Abstract
Terahertz (THz) spectral properties of human cornea are explored as a function of central corneal thickness (CCT) and corneal water content, and the clinical utility of THz-based corneal water content sensing is discussed. Three candidate corneal tissue water content (CTWC) perturbations, based on corneal physiology, are investigated that affect the axial water distribution and total thickness. The THz frequency reflectivity properties of the three CTWC perturbations were simulated and explored with varying system center frequency and bandwidths (Q-factors). The modeling showed that at effective optical path lengths on the order of a wavelength the cornea presents a lossy etalon bordered by air at the anterior and the aqueous humor at the posterior. The simulated standing wave peak-to-valley ratio is pronounced at lower frequencies and its effect on acquired data can be modulated by adjusting the bandwidth of the sensing system. These observations are supported with experimental spectroscopic data. The results suggest that a priori knowledge of corneal thickness can be utilized for accurate assessments of corneal tissue water content. The physiologic variation of corneal thickness with respect to the wavelengths spanned by the THz band is extremely limited compared to all other structures in the body making CTWC sensing unique amongst all proposed applications of THz medical imaging.
Keywords: Biological and medical imaging, medical diagnostics, clinical instruments, hydration interactions
I. Introduction
RECENT research in terahertz (THz) medical imaging suggests that active, standoff sensing systems based on THz frequency illumination may be ideal for mapping the distribution and movement of water near the surface of physiologic tissues [1]–[6]. The tissue of the cornea offers a unique opportunity for tissue water content (TWC) mapping due to its extreme homogeneity and low physiologic variation when compared to all other tissue systems in the human body [7]. Many diseases and pathologies, such as Fuchs' dystrophy [8], keratoconus [9], [10], and corneal graft rejection [11] are: defined by, proceeded by, or correlated with changes in corneal tissue water content (CTWC) and current clinical measurement methodologies do not allow for sufficiently early detection of these conditions [12].
The cornea is composed of water distributed throughout a protein matrix with a general increase in corneal tissue water content (CTWC) as a function of depth from the tissue surface. This blend of constituents and their large difference in dielectric function allows for high sensitivity measurement of temporal and/or spatial CTWC gradients. Proteins and other non-water constituents display refractive indices in the 1.5–1.9 range with low loss tangents and negligible frequency dependence [13]. In contrast, the dielectric function of water has a large imaginary component (when referenced to protein) and a real part that decreases monotonically with increasing frequency. Due to the large differences in electromagnetic properties of protein and water in the THz regime, deviations in water volume fractions characteristic of specific corneal pathologies can be measured with very high accuracy. In other words, a standoff THz imaging system will be very sensitive to changes in water based tissue constituents and very insensitive to changes in nonaqueous constituents.
To expand upon our earlier work [14], [15], we have developed three candidate axial tissue water gradient models with perturbations based on the hypothesized tissue water content distribution in varying disease states and examined their effect on the THz reflectivity of the cornea. Furthermore, we have explored the effect of thickness variation on the electromagnetic properties of the cornea. These simulations suggest that the cornea is unique among all other external human body structures. While the physiologic variation in thickness of the cornea (450 µm < tcornea < 650 µm) [16] is quite large with respect to optical wavelengths (<1 µm) [17] and high frequency ultrasonic wavelengths (< 30 µm) [18], [19], it is extremely uniform with respect to THz wavelengths. Furthermore, the tissue structure of the cornea is very ordered and presents a lossy homogenous medium to a THz frequency sensing system. These properties result in a well-defined curved etalon, and support the use of spectrally resolved measurement techniques where specific spectral signatures arise from macroscopic structure and not material properties. In other words, at THz frequencies, the cornea can be thought of as a curved thin film [20].
These properties allow one to rely on the following physiologically relevant range of parameters a priori when making measurements:
corneal thicknesses from 400 µm–700 µm;
tissue water content from 75% to 85%;
cornea bordered by air and a volume of water (aqueous humor. Fig. 1).
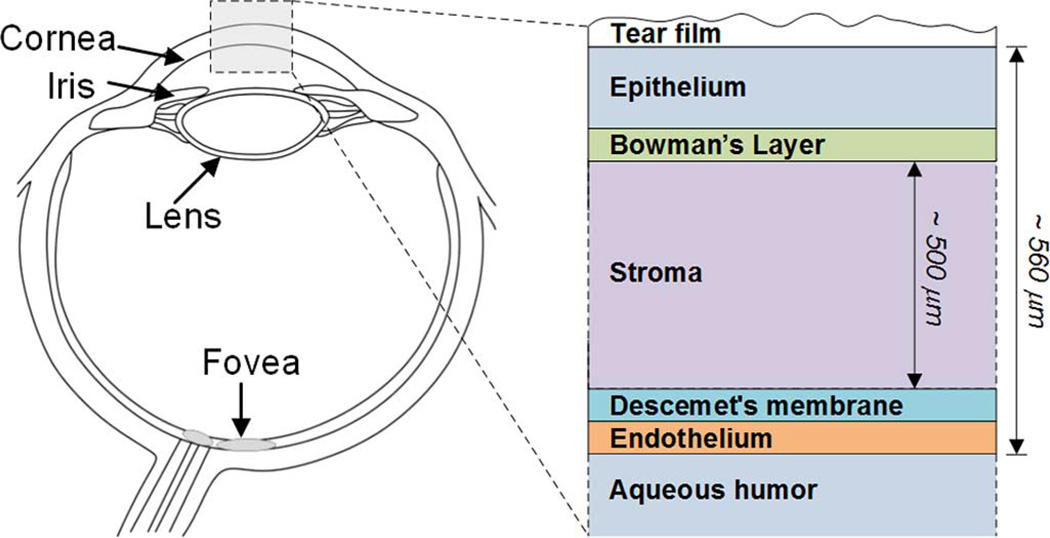
Fig. 1.
Corneal structure. The bulk of the cornea is composed of the Stroma. All other layers are between 5–15 µm (approximately optically thin at THz wavelengths). The cornea sits atop a body of water called the Aqueous humor.
This paper leverages these properties and explores the utility of millimeter wave and THz sensing and imaging systems to perform simultaneous CTWC and thickness measurements by analyzing the lossy etalon formed by the cornea and underlying body of water (aqueous humor). Finally, the expected utility of narrow band (Q = 50) and broadband (Q = 5) aggregate reflectometers centered at 100 and 525 GHz are examined in preparation for pre-clinical imaging in rabbit models, and the validity of the models are compared to spectroscopic measurements acquired in ex vivo porcine cornea.
II. Corneal Tissue Water Content
A. The Cornea
The cornea is the outermost structure of the eye (Fig. 1) and displays an average thickness in humans of ~580 µm [21]. The normal water content of the cornea is closely related to its visible wavelength (400–700 nm) transparency and refractive capabilities and typically contains ~78% water by volume [21]. It plays the leading role in collecting and focusing light on the retina, and provides 46 of the average 59 total diopters of refractive power in the eye [22].
The layered structure of the cornea is displayed in Fig. 1. The surface layer of the cornea (epithelium) is obscured by a thin layer of water called the tear film which is replenished by blinking, reflex tearing, and a number of other mechanisms [23]–[26]. The bottom layer of the cornea (endothelium) lies adjacent to a volume of water called the aqueous humor which is slightly more viscous than pure water but much less viscous than the vitreous humor [27]. The function of the endothelium is to regulate the water content of the stroma using the aqueous humor as a reservoir. While all the layers of the cornea are important for corneal health and visual acuity, all of the structures are optically thin with respect to THz wavelengths (< 15 µm) except for the stroma, which typically makes up ~90% of the total thickness (~ 522 µm). Due to the absorptive properties of liquid water, the majority of the interaction between THz illumination and corneal tissue occurs in the epithelium and the stroma. At longer wavelengths, such as millimeter waves, the skin depth increases to a point where the presence of the aqueous humor strongly contributes to the overall spectral response of the tissue. This reservoir of water behaves similar to a lossy electrical termination and, because of its impedance mismatch with the stroma, can create standing wave effects (Section IV-B) that can confound the overall water content measurement.
B. Role of Water Content in Corneal Diseases and Procedures
Many diseases of the eye measurably perturb corneal water content. Some, such as edema and corneal dystrophy, are diseases defined by the deterioration of the cornea's water-regulating process [8], [28], [29]. Others, such as keratoconus, have poorly understood mechanics, yet have been observed to exhibit non-uniform changes in water concentration of the cornea [30]. The water content of the cornea can also be perturbed by medical procedures, including Laser-Assisted in situ Keratomileusis (LASIK), Laser-Assisted Sub-Epithelial Keratectomy (LASEK), or corneal graft surgery [31]. In photorefractive surgeries, such as LASIK and LASEK, tissue ablation rates are strongly linked to corneal water content and errors in the measurement of TWC can be responsible for surgical over-correction [32]–[36]. In corneal graft surgeries, immune rejection is preceded by the formation of edema and the reversibility of rejection events tends to diminish with abnormal increases in TWC [31]. Furthermore, the survival rate of rejected grafts generally improves the earlier the formation of edema is detected [37].
C. Limitations of Clinically Accepted Measurement Techniques
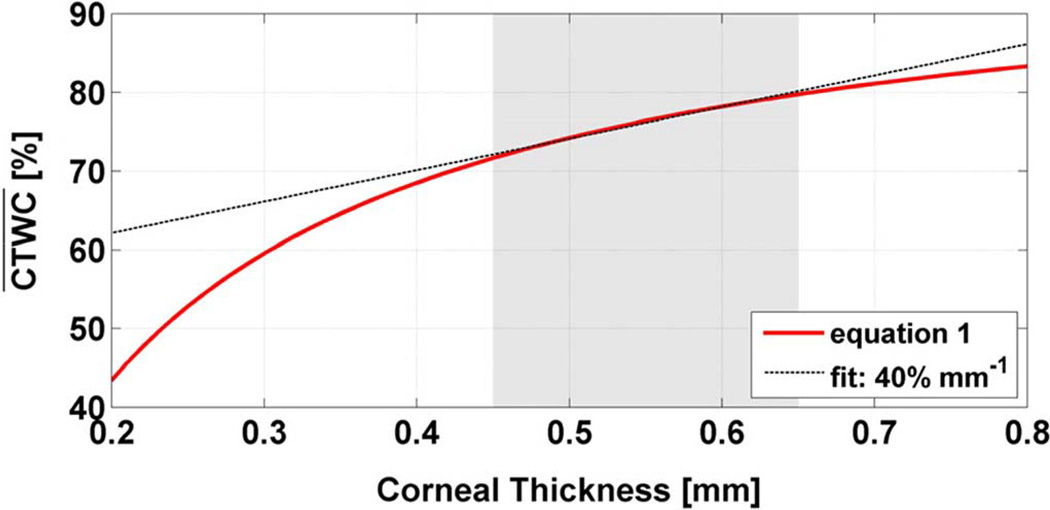
Currently available practice limits the in vivo measurement of CTWC to extrapolation using the central corneal thickness (CCT) measurements usually performed with ultrasound or optical coherence tomography (OCT) based pachymetry [12]. These operate on the assumption of a monotonically increasing relationship between CCT and the average water content of the cornea [38]
| (1) |
This relationship was established in 1965 from the empirical fit of 11 healthy ex vivo human corneas from a cornea bank and deviations of 20% or greater are seen in the data [39]. Additionally, the model does not account for physiologic corneal thickness variation [21]. For example, healthy patients with corneas too thin to be considered candidates for photorefractive surgery (< 450 µm, [40]) have predicted water content values of ~70%, a level not theoretically possible while the cornea is still attached to the eye. Additional inaccuracies plague the utility of the linear fit which predicts a dehydrated cornea thickness of ~127 µm [39] where the generally accepted thickness is >200 µm [38]. A plot of (1) is displayed in Fig. 2 where shaded area spans the physiologic range of thicknesses observed in healthy cornea and the dotted line is a linear fit to (1) within the physiologically relevant range (shaded region) with an approximate slope of 40%/mm.
Fig. 2.
Plot of (1) with physiologic relevant thickness denoted by the shaded region. A linear fit to (1) within the shaded region is represented by the dotted line.
