Abstract
The mechanism by which glucocorticoids alleviate renal inflammatory disorders remains incompletely understood. Here, we report that the efficacy of glucocorticoids in ameliorating FSGS depends on the capacity to expand myeloid-derived suppressor cells (MDSCs). After glucocorticoid treatment, the frequency of CD11b+HLA-DR−CD14−CD15+ MDSCs in peripheral blood rapidly increased in patients with glucocorticoid-sensitive FSGS but remained unchanged in patients with glucocorticoid-resistant FSGS. The frequency of CD11b+Gr-1+ MDSCs in mouse peripheral blood, bone marrow, spleen, kidney-draining lymph nodes (KDLNs), and kidney also increased after glucocorticoid treatment. The induced MDSCs from glucocorticoid-treated mice strongly suppressed T cells, dendritic cells, and macrophages but induced regulatory T cells in spleen, KDLNs, and kidney. Moreover, glucocorticoid treatment suppressed doxorubicin-induced T cell proliferation, dendritic cell and macrophage infiltration, and proinflammatory cytokine production, whereas this protective effect was largely abolished by depleting MDSCs using anti–Gr-1 antibody. Finally, the adoptive transfer of induced MDSCs into the doxorubicin-treated mice not only confirmed the protective role of MDSCs in doxorubicin-induced renal injury but also showed that the transferred MDSCs rapidly migrated into the lymphocyte-accumulating organs, such as the spleen and KDLNs, where they suppressed T cell proliferation. Taken together, these results demonstrate that glucocorticoid treatment ameliorates FSGS by expanding functional MDSCs and that this rapid elevation of MDSCs in peripheral blood may serve as an indicator for predicting the efficacy of glucocorticoid treatment.
Keywords: focal segmental glomerulosclerosis, renal protection, cell activation, chemokine, lymphocytes
FSGS, one of the leading causes of ESRD,1 accounts for approximately 20% of cases of nephrotic syndrome in children and 40% of such cases in adults.2 The causes of FSGS are very complicated, involving genetic abnormalities, immunologic abnormalities, viral infections, decreased nephron numbers, and hyperfiltration.3 Clinical observations suggested that FSGS is induced by immune factors, in which T cell dysfunction played an important role. In 1974, Shalhoub4 first hypothesized that idiopathic nephrotic syndrome (minimal-change disease and FSGS) was produced by T cell dysfunction, resulting in the secretion of chemical mediators toxic to the glomerular basement membrane. Various subsets of T cells, including CD8 cells and CD4 helper cells of Th1, Th2, IL17-producing Th17 and regulatory T cells (Tregs), are involved in glomerular disease.5–9 The cytokines released by Th17 cells further induce other cells to secret proinflammatory chemokines that attract leukocytes and activate resident glomerular cells.10
The initial treatment option of primary FSGS in children and young adults is often limited to high-dosage synthetic glucocorticoid (GC) analogues despite their multiple adverse effects.11,12 Approximately 30%–60% of patients with primary FSGS respond to an initial course.13,14 Although the mechanism remains unclear, the number of leukocytes in the peripheral blood of GC-sensitive patients was generally increased after GC treatment.13 In contrast, patients who are steroid resistant have a chronic disease and are at an increased risk of progressing to ESRD and complications related to uncontrolled nephrotic syndrome.11,13,15 GCs can also effectively promote apoptosis of leukemia cells, trigger complex anti-inflammatory actions, and induce apoptotic death of peripheral and developing T cells.16–18 However, although the molecular mechanisms underlying GC treatment have been extensively investigated, how exogenous GCs affect peripheral immune cells in FSGS or other renal diseases remains largely unknown.
Myeloid-derived suppressor cells (MDSCs) are a heterogeneous population of immature myeloid cells that can suppress T cell responses.19,20 In mice, the phenotype of MDSCs is characterized by the coexpression of Ly-6C/G and CD11b and can be subdivided into granulocytic CD11b+Ly-6G+Ly-6Clow MDSCs and monocytic CD11b+Ly-6G-Ly-6Chigh MDSCs. Granulocytic MDSCs suppress T cell proliferation primarily through reactive oxygen species, whereas monocytic MDSCs suppress T cell proliferation by high levels of nitric oxide produced from inducible nitric oxide synthase.19,21 Because of the lack of markers of mature myeloid and lymphoid cells, human MDSCs are generally defined as cells that express CD11b and the common myeloid marker CD33.22,23 While both groups of MDSCs are HLA-DRlow/neg and CD33+, human monocytic MDSCs are generally CD14 positive, and granulocytic MDSCs express CD15. Human MDSCs were reported for the first time in patients with head and neck cancer.24 Under tumor conditions, human MDSCs were widely found to be expanded.22,23,25,26 Recent studies indicated that the accumulation of MDSCs was also related to inflammatory and autoimmune diseases, including inflammatory bowel disease,27 type 1 diabetes,28 multiple sclerosis,29 and hepatitis B and C virus infection.30 However, the direct anti-inflammatory role and mechanism of MDSCs in FSGS remain inconclusive.
In the present study, we hypothesized that the expansion of immunosuppressive MDSCs might play an essential role in GC-mediated therapy for FSGS disease. The patients with FSGS were classified as GC sensitive or GC insensitive on the basis of clinical diagnosis. MDSCs in the peripheral blood of these patients with FSGS were assessed before and after GC treatment. The effects of MDSCs on experimental renal injury were determined by treating mice with GC or directly adoptive-transferring MDSCs into doxorubicin-treated mice. Our results demonstrated that MDSCs play a critical protective role against renal injury and serve as a major mechanism underlying the GC-based therapy for patients with FSGS.
RESULTS
Expansion of CD11b+HLA-DR−CD14−CD15+ and CD11b+HLA-DR−CD14+CD33+ Cells in Patients with FSGS Sensitive to GCs
Leukocytosis has long been recognized in GC-treated nephrotic diseases, although the underlying mechanism is largely unknown.31 To explore the molecular mechanism of this phenomenon, the peripheral blood of patients with FSGS was collected before and 7–14 days after GC therapy. These patients were then continuously treated with GCs. At the end of the 12–16 weeks of GC treatment, these patients were divided into GC-sensitive and GC-insensitive groups according to clinical diagnoses. Clinical measures and histologic injury at baseline are shown in Tables 1 and 2, respectively. In general, blood samples from 86 new patients with FSGS sensitive to GC treatment, 23 new patients insensitive to GC treatment, and 13 healthy people were collected. As shown in Table 1, urine protein (U-pro), total cholesterol (TC), and triglyceride (TG) were significantly downregulated in GC-sensitive patients after GC treatment, while serum albumin was significantly upregulated. In contrast, these measures did not significantly differ between GC-insensitive patients before and after GC treatment. There were also no significant differences between GC-sensitive patients and GC-insensitive patients for baseline clinical and pathologic features, including electron microscopy findings.
Table 1.
Clinical features of patients with FSGS
| Clinical Feature | GC-Sensitive Patients (n=86) | GC-Insensitive Patients (n=23) | Baseline Comparison P Value | ||||
|---|---|---|---|---|---|---|---|
| Before | After | P Value | Before | After | P Value | ||
| Age (yr) | 32.6±14.6 | 27.1±13.2 | 0.10 | ||||
| Men:women (n:n) | 57:29 | 17:6 | 0.49 | ||||
| Hypertension (%) | 10 (10.8) | 4 (17.4) | 0.47 | ||||
| U-pro (g/24 hr) | 5.46 (4.13–8.17) | 0.32 (0.20–0.38) | <0.001a | 5.46 (4.87–9.89) | 6.28 (3.05–8.37) | 0.83a | 0.53 |
| URBC (×104/ml) | 4 (1–10) | 3 (1–5) | <0.001a | 5(2–11) | 5(1–6) | 0.87a | 0.25 |
| SCr (μmol/L) | 88.0 (64.1–117.8) | 68.5 (52.8–82.4) | <0.001a | 70.7 (53.0–96.4) | 68.1 (53.9–100.8) | 0.89a | 0.12 |
| ALB (g/L) | 26.7±7.6 | 40.5±7.3 | <0.001b | 26.2±7.0 | 27.8±6.5 | 0.11b | 0.77 |
| TC (mmol/L) | 10.4±4.3 | 7.0±2.7 | <0.001b | 11.7±5.3 | 10.3±3.2 | 0.25b | 0.24 |
| TG (mmol/L) | 3.2±2.8 | 1.9±1.0 | <0.001b | 4.0±3.0 | 3.5±2.4 | 0.47b | 0.21 |
| MDSC (%) | 41.7 (36.5–47.1) | 58.9 (52.9–66.5) | <0.001a | 39.2 (34.8–45.4) | 40.7 (35.7–48.5) | 0.05a | 0.35 |
Sex ratio of patients was compared using chi-squared test. The incidence of hypertension was compared using Fisher exact test. Baseline age, albumin, total cholesterol, and triglycerides of GC-sensitive and GC-insensitive patients were compared using t test. Baseline urine protein, urine red blood cell, and serum creatinine of GC-sensitive and GC-insensitive patients were compared using Mann–Whitney test. Baseline comparison: GC-sensitive patients before versus GC-insensitive patients before. Hypertension is presented as number (percentage); urine protein, urine red blood cell, serum creatinine, and MDSC are presented as median (interquartile range). Age, albumin, total cholesterol, and trigylcerides are presented as mean±SD. U-pro, urine protein; URBC, urine red blood cell; SCr, serum creatinine; ALB, serum albumin; TC, total cholesterol; TG, triglyceride.
P values were calculated using Wilcoxon test.
P values were calculated using paired t test.
Table 2.
Morphologic findings of patients with FSGS
| Morphologic Findings | Total | GC-Sensitive Patients | GC-Insensitive Patients | P Value |
|---|---|---|---|---|
| Total cases (n) | 109 | 86 | 23 | |
| FSGS histologic subtype | 0.20 | |||
| Not otherwise specified | 48 (44.0) | 33 (38.4) | 15 (65.2) | |
| Collapsing | 5 (4.6) | 5 (5.8) | 0 | |
| Tip | 37 (33.9) | 32 (37.2) | 5 (21.7) | |
| Cellular | 8 (7.3) | 7 (8.1) | 1 (4.3) | |
| Perihilar | 11 (10.1) | 9 (10.5) | 2 (8.7) | |
| Glomeruli | 26 (21–35) | 26 (21–33) | 27 (15–42) | 0.62 |
| Global glomerulosclerosis (% glomeruli) | 0 (0–6) | 0 (0–5) | 0 (0–7) | 0.74 |
| Segmental glomerulosclerosis (%) | 10.0 (6.6–19.2) | 10.0 (6.1–17.1) | 16.7 (7.1–29.6) | 0.08 |
| Acute tubulointerstitial injury | ||||
| Absent | 12 (11.0) | 9 (10.5) | 3 (13.0) | |
| Mild | 69 (63.3) | 57 (66.3) | 12 (52.2) | |
| Moderate | 19 (17.4) | 13 (15.1) | 6 (26.1) | |
| Severe | 9 (8.3) | 7 (8.1) | 2 (8.7) | |
| Chronic tubulointerstitial injury | ||||
| Absent | 57 (52.3) | 46 (53.5) | 11 (47.8) | |
| Mild | 40 (36.7) | 31 (36.0) | 9 (39.1) | |
| Moderate | 9 (8.3) | 7 (8.1) | 2 (8.7) | |
| Severe | 3 (2.8) | 2 (2.3) | 1 (4.3) | |
| Vascular lesions | 62 (56.9) | 51 (59.3) | 11 (47.8) | 0.32 |
| Electron microscopy findings | ||||
| Podocyte foot process effacement (%) | 85 (73–85) | 80 (65–85) | 85 (80–85) | 0.24 |
Unless otherwise noted, data are presented as number (percentage) or the median (interquartile range). Data described as percentages were analyzed using chi-squared test or Fisher exact test. Other data were compared using Mann–Whitney or Kruskal–Wallis test.
