Abstract
Background. Human immunodeficiency virus (HIV)–infected individuals are at higher risk for chronic kidney disease than HIV-uninfected individuals. We investigated whether the inflammation present in treated HIV infection contributes to kidney dysfunction among HIV-infected men receiving highly active antiretroviral therapy.
Methods. The glomerular filtration rate (GFR) was directly measured (using iohexol) along with 12 markers of inflammation in Multicenter AIDS Cohort Study participants. Exploratory factor analysis was used to identify inflammatory processes related to kidney dysfunction. The estimated levels of these inflammatory processes were used in adjusted logistic regression analyses evaluating cross-sectional associations with kidney function outcomes.
Results. There were 434 HIV-infected men receiving highly active antiretroviral therapy and 200 HIV-uninfected men. HIV-infected men were younger (median age, 51 vs 53 years) and had higher urine protein-creatinine ratios (median, 98 vs 66 mg/g) but comparable GFRs (median, 109 vs 106 mL/min|1.73 m2). We found an inflammatory process dominated by markers: soluble tumor necrosis factor receptor 2, soluble interleukin 2 receptor α, soluble gp130, soluble CD27, and soluble CD14. An increase of 1 standard deviation in that inflammatory process was associated with significantly greater odds of GFR ≤90 mL/min/1.73 m2 (odds ratio, 2.0) and urine protein >200 mg/g (odds ratio, 2.3).
Conclusions. Higher circulating levels of immune activation markers among treated HIV-infected men may partially explain their higher burden of kidney dysfunction compared with uninfected men.
Keywords: HIV infection, inflammatory markers, chronic kidney disease, glomerular filtration rate, immune activation
Chronic immune activation is a feature of human immunodeficiency virus (HIV) infection and many chronic conditions. Proinflammatory cytokine levels, such as interleukin 6 (IL-6) and tumor necrosis factor (TNF) α, are elevated in HIV-infected individuals [1, 2] and can activate or enhance HIV replication [3–5] Even with highly active antiretroviral therapy (HAART)–induced viral suppression, immune activation persists with elevated levels of inflammatory cytokines that are predictive of disease outcomes [6]. Given this heightened inflammatory context, HIV-infected individuals may be at higher risk for diseases in which inflammation plays a contributory role.
Inflammation is considered a hallmark of chronic kidney disease (CKD), and higher levels of IL-6 and TNF-α have been noted in persons with CKD [7–9]. The extent to which proinflammatory cytokines are related to decline in glomerular filtration rate (GFR) remains unclear. High levels of inflammatory markers among persons with normal kidney function predict future CKD [10]. Furthermore, inflammation in the context of CKD is strongly associated with progression and CKD sequelae, such as cardiovascular disease [11] and anemia [12].
Compared with HIV-uninfected individuals, HIV-infected individuals are at higher risk for CKD [13–22]. Prior studies have established high prevalence of abnormal urine protein [23] and hyperfiltration [24] in HIV-infected compared with HIV-uninfected men, both indicators of early kidney damage and harbingers of future kidney function decline. Although other factors probably contribute to kidney damage among HIV-infected persons, inflammation that persists even in treated HIV infection may play a role. Most studies of this question have analyzed single inflammatory markers; however, inflammatory pathways are complex involving multiple markers. Using data from the Multicenter AIDS Cohort Study (MACS), we sought to characterize the relationship between inflammatory pathways and indicators of kidney damage among HIV-infected men and HIV-uninfected men at risk for HIV infection. We hypothesized that the heightened inflammation of HIV infection contributes to the higher risk of CKD in this population.
METHODS AND METHODS
Multicenter AIDS Cohort Study
The MACS is an ongoing multicenter prospective observational study established in 1984 of HIV-infected and HIV-uninfected men who have sex with men in Baltimore, Maryland–Washington, DC; Chicago, Illinois; Los Angeles, California; and Pittsburgh, Pennsylvania [25]. Semiannual visits include standardized questionnaires, physical examinations, and blood and urine collections for laboratory analyses and storage, with informed consent from each participant and approval from each local institutional review board.
