Abstract
Synthetic cannabinoids (SC), though not detected with routine urine toxicology screening, can cause severe metabolic derangements and widespread deleterious effects in multiple organ systems. The diversity of effects is related to the wide distribution of cannabinoid receptors in multiple organ systems. Both cannabinoid-receptor-mediated and non-receptor-mediated effects can result in severe cardiovascular, renal, and neurologic manifestations. We report the case of a 45-year-old African American male with ST-elevation myocardial infarction, subarachnoid hemorrhage, reversible cardiomyopathy, acute rhabdomyolysis, and severe metabolic derangement associated with the use of K2, an SC. Though each of these complications has been independently associated with SCs, the combination of these effects in a single patient has not been heretofore reported. This case demonstrates the range and severity of complications associated with the recreational use of SCs. Though now banned in the United States, use of systemic cannabinoids is still prevalent, especially among adolescents. Clinicians should be aware of their continued use and the potential for harm. To prevent delay in diagnosis, tests to screen for these substances should be made more readily available.
Keywords: Synthetic Cannabinoids, K2, Spice, subarachnoid hemorrhage, reversible cardiomyopathy, rhabdomyolysis, STEMI, multiorgan failure, JWH-018, JWH-073
Marijuana contains more than 60 different naturally occurring cannabinoids, the most biologically active being Δ9 tetrahydrocannabinol (1). Synthetic cannabinoids (SC) are laboratory-produced analogs of the naturally occurring cannabinoids. SCs are divided into seven major groups depending on chemical structure. ‘K2’, also known as ‘Spice’, is a popular SC product that also contains JWH-018 and JWH-073 metabolites and belongs to the naphthyl indoles group (2, 3). SC compounds are cannabinoid receptor (CB1 and CB2) agonists, and their metabolites also typically retain varying amounts of biological activity as agonists, neutral antagonists, or inverse agonists (3). CB1 receptors are present in abundance in the central nervous system, primarily in the basal ganglia, cerebellum, hippocampus, and cortex. CB2 receptors are present in peripheral tissues, principally in red blood cells, immune tissues, and spleen.
SCs have a wide variety of effects in humans. The most common are psychoactive effects, ranging from euphoria to anxiety, psychosis, and alterations in cognitive abilities. Other effects include nausea, vomiting, psychomotor agitation, muscle twitching, generalized convulsions, acute renal failure, hypertension, diaphoresis, palpitations/tachycardia, chest pain, and shortness of breath (3). There have also been case reports of ST-elevation myocardial infarction (STEMI), severe rhabdomyolysis, hypokalemia, and cannabinoid hyperemesis syndrome (4–6). Long-term effects include reduced brain volume, primarily in the hippocampus and amygdala, measured at 12 and 7%, respectively (3, 7). Other central effects include withdrawal symptoms, auditory or visual hallucinations, anxiety and insomnia, and stupor and suicidal ideation (3). We report a patient who experienced the following severe simultaneous complications attributed to the use of K2, an SC that has been banned in the United States since July 2012: subarachnoid hemorrhage, STEMI, reversible cardiomyopathy, rhabdomyolysis, and severe metabolic derangement.
