Abstract:
Improvements in micropore arterial line filter designs used for extracorporeal circulation are still needed because microbubbles larger than the rated pore sizes are being detected beyond the filter outlet. Linked to principles governing the function of micropore filters, fluid pressures contained in extracorporeal circuits also influence the behavior of gas bubbles and the extent to which they are carried in a fluid flow. To better understand the relationship between pressure and microbubble behavior, two ex vivo test circuits with and without inline resistance were designed to assess changes in microbubble load with changes in pressure. Ultrasound Doppler probes were used to measure and compare the quality and quantity of microbubbles generated in each test circuit. Analysis of microbubble load was separated into two distinct phases, the time periods during and immediately after bubble generation. Although microbubble number decreased similarly in both test circuits, changes in microbubble volume were significant only in the test circuit with inline resistance. The test circuit with inline resistance also showed a decrease in the rate of volume transferred across each ultrasound Doppler probe and the microbubble number and size range measured in the postbubble generation period. The present research proposes that fluid pressures contained in extracorporeal circuits may be used to affect gases in solution as a possible method to improve microbubble filtration during extracorporeal circulation.
Keywords: microbubble, arterial line filter, filtration, CPB equipment, patient safety
Morbidity and mortality associated with the accidental infusion of gaseous microemboli during cardiopulmonary bypass continues to be a concern, and although much has been done to advance the air handling capabilities of extracorporeal circuits, improvements in this area are still needed as the potential for microembolism persists alongside technological advances in perfusion system designs (1–7). More specifically, bubbles larger than the rated micron pores of arterial line filters have been detected beyond the filter outlet, highlighting an important deficiency in patient safety (8,9).
Using a numerical model to simulate trajectories of microbubbles through an arterial line filter, Fiore et al. (10) showed that although bubbles <100 μm were almost completely entrapped in the flow passing through the filter screen, a significant number of bubbles up to 1000 μm in size were also predicted to make contact with the filter material. The Fiore et al. study offers important insights into the microbubble size range that may be unable to reach the purge port by escaping the fluid flow and demonstrates the level of embolic stress load that may be placed on micropore screen filters. Although the numerical model overestimates the fluid viscosity normally seen during clinical perfusion and therefore the size range of microbubbles entrapped, the advent of warmer surgery with an emphasis on decreasing priming volumes and the use of higher hematocrit levels help bring outcomes of the Fiore et al. study closer to a reasonable estimation of microbubble capture within modern perfusion systems. Because the air handling performance of perfusion system components has been well studied (11–15), increased efforts to better understand inherent limitations of arterial line filters and how these insights may be used to improve the quality of perfusate returning to the patient are still needed.
A significant part of perfusion system performance in terms of its air handling ability is dependent on theories that relate among other things the blood flow rate, viscosity, pressure, temperature, and diffusion characteristics of gases. Understanding the physical relationships that connect these variables has helped guide improvements in perfusion systems and the development of foundations for clinical practice during cardiopulmonary bypass procedures. Central to this understanding are the effects of pressure on both dissolved and free gas forms in solution. According to Henry’s law, the amount of gas dissolved in a liquid is directly proportional to its partial pressure (16), but this relationship holds true only when temperature is constant. As the temperature of a liquid changes, the partial pressures exerted by gases dissolved in the liquid also vary as a result of changes in molecular motion. As the liquid’s temperature decreases, for example, a larger volume of gas must be dissolved in the liquid to exert the same partial pressure that was measured before the temperature drop; hence, gas solubility is said to increase. The opposite occurs as the temperature of the liquid increases and gas solubility is said to decrease. A good example of this in practice is the need to add CO2 while cooling during pH stat gas management.
Pressure is not only a principal factor in determining the rate and amount of gas that can dissolve, but it also influences the volume a free gas bubble occupies and the extent to which it is carried in a fluid flow. As a force, fluid pressure may counter best intentions in clinical practice causing an arterial line filter to fail, or it may be used as a method to enhance patient safety by promoting the reabsorption of free gases from the blood phase of a membrane oxygenator to the gas phase where it is more easily expelled. Boyle’s law states that when temperature remains constant, the volume of a confined gas varies inversely with its absolute pressure, explaining the compressible nature of gases. As pressure increases, volume decreases and vice versa. Boyle’s law can be described as:
where p is equal to the pressure of the system (atm), V is equal to the volume of the gas confined (L), and k is equal to the constant value representing the product of the volume and pressure in the system. At a constant temperature, Boyle’s law can also be used to determine the expected volume change for a given change in pressure and the other way around using:
where p1 · V1 represent the original pressure and volume of the system, and p2 · V2 are equal to the respective changes in pressure and volume.
The significance of the interconnections between these factors offers important insights into the clinical application of the pump oxygenator. In example, the relationship between pressure and the amount of gas dissolved in solution may be useful in providing an alternate conclusion to a recent study by Sleep et al. (17). High ventilation rates in the hypothermic and metabolically inactive circuit of the Sleep et al. study would help maximize the amount of gas dissolved at each temperature setting providing the substrate for potential bubble generation. Meanwhile, a constant pump flow with increasing viscosity at lower temperature would result in higher pressure drops across the resistive components in the circuit providing the physical means to pull any excess dissolved gases back out of solution. Although gas solubility increased as the temperature of the priming solution was decreased, increasing pressure drops across the oxygenator and arterial filter would aid the progressive expansion of existing microbubbles and allow any excess gases to re-emerge as the force maintaining their dissolved state diminished and a new equilibrium between pressure and the amount of gas in solution was reached. Although this may offer a plausible alternative explanation, the known theory it borrows from and the rational it lends to are both mimicked in the clinical setting. As the patient is cooled, the arterial pump flow and ventilation rate are lowered to match a decreasing metabolic demand while simultaneously limiting the pressure loss across the circuit that would naturally occur due to changes in viscosity.
To better understand how pressure affects the behavior of gases in a fluid flow and how these insights might link to possible improvements in microbubble filtration, two ex vivo test circuits were designed to assess changes in microbubble number and volume with changes in pressure. The test circuits were used to conduct two separate experiments with and without inline resistance. For the first experiment, a series of five trials was carried out to analyze changes in the quality and quantity of free gas forms along a 180-cm length of 3/8-inch polyvinyl chloride (PVC) tubing representing the connection between the filter outlet and arterial cannula. During the second experiment, which consisted of six trials, three inline resistors were added to the 180-cm length of tubing to evaluate and compare changes in microbubble number and volume across the generated pressure drops.
MATERIALS AND METHODS
Ex Vivo Test Circuit Design
The test circuits consisted of a Stockert SIII single roller-head pump (Stockert Instrument GmbH, Munich, Germany), two Gampt BCC200 Doppler ultrasound bubble counters calibrated with 3/8-inch probes (GAMPT mbH, Zappendorf, Germany), two Capiox RX25R filtered venous-cardiotomy reservoirs with sampling manifold (Terumo, Somerset, NJ), 1/2-inch PVC raceway tubing, 3/8-inch PVC tubing (total length 300 cm), three 1/4-inch PVC tubing resistor coils (total length 80 cm each), six 3/8-inch × 1/4-inch luered polycarbonate connectors, YSI 400 temperature probe, four pressure monitoring lines with three-way stopcocks, pressure isolator dome, a prebypass filter, and a 20-mL syringe.