Renal Substudy Design and Measurement of GFR
Direct measurement of GFR was performed in selected MACS participants from August 2008 to December 2010 [23]; briefly, HIV-infected men receiving HAART and HIV-uninfected men were selected from those without prior dialysis or transplant, recent diagnoses of cancer, or allergy to contrast agents. All men with hepatitis C virus (HCV) infection (defined by the presence of circulating anti-HCV antibody or HCV RNA, as described elsewhere [26]) were eligible. Plasma iohexol concentrations were measured using high-performance liquid chomatography (University of Rochester Medical Center GFR Laboratory) at 10, 30, 120, and 240 minutes after infusion of iohexol (5 mL; approximately 3200 mg) [27–29]. GFR was calculated using a 2-compartment model describing the fast and slow components of plasma iohexol concentration decay over time [27–30] and normalized to calculated body surface area [31]. Serum creatinine was measured with high-performance liquid chomatography [32]. Cystatin C was measured with immunonephelometry using a BN II analyzer (Siemens Diagnostics). The urine protein-creatinine ratio (uPCr) was calculated from a spot urine sample (Quest Diagnostics).
The present study included men with measured GFR for whom inflammatory markers were measured (see below) within the 5 years before GFR measurement. This interval was chosen to maximize sample size and because data from HIV-infected men on HAART in the MACS indicate very little change in biomarker levels beyond 1 year after suppression of HIV viral load [33].
Measurement of Inflammatory Markers
A total of 22 chemokines, cytokines, and soluble receptors and C-reactive protein (CRP) were measured at selected MACS visits. Of these markers, 12 were selected for the present study, based on literature review, preliminary analyses indicating likely candidates for relation to kidney disease, and detectability in ≥90% of samples: CRP, soluble TNF receptor 2 (sTNFr2), soluble interleukin 2 receptor α (sIL2rα), soluble gp130 (sgp130), soluble CD27 (sCD27), monocyte chemoattractant protein 1, interferon γ-induced protein 10 (IP10/CXCL10), TNF-α, IL-6, soluble CD14 (sCD14), interleukin 8 (CXCL8), and interleukin 10 (IL-10). (Markers not included in the analysis were interleukin 12p70, interleukin 1β, thymus- and activation-regulated chemokine, B-lymphocyte chemoattractant, B-cell activating factor, eotaxin, interferon γ, macrophage inflammatory protein 1β, monocyte chemoattractant protein 1, and interleukin 2).
Serum concentrations of IL-6, TNF-α, CXCL8, interleukin 10, CXCL10, and CCL2 were assessed using electrochemiluminesence-based multiplex assays (Meso-Scale Diagnostics; Proinflammatory 9-plex and Chemokine 7-plex) at a laboratory at the Johns Hopkins Bloomberg School of Public Health. The lower limit of detection for each plate-specific analyte was the concentration 2.5 standard deviations (SDs) above the background. Serum concentrations of sTNFr2, sIL2rα, sCD14, sgp130, and sCD27 were determined using a fluorescent bead-based multiplexed Luminex xMAP system at a laboratory at the University of California, Los Angeles, using a single assay lot (Fluorokine MAP; R&D Systems), a Bio-Plex 200 Luminex instrument, and Bio-Plex software (Bio-Rad). Concentrations of sIL2rα, sTNFr2, sgp130, and sCD14 were measured in a single panel (Human Biomarker Custom Premix Kit A). The lower limit of detection for each plate-specific analyte was the observed concentration of the lowest standard. CRP was measured at Quest Diagnostics with a high-sensitivity assay (lower limit of detection, 0.2 mg/L; Dade Behring).
Covariate Measurements
Hypertension, diabetes, and dyslipidemia were considered present if confirmed at ≥2 visits in any 1-year period before and including the GFR measurement. Hypertension was defined as a systolic blood pressure ≥140 mm Hg, a diastolic blood pressure ≥90 mm Hg, or receipt of antihypertensive medication with a self-reported history of hypertension. Similarly, diabetes mellitus was defined as a fasting glucose level ≥126 mg/dL or receipt of glucose-lowering medication with a history of a diagnosis of diabetes. Dyslipidemia was defined as receipt of lipid-lowering medication with a history of dyslipidemia or the presence of any of the following fasting measurements: total serum cholesterol ≥200 mg/dL, low-density lipoprotein ≥130 mg/dL, high-density lipoprotein <40 mg/dL, or triglycerides ≥150 mg/dL.
