Abstract
Objectives
The purpose of this study was to compare the microbial and clinical effects of mechanical debridement (MD) alone or in combination with the application of enamel matrix derivative (EMD) and sustained-release micro-spherical minocycline (MSM) for treatment of peri-implant mucosal infl ammation (PIMI).
Materials and Methods
Subjects with at least one implant with PIMI were included and divided into control and two different test groups. In all three groups, MD was performed. In the MSM group, following MD, MSM was placed subgingivally around the implants. In the EMD group, after MD, EMD was placed in the sulcus around the implants. Sampling of peri-implant crevicular fl uid for microbial analysis with real-time polymerase chain reaction and recording of probing depth (PD) and bleeding on probing (BOP) were performed prior to as well as two weeks and three months after treatment. Median values and interquartile range were estimated for each variable during the various assessment intervals of the study.
Results
In all groups, at two weeks and three months, the counts of Porphyromonas gingivalis decreased significantly compared to baseline. Levels of P. gingivalis were significantly reduced in MSM (P<0.001) and EMD (P=0.026) groups compared to the control group. Also, clinical parameters improved significantly at two weeks and three months. Reduction of PD was significant in MSM (P<0.001) and EMD (P<0.001) groups. The decrease in BOP in the MSM, EMD, and control groups was 60%, 50%, and 20%, respectively.
Conclusion
The use of MSM and EMD can be an adjunctive treatment for management of PIMI and improves clinical parameters and reduces P. gingivalis burden three months after treatment.
Keywords: Debridement, Infl ammation, Minocycline, Real-time polymerase chain reaction, Peri-implantitis
I. Introduction
One of the most common implant complications is peri-implant mucosal inflammation (PIMI), which is defined as inflammation resulting in resorption of alveolar bone1. It is the etiologic agent for the failure of 10% to 50% of implants at least one year after loading and is considered a major clinical problem2. In a systematic review, the prevalence of peri-implantitis was reported to be 8.6% after five years3. The clinical manifestation of peri-implantitis includes bleeding on probing (BOP), loss of supporting bone, increased peri-implant probing depth (PD), and suppuration4. Poor oral hygiene, history of periodontitis, and smoking are risk factors for peri-implantitis5. However, there is consensus regarding the infectious nature of peri-implantitis2. From a therapeutic point of view, decontamination of the implant surface and resolution of the infl ammatory process are the principal aims in the treatment of peri-implantitis6. A study by Loe et al.7 showed a similar cause-and-effect relationship between bacterial accumulation and incidence of peri-implant mucositis and gingivitis, and the therapeutic aims of peri-implantitis are based on evidence assimilated from the treatment of periodontitis. Implant screw design, in conjunction with surface modifications, has facilitated biofilm formation in cases of exposure to the oral cavity. Therefore, surface debridement is a major component of peri-implantitis treatment. However, a decrease in bacterial load to a level that allows healing to occur is difficult using only mechanical methods. As a result, adjunctive treatments, including use of antibiotics, antiseptics, and laser, have been used to improve non-surgical treatment8. Local debridement in conjunction with the use of systemic amoxicillin and metronidazole has resulted in the resolution of peri-implantitis lesions in dogs9. Slow-released antibiotics for the treatment of periodontal infections can also be useful for the treatment of peri-implantitis. Such devices consistently release high doses of antimicrobial agents into the sites in question and destroy bacterial biofilms that were not eliminated by mechanical debridement (MD). Local treatment of peri-implantitis with tetracycline fibers suppresses pathogens such as Actinobacillus actinomycetemcomitans, Prevotella intermedia, Porphyromonas gingivalis, and Tannerella forsythia up to 12 months after treatment10. In addition, there is clinical evidence regarding a gain in probing attachment level and a decrease in PD subsequent to the use of biodegradable sustained-release devices after initial treatment of peri-implantitis11. In a multi-center study, clinical efficacy in the management of periodontitis has been shown with adjunct administration of micro-spherical minocycline (MSM) in periodontal pockets of teeth12. Additional evidence supporting the efficacy of minocycline as an adjunct local antibiotic has been shown with the placement of minocycline-loaded strips in periodontal pockets13. Another treatment modality is the use of enamel matrix derivative (EMD). The results of recent human and animal studies on periodontal regeneration have shown that use of EMD on an already debrided and prepared root surface predictably leads to the formation of root cementum and alveolar bone14. The results of a study on dogs showed that EMD has a positive effect on bone regeneration after guided bone regeneration around the implants15. It should be noted that an EMD-containing solution (propylene glycol alginate or PGA) has a significant antimicrobial effect on periodontal pathogens16. In a clinical study, use of EMD in non-surgical procedures along with scaling and root planing (SRP) in moderate-to-severe periodontitis resulted in a 2-mm reduction in PD and a 44% reduction in BOP17. The ideal technique to define specific species in the oral biofilm is to determine quantity because the microbial differences between health and periodontal disease and before and after treatment of periodontal disease are quantitative rather than merely nothing the presence or absence of one or more species. Molecular techniques, such as real-time polymerase chain reaction (PCR), have made it possible to evaluate specific microorganisms and to identify those bacteria present in small numbers18. Until now, no study has evaluated the antimicrobial effects of EMD in the nonsurgical treatment of peri-implantitis, although a few studies have shown improvement in clinical parameters with its use. On the other hand, there has not been a randomized clinical trial on both clinical and microbial outcomes of adjunctive use of MSM in the nonsurgical treatment of peri-implant lesions. The present study was designed to compare these different therapeutic techniques, including MD alone and in conjunction with the use of local MSM or EMD, in terms of clinical and microbiological effects on an implant affected by PIMI.
