-2047.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wertman KF, Wyman AR, Botstein D. 1986. Host/vector interactions which affect the viability of recombinant phage lambda clones. Gene 49:253–262. doi: 10.1016/0378-1119(86)90286-6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.