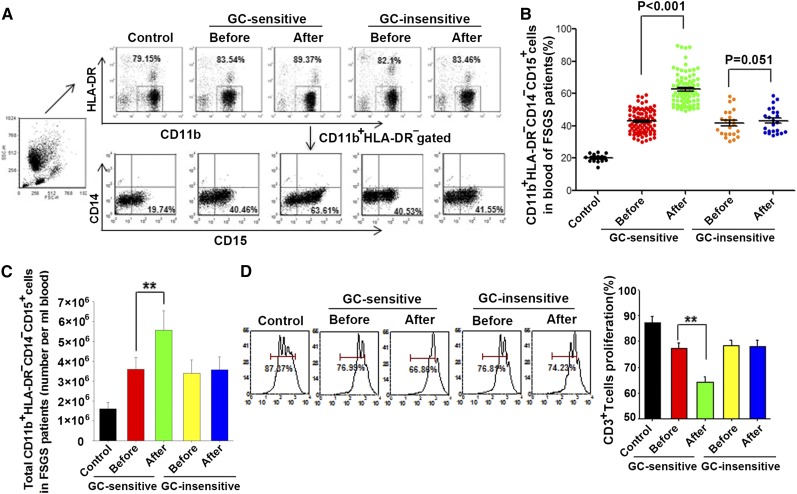
The levels of two MDSCs, CD11b+HLA-DR−CD14−CD15+ and CD11b+HLA-DR−CD14+CD33+ cells, were then analyzed in fresh peripheral blood of patients with FSGS before or 7–14 days after GC treatment. As shown in Figure 1, A–C, the level of CD11b+HLA-DR−CD14−CD15+ MDSCs significantly increased in GC-sensitive but not in GC-insensitive patients with FSGS. The induced cells in GC-sensitive patients with FSGS were mainly CD11b+HLA-DR−CD14−CD15+ cells, and CD11b+HLA-DR−CD14+CD33+ cells accounted for <2% (data not shown). To characterize the immune suppressive capacity of the expanded CD11b+HLA-DR−CD14−CD15+ cells, we isolated HLA-DR−CD15+ cells from peripheral blood of patients and co-cultured them with carboxyfluorescein succinimidyl ester (CFSE)–labeled T cells at the ratio of 1:2. As shown in Figure 1D, HLA-DR−CD15+ cells from GC-sensitive patients with FSGS strongly suppressed T cell proliferation.
Figure 1.
GC treatment induced expansion of CD11b+HLA-DR−CD14−CD15+ cells in peripheral blood samples from GC-sensitive but not GC-insensitive patients with FSGS. (A) Flow cytometry analysis of CD11b+HLA-DR-CD14-CD15+ cells in peripheral blood samples from normal controls (CTL, n=13) and patients with FSGS sensitive (n=86) and insensitive (n=23) to GC treatment. The blood samples were collected 7–14 days after GC treatment. Whole blood was centrifuged to get rid of plasma. The lower cells were lysed to remove red blood cells for further FACS analysis. In the FACS analysis, we first gated CD11b+HLA-DR- cells and then analyzed CD14-CD15+ cells. (B) Statistical analysis of the percentages of CD11b+HLA-DR−CD14−CD15+ MDSCs in peripheral blood samples from panel A. The MDSCs levels are presented as the percentage of CD14−CD15+ cells in total CD11b+HLA-DR−–gated cells (CD11b+HLA-DR−CD14−CD15+ cells are granulocytic MDSCs). (C) Absolute number of CD11b+HLA-DR−CD14-CD15+ MDSCs in 1 ml peripheral blood of patients with FSGS. (D) Suppressive effect of HLA-DR−-CD15+ MDSCs from the blood of GC-sensitive or GC-insensitive FSGS patients on T cell proliferation were measured as described in the Concise Methods. Sorted HLA-DR−CD15+ cells were co-cultured with CFSE-labeled CD3+ T cells isolated from PBMCs of healthy donors, and the capacity of CD3+ T cell proliferation was analyzed. The right histogram represents the statistical results and shown as the mean±SEM (n=3; four samples per group). **P<0.01.
We also assayed the MDSCs in the peripheral bloods of patients with minimal-change disease who are GC sensitive or GC insensitive before and after GC therapy. In this experiment, 48 patients with minimal-change disease were divided into GC-sensitive (43 of 48) and GC-insensitive (5 of 48) groups according to clinical diagnoses (Supplemental Table 1). At pathologic examination, the patients with minimal-change disease were diagnosed with normal appearance of glomeruli and lack of tubulointerstitial fibrosis on light microscopy, whereas these patients were diagnosed with extensive effacement of foot processes of the podocytes on electron microscopy. As shown in Supplemental Figure 1, A and B, the levels of CD11b+HLA-DR−CD14−CD15+ cells were significantly increased in the peripheral blood of GC-sensitive patients with minimal-change disease but not GC-insensitive patients with that disease. Moreover, the isolated HLA-DR−CD15+ cells from the peripheral blood of GC-sensitive patients with minimal-change disease also showed a strong inhibition on T cell proliferation (Supplemental Figure 1C). Therefore, the expansion of MDSCs is not specific to FSGS but likely a common phenomenon responding to effective treatment of GC.
CD11b+Gr-1+ Cells Expanded at the Early Stage of Doxorubicin-Induced Renal Injury in Mice
The pathologic features of ADR-induced renal injury are proteinuria, podocyte structural derangement, tubular injury, and the expansion of mononuclear cells and T cells in the interstitial compartment.7,9,32 In this experiment, 8-week-old male BALB/c mice that weighed 20–22 g were injected intravenously with saline (control) or doxorubicin, 10 mg/kg. As shown in the diagram of the experimental design (Supplemental Figure 2A), following doxorubicin administration, urine was collected on days 0, 3, 7, and 14, and the mice were euthanized and the sera were collected. Evidence collectively indicated that the doxorubicin-induced FSGS mice model was successfully developed. The doxorubicin-treated group of mice showed prominent increase in proteinuria and serum creatinine compared with the control (Supplemental Figure 2, B and C). The peak urinary albumin level was observed at 1 week. Electron microscopy and periodic acid-Schiff staining results (Supplemental Figure 2, D and E) revealed that the doxorubicin treatment caused a severe loss of foot processes, glomerular sclerosis, and tubular damage.
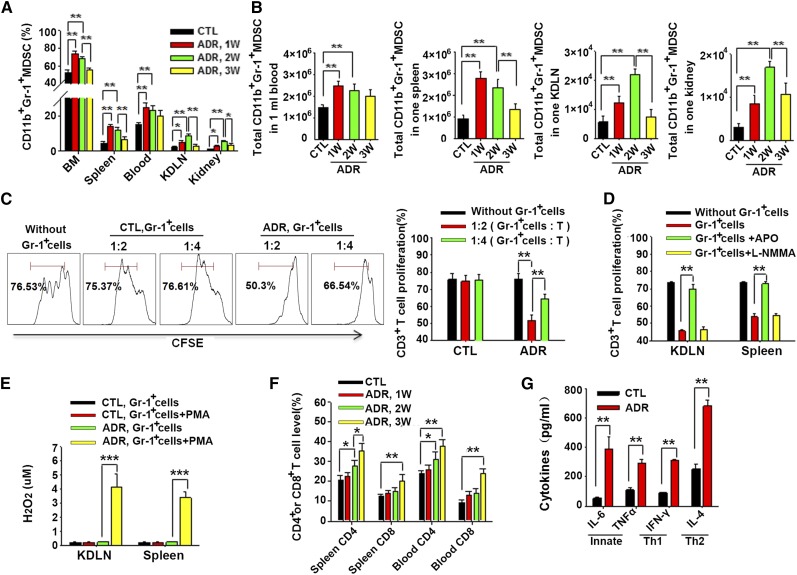
To examine the potential role of MDSCs in doxorubicin-induced renal injury, the levels of CD11b+Gr-1+ MDSCs33 were analyzed in the bone marrow (BM), spleen, blood, kidney-draining lymph nodes (KDLNs), and kidney over time. As shown in Figure 2, A and B, doxorubicin injection rapidly induced the accumulation of CD11b+Gr-1+ MDSCs in the BM, spleen, and blood at 1 week, and in KDLNs and kidney at 1 and 2 weeks, but the levels of CD11b+Gr-1+ MDSCs started to decrease later, suggesting the potential role of CD11b+Gr-1+ MDSCs at the early stage of doxorubicin-induced renal injury. To characterize the immune suppressive capacity of CD11b+Gr-1+ MDSCs derived from doxorubicin-treated mice, MDSCs were purified from mouse spleen after doxorubicin treatment and then co-cultured with CFSE-labeled T cells. As shown in Figure 2C, Gr-1+ cells purified from mouse spleen after doxorubicin treatment for 2 weeks strongly suppressed T cell proliferation in a dose-dependent manner. In agreement with that, MDSCs induced in doxorubicin-treated mice were mainly granulocytic MDSCs and suppressed T cell proliferation via reactive oxygen species pathway,34 addition of reactive oxygen species inhibitor apocynin (APO) but not nitric ocide inhibitor NG-methyl-L-arginine, acetate salt (L-NMMA) abolished the inhibitory effect of Gr-1+ cells (Figure 2D). A substantial amount of H2O2 was generated in Gr-1+ cells from doxorubicin-treated mice but not Gr-1+ cells from control mice following phorbol myristate acetate (PMA) stimulation (Figure 2E). We also assayed T cells and their activity in doxorubicin-treated mice and found that CD4+ and CD8+ T cells in the mouse spleen and blood (Figure 2F), as well as the secreted cytokine IL-6, TNF-α, IFN-γ, and IL-4 (Figure 2G) were upregulated.
Figure 2.