II. Materials and Methods
This randomized controlled trial study was designed as a double-blind, three-arm parallel-group to evaluate the effects of MSM (Arestine; Hansa Med Ltd., Mississauga, ON, Canada) and EMD (Emdogain; Straumann, Basel, Switzerland) in comparison with MD alone on the clinical and microbial profile of peri-implant infl ammatory diseases. We also used patients as a unit for randomization. Following approval of the study protocol by the Ethics Committee for Human Research at Tabriz University of Medical Sciences (protocol No.: 92111, IRCT20131103690), 96 adult subjects from a group of patients recalled for follow-up visits from September 2013 to March 2014 at the Department of Periodontics, Faculty of Dentistry, Tabriz University of Medical Science in Tabriz, Iran, were recruited for the present study. All of implants were of the same brand (Dentis, Daegu, Korea) with resorbable blasted media surface treatment.
Inclusion criteria: Adults 18 years of age or older, implant functional for at least one year, peri-implant mucositis and\or mild peri-implantitis defined as presence of BOP without soft tissue recession with or without minimal radiographic bone loss (≤2 mm), and PD≥4 mm in at least one site of the peri-implant arranged according to implant success index grade III, IV (Kadkhodazadeh and Amid classification)19. These grades are clinical manifestations rather than radiographic patterns.
Exclusion criteria: Use of systemic or local antibiotics in the past three months, regular intake of anti-inflammatory drugs in the past three months, any intervention for treatment of peri-implant infl ammation in the past three months, poor oral hygiene, smoking, pregnancy and lactation, severe periodontal disease, poorly uncontrolled diabetes or debilitating systemic disease, drug and alcohol addiction, or allergies to tetracycline-class drugs.
With an aim to identify remission in one positive site of six sites per implant, 23 patients per group (MSM, EMD, and control) were included in order to achieve a power of 80%, standard deviation 1.3 (Fisher's exact test), and 5% significance level. Patients were randomized using a web-based randomization software program (Research Randomizer; http://www.randomizer.org)20 and then randomly divided into one control group and two test groups. All subjects were instructed to use an effective home care program for oral hygiene and were randomly assigned to undergo one of the following treatment protocols. In the control group, mechanical subgingival debridement was carried out, which included threads using ultrasonic scaler instruments (Piezon 250; EMS Electro Medical Systems SA, Nyon, Switzerland); in addition, glycine-based powder air-polishing (Air-Flow Master, Perio Powder; EMS Electro Medical Systems SA) was used to remove subgingival biofilm. In the MSM group, after MD, 1 mg of minocycline hydrochloride microspheres was placed subgingivally in the affected sites after cessation of bleeding and isolation/drying of the implant site. In the EMD group, after MD and isolation of the peri-implant area, 1 mg of EMD was placed subgingivally in affected sites. According to Cumulative Interceptive Supportive Therapy (CIST), BOP-positive implant sites exhibit an increased PD (4 to 5 mm) and might or might not demonstrate suppuration; therefore, antiseptic therapy was delivered in addition to MD21. All patients were advised to avoid brushing and fl ossing of treated sites for seven days, thereby avoiding removal of Arestin and Emdogain from the site. After one week, patients resumed brushing these areas with a toothbrush soaked in 0.12% chlorhexidine twice a day. If more than one implant in the same patient was involved, treatment was delivered to all implants using the same protocol. Microbial analysis of gingival crevicular fl uid was carried out, and clinical parameters were recorded at baseline and two weeks and three months after treatment.
1. Peri-implant microbial collection and real-time PCR analysis
Each affected implant site was isolated with sterile cotton rolls (after removal of supragingival plaque with a plastic scaler), and subgingival samples were collected at the pre-determined location with the deepest probing pocket depth (PPD) around the implant site using sterile endodontic paper cones (#30). If bleeding occurred during removal of supragingival deposits, microbial sampling was postponed to the next session. The paper points were carefully inserted into the depth of the sulcus/pocket and kept in position for 15 seconds. The samples were inserted into a single labeled Eppendorf (Eppendorf AG, Hamburg, Germany) tube containing 1.5 mL of reduced ringer solution. Within 30 minutes after sampling, the tubes were transferred to the microbiology laboratory for subsequent real-time PCR measurement using a commercial gene probe (Primer Design Genesig kit; Primer Design Ltd., London, UK) test for evaluation of bacterial species (P. gingivalis).
Genomic DNA was extracted using a commercial DNA extraction kit (AmpliSens; Central Research Institute for Epidemiology, Moscow, Russia). In order to quantify the total counts of bacteria in the samples, quantitative real-time PCR was used with the Primer Design using Taqman probes that probe 3' and 5' ends labeled with dye6-carboxy-tetramethylrhodamine according to the manufacturer's instructions. Real-time PCR amplification protocols for bacterium consisted of an initial hot start at 95℃ for 10 minutes for enzyme activation, followed by 50 PCR cycles at 95℃ for 10 seconds for denaturation, and 60℃ for 60 seconds for annealing and extension, with fluorescence emissions monitored during the extension step. Standard curves were analyzed by comparing the universal primer set against a serial dilution of P. gingivalis genomic DNA. Based on the results obtained from the quantitative real-time PCR, the detection frequency of the species in the subgingival plaque was calculated. Real-time PCR and statistical analyses were performed by operators blind to the study design.
