Abstract
Objectives
To examine mortality patterns and dose-response relations between ionising radiation and mortality outcomes of a priori interest in 6409 uranium workers employed for at least 30 days (1951–1985), and followed through 2004.
Methods
Cohort mortality was evaluated through standardised mortality ratios (SMR). Linear excess relative risk (ERR) regression models examined associations between cause-specific mortality and exposures to internal ionising radiation from uranium deposition, external gamma and x-ray radiation, and radon decay products, while adjusting for non-radiologic covariates.
Results
Person-years at risk totalled 236 568 (mean follow-up 37 years), and 43% of the cohort had died. All-cause mortality was below expectation only in salaried workers. Cancer mortality was significantly elevated in hourly males, primarily from excess lung cancer (SMR=1.25, 95% CI 1.09 to 1.42). Cancer mortality in salaried males was near expectation, but lymphohaematopoietic malignancies were significantly elevated (SMR=1.52, 95% CI 1.06 to 2.12). A positive dose-response relation was observed for intestinal cancer, with a significant elevation in the highest internal organ dose category and a significant dose-response with organ dose from internal uranium deposition (ERR=1.5 per 100 µGy, 95% CI 0.12 to 4.1).
Conclusions
A healthy worker effect was observed only in salaried workers. Hourly workers had excess cancer mortality compared with the US population, although there was little evidence of a dose-response trend for any cancer evaluated except intestinal cancer. The association between non-malignant respiratory disease and radiation dose observed in previous studies was not apparent, possibly due to improved exposure assessment, different outcome groupings, and extended follow-up.
INTRODUCTION
The Fernald Feed Materials Production Center (FMPC) operated as a United States Department of Energy (US DOE) uranium processing facility from 1951 through 1989. The facility’s primary mission was to produce high-purity uranium metal necessary for nuclear weapons production and other defence missions. Operations involving storage and handling of radioactive materials began in 1951. By 1954, all main production plants, comprising a wide array of uranium fuel cycle chemical and metallurgical processes were fully operational. In addition to ionising radiation hazards typically associated with uranium production, subgroups of FMPC employees were potentially exposed to other sources, such as radon, thorium and low-level transuranic contamination. Non-radiologic hazards, such as acid gases (acids), solvents and dusts, were also present. The facility was operated by National Lead of Ohio (NLO) from 1951 to 1985. Production then slowed, and ceased in 1989.
Canu et al1, in an extensive literature review, found limited evidence of increased mortality from respiratory, lymphatic and haematopoietic cancers in workers occupationally exposed to uranium at a variety of facilities, including FMPC, and cited inadequate assessment of internal dose from uranium as a limitation of these studies. Several studies have examined health outcomes specifically among Caucasian male FMPC workers.2–5 These studies suggest increased risk of chronic non-malignant respiratory disease (NMRD) in relation to internal, and possibly external, radiation dose, and present equivocal results with respect to the relations between radiation dose and mortality from cancers of the lung, kidney, bladder and digestive tract.
The current study expanded the FMPC cohort to include all workers, added 15 years of vital status follow-up, and recalculated internal organ dose. Malignant and non-malignant respiratory and renal disease, cancers of the bladder, stomach and intestine (small intestine and colon, but not rectum), and lymphohaematopoietic cancers were of a priori interest based on the findings of previous studies of uranium production plant workers.1–6 Idiopathic pulmonary fibrosis (IPF) was examined based on previous findings of elevated risks associated with exposures to densely ionising radiation and metal dusts.7–10 Associations between these outcomes and internal ionising radiation from uranium deposition, gamma and x-ray radiation from external sources, and radon progeny were evaluated while adjusting for non-radiologic covariates.
METHODS
Cohort development
This research complied with the requirements of the Federal Policy for Protection of Human Subjects (10CFR745 or, where applicable, 45CFR46), and was reviewed by the National Institute for Occupational Safety and Health (NIOSH) Human Subjects Review Board to ensure that the rights and welfare of study subjects were protected.
The study population consisted of NLO workers employed 30 days or more at the FMPC from 1951 to 1985. The minimum employment criterion was used to prevent inclusion of occasional visitors and prospective hires who never actually worked at the site. The roster was developed from three sources of records, prioritised as follows: (1) the NLO Employee Database; (2) data from the original cohort assembled by Cragle et al3 and expanded by NIOSH using FMPC employment records and (3) the site’s Health Physics Information System (HIS-20). Information on site, medical and security databases was used to resolve inconsistencies among the primary sources. Work histories included employment dates, job title, organisation and building assignments.
Ascertainment of vital status
Vital status was previously determined by Cragle et al3 through 1989, using searches conducted by the Social Security Administration (SSA), Pension Benefits, Inc, and the National Death Index (NDI). Death certificates for that study were retrieved and coded to the eighth revision of the International Classification of Diseases (ICD), Adapted for Use in the United States.
For the current study, mortality was updated through 2004 by searches of the SSA’s death master file and NDI; cause of death coding was to the ICD revision in effect at time of death. Underlying cause of death was used for all analyses.
Exposure assessment
The exposure assessment is described in detail in Anderson et al11 and briefly here. The exposure assessors were blinded to case status.
Radiation exposures
Ionising radiation exposure from uranium and its by-products was of primary interest; the critical source term and exposure pathway varied by outcome. Multiple radiation sources were examined; because of known differences in biological effectiveness between radiation types, heterogeneous sources of ionising radiation exposure were not combined into a single exposure metric.
The current effort improved on the internal dose assessments used by previous studies of this cohort2–5 by using urine uranium concentration data from 1952 forward, and the latest uranium biokinetic and dosimetric models in combination with the most recent respiratory tract model to estimate exposures from internally deposited uranium compounds.12,13 The Internal Dose Evaluation Program (InDEP, SENES Oak Ridge, Inc, Center for Risk Analysis, Oak Ridge, Tennessee, USA) was used to calculate individual intakes and corresponding organ absorbed doses (hereafter referred to as ‘organ dose’) from urine bioassay obtained from HIS-20. Five target organs were selected for internal dose assessment based on the outcomes of a priori interest: lung, pancreas, lower large intestine, kidney and red bone marrow. Due to the large number of individual dose assessments required, some outcomes were analysed using doses calculated for similar or nearby target tissue. All intakes were assumed due to chronic inhalation of a moderately insoluble natural uranium aerosol, with activity median aerodynamic diameter of 10 µm.
External radiation exposure was quantified primarily using penetrating whole-body ‘dose’ values reported in HIS-20. Most penetrating exposure resulted from photons of energies between 30 and 1000 keV and anterior-to-posterior, or isotropic exposures geometries. Under these conditions, the recorded values provided reasonable approximations of whole-body equivalent dose.14,15 It was further assumed that the equivalent dose approximated tissue-absorbed dose in this analysis, although body attenuation would result in a moderate reduction in absorbed dose to ‘deep’ tissues (eg, red bone marrow). Exposure data were available in annual increments prior to 1981 and monthly thereafter. Information on external dose accrued at other facilities was abstracted from HIS-20 and the DOE Radiation Exposure Monitoring System. When records from these two sources conflicted, HIS-20 data were prioritised.
Radon decay products (RDP) exposure was estimated based on an exposure matrix developed and described by Hornung et al16 that used employment information to spatially and temporally locate workers in a matrix of RDP concentrations estimated by dispersion modelling. The estimation process included a Gaussian plume model and opportunistic dosimetry methods using legacy glass to develop radon source terms and estimate transport. Briefly, working-level months (WLM) were assigned to each worker based on his or her proximity over time to radium-bearing source terms.
Thorium exposures were qualitatively assessed by examination of work assignments, site process records and available air and bioassay data. Dichotomous assignments indicating exposure potentials were made for each work history segment, based on whether the subject’s work assignment (dates, job and location) coincided with thorium activities as evidenced by process records and limited monitoring data.
Non-radiologic exposures
All division, department, plant/building and job title combinations were enumerated and related to qualitative exposure potentials. Site process records, available industrial hygiene monitoring records and institutional knowledge were used to evaluate exposure potentials (ever/never) for chemicals potentially related to outcomes of a priori interest: asbestos, coal dust, acids (hydrofluoric acid, nitric acid, nitrogen dioxide), machining fluids (also called cutting fluids), miscellaneous dusts, miscellaneous laboratory chemicals, trichloroethylene, other solvents, vehicle exhausts and welding fumes (see online supplementary table S1).17,18
Socioeconomic status
Pay code (salaried vs hourly) was of interest because of risk differences seen in earlier studies, and because it can be associated with lifestyle factors, like smoking, that can confound exposure-response relations.19–21 Pay code was noted on most work history records in the employee database. The earliest pay code encountered in each worker’s employment history was used; only 5.7% of workers changed pay status while employed at Fernald.