Case description
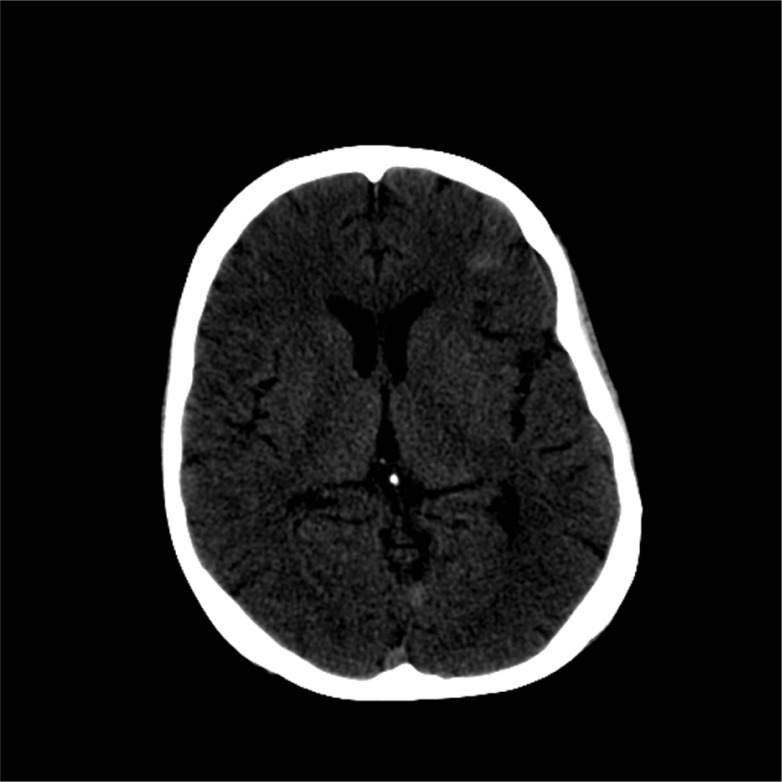
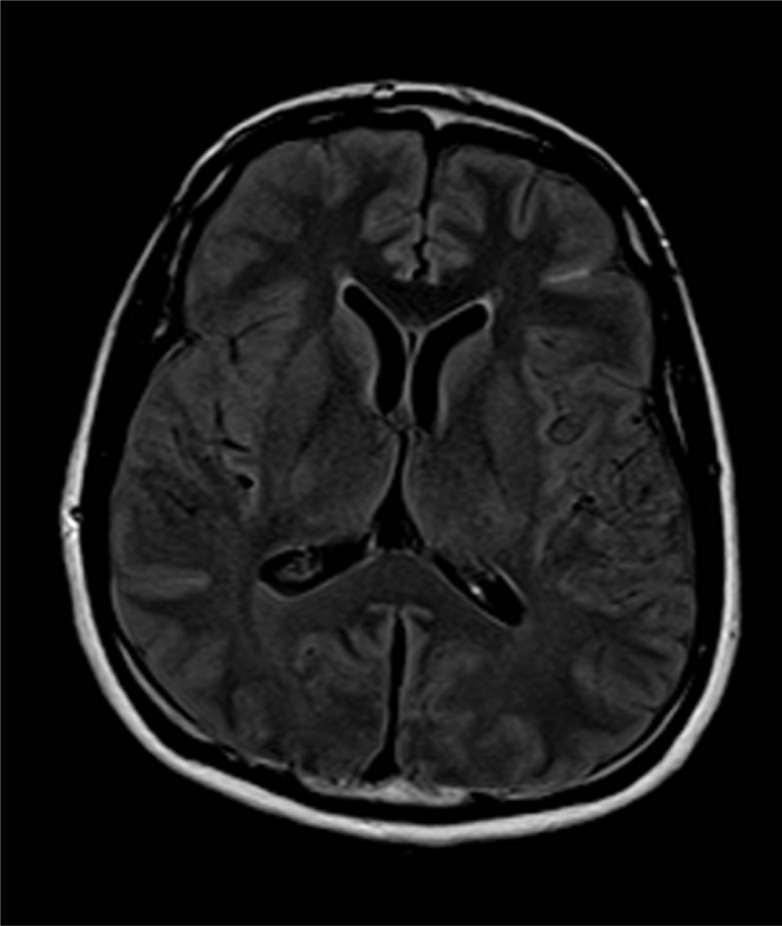
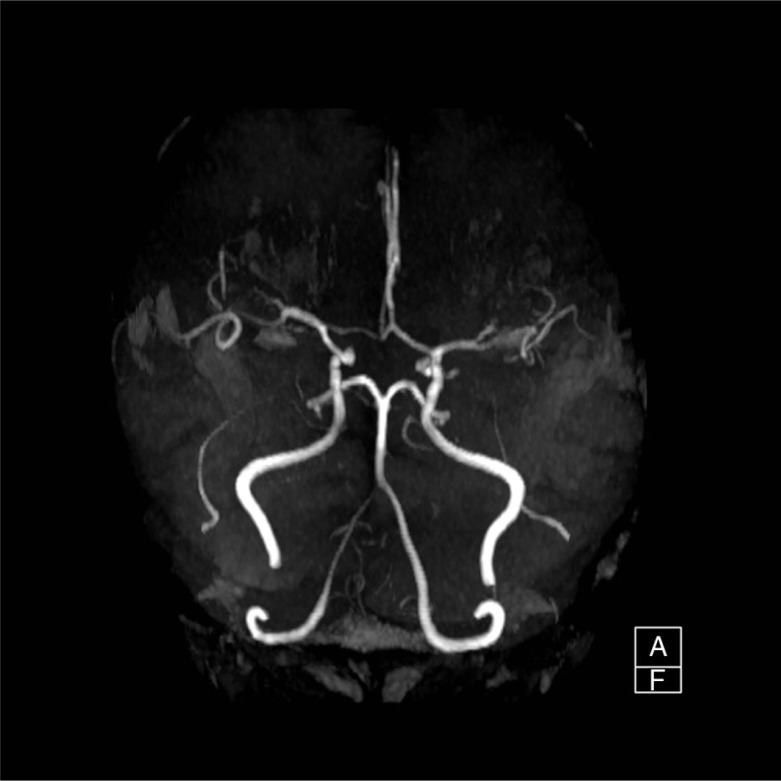
A 45-year-old African American male was found unresponsive by his roommate in their apartment. When emergency medical service (EMS) arrived, they noted a supply of K2 within arm's reach. Per available records from his primary care provider, the patient had a history of depression, anxiety, sleep disorder, hypertension, and substance abuse. On his last visit 4 months prior to hospitalization, the patient was found to have elevated blood sugar but no known history of diabetes and was not on any form of diabetic treatment. His home medications were only methadone and trazodone. There was no prior history of use of neuroleptic agents or serotonin selective reuptaker inhibitors. The patient did, however, have a history of intravenous heroin and inhalational cocaine abuse, but had been abstinent for the past year. Finally, he had been smoking cannabinoids regularly for 15 years and had recently started using K2 via a bong device twice daily 3 weeks prior to presentation. Initial vital signs showed normal blood pressure (125/81 mmHg), tachycardia (158 beats/min), tachypnea (30/min), high grade temperature (106.5°F), and hypoxia (90% O2 saturation on room air). The patient was obtunded, though bilateral pupils were equal and reactive to light. Muscular examination demonstrated normal tone with absence of rigidity. Reflexes were 1+ and symmetric with no clonus, and no focal neurological