Prevalent HCV was defined as a reactive HCV antibody or detectable HCV RNA level. HCV antibody-positive participants with undetectable plasma HCV RNA for >3 years were considered HCV uninfected. Plasma HIV RNA levels were measured using the Roche Amplicor assay (Hoffman-LaRoche), sensitive to 50 copies/mL. The CD4+ T-cell count was measured with 3-color flow cytometry [34]. Demographic and clinical characteristics were obtained from the GFR visit or, if missing, the closest visit within 1 year.
Kidney Outcome Definitions
Low kidney function was defined by GFR ≤90 mL/min/1.73 m2, consistent with Kidney Disease Improving Global Outcomes (KDIGO) CKD stage ≤2 [35]. This threshold was chosen because very few men in the MACS have a GFR <60 mL/min/1.73 m2, a more standard threshold for defining kidney disease. Hyperfiltration was defined as a GFR ≥140 mL/min/1.73 m2 for men aged ≤40 years, subtracting 1 mL/min/1.73 m2 from this threshold for each year of age >40, as described elsewhere [24]. High urine protein, an indicator of glomerular damage, was defined as uPCr >200 mg/g.
Statistical Analysis
Descriptive Analysis of Inflammatory Biomarkers
Biomarker values were natural log-transformed for comparison across HIV serostatus and GFR strata. Differences in biomarker distributions between HIV-infected and HIV-uninfected men and between men with GFRs ≤90 mL/min|1.73 m2 and those with GFRs >90 mL/min|1.73 m2 were assessed using nonparametric Kruskal–Wallis tests.
Handling of Inflammatory Biomarker Data Below the Lower Limit of Detection
Fifty-seven biomarker observations (from 7 markers) lay below their lower limits of detection. Because the exploratory factor analyses (EFAs; see below) required numerical values for all data, we imputed values for these 57 observations, using truncated log-normal distributions for each inflammatory marker. Specifically, we fit a normal distribution to the observed data, extrapolated the curve below the limit of detection, and drew values randomly from the distribution below the limit of detection.
EFA Methods
We used EFA to find combinations of inflammatory markers that indicated unique inflammatory processes. EFA identifies underlying processes by finding groups of markers that are highly correlated and thus likely to reflect the same process. To use EFA, biomarker values (observed and imputed) were standardized to create distributions with a mean of 0 and an SD of 1.
Assuming that biomarkers were linearly related to underlying inflammatory processes, the analyses yielded weighted linear combinations of the biomarkers (called factor scores) that represented the magnitude of the identified underlying inflammatory processes. The weights (hereafter called factor loadings) are thus, the strength and direction of the relationship between each inflammatory biomarker and the underlying inflammatory process. For the main analysis, we only retained identified inflammatory processes where the sum of the squared factor loadings were >1, indicating that the process was well described by the individual marker data. To assess the repeatability of our EFA-identified inflammatory processes, we performed the identical EFA on an HIV-uninfected secondary sample, comprising all HIV-uninfected men with biomarker measurements, using only the first MACS visit with measurements. The serum for these measurements was collected from June 1984 to September 2007. Fuller details of the EFA methods are presented in the Supplementary Appendix.
Analysis of the Relationship Between Inflammatory Processes and Kidney Outcomes
The estimated individual levels of the inflammatory processes were used as predictors in logistic regressions evaluating their associations with the following kidney outcomes: low versus normal GFR (≤90 vs >90 to <140 mL/min/1.73 m2); hyperfiltration (GFR ≥ 140 mL/min/1.73 m2 or age-corrected threshold if for men aged >40 years) versus normal GFR; and high versus normal urine protein level (>200 vs ≤200 mg/g). Relationships between the inflammatory processes and comorbid CKD risk factor outcomes, including diabetes, hypertension, and HCV, were assessed in separate logistic regressions. All models were adjusted for age >50 years, black race, and HIV serostatus. Analyses were done in the full sample as well as stratified by HIV status. Results were expressed as odds ratios (ORs) for the outcome associated with a 1-SD increase in the level of the inflammatory process. In sensitivity analyses, models for kidney outcomes were adjusted for CKD comorbid risk factors. Interactions between inflammatory processes and black race were also investigated.