The small linearized slope of (1) suggests that pachymetry is an insensitive measure of CTWC. Thus, while ultrasound or optical based pachymeters provide extremely accurate distance measurements (with axial resolution on the order of 10 microns [41]) the mapping from thickness to water content is extremely inaccurate and severely limits the utility of the technique. Furthermore, pachymetry is a point measurement system, thus precluding the possibility of determining the spatial distribution of water in corneal tissue.
To further explore the uncertainty in CTWC diagnostics, it is illustrative to compute the CTWC sensitivity required to detect specific diseases and pathologies. In our previous work [14], we computed the following sensitivities referencing [42]–[44] and (1): graft rejection 2.7%, Keratoconus 1.6%, Fuchs dystrophy 1.0%, and Refractive surgery 2.0%. However, if statistically significant CCT values are taken from a different set of research the computed required sensitivity for these pathologies are: Keratoconus 4% [45], Fuchs dystrophy 1.0% [46], and refractive surgery 8.5% [47]. Further, CCT measurements of in vivo healthy cornea suggest that average CTWC increases of <5% result in slight to no effect on the refractive, transparency, and mechanical functions of the cornea and diurnal percent increase in CTWC can occur during sleep [48]. The stark disagreement and inconsistency between published CCT values and estimated CTWC demonstrate the poor understanding between the coupling of thickness and CTWC and indicate that CTWC measurement is both a clinical and a basic research problem.
D. Diagnostic System Research
Many optical techniques for corneal water content have been researched, including (OCT) [49], near infrared reflectometry (laser Doppler) [50], and confocal Raman spectroscopy [51]. Of these only confocal Raman has provided an absolute measurement of corneal water content [52]. However, the scattering cross section of water is so low that measurements must use clinically unacceptable integration times or unsafe fluence levels [52]–[54]. The axial TWC measurements used in Section IV were obtained with confocal Raman spectrometer having a 25 mW laser at 514.2 nm. This was coupled to a microscope objective and yielded an energy density of ~1273.2 W/cm2 at the probing point within the cornea. The maximum permissible exposure (MPE) allowed by ANSI standard Z136.1 [51] at this wavelength is 1 mW/cm2 and while it is difficult to deduce from the text how the optics of the eye projected the remaining, unabsorbed beam onto the retina it is likely that MPE values were exceeded by a large margin.
The significant role that CTWC plays in visual acuity coupled with the limitations of clinically accepted techniques and current research present a key opportunity for THz reflectometry. The cornea is essentially a smooth vessel of water in a nearly lossless homogenous matrix on the surface of the body with physiologic variation in thickness that can be accounted for with companion optical methods. Given the tissue constituents, geometry, and location, CTWC measurement offers potentially higher accuracy than that of any other in vivo application studied in THz medical imaging to date. Furthermore, the required field of view (FOV) is small (~1 cm diameter) and does not change based on the pathology, disease, or injury of interest which allows for fast acquisition speed and the possibility of one type of system design for all ophthalmologic applications.
III. THz Electromagnetic Properties of Cornea
A. Model
In vivo, the corneal matrix is ordered and displays birefringence at optical wavelengths [55]. However, because optical anisotropy has not yet been demonstrated at THz wavelengths, we treat the cornea as a smooth, homogenous, isotropic medium whose dielectric constant is defined by axially varying tissue water content. Further, Collagen fibers, scars, vesicles, and other structures found in the cornea are very small (~50 µm) compared to the shortest wavelengths considered by this work (~500 µm) so their contributions to the THz electromagnetic properties of the cornea are considered negligible. Additionally, the surface of the cornea is optically smooth with respect to THz wavelengths with a root mean square (RMS) surface height standard deviation of ~0.129 µm [26].
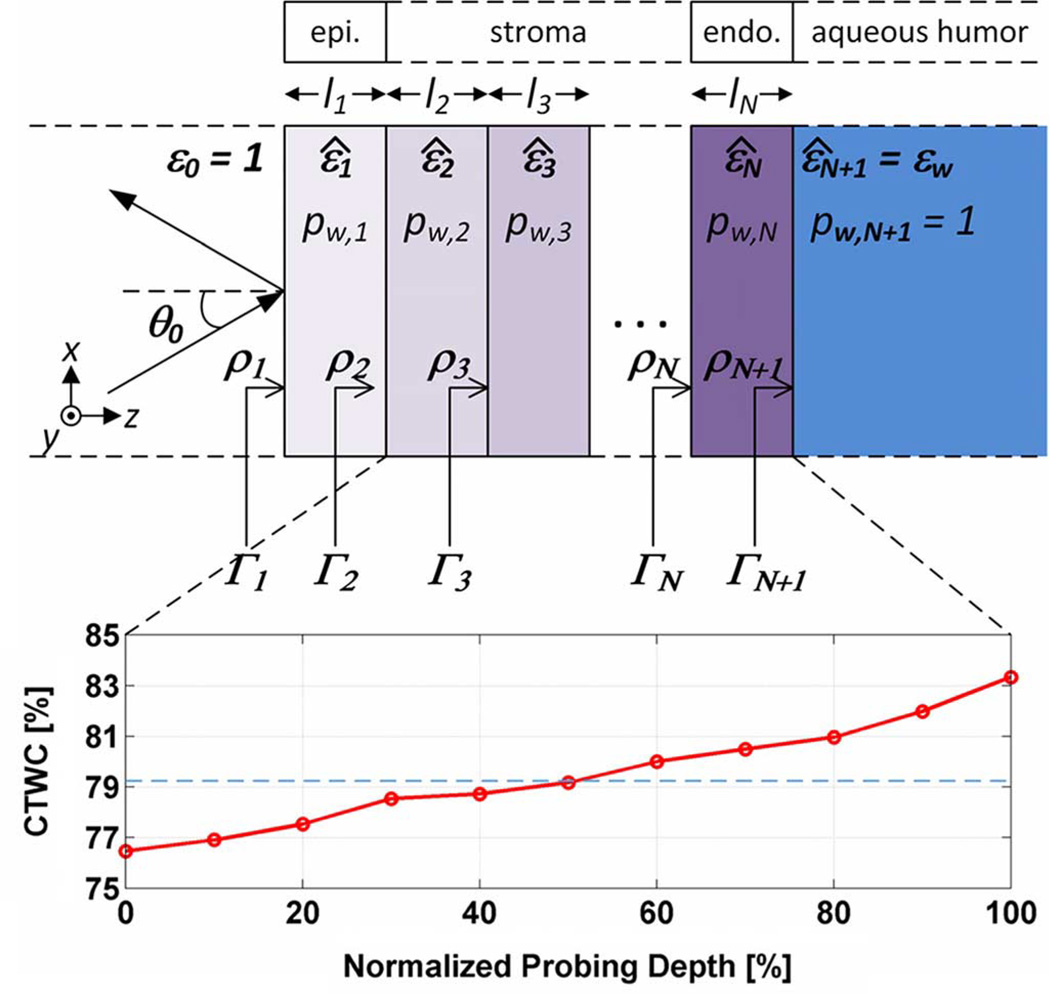
Although curved with a radius close to that of the typical eye, the THz radiation is assumed to be focused to a spot size much less than either the radius or the corneal diameter. Then, the cornea's electromagnetic properties can be modeled with stratified media [56] and effective media theories [57]. This is accomplished by discretizing the cornea into adjacent slabs of material whose dielectric function is homogenous and water-content dependent. The electric permittivity of each slab is then calculated using Bruggeman effective media theory [57]–[59] where the majority of the slabs represent the stroma due to its significant thickness as compared to all the other layers. Following computation of layer properties, the aggregate reflectivity is computed via the transfer-matrix method [60]. This approach is summarized in Fig. 3, where the first layer is occupied by the epithelium and the final layer is modeled as a half space of liquid water representing the aqueous humor (Fig. 1). Layers 1 through N + 1 represent the discretized, axially dependent CTWC.
Fig. 3.
Effective media model for cornea. The axial water content curve is representative of healthy cornea and has been adapted from [52]. The first layer in the stack represents the epithelium and the final layer is the aqueous humor (body of water).
The axially varying water content simulated via the stratified media model is adapted from [52] and was utilized in our previously published work [14], [15].
The data in [52] was obtained from rabbit eyes (N = 10) using confocal Raman spectroscopy. The trend is plotted in Fig. 3 as a function of normalized depth. The trend was generated by taking 11 evenly spaced points over the thickness of each cornea for 10 rabbits, converting their position to normalized thickness, and then averaging each point independently to produce the final presented trend.
An average tissue water content of the cornea of 79.4% was computed by averaging the trend along the normalized thickness axis. Note that this is higher than the accepted healthy cornea level of ~78%. Also note (discussed in greater detail in Section II-D) the fluence level utilized to acquire these measurements most likely perturbed tissue properties in the cornea which may have affected the overall accuracy of the measurement. However, to our knowledge, this is the most complete in vivo dataset of CTWC that currently exists.
The methodology for simulating the frequency dependent aggregate properties of tissue has been presented in prior publications [14], [15], [61]. A brief summary of this method is provided next.
The frequency dependent dielectric properties of water are captured by the double Debye model [62] [see (2)] with the same constitutive parameters and relaxation frequencies (ε0, ε1, ε∞, f1, f2) that were presented previously [14], [15]
| (2) |
The collagen and water of the cornea cannot be isolated volumetrically which allows the implementation of the binary-mixture Bruggeman model [57] [(3)] where εc is the frequency invariant dielectric constant of collagen, εw is the frequency dependent dielectric function of water computed with (2), pw,i is the water volume fraction, and ε̂i is the effective permittivity of the layer that satisfies the effective media relation in
| (3) |
The effective electrical length of the layer i is given in (4) where ε̂i is the complex, effective permittivity of layer i computed with (3), θi is the complex angle of refraction computed with Snell's law, and li is the layer physical thickness [60]
| (4) |
For the simulations presented below the discretized layer thickness was set at ~10 µm where reflectivity simulation results with varying layer thicknesses converged to a differential less than 10−6. Thinner layers resulted in insignificant increases in precision at the expense of increases in computational complexity. The Fresnel reflection coefficient (elementary reflection coefficients arising from boundary conditions) from layer i − 1 to layer i can be written as a function of the complex effective dielectric constants and the complex permittivities and electrical lengths (5) [63]
| (5) |
Note that the Fresnel coefficient has been written for TE polarization. This polarization has been shown to produce reflectivities and CTWC sensitivities larger than the TM polarization for any oblique incidence angle [7], [61].
The total aggregate electric field Г recursive reflection coefficient from layer i to the stack consisting of layers i + 1 to N + 1 is given in (6) where ГN + 1 is defined as the Fresnel coefficient between the final layer of the cornea and the aqueous humor and Г1 is the total reflection of the cornea [63]
| (6) |
B. Selection of Collagen Dielectric
Surprisingly, a large body of work on the THz dielectric properties of proteins does not yet exist in the literature and to our knowledge, a complete study of the spectroscopic properties of pure collagen do not exist. One of the most quoted studies on biologically relevant proteins is by Markelz and Heilweil, where a terahertz pulsed spectroscopic (TPS) system was used to assess the absorption coefficients of DNA, bovine serum albumin (BSA) and collagen, and the real index of DNA and BSA, between 0.1 and 2.0 THz [64]. They measured a BSA refractive index of ~1.7 with negligible loss tangent, suggesting that the real part of the dielectric constant is ~2.9. In the absence of direct spectroscopic measurements of pure collagen, the properties of BSA were deemed a good approximation and used throughout the present work.
