Abstract
Purpose of review
This article describes the importance of extra-hepatic systemic manifestations of chronic hepatitis C virus (HCV) infection.
Recent findings
While most HCV literature focuses on liver injury and fibrosis progression, a spectrum of systemic disease processes, collectively called CHASM (C Hepatitis Associated Systemic Manifestations) are present in a high proportion of infected persons. These include thyroid disease (Hashimoto’s thyroiditis, Graves disease, thyroid cancer), cardiovascular disease (atherosclerosis, carotid artery disease, coronary artery disease), renal disease (MPGN, glomerulosclerosis), eye disease (Mooren’s ulcers, sicca syndrome), skin disease (PCT, vasculitis, lichen planus), and lymphomas (NHL, splenic T-cell), and diabetes.
Summary
Mechanistic understanding of how HCV leads to CHASM processes could lead to development of new interventions. The role of early HCV treatment and cure may result in preventive strategies for a variety of complex disease states.
Keywords: HCV, extrahepatic, thyroid, vasculitis, lymphoma, diabetes, neurologic
INTRODUCTION
Hepatitis C virus infection results in a chronic disease state affecting over 170 million people worldwide. HCV is a hepatotropic virus with its primary site of replication in the liver. Viral kinetic models suggest that most infected individuals produce 10-13 trillion virions/day, primarily but not exclusively from hepatocytes. There is growing evidence that a variety of other tissues can support limited replication of HCV in sites outside the liver compartment. These infections may represent a tiny fraction of the viral population, but may be quite significant in the development and modulation of systemic extrahepatic disease processes. Furthermore, immune response to HCV viral proteins links active infection and replication with a host of immune-mediated processes that can result in injury to distant organs. Finally, the direct effects of HCV on hepatic metabolism may have significant impact on lipids, hormones, and counter-regulatory peptides that contribute to systemic disease states associated with the metabolic syndrome. In those with HIV infection, HCV infection in the liver may result in development of portal hypertension that exacerbates immune activation associated with shunting of blood flow. Taken collectively, understanding the disease processes directly and indirectly affected by HCV requires examination of disorders involving the thyroid gland, lymphocytes, skin, kidneys, pancreas, intestinal tract, the brain, eyes, blood vessels, and the heart. Overlapping organ and tissue involvement in those with HIV infection can lead to difficult diagnostic dilemmas and accelerated pathogenesis in those with HIV/HCV coinfection.
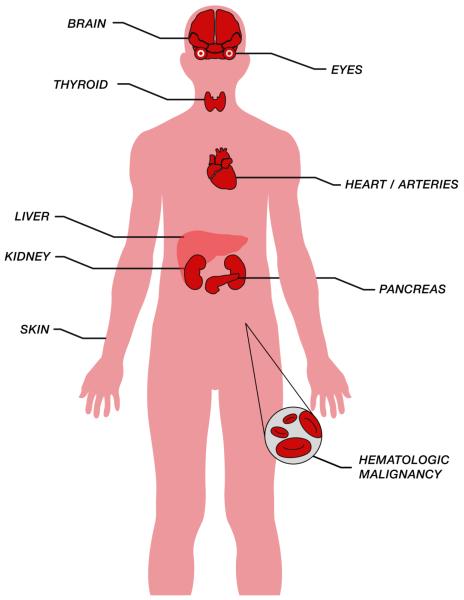
Epidemiologic support for the importance of extrahepatic disease processes to patient outcomes comes from recent studies that demonstrate an association between achievement of HCV cure (as evidenced by sustained viral response or SVR) and a decrease in both liver-related and all-cause mortality. Innes et al. reported mortality change in a Scottish cohort followed for a median time of 7.5 years. Non-liver mortality was reduced significantly with an adjusted hazard ratio of 0.68 compared to those who did not achieve viral clearance.1 A large study of HCV infected patients in the VA database demonstrated that non-liver related mortality was significantly reduced among patients who achieved SVR who had comorbidities that included coronary artery disease, diabetes, and hypertension. It was suggested that decreased chronic inflammation associated with HCV was a key factor in mortality decline.2 This finding was supported by data from a five center multinational study that included all consecutive patients enrolled between 1990 and 2003 with a median follow-up of 8.4 years. Sustained viral response was associated with an all-cause mortality hazard ratio of 0.26 and liver related mortality hazard ratio of 0.06. Both reductions were highly statistically significant.3 These studies point to the important role that HCV may play in non-hepatic causes of death. Therefore, an exploration of the specific effects HCV may have, directly and indirectly, on extrahepatic organs and tissues are warranted (see Figure 1). Since these processes may occur discretely or in combination within a single patient, we propose that they collectively be referred to as CHASM (C Hepatitis Associated Systemic Manifestations).
Figure 1.
Extra-hepatic sites of disease associated with HCV infection
Thyroid Disease
Following approval of interferon-alfa as treatment for HCV infection in 1991 came growing recognition that clinically significant thyroid disease was a common complication associated with therapy. Prospective studies demonstrated that up to 15% of all treated patients developed clinically recognizable thyroid disease and up to 40% developed thyroid antibodies. A typical pattern of disease was the development of severe thyrotoxicosis followed by development of hypothyroidism. Both Graves’ disease and Hashimoto’s thyroiditis were observed. These processes appear to have autoimmune etiology and are more common in women than men. The observation that discontinuation of interferon failed to resolve the condition led to the suggestion that interferon may trigger development of disease in those already predisposed to Graves’ and Hashimoto’s thyroid disease. (See Table 1). Subsequent epidemiologic studies established the link between HCV infection and incidence of thyroid antibodies, prior to exposure to interferon alfa. In a French series, Tran and colleagues reported a prevalence of 12.5% of HCV infected patients having thyroid antibodies, vs 1.7% in non-HCV infected controls (p= 0.21).4 This observation has been confirmed in numerous countries. Mechanistically, this phenomenon would be best explained by direct infection of thyroid tissues by HCV. Bartolome et al. reported detection of HCV RNA in thyroid tissue derived from HCV infected patients.5 Recently, Blackard et al. described productive infection of a human thyroid cell line (ML1) with HCV. The thyrocytes demonstrated expression of CD81 which is a major receptor for the HCV complex. Both positive and negative strand virus was identified in cell lysates. Interferon alfa inhibited HCV replication, and cell supernatants containing virus could infect native thyroid cells.6 It has been postulated that active infection with HCV leads to upregulation of CXCL10 expression in thyrocytes, which promotes recruitment of Th1 lymphocytes. These cells secrete interferon gamma and TNF alpha which further upregulate the immune cascade within the thyrocytes leading to active clinical disease manifestations.7
Table 1.