Statistical methods
Mortality was evaluated using the NIOSH Life Table Analysis System (LTAS.NET.22 Person-years at risk (PYAR) began on the latest of on 1 January 1951, the date cohort inclusion criteria (including 30 days of employment) were met, and the comparison rate file begin date. PYAR ended at the earlier of date of death and the study end date. A small number of workers lost to follow-up (n=30) were assumed to be alive at study end. PYAR were stratified by gender, race, age (in 5-year categories) and calendar year (in 5-year categories). US mortality rates (1940–2004) were used to estimate the expected numbers of deaths for all causes, all cancers and 92 cause-of-death categories.23 The standardised mortality ratio (SMR) was calculated as the ratio of the observed to the total number of expected deaths. Confidence limits were estimated based on a Poisson distribution for the observed deaths,24 with exact limits for outcomes with 10 or fewer deaths. Several outcomes for which rates were not available in the 1940–2004 file (IPF,25 asbestosis and silicosis) were evaluated using mortality rates from the US population (1960–2004). Since the FMPC was located in Ohio, mortality rates for Ohio (1960–2004) were also used. Directly standardised rate ratios (SRR) were used to ascertain differences by pay code.
Regression models were used to evaluate the relations between exposure and mortality from specific causes, adjusting for potential confounders. Only Caucasian males were included in these models because there were few workers (and deaths) from other demographic groups. Risk sets were defined using incidence density matching based on attained age.26 Within risk sets, exposure data were truncated at a cut-off date defined as the death date for cases and the date the worker reached the case’s death age for controls. Model parameters were estimated using conditional logistic regression based on the full risk sets, which is equivalent to a Cox proportional hazards analysis.27
For each outcome, regression models estimated the excess relative risk (ERR) associated with the radiologic exposures. Linear ERR models, which are often used to describe the effects of low-dose ionising radiation exposure, were preferred. Dose-response was further explored using categorical and natural (restricted cubic) spline models. Exposure categories (5 for outcomes with at least 50 cases; 3 otherwise) were based on the exposure distribution among the cases. Spline models placed knots at the 10th, 50th and 90th percentiles of the exposure distribution across all risk sets. Model fit was assessed using Akaike’s Information Criterion (AIC).28 All models were adjusted for birth year (using natural splines29) and pay code.
Univariate models examined radiation terms separately. To allow independent evaluation of the effects of each type of radiation exposure, multivariable models included terms for both organ dose and external radiation dose (and RDP exposure in WLM for models of lung cancer and chronic obstructive pulmonary disease (COPD)). For example, a multivariable ERR model (adjusted for pay code and birth year) was given by:
where t represents attained age, h0 (t) is the (unspecified) baseline hazard function (for unexposed hourly workers born in 1930), α1 is the log HR for salaried workers relative to hourly workers, α2 and α3 are spline parameters for birth year, and β, γ, and δ are the (age-, birth year- and pay code-adjusted) ERRs associated with one unit of organ dose, external dose and RDP exposure, respectively. In some models, the ERR was inestimable because either the dose parameter fell on the parameter space boundary (−1/maximum dose), or the model lacked convergence. In the former, the parameter estimate was assigned the lowest value that generated a non-negative HR, and a lower bound was not estimated; in the latter, no solution was reported. Consequently, log-linear models, which approximate linear models in the low-dose range, were considered and HRs reported. Because continuous linear trend estimators can be sensitive to outlying observations when exposure distributions are highly skewed,30 we assessed the impact of outliers using trimmed analyses that excluded risk set members with at least one exposure exceeding the exposure-specific 99th percentile.
For models of non-leukaemia outcomes, exposure lag periods of 0, 10 and 15 years were evaluated; however, when the number of cases was sparse, only a 10-year lag was evaluated because of the biological implausibility of a zero lag and the diminishing number of cases available at greater lags. For leukaemia models, chronic lymphocytic leukaemia was excluded because of possible differences in aetiology and latency and lags of 0, 2 and 5 years were assessed; however, deaths from lymphatic leukaemia not further specified were retained. The lag period was applied to all exposures in the model.
Assessment of selected potential non-radiologic confounders was included in univariate and multivariable modelling for outcomes with at least 30 cases (see online supplementary table S1). For agents to which at least 25% of the cohort was exposed, exposure duration to the agent was trichotomised as unexposed, low-duration (≤median exposure duration for exposed workers), and high-duration (the remainder of exposed workers). Less common exposures were dichotomised (ever/never to the cut-off date). Thorium, with only 150 workers assessed as exposed, was evaluated for potential confounding only in models of lung cancer and COPD. Potential confounders were added to the log-linear term in the base model one at a time, and confounding was evaluated by comparing dose coefficients from models including and excluding the potential confounding variables. Any variable resulting in more than a 20% change in the parameter estimate for one of the radiation terms was considered a confounder and retained in the model.
All regression modelling was performed using SAS (SAS Institute Inc, Cary, North Carolina, USA) based on recently published methods for modifying the Cox proportional hazards regression model to fit a linear ERR model using the NLMIXED procedure.31,32 Finally, diagnostics were run for lung cancer and COPD to assess collinearity among the three radiation terms (organ, external and RDP).
RESULTS
The study cohort comprised 6409 workers employed by NLO for at least 30 days (table 1). Collectively, there were 236 568 PYAR, for an average follow-up of 37 years. The cohort was largely male (85%) and Caucasian (95%). Workers’ mean age at first hire was 30.1 years. Results excluded six workers for whom pay code could not be determined. Although 40% of all workers were salaried, this ranged from 83% of Caucasian women to 20% of non-Caucasian men. Through 2004, a total of 2771 workers (43%) had died, with 860 of these deaths due to malignancies. Organ dose, external dose and RDP exposure distributions for the full cohort are summarised in table 1 and described in-depth in Anderson et al.11 A total of 348 workers had dose records at other facilities; the collective dose added from this offsite component was 1.29 person-Gy.
Table 1.
Cohort demographics and exposures by sex, race, and pay code*
| Male | Female | |||||||
|---|---|---|---|---|---|---|---|---|
| Caucasian† | Non-Caucasian | Caucasian‡ | Non-Caucasian | |||||
| Metric | Hourly | Salaried | Hourly | Salaried | Hourly | Salaried | Hourly | Salaried |
| Number | 3440 | 1771 | 193 | 47 | 153 | 731 | 30 | 38 |
| Person-years at risk | 125338 | 67615 | 6337 | 1487 | 4641 | 28839 | 912 | 1150 |
| Years of follow-up (mean, SD) | 36.4 (13.1) | 38.2 (12.4) | 32.8 (12.2) | 31.6 (9.7) | 30.3 (11.9) | 39.4 (12.3) | 30.4 (6.6) | 30.3 (7.1) |
| Percent deceased | 52.8 | 37.9 | 38.3 | 12.8 | 28.8 | 19.6 | 13.3 | 15.8 |
| Mean year of birth | 1928 | 1931 | 1935 | 1946 | 1938 | 1936 | 1951 | 1947 |
| Mean year of hire | 1959 | 1960 | 1965 | 1971 | 1970 | 1963 | 1973 | 1973 |
| Age at hire (mean, SD) | 31.6 (8.6) | 29.4 (8.9) | 29.7 (9.4) | 25.2 (5.9) | 32.1 (9.9) | 26.3 (7.8) | 22.0 (7.1) | 25.9 (9.5) |
| Years of employment (mean, SD) | 10.2 (9.9) | 8.53 (9.9) | 10.2 (9.4) | 4.69 (6.4) | 8.48 (8.6) | 7.19 (8.8) | 2.64 (5.4) | 3.42 (5.1) |
| Number of bioassay samples per worker (mean, SD) | 52.1 (57.1) | 18.3 (25.3) | 50.0 (50.4) | 12.7 (21.4) | 39.4 (51.9) | 10.5 (16.8) | 9.5 (20.3) | 5.4 (10.7) |
| Mean cumulative organ dose (µGy) | ||||||||
| Lung | 1552 | 388 | 965 | 138 | 67.9 | 296 | 34.5 | 154 |
| Kidney | 133 | 33.9 | 80.6 | 12.1 | 5.97 | 26.3 | 3.02 | 13.5 |
| Red bone marrow | 45.4 | 11.9 | 27.1 | 4.15 | 2.13 | 9.63 | 1.00 | 4.64 |
| Lower large intestine | 16.3 | 4.45 | 9.84 | 1.48 | 0.81 | 3.80 | 0.33 | 1.64 |
| Pancreas | 9.36 | 2.73 | 5.54 | 0.861 | 0.51 | 2.49 | 0.18 | 0.96 |
| Mean cumulative external dose (mGy) | 20.7 | 3.05 | 13.6 | 2.16 | 5.51 | 0.53 | 0.77 | 0.21 |
| Mean cumulative RDP (working level months) | 39.1 | 12.0 | 19.4 | 6.46 | 2.91 | 4.59 | 3.54 | 0.81 |
Workers employed at the Fernald Feed Materials Production Center at least 30 days between 1951–1985.