Doxorubicin treatment resulted in the elevation of the frequencies of T cells and MDSCs. (A) FACS were performed as described in the Concise Methods and the numbers of CD11b+ Gr-1+ MDSCs at various time points in the BM, spleen, blood, KDLNs, and kidney of doxorubicin-treated mice were determined. The MDSC counts are presented as percentages of CD11b+ Gr-1+ cells in total BM cells, splenocytes (Spleen), white blood cells (Blood), KDLN cells, and mononuclear cells in kidney (Kidney) after depletion of red blood cells (RBCs). (B) The absolute number of CD11b+Gr-1+ MDSCs at various time points in the BM, spleen, blood, KDLNs, and kidney of doxorubicin-treated mice. (C) Suppression of T cell proliferation by MDSCs isolated from the spleen of doxorubicin-treated mice for 2 weeks. Gr-1+ cells were incubated with CFSE-labeled T cells in different ratios. Gr-1+ cells isolated from the spleen of mice without doxorubicin treatment were used as control (CTL). The right histogram represents the statistical analysis from three independent experiments. (D) Gr-1+ cells were sorted from KDLN and spleen of mice with or without ADR treatment. The isolated cells were incubated with CFSE-labeled lymphocytes from spleen at the presence of APO or L-NMMA. CD3+ T cells proliferation from three independent experiments were analyzed with FACS. (E) Significant levels of reactive oxygen species were observed in KDLNs and spleen Gr-1+ cells from mice with doxorubicin treatment. The level of H2O2 was evaluated from three independent experiments and measured as described in the Concise Methods. (F) Doxorubicin-mediated expansion of CD4+ and CD8+ T cells in mouse spleen and peripheral blood at 1, 2, and 3 weeks after doxorubicin injection. CD4+ or CD8+ T cell counts are presented as percentages of CD4+ or CD8+ cells in total splenocytes or white blood cells after depletion ofng RBCs. (G) At 2 weeks after doxorubicin injection, serum was collected, and the levels of IL-6, TNF-α, IFN-γ, and IL-4 were measured by ELISA. Values are shown as the mean±SEM (n=3; six samples per group). *P<0.05; **P<0.01; ***P<0.001.
GCs Protect Mice from Doxorubicin-Induced Renal Injury and Induce the Expansion of MDSCs
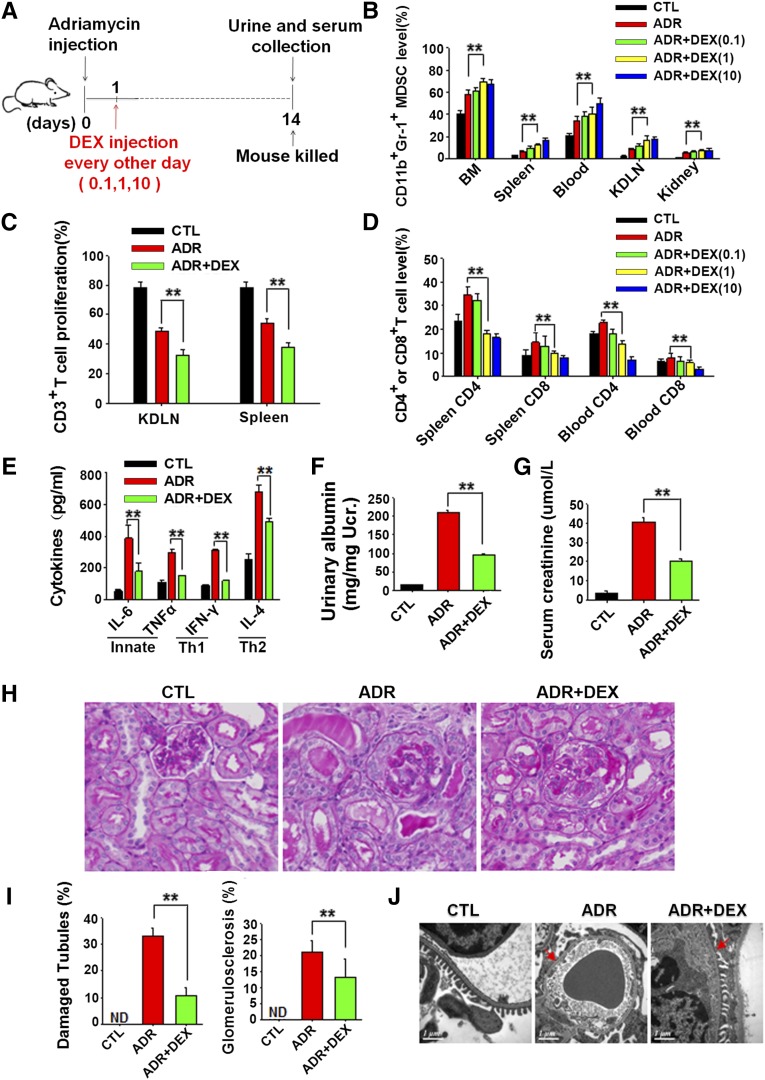
The effects of GCs on GN and their ability to expand the immunosuppressive MDSCs were examined. Mice were treated with synthetic dexamethasone at different concentrations (0.1, 1.0, 10 mg/kg) every other day after the induction of doxorubicin-induced mouse renal injury (Figure 3A). The results showed that dexamethasone treatment strongly increased the frequency of CD11b+Gr-1+MDSCs in mouse BM, spleen, blood, KDLNs, and kidney in a dose-dependent manner (Figure 3B). Furthermore, Gr-1+MDSCs isolated from the spleen and KDLNs of mice treated with 1 mg/kg dexamethasone also displayed the ability to inhibit T cell proliferation in vitro (Figure 3C). Accordingly, the populations of CD4+ and CD8+ T cells in mouse spleen and blood were strongly downregulated by dexamethasone treatment (Figure 3D). The serum levels of IL-6, TNF-α, IFN-γ, and IL-4 were examined and found to be significantly downregulated by GCs (Figure 3E). Next, we compared the doxorubicin-induced renal injury in mice treated with or without 1 mg/kg dexamethasone. As shown in Figure 3, F–I, dexamethasone treatment strongly attenuated the doxorubicin-induced renal injury in mice, accompanied by a lower levels of urinary albumin (Figure 3F) and serum creatinine (Figure 3G), less glomerular sclerosis and tubular damage (Figure 3, H and I), less foot processes loss or infusion (Figure 3J), and less body weight loss (Supplemental Figure 3). These results indicate that the protection that dexamethasone provides against doxorubicin-induced renal injury may depend on the induction of MDSCs.
Figure 3.
GCs protect mice from doxorubicin-induced renal injury and induce the expansion of MDSCs. (A) Diagram of the experimental design. Dexamethasone (DEX) (0.1, 1, 10 mg/kg) was injected intraperitoneally into mice 1 day after doxorubicin (Adriamycin [ADR] injection and injected every other day. Mice were administered 0.9% NaCl (CTL), doxorubicin (ADR), doxorubicinplus 0.1 mg/kg DEX (ADR+DEX [0.1]), doxorubicin plus 1 mg/kg DEX (ADR+DEX [1.0]), or doxorubicin plus 10 mg/kg DEX (ADR+DEX [10]). (B) The frequencies of CD11b+Gr-1+ MDSCs in mouse BM, spleen, peripheral blood, KDLNs, and kidney under different concentrations of DEX treatment. The MDSC counts are defined as percentages of CD11b+ Gr-1+ cells in total BM cells, splenocytes (Spleen), white blood cells (Blood), KDLN cells, and mononuclear cells in kidney (Kidney) after depletion of red blood cells (RBCs). (C) Suppression of T cell proliferation by MDSCs isolated from the KDLNs and spleens of doxorubicin-induced mice treated with 1 mg/kg DEX. CD3+ T cells proliferation from three independent experiments was analyzed. (D) The numbers of CD4+ and CD8+ T cells in mouse spleen and peripheral blood treated with different concentrations of DEX. CD4+ or CD8+ T cell counts are defined as percentages of CD4+ or CD8+ cells in total splenocytes or white blood cells after depletion of RBCs. (E) Serum IL-6, TNF-α, IFN-γ, and IL-4 levels of mice treated with 1 mg/kg DEX were measured by ELISA. (F) Urinary albumin levels in doxorubicin-treated mice that received or did not receive 1 mg/kg DEX treatment. (G) Serum creatinine levels in doxorubicin-treated mice treated with or without 1 mg/kg DEX. (H) Representative periodic acid-Schiff–stained kidney sections from doxorubicin-treated mice treated with or without 1 mg/kg DEX for 2 weeks (original magnification, ×400). (I) Quantitative analysis of glomerular sclerosis and tubular damage. Damaged tubules were determined by the presence of diffuse tubular dilation, intraluminal casts, tubular cell vacuolization and detachment in cortex and medulla. ND, not detected. (J) Representative electron microscopy photomicrographs of podocytes of ADR-treated mice treated with or without 1 mg/kg DEX. Red arrows indicate the loss of foot process or foot process fusion. Data are shown as the mean±SEM (n=3; six samples per group). **P<0.01.
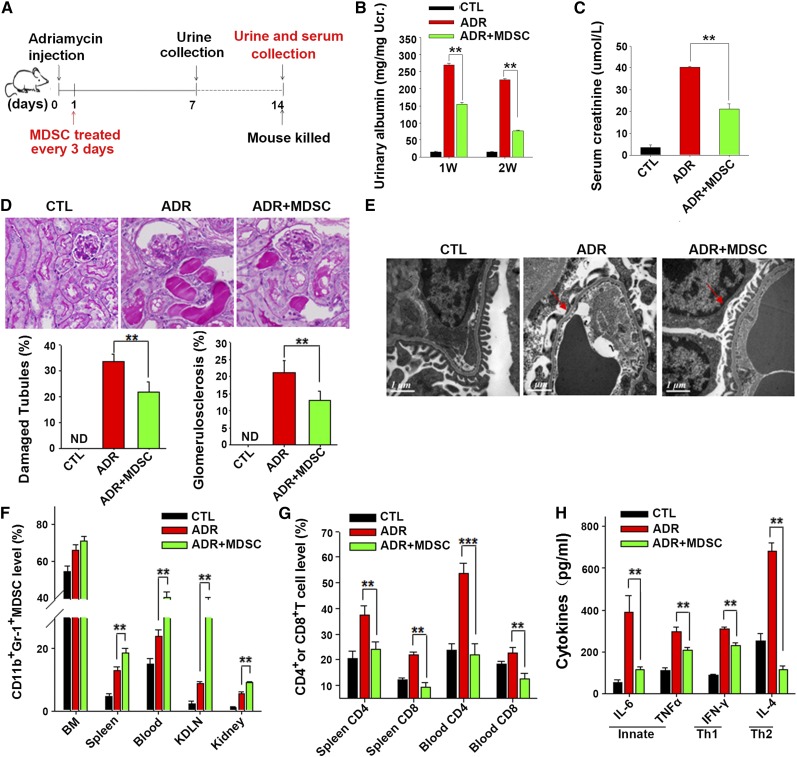
To directly view the effect of MDSCs on the attenuation of doxorubicin-induced renal injury in the mouse model, we isolated BM from BALB/c mice, induced the expansion of Bm-MDSCs by GM-CSF and IL-6,35 and then injected these BM-MDSCs into mice treated with doxorubicin every 3 days (Figure 4A). The transfer of BM-MDSCs strongly attenuated the doxorubicin-induced renal injury compared with that of mice without the BM-MDSC transfer. In the BM-MDSC group, mice showed a lower level of proteinuria (Figure 4B), lower serum creatinine (Figure 4C), less glomerular sclerosis and tubular damage (Figure 4D), less loss or fusion of foot processes (Figure 4E), and less body weight loss (Supplemental Figure 3), suggesting a protection of transferred BM-MDSCs against doxorubicin-induced renal injury. In concordance with this result, we found that adoptive transferred BM-MDSCs significantly upregulated the frequency of CD11b+Gr-1+ MDSCs in mouse Bm, spleen, blood, KDLNs, and kidney (Figure 4F). As expected, adoptive transfer of BM-MDSCs markedly decreased the numbers of CD4+ and CD8+ T cells in the spleen and blood (Figure 4G) and the levels of serum IL-6, TNF-α, IFN-γ, and IL-4 (Figure 4H) in doxorubicin-treated mice.