The following clinical parameters were recorded: (1) BOP: bleeding for up to 30 seconds after gentle probing and (2) PD: distance (mm) between the mucosal margin and the bottom of the sulcus.
2. Statistical analysis
Each variable was examined on a subject level. The Kolmogorov-Smirnov test was used to determine the normality of data. Median values and interquartile range were estimated for each variable during various assessment intervals of the study (baseline, two weeks, and three months after intervention). A Friedman test was carried out to compare the median values of nonparametric variables for different procedures during the time intervals. The Mann-Whitney U test was performed to evaluate variables between the test and control groups. P-values <0.05 were regarded as statistically significant. Statistic analysis was performed for SPSS for Windows version 16.0 (SPSS Inc., Chicago, IL, USA).
III. Results
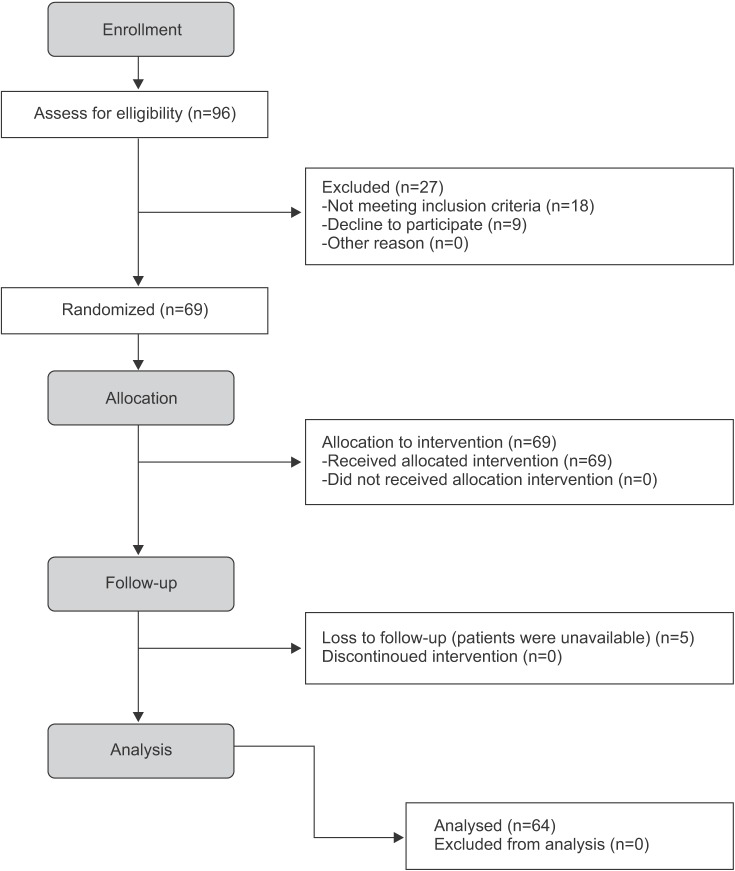
A total of 64 patients (20 subjects in the EMD, 23 subjects in the MSM, and 21 subjects in the control groups) participated in the study and were evaluated until the end of the follow-up period. Two patients in the control group and three patients in the EMD group were excluded from the study because they were not present at follow-up examinations.(Fig. 1) Table 1 shows the baseline demographic characteristics of all groups. There were no statistically significant differences in baseline demographics among the groups.
Fig. 1. Study flowchart.
Masumeh Faramarzi et al: Microbiological and clinical effects of enamel matrix derivative and sustained-release micro-spherical minocycline application as an adjunct to non-surgical therapy in peri-implant mucosal inflammation. J Korean Assoc Oral Maxillofac Surg 2015
Table 1. Demographic information of the study subjects at baseline.
| Demographic characteristic | MSM | EMD | Control |
|---|---|---|---|
| Number of patients | 23 | 20 | 21 |
| Gender (female/male) | 11/12 | 10/10 | 10/11 |
| Age (yr) | 48.4±2.9 | 49.9±2.9 | 45.6±2.9 |
| Location | |||
| Anterior max/posterior max | 2/9 | 3/8 | 3/8 |
| Anterior man/posterior man | 4/8 | 3/6 | 8/2 |
| Total number of implants | 3.4±0.6 | 3.0±0.5 | 3.7±0.6 |
| History of periodontitis | 11 (47.8) | 10 (50.0) | 10 (47.6) |
| Prosthesis (overdenture/bridge/crown) | 15/4/4 | 13/5/2 | 6/10/5 |
| History of GBR treatment | 6 | 7 | 4 |
| Surgical protocol (one/two stage) | 8/15 | 3/17 | 4/17 |
(MSM: micro-spherical minocycline, EMD: enamel matrix derivative, max: maxilla, man: mandible, GBR: guided bone regeneration)
Values are presented as number, mean±standard deviation, or number (%).