Therefore, in (3), εc = 2.9, and the absorption coefficient of the ith layer in the corneal tissue at any particular frequency can then be determined by (7), where and are the real and imaginary components respectively of the dielectric function of the ith layer
| (7) |
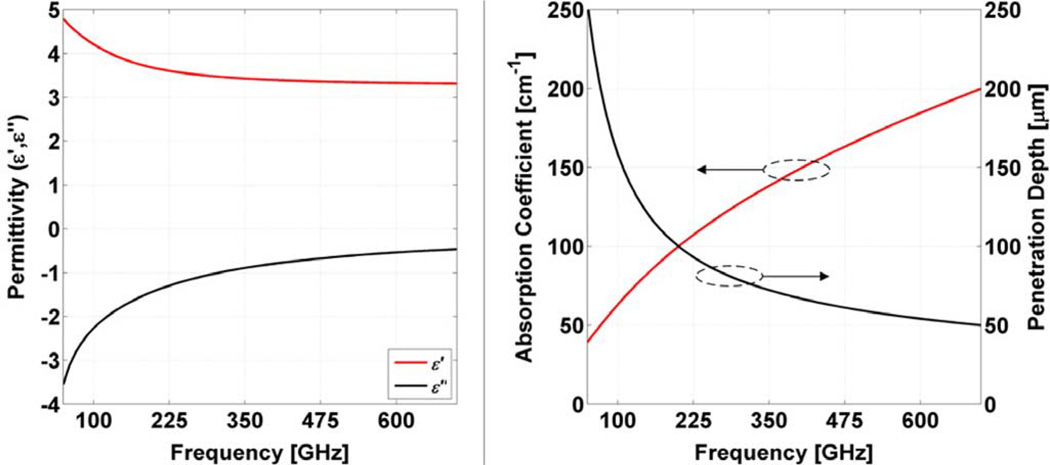
The aggregate dielectric function of corneal tissue, and the expected absorption coefficient and penetration depth are shown below in Fig. 4 over the frequency range utilized in the simulation results in Section IV.
Fig. 4.
Dielectric properties of cornea. (left) real and imaginary parts of the corneal tissue at 79.4% water by volume. (right) computed absorption coefficient and depth of penetration.
C. CTWC Sensitivity
In our earlier work we computed CTWC sensitivities as a function of frequency for a finite set of tissue water contents. For the present study, we are interested in the expected reflectivities and CTWC sensitivities at 100 GHz and 525 GHz corresponding to the operational frequencies of the two systems presented in the companion manuscript [65]. The band normalized power reflectivity of the cornea is described in (8) where Ss(f) is the source power spectral density, SD(f) is the detector spectral responsivity, pw is the water fraction, d is central corneal thickness, f is the illumination frequency, and Ωf is the system equivalent noise bandwidth (ENB). Equation (8) can be interpreted as the weighted average of the corneal THz reflectivity where the weights are determined by the spectral response of the system [7]
| (8) |
Note that (8) models a broadband measurement as an incoherent sum. The intrinsic sensitivity of the cornea reflectrometry system can be computed as the gradient of (8) with respect to parameters of interest. For example the CTWC sensitivity and corneal thickness sensitivity are given in (9) and (10), respectively
| (9) |
| (10) |
In one design of a sensing system, one could choose to maximize (9) and minimize (10) thus maximizing CTWC sensitivity while thus minimizing thickness sensitivity uncorrelated to CTWC. However, given the powerful a priori knowledge of corneal layer geometry one could design a system that is sensitive to both, similar to what is done in ellipsometry [66].
The CTWC sensitivity space described by (2)–(10) is rich with a number of parameters that can be varied, including CTWC gradient, thickness, incidence angle, window properties, etc. Additionally, the dynamics of CTWC gradients in vivo are not well known due to the lack of accurate, non-invasive CTWC sensing methodologies. To thoroughly explore this space three CTWC gradient models were developed that, to first order, capture CTWC gradient perturbations for all known corneal disease/pathology states.
D. CTWC Gradient Models
Water transport is regulated at the epithelium through water loss to the environment and moisture gain from tears and at the endothelium through the active exchange of water with the aqueous humor (Fig. 1). As discussed above, many corneal diseases significantly perturb CTWC and the perturbation often originates locally in either the epithelium or endothelium. Therefore, it is appropriate to analyze two cases: 1) where CTWC of the stroma adjacent to the epithelium is fixed and the CTWC of the stroma adjacent to the endothelium is varied and 2) where the opposite occurs. From here on these will be referred to as “Pinned Front” and “Pinned Back,” respectively. As a comparison, a third model entitled “global” was developed that modulates the CTWC of the entire stroma equally. Pachymetry treats changes in CTWC as occurring globally so the global model provides a benchmark against clinical results. These models are summarized in the following descriptions.
-
1)
Pinned front CTWC: This simulates the expected change in CTWC gradient for diseases such as Fuch's Dystrophy where the endothelium (Fig. 1) fails and the cornea starts to hyperhydrate from the backside. (H1(x) in (12).
-
2)
Pinned back CTWC: This simulates the expected change in CTWC gradient for dry eyes or perturbed front side CTWC from stromal ablation (Fig. 1) during LASIK/LASEK surgery. (H2(x) in (13)). Note that this model is a good approximation to the drying dynamics observed in [52].
-
3)
GLOBAL CTWC: This simulates the expected increase in corneal CTWC due to keratoconus where the entire volume of the cornea hyperhydrates. (H3(x) in (14).
Diffusion, which is thought to contribute to the overall distribution of TWC in the cornea [67], suggests that in gradient types 1 and 2 the deviation of CTWC from normal may be exponentially distributed. This behavior, as well as the global shifts, were modeled with the following equations:
| (11) |
| (12) |
| (13) |
| (14) |
In (11)–(14), x represents the normalized depth [0%, 100%], pw(x) represents the experimental data displayed in Fig. 3, g(x) represents the exponential deviation from normal pw(x), σ is defined such that the range of g(x) is bounded by [0, α] and α is the variable that modulates the CTWC and is defined such that the average CTWC, computed by taking the mean value of (12)–(14) are equal and satisfy the following equivalence relations:
| (15) |
| (16) |
Equivalence relations (15) and (16) state that if the candidate CTWC gradients are perturbed with the variable α as described by (11)–(14) then, while the water content at any particular depth might be significantly different, the average water content, as computed along the axis (15) will always be equal.
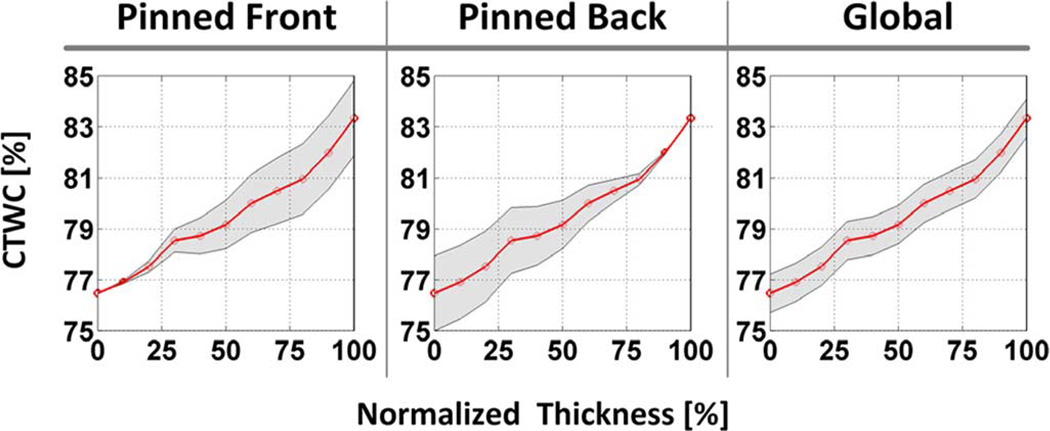
Gradients 1, 2, and 3 are displayed in the left, middle, and right panels, respectively, of Fig. 5 where the red line denotes axially varying CTWC (same as the curve in Fig. 3) and the shaded neighborhoods indicate the ranges explored in this simulation by varying the parameter α.
Fig. 5.
Candidate CTWC gradients for CTWC sensitivity simulations. (left) pinned front, (middle) pinned back, and (right) global.
Note that while the equivalence relation in (16) holds for all α, the water content at any point along the axis must remain within the range [0%, 100%] thus limiting α to the domain [−p(0), 100−p(100)] = [−76.77, 16.66]. Also note that while a CTWC range of [0%, 100%] is theoretically realizable, physiology limits the range somewhere closer to [75%, 85%] while the cornea is attached to the eye, although this is somewhat conjecture since non-invasive, in vivo water content determination (as discussed above) is severely limited.
Equations (11)–(16) enable a direct comparison for the expected THz properties of different pathologies through a shared, common, average CTWC which we denote here as . Currently, corneal pachymetry does not discriminate between distributions but gives a corneal tissue water content that is assumed to be an average of the entire depth. is used in Section IV as a basis of comparison of signals from different pathologies. For the remainder of the manuscript the following labeling is used
| (17) |
| (18) |
IV. Simulation Space Results
A. Theoretical Test Systems
A brute force exploration of the spectral (center frequency and bandwidth) dependence of (2)–(10) would be intractable for a single publication. Therefore, the following analysis was limited to incoherent direct detection of pre-defined center frequency and bandwidths and focused on the performance of two different quality factor values (Q = 5,50) centered at 100 GHz and 525 GHz. These values were chosen to mimic the two systems [7], [14], [15], [61], [68]–[70] that were used in our in vivo rabbit imaging trial discussed in the companion manuscript to this publication [65]. Additionally, as can be seen from the penetration depth data in Fig. 4, the 100 GHz system is more sensitive to the finite thickness of the cornea than the 525 GHz and comparison of the theoretical results enables exploration of the thin film like properties of the cornea.
The reflectivity of the whole space is calculated with a modified version of (8), given in (19), where the source and detector spectra have been combined into a single power spectral density (PSD) Sn(f).
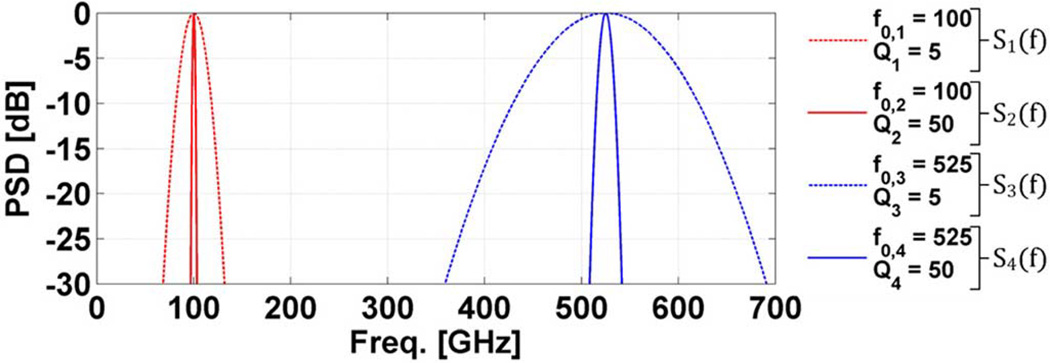
A plot of the four spectral densities is displayed in Fig. 6. A general case Gaussian spectrum was utilized for the PSDs with functional forms described in (19)–(21) where f0,n and Δfn are the center frequency and bandwidths respectively of Sn(f). and is defined (21) such that Δfn is the full-width at half-maximum (FWHM) of Sn(f)
| (19) |
| (20) |
| (21) |
Fig. 6.
System power spectral densities (PSD): center frequencies and quality factors with Q = 5, 50 at 100 and 525 GHz.
Note that while these Gaussian spectra are representations of the system spectra, they do not capture the asymmetry about the center frequency observed in the majority of THz imaging system implementations. For the following analysis, system PSDs S1,2(f). are referred to as millimeter wave spectra and PSDs S3,4(f) are referred to as THz spectra.