Figure 4.
Adoptively transferred MDSCs attenuated doxorubicin-induced mouse renal injury. (A) Diagram of the experimental design. MDSCs (5×106) induced from BM were injected into mice through the tail vein 1 day after doxorubicin treatment and injected every 3 days. Mice were divided into three groups: mice treated with saline (CTL), doxorubicin (Adriamycin; ADR) or doxorubicin plus BM-MDSCs (ADR+MDSC). (B) Urinary albumin levels in doxorubicin-treated mice with or without adoptively transferred MDSCs. (C) Serum creatinine levels in doxorubicin-treated mice with or without MDSC transfer. (D) Representative periodic acid-Schiff–stained kidney sections from doxorubicin-treated mice with or without MDSC transfer. The histograms represent quantitative analysis of glomerular sclerosis and tubular damage. Damaged tubules were determined by the presence of diffuse tubular dilation, intraluminal casts, tubular cell vacuolization, and detachment in cortex and medulla. ND, not detected (original magnification, ×400). (E) Representative electron microscopy photomicrographs of mouse podocytes of doxorubicin-treated mice with or without adoptively transferred MDSCs. Red arrows represent the loss of foot process or foot process fusion. (F) The frequencies of CD11b+Gr-1+ MDSCs in the BM, spleen, peripheral blood, KDLNs, and kidney of doxorubicin-treated mice with or without adoptively transferred BM-MDSCs. MDSC counts are presented as percentages of CD11b+ Gr-1+ cells in total BM cells, splenocytes (Spleen), blood cells (Blood), KDLN cells, and mononuclear cells in kidney (Kidney) after depletion of red blood cells (RBCs). (G) The inhibition of proliferation of CD4+ and CD8+ T cells in mouse spleen and peripheral blood by adoptively transferred MDSCs. CD4+ or CD8+ T cell counts are presented as percentages of CD4+ or CD8+ cells in total splenocytes or blood cells depleted of RBCs. (H) Serum IL-6, TNF-α, IFN-γ, and IL-4 levels of doxorubicin-treated mice with or without adoptively transferred BM-MDSCs were determined. Values are shown as the mean±SEM (n=3; five samples per group). **P<0.01.
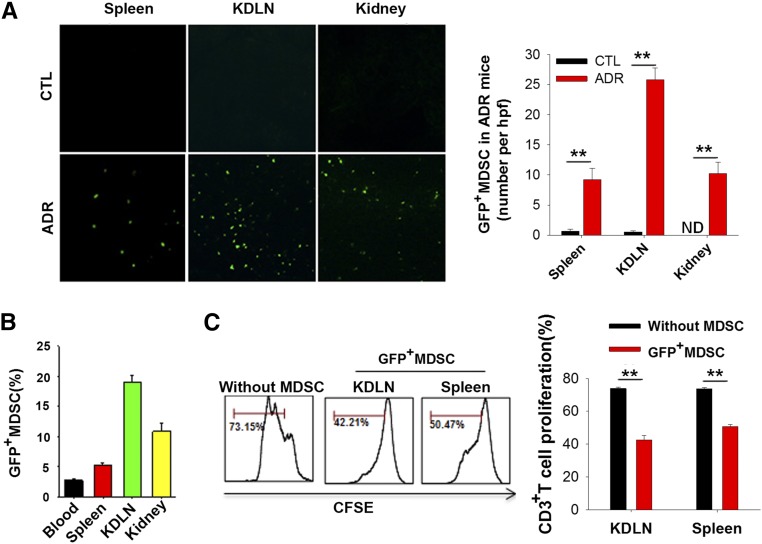
To trace the transferred MDSCs in mice under the condition of doxorubicin treatment, BM-MDSCs were induced from green fluorescent protein (GFP) transgenic mice. The tissue distribution of GFP-positive BM-MDSCs in the host mice was directly visualized following tissue dissection. In the doxorubicin-treated mice, GFP transgenic BM-MDSCs were readily detected in the mouse spleen, KDLN and kidney at 3 hours after injection (Figure 5, A and B). In contrast, no GFP transgenic BM-MDSCs were detected in the spleen, KDLN and kidney of mice without ADR treatment. To test the inhibitory capacity of the transferred MDSCs, GFP-positive MDSCs were isolated from mouse KDLN and spleen at 2 weeks after injection. As shown in Figure 5C, the isolated GFP-positive MDSCs significantly inhibited T cell proliferation. These results suggested that the transferred BM-MDSCs readily migrated to certain organs, particularly to where lymphocytes accumulated under inflammatory conditions.
Figure 5.
Recruitment of transferred MDSCs into the inflammatory sites. (A) Infiltration of transferred GFP+ BM-MDSCs into mouse spleen, KDLNs, and kidney under inflammatory conditions. BM-MDSCs obtained from GFP transgenic mice were injected intravenously into mice treated with or without doxorubicin (original magnification, ×200). The right panel represents the numbers of GFP+ BM-MDSCs. ND, not detected. Data are mean±SEM (n=3; six high-power field per group). (B) FACS analysis of GFP-positive BM-MDSCs in blood, spleen, KDLNs, and kidney. GFP+ MDSC counts are defined as percentages of GFP+ cells in total white blood cells (Blood), splenocytes (Spleen), KDLN cells, and mononuclear cells in kidney (Kidney) after depletion of red blood cells. (C) Infiltrated GFP+ BM-MDSCs in KDLN and spleen inhibited T cell proliferation. GFP+ BM-MDSCs were isolated from KDLNs and spleen of doxorubicin-treatedmice at week 2 after injection and then co-cultured with lymphocytes (MDSC:lymphocytes, 1:2) in the presence of ConA. CD3+ T cell proliferation was determined 4 days later. The right histogram represents the statistical analysis from three independent experiments (six samples per group). Data are shown as the mean±SEM. **P<0.01.
Depletion of MDSCs Aggravated Doxorubicin-Induced Renal Injury and Abolished the Protective Effect of GCs
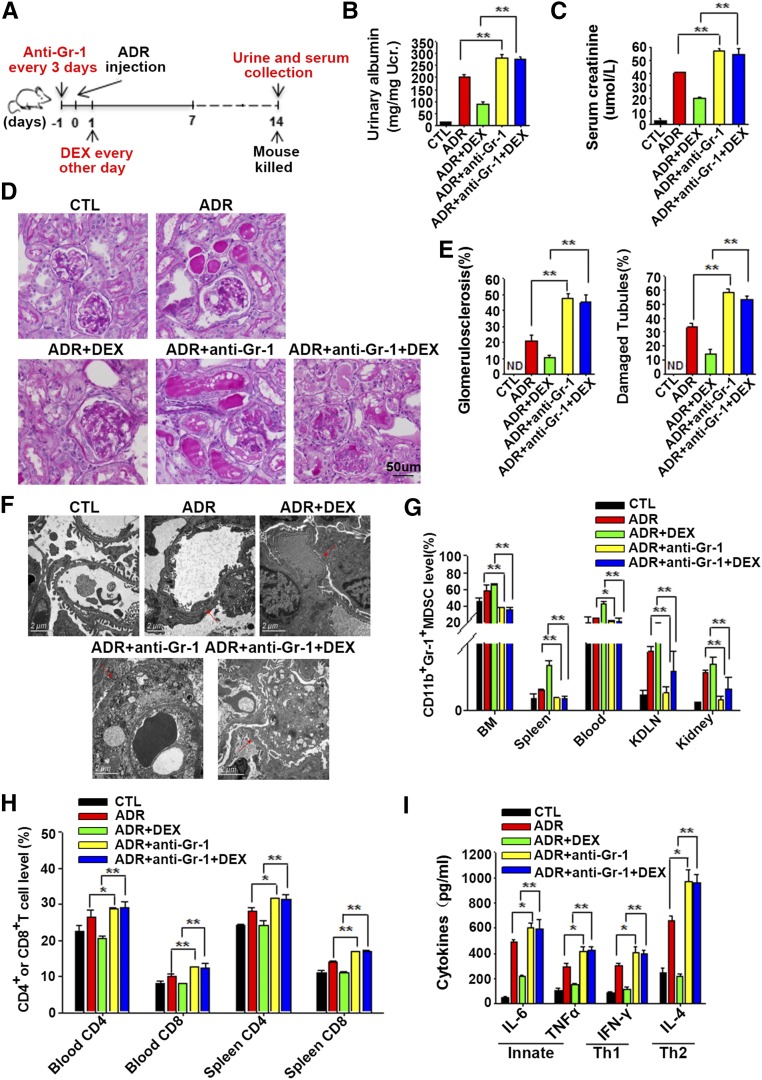
To confirm the protective effect of MDSCs on doxorubicin-induced renal injury, doxorubicin-induced mouse renal injury was monitored after depleting MDSCs using an anti–Gr-1 antibody.36 As shown in Figure 6A, mice were divided into five groups: mice administered with saline, mice treated with doxorubicin; mice treated with doxorubicin and dexamethasone; mice treated with doxorubicin and an anti–Gr-1 antibody; and mice treated with ADR, an anti–Gr-1 antibody and dexamethasone. Renal function of each mouse was measured at the end of the experiment. As shown in Figure 6, B and C, the levels of urinary albumin and serum creatinine were dramatically increased in mice with MDSC depletion compared with that of mice without MDSC depletion. Examination of renal pathologic findings showed that MDSC depletion in mice treated with doxorubicin resulted in more severe glomerular sclerosis and tubular damage (Figure 6, D and E), more loss of foot processes, and more vacuolization compared with mice without MDSC depletion (Figure 6F). Furthermore, as shown in Supplemental Figure 3, mice in the MDSC-depleted group showed more bodyweight loss.
Figure 6.