Effect of Cure with Interferon based regimens vs. DAA therapies
| PEG-INF + RBV | DAA based therapies | |
|---|---|---|
| Thyroid Disease | NO- May be exacerbated | Unknown |
| Eye Disease | Unknown | Unknown |
| Neurological Disease | Unknown | Unknown |
| CAD | Decreased | Unknown |
| Type 2 DM | Unknown | Unknown |
| Sjogren Syndrome | Possible improvement in sicca symptoms |
Unknown |
| B-cell NHL | May lead to regression | May lead to regression |
| Renal Disease | May improve | May improve |
| Skin Disease | Lichen planus (+/−) PCT (+/−) |
HCV-induced vasculitis |
Management of HCV-associated thyroid disease requires recognition of the process. All patients with HCV should have baseline TSH measurement to determine if hypo- or hyperthyroidism is present. Some experts also recommend evaluation of thyroid antibodies in all patients, to include TPO-AB and Tg-Ab since positive antibodies represent a risk factor for development of clinically relevant disease. In the increasingly rare event that interferon alfa is part of an HCV regimen, TSH levels should be monitored every three months. The predictive value of thyroid antibodies for clinical disease is not known at this time.8 Patients with incident thyroid disease should be managed medically. There are not sufficient data to determine long term effects of clearance of virus with interferon-free regimens.
Eye Disease
Early epidemiologic studies of HCV infected patients suggested a strong association between infection and certain relatively rare eye disorders. These include Mooren’s corneal ulcers and sicca syndrome.9 Decreased lacrimation leading to punctate keratitis and keratoconjunctivitis sicca are well described.10 Recent evidence suggests that HCV may be present in corneal cells from ocular donors infected with HCV.11 Other types of eye involvement include episcleritis, retinopathy and retinal vasculitis. HCV is in the tears of approximately 10% of those with HCV systemically.10
A prospective, cross-sectional trial examining intraocular pressure measurements HCV infected patients and controls revealed that mean tonometry was significant higher in HCV infected patients vs. blood donor controls.12 However, increased incidence of glaucoma has not been described to date.
Brain & Nervous System
Involvement of the brain and nervous system in HCV infection represents a relatively active area of research. Involvement of the brain and nervous tissue is potentially related to several distinct mechanisms including a) direct infection, b) indirect stimulation of neurotoxic cytokine pathways, and c) the development of central nervous system vasculitis.
The occurrence of neurocognitive changes associated with HCV is well-described and occurs in up to 50% of all HCV-infected persons. It manifests itself as fatigue and cognitive impairment.13,14 Neuroimaging supports the premise that HCV infection leads to changes associated with neurocognitive function aberrations.15 Direct infection and replication of HCV has been described by several investigators. Brain tissue and cerebrospinal fluid have positive and negative strand HCV RNA amplification indicative of active replication (as opposed to findings of only positive strand RNA which could be attributed to blood contamination).16-19 Letendre et al. studied HIV/HCV coinfected individuals and detected HCV by immune-based detection methods in astrocytes and macrophage-microglial cells.20 However, an in vitro study by Liu et al. attempted to infect human astrocytes with HCV. They found very restrictive replication despite the active binding and suggested that engagement of HCV with astrocytes led to strong IL-18 induction which led to direct neurotoxicity.21 Another mechanism that has been suggested for neurotoxicity is associated with toll like receptor (TLR) engagement. Activation of TLR2 following engagement of HCV core protein with dendritic cells appears to cause extracellular-signal kinase (ERK) mediated retraction of neuronal processes.22
In a post-mortem study of brain tissue of patients with HIV and HIV/HCV, investigators at Mt. Sinai hemisectioned brains and used a computer-aided virtual slide scanning system to examine arterioles and calculate an arteriolar sclerotic index. From this information, the authors determined the association of small vessel cerebrovascular disease (SVD) with various potential etiologic factors. The strongest independent risk factor of cerebral SVD was presence of HCV infection.23 This supports other studies that suggest that there are greater white matter abnormalities in those with HIV/HCV coinfection vs. HIV alone.24
Peripheral neuropathy associated with HCV infection is well-described and consists of sensory and sensorimotor symmetrical peripheral neuropathy. It is thought to be mediated through the development of cryoglobulinemia which leads to vasculitis-associated nerve ischemia.25 However, neuropathy has also been reported in those without cryoglobulinemia indicating that alternate mechanisms may be in play.26
A confounder in studies of HCV and neurocognitive dysfunction is the presence of liver disease. Hepatic fibrosis leads to development of porto-venous shunts that take portal blood derived from the gut to the systemic circulation. This blood contains neuroactive substances which cross the blood-brain barrier and lead to neurocognitive changes which are attributable to hepatic encephalopathy. These changes can be quite subtle and have been described as minimal hepatic encephalopathy or occult hepatic encephalopathy.27 Despite that, there is a growing body of evidence that HCV directly affects the brain and nerves independently of hepatic mediated processes.
Coronary Artery Disease & Atherosclerosis
The association between atherosclerosis and chronic hepatitis C infection has been described in the literature. Andinolfi et al. demonstrated that viral load and steatosis were independently associated with carotid atherosclerosis.28 Hsu et al. have demonstrated a clear link, showing that the eight-year cumulative incidences of ACS and ischemic stroke were significantly lower in patients with HCV who were treated (with peg-interferon and ribavirin) as compared to those in the untreated cohort (2.21% (95% CI 1.7% to 2.71%) vs 2.96% (95% CI 2.46% to 3.45%) for ACS (p=0.027), and 1.31% (95% CI 0.85% to 1.77%) vs 1.76% (95% CI 1.44% to 2.08%) for ischemic stroke (p=0.001).29 This is an overall estimated risk reduction of 23% in ACS and 38% in ischemic stroke. Petta et al. described a well-characterized cohort of 174 consecutive HCV genotype 1 infected patients and 174 non-HCV infected controls. Hepatic fibrosis associated with HCV infection was highly associated with presence of carotid plaques.30
Several mechanisms have been proposed, including direct damage from virions and immunological mechanisms. Serum HCV RNA levels have been shown to be independently associated with carotid atherosclerosis. Other studies suggest that HCV may replicate within cells found in atherosclerotic plaque.31
Other authors advocate for an immune-mediated process that leads to atherosclerosis. HCV may cause damage to arteries that creates a pro-inflammatory milieu.32 Specifically, Oliveira et al. measured elevated IL-6 and TNF-alpha in monoinfected patients with hepatitis C, and demonstrated a direct correlation with these pro-inflammatory markers and Framingham scores. The Framingham score suggested that HCV monoinfected, non-obese, non-diabetic patients have an intermediate cardiovascular risk, conferred by HCV alone. An analysis of the national VA database (ERCHIVES) among 237,069 persons including 111,436 HCV monoinfected and 1616 HCV/HIV coinfected patients also calculated Framingham scores and contrary to the prior study, failed to find higher Framingham risk scores for HCV-infected patients.33 However, the D.A.D. study and others have noted that the Framingham score may require adjustment in HIV-infected persons before use as a marker of actual cardiac risk.34 No prospective trials have yet validated actual cardiovascular risk however.