Masumeh Faramarzi et al: Microbiological and clinical effects of enamel matrix derivative and sustained-release micro-spherical minocycline application as an adjunct to non-surgical therapy in peri-implant mucosal inflammation. J Korean Assoc Oral Maxillofac Surg 2015
1. P. gingivalis counts
Throughout the study, the P. gingivalis counts in all three groups decreased significantly. Comparison of the control and MSM groups showed no significant differences at baseline (P=0.934); however, at two-week and three-month follow-up intervals, the differences were significant, with greater decreases in bacterial counts in the MSM group (P<0.001). (Table 2) A similar comparison between the control and EMD group showed that, despite absence of differences between the groups at baseline (P=0.082), the differences were significant at two-week (P=0.007) and three-month (P=0.026) intervals, with greater reductions in bacterial counts in the EMD group.(Table 3)
Table 2. Intra- and inter-group comparison of clinical and microbial parameters among MD and MD+microspherical minocyclin groups at baseline and two weeks and three months after treatment.
| Parameter | MD | MD+microspherical minocyclin | Inter-group comparison, P-value2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | 2 wk follow-up | 3 mo follow-up | Baseline | 2 wk follow-up | 3 mo follow-up | Baseline | 2 wk follow-up | 3 mo follow-up | |
| PD (mm) | 4.00 (5.00-6.00) | 5.00 (5.00-6.00) | 4.00 (5.00-6.00) | 4.00 (5.00-5.00) | 2.00 (2.00-2.00) | 2.00 (2.00-3.00) | 0.200 | 0.000 | 0.000 |
| P=0.0981 | P<0.0011 | ||||||||
| BOP (%) | 75.00 (75.00-100.00) | 75.00 (50.00-100.00) | 75.00 (50.00-100.00) | 75.00 (75.00-100.00) | 0.00 (0.00-25.00) | 0.00 (0.00-25.00) | 0.179 | 0.000 | 0.000 |
| P=0.1091 | P<0.0011 | ||||||||
| P. gingivalis (IU/mL) | 132.00 (29.00-221.50) | 114.00 (23.50-143.00) | 125.00 (26.00-160.50) | 115.00 (55.00-155.00) | 12.00 (0.00-36.00) | 21.00 (8.00-34.00) | 0.934 | <0.001 | <0.001 |
| P<0.0011 | P<0.0011 | ||||||||
(MD: mechanical debridement, PD: probing depth, BOP: bleeding on probing, P. gingivalis: Porphyromonas gingivalis)
1Friedman test. 2Mann-Whitney U test.
Values are presented as median (interquartile range).
Masumeh Faramarzi et al: Microbiological and clinical effects of enamel matrix derivative and sustained-release micro-spherical minocycline application as an adjunct to non-surgical therapy in peri-implant mucosal inflammation. J Korean Assoc Oral Maxillofac Surg 2015
Table 3. Intra- and inter-group comparison of clinical and microbial parameters among MD and MD+EMD groups at baseline and two weeks and three months after treatment.
| Parameter | MD | MD+EMD | Inter-group comparison, P-value2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | 2 wk follow-up | 3 mo follow-up | Baseline | 2 wk follow-up | 3 mo follow-up | Baseline | 2 wk follow-up | 3 mo follow-up | |
| PD (mm) | 4.00 (5.00-6.00) | 5.00 (5.00-6.00) | 4.00 (5.00-6.00) | 4.50 (4.00-5.00) | 4.00 (3.50-5.00) | 3.00 (2.00-4.37) | 0.209 | <0.001 | <0.001 |
| P=0.0981 | P<0.0011 | ||||||||
| BOP (%) | 75.00 (75.00-100.00) | 75.00 (50.00-100.00) | 75.00 (50.00-100.00) | 75.00 (75.00-100.00) | 75.00 (50.00-93.70) | 25.00 (0.00-50.00) | 0.602 | 0.234 | <0.001 |
| P=0.1091 | P<0.0011 | ||||||||
| P. gingivalis (IU/mL) | 132.00 (29.00-221.50) | 114.00 (23.50-143.00) | 125.00 (26.00-160.50) | 17.50 (0.00-91.75) | 0.00 (0.00-119.00) | 0.00 (0.00-271.50) | 0.082 | 0.007 | 0.026 |
| P<0.0011 | P<0.0011 | ||||||||
(MD: mechanical debridement, EMD: enamel matrix derivative, PD: probing depth, BOP: bleeding on probing, P. gingivalis: Porphyromonas gingivalis)
1Friedman test. 2Mann-Whitney U test.
Values are presented as median (interquartile range).