The GAMPT BCC200 with BCView software Version 3.4 is a self-calibrating ultrasound Doppler system capable of measuring microbubbles between 5 and 500 μm in size and macrobubbles up to 10 mL in volume at flow rates between .2 and 10 L/min. Each BCC200 monitor comes equipped with two ultrasound probes color-coded for identification purposes.
Ex Vivo Test Circuit Setup: Experiment 1
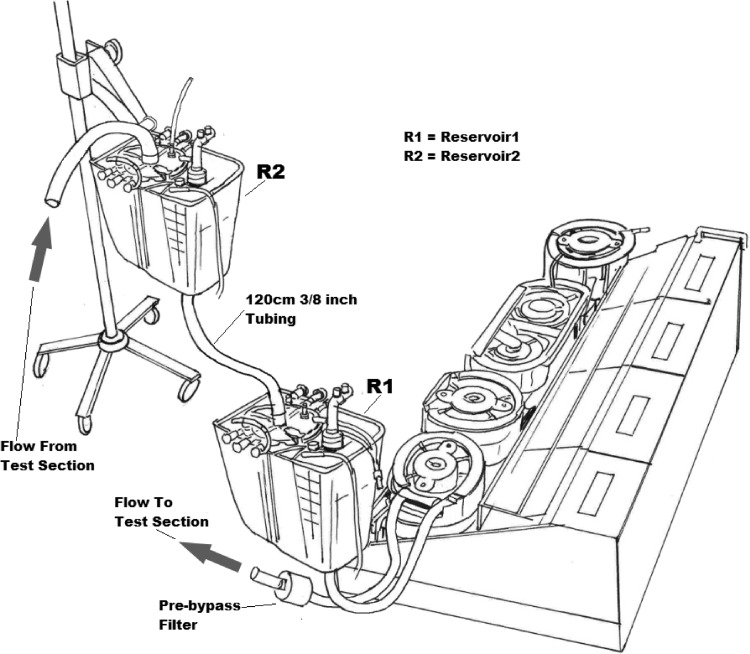
To limit the recirculation of microbubbles, a large volume test circuit with venous reservoir to cardiotomy reservoir connections was used. A single roller-head pump calibrated and occluded with 1/2-inch PVC raceway tubing was used to generate flow, whereas two Capiox RX25R filtered venous-cardiotomy reservoirs were used to recirculate the test circuits and act as degassing chambers. As depicted in Figures 1 and 2, inlet of the pump raceway tubing was connected to the venous outlet of reservoir R1. A prebypass filter joined the pump raceway outlet to a 180-cm length of 3/8-inch PVC tubing, which then terminated at the cardiotomy inlet port of reservoir R2. To complete the circuit, a 120-cm length of 3/8-inch PVC tubing connected the venous outlet of reservoir R2 to the cardiotomy inlet port of reservoir R1, while height differences between the two could be manipulated to facilitate gravity drainage from reservoir R2 to R1 during recirculation.
Figure 1.
Test circuit design showing flow direction to and from test section (test section not shown).
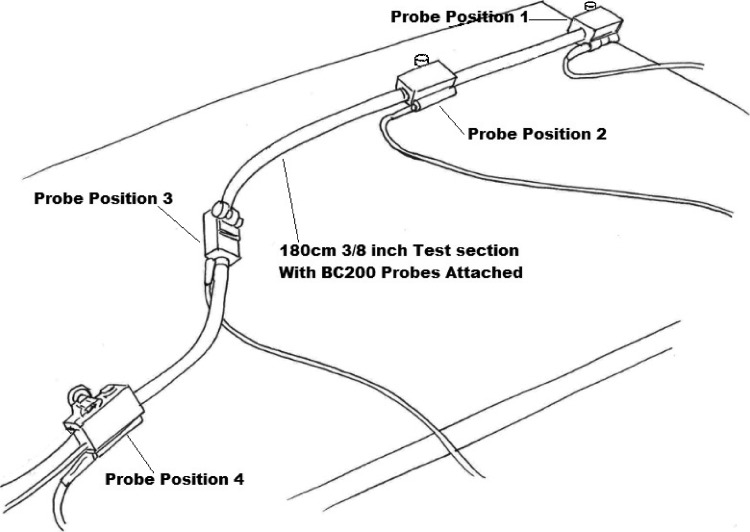
Figure 2.
Test section (180 cm) used for experiment 1 with BCC200 probes attached.
The BCC200 bubble counters were labeled “monitor 1” and “monitor 2” and the four ultrasound probes labeled to correspond with positions 1, 2, 3, and 4 were attached at equal distances from each other (35 cm apart) to the 180-cm length of tubing between the two reservoirs. The blue and red probes from monitor 1 occupied positions 1 and 2, whereas the blue and red probes from monitor 2 occupied positions 3 and 4, respectively. As shown in Figure 2, a smooth flat surface was used to minimize height differences along the test section to avoid any confounding effects that could result from differences in hydrostatic pressure between the four probe positions. Pressure loss in the test circuit was monitored from two luered ports, one just distal to the prebypass filter and the other just proximal to the cardiotomy inlet port of reservoir R2. Room air temperature measurements were taken from the venous inlet temperature monitoring port of reservoir R1.
The test circuit was primed with saline .9% solution (9.5 L) at room temperature (23.4°C) and circulated at 6 L/min until all four ultrasound Doppler probes were silent to confirm the absence of any circulating microbubbles. Both BCC200 bubble counters were set to monitor and record bubble sizes between 10 and 500 μm.
Conduct of Experiment 1
Once priming was complete, flow rate in the test circuit was maintained at 3.2 L/min and data collection with BCView software Version 3.4 was started before bubble generation to record a zero baseline at the start of each trial. Data collection continued for 3 minutes or until no bubble activity was observed after microbubble generation. Microbubble generation for measurement in the test circuit was achieved by rapping on the prebypass filter located proximally to the four ultrasound probes. The percussions were capable of causing the prebypass filter to release clusters of bubbles between 10 and 500 μm in size, which could easily be visualized during bubble generation because the measurements were taken in near real-time. A prebypass filter was selected as the source of microbubble generation for its apparent ease in producing bubble clusters in the targeted size range without saturating the probes or introducing over range gas volumes. The need to develop a method that could avoid introducing excessive bubble numbers and over range gas volumes was important to improve confidence in the accuracy of the measured volume. During bubble generation, the number of bubbles measured at probe position 1 for each trial was monitored using BCView software Version 3.4. In the first experiment, bubble generation continued until total bubble counts between 250 and 600 were collected at probe position 1. For the first four trials of the second experiment, bubble counts between 600 and 800 were collected. During trials 5 and 6 of the second experiment, total bubble counts of 1800 and 2200 were obtained.