Diabetes
Numerous studies have reported the association between glucose intolerance and chronic hepatitis C infection. Bahtiyar et al. demonstrated that up to 1/3 of patients with chronic hepatitis C develop type 2 diabetes (T2DM), which is a prevalence that is much higher as compared to the general population. It is also a higher prevalence as compared to those patients with other chronic liver diseases such as HBV, alcoholic liver disease, and primary biliary cirrhosis.35 Observational studies have noted that individuals with T2DM had a higher prevalence of HCV infection as compared to non-T2DM control groups (OR = 1.26, 95% CI 1.00–1.57).36 Interestingly, this association was not observed in an analysis of the NHASES 1999-2010 cohort. Diabetes and pre-diabetes was not associated with HCV antibody status.37 This conflicted with another groups analysis of the same cohort which did report that HCV was associated with an odds ratio of 2.3 for presence of diabetes.38 Key differences in the two analyses were the nature of the controls, the definitions of diabetes utilized and whether or not ALT level was controlled for. While the cohort studies are not conclusive, examination of the underlying pathophysiology provides some insights into why a true association may exist.
The mechanism(s) that link hyperinsulinemia and diabetes to HCV infections are an area of intensive investigation. Shlomai et al. examined the relationship between PGC-1α (Peroxisome proliferator-activated receptor-gamma co-activator 1 alpha) and HCV infection in Huh 7.5 cells. PGC-1α was induced in HCV-infected cells which was accompanied by increased expression of glucose-6 phosphatase and increased glucose production. A mitigating response to N-acetylcysteine suggests that HCV-induced oxidative stress led to PGC-1α upregulation.39 Host IL28b genotype has also been associated with presence of insulin resistance. TT variants were significantly more likely to demonstrate insulin resistance as evidenced by increased HOMA scores.40 TT patients are less likely to clear HCV spontaneously which would lead to an association between active persistent HCV infection and insulin resistance leading to Type 2 DM.
Diabetes may also alter the natural history of HCV infection. Type 2 diabetes is recognized as causing an altered cytokine milieu. Insulin resistance mediated by proinflammatory cytokines may contribute to quicker development of steatosis and fibrosis.41,42
Sjogren Syndrome
The pathogenesis of Sjogren syndrome (SS) is thought to be secondary to both host factors (e.g. genetic predisposition) and exogenous factors. Viral agents in particular have been proposed as likely exogenous factors, and hepatitis C has been linked to causation of SS in a subset of patients. The association was first described in 1992.43 A recent systematic review confirms the strong association even when strict criteria are utilized.44 The controversy lies in how to best define the association; does HCV truly act as a factor that leads to primary SS, or do the clinical symptoms seen represent a HCV-induced disease that mimics primary SS?45 Ramos-Casals et al. proposed that primary SS and HCV-induced SS (SS-HCV) were two separate processes, as distinguished by epidemiological, pathological, and immunological differences.46 It was noted that SS-HCV typically presented at an older age as compared to primary SS, and a reduced female:male ratio was noted for SS-HCV as compared to the primary SS group. On histological examination, the two groups essentially were the same. However they differed by immunologic profile. The SS-HCV group demonstrated a higher prevalence of cryoglobulinemia, rheumatoid factor, and hypocomplementemia, and positive ANA with negative Ro/La.
Few studies have addressed the role of treatment of HCV. Sicca syndrome was improved with interferon or interferon/ribavirin therapy in patients that had SVR in a small series.47 No data are available regarding DAA-based cures.
B-cell non-Hodgkin’s Lymphoma
The first report of NHL associated with HCV was in 1994 by Ferri et al.48 Numerous studies have since confirmed this association, although the degree association varies greatly by country. In the 1990s, investigators described a very high prevalence of HCV in patients with mixed cryoglobinemia.49 While mixed cryoglobinemia has since been shown to be implicated in a wide variety of clinical presentations, ranging from arthralgia to vasculitis to glomerulonephritis, it is highly associated with development of non-Hodgkin’s lymphoma (NHL). HCV infected patients with symptoms of mixed cryoglobulinemia are approximately 35 times more likely to develop NHL as compared to the general population.50 A meta-analysis by Dal Masa et al. evaluated 15 studies, and summarized that about 10% of NHL can be attributed to HCV infection in regions where HCV prevalence is high.51
Treatment of hepatitis C with peg-interferon and ribavirin combinations, led to complete and partial remissions of NHL among patients with HCV.52,53 Recently, case reports support the concept that interferon-free regimens (sofosbuvir + ribavirin) can be used to treat HCV which subsequently led to lymphoma regression.54 Prospective studies are lacking at this time however.
Most studies have focused on the association between B-cell NHL and HCV; however other studies suggest a possible link between more rare forms of lymphoma and HCV as well. The meta-analysis mentioned above by Dal Maso also demonstrated a connection between T-cell lymphomas and HCV infection. However, the studies were few and often with limited sample sizes, and requires further investigation.
Three mechanisms have been proposed that potentially explain the link between HCV and NHL induction: chronic antigenic stimulation, CD81 engagement on B and T cells (which may lead to activation of B cells), and direct infection of B cells by HCV.55 The authors suggest that two or more oncogenic signals are required to initiate uncontrolled B-cell replication.
Other Solid Organ Malignancies
Since many solid organ malignancies are relatively rare compared to the general population, it has been difficult to ascertain whether reported associations between HCV and solid organ tumors is real or artifactual. A recent abstract, utilizing data from the Kaiser Permanente Southern California tumor registry examined this association using over 14 million patient years of data. Overall, head and neck cancer, esophageal cancer, stomach cancer, colorectal cancer pancreatic cancer, lung cancer, prostate cancer, and renal cancer were increased. The dataset also supported increased risk of hematogenous malignancies including NHL and myeloma associated with HCV infection.56 Similar data are reported in the Chronic Hepatitis Cohort Study (CHeCS) which examined health records from 12,126 HCV infected persons and compared them to 122,795,010 records from the SEER cancer registry. Pancreatic cancer, rectal cancer, kidney cancer and lung cancer risk were all increased as well as NHL.57
Antonelli et al. first recognized the prevalence of thyroid carcinoma with hepatitis C infection in 1999.58 Subsequently, a large study examined the associations of HCV and a variety of cancers. The odds ratio for thyroid cancer association with HCV infection was 2.8 (95% C.I. 1.2-6.3, p= 0.01).59 There are no data regarding effect of HCV cure on thyroid cancer incidence at this time.. However, a retrospective analysis of the Veteran’s Administration database failed to find an association between HCV and thyroid cancer, though a strong association with thyroiditis was noted.60 Thyroid cancer was highly associated with HCV infection (P< 0.0001) in the Kaiser Permanente analysis as noted above.56 The difference in these study results may be due to high rates of comorbidities in the VA population and lower proportions of women, which may obscure the ability to detect HCV-associated thyroid cancer.