deficits were found. Cardiac auscultation elicited only regular tachycardia. Laboratory data showed (Table 1) marked hyperglycemia (Glucose 1,403 mg/dL), elevated creatinine (3.06 mg/dL), hypernatremia [Na-160 mmol/L (corrected 180.8)], hypokalemia (K 2.2 mmol/L), severe hypophosphatemia (PO4 0.7 mg/dL), mildly elevated cardiac enzymes (CK 387 U/L, CKMB 1.03 ng/mL, Troponin 0.569 ng/mL), hepatitis C Ab reactive, HIV Ag/Ab non-reactive, and Ab reactivity to hepatitis A and hepatitis B Surface Ag. The initial 12 Lead EKG showed sinus tachycardia (Heart rate 127/min) and first-degree atrioventricular block. Arterial blood gas showed anion gap metabolic acidosis, respiratory acidosis, and metabolic alkalosis. The lipid panel showed only hypertriglyceridemia (TG-309 mg/dL). Head CT scan (Fig. 1) showed subarachnoid hemorrhage and possible parenchymal hemorrhage in the left frontal lobe without soft tissue or bony lesions. Head MRI (Fig. 2) showed focal Fluid-attenuated inversion recovery (FLAIR) hyper-intense signal, also consistent with subarachnoid hemorrhage. Repeat head CT on the following day showed reduction in the conspicuity of the subarachnoid hemorrhage and no evidence of new bleeding. The patient has no evidence of trauma and was normotensive. The MRA brain (Fig. 3) showed normal vasculature and the cause of intracranial bleeding was attributed to SC use. No acute neurosurgical intervention was pursued. Seizure prophylaxis was administered for 21 days, and the mental status improved without focal neurological deficit.