RESULTS
MACS Renal Substudy
A total of 741 men underwent iohexol studies; 715 studies (96%) yielded satisfactory GFR measurements, comprising 457 HIV-infected and 258 HIV-uninfected men. As has been reported [23], the men in the renal substudy were similar to overall MACS participants but were more likely to be black (by design), to be HCV infected (by design), and to have slightly lower serum creatinine values (P < .01). Of these 715 men, 634 (434 HIV infected and 200 HIV uninfected) had inflammatory biomarker measurements and were included in the present analysis. Seventy-five percent of the biomarker measurements were from samples collected within 1.3 years of the GFR measurement.
Description of Included Men (GFR Sample)
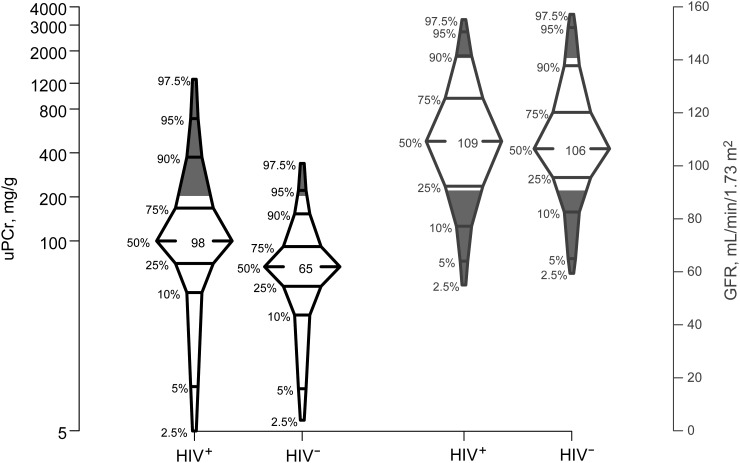
As shown in Table 1, the median age of the men was 52 years, their median body mass index (BMI) was 26 kg/m2, and 37% were black. Ninety-eight men had HCV infection; 30 others had cleared HCV. The HIV-infected men tended to be younger (51 vs 53 years) and had lower BMIs (26 vs 27 kg/m2), higher levels of cystatin C (0.79 vs 0.75 mg/L), and higher urine protein (uPCr 98 vs 65 mg/g) than HIV-uninfected men. Although the median GFR and serum creatinine levels did not differ by HIV serostatus, more HIV-infected men occupied both the higher (≥140 mL/min/1.73 m2) and lower (≤90 mL/min/m2) tails of the GFR distribution, compared with HIV-uninfected men (P = .04; Figure 1); Figure 1 also shows that HIV-infected men had higher uPCr values.
Table 1.