Results exclude four Caucasian males with unknown pay code (two of these were known to be deceased).
Results exclude two Caucasian females with unknown pay code (both were deceased).
FMPC, Feed Materials Production Center; PYAR, Person-years at risk; RDP, radon decay products.
Mortality results
For the full cohort, all-cause mortality was below expectation (SMR 0.91, 95% CI 0.88 to 0.95, n=2771) based on US population rates. However, all-cancer mortality was slightly elevated (SMR 1.06, 95% CI 0.99 to 1.14, n=860). All-cause and all-cancer mortality differed by race, sex and pay code (see online supplementary table S2). As there were few deaths among non-Caucasian workers, cause-specific mortality is reported by sex and pay code only (a priori outcomes, table 2; all outcomes, see online supplementary tables S3 and S4). Results were similar when Ohio referent rates were used (data not shown).
Table 2.
Observed deaths and standardised mortality ratios for all causes, all cancers and a priori outcomes by sex and pay code*, Fernald Feed Materials Production Center Cohort, 1951–2004†
| Males (n=5451) | Females (n=952) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hourly (n=3633) | Salaried (n=1818) | Hourly (n=183) | Salaried (n=769) | |||||||||
| Cause‡ | Obs | SMR | 95% CI | Obs | SMR | 95% CI | Obs | SMR | 95% CI | Obs | SMR | 95% CI |
| All causes | 1892 | 0.98 | 0.94 to 1.03 | 678 | 0.77 | 0.71 to 0.83 | 48 | 1.05 | 0.77 to 1.39 | 149 | 0.83 | 0.70 to 0.98 |
| All cancers | 575 | 1.15 | 1.06 to 1.24 | 211 | 0.90 | 0.79 to 1.03 | 13 | 1.05 | 0.56 to 1.79 | 59 | 0.97 | 0.74 to 1.25 |
| MN stomach | 18 | 1.15 | 0.68 to 1.82 | 12 | 1.77 | 0.91 to 3.09 | 0 | 0.00 | 0.00 to 13.2 | 2 | 1.93 | 0.23 to 6.96 |
| MN intestine | 50 | 1.16 | 0.86 to 1.53 | 14 | 0.71 | 0.39 to 1.18 | 2 | 1.66 | 0.20 to 6.00 | 4 | 0.82 | 0.22 to 2.09 |
| MN trachea, bronchus, lung | 223 | 1.25 | 1.09 to 1.42 | 52 | 0.62 | 0.46 to 0.81 | 5 | 2.01 | 0.65 to 4.68 | 17 | 1.13 | 0.66 to 1.81 |
| MN kidney | 12 | 0.95 | 0.49 to 1.66 | 3 | 0.49 | 0.10 to 1.44 | 0 | 0.00 | 0.00 to 18.2 | 3 | 2.99 | 0.62 to 8.75 |
| MN bladder and other urinary organs | 15 | 1.15 | 0.64 to 1.89 | 6 | 1.00 | 0.37 to 2.17 | 0 | 0.00 | 0.00 to 23.7 | 3 | 5.13 | 1.06 to 15.0 |
| Non-Hodgkin’s lymphoma | 18 | 0.97 | 0.58 to 1.54 | 12 | 1.33 | 0.69 to 2.32 | 2 | 4.41 | 0.53 to 16.0 | 0 | 0.00 | 0.00 to 1.71 |
| Hodgkin’s disease | 5 | 1.81 | 0.59 to 4.22 | 1 | 0.76 | 0.02 to 4.23 | 0 | 0.00 | 0.00 to 70.1 | 0 | 0.00 | 0.00 to 13.0 |
| Leukaemia | 17 | 0.92 | 0.54 to 1.48 | 15 | 1.71 | 0.95 to 2.81 | 0 | 0.00 | 0.00 to 9.23 | 3 | 1.61 | 0.33 to 4.70 |
| Multiple myeloma | 12 | 1.44 | 0.75 to 2.52 | 7 | 1.82 | 0.73 to 3.75 | 0 | 0.00 | 0.00 to 17.7 | 0 | 0.00 | 0.00 to 3.96 |
| Chronic obstructive pulmonary disease | 87 | 1.01 | 0.81 to 1.25 | 17 | 0.43 | 0.25 to 0.69 | 0 | 0.00 | 0.00 to 2.15 | 11 | 1.29 | 0.64 to 2.31 |
| Idiopathic pulmonary fibrosis | 11 | 1.77 | 0.88 to 3.17 | 3 | 1.01 | 0.21 to 2.94 | 1 | 7.40 | 0.19 to 41.2 | 0 | 0.00 | 0.00 to 6.10 |
| Chronic and unspecified nephritis and renal failure | 13 | 0.83 | 0.44 to 1.41 | 6 | 0.85 | 0.31 to 1.85 | 1 | 2.29 | 0.06 to 12.8 | 1 | 0.61 | 0.02 to 3.41 |
Four men and two women of unknown paycode were excluded.
Based on US population rates 1940–2004 for all outcomes except idiopathic pulmonary fibrosis (based on US population rates 1960–2004).
ICD codes were mapped to cause of death categories as described by Robinson and colleagues23 and as described on the NIOSH website (http://www.cdc.gov/niosh/ltas/rates.html).
COPD, chronic obstructive pulmonary disease; FMPC, Feed Materials Production Center; IPF, Idiopathic pulmonary fibrosis; MN, malignant neoplasm; Obs, observed number of deaths; SMR, standardised mortality ratio.
Males
Among hourly males (see online supplementary table S3), all-cause mortality was as expected (SMR=0.98, 95% CI 0.94 to 1.03, n=1892) but mortality from all cancers combined was elevated (SMR=1.15, 95% CI 1.06 to 1.24, n=575), primarily due to excess mortality from cancers of the lung (SMR=1.25, 95% CI 1.09 to 1.42, n=223) and digestive tract (SMR=1.21, 95% CI 1.03 to 1.43, n=148).
In salaried males, all-cause mortality was below expectation (SMR=0.77, 95% CI 0.71 to 0.83, n=678), with significant deficits in mortality from lung cancer (SMR=0.62, 95% CI 0.46 to 0.81, n=52), heart disease (SMR=0.69, 95% CI 0.60 to 0.79, n=209), and COPD (SMR=0.43, 95% CI 0.25 to 0.69, n=17). Mortality from all cancers was not elevated (SMR=0.90, 95% CI 0.79 to 1.03, n=211); however, mortality from lymphatic and haematopoietic tissue neoplasms was significantly elevated (SMR=1.52, 95% CI 1.06 to 2.12, n=35). Stomach cancer was not significantly elevated (SMR=1.77, 95% CI 0.91 to 3.09, n=12).
The SMR for the category encompassing cancers of the ‘peritoneum and other and unspecified digestive tract’ was significantly elevated among male workers, with nine deaths (SMR=3.51, 95% CI 1.61 to 6.67). Four of these were from peritoneal/retroperitoneal cancers and the remainder comprised miscellaneous digestive malignancies. Only one case of mesothelioma and one case of asbestosis were observed, both among hourly workers.
SRRs were significantly elevated for hourly compared with salaried males for a few causes of a priori interest: all cancers (SRR=1.29, 95% CI 1.10 to 1.52), including lung cancer (SRR=2.05, 95% CI 1.51 to 2.79); and COPD (SRR=2.37, 95% CI 1.40 to 4.00). Non-significantly elevated SRRs of at least 1.5 were seen for several a priori outcomes including Hodgkin’s disease and malignancies of the oesophagus, intestine, peritoneum and unspecified parts of the digestive system, and kidney, as well as a group of outcomes comprising pneumoconiosis and miscellaneous respiratory diseases. A non-significant twofold elevation was seen for brain cancer, not an outcome of a priori interest. Among non-cancer outcomes, the SRRs for ischaemic heart disease (SRR=1.38, 95% CI 1.16 to 1.64) and accidents (SRR=1.84, 95% CI 1.20 to 2.82) were significantly elevated.