Table 1.
Laboratory Data trend
| Laboratory data | Normal range | Day 1 | Day 3 | Day 7 | Discharge |
|---|---|---|---|---|---|
| Glucose | 74–106 mg/dL | 1,403 | 312 | 181 | 96 |
| BUN | 9–20 mg/dL | 23 | 22 | 9 | 8 |
| Creatinine | 0.70–1.50 mg/dL | 3.06 | 1.76 | 0.74 | 0.8 |
| Sodium | 137–145 mmol/L | 160 | 149 | 141 | 140 |
| Potassium | 3.5–5.1 mmol/L | 2.2 | 4.6 | 3.5 | 4 |
| Chloride | 98–107 mmol/L | 106 | 118 | 103 | 107 |
| Bicarbonate | 22–30 mmol/L | 20 | 13 | 32 | 26 |
| Calcium | 8.4–10.2 mg/dL | 10.8 | 7.1 | 7.1 | 9.8 |
| Phosphorous | 2.5–4.5 mg/dL | 0.7 | 4.7 | ||
| CK | 55–170 U/L | 387 | 32,391 | 301,901 | 124 |
| CKMB | 0.0–3.38 ng/mL | 1.03 | 30 | ||
| Troponin I | <0.034 ng/mL | 0.569 | 5.37 | ||
| WBC | 4.8–10.9 Thou/mm3 | 11.9 | 10.9 | 9.9 | 7.5 |
| Platelet | 130–400 Thou/mm3 | 230 | 115 | 66 | 235 |
| Hemoglobin | 13.5–17.5 mg/dL | 18 | 12.6 | 8.5 | 10.8 |
| Hematocrit | 41–53% | 59.3 | 36.5 | 25.4 | 34.1 |
Base line lab (8/23/14); creatinine – 0.95, BUN – 8.
Fig. 1.
CT scan Head without Contrast.
Fig. 2.
MRI Brain without contrast.
Fig. 3.
MRA Brain.
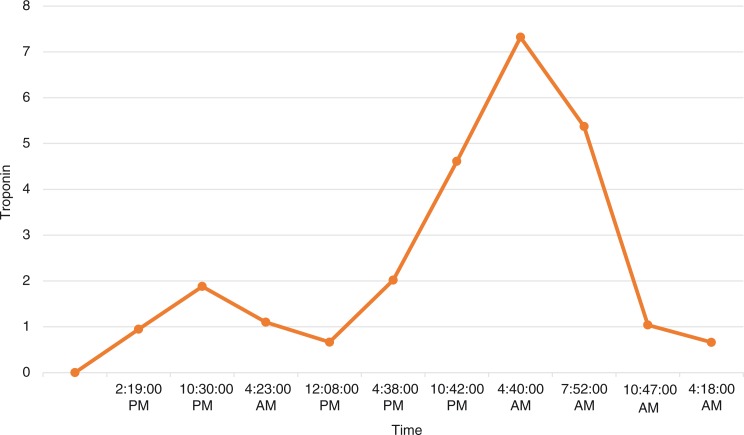
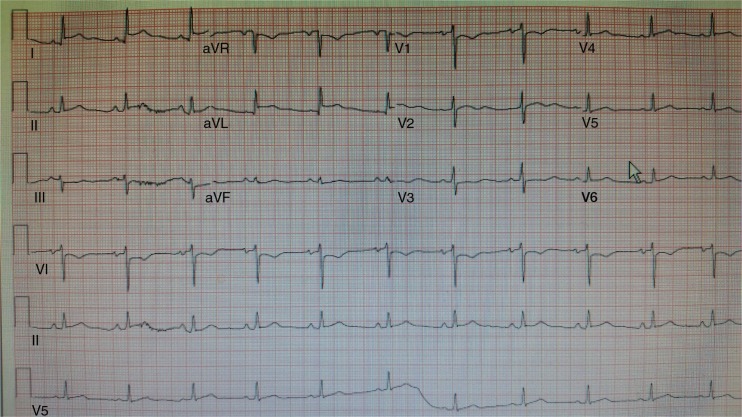
Serial troponin-I levels trended upward to a peak of 7.32 ng/mL (Fig. 4), and repeat EKG showed STEMI (Fig. 5) in leads aVL and I. Cardiac risk factors were cigarette smoking, hypertension, and recently diagnosed diabetes. The patient denied chest pain or shortness of breath. Anticoagulation and antiplatelet therapies were withheld due to the presence of an intracranial bleed, which also precluded coronary angiography. The patient remained hemodynamically stable. A 2D echocardiogram showed mild left ventricular dilation (end diastolic diameter 55 mm, end systolic diameter 49 mm, and LV posterior wall thickness 9 mm) with global hypokinesis and an ejection fraction of 15% (Echo-1). However, the LV EF spontaneously improved within 1 week to 65%, with normalization of the left ventricle size: end diastole 50 mm, end systole 27 mm, and posterior LV wall thickness 11 mm (Echo-2). As there was no evidence of sepsis, no coronary revascularization or anticoagulation, no significant history of alcohol abuse, and no apical ballooning on echocardiography, the transient cardiomyopathy attributed to K2-toxin-induced cardiomyopathy.