Demographic and Clinical Characteristics of HIV-Infected and HIV-Uninfected Men From the Multicenter AIDS Cohort Study
| Characteristic | Median (IQR)a |
||
|---|---|---|---|
| HIV Uninfected (n = 200) | HIV Infected (n = 434) | Overall (N = 634) | |
| Black, % | 38 | 37 | 37 |
| Age, yb | 53 (48–60) | 51 (46–57) | 52 (47–58) |
| Weight, kgb | 84 (75–94) | 79 (71–89) | 81 (72–91) |
| BMI, kg/m2b | 27 (24–30) | 26 (23–28) | 26 (24–29) |
| BSA, m2b | 2.0 (1.9–2.2) | 2.0 (1.9–2.1) | 2.0 (1.9–2.1) |
| Serum creatinine, mg/dL | 0.88 (0.78–1.02) | 0.87 (0.75–1.04) | 0.88 (0.76–1.03) |
| Cystatin C, mg/Lb,c | 0.75 (0.67–0.85) | 0.79 (0.69–0.94) | 0.77 (0.68–0.90) |
| uPCr, mg/gb,c | 65 (48–90) | 98 (69–165) | 85 (60–138) |
| GFR, mL/min/1.73 m2 | 106 (95–120) | 109 (92–125) | 107 (92–123) |
| HCV-infected, %c | 16 | 16 | 15 |
| History of risk factors, % | |||
| Diabetes mellitus | 14 | 17 | 16 |
| Hypertension | 59 | 56 | 57 |
| Dyslipidemiab | 78 | 92 | 87 |
| HIV-related factors | |||
| Time since HIV-infection, y | … | 19 (19–26) | … |
| Time since HAART initiation, y | … | 10 (7–12) | … |
| History of AIDS, % | … | 15 | … |
| HIV RNA <50 copies/mL, % | … | 80 | … |
| CD4+ T-cell count, cells/μL | … | 544 (388–741) | … |
Abbreviations: BMI, body mass index; BSA, body surface area; GFR, glomerular filtration rate; HAART, highly active antiretroviral therapy; HCV, hepatitis C virus; HIV, human immunodeficiency virus; IQR, interquartile range; uPCr, urine protein-creatinine ratio.
a Data represent median (IQR) values unless otherwise specified. Values were obtained from the kidney function assessment visit or the nearest visit before that.
b P < .05 (χ2 test or Kruskal–Wallis for comparison between HIV-uninfected and HIV-infected participants).
c Data were missing on HCV status for 4 participants, on cystatin C level, for 3, and on uPCr for 8.
Figure 1.
Percentile box plots displaying the distribution of glomerular filtration rate (GFR) and urine protein-creatinine ratio (uPCr) among the sample of human immunodeficiency virus (HIV)–infected (HIV+) and HIV-uninfected (HIV−) men from the Multicenter AIDS Cohort Study. The shaded areas represent proportion with GFR values ≥140 mL/min/1.73 m2 or ≤90 mL/min/1.73 m2 (right) and proportion with uPCr >200 mg/g (left), comparing HIV-infected with HIV-uninfected samples.
