Abstract
Background
The absence of a discrete mass, surrounding signal abnormality and solid enhancement are imaging features that have traditionally been used to differentiate soft tissue arteriovenous malformations from vascular tumors on MRI. We have observed that these findings are not uncommon in arteriovenous malformations, which may lead to misdiagnosis or inappropriate treatment.
Objective
To estimate the frequency of atypical MRI features in soft-tissue arteriovenous malformations and assess their relationship to lesion size, location, tissue type involved and vascular architecture.
Materials and methods
Medical records, MRI and histopathology were reviewed in consecutive patients with soft-tissue arteriovenous malformations in a multidisciplinary vascular anomalies clinic. Arteriovenous malformations were divided into those with and without “atypical” MRI findings (perilesional T2 signal abnormality, enhancement and/or a soft-tissue mass). Lesion location, size, tissue involved and vascular architecture were also compared between groups. Tissue stains were reviewed in available biopsy or resection specimens to assess relationships between MRI findings and histopathology.
Results
Thirty patients with treatment-naïve arteriovenous malformations were included. Fifteen lesions demonstrated atypical MRI. There was no difference in age, gender, lesion size or involved body part between the groups. However, more than half of the atypical lesions demonstrated multicompartmental involvement, and tiny intralesional flow voids were more common in atypical arteriovenous malformations. Histopathology also differed in atypical cases, showing densely packed endothelial cells with connective tissue architectural distortion and edema.
Conclusion
Arteriovenous malformations may exhibit features of a vascular tumor on MRI, particularly when multicompartmental and/or containing tiny internal vessels. These features are important to consider in suspected fast-flow vascular malformations and may have implications with respect to their treatment.
Keywords: Adults, Arteriovenous malformation, AVM, Children, Magnetic resonance imaging, Vascular anomaly
Introduction
Soft-tissue arteriovenous malformations are high-flow developmental vascular anomalies characterized by direct fistulous or plexiform connections between arteries and veins without normal intervening arterioles or capillaries [1–3]. They are more frequently isolated but occasionally syndromic, as seen in Parkes-Weber, Osler-Weber-Rendu and capillary malformation-arteriovenous malformation syndromes and RASA1 mutations. Lesions may come to clinical attention due to cosmetic deformity, pain, bleeding, thromboembolism and/or high-output heart failure. Although physical examination often reveals suggestive findings of pulsatility with overlying skin warmth and/or redness in soft-tissue arteriovenous malformations, MR imaging remains an important tool for their accurate diagnosis and treatment.
Like their counterparts in the central nervous system, the most conspicuous MRI abnormalities in soft-tissue arteriovenous malformations are tubular flow voids representing high flow within enlarged vascular channels. This observation, together with the absence of any surrounding soft-tissue signal abnormality, discrete soft-tissue mass or enhancement, permits accurate diagnosis given appropriate clinical history [2–5]. Importantly, these MR features of arteriovenous malformations have also been adopted by the International Society for the Study of Vascular Anomalies (ISSVA) classification, which states that arteriovenous malformations are characterized by “flow voids without parenchymal staining” [4]. However, a subset of arteriovenous malformations exhibits perilesional signal abnormalities, soft-tissue enhancement and/or mass-like features, atypical findings that make these lesions more difficult to differentiate from benign or malignant soft-tissue tumors, such as congenital hemangioma and angiosarcoma. These three MRI features, although felt to be uncommon in soft-tissue arteriovenous malformations, have been attributed to edema or a fibrofatty matrix within the tissues surrounding the arteriovenous malformation [6].
The goal of this study was to evaluate the frequency of these “atypical” MRI features of soft-tissue arteriovenous malformations in a cohort of patients undergoing evaluation at our vascular anomalies clinic, and to determine whether these atypical imaging features were associated with patient demographics, lesion location, lesion size or intralesional artery diameter. We also aimed to understand how these MRI findings were related to underlying histopathological differences in lesions that underwent resection or biopsy.