After microbubble generation and data collection, the test circuit was recirculated without disturbing the prebypass filter until the system was completely debubbled before the start of the next trial. The process of microbubble generation, data collection, and debubbling was repeated five times to establish average test values for statistical analysis. When the prebypass filter became significantly degassed to prevent sufficient generation of microbubble counts, a 20-mL syringe was used to inject bolus air into the test circuit. After bolus air injections, the BCC200 monitors were used to confirm that the fluid stream was free of circulating bubbles before the start of the next trial.
Ex Vivo Test Circuit Setup: Experiment 2
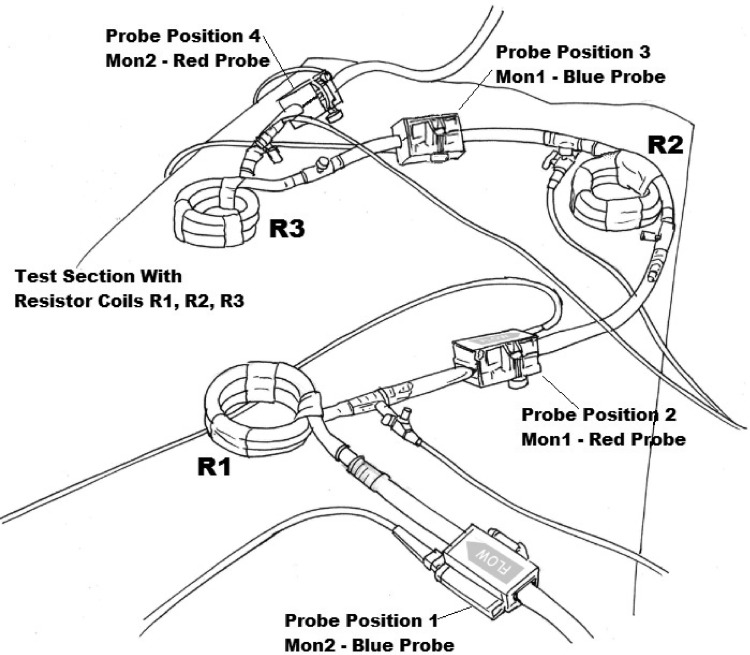
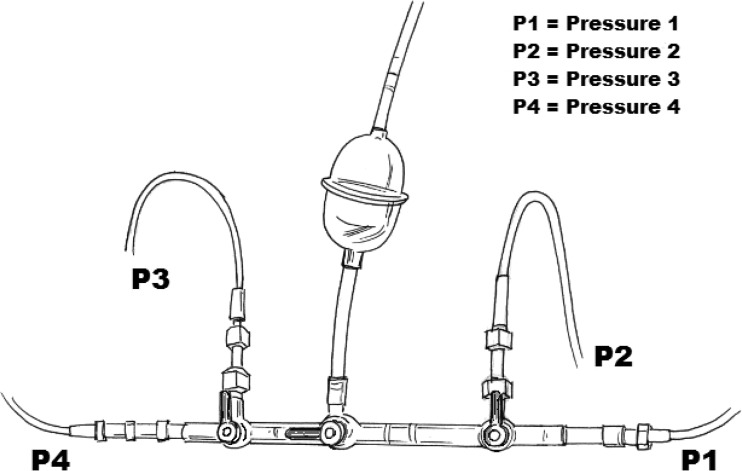
After the completion of experiment 1, the four ultrasound probes were removed and the 180-cm length of 3/8-inch tubing was modified to include three resistor coils each made from 80 cm of 1/4-inch PVC tubing. The resistor coils were placed 35 cm apart and joined to the original test circuit using six 3/8-inch × 1/4-inch luered polycarbonate connectors. The four ultrasound probes were then reattached at equal distances from each other (approximately 45 cm apart) before, between, and after the three resistor-coils as illustrated in Figure 3. Probe arrangement was also reorganized so that the blue probe of monitor 1 replaced the blue probe of monitor 2. The blue and red probe of monitor 2 now occupied positions 1 and 4, whereas the red and blue probes of monitor 1 occupied positions 2 and 3, respectively. In addition to the pressure monitoring site located immediately after the prebypass filter, three pressure monitoring lines were added to luered ports just distal to each resistor coil. The four monitoring lines were then connected and primed using a Capiox sampling manifold so that pressure readings from the four sites could be obtained using a single pressure transducer as shown in Figure 4. All other considerations for the setup of experiment 2 were duplicated as described for experiment 1.
Figure 3.
Test section (180 cm) with resistor coils used for experiment 2 showingBCC200 probe positions.
Figure 4.
Pressure monitoring system, showing setup to record from pressure 1 (P1).
Conduct of Experiment 2
Once priming was complete, flow rate in the modified test circuit was maintained at 3.2 L/min for each of the six trials performed. All other considerations for the conduct of experiment 2 were duplicated as described for experiment 1.
Data Analysis
PASW Statistics Version 18 (SPSS Inc., 2009, Chicago, IL) was used to generate descriptive and inferential statistics. Descriptive statistics were provided to summarize the means and standard deviations for microbubble number and volume in the two test circuits. Mean scores for microbubble number and volume in relation to probe position were also plotted. Dependent t tests (two-tailed) were used to compare changes in microbubble number and volume using the measurements taken from probe positions 1 and 4 as the paired sample for each experiment. Results were considered statistically significant at p < .05.
RESULTS
There were 44 paired measurements taken for microbubble count and volume over the course of both experiments in this study. The total microbubble number and volume measured were used to generate the descriptive statistics shown in Table 1. Pressure loss along the 180-cm test section with probes attached was 24 mmHg for the first experiment and 192 mmHg for the second experiment. Peak pressures were 27 mmHg in experiment 1 and 201 mmHg in experiment 2 with consecutive pressure readings of 133, 68, and 9 mmHg at the resistor coil outlets.
Table 1.
Mean microbubble number and volume ± SD in each experiment.