HIV-infected and HIV-uninfected men had similar prevalences of diabetes (16% overall), hypertension (57% overall) and HCV (16% overall), but HIV-infected men had a higher prevalence of dyslipidemia (92% vs 78%) Among HIV-infected men, the median CD4 + T-cell count was 544/µL, 80% had undetectable HIV RNA levels, 79% had been exposed to tenofovir disoproxil fumarate, and 65% had used tenofovir disoproxil fumarate within the past 6 months. The median time since HIV infection was 19 years and the median time receiving HAART was 10 years.
Inflammatory Biomarker Levels
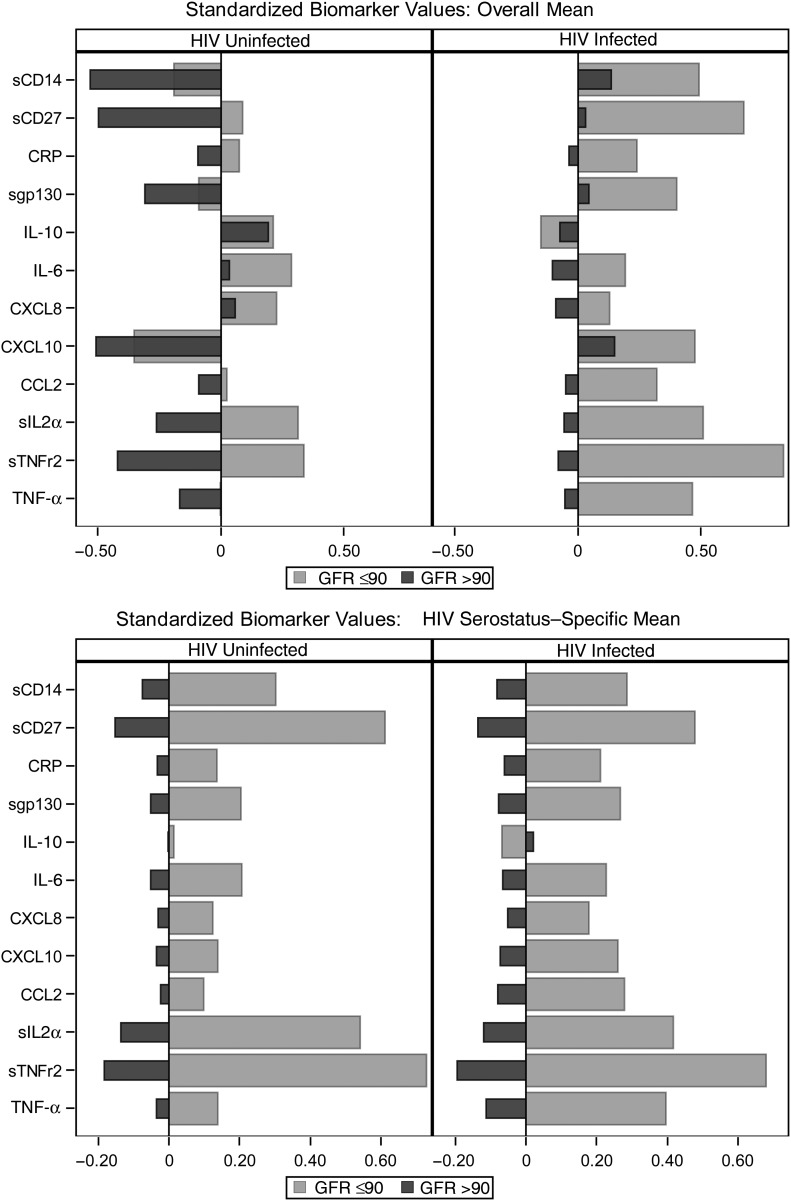
The standardized biomarker profiles are shown in Figure 2. The upper panel, with biomarker levels standardized to an overall mean, demonstrates that HIV-infected men generally had higher levels of inflammatory markers than HIV-uninfected men: sTNFr22, sIL2rα, sgp130, sCD27, CXCL10, TNF-α, sCD14, and IL-10 levels were all significantly higher among HIV-infected men. With the exception of IL-10, men with GFRs ≤90 mL/min/m2 had significantly higher levels of inflammatory markers than those with GFRs >90 mL/min/m2. When the biomarkers were standardized to a serostatus-specific mean (Figure 2, lower panel), the inflammatory marker profiles stratified by GFR ≤90 mL/min/m2 were similar between HIV-infected and HIV-uninfected men.
Figure 2.
Standardized biomarker profiles showing the average standardized levels of the 12 inflammatory biomarkers stratified by human immunodeficiency virus (HIV) serostatus and by glomerular filtration rate (GFR; ≤90 vs >90 mL/min/1.73 m2). Upper panel shows biomarker values standardized to an overall mean and standard deviation for each biomarker, ignoring HIV serostatus. Lower panel shows biomarker values standardized to a serostatus-specific mean for each biomarker. Abbreviations: CCL2, monocyte chemoattractant protein 1; CRP, C-reactive protein; CXCL8, interleukin 8; CXCL10, interferon γ–induced protein 10; IL-6, interleukin 6; IL-10, interleukin 10; sCD14, soluble CD14; sCD27, soluble CD27; sgp130, soluble gp130; sIL2rα, soluble interleukin 2 receptor α; sTNFr2, soluble tumor necrosis factor (TNF) receptor 2.
EFA Results
The correlations among the measured inflammatory markers are shown in Table 2, and the results from the EFA in Table 3. Three inflammatory processes were identified, accounting for 60% of the total variance in the biomarker data. Inflammatory process 1 was characterized mainly by the biomarkers sTNFr2, sIL2rα, sgp130, sCD27, and sCD14; inflammatory process 2, by IL-6, CXCL8, and TNF-α; and inflammatory process 3, by CXCL10 and CCL2.
Table 2.