Materials and methods
The Committee on Human Research provided institutional review board approval for this retrospective study. Patients diagnosed with soft-tissue arteriovenous malformations by the consensus of our multidisciplinary birthmark and vascular anomalies clinic (consisting of dermatologists, radiologists, surgeons and hematologists), were identified through a clinical database. Patients evaluated between 1995 and 2011 were included if they a) had no prior history of prior surgical or endovascular treatment, b) had chronic lesions with >1 year of symptoms, and c) had undergone diagnostic MRI including T1-, T2- and fat-suppressed, gadolinium-enhanced T1-weighted sequences in at least two planes within 6 months of their clinical evaluation. Patients with a history of trauma or a non-diagnostic or incomplete MRI were excluded.
Age and gender for each patient were extracted from the medical record. Two radiologists, one a fellow in interventional radiology (A.S.P.) and the other an attending (C.P.H.) with subspecialty certification in neuroradiology and expertise in vascular anomalies (10 years of experience), reviewed MRI studies for each subject, noting the presence or absence of perilesional T2 hyperintensity, perilesional enhancement, and any discrete soft tissue mass. Based on the presence or absence of these features, lesions were divided into those that were “typical” (lacking any of these three features) and those that were “atypical” (when one or more of these features was present). The following findings were also noted for each lesion:
Body part involved (head and/or neck, truncal or extremity)
Involved tissue type (subcutaneous, intramuscular or multicompartmental location of arteriovenous malformations)
Approximate size (greatest cross-sectional area in cm2)
Vascular architecture (small, indistinct flow voids less than 2 mm in diameter or distinct flow voids larger than 2 mm in diameter)
In order to correlate observed MRI findings with tissue histopathology, a dermatopathology fellow (J.M.S.) and a board-certified dermatopathologist (B.S.R with 20 years of experience) reviewed available histopathology slides in all included patients who underwent biopsy or excision after evaluation. Tissue architecture and arteriovenous malformation characteristics on hematoxylin and eosin (H&E) stains were documented.
Demographic and imaging features of typical and atypical lesions were compared using chi-square tests for equality of proportions and Student’s t-tests.
Results
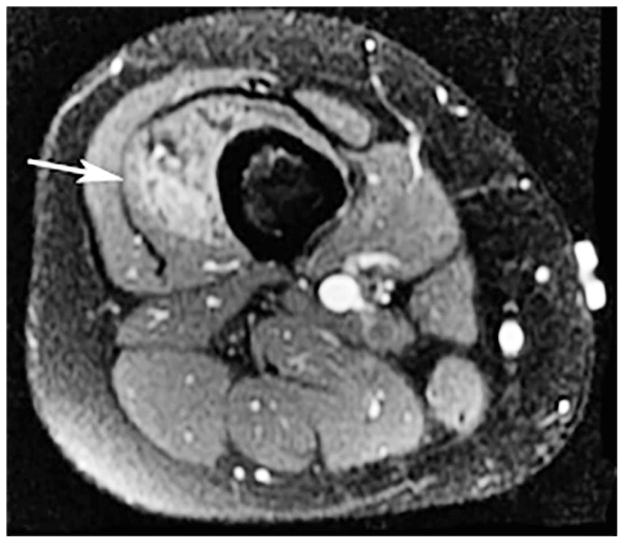
Thirty patients, each with a single soft-tissue arteriovenous malformation, were included in the study. Fifteen lesions were classified as typical and 15 lesions were classified as atypical based on MRI review. Representative examples of the typical lesions are illustrated in Figures 1–2. Representative examples of the atypical lesions are illustrated in Figures 3–5. Finally, a single patient demonstrated both features of a typical and atypical lesions (Fig 6).
Fig. 1.

A 6-year-old boy with history of painful right forearm arteriovenous malformations. Axial fat-suppressed gadolinium-enhanced T1-weighted image demonstrates a typical arteriovenous malformation (arrows) lesion with prominent flow voids and no enhancement or soft-tissue abnormalities
Fig. 2.