Depletion of MDSCs aggravated doxorubicin-induced podocyte injury and abolished the protective effect of GCs. (A) Diagram of the experimental design. The anti–Gr-1 antibody was injected intraperitoneally into mice 1 day before doxorubicinadministration and subsequently injected every 3 days. Dexamethasone (DEX) (1 mg/kg) was injected intraperitoneally 1 day after doxorubicin administration and injected every other day. Mice were divided into five groups: mice treated with saline (CTL); doxorubicin (Adriamycin [ADR]); doxorubicin plus DEX (ADR+DEX); doxorubicin plus anti–Gr-1 antibody (ADR + anti–Gr-1); or doxorubicin, anti–Gr-1 antibody, and DEX (ADR+anti–Gr-1+DEX). Urine and serum were collected at 2 weeks. (B) Urinary albumin levels in mice from the five different groups. (C and D) Serum creatinine levels (C) and representative periodic acid-Schiff–stained kidney sections (D) from different groups of mice (original magnification, ×400). (E) Quantitative analysis of glomerular sclerosis and tubular damage. Damaged tubules were determined by the presence of diffuse tubular dilation, intraluminal casts, tubular cell vacuolization, and detachment in cortex and medulla. ND, not detected. (F) Representative electron microscopy photomicrographs of mouse podocytes. Red arrows represent the loss of foot process or foot process fusion. (G) Frequencies of CD11b+Gr-1+ MDSCs in BM, spleen, peripheral blood, KDLNs, and kidney of different groups of mice. MDSC counts are presented as percentages of CD11b+Gr-1+ cells in total BM cells, splenocytes (Spleen), white blood cells (Blood), KDLN cells, and mononuclear cells in kidney (Kidney) after depletion of red blood cells (RBCs). (H) CD4+ and CD8+ T cell levels in mouse spleen and peripheral blood from different groups. CD4+ or CD8+ T cell counts are presented as percentages of CD4+ or CD8+ cells in total splenocytes or white blood cells after depletion of RBCs. (I) Levels of mouse serum IL-6, TNF-α, IFN-γ, and IL-4. Data are shown as the mean±SEM (n=3; six samples per group). *P<0.05; **P<0.01.
Next, the effect of anti–Gr-1 antibody administration on the numbers of MDSCs, T cells, and cytokines was examined. As expected, the administration of anti–Gr-1 antibody effectively downregulated the frequency of CD11b+Gr-1+MDSCs in BM, spleen, blood, KDLNs, and kidney (Figure 6G). The depletion of MDSCs led to a marked increase in the frequencies of CD4+ and CD8+ T cells in both spleen and blood of doxorubicin-treated mice (Figure 6H). The serum levels of IL-6, TNF-α, IFN-γ, and IL-4 were also increased in MDSC-depleted mice (Figure 6I). Interestingly, dexamethasone did not reverse doxorubicin-induced renal injury in MDSC-depleted mice, as indicated by severe loss of foot processes, glomerular sclerosis and tubular damage, more inflammatory T cell infiltration, and higher concentrations of proinflammatory cytokines, suggesting that MDSCs play an essential role in dexamethasone-mediated protection against doxorubicin-induced renal injury.
MDSCs Attenuated Doxorubicin-Induced Renal Injury via Inducing Tregs
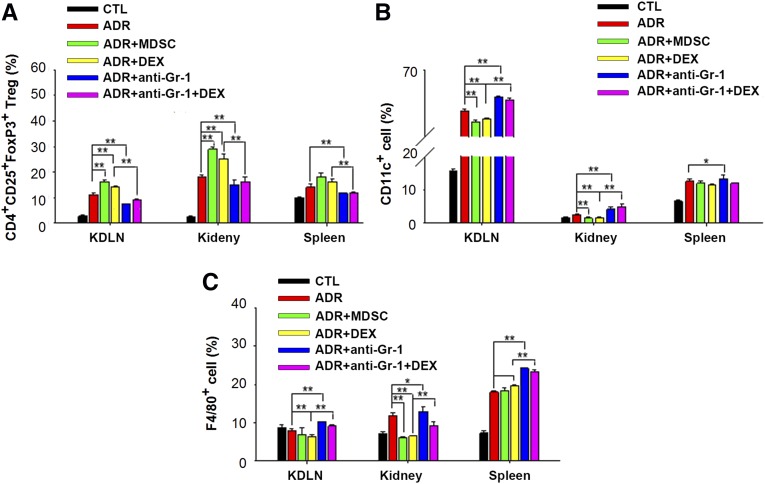
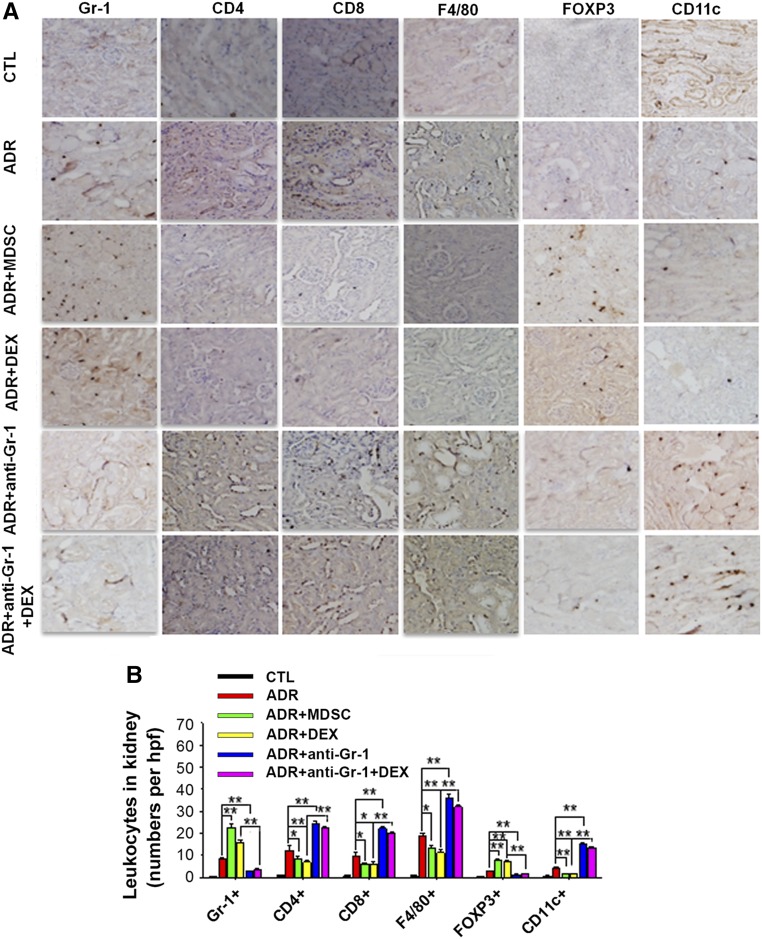
Given that MDSCs decreased the levels of proinflammatory cytokines in doxorubicin-treated mice, we next investigated whether the protective role of MDSCs depends on their capacity to induce Tregs or suppress other inflammatory leukocytes. As shown in Figure 7, A–C, and Supplemental Figure 4, transfer of MDSCs or injection of dexamethasone significantly induced the proliferation of CD4+CD25+FoxP3+ Tregs but decreased the number of CD11c+ and F4/80+ cells in KDLNs, kidney, and spleen of doxorubicin-treated mice. In contrast, depletion of MDSCs increased CD11c+ and F4/80+ macrophages but decreased CD4+CD25+FoxP3+ Tregs in KDLNs, kidney, and spleen of doxorubicin-treated mice. The immune staining also showed that transfer of MDSCs or injection of dexamethasone resulted in less infiltration of CD11c+ cells, F4/80+ macrophages, CD4+ T cells, and CD8+ T cells but more infiltration of Tregs in the mouse interstitium, whereas depletion of MDSCs resulted in more infiltration of F4/80+ macrophages, CD11c+ cells, CD4+ T cells, and CD8+ T cells but less infiltration of Tregs in mouse interstitium (Figure 8, A and B). These results suggest that MDSCs may attenuate doxorubicin-induced renal injuries by modulating Tregs and immune cells.
Figure 7.
MDSCs increased the level of Tregs but decreased the levels of inflammatory leukocytes in mouse KDLN, kidney, and spleen. (A–C) FACS analysis of CD4+CD25+FoxP3+ Tregs (A), CD11c+ cells (B), and F4/80+ cells (C) in KDLNs, kidney, and spleen from mice administered saline (CTL); doxorubicin (Adriamycin [ADR]); doxorubicin and BM-MDSCs (ADR+MDSC); doxorubicin and dexamethasone (DEX; ADR+DEX); doxorubicin and anti–Gr-1 antibody (ADR + anti–Gr-1); or doxorubicin, anti–Gr-1 antibody, and DEX (ADR+anti–Gr-1+DEX) for 2 weeks. The y-axis is defined as percentages of CD4+CD25+FoxP3+ cells (A), CD11c+ cells (B), and F4/80+ macrophages (C) in total splenocytes (Spleen), KDLN cells, and mononuclear cells in kidney (Kidney) after depletion of red blood cells, respectively. Data are shown as the mean±SEM (n=3; six samples per group). *P<0.05; **P<0.01.
Figure 8.
Infiltration of Tregs and immune cells in mouse kidney. (A) Representative photomicrographs of immunohistochemical staining of renal sections from different groups of mice (original magnification, ×200). (B) The numbers of infiltrated Tregs and leukocytes in mouse kidney. Data are shown as the mean±SEM (n=3; six high-power fields per group). *P<0.05; **P<0.01.
DISCUSSION
Although the cause of FSGS is complicated, it has long been considered a T cell–driven disease in which lymphokines, the "circulating factor," induce proteinuria and podocyte functional and structural derangement. In FSGS, monocytes and macrophages, T cells, and their mediators, such as Tregs, play an important role in the inflammatory process. The variety of inflammatory cells infiltrating into the interstitium and the correlation with the degree of renal insufficiency are important features of human and experimental FSGS.37–39 Doxorubicin-induced renal injury is a common mouse or rat model resembling FSGS.40 Although doxorubicin-induced renal injury is usually ascribed to a direct toxic effect of doxorubicin, an inflammatory mechanism for doxorubicin-induced podocyte injury has been extensively reported.7,40,41 Renal expression of TNF-α, IL-1β, and monocyte chemotactic protein-1 was increased in response to doxorubicin.41 Inflammatory CD8+ T cells7 and NF-κB40 play substantial roles in doxorubicin-induced renal injury. Using a doxorubicin-induced mouse model, Wang and coworkers42 reported the involvement of CD4+ T cells in the pathogenesis of the interstitial lesion in doxorubicin nephropathy. Amore et al.43 suggested that an imbalance of T cell subsets induced by doxorubicin is likely the initial step to the appearance of proteinuria and nephropathy. By inhibiting T cell activities, Tregs protected animals against chronic renal injury induced by doxorubicin.9
The main feature of MDSCs is an immunosuppressive function that MDSCs can suppress T cell proliferation and leukocyte inflammatory responses. However, although MDSCs have been reported to regulate innate and adaptive immunity in various inflammatory and autoimmune diseases, few studies have explored their roles in modulating chronic renal diseases. Here, we present the first evidence of the MDSCs responding to GC treatment and the effectiveness of GC treatment in human FSGS. Our results showed that MDSCs could be induced by GC treatment and protected mice against doxorubicin-induced renal functional impairment and structural derangement. Because the MDSCs expanded at the first week and then gradually reduced at later stage, the elevation of MDSCs may play a critical role in the initial host resistance to doxorubicin-induced renal injuries.