B. Millimeter Wave (100 GHz) Reflectivity
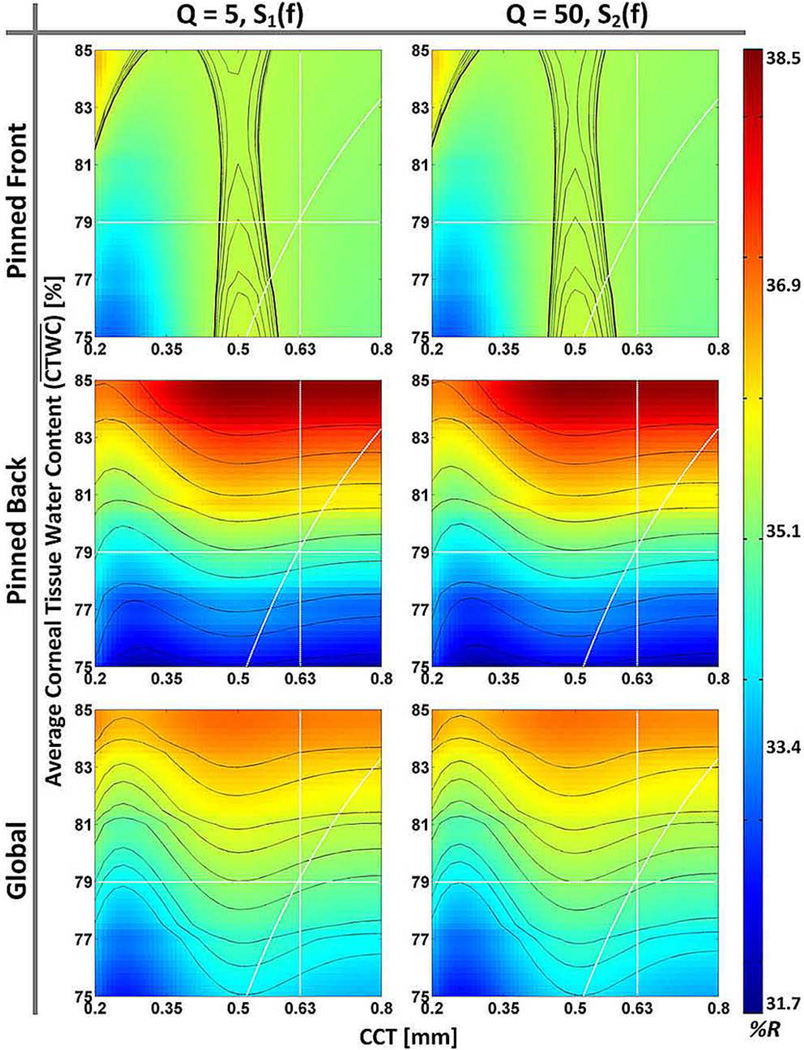
The expected corneal reflectivity space (8) for the band covered by the millimeter wave system are displayed in Fig. 7 for the three candidate CTWC gradient distributions and two system quality factors for a total of 6 unique pairs. The x-axis spans central corneal thickness (d in (8)–(10)) values from 0.2 to 0.8 mm and the y-axis spans average CTWC volume fractions () values (axial average of p in (8)–(10)) from 75% to 85%. The pixel values are plotted in units of % reflectivity as indicated by the color bar located on the right side of the figure, and all six figures are mapped using a shared color scale.
Fig. 7.
Dependence of corneal reflectivity on CTWC and thickness computed at 100 GHz for the cases of (top row) pinned front CTWC change (middle row), global CTWC change, and (bottom row) pinned back CTWC change. Each case was simulated with a source/detector Q of 5 and 50. All figures are displayed with a common colormap, with pixel intensities representing percent reflectivity.
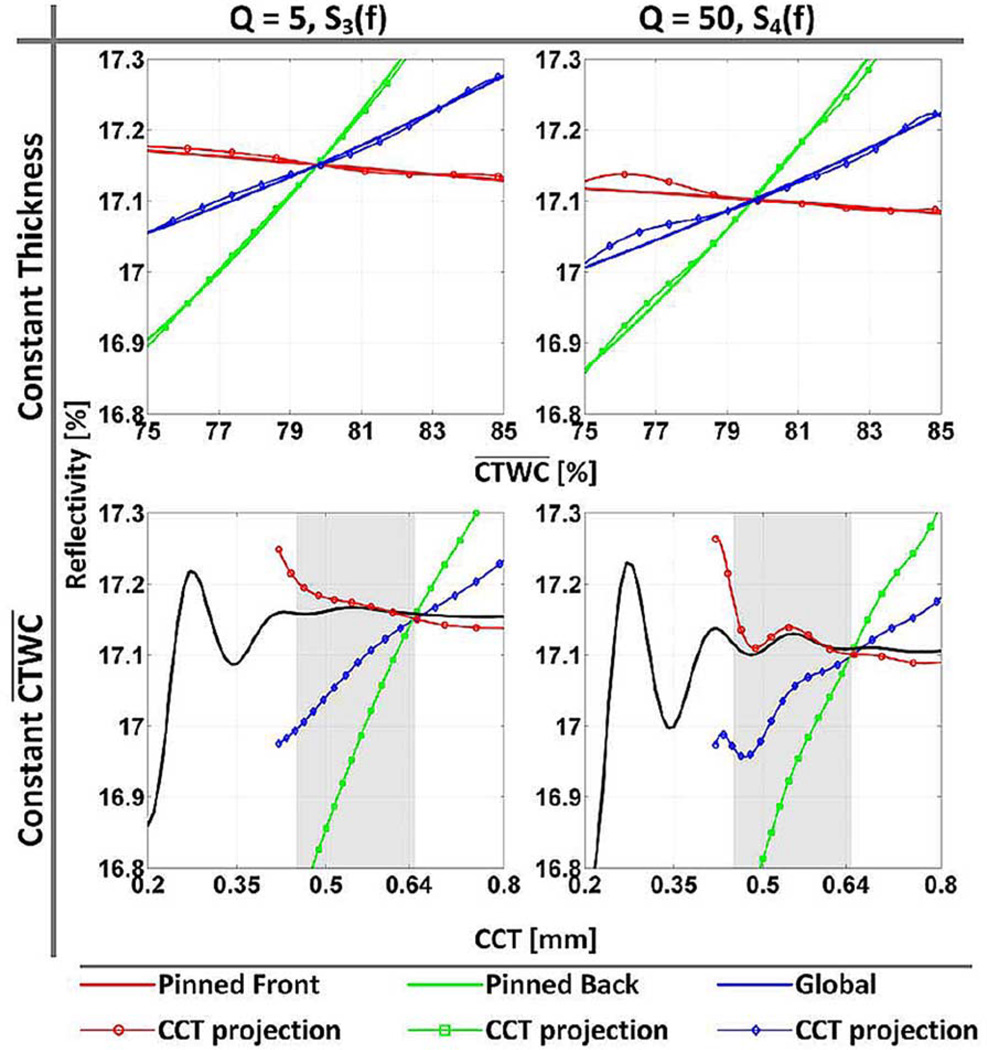
In addition to the space, the expected reflectivities of a human cornea with an average thickness (0.625 mm) were computed for discrete of 75%–85%, in increments of 1% for each of the three CTWC gradient generating functions [see (12)–(16)]. Isoreflection lines corresponding to those reflectivities were superimposed on their respective spaces (one space per generating function). Each contour line in the level set represents all possible CTWC/thickness pairs which yield the same reflectivity. Importantly, when the corneal thickness is unknown, the contour's range of y values represents all possible mapping to a given reflectivity. The isoreflection lines also summarize the impact of thickness sensitivity on sensing given sufficient system signal to noise ratio (SNR) as system sensitivity can be inferred from the contour line density. For the millimeter wave system under the Pinned Front case, determining without a measurement of the cornea's thickness is impossible. In the Global Shift case, the isoreflection lines span an interval of 1.5% (ml/ml), and in the Pinned Back case, the isoreflection lines span an interval of 0.6% (ml/ml).
Three white dotted lines are also superimposed on each contour map exploring three different variation profiles. The horizontal lines correspond to the expected corneal reflectivity when remains fixed at 79% and the thickness is varied. The vertical line explores the converse where the thickness is fixed at 0.625 mm and is varied from 75% to 85%). The curvilinear line indicates the profile described by (1) which predicts a coupled relationship between TWC and thickness.
As indicated by the color bar in Fig. 7, the reflectivity ranges from 31.7% to 38.5%. Further, while the entire reflectivity range is spanned by the pinned back gradient, the global gradient variation spans ~90% of the reflectivity range and the pinned front case spans ~80%. This result is somewhat logical as the high absorptivity of water confines the majority of the millimeter wave-tissue interaction to the cornea's top surface. In other words, these plots indicate that the modulation of the THz signal due to changes in top layers of the cornea is masking the contributions from the underlying layers. Consequently, the presence of the aqueous humor is somewhat screened by the loss incurred from transmission through the upper layers.
Of interest is the apparent invariance of corneal reflectivity to system bandwidth. The maps for the Q = 5 and Q = 50 systems are nearly identical, and the discernable differences appear only upon study of the isoreflection lines in the pinned front case. This is due to the extremely thin geometry of the cornea with respect to the wavelength within the cornea: ncornea ~ 2.91 − j1.45, λcornea (f = 100 GHz) = λc,100 ~1.03 mm > tcornea. At these frequencies m · λcornea/2 > tcornea for all integers m > 1, thus only one longitudinal mode is supported by the lossy cavity, and the difference between integrating over a larger bandwidth (Q = 5) and smaller bandwidth (Q = 50) does not change the number of accessed longitudinal modes.
This concept is further explored by analyzing pixel intensities along the white dotted lines in Fig. 7, which are displayed in Fig. 8. The constant thickness (0.625 mm), varying profiles are displayed in the top row and the constant (79.4%), varying thickness profiles are displayed in the bottom row for the pinned front (solid red), pinned back (solid green), and global (solid blue) CTWC variation cases. Further, the CCT projections for each CTWC gradient-system Q pair are plotted in a dotted line style with color corresponding to its associated CTWC gradient. These plots allow for a straightforward comparison between the one fixed parameter, one varied parameter behavior, and the coupled behavior predicted by (1). The shaded region indicates the average range of thicknesses observed in human patients.
Fig. 8.
Millimeter-wave corneal reflectivity profiles. (top row) constant thickness, varying tissue water content. (right) constant thickness, varying CTWC. These profiles are plotted in solid lines for the three different variation types; pinned front, pinned back, and global. The dotted lines are the projection of (1) onto each axis.
The fixed thickness, varying curves all display monotonic, positive slope behavior, and the slope increases as the bulk of the change in CTWC gradient occurs closer to the front surface indicating, again, that surface layers dominate the electromagnetic properties. The CCT projections closely follow the constant thickness-changing variation for both the global and pinned back cases but deviate somewhat in the pinned front case. The pinned front CCT projection displays both positive and negative slopes suggesting that diseases such as Fuch's dystrophy may in fact present a millimeter wave system with a decreasing signal for an increase in both thickness and .
The constant , varying corneal thickness curves resemble that of a lossy etalon formed by the air/cornea and cornea/aqueous humor interfaces separated by a distance equal to the corneal thickness. This behavior is evidenced by the peak of each CTWC gradient case occurring at ~500 µm (λc,100/2). The three CTWC variation cases scale with corneal thickness variation and converge at the healthy CTWC percentage (79.4%) thus the three profiles are all represented by the same solid line (black) in Fig. 8. Note that the integer multiple of wavelength fraction relationships are approximate since any candidate percent CTWC is axially averaged. Due to the gradient, the cornea index is lower in the posterior and greater in the anterior region. Furthermore, due to absorption the overall response is affected more by the posterior index.
Similar to previously discussed results, the CCT cuts demonstrate marked differences between the three CTWC cases. Again, pinned back and global gradients demonstrate monotonic, positive slopes while the pinned front case resembles a highly attenuated standing wave, albeit with a slight decrease in the distance that yields peak reflectivity. One would expect the thickness of the cornea to increase as the TWC is increased, thus the millimeter-wave system may be able to delineate between pinned front and the pinned back, global cases if millimeter reflectometry trends were available. These findings may assist in the diagnosis of Fuch's dystrophy and corneal graft rejection.