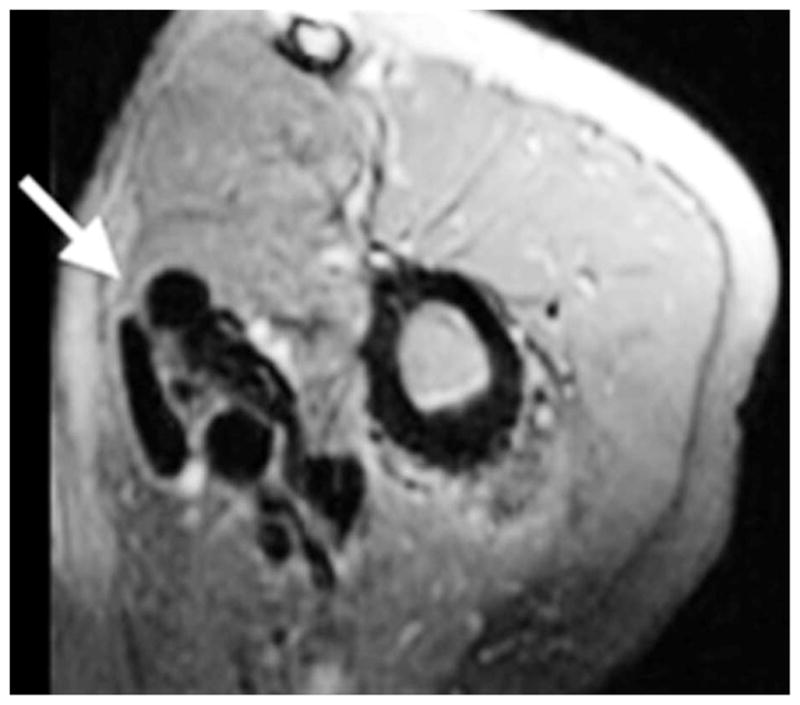
A 27-year-old man with a history of painful left upper arm arteriovenous malformation. Axial fat-suppressed gadolinium-enhanced T1-weighted image demonstrates a typical arteriovenous malformation (arrow) lesion with prominent flow voids and no enhancement or soft-tissue abnormalities.
Fig. 3.

A 2-year-old girl with a history of painful pulsatile upper lip arteriovenous malformation. Axial fat-suppressed gadolinium-enhanced T1-weighted image demonstrates an atypical arteriovenous malformation (arrows) lesion with a discrete soft-tissue mass, enhancement and tiny intralesional flow voids
Fig. 5.
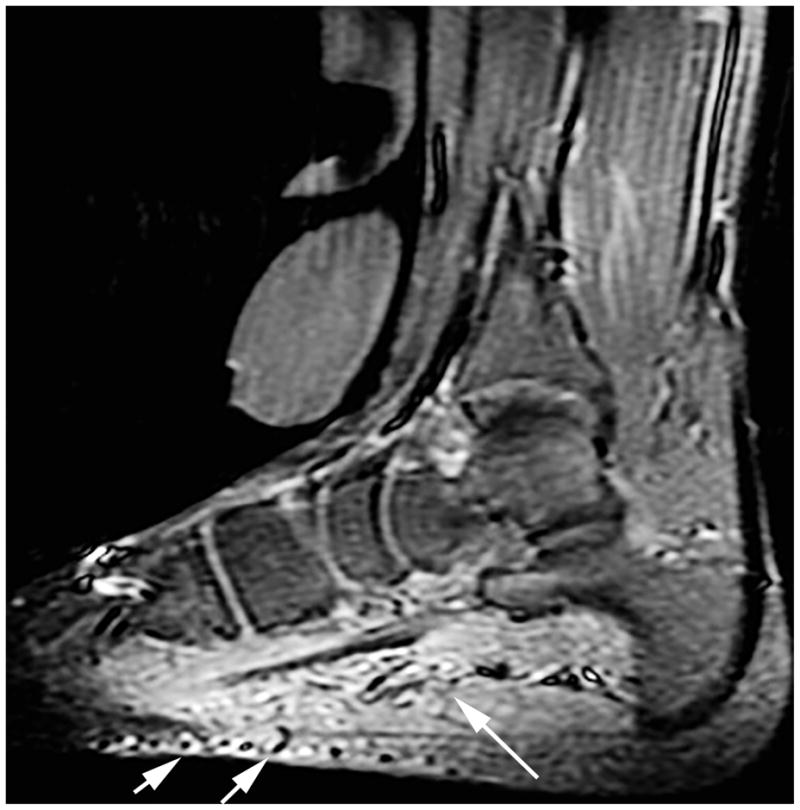
An 11-year-old girl with a history of arteriovenous malformations within the plantar foot. Sagittal STIR images demonstrate an atypical arteriovenous malformation (arrows) with tiny indistinct flow voids, soft-tissue hyperintensity and mass-like characteristics of the lesion
Fig. 6.