| Experiment | Mean Microbubble Number | Minimum/Maximum Score | Mean Microbubble Volume (μL) | Minimum/Maximum Score | |
|---|---|---|---|---|---|
| 1 | 370.50 ± 126.086 | 215/646 | .290600 ± .0975524 | .1360/.4360 | |
| 2 | 1017.46 ± 588.690 | 445/2279 | .921083 ± .7171217 | .1920/2.5900 |
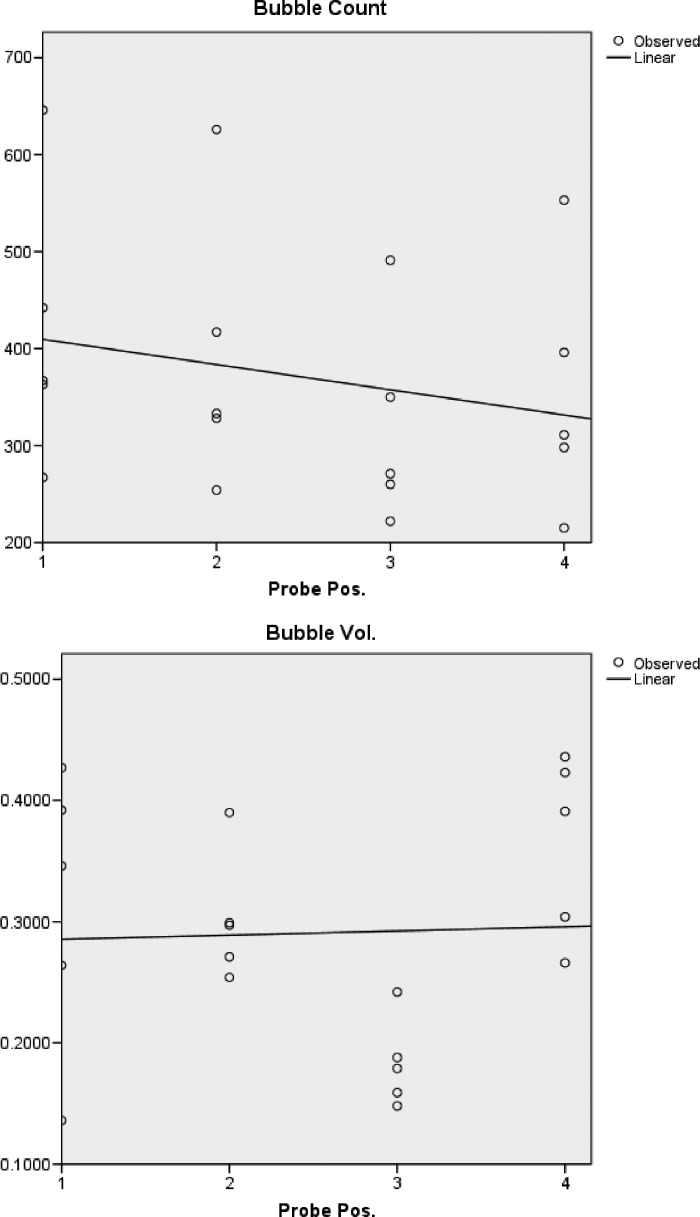
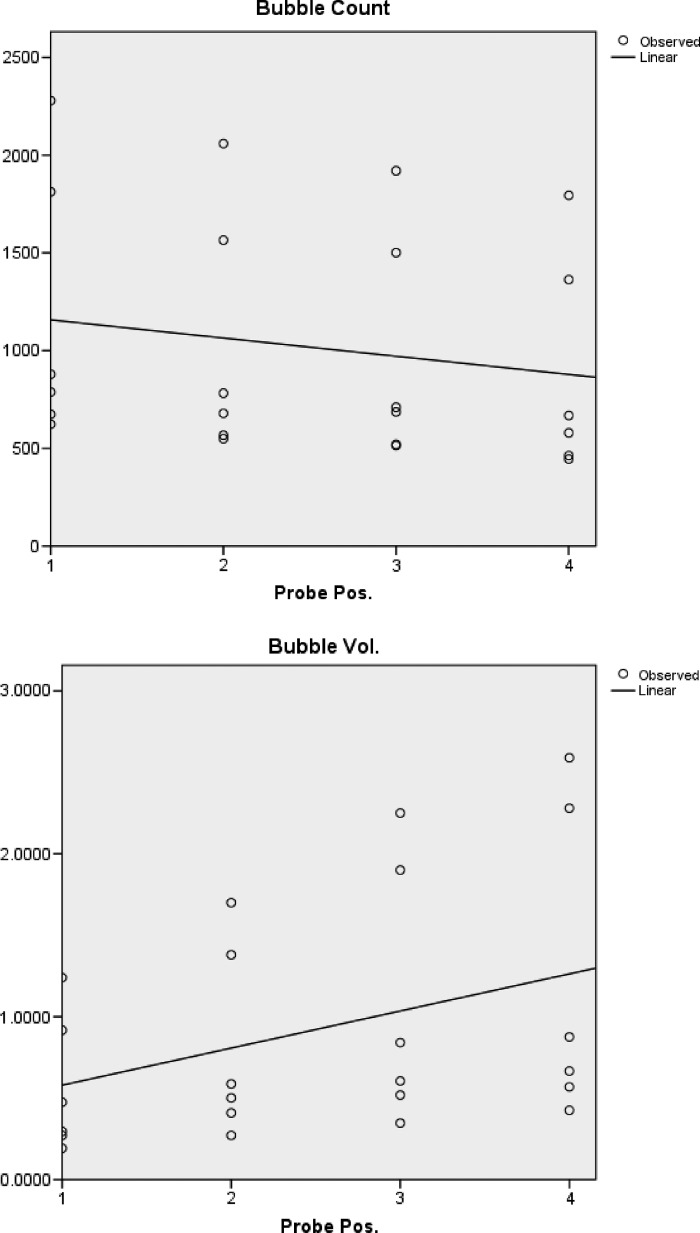
Although changes in microbubble count between probe positions 1 and 4 in the first experiment were significant (t [4] = 7.29, p < .05), changes in microbubble volume were not (Table 2). This contrasts significant findings in the second experiment when higher pressure drops were used. In experiment 2, changes in both microbubble number (t [5] = 5.116, p < .05) and volume (t [5] = −2.868, p < .05) reached statistically significant values shown in Table 3. This is further appreciated when comparing the linear regression lines plotted in Figures 5 and 6. As illustrated, both experiments showed a similar reduction in microbubble number, whereas microbubble volume in the first experiment remained relatively constant with lower pressure loss in comparison to the second experiment in which volume increased in a linear fashion across the generated pressure drops.
Table 2.
Mean change in microbubble number and volume between probe positions 1 and 4 during bubble generation and postbubble generation periods for experiment 1.
| Experiment 1 | Probe Position 1 | Probe Position 4 | Significance (two-tailed) | |
|---|---|---|---|---|
| Total mean number | 417.00 | 354.60 | .002 | |
| Total mean volume (μL) | .313 | .364 | .069 | |
| Mean bubble generation period (seconds) | 33.8 | |||
| Mean number | 313.40 | 271.8 | .001 | |
| Mean volume (μL) | .257 | .303 | .120 | |
| Mean postbubble generation period (seconds) | 121.2 | |||
| Mean number | 103.40 | 82.8 | .025 | |
| Mean volume (μL) | .056 | .060 | .641 |
Table 3.
Mean change in microbubble number and volume between probe positions 1 and 4 during bubble generation and postbubble generation periods for experiment 2.
| Experiment 2 | Probe Position 1 | Probe Position 4 | Significance (two-tailed) | |
|---|---|---|---|---|
| Total mean number | 1175.83 | 885.33 | .004 | |
| Total mean volume (μL) | .565 | 1.234 | .035 | |
| Mean bubble Generation period (seconds) | 32.7 | |||
| Mean number | 1150 | 877.50 | .004 | |
| Mean volume (μL) | .559 | 1.097 | .012 | |
| Mean postbubble Generation period (seconds) | 30.3 | |||
| Mean number | 25.83 | 7.83 | .076 | |
| Mean volume (μL) | .006 | .137 | .324 |
Figure 5.
Mean microbubble number and volume in microliters measured at each probe position in experiment 1.
Figure 6.
Mean microbubble number and volume in microliters measured at each probe position in experiment 2.