C. Reflective THz Imaging System
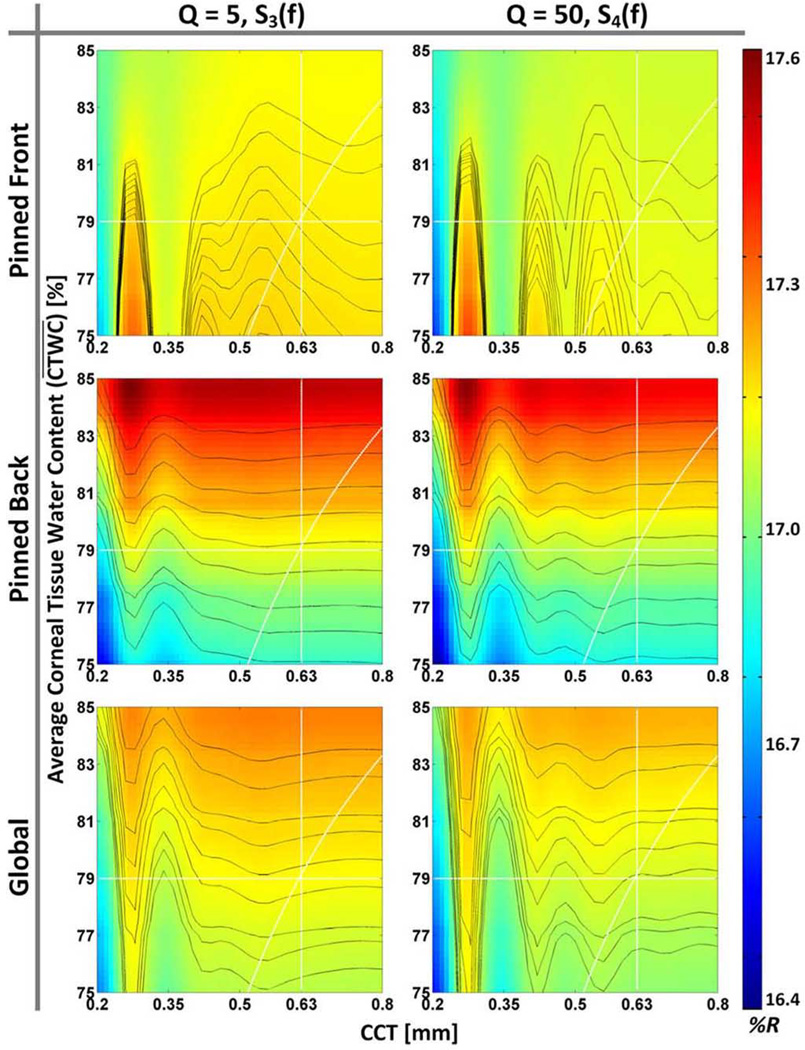
The expected corneal TWC-thickness reflectivity space (8) for the band covered by the 525 GHz system for the three candidate TWC distributions and two system quality factors are shown in Fig. 9. The spaces have the same x limits (0.2–0.8 mm) and y limits (75%–85% hydration) as Fig. 7. Contour lines representing hydration-thickness sets with identical reflectivities were superimposed in the same manner as Fig. 7, as well as the pixel intensity paths exploring thickness variation, variation, and simultaneous and thickness variation.
Fig. 9.
Dependence of corneal reflectivity on CTWC and thickness computed at 525 GHz for the cases of (top row) pinned front CTWC change (middle row), global CTWC change, and (bottom row) pinned back CTWC change. Each case was simulated with a source/detector Q of 5 and 50. All figures are displayed with a common colormap, with pixel intensities representing percent reflectivity.
The six plots in Fig. 9 are displayed with a shared color map and the reflectivity ranges from 16.4% to 17.6%. The plots in Fig. 9 display overall lower reflectivity and a smaller reflectivity range than that displayed in Fig. 7 due to the significant decrease in water's permittivity and the intrinsic sensitivity (dR/dp) when comparing center frequencies 100 and 525 GHz.
Similar to the millimeter wave system, the entire reflectivity range is spanned by the pinned back case. However, the pinned front case spans a larger reflectivity space than that of the global variation case, demonstrating 80% and 70% of the total range, respectively.
There is also a noticeable difference in the thickness/CTWC maps between the Q = 5 and Q = 50 candidate systems. At 525 GHz, the approximate refractive index of the cornea is ncornea ~2.1 − j0.45 resulting in a central illumination wavelength in the cornea of λcornea (f = 525 GHz) = λc,525 ~0.272 mm in numerous integer multiples of ~λc,525/2, a result that issupported by the lossy cornea cavity. This effect can be seen by comparing the spaces for the two quality factors at each CTWC gradient case where each Q = 50 space displays a larger range than its Q = 5 counterpart. This effect is also observed in the profiles of the contour lines that indicate the local extremum of the space.
Pixel intensities from the indicated profiles are shown in Fig. 10 with the same reference points (constant thickness = 0.625 µm, constant CTWC = 79%) and set of Qs (Q = 5, Q = 50) as the millimeter wave reflectometer and are displayed in the same arrangement as Fig. 8. The broad band (Q = 5) fixed thickness, varying shows monotonic increases in reflectivity as is increased for the pinned back and global variation cases and monotonic decreases in reflectivity for the pinned front case. Similar to the 100 GHz system, the CCT projections are aligned with the constant thickness, varying TWC profiles although the deviation is less, especially for the pinned front case. Increasing Q still results in little deviation between the constant thickness paths and the CCT projections, although an increase in path curvature is evident.
Fig. 10.
525 GHz corneal reflectivity profiles. (top row) constant thickness, varying CTWC and (bottom row) constant CTWC, varying thickness.
The fixed , varying thickness paths show significant attenuated standing wave behavior with the first visible peak occurring at 0.276 mm (~2 · 1/2 · λc,525); the approximate wavelength of illumination inside the cornea. The first valley is at ~5/4 · λc,525 and there appears to be a second peak at ~3/2 · λc,525 although the high absorption constant of water at these frequencies is overpowered the constructive interference arising from multiple passes within the cornea. Decreasing the bandwidth (Q = 50) leads to an amplification of standing wave effects as evidenced by the plot in the bottom right of Fig. 10. The extinction ratio is larger and local maxima are discernable at ~2 · 1/2 · λc,525, ~3 · 1/2 · λc,525, and ~4 · 1/2 · λc,525.
The CCT projections are also markedly different from those observed with the millimeter wave system. In the Q = 5 case, pinned back and global TWC distributions show monotonically increasing behavior while the pinned front shows montonically decreasing behavior. In the narrow band case (Q = 50), the pinned back case displays significant increases in reflectivity while both the global and pinned front cases show standing wave like behavior.
D. Discussion
Like the millimeter wave system, the THz system cannot be used to determine CTWC in the pinned front scenario without a measurement of the cornea's thickness. However, in stark contrast to the 100 GHz system, in the global shift case, the THz systems' isoreflection lines span a diagnostically irrelevant interval of ~0.2% (ml/ml), and in the pinned back case, the isoreflection lines span an interval well below 0.1% (ml/ml). Given these results, the THz system can sense CTWC for diagnostic applications in the Global Shift and Pinned Back cases without an accompanying thickness measurement.
Similar to Fig. 8, an etalon-like effect is observed in the constant CTWC plot of Fig. 10. The reflectivity varies as the cornea-air interface and cornea-aqueous humor interfaces are moved apart due to interference effects that appear and disappear with the varying optical path length. However, in contrast to the millimeter wave corneal paths (Fig. 8), the Q = 5, THz system has a minor variation in reflectivity due to thickness variation in the physiologically relevant thickness range. This behavior is due to both the differences in the cornea's skin depth and to the broadband nature of the Q = 5, THz system. At 525 GHz, the skin depth of the cornea is significantly smaller than that at 100 GHz (0.58 mm and 1.1 mm, respectively), causing the front layers of the cornea to more strongly mask effects from the back layers of the cornea. This phenomena causes the interference effects formed by the cornea's thickness to be negligible relative to the cornea–air reflection, making the CTWC measurement for THz systems less sensitive to thickness variations than that acquired with the millimeter wave systems.
The broadband nature of the THz system also reduces the etalon-like interference effect, because the sign of the interference effects is frequency dependent; optical path length varies with frequency, and the variations in the optical path length phase shift different frequencies by different amounts. As a result, systems that span multiple frequencies sum interference effects that oscillate in sign (which cancel one another), meaning that the interferences’ relative contribution to the reflectivity decreases as the operating bandwidth grows.
One interesting point that arises from this analysis is the differences between the predicted CTWC sensitivities calculated with each gradient type. This finding presents an excellent opportunity for THz corneal sensing as there are no one-to-one mappings between axial CTWC distributions and pachymetry measurements, and the simulations suggest that an ensemble of THz reflectivity measurements may allow one to ascertain this information.
V. Spectroscopic Measurements
To explore the utility of the pinned front, pinned back, and global CTWC gradient types, the expected change in reflectivity for a change in water content as a function of frequency was computed for each type and then compared to experimental data extracted from measurements of ex vivo corneas. These corneas were prepared by immersing them in polyethylene glycol (PEG) solutions of varying concentrations for 3 days to produc water concentrations (confirmed by dry weight to wet weight ratios) ranging from 78.8% to 91.5%. 78.8% was the lowest concentration achievable that still produced a smooth surface where surface roughness would not affect the measured reflectivity. 91.5% somewhat exceeds the physiologically relevant range, but the reflectivity curves are approximately linear over a broad range and additional data points improved the quality of fit.
In addition to the three CTWC gradient types, a half space model was generated using the Double Debye and Bruggeman described in (2)–(16) and compared to the gradient types. This model was generated for two reasons.
-
1)
It was hypothesized that the perturbation of CTWC ex vivo through the use of PEG solutions led to reduction of the axial water content gradient due to the absence of an active endothelium layer thus improving utility of a half space.
-
2)
It is instructive to evaluate the results of a simplified model as compared to a more complicated, ostensibly more accurate model although the accuracy of the half space in vivo is untested.
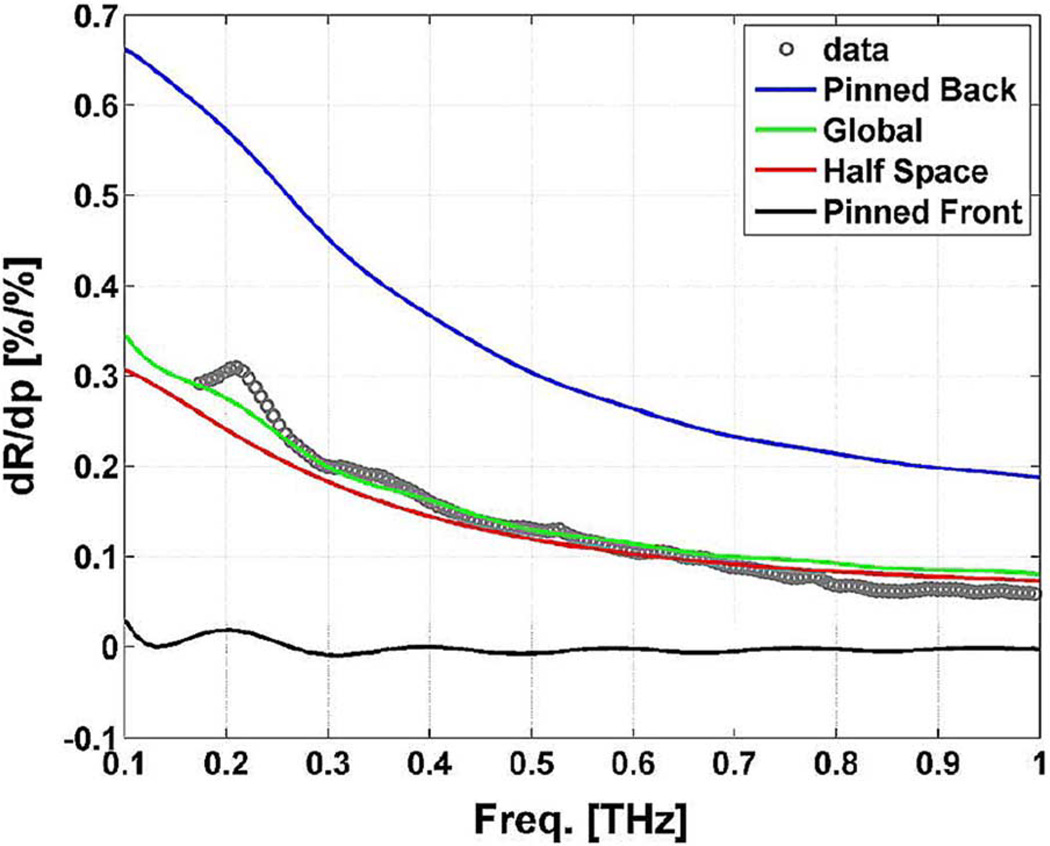
The results are displayed in Fig. 11. The data was acquired with a THz TDS system with spectral operating limits spanning ~180–1000 GHz, which does not allow for investigation of 100 GHz properties but allows for analysis of trends dependent on illumination frequency. Because in vivo axial gradients are difficult to replicate ex vivo and impossible to verify non-destructively the hyperhydration/dehydration protocol was designed to modulate the CTWC globally and reduce the magnitude of the axial gradient. The experimental results support the intended setup. The pinned front gradient type demonstrates low sensitivity and a standing wave like behavior that continues throughout the entire computational domain. The pinned back gradient type demonstrates the largest sensitivity due to the largest changes in CTWC occurring in the top layer of the cornea. The global shift gradient type and half space produce similar results and demonstrate very good agreement with the experimental data. This agreement provides strong evidence that the Bruggeman, Debye, and stratified media theories are appropriate for corneal modeling and, more specifically, that the Bruggeman model enables an accurate, and straightforward method to calculate numerical derivatives in the reflectivity space.