Females
Among hourly females (see online supplementary table S4), all-cause mortality (SMR=1.05, 95% CI 0.77 to 1.39, n=48) and all-cancer mortality were as expected (SMR=1.05, 95% CI 0.56 to 1.79, n=13). In salaried females, all-cause mortality was reduced (SMR=0.83, 95% CI 0.70 to 0.98, n=149), mostly from a significant deficit of heart disease (SMR=0.55, 95% CI 0.35 to 0.82, n=24); mortality from cancers of the urinary tract was the only cause of a priori interest that was significantly elevated (SMR=3.78, 95% CI 1.39 to 8.23, n=6), with three deaths from kidney cancer and three from bladder cancer. Sparse deaths among hourly females precluded statistical comparison of standardised rates between hourly and salaried females.
Regression analyses
For each outcome, the lagged distributions of radiation metrics within risk sets are provided in online supplementary table S5. Untrimmed distributions were highly skewed. Maximum values were reduced in 1% trimmed risk sets, but the trimmed distributions remained quite skewed. Diagnostics indicated a low degree of collinearity among the three radiation terms for both lung cancer (maximum condition index 2.76) and COPD (maximum condition index 2.82).
For COPD and intestinal cancer, models with 10-year lags fit best, as determined by the AIC (data not shown). For lung cancer, the unlagged model fits slightly better than the model with a 10-year lag; however, a 10-year lag was selected because of biological plausibility. Similarly, leukaemia exposures were lagged 2 years, although unlagged models had a slightly improved fit.
Estimates from the univariate and multivariable regression models are displayed in tables 3 and 4, respectively, for outcomes with at least 25 cases, and in online supplementary tables S6 and S7, respectively, for other outcomes of a priori interest. Point estimates from dose-response analyses were scaled according to the observed range of internal exposure for cases and controls; therefore, effects from internal deposition are reported in units smaller than those used for external radiation.
Table 3.
Summary of univariate Cox proportional hazards regression models for Caucasian male Fernald workers (mortality through 2004) for outcomes of a priori interest with at least 25 cases
| Organ dose (µGy)* | External dose (mGy) | Radon decay products (WLM) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Model† | EST | PL 95% CI‡ | Model | EST | PL 95% CI‡ | Model | EST | PL 95% CI‡ | |||
| MN lung | U1 | Unexposed (25)§ | Ref | U1 | Unexposed (94) | Ref | U1 | 0.0–0.51 (53) | Ref | |||
| >0.0–14 (61) | 0.047 | (−0.33 to 0.68) | >0.0–2.0 (31) | −0.047 | (−0.36 to 0.39) | >0.51–5.4 (54) | −0.21 | (−0.45 to 0.13) | ||||
| >14–170 (61) | −0.16 | (−0.46 to 0.34) | >2.0–9.0 (54) | −0.13 | (−0.37 to 0.19) | >5.4–20 (54) | −0.33 | (−0.53 to −0.034) | ||||
| >170–1500 (61) | −0.34 | (−0.58 to 0.053) | >9.0–40 (46) | −0.26 | (−0.48 to 0.049) | >20–42 (55) | −0.22 | (−0.46 to 0.13) | ||||
| >1500 (61) | −0.094 | (−0.42 to 0.45) | >40 (44) | 0.019 | (−0.30 to 0.47) | >42 (53) | −0.25 | (−0.49 to 0.089) | ||||
| U2 | ERR (per 100 µGy) | 0.0021 | (−0.00062 to 0.0064) | U2 | ERR (per 100 mGy) | 0.17 | (−0.18 to 0.68) | U2 | ERR (per 10 WLM) | −0.0061 | (−0.013 to 0.0046) | |
| U3 | ERR (per 100 µGy), 1%¶ | 0.00087 | (−0.0034 to 0.0071) | U3 | ERR (per 100 mGy), 1% | 0.12 | (−0.32 to 0.74) | U3 | ERR (per 10 WLM), 1% | 0.00062 | (−0.011 to 0.020) | |
| U4 | HR (at 100 µGy) | 1.002 | (0.999 to 1.005) | U4 | HR (at 100 mGy) | 1.2 | (0.79 to 1.7) | U4 | HR (at 10 WLM) | 0.993 | (0.98 to 1.005) | |
| COPD | U1 | 0.0–24 (20) | Ref | U1 | Unexposed (27) | Ref | U1 | 0.0–4.3 (20) | Ref | |||
| >24–150 (21) | 0.64 | (−0.092 to 2.0) | >0.0–2.0 (12) | 0.20 | (−0.40 to 1.3) | >4.3–14 (20) | 0.67 | (−0.084 to 2.1) | ||||
| >150–650 (21) | 0.46 | (−0.20 to 1.7) | >2.0–16 (24) | −0.094 | (−0.48 to 0.57) | >14–30 (21) | 0.60 | (−0.12 to 1.9) | ||||
| >650–2100 (19) | 0.58 | (−0.15 to 1.9) | >16–49 (20) | 0.15 | (−0.38 to 1.1) | >30–45 (21) | 1.1 | (0.16 to 2.9) | ||||
| >2100 (21) | 0.54 | (−0.17 to 1.9) | >49 (19) | 0.62 | (−0.15 to 2.0) | >45 (20) | 0.42 | (−0.23 to 1.6) | ||||
| U2 | ERR (per 100 µGy) | −0.0018 | (NE to 0.0027) | U2 | ERR (per 100 mGy) | 0.48 | (−0.17 to 1.6) | U2 | ERR (per 10 WLM) | −0.011 | (−0.013 to 0.0068) | |
| U3 | ERR (per 100 µGy), 1% | 0.0015 | (−0.0056 to 0.014) | U3 | ERR (per 100 mGy), 1% | 0.58 | (−0.24 to 2.0) | U3 | ERR (per 10 WLM), 1% | −0.017 | (−0.017 to 0.012) | |
| U4 | HR (at 100 µGy) | 0.997 | (0.99 to 1.002) | U4 | HR (per 100 mGy) | 1.4 | (0.79 to 2.3) | U4 | HR (at 10 WLM) | 0.989 | (0.96 to 1.01) | |
| MN intestine | U1 | 0.0–0.01 (12) | Ref | U1 | Unexposed (19) | Ref | ||||||
| >0.01–0.98 (12) | −0.28 | (−0.68 to 0.65) | >0.0–3.0 (9) | −0.077 | (−0.60 to 1.0) | |||||||
| >0.98–4.4 (13) | 0.21 | (−0.46 to 1.8) | >3.0–9.1 (12) | 0.36 | (−0.37 to 1.8) | |||||||
| >4.4–36 (11) | −0.22 | (−0.67 to 0.86) | >9.1–28 (10) | 0.28 | (−0.44 to 1.8) | |||||||
| >36 (13) | 1.7 | (0.16 to 5.3) | >28 (11) | 0.13 | (−0.50 to 1.5) | |||||||
| U2 | ERR (per 100 µGy) | 1.5 | (0.12 to 4.1) | U2 | ERR (per 100 mGy) | −0.31 | (NE to 0.82) | |||||