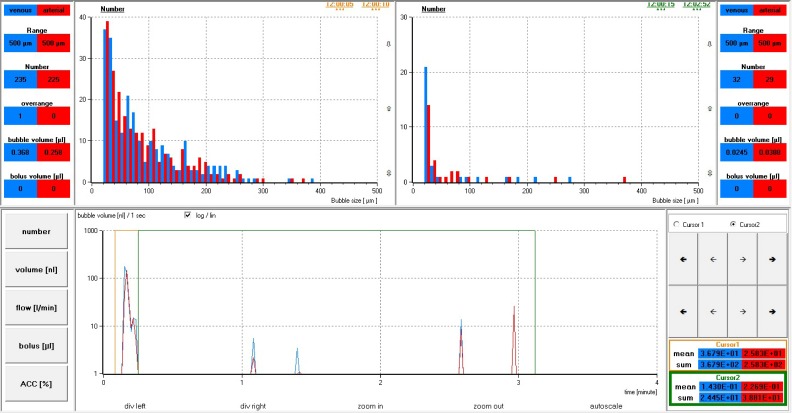
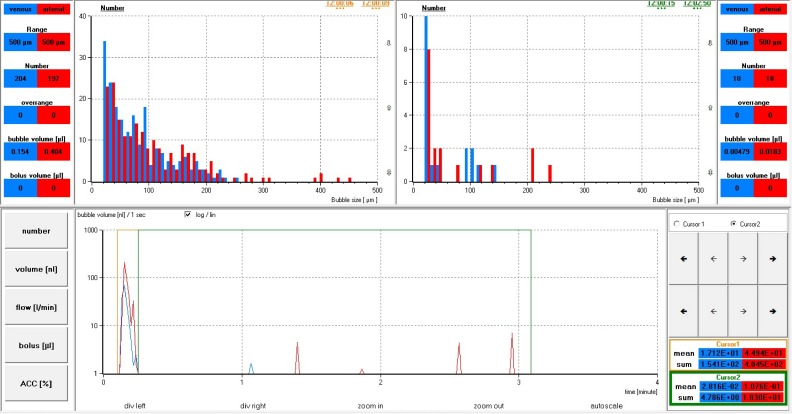
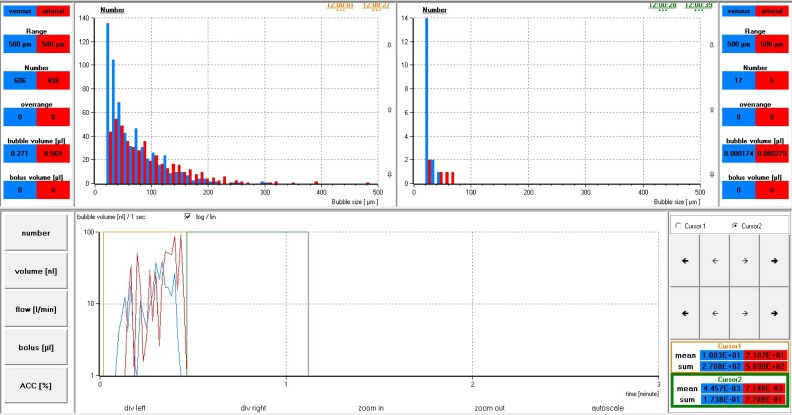
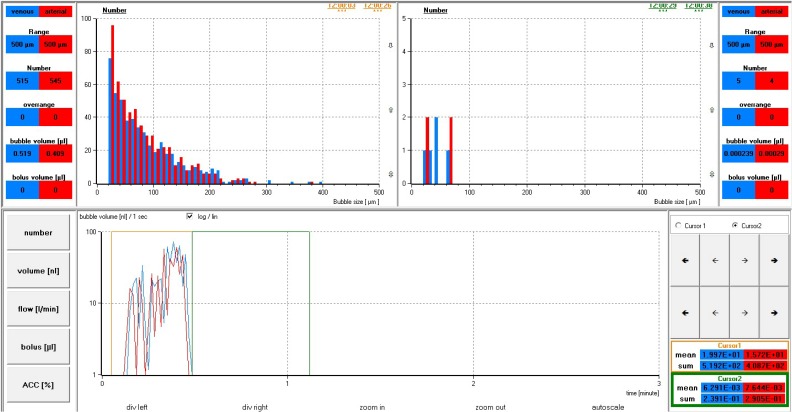
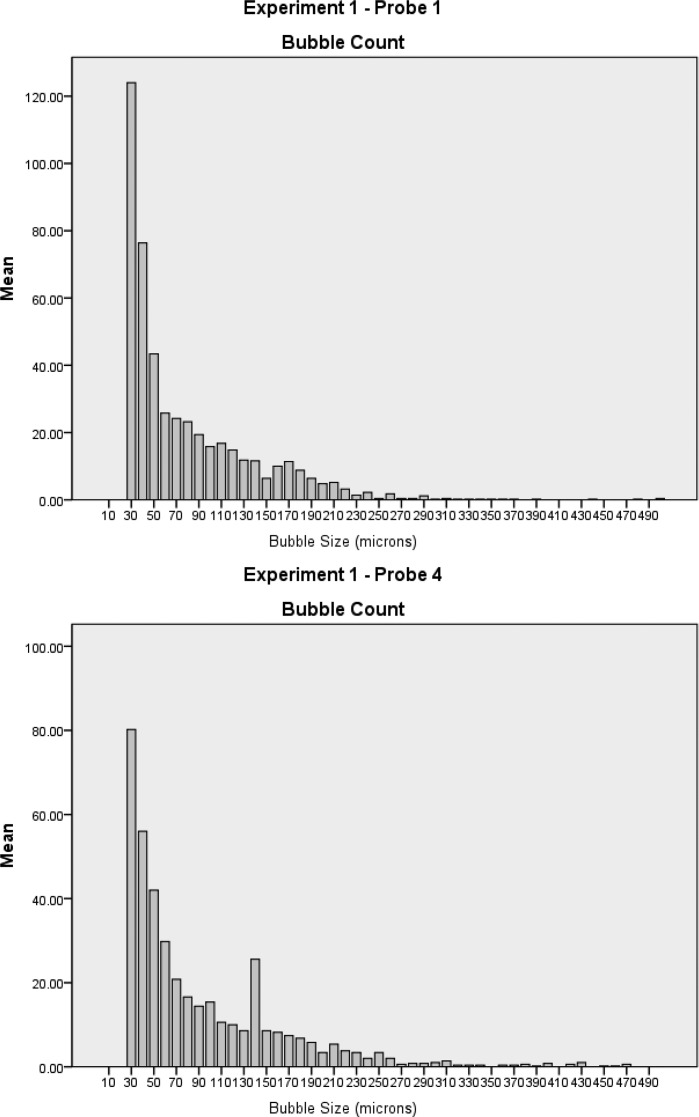
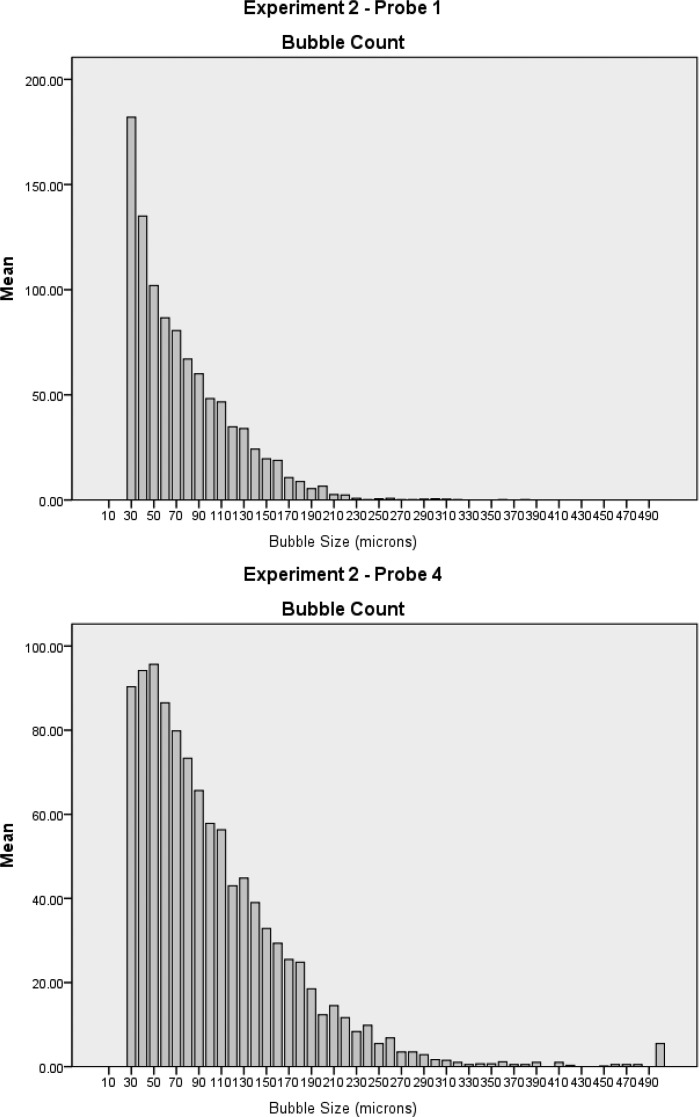
Table 4 provides microbubble measurements including over range counts and bolus volumes at each probe position for both experiments, whereas Figures 7 through 10 are used as examples to compare changes in microbubble number and volume among the four probe positions over time. Area under the gold cursor in each figure with corresponding histogram represents the period of bubble generation. Area under the green cursor with corresponding histogram represents the time taken for each test circuit to naturally degas. As demonstrated in Figures 7 through 10, bubble clusters with a volume greater than 1 nL/sec appear on the timeline as spikes under each cursor, whereas total individual bubble counts are included in the respective histograms for each time period. Figures 11 and 12 represent cumulative bubble counts by micron size between probe positions 1 and 4 in both experiments.
Table 4.
Microbubble measurements including over range and bolus volume counts in each experiment.
| Experiment 1 |
Experiment 2 |
|||||||
|---|---|---|---|---|---|---|---|---|
| Probe Position 1 | Probe Position 2 | Probe Position 3 | Probe Position 4 | Probe Position 1 | Probe Position 2 | Probe Position 3 | Probe Position 4 | |
| Trial 1 | ||||||||
| MB number | 267 | 254 | 222 | 215 | 623 | 549 | 520 | 463 |
| MB volume (mL) | .392 | .297 | .159 | .423 | .271 | .409 | .519 | .569 |
| Over range (>500 μm) | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Bolus volume (μL) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Trial 2 | ||||||||
| MB number | 367 | 328 | 260 | 298 | 788 | 679 | 686 | 579 |
| MB volume (μL) | .136 | .254 | .179 | .266 | .295 | .501 | .605 | .666 |
| Over range (>500 μm) | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| Bolus volume (μL) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Trial 3 | ||||||||
| MB number | 646 | 626 | 491 | 553 | 879 | 782 | 710 | 668 |
| MB volume (μL) | .346 | .39 | .242 | .391 | .475 | .587 | .841 | .876 |
| Over range (>500 μm) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Bolus volume (μL) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Trial 4 | ||||||||
| MB number | 442 | 417 | 350 | 396 | 674 | 567 | 514 | 445 |
| MB volume (μL) | .427 | .299 | .188 | .436 | .192 | .271 | .347 | .425 |
| Over range (μ500 μm) | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Bolus volume (μL) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Trial 5 | ||||||||