Fig. 11.
CTWC sensitivity fit to data confirming the validity of the Bruggeman model. Note the effective frequency invariance of corneal tissue constituents. Data obtained from [15].
VI. Comments on the Corneal Model
There are a number or key points regarding the applicability of the presented model. First, that the model assumes no local corneal curvature and uniform plane wave illumination. Real corneas are curved and a significant majority of THz imaging systems illuminate with a Gaussian or quasi-Gaussian beam focused by a broadband reflective or refractive optic. Additionally, only the center of the cornea is considered. Pachymetry is based entirely on central corneal thickness measurements, and it is currently impossible to apply theory at the periphery of the cornea. To this end, our model is sufficient at this stage of development. Future work will have to investigate the limitations of this empirical relationship.
Second, there are currently no means of independently verifying the accuracy of the three gradient cases considered because of the inability to axially resolve CTWC distributions in vivo without invasive methods. It should be noted however (as discussed above in Section III.D), that endothelial diseases such as Fuchs’ dystrophy are known to first modulate the water content of the endothelium, and conditions such as dry eyes are known to first modulate the epithelium. Thus while the exact axial variation cannot be verified in vivo, at minimum the hypothesized axial location of perturbation and subsequent modulation in THz properties can be explored.
Third, this model has neglected to include the tear film (Fig. 1). The tear film cannot be replicated ex vivo [71], [72] and its thickness is notoriously difficult to measure in vivo [23]–[26]. The presence of the window in our in vivo animal experiments [65] physically prevented the tear film from forming between the window and cornea, and the presented simulations were designed to model this behavior. Future modeling work will evaluate the effect of the tear film on reflectometry measurements once accurate tear film thickness measurements have been experimentally implemented.
Finally it should be noted that the analysis used a healthy CCT value of 0.625 mm which is what (1) predicts for a value of 79.4% extracted from the data in [52]. This highlights the consistency issues outlined in Section II.C. where healthy CCT measurements have yielded average values of 0.565 mm and ex vivo thermogravimetric analysis has yielded ~78%. Since in vivo measurements are the most accurate and most relevant for the proposed applications and (1) cannot reconcile the discrepancy between in vivo and ex vivo measurements it would be prudent to examine all combinations of healthy CCT and CTWC gradients, and . However, due to space considerations and the relative novelty of in vivo measurement compared to in vivo CCT the analysis was been restricted to a value of 79.4%. The analysis of one pair serves to illustrate the unique, thin film nature of the cornea and it is assumed that the reader can extrapolate the electromagnetic properties of varying pairs from the figures.
VII. Conclusion
An extensive 1D plane wave study of the electromagnetic properties of the cornea was presented. Corneal tissue water content (CTWC) was modeled with a combination of Debye, Bruggeman, and stratified media theory. Reflectivities as a function of frequency, CTWC level, CTWC gradient type, and corneal thickness were computed and then the aggregate reflectivities of subsets of these spaces were explored with four different systems; two centered at 100 GHz and two centered at 525 GHz. Additionally each system was simulated broad band (Q = 5) and narrow band (Q = 50). The results elucidate the complex relationship between TWC, corneal thickness, and RF reflectivity, and demonstrate the utility of different frequency bands and bandwidths.
A key development in the THz sensing of corneal tissue water content presented by this work is the significant difference in THz reflectivity properties of cornea when the variation in CTWC is localized to the epithelial layer, endothelial layer, or is distributed evenly throughout the thickness dimension of the cornea. Localization of CTWC as a function of pathology has been hypothesized but few models exist due to the inability to quantify in vivo CTWC gradients. To our knowledge this paper presents the first ever CTWC gradient models and the first identification of THz reflectivity variations as a function of CTWC perturbations locations.
Numerous published studies explore the theoretical THz electromagnetic properties of tissue, primarily skin, through application of some forms of effective media theory 1D wave mechanics. While the utility of these studies as general case tools is manifest, they cannot be used to calculate the constituents of a specific tissue of interest due to the large physiologic variation of layer thickness, TWC axial distribution, scattering structure distribution, etc. leading to large intrapatient and interpatient variation. Accurate conversion of reflectometry data to axial/spatial distributions of tissue parameters requires a priori knowledge of constituent distribution which may be all but impossible, or at the very least impractical, to acquire in a clinical setting.
The uniqueness of the cornea among all other external structures of the human body lies in its relative lack of geometric and tissue constituent physiologic variability with respect to a THz wavelength. Cornea is composed entirely of water diffused in a low loss collagen matrix and bounded on the posterior side by a body of water. In other words, with respect to THz frequencies, cornea is a lossy thin film lying on top of a lossy termination (aqueous humor). Thus, cornea can present spectrally varying information arising from tissue structure that can be utilized, along with a priori knowledge of tissue structure, to generate high sensitivity, high accuracy maps of CTWC in vivo. Further, due to the thin film properties, reflectivity measurements at multiple frequencies can be combined to deliver simultaneous corneal thickness measurements and thickness resolved CTWC measurements. This measurement capability would be unprecedented in ophthalmology and has the potential to revolutionize the field through early detection of corneal pathology/disease/ injury that are correlated with tissue edema.
Acknowledgments
This work was supported in part by the National Eye Institute under Grant R01EY021590 and by the National Institute of Biomedical Imaging and Bioengineering (NIBIB) under Grant R21EB015084 and Grant R21EB016896.
Biographies

Zachary D. Taylor (S'06–M'09) received the B.S. degree in electrical engineering from the University of California (UCLA), Los Angeles, CA, USA, in 2004, and the M.S. and Ph.D. degrees in electrical engineering from the University of California (UCSB), Santa Barbara, CA, USA, in 2006 and 2009, respectively.
He is an adjunct assistant professor with the University of California (UCLA), Los Angeles, CA, USA, with appointments in the Department of Bioengineering, Department of Surgery, and Department of Head and Neck Surgery. He is currently conducting biomedical THz imaging research in collaboration with the departments of Neurosurgery, Neuropathology, Pathology, General surgery, and Ophthalmology at the UCLA David Geffen School of Medicine. Additionally, he is working on THz system design, THz optics design, and image processing techniques to improve THz image acquisition rates.

James Garritano received the B.S. degrees in electrical engineering and iobchemistry from the University of California (UCLA), Los Angeles, CA, USA, in 2012, and is currently a graduate student Medical Imaging Informatics track in the Department of Bioengineering at UCLA and a researcher in the Center for Advanced Surgical and Interventional Technology. His research interests are THz medical imaging, image-guided therapy, image analysis, image registration, sensor fusion, and fluorescence-lifetime imaging.

Shijun Sung received the B.S. degrees in electrical engineering University of California (UCLA), Los Angeles, CA, USA, in 2011, and is currently a graduate student in the Department of Electrical Engineering and performs research with a number of faculty members and researchers in the Department of Bioengineering and in the David Geffen school of medicine. His research interests included THz medical imaging, THz optical and quasi-optical design, THz system design, and corneal water content sensing.

Neha Bajwa received the B.S. degree in biomedical engineering from The Johns Hopkins University, Baltimore, MD, USA, with a concentration in cell tissue engineering, in 2008, and the M.S. degree in biomedical engineering from the University of California (UCLA), Los Angeles, CA, USA, in 2010, and is currently working toward the Ph.D. degree in biomedical engineering at UCLA..
She has focused her research on THz imaging for diagnostic applications in cornea and burn models. Additionally, she is investigating THz imaging for cancer margin detection and fusing THz burn data with MRI imagery.

David B. Bennett (S'11) received the B.S. degree in physics from University of California, (UCSB), Santa Barbara, CA, USA, in 2006, and the M.S. and Ph.D. degrees in electrical engineering from University of California (UCLA), Los Angeles, CA, USA, in 2008 and 2011, respectively. His thesis work produced the initial analysis of the THz properties of corneal tissue and he performed some of the first in vivo experiments of THz frequency corneal reflectivity.
He is currently with Fitbit, San Francisco, CA, USA.

Bryan N. Nowroozi received the Ph.D. degree in ecology and evolutionary biology from Brown University, Providence, RI, USA, in 2011.
He was a postdoctoral researcher in the UCLA Department of Bioengineering and the assistant director of graduate education at the UCLA Center for Advanced Surgical and Interventional Technology. His research interests are in medical imaging, haptic feedback, and biomechanics. He currently with Mimeo Labs Inc., Santa Monica, CA, USA.

Priyamvada Tewari received the B.S. and M.S. (Integrated Honors) degrees in biophysics from Panjab University, Haryana, India, in 2006, and the Ph.D. degree from the Department of Bioengineering, University of California (UCLA), Los Angeles, CA, USA, in 2012. The topic of her dissertation was Terahertz imaging for early detection of skin disorders and pathologies including cancer, burns, and hydration.
She is currently with Elsevier Life Science Solutions, San Francisco, CA, USA.

James W. Sayre received the Ph.D. degree in bio-statistics in the department of Biostatistics, University of California (UCLA), Los Angeles, CA, USA, in 1977.
He is an Adjunct Professor of Biostatistics & Radiological Sciences at UCLA. His current research interests include computational statistics and database management, clinical trials, statistical methodology in medical diagnostic systems.

Jean Pierre Hubschman trained in Ophthalmology at the Hopital La Timone in Marseille France from 1991 to 1996 and received the M.D. degree from Aix-Marseille University, France, in 1996.
He did a Fellowship in vitreo-retinal surgery from 1996 to 1998 and then headed the Retina department of the Ophthalmologic Center of Saint Jean de Luz—France, the Policlinique Aguilera Biarritz—France and the Policlinica Guipozcoa San Sebastian, Spain. In 2007, he joined the Retina division of the Jules Stein Eye Institute, University of California (UCLA), Los Angeles, CA, USA, as an Assistant Professor of Ophthalmology. His current research interests include development of minimally invasive surgical tools and techniques, surgical robotic systems, and ocular imaging technologies.
Dr. Hubschman is a member of the American Research Society in Ophthalmology, the European Research Society in Ophthalmology and Vision, and the American Society of Cataract and Refractive Surgery.

Sophie X. Deng received her doctor of medicine and doctor of philosophy degrees at the University of Rochester School of Medicine and Dentistry, Rochester, NY, USA, where she completed the rigorous Medical Scientist Training Program.