Correlations Between the 12 Cytokines, Chemokines, and Soluble Receptors in the GFR Sample and the HIV-Uninfected Secondary Sample
| Correlation |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Marker | sCD14 | sCD27 | CRP | sgp130 | IL-10 | IL-6 | CXCL8 | CXCL10 | CCL2 | sIL2rα | sTNFr2 | TNF-α |
| GFR sample (n = 634) | ||||||||||||
| sCD14 | 1.00 | 0.43a,b | 0.24b | 0.46a,b | 0.00 | 0.09 | 0.01 | 0.19b | 0.04 | 0.42a,b | 0.51a,b | 0.19b |
| sCD27 | … | 1.00 | 0.17b | 0.42a,b | 0.10c | 0.19b | 0.05 | 0.40a,b | −0.01 | 0.70a,b | 0.78a,b | 0.38a,b |
| CRP | … | … | 1.00 | 0.07 | 0.10c | 0.28b | 0.09c | −0.01 | −0.02 | 0.25b | 0.28b | 0.17b |
| sgp130 | … | … | … | 1.00 | −0.01 | 0.08c | 0.02 | 0.23b | 0.03 | 0.46a,b | 0.54a,b | 0.14b |
| IL-10 | … | … | … | … | 1.00 | 0.27b | 0.08c | 0.14b | 0.07 | 0.19b | 0.14b | 0.27b |
| IL-6 | … | … | … | … | … | 1.00 | 0.52a,b | 0.12c | 0.06 | 0.23b | 0.25b | 0.56a,b |
| CXCL8 | … | … | … | … | … | … | 1.00 | −0.03 | 0.08 | 0.06 | 0.08c | 0.39b |
| CXCL10 | … | … | … | … | … | … | … | 1.00 | 0.40a,b | 0.41a,b | 0.47a,b | 0.3a,b |
| CCL2 | … | … | … | … | … | … | … | … | 1.00 | 0.07 | 0.09c | 0.08 |
| sIL2rα | … | … | … | … | … | … | … | … | … | 1.00 | 0.79a,b | 0.41a,b |
| sTNFr2 | … | … | … | … | … | … | … | … | … | … | 1.00 | 0.44a,b |
| TNF-α | … | … | … | … | … | … | … | … | … | … | … | 1.00 |
| HIV-uninfected secondary sample (n = 247) | ||||||||||||
| sCD14 | 1.00 | 0.31a,b | 0.20c | 0.29b | 0.02 | 0.09 | 0.01 | 0.15c | 0.02 | 0.35a,b | 0.39a,b | 0.14c |
| sCD27 | 1.00 | 0.06 | 0.40a,b | 0.00 | 0.15c | −0.00 | 0.43a,b | −0.12 | 0.70a,b | 0.78a,b | 0.21b | |
| CRP | … | … | 1.00 | 0.02 | 0.13c | 0.33a,b | 0.07 | 0.01 | 0.12 | 0.20c | 0.18c | 0.13c |
| sgp130 | … | … | … | 1.00 | −0.08 | 0.12 | 0.01 | 0.30a,b | −0.10 | 0.36a,b | 0.48a,b | 0.12 |
| IL-10 | … | … | … | … | 1.00 | 0.19c | 0.09 | 0.13b | 0.05 | 0.12 | 0.05 | 0.28b |
| IL-6 | … | … | … | … | … | 1.00 | 0.35a,b | 0.23b | 0.05 | 0.19c | 0.23b | 0.57a,b |
| CXCL8 | … | … | … | … | … | 1.00 | 0.03 | 0.21b | 0.01 | 0.07 | 0.33a,b | |
| CXCL10 | … | … | … | … | … | … | … | 1.00 | 0.06 | 0.38a,b | 0.53a,b | 0.29b |
| CCL2 | … | … | … | … | … | … | … | … | 1.00 | 0.02 | 0.02 | 0.08 |
| sIL2rα | … | … | … | … | … | … | … | … | … | 1.00 | 0.73a,b | 0.24b |
| sTNFr2 | … | … | … | … | … | … | … | … | … | … | 1.00 | 0.30a,b |
| TNF-α | … | … | … | … | … | … | … | … | … | … | … | 1.00 |
Abbreviations: CCL2, monocyte chemoattractant protein 1; CRP, C-reactive protein; CXCL8, interleukin-8; CXCL10, interferon γ–induced protein 10; GFR, glomerular filtration rate; HIV, human immunodeficiency virus; IL-6, interleukin 6; IL-10, interleukin 10; sCD14, soluble CD14; sCD27, soluble CD27; sgp130, soluble gp130; sIL2rα, soluble interleukin 2 receptor α; sTNFr2, soluble TNF receptor 2; TNF, tumor necrosis factor.
a Correlation coefficient ≥3.0 (moderately strong relationship).
b P < .001.
c .001 ≤ P < .05.
