Abstract
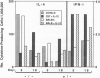
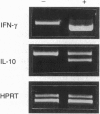
Interleukin 12 (IL-12) is a powerful stimulus for the growth of activated T and natural killer cells, their generation of interferon gamma (IFN-gamma), and the differentiation of T helper type 1 (Th1) effector cells from naive precursors in vitro. These activities are consistent with the capacity of exogenous IL-12 to heal otherwise susceptible BALB/c mice infected with the intramacrophage parasite Leishmania major. Using this characterized model of CD4 cell subset differentiation, we examined the immunologic effects of IL-12 administered either at the time of infection, when naive T cells are primed, or after 14 days of infection, by which time CD4+ subset differentiation has occurred. Given with the inoculation of parasites, IL-12 induced IFN-gamma and IL-10 and markedly suppressed IL-4. Effects on IL-10 and IL-4 were comparable in mice with homozygous disruption of the IFN-gamma gene (IFN-gamma 0/0), and suppression of IL-4 was unchanged by administration of neutralizing anti-IL-10 antibody. Induction of IFN-gamma and IL-10 mRNA by IL-12 also occurred in infected SCID mice. Given after day 14 of infection, however, IL-12 not only induced IFN-gamma and IL-10 but also induced IL-4 in normal and IFN-gamma 0/0 mice. These data demonstrate direct effects of IL-12 independent of IFN-gamma, IL-10, and IL-4 and demonstrate that the ineffectiveness of IL-12 administered following infection with L. major correlates with resistance of differentiated Th2 cells to the IL-4-suppressing activity of IL-12.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abrams J. S., Roncarolo M. G., Yssel H., Andersson U., Gleich G. J., Silver J. E. Strategies of anti-cytokine monoclonal antibody development: immunoassay of IL-10 and IL-5 in clinical samples. Immunol Rev. 1992 Jun;127:5–24. doi: 10.1111/j.1600-065x.1992.tb01406.x. [DOI] [PubMed] [Google Scholar]
- Afonso L. C., Scharton T. M., Vieira L. Q., Wysocka M., Trinchieri G., Scott P. The adjuvant effect of interleukin-12 in a vaccine against Leishmania major. Science. 1994 Jan 14;263(5144):235–237. doi: 10.1126/science.7904381. [DOI] [PubMed] [Google Scholar]
- Brunda M. J., Luistro L., Warrier R. R., Wright R. B., Hubbard B. R., Murphy M., Wolf S. F., Gately M. K. Antitumor and antimetastatic activity of interleukin 12 against murine tumors. J Exp Med. 1993 Oct 1;178(4):1223–1230. doi: 10.1084/jem.178.4.1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan S. H., Perussia B., Gupta J. W., Kobayashi M., Pospísil M., Young H. A., Wolf S. F., Young D., Clark S. C., Trinchieri G. Induction of interferon gamma production by natural killer cell stimulatory factor: characterization of the responder cells and synergy with other inducers. J Exp Med. 1991 Apr 1;173(4):869–879. doi: 10.1084/jem.173.4.869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chehimi J., Starr S. E., Frank I., Rengaraju M., Jackson S. J., Llanes C., Kobayashi M., Perussia B., Young D., Nickbarg E. Natural killer (NK) cell stimulatory factor increases the cytotoxic activity of NK cells from both healthy donors and human immunodeficiency virus-infected patients. J Exp Med. 1992 Mar 1;175(3):789–796. doi: 10.1084/jem.175.3.789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chua A. O., Chizzonite R., Desai B. B., Truitt T. P., Nunes P., Minetti L. J., Warrier R. R., Presky D. H., Levine J. F., Gately M. K. Expression cloning of a human IL-12 receptor component. A new member of the cytokine receptor superfamily with strong homology to gp130. J Immunol. 1994 Jul 1;153(1):128–136. [PubMed] [Google Scholar]