A 23-year-old woman with a history of painful arteriovenous malformations within the right thigh. T2 fat-suppressed images demonstrate features of a typical and atypical arteriovenous malformation in the same patient. a. In the distal thigh, at the level of the nidus, there are tiny indistinct flow voids, soft-tissue hyperintensity and mass-like characteristics of the lesion. b. In the proximal thigh, there is a distinct large feeding vessel with flow void, and lack of associated soft-tissue mass
Demographic data and imaging characteristics are summarized for the typical and atypical groups in Table 1. There was no significant difference in patient age or gender, lesion size or involved body part on MRI, between the two groups. However, both the tissue types involved and the intralesional vascular architecture differed between atypical and typical arteriovenous malformations, with multicompartmental lesions and lesions with smaller flow voids more common in the atypical group. Qualitatively, intralesional flow voids in atypical lesions were often small and filiform in morphology (<1 mm) (Fig. 1). All 15 atypical lesions showed T2 hyperintensity and enhancement, and 13/15 (86.6%) were associated with a discrete soft-tissue mass that was distinct from the abnormal vessels (usually within adjacent muscles). Enhancement patterns and T2 hyperintensity were mixed; however, enhancement was evident outside of the vascular structures within muscular and other soft tissues surrounding the nidus. Most atypical lesions were isointense or slightly hypointense to normal muscle on T1-weighted images. None demonstrated intrinsic T1 shortening to suggest the presence of fat or thrombus.
Table 1.
Clinical and imaging features of soft tissue arteriovenous malformations (AVMs)
| Atypical AVMs (n=15) | Typical AVMs (n=15) | P-value | |
|---|---|---|---|
|
|
|||
| Demographic data | |||
| Age | 19.6 +/− 15.1 years | 21.1 +/− 11.9 years | 0.76 |
| Range | 0.4–51 years | 4–50 years | |
| Median | 20 years | 20 years | |
| Interquartile Range | 28 years | 13 years | |
| Gender (F:M) | 10:5 | 9:6 | 0.70 |
|
| |||
| Arteriovenous malformation location | |||
| Head / neck | 7 | 9 | 0.48 |
| Trunk | 2 | 3 | |
| Extremities | 6 | 3 | |
|
| |||
| Tissue type(s) involved | |||
| Subcutaneous | 3 | 9 | 0.01* |
| Intramuscular | 4 | 5 | |
| Multicompartmental | 8 | 1 | |
|
| |||
| Cross-sectional area | 18.7 +/− 13.8 cm | 24.8 +/− 22.4 cm | 0.37 |
|
| |||
| Luminal diameter <2 mm | 11 | 14 | <0.01* |
Statistically significant (P<0.05)
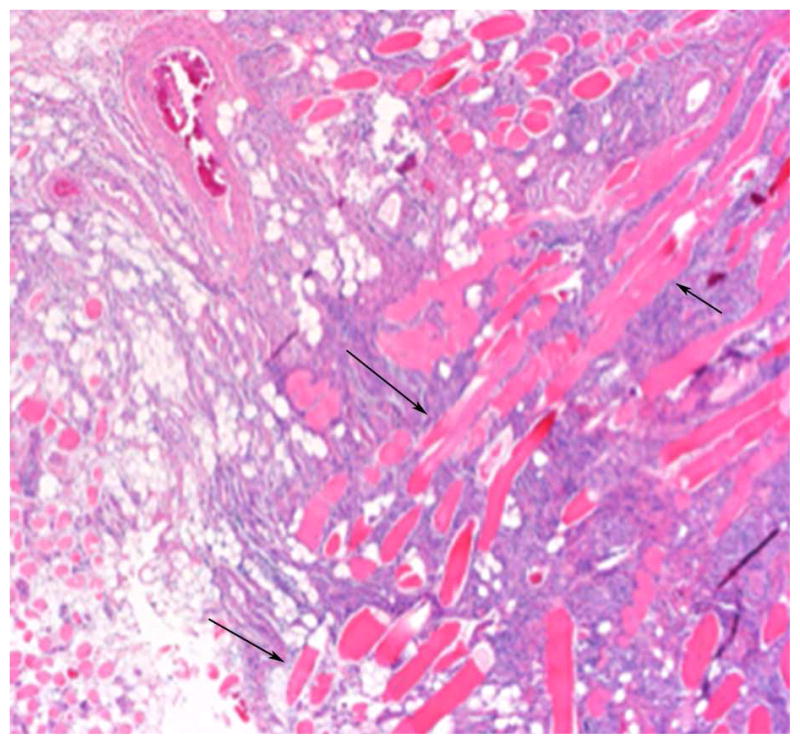
Twelve of 30 patients had pathological specimens available for review. Eight of these cases had atypical MRI findings. Concordant with the MRI findings, H&E stains in these cases demonstrated two distinctive histopathological patterns. In the first pattern, broad sheets of densely packed, plump endothelial cells infiltrate between connective tissue elements, splaying apart collagen bundles, muscle fibers and adipocytes. These endothelial cells form vascular spaces with often inconspicuous or slit-like lumens, only some of which appear to contain erythrocytes (Fig. 7). In the second pattern, small jagged vessels are set within connective tissue fibers that are widely separated by interstitial edema. Both of these patterns are unified by small or poorly formed vessels with architectural distortion of native connective tissue elements. By contrast, cases with typical MRI findings revealed proliferations of well-formed, variably sized vessels without distortion of the surrounding connective tissue (Fig. 8).