As an immune suppressor, GCs are effective anti-inflammatory treatments for asthma, rheumatoid arthritis, inflammatory bowel disease, and autoimmune diseases.44 FSGS has long been regarded as a T cell–driven condition.4 Systemic administration of GCs can result in GN remission.45 Accompanied by the remission of GN, a certain degree of leukocytosis has been observed in patients with nephrotic syndrome after GC treatment.31,46 GCs can suppress inflammation and immune function through various mechanisms, including interfering with the function of T lymphocytes, reducing the recruitment of monocytes and macrophages, and inhibiting the release of inflammatory cytokines by immune cells.47–49 A recent study by Varga et al.48 reported that GC treatment could induce an anti-inflammatory monocyte subset in mice that resemble myeloid-derived suppressor cells. By assessing the MDSCs in patients with FSGS who are sensitive or insensitive to GCs, we demonstrate for the first time that the expansion of immunosuppressive MDSCs may be a major mechanism underlying the protective or therapeutic effect of GCs in FSGS. Compared with GC-insensitive patients with FSGS, those who are GC sensitive produce significantly more MDSCs at the early stage of GC treatment. These rapidly induced MDSCs would suppress the proliferation and activation of T cells and thus attenuate the inflammation and renal injury in GC-sensitive patients. Given that MDSCs will eventually differentiate into mature neutrophils and monocytes, this may explain why leukocytosis is often observed after GC treatment in patients with nephritic syndrome patients.33
We also confirmed the essential role of MDSCs in the GC-mediated protection of renal injury using a doxorubicin-induced mouse renal injury model. Although the response of doxorubicin-treated mice to steroid has not been characterized, several studies have shown altered expression of plasma proteins50 and podocyte proteins51 and an attenuation of glomerular filtration barrier damage52 in rat nephropathy model following steroid treatment. Our results show that dexamethasone treatment strongly promotes the expansion of MDSCs in mouse BM, spleen, peripheral blood, KDLNs, and kidney in the presence of doxorubicin. Of note, when MDSCs in mice are depleted by injecting the anti–Gr-1 antibody, the effect of dexamethasone on ameliorating doxorubicin-induced renal injury and suppressing T cell–mediated immune response is largely abolished. These results confirm that the generation of immunosuppressive MDSCs may serve as an important mechanism by which GCs suppress chronic inflammation and autoimmunity. A cell tracing experiment provides direct evidence that under inflammation, exogenous MDSCs can migrate to lymphocyte-accumulating organs, such as the spleen and KDLNs, where they suppress the proliferation of T cells. Given that the effect of GCs on attenuating renal injuries and chronic inflammation depends on MDSCs, the level of MDSCs in patients’ peripheral blood after GC treatment may provide an early indicator to predict the efficacy of GC-based therapy for individual patients with FSGS.
In the present study, MDSCs were depleted by anti–Gr-1 antibody treatment. Anti–Gr-1 antibody also reduced the production of neutrophils and monocytes. Because neutrophils and monocytes are major proinflammatory cells under acute inflammation, deficient production of neutrophils and monocytes should attenuate the inflammation and the inflammation-induced tissue injury. However, our data clearly showed that anti–Gr-1 antibody treatment largely abolished the protective effect of dexamethasone on doxorubicin-induced renal injury. The mice treated with dexamethasone and anti–Gr-1 antibody showed more severe doxorubicin-induced renal injury and inflammation compared with the mice treated with dexamethasone alone. The effect of anti–Gr-1 antibody on aggravating doxorubicin-induced renal injury suggests that reduction of neutrophils and monocytes by anti–Gr-1 antibody plays a minor role in abolishing the protection of dexamethasone on doxorubicin-induced renal injury.
In addition to the abnormalities of subsets of T cells in FSGS, the role of cytokines in the development of FSGS has also attracted attention. IL-4 production by Th2 cells was upregulated in patients with membranous nephropathy and correlated with the severity of proteinuria.53 Yano et al.54 found that the levels of IL-4, IL-10, IFN-γ, IL-1β, and IL-12 in the supernatants of peripheral blood mononuclear cells from patients with non-IgA mesangial proliferative GN are higher than those in healthy controls and that the level of IFN-γ correlated with the degree of renal injury. Futrakul et al.55 reported that the immune imbalance caused by decreased anti-inflammatory factor dominated the mechanism of FSGS. In the present study, we also found an increase in IL-6, TNF-α, IFN-γ, and IL-4 levels in mice with doxorubicin-induced renal injury, whereas adoptive transferred BM-MDSCs or MDSC-expanding dexamethasone strongly decreased the levels of these cytokines. Thus, MDSCs may attenuate doxorubicin-induced mouse renal injuries by controlling the production of inflammatory cytokines. In summary, our study demonstrates that GC-induced or adoptively transferred MDSCs protect mice from doxorubicin-induced injury via suppressing CD4+ and CD8+ T cells, CD11c+ dendritic cells, and F4/80+ macrophages but promoting anti-inflammatory Tregs. The number of MDSCs in peripheral blood of patients with FSGS after GC treatment may serve as an indicator for predicting the efficacy of GC treatment.
CONCISE METHODS
Patients
The Human Subjects Committee of Jinling Hospital, Nanjing University School of Medicine, Nanjing, China, approved all protocols concerning the use of patient samples in this study. Each donor provided a signed consent form. Blood samples were collected from consenting healthy donors and patients with FSGS (with signed consent form) for MDSC analysis. All patients were diagnosed by renal biopsy at National Clinical Research Center of Kidney Diseases, Jinling Hospital, Nanjing University School of Medicine. The patients were selected according to the following criteria: (1) proteinuria >0.4 g/24 hours; (2) serum creatinine <265.2 μmol/L; (3) global glomerulosclerosis with <50% of glomeruli; and (4) absence of obesity, diabetes mellitus, hepatitis C virus infection, hepatitis, and malignant tumors. In this study, 13 normal controls, 86 patients with FSGS and 43 patients with minimal-change disease sensitive to GC treatment, and 23 patients with FSGS and 5 patients with minimal-changed disease insensitive to GC treatment were enrolled.
Mice
Animal maintenance and experimental procedures were carried out in accordance with the National Institutes of Health Guidelines for Use of Experimental Animals and approved by the Medicine Animal Care Committee of Nanjing University. Eight-week-old male BALB/c mice that weighed 20–22 g were obtained from the Model Animal Research Center (Nanjing, China).
Reagents and Antibodies
Murine IL-6 and GM-CSF cytokines were obtained from PEPROTECH (Rocky Hill, NJ). Doxorubicin hydrochloride, dexamethasone, APO, and L-NMMA were obtained from Sigma-Aldrich (St. Louis, MO). PE-conjugated anti-mouse CD11b and PerCP/Cy5.5 anti-mouse Gr-1 were purchased from Biolegend (San Diego, CA). FITC-conjugated rat anti-mouse CD4 and CD8a antibodies were purchased from BD Biosciences (Franklin Lakes, NJ). APC-conjugated anti-mice CD3, APC-conjugated anti-human HLA-DR, Fluor 488-conjugated anti-human CD14, PE-conjugated anti-human CD11b, and PerCP/Cy5.5 anti-human CD15 were purchased from Biolegend. Purified anti-mouse CD4, CD8a, F4/80, and Ly-6G/Ly-6C (Gr-1) antibodies for immunohistochemistry were also purchased from Biolegend. Purified anti-mouse CD11c and FoxP3 antibodies for immunohistochemistry were purchased from eBioscience.
Flow Cytometry Analysis
KDLN cells and spleen cells were isolated from mice using standard methods. In brief, KDLNs and spleen were finely minced, grinded, passed through 70-μm nylon cell strainer, centrifuged, and depleted the red blood cells. The remaining cells were used for FACS analysis. Whole blood was centrifuged to get rid of plasma. The lower cells were lysed to remove red blood cells for FACS analysis. Mononuclear cells of kidney were prepared with a little modification as described by Gentle et al.56 In brief, kidney was minced and digested with RPMI-1640 containing 1 mg/ml collagenase D and 100 U/ml DNase I for 45 minutes at 37°C. The digestion was then passed through a 70-μm nylon cell strainer and centrifuged at 50g for 5 minutes to get rid of large pieces of tissues. The supernatants were then centrifuged at 350g for 5 minutes and pellets were washed with flow buffer (PBS containing 2% FBS and 2 mM EDTA), resuspended in 40% Percoll (Sigma-Aldrich) in RPMI 1640 medium, and overlaid on 66.7% Percoll solution. Gradient separation was carried out at 750g for 20 minutes at room temperature. Pellets were washed extensively with Hank buffer devoid of Ca2+ and Mg2+ (HBSS) before FACS. Flow analysis was conducted using a BD FACScalibur device and analyzed with FCS express V3. After washing with HBSS, 5×105 cells from BM, blood, spleen, KDLNs, and kidney were blocked using 1% BSA at 4°C for 30 minutes. Antibodies were added for incubation for another 30 minutes at 4°C.
Adoptive Transfer of BM-Derived MDSCs
BM-derived MDSCs were prepared as previously described.49 In brief, cells were plated into dishes using RPMI 1640 medium supplemented with 2 mM L-glutamine, 10 mM HEPES, 20 µM 2-mercaptoethanol, 150 U/ml streptomycin, 200 U/ml penicillin, and 10% FBS and stimulated with combinations of GM-CSF (40 ng/ml) and IL-6 (40 ng/ml). Cells were cultured at 37°C in a 5% CO2-humidified atmosphere for 4 days, and 5×106 induced BM-MDSCs were injected into the tail vein of each BALB/c mouse. Mice were divided into three groups (five mice in each group): (1) the control group (which received saline only), (2) the doxorubicin group (which received doxorubicin only), and (3) the doxorubicin+MDSC group. Doxorubicin was injected via the tail vein of each nonanesthetized mouse (10 mg/kg). Mice in the control group were treated with saline only. Body weights were measured daily. BM, blood, spleen, and kidney samples were obtained from mice in each group.
MDSC Depletion Assay
MDSCs were depleted in vivo by intraperitoneal injections of 100 μg of a purified anti-mouse Ly-6G/Ly-6C (Gr-1, clone RB6–8C5) antibody (BD Biosciences) per mouse every 3 days.36 Control mice were given equivalent amounts of purified rat IgG (BD Biosciences). The efficiency of antibody administration was confirmed by FACS.