| MB number | 363 | 333 | 271 | 311 | 1812 | 1565 | 1501 | 1363 |
| MB volume (μL) | .264 | .271 | .148 | .304 | .917 | 1.38 | 1.9 | 2.59 |
| Over range (>500 μm) | 0 | 0 | 0 | 0 | 0 | 3 | 8 | 21 |
| Bolus volume (μL) | 0 | 0 | 0 | 0 | 0 | 6 | 9 | 17 |
| Trial 6 | ||||||||
| MB number | — | — | — | — | 2279 | 2059 | 1920 | 1794 |
| MB volume (μL) | — | — | — | — | 1.24 | 1.7 | 2.25 | 2.28 |
| Over range (>500 μm) | — | — | — | — | 1 | 6 | 11 | 12 |
| Bolus volume (μL) | — | — | — | — | 2 | 6 | 16 | 3 |
MB, microbubble.
Figure 7.
Experiment 1 results for monitor 1 probe positions 1 (blue) and 2 (red). Bottom portion showing volume transfer (nL /sec) during bubble generation (gold cursor) and degassing periods (green cursor). Top portion showing histograms for bubble counts corresponding with bubble generation (gold) and degassing periods (green).
Figure 8.
Experiment 1 results for monitor 2 probe positions 3 (blue) and 4 (red). Bottom portion showing volume transfer (nL /sec) during bubble generation (gold cursor) and degassing periods (green cursor). Top portion showing histograms for bubble counts corresponding with bubble generation (gold) for degassing periods (green).
Figure 9.
Experiment 2 results for monitor 2 probe positions 1 (blue) and 4 (red). Bottom portion showing volume transfer (nL /sec) during bubble generation (gold cursor) and degassing periods (green cursor). Top portion showing histograms for bubble counts corresponding with bubble generation (gold) and degassing periods (green).
Figure 10.
Experiment 2 results for monitor 1 probe positions 2 (red) and 3 (blue). Bottom portion showing volume transfer (nL /sec) during bubble generation (gold cursor) and degassing periods (green cursor). Top portion showing histograms for bubble counts corresponding with bubble generation (gold) and degassing periods (green).
Figure 11.
Cumulative bubble counts by micron size in experiment 1 between probe positions 1 and 4.
Figure 12.
Cumulative bubble counts by micron size in experiment 2 between probe positions 1 and 4.
Of notable interest is the effect pressure had on the rate of volume transferred, the time required for degassing, and the microbubble number and size range measured in the postbubble generation period. Although volume transfer in the first experiment exceeded 100 nL/sec during bubble generation with spikes greater than 5 nL/sec in the postbubble generation period (Figures 7 and 8), peak transfer for experiment 2 remained below 100 nL/sec and less than 1 nL/sec in the postbubble generation period (Figures 9 and 10). Higher peak pressures showed greater gas compression with a decrease in volume transfer in both time periods for the second experiment as compared with the first experiment.