Masumeh Faramarzi et al: Microbiological and clinical effects of enamel matrix derivative and sustained-release micro-spherical minocycline application as an adjunct to non-surgical therapy in peri-implant mucosal inflammation. J Korean Assoc Oral Maxillofac Surg 2015
2. Clinical parameters
The mean value of PD at baseline evaluation was 4 mm for minocycline and control groups and 4.5 mm for the EMD group. The control group did not significantly change until the end of the study, when even the average variation increased. However, this increase was not statistically significant. In the minocycline group, this amount was reduced to 2 mm after two weeks and was maintained until the end of the study, which was found to be statistically significant compared to the baseline (P<0.001) and control (P<0.001). In the EMD group after three months, this amount was decreased to 3 mm, which was statistically significant compared to the baseline (P<0.001) and control (P<0.001). At the end of three months, patients who received minocycline and EMD followed by MD showed significant reductions of BOP in pockets around dental implants in comparison with the baseline (P<0.001) and control groups (P<0.001). The amount of decrease in minocycline and EMD groups was 60% and 50%, respectively. Reduction of BOP in the control group at the end of three months was 20%, and none of the patients in the study were free of BOP.(Tables 2, 3)
IV. Discussion
The aim of the present randomized clinical trial was to compare the effects of intra-sulcular application of sustained-release MSM and EMD after MD and MD alone on microbial and clinical parameters in the treatment of PIMI. The main outcome of the present study was changes in the counts of P. gingivalis microorganisms. The results showed that all three protocols had a significant effect on decreasing P. gingivalis counts at two-week and three-month intervals compared to baseline; improvement in clinical parameters was also significant with the adjunctive use of MSM and EMD compared to MD alone. Microorganisms are important etiologic factors in periodontal disease, and the composition of microbiota in peri-implant areas affects the future health status of the area22. The submucosal microbiota in implants with clinically healthy peri-implant marginal tissues mainly consists of Gram-positive cocci and rods. In contrast, diseased dental implant microbiota in animals mainly consists of periodontal pathogens such as A. actinomycetemcomitans and P. gingivalis23. P. gingivalis was identified in the peri-implant crevicular fluid of all subjects and is consistent with previous studies in which a possible relationship was found between this microorganism and peri-implant lesions. P. gingivalis is one of the microorganisms that has been extensively studied. It is an anaerobic Gram-negative bacteria species with different virulence factors, including proteases, fimbriae, lipopolysaccharide, and capsule, which enable the pathogen to induce peri-implantitis. It can invade host cells and survive and induces an inflammatory response and destruction of extracellular matrix and bone24. The principal aim of treatment of peri-implantitis is to resolve inflammation and stop disease progression. The etiology of peri-implantitis is similar to that of periodontitis. Therefore, the treatment process is similar in both cases, i.e., anti-infection therapy25. Various methods have been used to treat peri-implantitis. For example, lasers and photodynamic therapy have been used for decontamination of implant surfaces during surgical and regenerative treatment of peri-implantitis. Carbon dioxide laser has been successful in eliminating pathogens, especially Streptococcus sanguis and P. gingivalis, from the surface of titanium implants and does not induce surface changes, increase surface temperature, or prevent cellular adhesion to irradiated surfaces26. Regarding the mechanical method used in this study, MD was performed using an ultrasonic scaler with a metal tip and air-powder polishing. A study performed by Sahm et al.27 on the use of air-powder polishing of implants with rough surfaces showed that the bleeding index decreased to a greater extent in comparison with the use of carbon curettes. Park et al.28 also showed that in vitro use of an ultrasonic scaler with a metal tip on implants with rough surfaces creates smoother surfaces and removes bacterial biofilm more effectively than ultrasonic scalers with a plastic tip. Almost all antibiotics that have been used in the treatment of peri-implantitis belong to the group of tetracyclines. Tetracycline HCl has been used in polymeric fibers in a study by Mombelli et al.10 in partially edentulous patients with peri-implantitis with PPD greater than 5 mm around the implants. Over 12 months, a mean reduction in PD of 1.25 mm was reported. During the follow-up period (1, 3, 6, and 12 months), improvement in clinical parameters of PPD and modified bleeding index was considerably improved compared to baseline values. Mean counts of cultivable anaerobic bacteria at 1, 3, and 6 month postoperative intervals were significantly less than those at baseline. This decrease in the frequency of detection of P. intermedia/nigrescens, Fusobacterium sp., Bacteroides forsythus, and Campylobacter rectus was significant. However, in relation to A. actinomycetemcomitans, P. gingivalis, and Eikenella corrodens, these pathogens were found to have very low frequencies at baseline and therefore did not exhibit significant decrease10. To date, clinical studies have also shown the effects of adjunctive use of minocycline on the microbial and clinical results of treatment of peri-implantitis. Renvert carried out a 12-month controlled clinical trial to compare minocycline microspheres and chlorhexidine in the treatment of initial infections around implants and showed that adjunctive use of minocycline microspheres resulted in reduction in PD and bleeding. However, adjunctive use of chlorhexidine alone resulted in a limited decrease in bleeding score. The study failed to show a significant difference in bacterial load between the two groups29. Persson et al.30 also studied the antimicrobial effect of topical application of Arestin in the treatment of peri-implantitis. In his study, DNA-DNA checkerboard hybridization was used to analyze multiple samples of bacteria. At 180 days after the study, the bacterial loads of T. forsythia, P. gingivalis, and Treponema denticola were reduced. It was observed that the effect of Arestin on A. actinomycetemcomitans was greater than its effects on the other pathogens30.
Renvert et al.31 evaluated the effect of local application of minocycline in patients with bone loss (less than 3 mm in a 10 to 12 year period). PPD was reduced from 5.0 to 4.4 mm at 12 months in the deepest probing areas. The mean scores of BOP around the implants during the first month decreased from 88% to 40%; however, it increased to 71% during the following 12 months. In the present study, the group treated with minocycline exhibited significantly lower P. gingivalis counts at two-week and three-month follow-ups compared to baseline, consistent with the results of previous studies. This decrease was also significant in comparison with a decrease in P. gingivalis count in the control group. Another adjunctive technique in the treatment of peri-implantitis is the local use of EMD. Periodontal regeneration with EMD is based on the hypothesis that its use in periodontal lesions depends on a process similar to the development of tooth-supporting structures during tooth formation. The enamel matrix is composed of a number of proteins, 90% of which is amelogenin. These proteins induce periodontal attachment development during tooth formation14. Gestrelius et al.32 showed that EMD precipitates from its PGA vehicle at physiologic pH in vivo and in vitro, forms a covering on the root surface, and produces insoluble compositions that last up to two weeks, which seems to be sufficient for the re-colonization of the root surface with the fibroblasts of the periodontal ligament. Studies have shown the effect of EMD on suppression of P. gingivalis in vitro. Spahr et al.16 showed the inhibitory effect of EMD on the growth of Gram-negative periodontal pathogens, including P. gingivalis, A. actinomycetemcomitans, and P. intermedia in vitro. Schou et al.23 also showed antimicrobial effects of Emdogain gel on P. gingivalis and attributed this result to the PGA in the composition of gel. However, there is insufficient evidence regarding the antimicrobial effects of EMD in the treatment of peri-implantitis even though previous studies have shown improvements in clinical parameters with its use. In the present study, P. gingivalis count in the group treated with EMD decreased significantly at two-week and three-month intervals.