Fig. 7.
A 27-year-old woman with chronic painful left forehead arteriovenous malformations status postsurgical excision. Hematoxylin and eosin stains of soft-tissue “atypical” arteriovenous malformation specimens. a Diffuse proliferation of small, crowded blood vessels permeates the connective tissue (40x). b Skeletal muscle fibers (arrows) are widely separated by small vessels and vascular lumina are not conspicuous (100x)
Fig. 8.
A 30-year-old woman with chronic painful left temporal arteriovenous malformations status postsurgical excision. Hematoxylin and eosin stains of soft-tissue “typical” arteriovenous malformation specimens. a Variably sized vessels (arrows) throughout the subcutis (40x). b Individual, well-formed vessels (arrows) lie between skeletal muscle fibers, without disruption of the overall muscular architecture (100x)
Discussion
The conventionally described MRI findings in soft-tissue arteriovenous malformations are vascular flow voids without associated T2 hyperintensity, enhancement or mass effect on surrounding tissues. In this series of 30 patients with treatment-naïve arteriovenous malformations, we found that 50% of lesions exhibited one or more of these features. These abnormalities frequently occurred together and were more common in lesions that spanned multiple tissue compartments and/or exhibited tiny intralesional arteries. Awareness of these MRI characteristics may help radiologists recognize these lesions as consistent with arteriovenous malformation and avoid pitfalls or misdiagnosis of other benign or malignant vascular masses.
Based on a review of the available tissue specimens in our patients, we also found that biopsies in lesions with these atypical findings exhibited two distinct histopathological phenotypes, both of which differed significantly from arteriovenous malformations without these features. In contrast to typical arteriovenous malformations, which showed well-formed vessels without significant surrounding tissue architectural distortion, atypical arteriovenous malformations showed either an infiltrative pattern of densely packed vessels or small vessels with abnormal morphology within a milieu of interstitial edema.
Multicompartmental arteriovenous malformations more commonly exhibited atypical, tumor-like abnormalities on MRI. All seven of the multicompartmental lesions in our series demonstrated subcutaneous and intramuscular involvement, and one of these lesions additionally demonstrated pleural involvement. An additional common observation within these lesions was tiny flow voids with a permeative appearance, suggesting that these lesions may exhibit a unique angioarchitecture. This was confirmed on histopathological review of the atypical cases, which revealed architectural distortion and edema within the connective tissues surrounding the arteriovenous malformation and, in some cases, abnormal vascular morphology.