| U3 | ERR (per 100 µGy), 1% | 2.1 | (0.13 to 5.7) | U3 | ERR (per 100 mGy), 1% | −0.30 | (−0.75 to 1.3) | |||||
| U4 | HR (at 100 µGy) | 1.7 | (0.92 to 2.7) | U4 | HR (at 100 mGy) | 0.74 | (0.23 to 1.8) | |||||
| MN pancreas | U1 | 0.0–0.13 (14) | Ref | U1 | 0.0–<2.0 (20) | Ref | ||||||
| >0.13–1.5 (13) | 0.49 | (−0.32 to 2.2) | 2.0–<7.0 (11) | 0.37 | (−0.37 to 1.8) | |||||||
| >1.5 (14) | 0.25 | (−0.44 to 1.9) | ≥7.0 (1) | −0.31 | (−0.70 to 0.50) | |||||||
| U2 | ERR (per 100 µGy) | −0.42 | (NE to 2.6) | U2 | ERR (per 100 mGy) | −0.31 | (NE to 0.82) | |||||
| U3 | ERR (per 100 µGy), 1% | 0.87 | (−1.6 to 8.5) | U3 | ERR (per 100 mGy), 1% | −0.78 | (NE to 0.39) | |||||
| U4 | HR (at 100 µGy) | 0.5 | (0.011 to 3.2) | U4 | HR (at 100 mGy) | 0.57 | (0.10 to 1.8) | |||||
| NHL | U1 | 0.0–0.26 (9) | Ref | U1 | Unexposed (10) | Ref | ||||||
| >0.26–7.2 (10) | 0.25 | (−0.52 to 2.4) | >0.0–9.9 (10) | 0.16 | (−0.55 to 2.1) | |||||||
| >7.2 (11) | 0.32 | (−0.52 to 2.7) | >9.9 (10) | 0.39 | (−0.52 to 3.1) | |||||||
| U2 | ERR (per 100 µGy) | 0.33 | (−0.065 to 1.6) | U2 | ERR (per 100 mGy) | 0.0010 | (−0.31 to 3.0) | |||||
| U3 | ERR (per 100 µGy), 1% | −0.24 | (−0.25 to 1.2) | U3 | ERR (per 100 mGy), 1% | −0.78 | (NE to 2.6) | |||||
| U4 | HR (at 100 µGy) | 1.2 | (0.88 to 1.5) | U4 | HR (at 100 mGy), 1% | 1.0 | (0.19 to 3.0) | |||||
| Leukaemia (excluding CLL) | U1 | 0.0–0.22 (10) | Ref | U1 | Unexposed (14) | Ref | ||||||
| >0.22–2.8 (8) | −0.042 | (−0.64 to 1.5) | >0.0–7.0 (7) | −0.50 | (−0.81 to 0.24) | |||||||
| >2.8 (10) | −0.38 | (−0.76 to 0.64) | >7.0 (7) | −0.42 | (−0.80 to 0.55) | |||||||
| U2 | ERR (per 100 µGy) | −0.061 | (NE to 0.25) | U2 | ERR (per 100 mGy) | 0.33 | (−0.27 to 2.8) | |||||
| U3 | ERR (per 100 µGy), 1% | −0.23 | (NE to 0.26) | U3 | ERR (per 100 mGy), 1% | −0.72 | (NE to 1.4) | |||||
| U4 | HR (at 100 µGy) | 0.27 | (0.022 to 0.96) | U4 | HR (at 100 mGy) | 1.8 | (0.54 to 4.2) | |||||
| MN stomach | U1 | 0.0–0.05 (9) | Ref | U1 | 0.0–1.0 (9) | Ref | ||||||
| >0.05–6.2 (9) | −0.32 | (−0.75 to 0.87) | >1.0–3.0 (8) | 1.2 | (−0.19 to 5.2) | |||||||
| >6.2 (10) | 0.65 | (−0.41 to 3.8) | >3.0 (11) | 0.33 | (−0.50 to 2.7) | |||||||
| U2 | ERR (per 100 µGy) | 0.38 | (−0.20 to 5.7) | U2 | ERR (per 100 mGy) | 1.1 | (−0.31 to 5.9) | |||||
| U3 | ERR (per 100 µGy), 1% | −0.80 | (NE to 7.6) | U3 | ERR (per 100 mGy), 1% | 0.39 | (−0.81 to 5.1) | |||||
| U4 | HR (at 100 µGy) | 1.2 | (0.21 to 2.6) | U4 | HR (at 100 mGy) | 1.8 | (0.45 to 4.7) | |||||
Target organ varied by outcome (see online supplementary table S5); estimates of cumulative exposure incorporate a 10-year lag period for non-leukaemia outcomes (2-year lag period for leukaemia excluding CLL).
All univariate models are adjusted for pay code (hourly/salaried) and birth year (spline terms) and consider one radiologic exposure at a time. Model U1 is linear ERR with a categorical treatment of exposure; model U2 is linear ERR with continuous treatment of exposure; model U3 is like model U2, but incorporates a 1% trim; and model U4 is log-linear with a continuous treatment of exposure. Models of lung cancer and COPD are additionally adjusted for occupational exposure to acids (external dose model only); the organ dose model of NHL is additionally adjusted for occupational exposure to trichloroethylene.
Profile likelihood 95% CIs.
Number of cases in parentheses.
1% denotes a 1% trimmed model which excluded risk sets for which the case exceeded the 99th percentile and all controls that exceed the 99th percentile.
CLL, chronic lymphocytic leukaemia; COPD, chronic obstructive pulmonary disease; ERR, excess relative risk; EST, estimate; MN, malignant neoplasm; NE, not estimable; NHL, non-Hodgkin’s lymphoma; WLM, working-level months.
Table 4.
Summary of multivariable Cox proportional hazards categorical regression modelling for Caucasian male Fernald workers (mortality through 2004) for outcomes of a priori interest with at least 25 cases
| Outcome | Model | Organ dose (µGy)* | EST | PL 95% CI‡ | External dose (mGy) | EST | PL 95% CI‡ | Radon decay products (WLM) | EST | PL 95% CI‡ |
|---|---|---|---|---|---|---|---|---|---|---|
| MN lung | M1 | Unexposed | Ref | Unexposed | Ref | 0.0–0.51 | Ref | |||
| >0.0–14 | 0.17 | (−0.25 to 0.79) | >0.0–2.0 | −0.049 | (−0.36 to 0.34) | >0.51–5.4 | −0.22 | (−0.55 to 0.13) | ||
| >14–170 | 0.0056 | (−0.38 to 0.56) | >2.0–9.0 | −0.018 | (−0.31 to 0.30) | >5.4–20 | −0.30 | (−0.70 to 0.089) | ||
| >170–1500 | −0.21 | (−0.54 to 0.25) | >9.0–40 | −0.084 | (−0.41 to 0.24) | >20–42 | −0.16 | (−0.58 to 0.29) | ||
| >1500 | 0.027 | (−0.37 to 0.60) | >40 | 0.17 | (−0.19 to 0.64) | >42 | −0.16 | (−0.60 to 0.31) | ||
| M2 | ERR (per 100 µGy) | 0.0022 | (−0.00093 to 0.0070) | ERR (per 100 mGy) | 0.13 | (−0.26 to 0.68) | ERR (per 10 WLM) | −0.0070 | (−0.013 to 0.0068) | |
| M3 | ERR (per 100 µGy), 1%§ | 0.00064 | (−0.0044 to 0.0073) | ERR (per 100 mGy), 1% | 0.093 | (−0.40 to 0.75) | ERR (per 10 WLM), 1% | 0.0011 | (−0.012 to 0.023) | |
| M4 | HR (at 100 µGy) | 1.002 | (0.999 to 1.005) | HR (at 100 mGy) | 1.2 | (0.76 to 1.7) | HR (at 10 WLM) | 0.999 | (0.97 to 1.004) | |