Use of EMD in the surgical treatment of periodontal lesions resulted in significant decrease in PD and clinical attachment level (CAL) gain compared to the control group (without the use of EMD). The effects of EMD in reduction of PD and BOP could be attributed to the various anti-inflammatory and anti-antimicrobial effects of EMD. This observation has been documented in various studies33. In a study by Wennström and Lindhe34 on SRP with or without non-surgical application of EMD, areas treated with EMD exhibited better regeneration and postoperative pain levels with less BOP compared to SRP alone. In a study by Mombelli et al.35, adjunctive therapy with EMD with SRP in patients with moderate-to-severe periodontitis (BOP of 90% and a mean pocket depth of 7.3 mm) resulted in a decrease of 2.8 mm in PD and a decrease of 30% in BOP. Sculean investigated peri-implantitis treatment with ethylenediaminetetraacetic acid decontamination and application of EMDs. In the study, intrabony defects in three patients were treated with open flap surgery. One year after treatment, reduction in PD, gain in CAL, and radiographic bone fill was observed in all patients, and the results were sustained up to three years36. However, no study has been conducted on the nonsurgical use of EMD in the treatment of peri-implantitis.
In the present study, the mean reduction in BOP for minocycline and EMD groups was 60% and 50%, respectively, while the control group showed a BOP reduction of only 20%. At three months, the control group showed no significant change in PD, but the minocycline and EMD groups showed 2 mm and 1.5 mm respective reduction of PD. The differences in BOP and PD reduction in this study compared with previous studies might be due to differences in initial PD, amount of bone loss, plaque control, and various demographic characteristics, as well as the type and method of baseline treatment.
Detection of microorganisms varies significantly with the effect of methodology. Real-time PCR was used in the present study.
Different techniques are available to evaluate microbial samples of the dental plaque. Bacterial culture is considered a gold standard diagnostic test for the quantitative evaluation of microorganisms colonizing the oral cavity although it has some limitations compared to real-time PCR. The culture process is time-consuming and has low sensitivity. Real-time PCR is more reliable and is capable of identifying a small number of microorganisms. However, it requires extraction of bacterial DNA, which is difficult and costly. In addition, contrary to the culturing technique, real-time PCR identifies nonviable bacteria in samples because there are no differences between intact DNA in viable and nonviable cells37,38,39.
The limitations of our study are the small scale and relatively small number of patients. Moreover, the studied patients were heterogeneous in terms of rate of plaque control during the study. Therefore, we recommend future studies with the same concept and an increased number of patients to ensure the ability to detect differences with stronger trends. Also, more studies on treatment modalities for PIMI with longer term follow-up are necessary because PIMI is a chronic disease that requires repeat treatment procedures.
V. Conclusion
The results of the present study showed that the use of MSM and EMD can be considered as an adjunctive therapy for the MD in the non-surgical management of these lesions, resulting in a decrease in P. gingivalis count and improvement in clinical parameters at least three months after treatment.
Footnotes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
References
- 1.Misch CE, Perel ML, Wang HL, Sammartino G, Galindo-Moreno P, Trisi P, et al. Implant success, survival, and failure: the International Congress of Oral Implantologists (ICOI) Pisa Consensus Conference. Implant Dent. 2008;17:5–15. doi: 10.1097/ID.0b013e3181676059. [DOI] [PubMed] [Google Scholar]
- 2.Esposito M, Hirsch JM, Lekholm U, Thomsen P. Biological factors contributing to failures of osseointegrated oral implants. (II). Etiopathogenesis. Eur J Oral Sci. 1998;106:721–764. doi: 10.1046/j.0909-8836..t01-6-.x. [DOI] [PubMed] [Google Scholar]
- 3.Pjetursson BE, Tan K, Lang NP, Brägger U, Egger M, Zwahlen M. A systematic review of the survival and complication rates of fixed partial dentures (FPDs) after an observation period of at least 5 years. Clin Oral Implants Res. 2004;15:667–676. doi: 10.1111/j.1600-0501.2004.01120.x. [DOI] [PubMed] [Google Scholar]
- 4.Heitz-Mayfield LJ. Peri-implant diseases: diagnosis and risk indicators. J Clin Periodontol. 2008;35(8 Suppl):292–304. doi: 10.1111/j.1600-051X.2008.01275.x. [DOI] [PubMed] [Google Scholar]