The histopathological features seen in our cases with atypical MRI findings are similar to those recently described in intramuscular capillary-type hemangiomas [7], which also demonstrate aggregates and sheets of small vessels lined by plump endothelial cells that splay apart individual muscle fibers. However, not all of our cases were primarily intramuscular, and in some cases the distortion of connective tissue elements seen on histological analysis was due to interstitial edema, rather than to vascular infiltration. Thus, the cases with atypical MRI findings described herein may represent a broad radiographic category, with intramuscular capillary-type hemangiomas representing one lesion typical of this category.
Congenital arteriovenous malformations usually progress slowly, but a certain subset may become symptomatic because of episodes of sudden growth and/or inflammation. Recently, a series by Meijer-Jorna et al. [8] suggested that symptomatic surgical lesions, most of which were arteriovenous malformations, demonstrate endothelial microproliferation with immature histology. A subsequent study by the same group [9] showed microvascular proliferation and immaturity of cells uniquely noted in arteriovenous malformations. Expression of VEGF-A, angiopoietin-1, Ki-67, p16 and p21/27 were overall significantly increased in immature areas within arteriovenous malformations. Although not studied in our analysis, the lesions described in our series may represent an imaging correlate to the arteriovenous malformations described by Meijer-Jorna et al. [8], which could be investigated in the future. Moreover, with a larger sample size, correlation with clinical symptoms and recurrence could also be analyzed.
A newly described clinical phenotype of brain arteriovenous malformation [10], termed “cerebral proliferative angiopathy” merits discussion in that this lesion may also may be regarded as distinct from “classic” brain arteriovenous malformation in angioarchitecture and imaging presentation. These brain lesions show diffuse angiogenesis with multiple arterial feeders (without dominance) feeding a large nidus, with small draining veins. The lesions also enhance with administration of gadolinium chelate, consistent with microvascular leakage into the surrounding interstitium related to the histopathological changes. The tiny flow voids observed in the atypical lesions in our study exhibited a unique angioarchitecture and histopathological profile, perhaps similar to cerebral proliferative angiopathies in the central nervous system. Further histopathological and angiogenic markers could be screened for in the future to determine if the MRI features we described correlate with these markers.
Our study has a number of limitations. First, our sample size is low thereby preventing significant correlations between lesion features. Second, our study design was retrospective as it was inspired by several years of empiric observation by the authors. Third, histological data were available for less than half of the lesions. The atypical arteriovenous malformations described here serve to expand the differential for the radiologist. Some of the features we describe in these arteriovenous malformations could have been misdiagnosed as other lesions. While the imaging features we have identified alone may not ultimately obviate surgical excision or biopsy, they should make the radiologist aware of alternative possibilities other than neoplasm and potentially obviate biopsy when the clinical presentation is sufficiently diagnostic. Future studies are needed to further characterize the imaging and clinical features of these lesions. Moreover, the atypical lesions could portend a different clinical outcome and presentation compared to typical arteriovenous malformations. Again, with our small sample size and limited data set, these features relating to clinical presentation and treatment could not be assessed to any significance.
Conclusion
Soft-tissue arteriovenous malformations may exhibit features of a vascular tumor on MRI, particularly when multicompartmental and/or containing small (<2 mm) internal vessels. Atypical features of perilesional T2 hyperintensity, enhancement and mass effect may be related to microproliferation of immature cells and/or the presence of interstitial edema. These features are important to consider when reviewing MRI in the patient with suspected fast-flow vascular malformation.
Fig. 4.

A 30-year-old woman with a history of painful right posterior neck arteriovenous malformations. Axial fat-suppressed gadolinium-enhanced T1-weighted image demonstrates an atypical arteriovenous malformation (arrow) lesion with a discrete soft-tissue mass, enhancement and tiny intralesional flow voids
Footnotes
Conflicts of interest Dr. Frieden is a consultant for Pierre Fabre. Drs. Patel, Schulman, Ruben, Hoffman, Dowd and Hess have no disclosures.