Dexamethasone Treatment and Renal Function
Mice were divided into three groups (five mice in each group): (1) the control group (which received saline only), (2) the doxorubicin group (which received doxorubicin only), and (3) the doxorubicin+dexamethasone group. Doxorubicin was injected via the tail vein of each nonanesthetized mouse (10 mg/kg). Different concentrations of dexamethasone (0.1, 1, and 10 mg/kg) were injected intraperitoneally into mice each day. BM, blood, spleen, and kidney samples were obtained from mice in each group. Renal function was assessed by measuring urine albumin and creatinine. Urine was collected from each mouse for 24 hours. Urine albumin was measured using a mouse albumin ELISA kit (Bethyl Laboratories, Montgomery, TX). Urine and serum creatinine was determined as previously described.57
T Cell Proliferation Assays
To obtain high purity in MDSCs from PBMCs or spleen and KDLNs, a cell isolation kit (Miltenyi Biotec, Bergisch Gladbach, Germany) was used according to the manufacturer’s instructions. For the mouse T cell inhibition assay, splenocytes were first separated with lymphocyte separation medium. Lymphocytes were labeled with CFSE according to the manufacturer’s instructions (Invitrogen, Carlsbad, CA). CFSE-labeled lymphocytes were stimulated with ConA (Sigma-Aldrich), and lymphocytes were co-cultured at 2:1 and 4:1 ratios with purified MDSCs in 96-well flat bottom plates. On the fourth day, the cultured cells were incubated with APC-conjugated anti-CD3 antibody. APC-positive cells were gated for further CFSE-labeled T cell proliferation analysis. For human HLA-DR−CD15+ cell inhibiting assay, anti–HLA-DR microbeads for the depletion of HLA-DR expressing cells from human peripheral blood and CD15-conjugated microbeads for positive selection of CD15+ cells from human peripheral blood were purchased from Miltenyi Biotec. T cells were sorted from PBMCs of healthy donors by anti-CD3 microbeads (Miltenyi Biotec), labeled with CFSE, and seeded in 96-well plates with HLA-DR−CD15+ cells at a 2:1 ratio. T cell proliferation was induced by anti-CD3/CD28 stimulation beads (Invitrogen). T cell proliferation was analyzed by flow cytometry on day 4.
Electron Microscopy Examination of the Kidney
Renal tissues (l mm3 size) were fixed in cold 3.75% glutaraldehyde for 4 hours. After washing in 0.1 M phosphate buffer (pH, 7.5) five to six times, tissues were fixed in 2% osmium tetroxide for 2 hours and then dehydrated in acetone and ethanol and embedded. Ultrathin sections (80–90 nm) were stained with uranyl acetate and lead citrate. The sections were viewed on a Hitachi 7500 transmission electron microscope (Hitachi Co., Japan).
Histology and Immunohistochemistry
Coronal sections of renal tissue were stained with periodic acid–Schiff. Glomerulosclerosis and damaged tubules were evaluated using methods described previously.58 Immunohistochemical staining was performed using a routine protocol.9 Primary antibodies were all from Biolegend. The secondary biotinylated rabbit anti-rat immunoglobulin was from Dako (Carpinteria, CA).
Tracing of BM-Derived MDSCs in Mice Treated with Doxorubicin
Fluorescent BM-MDSCs were obtained from GFP transgenic mice after GM-CSF and IL-6 induction and injected intravenously (5×106 per mouse) into doxorubicin-treated mice. For tracing the location of fluorescently labeled BM-MDSCs, mice were euthanized 3 hours after injection. The spleens, KDLNs, and kidneys were removed to prepare frozen sections or FACS analysis. For inhibiting assay, MDSCs (5×106) induced from BM of GFP transgenic mice were injected into mice through the tail vein 1 day after doxorubicin treatment and injected every 3 days. At 2 weeks, GFP-positive MDSCs were sorted from KDLN and spleen and co-cultured with T cells for T cell proliferation analysis.
Detection of Reactive Oxygen Species
H2O2 was quantified using Amplex Red Hydrogen Peroxide/Peroxidase Assay Kit (Molecular Probes) according to the manufacturer’s protocol. Briefly, 2×104 cells were resuspended in HBSS (Sigma-Aldrich). After addition of 30 ng/ml phorbol myristate acetate, the absorbance at 560 nm was measured using a microplate plate reader (Molecular Devices SpectraMax M2) at 37°C. Absorbance results were normalized to a standard curve generated by serial dilutions of 20 mM H2O2.
Measurement of Serum Cytokine Levels
Whole blood was collected without anticoagulant and the serum was isolated by centrifugation. Serum levels of IL-4, IL-6, TNF-α, and IFN-γ were determined using ELISA kits (R&D Systems). The absorbance was measured using wavelength correction (A450 nm) with a microplate reader (Bio-Rad).
Statistical Analyses
All of the data were analyzed using statistical software SPSS 19.0. Variables are expressed as the mean±SEM or mean±SD. Normally distributed variables were compared using t test. More than two group differences were analyzed by ANOVA. Nonparametric variables were expressed as the median (interquartile range) and were compared using Mann–Whitney or Kruskal–Wallis test. Data described as percentage were analyzed using a chi-squared test or Fisher exact test. The paired data were analyzed by paired t test or Wilcoxon test. The reported P value was two sided. P<0.05 was considered to be statistically significant.
Disclosures
None.
Acknowledgments
This work was supported by grants from National Basic Research Program of China (973 Program, 2012CB517603 and 2011CB504803), National Natural Science Foundation of China (no. 31301061), Natural Science Foundation of Jiangsu Province (no. BK20130564) and Specialized Research Fund for the Doctoral Program of Higher Education (20130091120037).
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2014050468/-/DCSupplemental.
References
- 1.Troyanov S, Wall CA, Miller JA, Scholey JW, Cattran DC, Toronto Glomerulonephritis Registry Group : Focal and segmental glomerulosclerosis: Definition and relevance of a partial remission. J Am Soc Nephrol 16: 1061–1068, 2005 [DOI] [PubMed] [Google Scholar]
- 2.Kitiyakara C, Kopp JB, Eggers P: Trends in the epidemiology of focal segmental glomerulosclerosis. Semin Nephrol 23: 172–182, 2003 [DOI] [PubMed] [Google Scholar]
- 3.Barisoni L, Schnaper HW, Kopp JB: Advances in the biology and genetics of the podocytopathies: implications for diagnosis and therapy. Arch Pathol Lab Med 133: 201–216, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shalhoub RJ: Pathogenesis of lipoid nephrosis: A disorder of T-cell function. Lancet 2: 556–560, 1974 [DOI] [PubMed] [Google Scholar]
- 5.Kim AH, Markiewicz MA, Shaw AS: New roles revealed for T cells and DCs in glomerulonephritis. J Clin Invest 119: 1074–1076, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Turner JE, Paust HJ, Steinmetz OM, Panzer U: The Th17 immune response in renal inflammation. Kidney Int 77: 1070–1075, 2010 [DOI] [PubMed] [Google Scholar]
- 7.Wang Y, Wang YP, Tay YC, Harris DC: Role of CD8(+) cells in the progression of murine adriamycin nephropathy. Kidney Int 59: 941–949, 2001 [DOI] [PubMed] [Google Scholar]
- 8.Le Berre L, Bruneau S, Naulet J, Renaudin K, Buzelin F, Usal C, Smit H, Condamine T, Soulillou JP, Dantal J: Induction of T regulatory cells attenuates idiopathic nephrotic syndrome. J Am Soc Nephrol 20: 57–67, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang YM, Zhang GY, Wang Y, Hu M, Wu H, Watson D, Hori S, Alexander IE, Harris DC, Alexander SI: Foxp3-transduced polyclonal regulatory T cells protect against chronic renal injury from adriamycin. J Am Soc Nephrol 17: 697–706, 2006 [DOI] [PubMed] [Google Scholar]
- 10.Turner JE, Paust HJ, Steinmetz OM, Peters A, Riedel JH, Erhardt A, Wegscheid C, Velden J, Fehr S, Mittrücker HW, Tiegs G, Stahl RAK, Panzer U: CCR6 recruits regulatory T cells and Th17 cells to the kidney in glomerulonephritis. J Am Soc Nephrol 21: 974–985, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rydel JJ, Korbet SM, Borok RZ, Schwartz MM: Focal segmental glomerular sclerosis in adults: presentation, course, and response to treatment. Am J Kidney Dis 25: 534–542, 1995 [DOI] [PubMed] [Google Scholar]
- 12.Braun N, Schmutzler F, Lange C, Perna A, Remuzzi G, Risler T, Willis NS: Immunosuppressive treatment for focal segmental glomerulosclerosis in adults. Cochrane Database Syst Rev (3): CD003233, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Abrantes MM, Cardoso LSB, Lima EM, Penido Silva JM, Diniz JS, Bambirra EA, Oliveira EA: Predictive factors of chronic kidney disease in primary focal segmental glomerulosclerosis. Pediatr Nephrol 21: 1003–1012, 2006 [DOI] [PubMed] [Google Scholar]
- 14.Moranne O, Watier L, Rossert J, Stengel B, GN-Progress Study Group : Primary glomerulonephritis: an update on renal survival and determinants of progression. QJM 101: 215–224, 2008 [DOI] [PubMed] [Google Scholar]
- 15.Bakir AA, Share DS, Levy PS, Arruda JAL, Dunea G: Focal segmental glomerulosclerosis in adult African Americans. Clin Nephrol 46: 306–311, 1996 [PubMed] [Google Scholar]
- 16.Greenstein S, Ghias K, Krett NL, Rosen ST: Mechanisms of glucocorticoid-mediated apoptosis in hematological malignancies. Clin Cancer Res 8: 1681–1694, 2002 [PubMed] [Google Scholar]
- 17.Schmidt S, Rainer J, Ploner C, Presul E, Riml S, Kofler R: Glucocorticoid-induced apoptosis and glucocorticoid resistance: molecular mechanisms and clinical relevance. Cell Death Differ 11[Suppl 1]: S45–S55, 2004 [DOI] [PubMed] [Google Scholar]
- 18.Wüst S, van den Brandt J, Tischner D, Kleiman A, Tuckermann JP, Gold R, Lühder F, Reichardt HM: Peripheral T cells are the therapeutic targets of glucocorticoids in experimental autoimmune encephalomyelitis. J Immunol 180: 8434–8443, 2008 [DOI] [PubMed] [Google Scholar]