Fig. 4.
Troponin trend.
Fig. 5.
12 lead EKG.
The hospital course was further complicated by respiratory failure requiring intubation for ventilator support. The patient had hyperthermia at the time of admission without leukocytosis or bandemia. The patient remained afebrile for the remainder of his hospital stay; two sets of blood and sputum culture were negative, and urine analysis was normal. Abdominal CT scan showed normal kidneys and no source of infection. Abdominal ultrasound failed to demonstrate cholelithiais or cholecystitis. Chest X-ray was normal on admission, though on day 3, new perihilar and bibasilar opacities appeared. The patient was initially treated empirically with broad-spectrum antibiotic therapy, which was changed to ampicillin sulbactam for presumed aspiration pneumonia. On presentation, the patient met the criteria for systemic inflammatory response syndrome (SIRS), with hyperthermia, tachypnea, and tachycardia, but no sources of infection were evident. When he was later diagnosed with pneumonia, no SIRS criteria were present. Throughout the hospitalization, the patient remained normotensive.
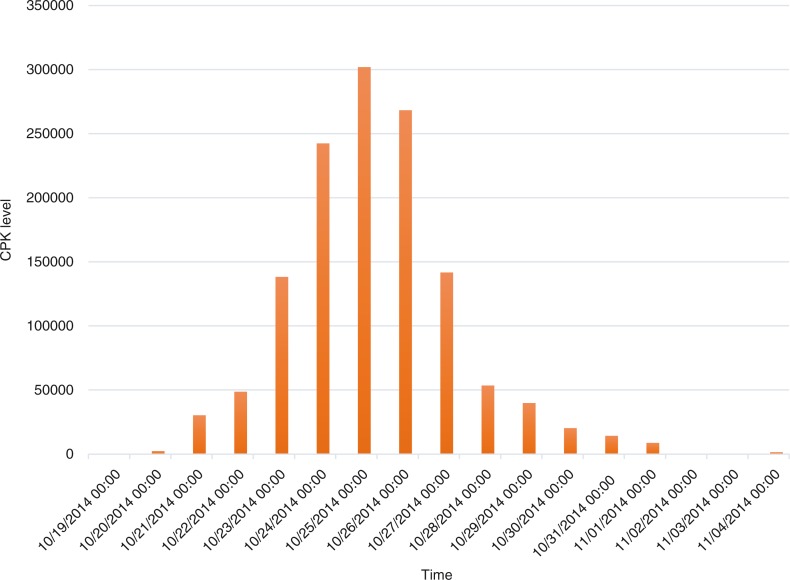
The creatine phosphokinase (CPK) level continued to trend upward, reaching a peak of 301,901 U/L over 2 weeks (Fig. 6). With daily continuous renal replacement therapy, the CPK level decreased progressively over the ensuing 2 weeks, as the renal function returned to normal (creatinine 0.95 mg/dL) as well. Urine toxicology returned positive for methadone; toxicology screen for cannabinoids, flunitrazepam, and gamma hydroxybutarate were all negative. After a 3-week hospitalization, the patient recovered entirely to baseline and was discharged home.