- Clerici M., Lucey D. R., Berzofsky J. A., Pinto L. A., Wynn T. A., Blatt S. P., Dolan M. J., Hendrix C. W., Wolf S. F., Shearer G. M. Restoration of HIV-specific cell-mediated immune responses by interleukin-12 in vitro. Science. 1993 Dec 10;262(5140):1721–1724. doi: 10.1126/science.7903123. [DOI] [PubMed] [Google Scholar]
- Coffman R. L., Varkila K., Scott P., Chatelain R. Role of cytokines in the differentiation of CD4+ T-cell subsets in vivo. Immunol Rev. 1991 Oct;123:189–207. doi: 10.1111/j.1600-065x.1991.tb00611.x. [DOI] [PubMed] [Google Scholar]
- Dalton D. K., Pitts-Meek S., Keshav S., Figari I. S., Bradley A., Stewart T. A. Multiple defects of immune cell function in mice with disrupted interferon-gamma genes. Science. 1993 Mar 19;259(5102):1739–1742. doi: 10.1126/science.8456300. [DOI] [PubMed] [Google Scholar]
- Desai B. B., Quinn P. M., Wolitzky A. G., Mongini P. K., Chizzonite R., Gately M. K. IL-12 receptor. II. Distribution and regulation of receptor expression. J Immunol. 1992 May 15;148(10):3125–3132. [PubMed] [Google Scholar]
- Finkelman F. D., Madden K. B., Cheever A. W., Katona I. M., Morris S. C., Gately M. K., Hubbard B. R., Gause W. C., Urban J. F., Jr Effects of interleukin 12 on immune responses and host protection in mice infected with intestinal nematode parasites. J Exp Med. 1994 May 1;179(5):1563–1572. doi: 10.1084/jem.179.5.1563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gately M. K., Desai B. B., Wolitzky A. G., Quinn P. M., Dwyer C. M., Podlaski F. J., Familletti P. C., Sinigaglia F., Chizonnite R., Gubler U. Regulation of human lymphocyte proliferation by a heterodimeric cytokine, IL-12 (cytotoxic lymphocyte maturation factor). J Immunol. 1991 Aug 1;147(3):874–882. [PubMed] [Google Scholar]
- Gazzinelli R. T., Hieny S., Wynn T. A., Wolf S., Sher A. Interleukin 12 is required for the T-lymphocyte-independent induction of interferon gamma by an intracellular parasite and induces resistance in T-cell-deficient hosts. Proc Natl Acad Sci U S A. 1993 Jul 1;90(13):6115–6119. doi: 10.1073/pnas.90.13.6115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Germann T., Gately M. K., Schoenhaut D. S., Lohoff M., Mattner F., Fischer S., Jin S. C., Schmitt E., Rüde E. Interleukin-12/T cell stimulating factor, a cytokine with multiple effects on T helper type 1 (Th1) but not on Th2 cells. Eur J Immunol. 1993 Aug;23(8):1762–1770. doi: 10.1002/eji.1830230805. [DOI] [PubMed] [Google Scholar]
- Heinzel F. P., Sadick M. D., Mutha S. S., Locksley R. M. Production of interferon gamma, interleukin 2, interleukin 4, and interleukin 10 by CD4+ lymphocytes in vivo during healing and progressive murine leishmaniasis. Proc Natl Acad Sci U S A. 1991 Aug 15;88(16):7011–7015. doi: 10.1073/pnas.88.16.7011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinzel F. P., Schoenhaut D. S., Rerko R. M., Rosser L. E., Gately M. K. Recombinant interleukin 12 cures mice infected with Leishmania major. J Exp Med. 1993 May 1;177(5):1505–1509. doi: 10.1084/jem.177.5.1505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsieh C. S., Macatonia S. E., Tripp C. S., Wolf S. F., O'Garra A., Murphy K. M. Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science. 1993 Apr 23;260(5107):547–549. doi: 10.1126/science.8097338. [DOI] [PubMed] [Google Scholar]
- Manetti R., Gerosa F., Giudizi M. G., Biagiotti R., Parronchi P., Piccinni M. P., Sampognaro S., Maggi E., Romagnani S., Trinchieri G. Interleukin 12 induces stable priming for interferon gamma (IFN-gamma) production during differentiation of human T helper (Th) cells and transient IFN-gamma production in established Th2 cell clones. J Exp Med. 1994 Apr 1;179(4):1273–1283. doi: 10.1084/jem.179.4.1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKnight A. J., Zimmer G. J., Fogelman I., Wolf S. F., Abbas A. K. Effects of IL-12 on helper T cell-dependent immune responses in vivo. J Immunol. 1994 Mar 1;152(5):2172–2179. [PubMed] [Google Scholar]