| COPD | M1 | 0.0–24 | Ref | Unexposed | Ref | 0.0–4.3 | Ref | |||
| >24–150 | 0.71 | (−0.36 to 2.6) | >0.0–2.0 | 0.21 | (−0.69 to 2.2) | >4.3–14 | 0.57 | (−0.47 to 2.4) | ||
| >150–650 | 0.50 | (−0.40 to 2.3) | >2.0–16 | −0.50 | (−0.96 to 0.51) | >14–30 | 0.46 | (−0.44 to 2.2) | ||
| >650–2100 | 0.70 | (−0.37 to 2.8) | >16–49 | −0.31 | (−1.5 to 1.2) | >30–45 | 1.3 | (−0.05 to 4.1) | ||
| >2100 | 0.36 | (−0.87 to 2.3) | >49 | 0.58 | (−0.89 to 3.2) | >45 | 0.36 | (−0.64 to 2.1) | ||
| M2 | ERR (per 100 µGy) | −0.0062 | (−0.0065 to 0.000062) | ERR (per 100 mGy) | 0.54 | (−0.00025 to 1.4) | ERR (per 10 WLM) | −0.018 | (−0.018 to 0.0027) | |
| M3 | ERR (per 100 µGy), 1% | 0.000048 | (−0.0059 to 0.013) | ERR (per 100 mGy), 1% | 0.45 | (−0.38 to 1.8) | ERR (per 10 WLM), 1% | −0.017 | (−0.017 to 0.025) | |
| M4 | HR (per 100 µGy) | 0.996 | (0.99 to 1.002) | HR (per 100 mGy) | 1.7 | (0.93 to 2.8) | HR (at 10 WLM) | 0.99 | (0.96 to 1.01) | |
| MN intestine | M1 | 0.0–0.01 | Ref | Unexposed | Ref | |||||
| >0.01–0.98 | −0.39 | (−0.78 to 0.49) | >0.0–3.0 | −0.20 | (−0.70 to 0.74) | |||||
| >0.98–4.4 | 0.24 | (−0.51 to 1.9) | >3.0–9.1 | 0.38 | (−0.38 to 1.8) | |||||
| >4.4–36 | −0.24 | (−0.78 to 0.91) | >9.1–28 | −0.18 | (−1.2 to 1.0) | |||||
| >36 | 1.7 | (0.17 to 5.7) | >28 | −0.23 | (−1.0 to 0.73) | |||||
| M2 | ERR (per 100 µGy) | NS | ERR (per 100 mGy) | NS | ||||||
| M3 | ERR (per 100 µGy), 1% | NS | ERR (per 100 mGy), 1% | NS | ||||||
| M4 | HR (at 100 µGy) | 2 | (1.05 to 3.2) | HR (at 100 mGy) | 0.57 | (0.18 to 1.4) | ||||
| MN pancreas | M1 | 0.0–0.13 | Ref | 0.0–<2.0 | Ref | |||||
| >0.13–1.5 | 0.44 | (−0.36 to 2.2) | 2.0–<7.0 | 0.36 | (−0.54 to 2.3) | |||||
| >1.5 | 0.46 | (−0.38 to 2.1) | ≥7.0 | −0.52 | (−0.96 to 0.52) | |||||
| M2 | ERR (per 100 µGy) | NS | ERR (per 100 mGy) | NS | ||||||
| M3 | ERR (per 100 µGy), 1% | NS | ERR (per 100 mGy), 1% | NS | ||||||
| M4 | HR (at 100 µGy) | 0.61 | (0.015 to 3.5) | HR (at 100 mGy) | 0.59 | (0.11 to 1.9) | ||||
| NHL | M1 | 0.0–0.26 | Ref | Unexposed | Ref | |||||
| >0.26–7.2 | 0.19 | (−0.65 to 4.1) | >0.0–9.9 | 0.21 | (−0.59 to 3.9) | |||||
| >7.2 | 0.090 | (−0.79 to 4.3) | >9.9 | 0.42 | (−1.3 to 4.9) | |||||
| M2 | ERR (per 100 µGy) | NS | ERR (per 100 mGy) | NS | ||||||
| M3 | ERR (per 100 µGy), 1% | NS | ERR (per 100 mGy), 1% | NS | ||||||
| M4 | HR (at 100 µGy) | 1.2 | (0.89 to 1.5) | HR (at 100 mGy) | 0.77 | (0.14 to 2.5) | ||||
| Leukaemia (excluding CLL) | M1 | 0.0–0.22 | Ref | Unexposed | Ref | |||||
| >0.22–2.8 | 0.095 | (−0.55 to 1.4) | >0.0–7.0 | −0.47 | (−0.89 to 0.25) | |||||
| >2.8 | −0.20 | (−0.70 to 0.61) | >7.0 | −0.36 | (−0.89 to 0.57) | |||||
| M2 | ERR (per 100 µGy) | NS | ERR (per 100 mGy) | NS | ||||||
| M3 | ERR (per 100 µGy), 1% | NS | ERR (per 100 mGy), 1% | NS | ||||||
| M4 | HR (at 100 µGy) | 0.18 | (0.012 to 0.80) | HR (at 100 mGy) | 2.8 | (0.87 to 6.0) | ||||
| MN stomach | M1 | 0.0–0.05 | Ref | 0.0–1.0 | Ref | |||||
| >0.05–6.2 | −0.51 | (−0.93 to 0.86) | >1.0–3.0 | 1.1 | (−0.20 to 4.9) | |||||
| >6.2 | 0.62 | (−0.66 to 4.8) | >3.0 | 0.15 | (−0.76 to 2.0) | |||||
| M2 | ERR (per 100 µGy) | 0.041 | (−0.20 to 5.6) | ERR (per 100 mGy) | 1.0 | (−0.31 to 6.1) | ||||
| M3 | ERR (per 100 µGy), 1% | NS | ERR (per 100 mGy), 1% | NS | ||||||
| M4 | HR (at 100 µGy) | 1.002 | (0.16 to 2.5) | HR (at 100 mGy) | 1.8 | (0.42 to 4.9) |
Target organ varied by outcome (see online supplementary table S5); estimates of cumulative exposure incorporate a 10-year lag period for non-leukaemia outcomes (2-year lag period for leukaemia excluding CLL).
All multivariable models are adjusted for pay code (hourly/salaried) and birth year (spline terms) and consider all radiologic exposures simultaneously. Model M1 is linear ERR with a categorical treatment of exposure; model M2 is linear ERR with continuous treatment of exposure; model M3 is like model M2, but incorporates a 1% trim; and model M4 is log-linear with a continuous treatment of exposure. Models of lung cancer and COPD are additionally adjusted for occupational exposure to acids; models of NHL are additionally adjusted for occupational exposure to trichloroethylene.
Profile likelihood 95% CIs.
1% denotes a 1% trimmed model which excluded risk sets for which the case exceeded the 99th percentile and all controls that exceed the 99th percentile.
CLL, chronic lymphocytic leukaemia; COPD, chronic obstructive pulmonary disease; ERR, excess relative risk; EST, estimate; MN, malignant neoplasm; NE, not estimable; NS, no solution found; NHL, non-Hodgkin’s lymphoma; RDP, Radon decay products; WLM, working-level months.
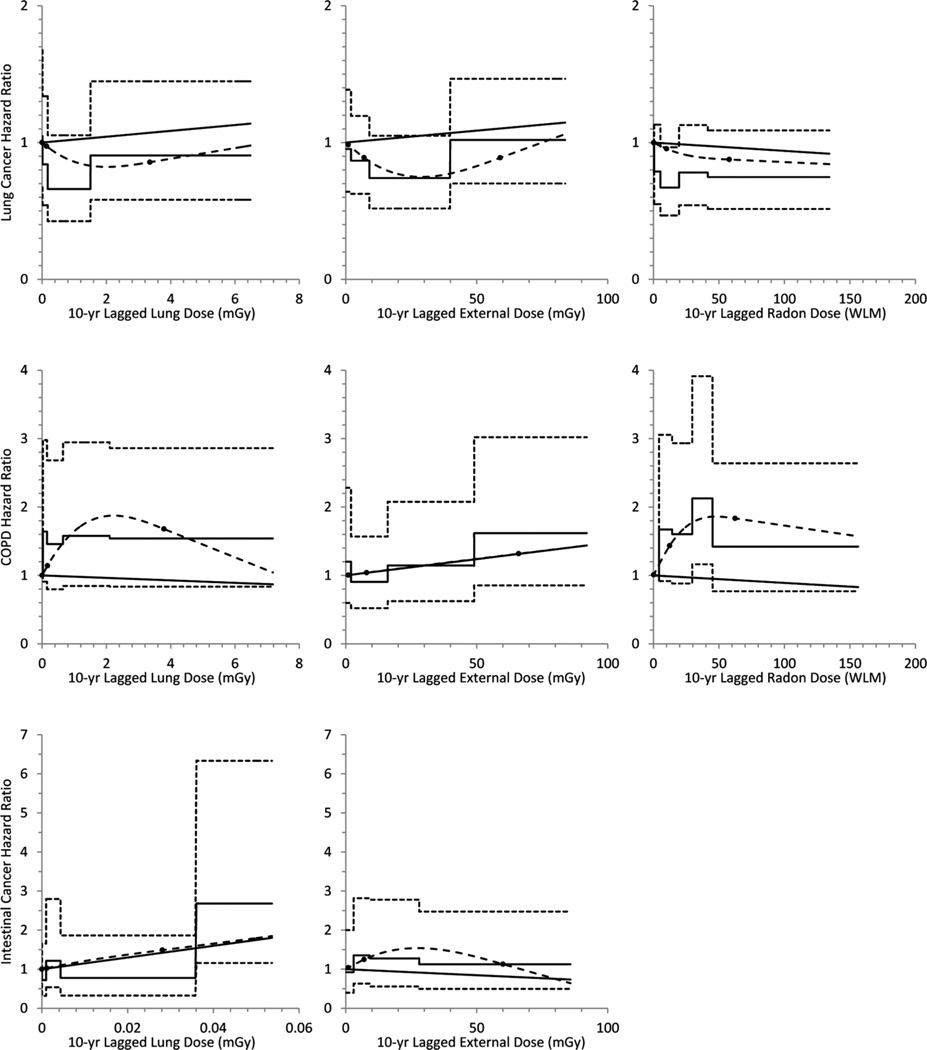
The categorical, spline and linear (untrimmed) models for outcomes with at least 50 cases are graphically displayed in figure 1. Graphs were truncated at the 95th percentile because data were sparse above that point, and distributions highly right-skewed.
Figure 1.
Categorical, linear and natural (restricted cubic) regression spline models evaluating exposure-response relations for lung cancer, COPD and intestinal cancer (outcomes of a priori interest with at least 50 cases) for Caucasian male Fernald workers (1951–2004).