She did her residency in ophthalmology at the Illinois Eye and Ear Infirmary, Chicago, IL, USA. She subsequently completed her fellowship in Cornea, External Ocular Disease and Refractive Surgery at the Jules Stein Eye Institute.She is an Associate Professor in the Cornea Division at the Jules Stein Eye Institute, University of California (UCLA), Los Angeles, CA, USA. She is a specialist in corneal and external ocular diseases, and cataracts. Her surgical areas include endothelial keratoplasty (DSEK and DMEK), deep anterior lamellar keratoplasty (DALK), penetrating keratoplasty, limbal stem cell transplantation, artificial cornea and cataract. She is the director of the Conea Biology Laboratory at the Jules Stein Eye Institute. Her research focuses on corneal epithelial stem cells regulation, deficiency and regeneration. Her research aims to improve the current treatment for patients with limbal stem cell deficiency by using stem cell therapy to restore vision. In addition, she conducts clinical studies to develop new imaging and molecular tests to accurately diagnose and stage limbal stem cell deficiency.

Elliott R. Brown (M'92–SM'97–F'00) received the Ph.D. degree in applied physics from the California Institute of Technology, Pasadena, CA, USA, in 1985.
He did his post-doctoral work at Lincoln Laboratory, Massachusetts Institute of Technology. He is a Professor of Physics and Electrical Engineering at Wright State University WSU), Dayton, OH, USA, where he holds the Ohio Research Scholars Endowed Chair in Sensors Physics. He conducts research and teaching courses in RF and THz sensor science and technology, and in solid-state physics and engineering. His THz research encompasses several topics including ultra-low-noise rectifiers, photomixing sources, the THz phenomenology of biomaterials, and THz remote sensor and imager design and simulation. Other areas of research include multifunctional RF electronics and systems, biomedical ultrasonic imaging in and around hard tissue (in collaboration with the UCLA Medical and Dental Schools), and electronic and photonic transport in nanostructures. Prior to WSU, he was a Professor of Electrical and Computer Engineering at the University of California, (Santa Barbara and Los Angeles campuses), and prior to that was a Program Manager at DARPA in the Electronics Technology Office, Arlington, VA, USA.
Dr. Brown is a Fellow of the American Physical Society (since 2007). In 1998, he received an Award for Outstanding Achievement from the U.S. Office of the Secretary of Defense.

Warren S. Grundfest received the M.D. degree from the College of Physicians & Surgeons, Columbia University, New York, NY, USA, in 1980, and trained in General Surgery at University of California (UCLA), Los Angeles, CA, USA, and Cedars-Sinai Medical Center, Los Angeles, CA, USA, in 1985.
He is a Professor of Bioengineering at the University of California (UCLA), Los Angeles, CA, USA. He is also a Professor of Electrical Engineering and Professor of Surgery at UCLA. His current research interests include excimer lasers for medical applications, optical diagnostic procedures, minimally invasive surgical tools, haptic feedback and ultrasound imaging.
Dr. Grundfest was elected Fellow, American Institute of Medical & Biologic Engineers (AIMBE), for pioneering development and dissemination of minimally invasive surgery in 1996. In the same year he was elected Fellow, Society of Photo-Optical Instrumentation Engineers (SPIE), for his distinguished & valuable contributions to the field of optical engineering in medicine & biology.
Footnotes
Color versions of one or more of the figures in this paper are available online at http://ieeexplore.ieee.org.
Contributor Information
Zachary D. Taylor, Email: zdeis@seas.ucla.edu, Department of Bioengineering, University of California (UCLA), Los Angeles, CA 90095 USA, and also with the Center for Advanced Surgical and Interventional Technology (CASIT), University of California (UCLA), Los Angeles, CA 90095 USA.
James Garritano, Department of Bioengineering, University of California (UCLA), Los Angeles, CA 90095 USA, and also with the Center for Advanced Surgical and Interventional Technology (CASIT), University of California (UCLA), Los Angeles, CA 90095 USA.
Shijun Sung, Department of Electrical Engineering, University of California (UCLA), Los Angeles, CA 90095 USA.
Neha Bajwa, Department of Bioengineering, University of California (UCLA), Los Angeles, CA 90095 USA, and also with the Center for Advanced Surgical and Interventional Technology (CASIT), University of California (UCLA), Los Angeles, CA 90095 USA.
David B. Bennett, Department of Electrical Engineering, University of California (UCLA), Los Angeles, CA 90095 USA. He is now with Fitbit, San Francisco, CA 94105 USA.
Bryan Nowroozi, Department of Bioengineering, University of California (UCLA), Los Angeles, CA 90095 USA, and also with the Center for Advanced Surgical and Interventional Technology (CASIT), University of California (UCLA), Los Angeles, CA 90095 USA. He is now with Mimeo Labs Inc, Santa Monica, CA 90404 USA.
Priyamvada Tewari, Department of Bioengineering, University of California (UCLA), Los Angeles, CA 90095 USA, and also with the Center for Advanced Surgical and Interventional Technology (CASIT), University of California (UCLA), Los Angeles, CA 90095 USA. She is now with Elsevier Life Science solutions, San Francisco, CA 94105 USA.
James Sayre, Department of Biostatistics, University of California (UCLA), Los Angeles, CA 90095 USA.
Jean-Pierre Hubschman, Department of Ophthalmology, University of California (UCLA), Los Angeles, CA 90095 USA.
Sophie Deng, Department of Ophthalmology, University of California (UCLA), Los Angeles, CA 90095 USA.
Elliott R. Brown, Department. of Electrical Engineering, Wright State University, Dayton, OH 45435 USA.
Warren S. Grundfest, Department of Bioengineering, University of California (UCLA), Los Angeles, CA 90095 USA, and also with the Center for Advanced Surgical and Interventional Technology (CASIT), University of California (UCLA), Los Angeles, CA 90095 USA.
References
- 1.Ferguson B, Wang S, Gray D, Abbott D, Zhang XC. Identification of biological tissue using chirped probe THz imaging. Microelectron. J. 2002;33:1043–1051. [Google Scholar]
- 2.Han PY, Cho GC, Zhang XC. Time-domain transillumination of biological tissues with terahertz pulses. Opt. Lett. 2000;25:242–244. doi: 10.1364/ol.25.000242. [DOI] [PubMed] [Google Scholar]
- 3.LöFfler T, Bauer T, Siebert K, Roskos H, Fitzgerald A, Czasch S. Terahertz dark-field imaging of biomedical tissue. Opt. Express. 2001;9:616–621. doi: 10.1364/oe.9.000616. [DOI] [PubMed] [Google Scholar]
- 4.Ferguson B, Wang S, Gray D, Abbot D, Zhang XC. T-ray computed tomography. Opt. Lett. 2002;27:1312–1314. doi: 10.1364/ol.27.001312. [DOI] [PubMed] [Google Scholar]
- 5.Arnone DD, et al. Appl. THz Technol. to Medical Imag. Munich, Germany: 1999. pp. 209–219. [Google Scholar]
- 6.Hoshina H, Hayashi A, Miyoshi N, Fukunaga Y, Miyamaru F, Otani C. 2009 34th Int. Conf. on Infrared, Millimeter, and THz Waves (IRMMW-THz 2009) 2009. Terahertz pulsed imaging of frozen biological tissues, in; pp. 1–2. [Google Scholar]
- 7.Taylor ZD, et al. THz Medical Imaging: In vivo Hydration Sensing. IEEE Trans. THz Sci. Technol. 2011;1:201–219. doi: 10.1109/TTHZ.2011.2159551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Adamis AP, Filatov V, Tripathi BJ, Tripathi RAMC. Fuchs' endothelial dystrophy of the cornea. Survey of Ophthalmol. 1993;38:149–168. doi: 10.1016/0039-6257(93)90099-s. 9// [DOI] [PubMed] [Google Scholar]
- 9.Krachmer JH, Feder RS, Belin MW. Keratoconus and related noninflammatory corneal thinning disorders. Survey of Ophthalmology. 1984;28:293–322. doi: 10.1016/0039-6257(84)90094-8. 1// [DOI] [PubMed] [Google Scholar]
- 10.Rabinowitz YS. Keratoconus. Survey of Ophthalmol. 1998;42:297–319. doi: 10.1016/s0039-6257(97)00119-7. 1// [DOI] [PubMed] [Google Scholar]
- 11.Panda A, Vanathi M, Kumar A, Dash Y, Priya S. Corneal graft rejection. Survey of Ophthalmol. 2007;52:375–396. doi: 10.1016/j.survophthal.2007.04.008. 7// [DOI] [PubMed] [Google Scholar]
- 12.Lackner B, Schmidinger G, Pieh S, Funovics MA, Skorpik C. Repeatability and reproducibility of central corneal thickness measurement with Pentacam, Orbscan, and ultrasound. Optom. Vis. Sci. 2005 Oct;82:892–899. doi: 10.1097/01.opx.0000180817.46312.0a. [DOI] [PubMed] [Google Scholar]
- 13.Woodward RM, et al. Terahertz pulse imaging in reflection geometry of human skin cancer and skin tissue. Phys. in Medicine and Biol. 2002;47:3853. doi: 10.1088/0031-9155/47/21/325. [DOI] [PubMed] [Google Scholar]
- 14.Bennett D, et al. Assessment of corneal hydration sensing in the terahertz band: In vivo results at 100 GHz. J. Biomed. Opt. 2012;17:097008–1. doi: 10.1117/1.JBO.17.9.097008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bennett DB, et al. Terahertz sensing in corneal tissues. J. Biomed. Opt. 2011;16 doi: 10.1117/1.3575168. Art id 057003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lackner B, Schmidinger G, Pieh S, Funovics MA, Skorpik C. Repeatability and reproducibility of central corneal thickness measurement with Pentacam, Orbscan, and ultrasound. Optometry & Vision Sci. 2005;82:892–899. doi: 10.1097/01.opx.0000180817.46312.0a. [DOI] [PubMed] [Google Scholar]
- 17.Huang D, et al. Optical coherence tomography. Science. 1991 Nov 22;254:1178–1181. doi: 10.1126/science.1957169. 1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pavlin CJ, Sherar MD, Foster FS. Subsurface ultrasound microscopic imaging of the intact eye. Ophthalmol. 1990;97:244–250. doi: 10.1016/s0161-6420(90)32598-8. 2// [DOI] [PubMed] [Google Scholar]
- 19.Pavlin CJ, Harasiewicz K, Sherar MD, Foster FS. Clinical use of ultrasound biomicroscopy. Ophthalmol. 1991;98:287–295. doi: 10.1016/s0161-6420(91)32298-x. 3// [DOI] [PubMed] [Google Scholar]
- 20.McCrackin FL, Passaglia E, Stromberg RR, Steinberg HL. Measurement of the thickness and refractive index of very thin films and the optical properties of surfaces by ellipsometry. J. Res. Nat. Bur. Sec. A. 1963;67 doi: 10.6028/jres.067A.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Doughty MJ, Zaman ML. Human corneal thickness and its impact on intraocular pressure measures: A review and meta-analysis approach. Survey of Ophthalmol. 2000;44:367–408. doi: 10.1016/s0039-6257(00)00110-7. 3// [DOI] [PubMed] [Google Scholar]