In addition, higher rates of pressure loss were associated with a decrease in the volume and number of microbubbles measured in the postbubble generation period. The total mean volume and number measured in experiment 2 at probe position 1 during bubble generation reached 559 nL (n = 1150) compared with 257 nL (n = 313.4) for experiment 1 (Tables 2 and 3). This contrasts with the mean volume and number measured in both experiments at probe position 1 in the postbubble generation period. In experiment 1, 56 nL (n = 103.4) were measured as compared with 6 nL (n = 25.83) in experiment 2, representing a decrease in measured volume between the two time periods by approximately two orders of magnitude in the circuit with inline resistance. Whereas the microbubble load generated was greater in experiment 2, the length of time required to degas in the postbubble generation period as well as the microbubble number and size range measured were all greater in experiment 1 (Tables 2 and 3).
DISCUSSION
As its primary focus, this study was used to model the effects of pressure on gases contained in a fluid flow to gain insight on possible improvements in microbubble filtration. At the outset of this study, it was uncertain how the resistor coils would affect the quality and quantity of microbubbles produced. It was questioned whether the smaller tubing size of the coils would force more bubbles to contact each other and affect the amount of coalescence taking place, but results for experiment 1 without resistor coils demonstrated a similar effect. A higher number of smaller bubbles measured at probe position 1 as compared with a lower number of larger bubbles having a similar volume measured at probe position 4 suggest coalescence as a dominant behavior in experiment 1. The fact that this effect was reproduced in all five trials of the first experiment further supports coalescence as a principle behavior.
The phenomenon of coalescence also appeared to increase further during the second experiment when larger microbubble counts were purposely generated. Table 4 shows an exponential increase in the number of detected free gas volumes larger than 500 μm for trials 5 and 6 as compared with the first four trials of experiment 2 when lower bubble counts were produced. As the number of microbubbles in a cluster increases, it seems logical that the chance of coalescence between them would also increase. The significance of coalescence and the role it may play in the development of larger bubbles forms an important finding of this study, indicating that when left relatively undisturbed in a fluid flow, microbubbles in close proximity to each other have a tendency to combine and increase in size. This contrasts with what we know to happen when the fluid flow is disrupted such as when it passes through an arterial line filter and large numbers of smaller bubbles are produced (13).
Principles surrounding laminar flow can be used to help explain in part the assumed behavior of coalescence observed in this study. Laminar flow in a tube consists of several thin concentric layers (lamina) or streamlines that force the particles they contain to travel in a straight path parallel to the wall of the tubing. A velocity gradient exists between each lamina so that fluid flow is fastest in the center streamline and then steadily decreases until it approaches zero where the outermost lamina comes in contact with the tubing wall. Within a laminar flow, the buoyant force of an air bubble works in opposition to the drag force generated by the flow passing over the spherical surfaces of the bubble. As a bubble increases in size, a higher drag force is required to counter its increasing buoyancy to maintain the bubble within the fluid stream. At lower flow velocities, buoyancy can overcome the drag force, causing the bubble to rise up and escape from the center streamline. However, as the speed and/or viscosity of the fluid flow increases, the drag force becomes more dominant and capable of entrapping even larger bubbles. At the same time, fluid pressures generated by the resistance to flow also compress the bubble, decreasing its size and the drag force required to maintain its position in the fluid stream. Combined, these principles encourage the phenomenon of coalescence as entrapped clusters of bubbles too small to escape the fluid flow may accumulate in close proximity to each other.
Microbubble volume in this study was also shown to increase disproportionately across the test circuits as microbubble number decreased, indicating another effect in addition to coalescence. Considering the absolute pressure drop observed in experiment 2 (25%) in comparison to the observed volume change (>200%), it becomes apparent that an additional source of gas was being added to the volume measured. Because there was no other external gas source, the additional volume would have to come from gases dissolved in solution. Volume expansion in the test circuit can arise from pressure drops created by the resistor coils themselves or by localized pressure drops that result from turbulences in the fluid flow. This effect is also present in experiment 1 but to a lesser degree. The Gampt monitor uses probes that slightly compress the tubing when attached and may cause turbulences distal to each probe, especially at the lower fluid viscosities used in this study. Although there was an absolute pressure drop of 3% between probes 1 and 4 in the first experiment, the volume expansion measured at probe 4 was approximately 15% greater than the volume measured at probe 1 in the bubble generation period and 7% in the postbubble generation period. The excess increase in volume suggests the presence of localized pressure drops, accounting for approximately 12% and 4% of the observed volume expansion in the respective time periods while using a 3/8-inch .9% saline flow of 3.2 LPM.
Although study results showed that volume transfer across the test section was affected by peak pressure, an increased pressure loss was associated with a decrease in the time required for degassing as well as the microbubble number and size range measured in the postbubble generation period. It is suggested that higher rates of decompression across the test section in experiment 2 caused a rapid removal of gases dissolved in solution, allowing the generated free gas forms to absorb and coalesce with other circulating microbubbles to effectively eliminate them from the fluid flow. While circulation through the large volume test circuit allowed the generated free gas forms to escape, a steady decrease in the amount of gases dissolved in solution helped to stabilize the fluid flow across each pressure drop, prompting a decrease in the amount of microbubble activity measured in the postbubble generation period. In relation to methods used for microbubble filtration during extracorporeal circulation (ECC), these findings support the use and possible benefit of a sequential filter arrangement as previously reported by Herbst et al. (16). It is proposed that the new design makes it possible for pressure changes across the first micropore screen to aid the development of larger free gas forms, while a second screen placed in series could be used to capture the larger bubbles and provide an opportunity to direct them away from the fluid stream returning to the patient. Although this demonstrates one example of how pressure might be used to affect the micropore filtration of gas bubbles, the model used in this study was not intended to confirm additional possibilities that could be created by varying the effective micropore size between sequentially placed filter screens.
Summarizing the events that may contribute to the microbubbles that pass through an arterial filter toward the patient, it could be explained that flow at the filter inlet is disrupted as mass deceleration across the larger volume filter housing occurs. Bubbles large enough to overcome the drag force acting on them rise up and escape through an appropriately located purge port, whereas smaller bubbles maintained in the flow impact on the filter screen and are either held in place as an available source for dissolved gases, pass through undisturbed or are broken down into smaller particles by the micropore interface. Although a pressure drop at the distal surface of the filter screen promotes volume expansion, clusters of microbubbles are also prone to coalesce and increase in size as flow velocity is re-established in the fluid steam returning to the patient. Gases contained in the flow may also undergo further volume expansion as they traverse another pressure drop at the arterial cannula outlet. The relevance of this may deepen an appreciation for the extracorporeal circuit as a source of free gas transfer to the patient and helps direct attention to the realities of practice for the clinical perfusionist and the critical issues being faced (18). Although blood sampling and drug administrations are necessary to maintain the patient on ECC, current limitations in arterial line filters make it difficult to guarantee complete safety during these maneuvers and therefore warrant increased attention to perfusionist technique (9,15).