- Morris S. C., Madden K. B., Adamovicz J. J., Gause W. C., Hubbard B. R., Gately M. K., Finkelman F. D. Effects of IL-12 on in vivo cytokine gene expression and Ig isotype selection. J Immunol. 1994 Feb 1;152(3):1047–1056. [PubMed] [Google Scholar]
- Reiner S. L., Wang Z. E., Hatam F., Scott P., Locksley R. M. TH1 and TH2 cell antigen receptors in experimental leishmaniasis. Science. 1993 Mar 5;259(5100):1457–1460. doi: 10.1126/science.8451641. [DOI] [PubMed] [Google Scholar]
- Reiner S. L., Zheng S., Corry D. B., Locksley R. M. Constructing polycompetitor cDNAs for quantitative PCR. J Immunol Methods. 1993 Sep 27;165(1):37–46. doi: 10.1016/0022-1759(93)90104-f. [DOI] [PubMed] [Google Scholar]
- Reiner S. L., Zheng S., Wang Z. E., Stowring L., Locksley R. M. Leishmania promastigotes evade interleukin 12 (IL-12) induction by macrophages and stimulate a broad range of cytokines from CD4+ T cells during initiation of infection. J Exp Med. 1994 Feb 1;179(2):447–456. doi: 10.1084/jem.179.2.447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadick M. D., Heinzel F. P., Holaday B. J., Pu R. T., Dawkins R. S., Locksley R. M. Cure of murine leishmaniasis with anti-interleukin 4 monoclonal antibody. Evidence for a T cell-dependent, interferon gamma-independent mechanism. J Exp Med. 1990 Jan 1;171(1):115–127. doi: 10.1084/jem.171.1.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmitt E., Hoehn P., Germann T., Rüde E. Differential effects of interleukin-12 on the development of naive mouse CD4+ T cells. Eur J Immunol. 1994 Feb;24(2):343–347. doi: 10.1002/eji.1830240211. [DOI] [PubMed] [Google Scholar]
- Seder R. A., Gazzinelli R., Sher A., Paul W. E. Interleukin 12 acts directly on CD4+ T cells to enhance priming for interferon gamma production and diminishes interleukin 4 inhibition of such priming. Proc Natl Acad Sci U S A. 1993 Nov 1;90(21):10188–10192. doi: 10.1073/pnas.90.21.10188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sypek J. P., Chung C. L., Mayor S. E., Subramanyam J. M., Goldman S. J., Sieburth D. S., Wolf S. F., Schaub R. G. Resolution of cutaneous leishmaniasis: interleukin 12 initiates a protective T helper type 1 immune response. J Exp Med. 1993 Jun 1;177(6):1797–1802. doi: 10.1084/jem.177.6.1797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tripp C. S., Wolf S. F., Unanue E. R. Interleukin 12 and tumor necrosis factor alpha are costimulators of interferon gamma production by natural killer cells in severe combined immunodeficiency mice with listeriosis, and interleukin 10 is a physiologic antagonist. Proc Natl Acad Sci U S A. 1993 Apr 15;90(8):3725–3729. doi: 10.1073/pnas.90.8.3725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z. E., Reiner S. L., Zheng S., Dalton D. K., Locksley R. M. CD4+ effector cells default to the Th2 pathway in interferon gamma-deficient mice infected with Leishmania major. J Exp Med. 1994 Apr 1;179(4):1367–1371. doi: 10.1084/jem.179.4.1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagida T., Kato T., Igarashi O., Inoue T., Nariuchi H. Second signal activity of IL-12 on the proliferation and IL-2R expression of T helper cell-1 clone. J Immunol. 1994 May 15;152(10):4919–4928. [PubMed] [Google Scholar]
- Yoshimoto T., Paul W. E. CD4pos, NK1.1pos T cells promptly produce interleukin 4 in response to in vivo challenge with anti-CD3. J Exp Med. 1994 Apr 1;179(4):1285–1295. doi: 10.1084/jem.179.4.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Waal Malefyt R., Yssel H., de Vries J. E. Direct effects of IL-10 on subsets of human CD4+ T cell clones and resting T cells. Specific inhibition of IL-2 production and proliferation. J Immunol. 1993 Jun 1;150(11):4754–4765. [PubMed] [Google Scholar]