- 5.Apse P, Ellen RP, Overall CM, Zarb GA. Microbiota and crevicular fluid collagenase activity in the osseointegrated dental implant sulcus: a comparison of sites in edentulous and partially edentulous patients. J Periodontal Res. 1989;24:96–105. doi: 10.1111/j.1600-0765.1989.tb00863.x. [DOI] [PubMed] [Google Scholar]
- 6.Serino G, Ström C. Peri-implantitis in partially edentulous patients: association with inadequate plaque control. Clin Oral Implants Res. 2009;20:169–174. doi: 10.1111/j.1600-0501.2008.01627.x. [DOI] [PubMed] [Google Scholar]
- 7.Loe H, Theilade E, Jensen SB. Experimental gingivitis in man. J Periodontol. 1965;36:177–187. doi: 10.1902/jop.1965.36.3.177. [DOI] [PubMed] [Google Scholar]
- 8.Renvert S, Roos-Jansåker AM, Claffey N. Non-surgical treatment of peri-implant mucositis and peri-implantitis: a literature review. J Clin Periodontol. 2008;35(8 Suppl):305–315. doi: 10.1111/j.1600-051X.2008.01276.x. [DOI] [PubMed] [Google Scholar]
- 9.Ericsson I, Persson LG, Berglundh T, Edlund T, Lindhe J. The effect of antimicrobial therapy on periimplantitis lesions. An experimental study in the dog. Clin Oral Implants Res. 1996;7:320–328. doi: 10.1034/j.1600-0501.1996.070404.x. [DOI] [PubMed] [Google Scholar]
- 10.Mombelli A, Feloutzis A, Brägger U, Lang NP. Treatment of peri-implantitis by local delivery of tetracycline. Clinical, microbiological and radiological results. Clin Oral Implants Res. 2001;12:287–294. doi: 10.1034/j.1600-0501.2001.012004287.x. [DOI] [PubMed] [Google Scholar]
- 11.Büchter A, Meyer U, Kruse-Lösler B, Joos U, Kleinheinz J. Sustained release of doxycycline for the treatment of peri-implantitis: randomised controlled trial. Br J Oral Maxillofac Surg. 2004;42:439–444. doi: 10.1016/j.bjoms.2004.06.005. [DOI] [PubMed] [Google Scholar]
- 12.Williams RC, Paquette DW, Offenbacher S, Adams DF, Armitage GC, Bray K, et al. Treatment of periodontitis by local administration of minocycline microspheres: a controlled trial. J Periodontol. 2001;72:1535–1544. doi: 10.1902/jop.2001.72.11.1535. [DOI] [PubMed] [Google Scholar]
- 13.Leung WK, Jin L, Yau JY, Sun Q, Corbet EF. Microflora cultivable from minocycline strips placed in persisting periodontal pockets. Arch Oral Biol. 2005;50:39–48. doi: 10.1016/j.archoralbio.2004.08.002. [DOI] [PubMed] [Google Scholar]
- 14.Hammarström L, Heijl L, Gestrelius S. Periodontal regeneration in a buccal dehiscence model in monkeys after application of enamel matrix proteins. J Clin Periodontol. 1997;24:669–677. doi: 10.1111/j.1600-051x.1997.tb00248.x. [DOI] [PubMed] [Google Scholar]
- 15.Casati MZ, Sallum EA, Nociti FH, Jr, Caffesse RG, Sallum AW. Enamel matrix derivative and bone healing after guided bone regeneration in dehiscence-type defects around implants. A histomorphometric study in dogs. J Periodontol. 2002;73:789–796. doi: 10.1902/jop.2002.73.7.789. [DOI] [PubMed] [Google Scholar]
- 16.Spahr A, Lyngstadaas SP, Boeckh C, Andersson C, Podbielski A, Haller B. Effect of the enamel matrix derivative Emdogain on the growth of periodontal pathogens in vitro. J Clin Periodontol. 2002;29:62–72. doi: 10.1034/j.1600-051x.2002.290110.x. [DOI] [PubMed] [Google Scholar]
- 17.Gutierrez MA, Mellonig JT, Cochran DL. Evaluation of enamel matrix derivative as an adjunct to non-surgical periodontal therapy. J Clin Periodontol. 2003;30:739–745. doi: 10.1034/j.1600-051x.2003.00374.x. [DOI] [PubMed] [Google Scholar]
- 18.Haffajee AD, Socransky SS. Microbiology of periodontal diseases: introduction. Periodontol 2000. 2005;38:9–12. doi: 10.1111/j.1600-0757.2005.00112.x. [DOI] [PubMed] [Google Scholar]
- 19.Kadkhodazadeh M, Amid R. Evaluation of peri-implant tissue health using a scoring system. J Implant Adv Clin Dent. 2012;4:51–57. [Google Scholar]
- 20.Research Randomizer [Internet] Place unknown: Geoffrey C. Urbaniak and Scott Plous; 1997-2015. [cited 2009 Aug 18]. Available from: https://www.randomizer.org. [Google Scholar]
- 21.Lang NP, Berglundh T, Heitz-Mayfield LJ, Pjetursson BE, Salvi GE, Sanz M. Consensus statements and recommended clinical procedures regarding implant survival and complications. Int J Oral Maxillofac Implants. 2004;19(Suppl):150–154. [PubMed] [Google Scholar]
- 22.Mombelli A, van Oosten MA, Schurch E, Jr, Land NP. The microbiota associated with successful or failing osseointegrated titanium implants. Oral Microbiol Immunol. 1987;2:145–151. doi: 10.1111/j.1399-302x.1987.tb00298.x. [DOI] [PubMed] [Google Scholar]