Fig. 6.
CPK trend.
Discussion
This case illustrates the range and severity of complications associated with the recreational use of synthetic marijuana. Though it is the third most reported substance used by US high school students (12%) after alcohol (57%) and natural marijuana (39%), less than half of the emergency medicine physicians surveyed in December 2010 had ever heard of Spice (34%) or K2 (49%). Only 20% felt adequately prepared to care for a patient with acute Spice or K2 intoxication (8).
There have been reported cases of ischemic stroke with use of SC (9, 10). The proposed mechanisms include cardiac arrhythmia with embolism and reversible cerebral vasoconstriction syndrome (9). A case of intracranial bleeding associated with SC use was reported in the Japanese literature in 2014 (11). This particular patient had a history of arteriovenous malformation (AVM) resection, 10 months prior and presented with bleeding in the same area, along with severe rhabdomyolysis (11). Our patient had normal blood pressure on initial presentation, no evidence of trauma by history or imaging, and no significant aneurysmal lesion on MRA. We assert that this is the first reported case of subarachnoid hemorrhage associated with K2 in the United States.
The mechanism linking synthetic marijuana and myocardial infarction is not clear. It has been proposed that SC has a deleterious effect on the coronary microcirculation, resulting in the reversible slow coronary flow phenomenon and increased sympathetic tone (12). A randomized double-blind, cross-over study by (13) found that smoking marijuana decreases myocardial oxygen delivery, increases myocardial oxygen demand, and decreases the time to develop angina during exercise. Whether increased platelet aggregation plays a role in marijuana-related adverse effects remains to be elucidated (12). Though our patient had pre-existing cardiac risk factors, and the possibility of atherosclerotic thrombus formation with occlusion of the coronary artery and plaque rupture exists, it seems more likely that the use of K2 precipitated or aggravated the acute coronary event. We lack the evidence of cardiac catheterization and coronary angiography to confirm this suspected mechanism.
Emotional stressors are associated with stress cardiomyopathy and may be correlated with a hyper-catecholaminergic state. An alternative mechanism linking cardiomyopathy to psychological stress may be the endocannabinoid system. Cases have been reported with reversible cardiomyopathy with temporal association with inhalation marijuana use (14). Cannabinoids reduce cardiomyocyte contractility by direct (non-receptor mediated) and CB1-mediated effects. A possible link between cannabis use and recurrent stress cardiomyopathy has been proposed (15). In addition, Partha et al. reports that CB1 receptors promote oxidative stress and cell death in murine models of doxorubicin-induced cardiomyopathy and human cardiomyocytes (16). Based on this indirect evidence, it is hypothesized that reversible cardiomyopathy in our patient may have represented cardiotoxicity due to a cannabis-related, CB1-mediated signaling mechanism along with a hyper-catecholaminergic state in a patient with recent myocardial infarction.
In addition, there have been reported cases of rhabdomyolysis with SC use (5, 11). Our patient had severe hypophosphatemia, hypokalemia, and hyperthermia on presentation, all of which may contribute to acute rhabdomyolysis, along with his acute illness and immobilization. SC has also been postulated to have serotonin-like or weak monoamine oxidase inhibitor properties (17, 18). This suggests the possibility of serotonin syndrome, though there have been no case reports linking that condition to the use of SC. Finally, our patient had high temperature on initial presentation but not thereafter, and there was no rigidity. Like other reported cases of rhabdomyolysis associated with SC, K2 metabolites may have contributed to the significant rise of CPK over time in our patient; further research is necessary to understand the underlining mechanism and elucidate specific treatments.