- 22.Gullstrand A. Appendix. In: Helmholtz HV, editor. Handbuch Der Physiologischen Optik. Vol. 1. New York: Dover; pp. 351–352. 351–352. [Google Scholar]
- 23.Azartash K, Kwan J, Paugh JR, Nguyen AL, Jester JV, Gratton E. Pre-corneal tear film thickness in humans measured with a novel technique. Mol. Vis. 2011;17:756–767. [PMC free article] [PubMed] [Google Scholar]
- 24.King-Smith PE, Fink BA, Fogt N, Nichols KK, Hill RM, Wilson GS. The thickness of the human precorneal tear film: Evidence from reflection spectra. Invest. Ophthalmol. Vis. Sci. 2000 Oct;41:3348–3359. [PubMed] [Google Scholar]
- 25.Dong J, Wu Q, Wang XG. Measurement of central corneal thickness and pre-corneal tear film thickness of rabbits using the scheimpflug system. Int. J. Ophthalmol. 2013;6:584–587. doi: 10.3980/j.issn.2222-3959.2013.05.05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.King-Smith PE, Kimball SH, Nichols JJ. Tear film interferometry and corneal surface roughness. Invest. Ophthalmol. Vis. Sci. 2014 Apr;55:2614–2618. doi: 10.1167/iovs.14-14076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Heys JJ, Barocas VH, Taravella MJ. Modeling passive mechanical interaction between aqueous humor and iris. J. Biomech. Eng. 2001;123:540–547. doi: 10.1115/1.1411972. [DOI] [PubMed] [Google Scholar]
- 28.Riazuddin SA, et al. Missense mutations in TCF8 cause late-onset fuchs corneal dystrophy and interact with FCD4 on chromosome 9p. Amer. J. Human Genetics. 86:45–53. doi: 10.1016/j.ajhg.2009.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Klintworth G. Corneal dystrophies. Orphanet J. Rare Diseases. 2009;4:7. doi: 10.1186/1750-1172-4-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yue BY, Sugar J, Schrode K. Histochemical studies of keratoconus. Curr. Eye Res. 1988 Jan;7:81–86. doi: 10.3109/02713688809047024. [DOI] [PubMed] [Google Scholar]
- 31.Borderie VM, Touzeau O, Bourcier T, Allouch CE, Zito E, Laroche L. Outcome of graft central thickness after penetrating keratoplasty. Ophthalmol. 2005;112:626–633. doi: 10.1016/j.ophtha.2004.11.044. [DOI] [PubMed] [Google Scholar]
- 32.Dougherty PJ, Wellish KL, Maloney RK. Excimer laser ablation rate and corneal hydration. Am. J. Ophthalmol. 1994 Aug 15;118:169–76. doi: 10.1016/s0002-9394(14)72896-x. [DOI] [PubMed] [Google Scholar]
- 33.de Souza IR, de Souza AP, de Queiroz AP, Figueiredo P, Jesus RS, Kara-Jose N. Influence of temperature and humidity on laser in situ keratomileusis outcomes. J. Refract. Surg. 2001 Mar-Apr;17:S202–S204. doi: 10.3928/1081-597X-20010302-11. [DOI] [PubMed] [Google Scholar]
- 34.Fisher BTBS, Masiello KABS, Goldstein MHMD, Hahn DWPD. Assessment of transient changes in corneal hydration using confocal Raman spectroscopy. Cornea. 2003;22:363–370. doi: 10.1097/00003226-200305000-00016. [DOI] [PubMed] [Google Scholar]
- 35.Bende T, Seiler T, Wollensak J. Side effects in excimer corneal surgery: Corneal thermal gradients. Graefe's Arch. Clinical and Experimental Ophthalmol. 1988;226 doi: 10.1007/BF02181196. [DOI] [PubMed] [Google Scholar]
- 36.Farah SG, Azar DT. Visual outcomes after primary LASIK. In: Azar DT, Koch DD, editors. LASIK: Fundamentals, Surgical Techniques, and Complications. New York: Marcel Dekker; 2003. p. 506. xiv. [Google Scholar]
- 37.Ytteborg J, Dohlman CH. Corneal edema and intraocular pressure: II. Clinical results. Arch. Ophthalmol. 1965;74:477–484. doi: 10.1001/archopht.1965.00970040479008. [DOI] [PubMed] [Google Scholar]
- 38.Brugin E, Ghirlando A, Gambato C, Midena E. Central corneal thickness: Z-ring corneal confocal microscopy versus ultrasound pachymetry. Cornea. 2007;26:303–307. doi: 10.1097/ICO.0b013e31802e1dea. [DOI] [PubMed] [Google Scholar]
- 39.Ytteborg J, Dohlman CH. Corneal edema and intraocular pressure: II. Clinical results. Arch. Ophthalmol. 1965 Oct 1;74:477–484. doi: 10.1001/archopht.1965.00970040479008. 1965. [DOI] [PubMed] [Google Scholar]
- 40.Bamashmus MA, Saleh MF, Awadalla MA. Reasons for not performing keratorefractive surgery in patients seeking refractive surgery in a hospital-based cohort in “yemen”. Middle East Afr. J. Ophthalmol. 2010 Oct;17:349–353. doi: 10.4103/0974-9233.71605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Izatt JA, et al. MIcrometer-scale resolution imaging of the anterior eye in vivo with optical coherence tomography. Arch. Ophthalmol. 1994;112:1584–1589. doi: 10.1001/archopht.1994.01090240090031. [DOI] [PubMed] [Google Scholar]
- 42.Gromacki SJ, Barr JT. Central and peripheral corneal thickness in keratoconus and normal patient groups. Optometry & Vision Sci. 1994;71:437–441. doi: 10.1097/00006324-199407000-00003. [DOI] [PubMed] [Google Scholar]
- 43.McDonnell PJ, Enger C, Stark WJ, Stulting RD Stulting, Collaborative Corneal Transplantation Study Group. Corneal thickness changes after high-risk penetrating keratoplasty. Arch. Ophthalmol. 1993 Oct;111:1374–1381. doi: 10.1001/archopht.1993.01090100082032. [DOI] [PubMed] [Google Scholar]
- 44.Mandell RB, Polse KA, Brand RJ, Vastine D, Demartini D, Flom R. Corneal hydration control in Fuchs' dystrophy. Invest Ophthalmol. Vis. Sci. 1989 May;30:845–852. [PubMed] [Google Scholar]
- 45.Uçakhan ÖÖ, Özkan M, Kanpolat A. Corneal thickness measurements in normal and keratoconic eyes: Pentacam comprehensive eye scanner versus noncontact specular microscopy and ultrasound pachymetry. J. Cataract & Refractive Surgery. 2006;32:970–977. doi: 10.1016/j.jcrs.2006.02.037. 6// [DOI] [PubMed] [Google Scholar]
- 46.Mandell RB, Polse KA, Brand RJ, Vastine D, Demartini D, Flom R. Corneal hydration control in Fuchs' dystrophy. Investig. Ophthalmol. & Visual Sci. 1989 May 1;30:845–852. 1989. [PubMed] [Google Scholar]
- 47.Chakrabarti HS, Craig JP, Brahma A, Malik TY, McGhee CNJ. Comparison of corneal thickness measurements using ultrasound and Orbscan slit-scanning topography in normal and post-LASIK eyes. J. Cataract & Refractive Surgery. 2001;27:1823–1828. doi: 10.1016/s0886-3350(01)01089-6. 11// [DOI] [PubMed] [Google Scholar]
- 48.Ehlers N, Bramsen T. Central thickness in corneal disorders. Act. Ophthalmol. 1978;56:412–416. doi: 10.1111/j.1755-3768.1978.tb05694.x. [DOI] [PubMed] [Google Scholar]
- 49.Bechmann M, et al. Central corneal thickness measurement with a retinal optical coherence tomography device versus standard ultrasonic pachymetry. Cornea. 2001;20:50–54. doi: 10.1097/00003226-200101000-00010. [DOI] [PubMed] [Google Scholar]
- 50.Hitzenberger CK, Drexler W, Fercher AF. Measurement of corneal thickness by laser Doppler interferometry. Investig. Ophthalmol. & Visual Sci. 1992 Jan 1;33:98–103. 1992. [PubMed] [Google Scholar]
- 51.ANSI. Z136.1—Safe use of lasers. 2007 ed. [Google Scholar]
- 52.Bauer NJ, Wicksted JP, Jongsma FH, Mar WF, Hendrikse F, Motamedi M. Noninvasive assessment of the hydration gradient across the cornea using confocal Raman spectroscopy. Invest. Ophthalmol. Vis. Sci. 1998 Apr;39:831–835. [PubMed] [Google Scholar]
- 53.Bauer NJ, Hendrikse F, March WF. In vivo confocal Raman spectroscopy of the human cornea. Cornea. 1999 Jul;18:483–8. doi: 10.1097/00003226-199907000-00015. [DOI] [PubMed] [Google Scholar]
- 54.Fisher BT, Masiello KA, Goldstein MH, Hahn DW. Assessment of transient changes in corneal hydration using confocal Raman spectroscopy. Cornea. 2003 May;22:363–370. doi: 10.1097/00003226-200305000-00016. [DOI] [PubMed] [Google Scholar]
- 55.Knighton RW, Huang X-R. Linear birefringence of the central human cornea. Investig. Ophthalmol. Visual Sci. 2002 Jan 1;43:82–86. 2002. [PubMed] [Google Scholar]
- 56.Yeh P, Yariv A, Hong C-S. Electromagnetic propagation in periodic stratified media. I. General theory. J. the Optical Society of America. 1977;67:423–438. 1977/04/01. [Google Scholar]
- 57.Niklasson GA, Granqvist CG, Hunderi O. Effective medium models for the optical properties of inhomogeneous materials. Appl. Opt. 1981;20:26–30. doi: 10.1364/AO.20.000026. [DOI] [PubMed] [Google Scholar]
- 58.Landauer R. Electrical conductivity in inhomogeneous media. AIP Conf. Proc. 1978;40:2–45. [Google Scholar]
- 59.Karkkainen KK, Sihvola AH, Nikoskinen KI. Effective permittivity of mixtures: Numerical validation by the FDTD method. IEEE Trans. Geosci. Remote Sensing. 2000;38:1303–1308. [Google Scholar]
- 60.Born M, Wolf E. Principles of Optics: Electromagnetic Theory of Propagation, Interference and Diffraction of Light. Cambridge, U.K: Cambridge University Press; 1980. [Google Scholar]
- 61.Bennett DB, Li W, Taylor ZD, Grundfest WS, Brown ER. Stratified media model for terahertz reflectometry of the skin. IEEE Sensors J. 2010;11 1530-437X. [Google Scholar]
- 62.Xu J, Plaxco KW, Allen SJ, Bjarnason JE, Brown ER. 0.15-3.72 THz absorption of aqueous salts and saline solutions. Appl. Phys. Lett. 2007;90 031908-031908-3. [Google Scholar]
- 63.Orfanidis SJ. Electromagnetic Waves and Antennas. Vol. 1 New Brunswick, NJ, USA: Rutgers Univ; 2014. [Google Scholar]
- 64.Markelz AG, Roitberg A, Heilweil EJ. Pulsed terahertz spectroscopy of DNA, bovine serum albumin and collagen between 0.1 and 2.0 THz. Chemical Phys. Lett. 2000;320:42–48. [Google Scholar]
- 65.Taylor ZD, et al. THz sensing of corneal tissue water content: In vivo sensing and imaging results. IEEE Trans. THz Sci. Technol. 2015;5 doi: 10.1109/TTHZ.2015.2392628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Azzam RMA, Bashara NM. Ellipsometry and Polarized Light. Amsterdam, the Netherlands: Elsevier Science; 1987. [Google Scholar]
- 67.Fatt I, Goldstick TK. Dynamics of water transport in swelling membranes. J. Colloid Sci. 1965;20:962–989. doi: 10.1016/0095-8522(65)90068-1. 12// [DOI] [PubMed] [Google Scholar]
- 68.Taylor ZD, Singh RS, Brown ER, Bjarnason JE, Hanson MP, Gossard AC. Analysis of pulsed THz imaging using optical character recognition. IEEE Sensors J. 2009;9:3–8. [Google Scholar]
- 69.Tewari P, et al. In vivo terahertz imaging of rat skin burns. J. Biomed. Opt. 2012;17:040503–3. doi: 10.1117/1.JBO.17.4.040503. [DOI] [PubMed] [Google Scholar]
- 70.Taylor ZD, et al. Reflective terahertz imaging of porcine skin burns. Opt. Lett. 2008;33:1258–1260. doi: 10.1364/ol.33.001258. [DOI] [PubMed] [Google Scholar]
- 71.Spoler F, Frentz M, Schrage NF. Towards a new in vitro model of dry eye: The ex vivo eye irritation test. Dev. Ophthalmol. 2010;45:93–107. doi: 10.1159/000315023. [DOI] [PubMed] [Google Scholar]
- 72.Glass DH, Roberts CJ, Litsky AS, Weber PA. A viscoelastic biomechanical model of the cornea describing the effect of viscosity and elasticity on hysteresis. Investig. Ophthalmology & Visual Sci. 2008 Sep;49:3919–3926. doi: 10.1167/iovs.07-1321. 2008. [DOI] [PubMed] [Google Scholar]