Following observations of rising rates of renal cell carcinoma (RCC), investigators at Henry Ford Medical Center initiated a prospective evaluation of new RCCs and compared them to newly diagnosed colon cancer controls. HCV infection was highly associated with development of RRC (OR= 24.2).61 This finding is supported by the Kaiser Permanente retrospective registry as well.56
Other than the large cohort studies described above, there are limited data regarding the association between pancreatic cancer and HCV infection and the finding are mixed. However, a metanalysis examining 8 case-control studies did find that anti-HCV seropositivity was associated with development of pancreatic adenocarcinoma (OR 1.21, 95% CI 1.02-1.44).
Renal
A spectrum of HCV-related renal disease has been reported in the literature, including cryoglobulinemia leading to membranoproliferative glomerulonephritis (MPGN), non-cryoglobulinemia MPGN, IgA nephropathy, post-infectious glomerulonephritis, focal and segmental glomerulosclerosis, fibrillary glomerulonephritis, and immunotactoid glomerulopathy.62
Mixed cryoglobulinemia is most commonly associated with hepatitis C, in which the cryoglobulins consist mainly of rheumatoid factor and IgG.63 Cryoglobulins have been detected in about 50% of patients with HCV infection; however only about 1% of these patients have high levels of cryoglobulins associated with known disease processes.64 In terms of renal disease, cryoglobulinemia manifests with mild to moderate renal insufficiency with microscopic hematuria and proteinuria. Renal biopsy shows histology consistent with MPGN65 , with immune complex deposition frequently observed in the glomeruli.66
In terms of therapy, most treatments have targeted either the immune response or viral replication, or both. Two controlled trials have demonstrated improvements in cryoglobulins, rheumatoid factor, and creatinine levels with treatment with interferon alpha.67,68 Antiviral therapy (with interferon and ribavirin) has been shown to be successful for both HCV-related renal disease with cryoglobulinemia and HCV related MPGN, however, relapse rates are often high.65,69,70 However, with the advent of DAA’s, some authors have published case reports with promising results. For example, De Nicola et al. reported a case of HCV-1 related cryoglobulinemic membranoproliferative glomerulonephritis, with severe nephritic syndrome and rapidly progressive renal failure with treatment, who had complete resolution of her acute renal failure with combination therapy of telaprevir (DAA), PEG-IFN, and RBV.71
Dermatologic Manifestations
There are three main types of skin lesions that have been linked with hepatitis C infection: vasculitis associated with mixed essential cryoglobulinemia, porphyria cutanea tarda, and lichen planus.
Vasculitis, mixed essential cryoglobulinemia
As explained previously, cryoglobulinemia has been described in about 50% of patients with HCV, and is thought to trigger development of the vasculitis. Cryoglobulinemic vasculitis is classified as a small-vessel vasculitis, with clinical symptoms that include palpable purpura on the lower extremities, as well as arthralgias and weakness.
Previously, mixed cryoglobulinemia was treated with PEG-interferon and ribavirin. Rituximab was utilized as a second-line treatment option (or adjunct) if interferon and ribavirin failed.72 However, with DAA now becoming available, cohorts have begun to study combination treatments with DAA’s. A case report by Urraro et al. demonstrated complete remission of HCV-induced vasculitis with a combination treatment of rituximab, PEG-interferon, ribavirin, and a DAA (boceprevir).73 Further studies are warranted to validate the effectiveness of the combined therapy, since this was a single case report.
PCT
Porphyria cutanea tarda (PCT) is the most common form of porphyria. PCT is secondary to the decreased activity of the enzyme uroporphyrinogen decarboxylase .74 However, defects of the enzyme alone do not necessarily produce the phenotypic symptoms seen in PCT. Rather, exogenous factors such as iron overload, alcohol, and estrogen are required to produce manifestations of PCT, which includes blistering of the hands, forearms, and/or face, as well as cutaneous fragility.75 Numerous studies have shown an association between hepatitis C infection and PCT, as well as development of PCT at an earlier age in HCV-infected patients as compared to those without HCV.76 A systematic review found that 50% of patients with the diagnosis of PCT have HCV infection. Conversely, PCT occurs in 1-5% of those with chronic HCV.77 HIV may be a significant cofactor in development of PCT.78 The mechanistic underpinning of these observations remains unknown.
Studies have shown mixed results regarding remission of PCT with interferon based treatments. Several case reports demonstrated resolution of the skin lesions and normalization of urine porphyrins and serum liver enzymes and ferritin with interferon treatment.79,80 However, other studies refute these findings.81 To the authors’ knowledge, no cohorts have yet analyzed the effect of DAA therapy in regards to resolution of PCT.
Lichen planus
Lichen planus is a chronic, inflammatory, mucocutaneous skin disease. The lesions are most commonly seen in the extremities, as well as nails, face, scalp, and genitalia. Lesions of the mucosa are most commonly seen in the oral cavity, nasal membranes, esophagus, stomach, bladder, vagina, and glans penis.82 Studies have shown a 9.8-23% prevalence of hepatitis C seropositivity in individuals with lichen planus.83
The mechanism behind HCV and lichen planus has not been clearly elucidated; however it is suspected that it is an immune-mediated mechanism. Shared heptapeptides have been cited as a possible immunogen between several viruses including HCV and desmoglein-3 as well as other skin epitopes.84 Despite this hypothesis, no clear pattern of resolution of lichen planus has been demonstrated with interferon treatment for HCV. Studies have shown conflicting results. Doutre et al. suggested that treatment with interferon alpha completely cleared lichen planus.85 However, other studies have demonstrated that interferon may aggravate existing lichen planus.86,87,88 There are no reported cases of DAA treatment and the outcome of lichen planus to date.
CONCLUSION
There are multiple systemic manifestations of HCV infection that can collectively be grouped and identified as part of CHASM syndrome. Many are unrecognized and poorly characterized in terms of pathophysiology and treatment effects. In patients with concomitant HIV infection, the clinical presentation may be complicated by overlapping risks related to immune activation, increased gut permeability, and direct/indirect HIV effects on end organs. Furthermore, there is significantly overlap of HIV end-organ and HCV extrahepatic pathogenesis which often significantly increases the risk of these diseases in the HIV/HCV co-infected host. There is an urgent need to address these disease processes because improved understanding may lead to earlier recognition, treatment and perhaps resolution or even disease prevention. Furthermore, the current focus by payers on liver disease stage as a gateway to DAA access lacks recognition of the burden of HCV mediated disease outside of the liver. Further study to show associations with SVR and decreased extrahepatic clinical outcomes is desperately needed to expand access to DAA.