- 23.Schou S, Holmstrup P, Keiding N, Fiehn NE. Microbiology of ligature-induced marginal inflammation around osseointegrated implants and ankylosed teeth in cynomolgus monkeys (Macaca fascicularis) Clin Oral Implants Res. 1996;7:190–200. doi: 10.1034/j.1600-0501.1996.070301.x. [DOI] [PubMed] [Google Scholar]
- 24.Holt SC, Kesavalu L, Walker S, Genco CA. Virulence factors of Porphyromonas gingivalis. Periodontol 2000. 1999;20:168–238. doi: 10.1111/j.1600-0757.1999.tb00162.x. [DOI] [PubMed] [Google Scholar]
- 25.Lang NP, Wilson TG, Corbet EF. Biological complications with dental implants: their prevention, diagnosis and treatment. Clin Oral Implants Res. 2000;11(Suppl 1):146–155. doi: 10.1034/j.1600-0501.2000.011s1146.x. [DOI] [PubMed] [Google Scholar]
- 26.Kato T, Kusakari H, Hoshino E. Bactericidal efficacy of carbon dioxide laser against bacteria-contaminated titanium implant and subsequent cellular adhesion to irradiated area. Lasers Surg Med. 1998;23:299–309. doi: 10.1002/(sici)1096-9101(1998)23:5<299::aid-lsm10>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 27.Sahm N, Becker J, Santel T, Schwarz F. Non-surgical treatment of peri-implantitis using an air-abrasive device or mechanical debridement and local application of chlorhexidine: a prospective, randomized, controlled clinical study. J Clin Periodontol. 2011;38:872–878. doi: 10.1111/j.1600-051X.2011.01762.x. [DOI] [PubMed] [Google Scholar]
- 28.Park JB, Jang YJ, Koh M, Choi BK, Kim KK, Ko Y. In vitro analysis of the efficacy of ultrasonic scalers and a toothbrush for removing bacteria from resorbable blast material titanium disks. J Periodontol. 2013;84:1191–1198. doi: 10.1902/jop.2012.120369. [DOI] [PubMed] [Google Scholar]
- 29.Renvert S, Lessem J, Lindahl C, Svensson M. Treatment of incipient peri-implant infections using topical minocycline microspheres versus topical chlorhexidine gel as an adjunct to mechanical debridement. J Int Acad Periodontol. 2004;6(4 Suppl):154–159. [PubMed] [Google Scholar]
- 30.Persson GR, Salvi GE, Heitz-Mayfield LJ, Lang NP. Antimicrobial therapy using a local drug delivery system (Arestin) in the treatment of peri-implantitis. I: microbiological outcomes. Clin Oral Implants Res. 2006;17:386–393. doi: 10.1111/j.1600-0501.2006.01269.x. [DOI] [PubMed] [Google Scholar]
- 31.Renvert S, Lessem J, Dahlén G, Lindahl C, Svensson M. Topical minocycline microspheres versus topical chlorhexidine gel as an adjunct to mechanical debridement of incipient peri-implant infections: a randomized clinical trial. J Clin Periodontol. 2006;33:362–369. doi: 10.1111/j.1600-051X.2006.00919.x. [DOI] [PubMed] [Google Scholar]
- 32.Gestrelius S, Andersson C, Lidström D, Hammarström L, Somerman M. In vitro studies on periodontal ligament cells and enamel matrix derivative. J Clin Periodontol. 1997;24:685–692. doi: 10.1111/j.1600-051x.1997.tb00250.x. [DOI] [PubMed] [Google Scholar]
- 33.Bosshardt DD. Biological mediators and periodontal regeneration: a review of enamel matrix proteins at the cellular and molecular levels. J Clin Periodontol. 2008;35(8 Suppl):87–105. doi: 10.1111/j.1600-051X.2008.01264.x. [DOI] [PubMed] [Google Scholar]
- 34.Wennström JL, Lindhe J. Some effects of enamel matrix proteins on wound healing in the dento-gingival region. J Clin Periodontol. 2002;29:9–14. doi: 10.1034/j.1600-051x.2002.290102.x. [DOI] [PubMed] [Google Scholar]
- 35.Mombelli A, Brochut P, Plagnat D, Casagni F, Giannopoulou C. Enamel matrix proteins and systemic antibiotics as adjuncts to non-surgical periodontal treatment: clinical effects. J Clin Periodontol. 2005;32:225–230. doi: 10.1111/j.1600-051X.2005.00664.x. [DOI] [PubMed] [Google Scholar]
- 36.Sculean A, Windisch P, Auschill TM, Döri F. Treatment of peri-implantitis with EDTA decontamination and application of an enamel matrix protein derivate-a report of 3 case. Periodontal Pract Today. 2004;1:237–245. [Google Scholar]
- 37.Boutaga K, van Winkelhoff AJ, Vandenbroucke-Grauls CM, Savelkoul PH. Comparison of real-time PCR and culture for detection of Porphyromonas gingivalis in subgingival plaque samples. J Clin Microbiol. 2003;41:4950–4954. doi: 10.1128/JCM.41.11.4950-4954.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Boutaga K, van Winkelhoff AJ, Vandenbroucke-Grauls CM, Savelkoul PH. The additional value of real-time PCR in the quantitative detection of periodontal pathogens. J Clin Periodontol. 2006;33:427–433. doi: 10.1111/j.1600-051X.2006.00925.x. [DOI] [PubMed] [Google Scholar]
- 39.Sakamoto M, Takeuchi Y, Umeda M, Ishikawa I, Benno Y. Rapid detection and quantification of five periodontopathic bacteria by real-time PCR. Microbiol Immunol. 2001;45:39–44. doi: 10.1111/j.1348-0421.2001.tb01272.x. [DOI] [PubMed] [Google Scholar]