Metabolic derangements with hypokalemia, insulin resistance, and acute kidney injury have all been reported in association with SC use (19, 20). The close relationship between CB2 receptors and obesity-associated inflammation, insulin resistance, and liver disease has resulted in significant interest in developing CB2 receptor antagonists as a potential treatment of the metabolic syndrome (19, 21). However, it is not clear how acute intake of SC can result in such dramatic metabolic derangements, and further investigation is necessary to confirm any causal relationships.
Limitations and conclusion
Unfortunately, specific urine toxicology testing for K2 metabolites was not available in our patient. However, once the patient regained alertness, he confirmed the daily use of K2 for several weeks prior to presentation. This case highlights the diverse spectrum and severity of abnormalities associated with the use of SCs, and the potential for all of these deleterious cardiovascular, neurological, and metabolic effects to occur simultaneously. Physicians should consider appropriate laboratory testing in patients with suspected substance abuse. Further investigation is needed to explore the diverse toxic effects and propose specific treatment of these SCs.
Acknowledgement
We would like to thank Dr. Robin Shrestha MD, Critical care fellow at University of Maryland Medical Center, Dr. Thaddeus Pula MD, (Neurology) and Dr. Sasa Machala MD (Critical care) at University of Maryland Medical Center Midtown Campus.
Conflict of interest and funding
The authors have not received any funding or benefits from industry or elsewhere to conduct this study.
References
- 1.Dewey WL. Cannabinoid pharmacology. Pharmacol Rev. 1986;38:151–78. doi: 10.1016/0378-8741(87)90061-4. [DOI] [PubMed] [Google Scholar]
- 2.Rosenbaum CD, Carreiro SP, Babu KM. Here today, gone tomorrow … and back again? A review of herbal marijuana alternatives (K2, Spice), synthetic cathinones (bath salts), kratom, salvia divinorum, methoxetamine, and piperazines. J Med Toxicol. 2012;8:15–32. doi: 10.1007/s13181-011-0202-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Seely KA, Lapoint J, Moran JH, Fattore L. Spice drugs are more than harmless herbal blends: A review of the pharmacology and toxicology of synthetic cannabinoids. Prog Neuropsychopharmacol Biol Psychiatry. 2012;39:234–43. doi: 10.1016/j.pnpbp.2012.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mir A, Obafemi A, Young A, Kane C. Myocardial infarction associated with use of the synthetic cannabinoid K2. Pediatrics. 2011;128:e1622–7. doi: 10.1542/peds.2010-3823. [DOI] [PubMed] [Google Scholar]
- 5.Durand D, Delgado LL, de la Parra-Pellot DM, Nichols-Vinueza D. Psychosis and severe rhabdomyolysis associated with synthetic cannabinoid use. Clin Schizophr Relat Psychoses. 2013;8:1–13. doi: 10.3371/CSRP.DUDE.031513. [DOI] [PubMed] [Google Scholar]
- 6.Ukaigwe A, Karmacharya P, Donato A. A gut gone to pot: A case of cannabinoid hyperemesis syndrome due to K2, a synthetic cannabinoid. Case Rep Emerg Med. 2014;2014:167098. doi: 10.1155/2014/167098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yücel M, Solowij N, Respondek C, Whittle S, Fornito A, Pantelis C, et al. Regional brain abnormalities associated with long-term heavy cannabis use. Archives of General Psychiatry. 2008;65:694–701. doi: 10.1001/archpsyc.65.6.694. [DOI] [PubMed] [Google Scholar]