KEY POINTS.
Systemic extrahepatic complications of HCV comprise a spectrum of disease states in many organs and systems
Effective treatment of HCV may reduce or eliminate some but not all of these systemic complications
Further research into early treatment intervention as a prevention strategy for systemic disease is warranted.
Acknowledgments
The authors are grateful for graphic illustration support provided by Douglass Rouster and copy editing by Susan Rouster. This work was supported in part by NIDDK K24 070528 (K.E.S).
Footnotes
Compliance with Ethics Guidelines
Conflict of Interest
Amy C. Sherman and Kenneth E. Sherman declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
REFERENCES
Papers of particular interest, published recently, have been highlighted as:
• Of importance
•• Of major importance
- 1.Innes HA, McDonald SA, Dillon JF, et al. Toward a more complete understanding of the association between a hepatitis C sustained viral response and cause-specific outcomes. Hepatology. 2015 doi: 10.1002/hep.27766. [DOI] [PubMed] [Google Scholar]
- ** 2.Backus LI, Boothroyd DB, Phillips BR, Belperio P, Halloran J, Mole LA. A sustained virologic response reduces risk of all-cause mortality in patients with hepatitis C. Clin Gastroenterol Hepatol. 2011;9:509–16. e1. doi: 10.1016/j.cgh.2011.03.004. This paper demonstrated that effective HCV treatment had a significant effect on not just liver related mortality, but upon all cause mortality.
- 3.van der Meer AJ, Veldt BJ, Feld JJ, et al. Association between sustained virological response and all-cause mortality among patients with chronic hepatitis C and advanced hepatic fibrosis. JAMA. 2012;308:2584–93. doi: 10.1001/jama.2012.144878. [DOI] [PubMed] [Google Scholar]
- 4.Tran A, Quaranta JF, Benzaken S, et al. High prevalence of thyroid autoantibodies in a prospective series of patients with chronic hepatitis C before interferon therapy. Hepatology. 1993;18:253–7. [PubMed] [Google Scholar]
- 5.Bartolome J, Rodriguez-Inigo E, Quadros P, et al. Detection of hepatitis C virus in thyroid tissue from patients with chronic HCV infection. J Med Virol. 2008;80:1588–94. doi: 10.1002/jmv.21269. [DOI] [PubMed] [Google Scholar]
- 6.Blackard JT, Kong L, Huber AK, Tomer Y. Hepatitis C virus infection of a thyroid cell line: implications for pathogenesis of hepatitis C virus and thyroiditis. Thyroid : official journal of the American Thyroid Association. 2013;23:863–70. doi: 10.1089/thy.2012.0507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ferri C, Sebastiani M, Giuggioli D, et al. Hepatitis C virus syndrome: A constellation of organ- and non-organ specific autoimmune disorders, B-cell non-Hodgkin’s lymphoma, and cancer. World J Hepatol. 2015;7:327–43. doi: 10.4254/wjh.v7.i3.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tomer Y, Menconi F. Interferon induced thyroiditis. Best practice & research Clinical endocrinology & metabolism. 2009;23:703–12. doi: 10.1016/j.beem.2009.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wilson SE, Lee WM, Murakami C, Weng J, Moninger GA. Mooren’s corneal ulcers and hepatitis C virus infection. N Engl J Med. 1993;329:62. doi: 10.1056/NEJM199307013290118. [DOI] [PubMed] [Google Scholar]
- 10.Jacobi C, Wenkel H, Jacobi A, Korn K, Cursiefen C, Kruse FE. Hepatitis C and ocular surface disease. Am J Ophthalmol. 2007;144:705–11. doi: 10.1016/j.ajo.2007.07.028. [DOI] [PubMed] [Google Scholar]
- 11.Heck E, Dingrando A, Proctor C, Cavanagh HD. Viral HCV RNA reactivity of corneal cells in plasma HCV nucleic acid-positive eye donors. Cornea. 2013;32:506–7. doi: 10.1097/ICO.0b013e318268d6d6. [DOI] [PubMed] [Google Scholar]
- 12.Zeni LP, Viera PD, Michalczuk MT, Birkhan OA, Vilela MA, Alvares-da-Silva MR. Hepatitis C virus induces abnormalities in surface and intraocular pressure: a comparative study. Eur J Gastroenterol Hepatol. 2013;25:411–5. doi: 10.1097/MEG.0b013e32835bc2f1. [DOI] [PubMed] [Google Scholar]
- 13.Laskus T, Radkowski M, Adair DM, Wilkinson J, Scheck AC, Rakela J. Emerging evidence of hepatitis C virus neuroinvasion. AIDS. 2005;19(Suppl 3):S140–4. doi: 10.1097/01.aids.0000192083.41561.00. [DOI] [PubMed] [Google Scholar]
- 14.Kramer L, Bauer E, Funk G, et al. Subclinical impairment of brain function in chronic hepatitis C infection. J Hepatol. 2002;37:349–54. doi: 10.1016/s0168-8278(02)00172-1. [DOI] [PubMed] [Google Scholar]
- 15.Weissenborn K, Krause J, Bokemeyer M, et al. Hepatitis C virus infection affects the brain-evidence from psychometric studies and magnetic resonance spectroscopy. J Hepatol. 2004;41:845–51. doi: 10.1016/j.jhep.2004.07.022. [DOI] [PubMed] [Google Scholar]
- 16.Radkowski M, Wilkinson J, Nowicki M, et al. Search for hepatitis C virus negative-strand RNA sequences and analysis of viral sequences in the central nervous system: evidence of replication. J Virol. 2002;76:600–8. doi: 10.1128/JVI.76.2.600-608.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Laskus T, Radkowski M, Bednarska A, et al. Detection and analysis of hepatitis C virus sequences in cerebrospinal fluid. J Virol. 2002;76:10064–8. doi: 10.1128/JVI.76.19.10064-10068.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vargas HE, Laskus T, Radkowski M, et al. Detection of hepatitis C virus sequences in brain tissue obtained in recurrent hepatitis C after liver transplantation. Liver Transpl. 2002;8:1014–9. doi: 10.1053/jlts.2002.36393. [DOI] [PubMed] [Google Scholar]
- 19.Sherker AH, Twu JS, Reyes GR, Robinson WS. Presence of viral replicative intermediates in the liver and serum of patients infected with hepatitis C virus. J Med Virol. 1993;39:91–6. doi: 10.1002/jmv.1890390203. [DOI] [PubMed] [Google Scholar]
- * 20.Letendre S, Paulino AD, Rockenstein E, et al. Pathogenesis of hepatitis C virus coinfection in the brains of patients infected with HIV. J Infect Dis. 2007;196:361–70. doi: 10.1086/519285. Key paper describing mechanisms on brain dysfunction related to HCV in the HIV-infected patient.