At the heart of this enquiry are questions concerning the behavior of gases in solution and how this might contribute to deficiencies in current filter designs. An important question in this regard is why microbubbles larger then the rated micron pores of arterial line filters are being detected distal to the filter’s outlet? With respect to the outcomes of this study and the relevant theory previously described (16), it is reasonable to assume that in addition to the free gas transfer that occurs during instances when the bubble point pressure of a wetted pore is exceeded, larger bubbles may appear beyond the filter screen as a result of pressure drops that allow dissolved gases to re-emerge and coalesce with other existing free gas forms. As well, microbubbles exposed to a higher prescreen pressure may be sufficiently compressed to pass through a wetted pore undisturbed and then expand according to the pressure drop at the pores distal surface. It is also possible that larger bubbles impacting on the filter material break apart only to reform again in the fluid stream returning to the patient. For completeness, increasing temperatures and dissolved gas concentrations may also theoretically precipitate an increase in microbubble formation, especially in areas of lower pressure where the pressure drop serves as an additive force to augment the effects of decreasing gas solubility. In reality, it is more likely that at different time points, various combinations of these mechanisms contribute to the larger bubbles that can culminate downstream from the arterial filter outlet.
CONCLUSION
The behaviors of gas in solution are complex and the ways this might contribute to deficiencies in current methods for microbubble filtration are multifaceted. Although the quality and quantity of free gases in solution can readily change according to surrounding conditions, the filter material used for its removal incorporates a static position with fixed dimensions that is unable to cope effectively with the dynamics of microbubbles in a fluid flow. Gases may be moved in and out of solution and the free forms they derive can coalesce to increase in volume and then break down again into numerous particles of varying size. What is needed to improve this situation is a method that can disrupt the changing nature of gases. With its focus on the effects of pressure, this article raises a relevant question: can pressure at the micron pore interface be manipulated to improve microbubble filtration? The present research proposes that pressure as a force can be used to disrupt the changing nature of gases in a fluid flow and may provide a method to improve microbubble filtration during ECC. Further research with new arterial line filter designs that are able to take advantage of this method are needed to confirm this hypothesis.
ACKNOWLEDGMENTS
I thank Robert Klaua and Grit Oblonczek at GAMPT mbH for their kind assistance and advice in troubleshooting the integration and use of the BCC200 monitors for this research.
REFERENCES
- 1.Guan Y, Su X, McCoach R, Wise R, Kunselman A, Undar A.. Evaluation of Quadrox-i® adult hollow fiber oxygenator with integrated arterial filter. J Extra Corpor Technol. 2010;42:134–138. [PMC free article] [PubMed] [Google Scholar]
- 2.Mathis RK, Lin J, Dogal NM, et al. Evaluation of four pediatric cardiopulmonary bypass circuits in terms of perfusion quality and capturing gaseous microemboli. Perfusion. 2012;27:470–479. [DOI] [PubMed] [Google Scholar]
- 3.Lin J, Dogal NM, Mathis RK, Qiu F, Kunselman A, Ündar A.. Evaluation of Quadrox-i and Capiox FX neonatal oxygenators with integrated arterial filters in eliminating gaseous microemboli and retaining hemodynamic properties during simulated cardiopulmonary bypass. Perfusion. 2012;27:235–243. [DOI] [PubMed] [Google Scholar]
- 4.Salavitabar A, Qiu F, Kunselman A, Ündar A.. Evaluation of the Quadrox-I neonatal oxygenator with an integrated arterial filter. Perfusion. 2010;25:409–415. [DOI] [PubMed] [Google Scholar]
- 5.Qiu F, Peng S, Kunselman A, Ündar A.. Evaluation of Capiox FX05 oxygenator with an integrated arterial filter on trapping gaseous microemboli and pressure drop with open and closed purge line. Artif Organs. 2010;34:1053–1057. [DOI] [PubMed] [Google Scholar]
- 6.Gomez D, Preston TJ, Olshove VF, Phillips AB, Galantowicz ME.. Evaluation of air handling in a new generation neonatal oxygenator with integral arterial filter. Perfusion. 2009;24:107–112. [DOI] [PubMed] [Google Scholar]
- 7.Perthel M, Kseibi S, Sagebiel F, Alken A, Laas J.. Comparison of conventional extracorporeal circulation and minimal extracorporeal circulation with respect to microbubbles and microembolic signals. Perfusion. 2005;20:329–333. [DOI] [PubMed] [Google Scholar]
- 8.Liu S, Newland RF, Tully PJ, Tuble SC, Baker RA.. In vitro evaluation of gaseous microemboli handling of cardiopulmonary bypass circuits with and without integrated arterial line filters. J Extra Corpor Technol. 2011;43:107–114. [PMC free article] [PubMed] [Google Scholar]
- 9.Lynch JE, Wells C, Akers T, et al. Monitoring microemboli during cardiopulmonary bypass with the EDAC® quantifier. J Extra Corpor Technol. 2010;42:212–218. [PMC free article] [PubMed] [Google Scholar]
- 10.Fiore GB, Morbiducci U, Ponzini R, Redaelli A.. Bubble tracking through computational fluid dynamics in arterial line filters for cardiopulmonary bypass. ASAIO J. 2009;55:438–444. [DOI] [PubMed] [Google Scholar]
- 11.Jones TJ, Deal DD, Vernon JC, Blackburn N, Stump DA.. How effective are cardiopulmonary bypass circuits at removing gaseous microemboli? J Extra Corpor Technol. 2002;34:34–39. [PubMed] [Google Scholar]
- 12.Dickinson TA, Riley JB, Crowley JC, Zabetakis PM.. In vitro evaluation of the air separation ability of four cardiovascular manufacturer extracorporeal circuit designs. J Extra Corpor Technol. 2006;38:206–213. [PMC free article] [PubMed] [Google Scholar]
- 13.Myers GJ, Voorhees C, Haynes R, Eke B.. Post-arterial filter gaseous microemboli activity of five integral cardiotomy reservoirs during venting: An in vitro study. J Extra Corpor Technol. 2009;41:20–27. [PMC free article] [PubMed] [Google Scholar]
- 14.Riley JB.. Arterial line filters rankedfor gaseousmicro-emboli separation performance: An in vitro study. J Extra Corpor Technol. 2008;40:21–26. [PMC free article] [PubMed] [Google Scholar]
- 15.Lynch JE, Riley JB.. Microemboli detection on extracorporeal bypass circuits. Perfusion. 2008;23:23–32. [DOI] [PubMed] [Google Scholar]
- 16.Herbst DP, Najm HK.. Development of a new arterial-line filter design using computational fluid dynamics analysis. J Extra Corpor Technol. 2012;44:140–145. [PMC free article] [PubMed] [Google Scholar]
- 17.Sleep J, Syhre I, Evans E.. Blood temperature management and gaseous microemboli creation: An in-vitro analysis. J Extra Corpor Technol. 2010;42:219–222. [PMC free article] [PubMed] [Google Scholar]
- 18.Taylor RL, Borger MA, Weisel RD, et al. Cerebral microemboli during cardiopulmonary bypass: Increased emboli during perfusionist intervention. Ann Thorac Surg. 1999;68:89–93. [DOI] [PubMed] [Google Scholar]