In the categorical univariate and multivariable models, only for intestinal cancer did any organ dose category differ significantly from baseline, with an elevation in the highest category (>36 µGy). This finding was echoed by a positive, significant relation between organ dose and intestinal cancer in the univariate linear model (ERR=1.5 per 100 µGy, 95% CI 0.12 to 4.1); no solution was found for the multivariable linear model, but a positive, significant relation was seen for the multivariable log-linear model. Although no strong monotonic trend was evident, the linear model provided a reasonably good fit (evaluated using the AIC) for the relation between intestinal cancer and organ dose (figure 1). No other significant positive associations were observed. The only other statistically significant result was a negative relation between leukaemia and organ dose that was limited to the univariate and multivariable log-linear models.
A number of potential confounders were examined in the linear models. For both lung cancer and COPD, exposure to acids (trichotomised duration of exposure to hydrofluoric acid, nitric acid and/or nitrogen dioxide) was a confounder. Both trichloroethylene and laboratory chemicals were confounders for non-Hodgkin’s lymphoma; as the model would not converge with both terms included, trichloroethylene, which had the larger effect, was retained. No other non-radiologic exposure met the threshold for inclusion in the final models.
DISCUSSION
This update of the FMPC cohort extended follow-up and improved exposure characterisation. Overall mortality outcomes among Caucasian males, including differences by pay code, are generally consistent with those observed previously by Cragle et al.3 As in that study, all-cause mortality results suggest a healthy worker effect only in salaried employees and elevated all-cancer SMR only in hourly employees. The previously observed stomach cancer excess in salaried males has lessened somewhat and is no longer statistically significant, while the statistically significant lung cancer excess in hourly employees persists. A new finding is excess mortality from malignancies of the peritoneum and other and unspecified digestive tract sites. No worker dying from these causes held a job judged to have potential for asbestos exposure, and only single deaths from asbestosis and mesothelioma were reported.
Previous studies of FMPC employees were restricted to Caucasian males. Notable among females in this study was a statistically significant excess of urinary tract malignancies in salaried workers, but deaths were too sparse to permit further analysis. Results of regression analyses differ from those of previous examinations of Caucasian male FMPC workers. The positive, statistically significant relations between lung cancer and external and internal radiation dose seen in some previous studies were not observed here. Although the updated SMR for lung cancer was significantly elevated among hourly Caucasian males, no significant relations were seen between this outcome and any radiation metric. Cragle et al3 suggested that the relation between lung cancer and external dose category observed in the previous study was driven by excesses in the highest-dose categories. In the current study, no trend was observed, and although the highest category had a positive parameter estimate, the effect was far from statistically significant.
Previous studies also tended to group mortality outcomes because of case scarcity, some grouping NMRD very broadly2,3 and others grouping most lymphohaematopoietic malignancies together for exposure-response analyses.4,5 The current study used smaller outcome groupings for greater aetiologic homogeneity, but continued to be hampered by case scarcity in relation to the number of exposures considered. Chronic NMRD was previously associated with internal dose; in the current study, COPD, the largest subgroup of NMRD deaths, was not significantly associated with any type of radiation exposure.
A new finding of interest was the relation between intestinal cancer and organ dose. Although no non-radiologic exposure was retained in the models of intestinal cancer and organ dose, incomplete control for confounding by dust or some other chemical cannot be ruled out.
The negative, significant relation between leukaemia and organ dose, not observed in previous studies of this workforce, reflects clustering of cases in the very low-dose region and the preponderance of controls in the right tail. Dose uncertainty resulting in exposure misclassification may explain the observed risk attenuation. Another potential explanation is induction of leukaemia at lower doses among susceptible individuals.33–35
A number of factors likely contributed to between-study discrepancies. The additional follow-up ascertained new cases for many outcomes, including 155 additional lung cancer deaths in Caucasian males; differences in dose distribution among these new cases compared with those ascertained earlier could alter exposure-response results. Furthermore, exposures were evaluated differently in this study, with incorporation of external dose accrued at other facilities and assessment of absorbed organ dose. One previous study considered trichotomised estimated exposures to uranium dust.2 Another used estimated lung burdens from in vivo (whole-body counting) data and urinalysis data to represent internal dose for all-health outcomes studied, and analysed the effects of internal and external doses separately.3 By contrast, the software used in the current investigation facilitated calculation of absorbed doses for organs appropriate to the outcomes of interest, and radiation terms were considered both jointly and separately. Uranium urine concentrations were used to estimate radiation dose; however, excreta values may be a marker for other agents integral to the uranium compounds absorbed by exposed individuals. The concomitant effects are indistinguishable; hence, any observed association cannot be solely attributed to the effects of ionising radiation. There is sparse information on the relative biological effectiveness of uranium alpha particles for induction of some malignancies (eg, haematopoietic and digestive cancers); therefore, cautious interpretation of risks from uranium assimilation is warranted.
None of the previous studies analysed the effects of RDP. The suggestion by Hornung et al16 that RDP may be the predominant radiologic exposure associated with lung cancer among FMPC workers was not borne out in this study. RDP exposure showed a non-significant negative relation with lung cancer; the direction of the response changed when either workers employed for less than a year were excluded (data not shown) or when outliers were trimmed. The RDP estimates, based on location rather than individual dose data, have substantial uncertainty, and involve assumptions about workers’ locations, seasonal and diurnal variations in wind and temperature, the difference in RDP levels outside and within buildings and more. These assumptions hinder definitive evaluation of the relations between RDP and lung cancer at the facility. The current study was the first to evaluate all three radiation terms (external dose, organ dose and RDP) simultaneously.
Although both radiologic and non-radiologic exposures were considered, incomplete control for potential confounders remains an issue. The scarcity of exposed cases for some outcomes, and the prioritisation of radiation exposures resulted in limited statistical power to evaluate the role of nonradiologic exposures in the models. Furthermore, the nonradiologic exposures were evaluated using only a summary metric for duration of dichotomised exposure potential, with no metrics for intensity or temporality included.
Interpretation is also hindered by incomplete control for smoking and other lifestyle factors. Electronic smoking data were limited, and the collection of hard-copy smoking data, which were also sporadic, was deemed impractical. While inclusion of pay code in all models likely effects partial control for smoking, and mortality findings for lifestyle-related outcomes not evaluated a priori do not suggest an extremely strong role for lifestyle-related factors, individual smoking data would permit a more thorough assessment of the role of smoking in the elevated lung cancer SMR among hourly workers.
In summary, with 15 additional years of follow-up, mortality patterns have not changed greatly among Caucasian male FMPC workers. Further assessment of increased risks of bladder and renal cancers among Caucasian females might be enhanced by additional mortality follow-up or use of a cancer incidence design. While follow-up is now substantial for this cohort, average radiation doses are low and the cohort is relatively small, resulting in limited statistical power for sparse outcomes like leukaemia. Re-examination after another 10 years of follow-up have accrued may be warranted.
Supplementary Material
What this paper adds.
-
▸
Information on the risks from occupational exposures to uranium is limited.
-
▸
Earlier studies of workers exposed to soluble and insoluble uranium compounds while employed at the Fernald Feed Materials Production Center in Ohio, United States, were limited by short follow-up periods and the exclusion of female and non-Caucasian employees.
-
▸
We expanded the cohort to include all workers employed for 30 days or more (1951–1985) and extended vital status follow-up by 15 years.
-
▸
Consistent with previous studies, the healthy worker effect was observed only for salaried employees, and elevated all-cancer mortality was observed only for hourly employees.
-
▸
In regression analyses evaluating the relations between increasing dose from assimilated uranium (as estimated from available bioassay information) and mortality from outcomes of a priori interest, we only observed significant elevations in the risk of intestinal cancer.
Acknowledgements
The Internal Dose Evaluation Program (InDEP) was developed under a contract with NIOSH by SENES Oak Ridge, Inc, Center for Risk Analysis, Oak Ridge, TN.
Funding Funding for this study was provided through an agreement between the US Department of Energy (DOE) and the US Department of Health and Human Services (DHHS). The study was made possible by the cooperation and support of the DOE and their employees and contractors.
Footnotes
Disclaimer The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institute for Occupational Safety and Health.
Competing interests None.
Ethics approval The study protocol was approved by the Human Subjects Review Board of the National Institute for Occupational Safety and Health.
Provenance and peer review Not commissioned; externally peer reviewed.