- 21.Liu Z, Zhao F, He JJ. Hepatitis C virus (HCV) interaction with astrocytes: nonproductive infection and induction of IL-18. J Neurovirol. 2014;20:278–93. doi: 10.1007/s13365-014-0245-7. [DOI] [PubMed] [Google Scholar]
- 22.Paulino AD, Ubhi K, Rockenstein E, et al. Neurotoxic effects of the HCV core protein are mediated by sustained activation of ERK via TLR2 signaling. J Neurovirol. 2011;17:327–40. doi: 10.1007/s13365-011-0039-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Morgello S, Murray J, Van Der Elst S, Byrd D. HCV, but not HIV, is a risk factor for cerebral small vessel disease. Neurology(R) neuroimmunology & neuroinflammation. 2014;1:e27. doi: 10.1212/NXI.0000000000000027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jernigan TL, Archibald SL, Fennema-Notestine C, et al. Clinical factors related to brain structure in HIV: the CHARTER study. J Neurovirol. 2011;17:248–57. doi: 10.1007/s13365-011-0032-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nemni R, Corbo M, Fazio R, Quattrini A, Comi G, Canal N. Cryoglobulinaemic neuropathy. A clinical, morphological and immunocytochemical study of 8 cases. Brain : a journal of neurology. 1988;111(Pt 3):541–52. doi: 10.1093/brain/111.3.541. [DOI] [PubMed] [Google Scholar]
- 26.Lidove O, Cacoub P, Maisonobe T, et al. Hepatitis C virus infection with peripheral neuropathy is not always associated with cryoglobulinaemia. Annals of the rheumatic diseases. 2001;60:290–2. doi: 10.1136/ard.60.3.290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bajaj JS, Wade JB, Sanyal AJ. Spectrum of neurocognitive impairment in cirrhosis: Implications for the assessment of hepatic encephalopathy. Hepatology. 2009;50:2014–21. doi: 10.1002/hep.23216. [DOI] [PubMed] [Google Scholar]
- 28.Adinolfi LE, Restivo L, Zampino R, et al. Chronic HCV infection is a risk of atherosclerosis. Role of HCV and HCV-related steatosis. Atherosclerosis. 2012;221:496–502. doi: 10.1016/j.atherosclerosis.2012.01.051. [DOI] [PubMed] [Google Scholar]
- 29.Hsu YC, Ho HJ, Huang YT, et al. Association between antiviral treatment and extrahepatic outcomes in patients with hepatitis C virus infection. Gut. 2015;64:495–503. doi: 10.1136/gutjnl-2014-308163. [DOI] [PubMed] [Google Scholar]
- * 30.Petta S, Torres D, Fazio G, et al. Carotid atherosclerosis and chronic hepatitis C: a prospective study of risk associations. Hepatology. 2012;55:1317–23. doi: 10.1002/hep.25508. Important study that demonstrates relationship between HCV and atherosclerotic disease.
- 31.Boddi M, Abbate R, Chellini B, et al. HCV infection facilitates asymptomatic carotid atherosclerosis: preliminary report of HCV RNA localization in human carotid plaques. Dig Liver Dis. 2007;39(Suppl 1):S55–60. doi: 10.1016/s1590-8658(07)80012-0. [DOI] [PubMed] [Google Scholar]
- 32.Oliveira CP, Kappel CR, Siqueira ER, et al. Effects of hepatitis C virus on cardiovascular risk in infected patients: a comparative study. International journal of cardiology. 2013;164:221–6. doi: 10.1016/j.ijcard.2011.07.016. [DOI] [PubMed] [Google Scholar]
- 33.Chew KW, Bhattacharya D, McGinnis K, et al. Coronary Heart Disease Risk by Framingham Risk Score in Hepatitis C and HIV/Hepatitis C Coinfected Persons. AIDS Res Hum Retroviruses. 2015 doi: 10.1089/aid.2014.0284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Friis-Moller N, Thiebaut R, Reiss P, et al. Predicting the risk of cardiovascular disease in HIV-infected patients: the data collection on adverse effects of anti-HIV drugs study. European journal of cardiovascular prevention and rehabilitation : official journal of the European Society of Cardiology, Working Groups on Epidemiology & Prevention and Cardiac Rehabilitation and Exercise Physiology. 2010;17:491–501. doi: 10.1097/HJR.0b013e328336a150. [DOI] [PubMed] [Google Scholar]
- 35.Bahtiyar G, Shin JJ, Aytaman A, Sowers JR, McFarlane SI. Association of diabetes and hepatitis C infection: epidemiologic evidence and pathophysiologic insights. Current diabetes reports. 2004;4:194–8. doi: 10.1007/s11892-004-0023-7. [DOI] [PubMed] [Google Scholar]
- 36.Huang JF, Dai CY, Hwang SJ, et al. Hepatitis C viremia increases the association with type 2 diabetes mellitus in a hepatitis B and C endemic area: an epidemiological link with virological implication. Am J Gastroenterol. 2007;102:1237–43. doi: 10.1111/j.1572-0241.2007.01181.x. [DOI] [PubMed] [Google Scholar]
- 37.Ruhl CE, Menke A, Cowie CC, Everhart JE. Relationship of hepatitis C virus infection with diabetes in the U.S. population. Hepatology. 2014;60:1139–49. doi: 10.1002/hep.27047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Younossi ZM, Stepanova M, Nader F, Younossi Z, Elsheikh E. Associations of chronic hepatitis C with metabolic and cardiac outcomes. Aliment Pharmacol Ther. 2013;37:647–52. doi: 10.1111/apt.12234. [DOI] [PubMed] [Google Scholar]
- 39.Shlomai A, Rechtman MM, Burdelova EO, et al. The metabolic regulator PGC-1alpha links hepatitis C virus infection to hepatic insulin resistance. J Hepatol. 2012;57:867–73. doi: 10.1016/j.jhep.2012.06.021. [DOI] [PubMed] [Google Scholar]
- 40.Stattermayer AF, Rutter K, Beinhardt S, et al. Association of the IL28B genotype with insulin resistance in patients with chronic hepatitis C. J Hepatol. 2012;57:492–8. doi: 10.1016/j.jhep.2012.04.036. [DOI] [PubMed] [Google Scholar]
- 41.Skowronski M, Zozulinska D, Juszczyk J, Wierusz-Wysocka B. Hepatitis C virus infection: evidence for an association with type 2 diabetes. Diabetes care. 2006;29:750. doi: 10.2337/diacare.29.03.06.dc05-2263. author reply 1. [DOI] [PubMed] [Google Scholar]