- 8.Center for Substance Abuse Research. Synthetic marijuana third most reported substance used by U.S. high school students. CESAR Fax. 2013;22(17) http://www.cesar.umd.edu/cesar/cesarfax/vol22/22-17.pdf. [Google Scholar]
- 9.McSherry JW. Spice, pot, and stroke. Neurology. 2014;82:2147. doi: 10.1212/WNL.0000000000000508. [DOI] [PubMed] [Google Scholar]
- 10.Freeman MJ, Rose DZ, Myers MA, Gooch CL, Bozeman AC, Burgin WS. Ischemic stroke after use of the synthetic marijuana “spice”. Neurology. 2013;81:2090–3. doi: 10.1212/01.wnl.0000437297.05570.a2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tanei T, Morita Y, Yashima A, Wakita H, Takebayashi S, Nakahara N, et al. Severe rhabdomyolysis and intracranial hemorrhage associated with synthetic cannabinoid: A case report. No Shinkei Geka. 2014;42:867–71. [PubMed] [Google Scholar]
- 12.Thomas G, Kloner RA, Rezkalla S. Adverse cardiovascular, cerebrovascular, and peripheral vascular effects of marijuana inhalation: What cardiologists need to know. Am J Cardiol. 2014;113:187–90. doi: 10.1016/j.amjcard.2013.09.042. [DOI] [PubMed] [Google Scholar]
- 13.Gottschalk LA, Aronow WS, Prakash R. Effect of marijuana and placebo-marijuana smoking on psychological state and on psychophysiological cardiovascular functioning in angina patients. Biol Psychiatry. 1977;12:255–e266. [PubMed] [Google Scholar]
- 14.Ting JYS. Reversible cardiomyopathy associated with acute inhaled marijuana use in a young adult. Clin Toxicol (Phila) 2007;45:432–4. doi: 10.1080/15563650601073587. [DOI] [PubMed] [Google Scholar]
- 15.Kaushik M, Alla VM, Madan R, Arouni AJ, Mohiuddin SM. Recurrent stress cardiomyopathy with variable regional involvement: Insights into etiopathogenetic mechanisms. Circulation. 2011;124:e556–7. doi: 10.1161/CIRCULATIONAHA.111.059329. [DOI] [PubMed] [Google Scholar]
- 16.Mukhopadhyay P, Rajesh M, Bátkai S, Patel V, Kashiwaya Y, Liaudet L, et al. CB1 cannabinoid receptors promote oxidative stress and cell death in murine models of doxorubicin-induced cardiomyopathy and in human cardiomyocytes. Cardiovasc Res. 2010;85:773–84. doi: 10.1093/cvr/cvp369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.El-Subbagh H, Wittig T, Decker M, Elz S, Nieger M, Lehmann J. Dopamine/serotonin receptor ligands. Part IV [1]: Synthesis and pharmacology of novel 3-benzazecines and 3-benzazonines as potential 5-HT2A and dopamine receptor ligands. Arch Pharm (Weinheim) 2002;335:443–8. doi: 10.1002/1521-4184(200212)335:9<443::AID-ARDP443>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 18.Fisar Z. Inhibition of monoamine oxidase activity by cannabinoids. Naunyn Schmiedebergs Arch Pharmacol. 2010;381:563–72. doi: 10.1007/s00210-010-0517-6. [DOI] [PubMed] [Google Scholar]
- 19.Hermanns-Clausen M, Kneisel S, Szabo B, Auwärter V. Acute toxicity due to the confirmed consumption of synthetic cannabinoids: Clinical and laboratory findings. Addiction. 2013;108:534–44. doi: 10.1111/j.1360-0443.2012.04078.x. [DOI] [PubMed] [Google Scholar]
- 20.Bhanushali GK, Jain G, Fatima H, Leisch LJ. Thornley-Brown D. AKI associated with synthetic cannabinoids: A case series. Clin J Am Soc Nephrol. 2013;8:5236. doi: 10.2215/CJN.05690612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Deveaux V, Cadoudal T, Ichigotani Y, Teixeira-Clerc F, Louvet A, Manin S, et al. Cannabinoid CB2 receptor potentiates obesity-associated inflammation, insulin resistance and hepatic steatosis. PLoS One. 2009;4:e5844. doi: 10.1371/journal.pone.0005844. [DOI] [PMC free article] [PubMed] [Google Scholar]