- 19.Gabrilovich DI, Ostrand-Rosenberg S, Bronte V: Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol 12: 253–268, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Movahedi K, Guilliams M, Van den Bossche J, Van den Bergh R, Gysemans C, Beschin A, De Baetselier P, Van Ginderachter JA: Identification of discrete tumor-induced myeloid-derived suppressor cell subpopulations with distinct T cell-suppressive activity. Blood 111: 4233–4244, 2008 [DOI] [PubMed] [Google Scholar]
- 21.Bronte V, Zanovello P: Regulation of immune responses by L-arginine metabolism. Nat Rev Immunol 5: 641–654, 2005 [DOI] [PubMed] [Google Scholar]
- 22.Zea AH, Rodriguez PC, Atkins MB, Hernandez C, Signoretti S, Zabaleta J, McDermott D, Quiceno D, Youmans A, O’Neill A, Mier J, Ochoa AC: Arginase-producing myeloid suppressor cells in renal cell carcinoma patients: A mechanism of tumor evasion. Cancer Res 65: 3044–3048, 2005 [DOI] [PubMed] [Google Scholar]
- 23.Corzo CA, Cotter MJ, Cheng P, Cheng F, Kusmartsev S, Sotomayor E, Padhya T, McCaffrey TV, McCaffrey JC, Gabrilovich DI: Mechanism regulating reactive oxygen species in tumor-induced myeloid-derived suppressor cells. J Immunol 182: 5693–5701, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pak AS, Wright MA, Matthews JP, Collins SL, Petruzzelli GJ, Young MRI: Mechanisms of immune suppression in patients with head and neck cancer: Presence of CD34(+) cells which suppress immune functions within cancers that secrete granulocyte-macrophage colony-stimulating factor. Clin Cancer Res 1: 95–103, 1995 [PubMed] [Google Scholar]
- 25.Daud AI, Mirza N, Lenox B, Andrews S, Urbas P, Gao GX, Lee JH, Sondak VK, Riker AI, Deconti RC, Gabrilovich D: Phenotypic and functional analysis of dendritic cells and clinical outcome in patients with high-risk melanoma treated with adjuvant granulocyte macrophage colony-stimulating factor. J Clin Oncol 26: 3235–3241, 2008 [DOI] [PubMed] [Google Scholar]
- 26.Hoechst B, Ormandy LA, Ballmaier M, Lehner F, Krüger C, Manns MP, Greten TF, Korangy F: A new population of myeloid-derived suppressor cells in hepatocellular carcinoma patients induces CD4(+)CD25(+)Foxp3(+) T cells. Gastroenterology 135: 234–243, 2008 [DOI] [PubMed] [Google Scholar]
- 27.Zhang J, Wang B, Zhang W, Wei Y, Bian Z, Zhang CY, Li L, Zen K: Protein tyrosine phosphatase 1B deficiency ameliorates murine experimental colitis via the expansion of myeloid-derived suppressor cells. PLoS ONE 8: e70828, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Xia S, Sha H, Yang L, Ji Y, Ostrand-Rosenberg S, Qi L: Gr-1+ CD11b+ myeloid-derived suppressor cells suppress inflammation and promote insulin sensitivity in obesity. J Biol Chem 286: 23591–23599, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cripps JG, Gorham JD: MDSC in autoimmunity. Int Immunopharmacol 11: 789–793, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen S, Akbar SM, Abe M, Hiasa Y, Onji M: Immunosuppressive functions of hepatic myeloid-derived suppressor cells of normal mice and in a murine model of chronic hepatitis B virus. Clin Exp Immunol 166: 134–142, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Danowski TS, Weigand FA, Greenman L, Gailani S, Greenberg WV, Mateer FM: Corticotropin (ACTH) therapy of nephrotic syndrome in children. II. Laboratory findings in one hundred six instances. AMA J Dis Child 93: 604–614, 1957 [DOI] [PubMed] [Google Scholar]
- 32.Wang Y, Wang YP, Tay YC, Harris DC: Progressive doxorubicin nephropathy in mice: Sequence of histologic and immunohistochemical events. Kidney Int 58: 1797–1804, 2000 [DOI] [PubMed] [Google Scholar]
- 33.Gabrilovich DI, Nagaraj S: Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 9: 162–174, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Youn JI, Nagaraj S, Collazo M, Gabrilovich DI: Subsets of myeloid-derived suppressor cells in tumor-bearing mice. J Immunol 181: 5791–5802, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li L, Zhang J, Diao W, Wang D, Wei Y, Zhang CY, Zen K: MicroRNA-155 and MicroRNA-21 promote the expansion of functional myeloid-derived suppressor cells. J Immunol 192: 1034–1043, 2014 [DOI] [PubMed] [Google Scholar]
- 36.Qu P, Yan C, Blum JS, Kapur R, Du H: Myeloid-specific expression of human lysosomal acid lipase corrects malformation and malfunction of myeloid-derived suppressor cells in lal-/- mice. J Immunol 187: 3854–3866, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bricio T, Molina A, Egido J, Gonzalez E, Mampaso F: IL-1-like production in adriamycin-induced nephrotic syndrome in the rat. Clin Exp Immunol 87: 117–121, 1992 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ginevri F, Trivelli A, Mutti A, Bergamaschi E, Fabbretti G, Callea F, Salvidio G, Altieri P, Perfumo F, Ghiggeri GM: Progression of chronic adriamycin nephropathy in leukopenic rats. Nephron 63: 79–88, 1993 [DOI] [PubMed] [Google Scholar]
- 39.Chen A, Sheu LF, Ho YS, Lin YF, Chou WY, Chou TC, Lee WH: Experimental focal segmental glomerulosclerosis in mice. Nephron 78: 440–452, 1998 [DOI] [PubMed] [Google Scholar]
- 40.Rangan GK, Wang Y, Tay YC, Harris DC: Inhibition of nuclear factor-kappaB activation reduces cortical tubulointerstitial injury in proteinuric rats. Kidney Int 56: 118–134, 1999 [DOI] [PubMed] [Google Scholar]
- 41.Liu S, Jia Z, Zhou L, Liu Y, Ling H, Zhou SF, Zhang A, Du Y, Guan G, Yang T: Nitro-oleic acid protects against adriamycin-induced nephropathy in mice. Am J Physiol Renal Physiol 305: F1533–F1541, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang Y, Wang Y, Feng X, Bao S, Yi S, Kairaitis L, Tay YC, Rangan GK, Harris DC: Depletion of CD4(+) T cells aggravates glomerular and interstitial injury in murine adriamycin nephropathy. Kidney Int 59: 975–984, 2001 [DOI] [PubMed] [Google Scholar]
- 43.Amore A, Mazzucco G, Cavallo F, Forni G, Gianoglio B, Motta M, Peruzzi L, Novelli F, Porcellini MG, Cesano G, Coppo R: Adriamycin-induced proteinuria in nude mice: an immune-system-mediated toxic effect. Nephrol Dial Transplant 11: 1012–1018, 1996 [PubMed] [Google Scholar]
- 44.Barnes PJ, Adcock IM: Glucocorticoid resistance in inflammatory diseases. Lancet 373: 1905–1917, 2009 [DOI] [PubMed] [Google Scholar]
- 45.Suda T, Chida K, Matsuda H, Hashizume H, Ide K, Yokomura K, Suzuki K, Kuwata H, Miwa S, Nakano H, Fujisawa T, Enomoto N, Matsushita A, Nakamura H: High-dose intravenous glucocorticoid therapy abrogates circulating dendritic cells. J Allergy Clin Immunol 112: 1237–1239, 2003 [DOI] [PubMed] [Google Scholar]
- 46.Lee RW, Creed TJ, Schewitz LP, Newcomb PV, Nicholson LB, Dick AD, Dayan CM: CD4+CD25(int) T cells in inflammatory diseases refractory to treatment with glucocorticoids. J Immunol 179: 7941–7948, 2007 [DOI] [PubMed] [Google Scholar]
- 47.Rhen T, Cidlowski JA: Antiinflammatory action of glucocorticoids—new mechanisms for old drugs. N Engl J Med 353: 1711–1723, 2005 [DOI] [PubMed] [Google Scholar]
- 48.Varga G, Ehrchen J, Tsianakas A, Tenbrock K, Rattenholl A, Seeliger S, Mack M, Roth J, Sunderkoetter C: Glucocorticoids induce an activated, anti-inflammatory monocyte subset in mice that resembles myeloid-derived suppressor cells. J Leukoc Biol 84: 644–650, 2008 [DOI] [PubMed] [Google Scholar]
- 49.Marigo I, Bosio E, Solito S, Mesa C, Fernandez A, Dolcetti L, Ugel S, Sonda N, Bicciato S, Falisi E, Calabrese F, Basso G, Zanovello P, Cozzi E, Mandruzzato S, Bronte V: Tumor-induced tolerance and immune suppression depend on the C/EBPbeta transcription factor. Immunity 32: 790–802, 2010 [DOI] [PubMed] [Google Scholar]
- 50.Ai S, Zheng J, Lin Q, Chen R: Proteomic analysis indicates altered expression of plasma proteins in a rat nephropathy model. Clin Exp Nephrol 17: 24–31, 2013 [DOI] [PubMed] [Google Scholar]
- 51.Xing Y, Ding J, Fan Q, Guan N: Diversities of podocyte molecular changes induced by different antiproteinuria drugs. Exp Biol Med (Maywood) 231: 585–593, 2006 [DOI] [PubMed] [Google Scholar]
- 52.Zheng J, Gong J, Zhang A, Li S, Zeng Z, Han Y, Gan W: Attenuation of glomerular filtration barrier damage in adriamycin-induced nephropathic rats with bufalin: An antiproteinuric agent. J Steroid Biochem Mol Biol 129: 107–114, 2012 [DOI] [PubMed] [Google Scholar]
- 53.Masutani K, Taniguchi M, Nakashima H, Yotsueda H, Kudoh Y, Tsuruya K, Tokumoto M, Fukuda K, Kanai H, Hirakata H, Iida M: Up-regulated interleukin-4 production by peripheral T-helper cells in idiopathic membranous nephropathy. Nephrol Dial Transplant 19: 580–586, 2004 [DOI] [PubMed] [Google Scholar]
- 54.Yano N, Endoh M, Nomoto Y, Sakai H, Fadden K, Rifai A: Phenotypic characterization of cytokine expression in patients with IgA nephropathy. J Clin Immunol 17: 396–403, 1997 [DOI] [PubMed] [Google Scholar]
- 55.Futrakul N, Siriviriyakul P, Panichakul T, Butthep P, Patumraj S, Futrakul P: Glomerular endothelial cytotoxicity and dysfunction in nephrosis with focal segmental glomerulosclerosis. Clin Hemorheol Microcirc 29: 469–473, 2003 [PubMed] [Google Scholar]
- 56.Gentle ME, Shi S, Daehn I, Zhang T, Qi H, Yu L, D’Agati VD, Schlondorff DO, Bottinger EP: Epithelial cell TGFβ signaling induces acute tubular injury and interstitial inflammation. J Am Soc Nephrol 24: 787–799, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.He W, Kang YS, Dai C, Liu Y: Blockade of Wnt/β-catenin signaling by paricalcitol ameliorates proteinuria and kidney injury. J Am Soc Nephrol 22: 90–103, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cao Q, Wang Y, Zheng D, Sun Y, Wang Y, Lee VW, Zheng G, Tan TK, Ince J, Alexander SI, Harris DC: IL-10/TGF-beta-modified macrophages induce regulatory T cells and protect against adriamycin nephrosis. J Am Soc Nephrol 21: 933–942, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]