- 42.Safi SZ, Shah H, Siok Yan GO, Qvist R. Insulin resistance provides the connection between hepatitis C virus and diabetes. Hepat Mon. 2015;15:e23941. doi: 10.5812/hepatmon.23941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Haddad J, Deny P, Munz-Gotheil C, et al. Lymphocytic sialadenitis of Sjogren’s syndrome associated with chronic hepatitis C virus liver disease. Lancet. 1992;339:321–3. doi: 10.1016/0140-6736(92)91645-O. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang Y, Dou H, Liu G, et al. Hepatitis C virus infection and the risk of Sjogren or sicca syndrome: a meta-analysis. Microbiology and immunology. 2014;58:675–87. doi: 10.1111/1348-0421.12202. [DOI] [PubMed] [Google Scholar]
- 45.De Vita S, Damato R, De Marchi G, Sacco S, Ferraccioli G. True primary Sjogren’s syndrome in a subset of patients with hepatitis C infection: a model linking chronic infection to chronic sialadenitis. Isr Med Assoc J. 2002;4:1101–5. [PubMed] [Google Scholar]
- 46.Ramos-Casals M, Loustaud-Ratti V, De Vita S, et al. Sjogren syndrome associated with hepatitis C virus: a multicenter analysis of 137 cases. Medicine (Baltimore) 2005;84:81–9. doi: 10.1097/01.md.0000157397.30055.c9. [DOI] [PubMed] [Google Scholar]
- 47.Doffoel-Hantz V, Loustaud-Ratti V, Ramos-Casals M, et al. [Evolution of Sjogren syndrome associated with hepatitis C virus when chronic hepatitis C is treated by interferon or the association of interferon and ribavirin] La Revue de medecine interne / fondee par la Societe nationale francaise de medecine interne. 2005;26:88–94. doi: 10.1016/j.revmed.2004.10.020. [DOI] [PubMed] [Google Scholar]
- 48.Ferri C, Caracciolo F, Zignego AL, et al. Hepatitis C virus infection in patients with non-Hodgkin’s lymphoma. Br J Haematol. 1994;88:392–4. doi: 10.1111/j.1365-2141.1994.tb05036.x. [DOI] [PubMed] [Google Scholar]
- 49.Pascual M, Perrin L, Giostra E, Schifferli JA. Hepatitis C virus in patients with cryoglobulinemia type II. J Infect Dis. 1990;162:569–70. doi: 10.1093/infdis/162.2.569. [DOI] [PubMed] [Google Scholar]
- 50.Monti G, Pioltelli P, Saccardo F, et al. Incidence and characteristics of non-Hodgkin lymphomas in a multicenter case file of patients with hepatitis C virus-related symptomatic mixed cryoglobulinemias. Arch Intern Med. 2005;165:101–5. doi: 10.1001/archinte.165.1.101. [DOI] [PubMed] [Google Scholar]
- 51.Dal Maso L, Franceschi S. Hepatitis C virus and risk of lymphoma and other lymphoid neoplasms: a meta-analysis of epidemiologic studies. Cancer Epidemiol Biomarkers Prev. 2006;15:2078–85. doi: 10.1158/1055-9965.EPI-06-0308. [DOI] [PubMed] [Google Scholar]
- 52.Hermine O, Lefrere F, Bronowicki JP, et al. Regression of splenic lymphoma with villous lymphocytes after treatment of hepatitis C virus infection. N Engl J Med. 2002;347:89–94. doi: 10.1056/NEJMoa013376. [DOI] [PubMed] [Google Scholar]
- 53.Vallisa D, Bernuzzi P, Arcaini L, et al. Role of anti-hepatitis C virus (HCV) treatment in HCV-related, low-grade, B-cell, non-Hodgkin’s lymphoma: a multicenter Italian experience. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2005;23:468–73. doi: 10.1200/JCO.2005.06.008. [DOI] [PubMed] [Google Scholar]
- 54.Sultanik P, Klotz C, Brault P, Pol S, Mallet V. Regression of an HCV-associated disseminated marginal zone lymphoma under IFN-free antiviral treatment. Blood. 2015;125:2446–7. doi: 10.1182/blood-2014-12-618652. [DOI] [PubMed] [Google Scholar]
- 55.Marcucci F, Mele A. Hepatitis viruses and non-Hodgkin lymphoma: epidemiology, mechanisms of tumorigenesis, and therapeutic opportunities. Blood. 2011;117:1792–8. doi: 10.1182/blood-2010-06-275818. [DOI] [PubMed] [Google Scholar]
- 56.Nyberg AHCJ, Shi JM, Cheetham TC, Chiang KM, Haque R, Younossi ZM, Nybert LM. International Liver Congress (EASL) Vienna, Austria: 2015. INCREASED CANCER RATES IN PATIENTS WITH CHRONIC HEPATITIS C: AN ANALYSIS OF THE CANCER REGISTRY. 2015. p. Abs #0058. [Google Scholar]
- 57.Allison RD, Tong X, Moorman AC, et al. Increased Incidence of Cancer and Cancer-related Mortality Among Persons with Chronic Hepatitis C Infection, 2006-2010. J Hepatol. 2015 doi: 10.1016/j.jhep.2015.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Antonelli A, Ferri C, Fallahi P. Thyroid cancer in patients with hepatitis C infection. JAMA. 1999;281:1588. doi: 10.1001/jama.281.17.1588. [DOI] [PubMed] [Google Scholar]
- 59.Montella M, Crispo A, de Bellis G, et al. HCV and cancer: a case-control study in a high-endemic area. Liver. 2001;21:335–41. doi: 10.1034/j.1600-0676.2001.210506.x. [DOI] [PubMed] [Google Scholar]
- 60.Giordano TP, Henderson L, Landgren O, et al. Risk of non-Hodgkin lymphoma and lymphoproliferative precursor diseases in US veterans with hepatitis C virus. JAMA. 2007;297:2010–7. doi: 10.1001/jama.297.18.2010. [DOI] [PubMed] [Google Scholar]
- 61.Gonzalez HC, Lamerato L, Rogers CG, Gordon SC. Chronic hepatitis C infection as a risk factor for renal cell carcinoma. Dig Dis Sci. 2015;60:1820–4. doi: 10.1007/s10620-015-3521-3. [DOI] [PubMed] [Google Scholar]
- 62.Meyers CM, Seeff LB, Stehman-Breen CO, Hoofnagle JH. Hepatitis C and renal disease: an update. Am J Kidney Dis. 2003;42:631–57. doi: 10.1016/s0272-6386(03)00828-x. [DOI] [PubMed] [Google Scholar]
- 63.Agnello V. Therapy for cryoglobulinemia secondary to hepatitis C virus: the need for tailored protocols and multiclinic studies. J Rheumatol. 2000;27:2065–7. [PubMed] [Google Scholar]