Contributor Information
Anand S. Patel, Email: anand.patel@ucsf.edu, Department of Radiology and Biomedical Imaging, University of California, San Francisco, 505 Parnassus Ave., M-361, San Francisco, CA 94143, USA. Department of Radiology, University of California, San Francisco, Birthmarks and Vascular Anomalies Clinic, San Francisco, CA
Joshua M. Schulman, Department of Pathology, University of California, San Francisco, Birthmarks and Vascular Anomalies Clinic, San Francisco, CA
Beth S. Ruben, Department of Pathology, University of California, San Francisco, Birthmarks and Vascular Anomalies Clinic, San Francisco, CA
William Y. Hoffman, Department of Plastic Surgery, University of California, San Francisco, Birthmarks and Vascular Anomalies Clinic, San Francisco, CA
Christopher F. Dowd, Department of Radiology, University of California, San Francisco, Birthmarks and Vascular Anomalies Clinic, San Francisco, CA
Ilona J. Frieden, Department of Dermatology, University of California, San Francisco, Birthmarks and Vascular Anomalies Clinic, San Francisco, CA
Christopher P. Hess, Department of Radiology, University of California, San Francisco, Birthmarks and Vascular Anomalies Clinic, San Francisco, CA
References
- 1.Donnelly LF, Adams DM, Bisset GS., 3rd Vascular malformations and hemangiomas: a practical approach in a multidisciplinary clinic. AJR Am J Roentgenol. 2000;174:597–608. doi: 10.2214/ajr.174.3.1740597. [DOI] [PubMed] [Google Scholar]
- 2.Dubois J, Alison M. Vascular anomalies: what a radiologist needs to know. Pediatr Radiol. 2010;40:895–905. doi: 10.1007/s00247-010-1621-y. [DOI] [PubMed] [Google Scholar]
- 3.Konez O, Burrows PE. Magnetic resonance of vascular anomalies. Magn Reson Imaging Clin North Am. 2002;10:363–388. vii. doi: 10.1016/s1064-9689(01)00009-5. [DOI] [PubMed] [Google Scholar]
- 4.Enjolras O, Wassef M, Chapot R. Color atlas of vascular tumors and vascular malformations. Cambridge University Press; Cambridge, UK: 2007. [Google Scholar]
- 5.Hovius SE, Borg DH, Paans PR, Pieterman H. The diagnostic value of magnetic resonance imaging in combination with angiography in patients with vascular malformations: a prospective study. Ann Plast Surg. 1996;37:278–285. doi: 10.1097/00000637-199609000-00008. [DOI] [PubMed] [Google Scholar]
- 6.Robertson RL, Robson CD, Barnes PD, Burrows PE. Head and neck vascular anomalies of childhood. Neuroimaging Clin North Am. 1999;9:115–132. [PubMed] [Google Scholar]
- 7.Yilmaz S, Kozakewich HP, Alomari AI, et al. Intramuscular capillary-type hemangioma: radiologic-pathologic correlation. Pediatr Radiol. 2014;44:558–5658. doi: 10.1007/s00247-014-2876-5. [DOI] [PubMed] [Google Scholar]
- 8.Meijer-Jorna LB, van der Loos CM, de Boer OJ, et al. Microvascular proliferation in congenital vascular malformations of skin and soft tissue. J Clin Pathol. 2007;60:798–803. doi: 10.1136/jcp.2006.038885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Meijer-Jorna LB, van der Loos CM, Teeling P, et al. Proliferation and maturation of microvessels in arteriovenous malformations--expression patterns of angiogenic and cell cycle-dependent factors. J Cutan Pathol. 2012;39:610–620. doi: 10.1111/j.1600-0560.2012.01913.x. [DOI] [PubMed] [Google Scholar]
- 10.Saliou G, Krings T, Rutgers DR, et al. PWI-MRI and contrast extravasation in brain AVM help to estimate angiogenic activity. Neuroradiology. 2011;53:793–800. doi: 10.1007/s00234-011-0882-y. [DOI] [PubMed] [Google Scholar]