REFERENCES
- 1.Canu IG, Ellis ED, Tirmarche M. Cancer risk in nuclear workers occupationally exposed to uranium—emphasis on internal exposure. Health Physics. 2008;94:1–17. doi: 10.1097/01.HP.0000281195.63082.e3. [DOI] [PubMed] [Google Scholar]
- 2.Wilson J. An epidemiologic investigation of non-malignant respiratory disease among workers at a uranium mill. The University of North Carolina; Chapel Hill: Ann Arbor, MI: University Microfilms International; 1983. [Google Scholar]
- 3.Cragle DL, Watkins JP, Ingle JN, et al. Mortality among a cohort of white male workers at a uranium processing plant: Fernald Feed Materials Production Center, 1951–1989. Oak Ridge, TN: Center for Epidemiologic Research, Oak Ridge Institute for Science and Education; 1996. [Google Scholar]
- 4.Ritz B. Radiation exposure and cancer mortality in uranium processing workers. Epidemiology. 1999;10:531–538. [PubMed] [Google Scholar]
- 5.Ritz B. Cancer mortality among workers exposed to chemicals during uranium processing. J Occup Environ Med. 1999;41:556–566. doi: 10.1097/00043764-199907000-00004. [DOI] [PubMed] [Google Scholar]
- 6.Dupree-Ellis E, Watkins J, Ingle JN, et al. External radiation exposure and mortality in a cohort of uranium processing workers. Am J Epidemiol. 2000;152:91–95. doi: 10.1093/aje/152.1.91. [DOI] [PubMed] [Google Scholar]
- 7.Archer VE, Renzetti AD, Doggett RS, et al. Chronic diffuse interstitial fibrosis of the lung in uranium miners. J Occup Environ Med. 1998;40:460–474. doi: 10.1097/00043764-199805000-00009. [DOI] [PubMed] [Google Scholar]
- 8.Newman LS, Mroz MM, Ruttenbur AJ. Lung fibrosis in plutonium workers. Radiat Res. 2005;164:123–131. doi: 10.1667/rr3407. [DOI] [PubMed] [Google Scholar]
- 9.Hubbard R, Lewis S, Richards K, et al. Occupational exposure to metal or wood dust and aetiology of cryptogenic fibrosing alveolitis. Lancet. 1996;347:284–289. doi: 10.1016/s0140-6736(96)90465-1. [DOI] [PubMed] [Google Scholar]
- 10.Scott J, Johnston I, Britton J. What causes cryptogenic fibrosing alveolitis? A case-control study of environmental exposure to dust. BMJ. 1990;301:1015–1017. doi: 10.1136/bmj.301.6759.1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Anderson J, Daniels RD, Fleming DA, et al. Exposure assessment for a cohort of workers at a uranium processing facility. J Expo Sci Environ Epidemiol. 2012;22:324–330. doi: 10.1038/jes.2012.20. [DOI] [PubMed] [Google Scholar]
- 12.ICRP. Age-dependent doses to members of the public from intake of radionuclides: part 43 ingestion dose coefficients. New York: Elsevier Science Inc.; 1995. Publication 69. [Google Scholar]
- 13.ICRP. Human respiratory tract model for radiological protection. New York: Elsevier Science Inc.; 1994. Publication 66. [Google Scholar]
- 14.Thierry-Chef I, Marshall M, Fix JJ, et al. The 15-country collaborative study of cancer risk among radiation workers in the nuclear industry: study of errors in dosimetry. Radiat Res. 2007;167:380–395. doi: 10.1667/RR0552.1. [DOI] [PubMed] [Google Scholar]
- 15.Daniels RD, Schubauer-Berigan MK. Bias and uncertainty of penetrating photon dose measured by film dosemeters in an epidemiological study of US nuclear workers. Radiat Prot Dosimetry. 2005;113:275–289. doi: 10.1093/rpd/nch470. [DOI] [PubMed] [Google Scholar]
- 16.Hornung RW, Pinney SM, Lodwick J, et al. Estimation of radon exposures to workers at the Fernald Feed Materials Production Center 1952–1988. J Expo Sci Environ Epidemiol. 2008;18:512–523. doi: 10.1038/sj.jes.7500645. [DOI] [PubMed] [Google Scholar]
- 17.NIOSH. NIOSH pocket guide to chemical hazards. Cincinnati, OH: 2009. [PubMed] [Google Scholar]
- 18.Fleming LE, Beckett WS. Occupational and environmental disease of the gastrointestinal system. In: Rom WM, editor. Occupational and environmental medicine. 3rd edn. Philadelphia: Lippincott-Raven; 1998. pp. 633–647. [Google Scholar]
- 19.Graham H, Francis B, Inskip HM, et al. Socioeconomic lifecourse influences on women’s smoking status in early adulthood. J Epidemiol Community Health. 2006;60:228–233. doi: 10.1136/jech.2005.039784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Paavola M, Vartiainen E, Haukkala A. Smoking from adolescence to adulthood: the effects of parental and own socioeconomic status. Eur J Public Health. 2004;14:417–421. doi: 10.1093/eurpub/14.4.417. [DOI] [PubMed] [Google Scholar]
- 21.Fujishiro K, Stukovsky KD, Roux AD, et al. Occupational gradients in smoking behavior and exposure to workplace environmental tobacco smoke: the multi-ethnic study of atherosclerosis. J Occup Environ Med. 2012;54:136–145. doi: 10.1097/JOM.0b013e318244501e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schubauer-Berigan MK, Hein MJ, Raudabaugh WM, et al. Update of the NIOSH Life Table Analysis System: a person-years analysis program for the Windows computing environment. Am J Indust Med. 2011;54:915–924. doi: 10.1002/ajim.20999. [DOI] [PubMed] [Google Scholar]
- 23.Robinson CF, Schnorr TM, Cassinelli RT, II, et al. Tenth revision U.S. mortality rates for use with the NIOSH Life Table Analysis System. J Occup Environ Med. 2006;48:662–667. doi: 10.1097/01.jom.0000229968.74906.8f. [DOI] [PubMed] [Google Scholar]
- 24.Rothman KJ. Modern epidemiology. Boston, MA: Little, Brown, and Company; 1986. [Google Scholar]
- 25.Schubauer-Berigan MK, Daniels RD, Pinkerton LE. Radon exposure and mortality among white and American Indian uranium miners: an update of the Colorado Plateau cohort. Am J Epidemiol. 2009;169:718–730. doi: 10.1093/aje/kwn406. [DOI] [PubMed] [Google Scholar]
- 26.Langholz B, Goldstein L. Risk set sampling in epidemiologic cohort studies. Statist Sci. 1996;11:35–53. [Google Scholar]
- 27.Callas PW, Pastides H, Hosmer DW. Empirical comparisons of proportional hazards, Poisson, and logistic regression modeling of occupational cohort data. Am J Ind Med. 1998;33:33–47. doi: 10.1002/(sici)1097-0274(199801)33:1<33::aid-ajim5>3.0.co;2-x. [DOI] [PubMed] [Google Scholar]
- 28.Akaike H. Information theory and an extension of the maximum likelihood principle. In: Petrov BN, Csaki F, editors. Second international symposium on information theory. Budapest: Akademiai Kiado; 1973. pp. 267–281. [Google Scholar]
- 29.Hein MJ, Schubauer-Berigan MK, Deddens JA. Evaluating bias from birth-cohort effects in the age-based Cox proportional hazards model. Epidemiology. 2011;22:249–256. doi: 10.1097/EDE.0b013e3182093912. [DOI] [PubMed] [Google Scholar]
- 30.Rosario AS, Wellmann J, Heid IM, et al. Radon epidemiology: continuous and categorical trend estimators when the exposure distribution is skewed and outliers may be present. J Toxicol Environ Health A. 2006;69:681–700. doi: 10.1080/15287390500261190. [DOI] [PubMed] [Google Scholar]
- 31.Langholz B, Richardson DB. Fitting general relative risk models for survival time and matched case-control analysis. Am J Epidemiol. 2010;171:377–383. doi: 10.1093/aje/kwp403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Richardson DB. A simple approach for fitting linear relative rate models in SAS. Am J Epidemiol. 2008;168:1333–1338. doi: 10.1093/aje/kwn278. [DOI] [PubMed] [Google Scholar]
- 33.Nakamura N. A hypothesis: radiation-related leukemia is mainly attributable to the small number of people who carry pre-existing clonally expanded preleukemic cells. Radiat Res. 2005;163:258–265. doi: 10.1667/rr3311. [DOI] [PubMed] [Google Scholar]
- 34.Hall EJ, Brenner DJ, Worgul B, et al. Genetic susceptibility to radiation. Adv Space Res. 2005;35:249–253. doi: 10.1016/j.asr.2004.12.032. [DOI] [PubMed] [Google Scholar]
- 35.Daniels RD, Bertke S, Waters KM, et al. Risk of leukaemia mortality from exposure to ionising radiation in US nuclear workers: a pooled case-control study. Occup Environ Med. 2013;70:41–48. doi: 10.1136/oemed-2012-100906. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.