- 64.Pawlotsky JM, Ben Yahia M, Andre C, et al. Immunological disorders in C virus chronic active hepatitis: a prospective case-control study. Hepatology. 1994;19:841–8. [PubMed] [Google Scholar]
- 65.Johnson RJ, Gretch DR, Yamabe H, et al. Membranoproliferative glomerulonephritis associated with hepatitis C virus infection. N Engl J Med. 1993;328:465–70. doi: 10.1056/NEJM199302183280703. [DOI] [PubMed] [Google Scholar]
- 66.Agnello V. Mixed cryoglobulinaemia after hepatitis C virus: more and less ambiguity. Annals of the rheumatic diseases. 1998;57:701–2. doi: 10.1136/ard.57.12.701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Misiani R, Bellavita P, Fenili D, et al. Interferon alfa-2a therapy in cryoglobulinemia associated with hepatitis C virus. N Engl J Med. 1994;330:751–6. doi: 10.1056/NEJM199403173301104. [DOI] [PubMed] [Google Scholar]
- 68.Ferri C, Marzo E, Longombardo G, et al. Interferon-alpha in mixed cryoglobulinemia patients: a randomized, crossover-controlled trial. Blood. 1993;81:1132–6. [PubMed] [Google Scholar]
- 69.Casato M, Agnello V, Pucillo LP, et al. Predictors of long-term response to high-dose interferon therapy in type II cryoglobulinemia associated with hepatitis C virus infection. Blood. 1997;90:3865–73. [PubMed] [Google Scholar]
- 70.Gragnani L, Fognani E, Piluso A, et al. Long-term effect of HCV eradication in patients with mixed cryoglobulinemia: a prospective, controlled, open-label, cohort study. Hepatology. 2015;61:1145–53. doi: 10.1002/hep.27623. [DOI] [PubMed] [Google Scholar]
- 71.De Nicola S, Aghemo A, Campise MR, et al. Telaprevir in a patient with chronic hepatitis C and cryoglobulinemic glomerulonephritis. Antivir Ther. 2014;19:527–31. doi: 10.3851/IMP2684. [DOI] [PubMed] [Google Scholar]
- 72.Petrarca A, Rigacci L, Caini P, et al. Safety and efficacy of rituximab in patients with hepatitis C virus-related mixed cryoglobulinemia and severe liver disease. Blood. 2010;116:335–42. doi: 10.1182/blood-2009-11-253948. [DOI] [PubMed] [Google Scholar]
- 73.Urraro T, Gragnani L, Piluso A, et al. Combined treatment with antiviral therapy and rituximab in patients with mixed cryoglobulinemia: review of the literature and report of a case using direct antiviral agents-based antihepatitis C virus therapy. Case reports in immunology. 2015;2015:816424. doi: 10.1155/2015/816424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Elder GH. Porphyria cutanea tarda. Semin Liver Dis. 1998;18:67–75. doi: 10.1055/s-2007-1007142. [DOI] [PubMed] [Google Scholar]
- 75.Bonkovsky HL, Barnard GF. The Porphyrias. Current treatment options in gastroenterology. 2000;3:487–500. doi: 10.1007/s11938-000-0038-3. [DOI] [PubMed] [Google Scholar]
- 76.Cribier B, Chiaverini C, Dali-Youcef N, et al. Porphyria cutanea tarda, hepatitis C, uroporphyrinogen decarboxylase and mutations of HFE gene. A case-control study. Dermatology. 2009;218:15–21. doi: 10.1159/000173696. [DOI] [PubMed] [Google Scholar]
- 77.Gisbert JP, Garcia-Buey L, Pajares JM, Moreno-Otero R. Prevalence of hepatitis C virus infection in porphyria cutanea tarda: systematic review and meta-analysis. J Hepatol. 2003;39:620–7. doi: 10.1016/s0168-8278(03)00346-5. [DOI] [PubMed] [Google Scholar]
- 78.O’Connor WJ, Murphy GM, Darby C, et al. Porphyrin abnormalities in acquired immunodeficiency syndrome. Archives of dermatology. 1996;132:1443–7. [PubMed] [Google Scholar]
- 79.Siegel LB, Eber BB. Porphyria cutanea tarda remission. Ann Intern Med. 1994;121:308–9. doi: 10.7326/0003-4819-121-4-199408150-00018. [DOI] [PubMed] [Google Scholar]
- 80.Takikawa H, Yamazaki R, Shoji S, Miyake K, Yamanaka M. Normalization of urinary porphyrin level and disappearance of skin lesions after successful interferon therapy in a case of chronic hepatitis C complicated with porphyria cutanea tarda. J Hepatol. 1995;22:249–50. doi: 10.1016/0168-8278(95)80438-2. [DOI] [PubMed] [Google Scholar]
- 81.Furuta M, Kaito M, Gabazza E, et al. Ineffective interferon treatment of chronic hepatitis C-associated porphyria cutanea tarda, but with a transient decrease in HCV RNA levels. J Gastroenterol. 2000;35:60–2. doi: 10.1007/pl00009978. [DOI] [PubMed] [Google Scholar]
- 82.Nagao Y, Sata M. Hepatitis C virus and lichen planus. J Gastroenterol Hepatol. 2004;19:1101–13. doi: 10.1046/j.1440-1746.2003.03324.x. [DOI] [PubMed] [Google Scholar]
- 83.Poljacki M, Gajinov Z, Ivkov M, Matic M, Golusin Z. [Skin diseases and hepatitis virus C infection] Medicinski pregled. 2000;53:141–5. [PubMed] [Google Scholar]
- 84.Lucchese A. A potential peptide pathway from viruses to oral lichen planus. J Med Virol. 2015;87:1060–5. doi: 10.1002/jmv.24131. [DOI] [PubMed] [Google Scholar]
- 85.Doutre MS, Beylot C, Couzigou P, Long P, Royer P, Beylot J. Lichen planus and virus C hepatitis: disappearance of the lichen under interferon alfa therapy. Dermatology. 1992;184:229. doi: 10.1159/000247552. [DOI] [PubMed] [Google Scholar]
- 86.Protzer U, Ochsendorf FR, Leopolder-Ochsendorf A, Holtermuller KH. Exacerbation of lichen planus during interferon alfa-2a therapy for chronic active hepatitis C. Gastroenterology. 1993;104:903–5. doi: 10.1016/0016-5085(93)91029-h. [DOI] [PubMed] [Google Scholar]
- 87.Boccia S, Gamberini S, Dalla Libera M, Strumia R, Venturini D. Lichen planus and interferon therapy for hepatitis C. Gastroenterology. 1993;105:1921–2. doi: 10.1016/0016-5085(93)91099-4. [DOI] [PubMed] [Google Scholar]
- 88.Barreca T, Corsini G, Franceschini R, Gambini C, Garibaldi A, Rolandi E. Lichen planus induced by interferon-alpha-2a therapy for chronic active hepatitis C. Eur J Gastroenterol Hepatol. 1995;7:367–8. [PubMed] [Google Scholar]