Table 3.
Factor Loadingsa Showing the Strength of Relationship Between Factors and Individual Biomarkers, Comparing Results Between GFR and HIV-Uninfected Secondary Samples
| Biomarker | Factor Loadings for Inflammatory Processes in GFR Sample |
Factor Loadings for Inflammatory Processes in Secondary Sample |
||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 1 | 2 | 3 | |
| sTNFr2 | 0.87b,c | 0.05 | 0.06 | 0.86c | 0.05 | 0.08 |
| sCD27 | 0.80b,c | 0.01 | 0.01 | 0.86c | −0.05 | −0.06 |
| sIL2rα | 0.77b,c | 0.07 | 0.06 | 0.77c | −0.01 | 0.18 |
| sgp30 | 0.66b,c | −0.13 | −0.06 | 0.66c | −0.06 | −0.19 |
| sCD14 | 0.66b,c | −0.06 | −0.14 | 0.46 | −0.17 | 0.44b |
| CRP | 0.29 | 0.25 | −0.24 | −0.00 | 0.10 | 0.81b,c |
| IL-6 | −0.02 | 0.78b,c | −0.04 | 0.08 | 0.73b,c | 0.14 |
| CXCL8 | −0.14 | 0.64b,c | −0.06 | −0.14 | 0.66b | 0.02 |
| TNF-α | 0.15 | 0.61b,c | 0.18 | 0.18 | 0.76b,c | −0.05 |
| IL-10 | −0.04 | 0.30 | 0.15 | −0.06 | 0.41b | 0.15 |
| CXCL10 | 0.19 | −0.02 | 0.62b,c | 0.58b,c | 0.26 | −0.21 |
| CCL2 | −0.18 | 0.02 | 0.55b | −0.19 | 0.20 | 0.43b |
| Variance explained, % | 33.56 | 15.25 | 11.02 | 30.24 | 15.58 | 9.15 |
| Cumulative variance | 33.56 | 48.80 | 59.82 | 30.24 | 45.82 | 54.97 |
Abbreviations: CCL2, monocyte chemoattractant protein 1; CRP, C-reactive protein; CXCL8, interleukin 8; CXCL10, interferon γ–induced protein 10; EFA, exploratory factor analysis; GFR, glomerular filtration rate; HIV, human immunodeficiency virus; IL-6, interleukin 6; IL-10, interleukin 10; sCD14, soluble CD14; sCD27, soluble CD27; sgp30, soluble gp30; sIL2rα, soluble interleukin 2 receptor α; sTNFr2, soluble tumor necrosis factor receptor 2; TNF, tumor necrosis factor.
a See “EFA Methods” in Statistical Analysis section.
b Salient factors using criterion of factor loadings >0.4.
c Salient factors using combined criteria of factor loadings >0.4 and α <.1 from maximum likelihood factor analysis.
Repeatability of the EFA Results (Secondary Sample)
EFA was also performed on inflammatory biomarker data from 247 HIV-uninfected men with a median age of 40 years and a median BMI of 25 kg/m2; 39% were black. Thirteen percent had a history of hypertension and 19% were HCV positive. Of these 247 men, 197 had later biomarker measurements included in the GFR sample described above. However, the biomarker measurements for this sample were from serum collected earlier, and the specimens were distinct from those analyzed in the GFR sample. The correlations among inflammatory markers (Table 2) as well as the factor loadings (Table 3) for the secondary sample were very similar to those for the GFR sample, with the exception of markers contributing to inflammatory process 3; inconsistent results with respect to CXCL10, CRP, and sCD14 factor loadings rendered inflammatory process 3 less reliable.
Relationship Between Renal Outcomes and Inflammatory Processes
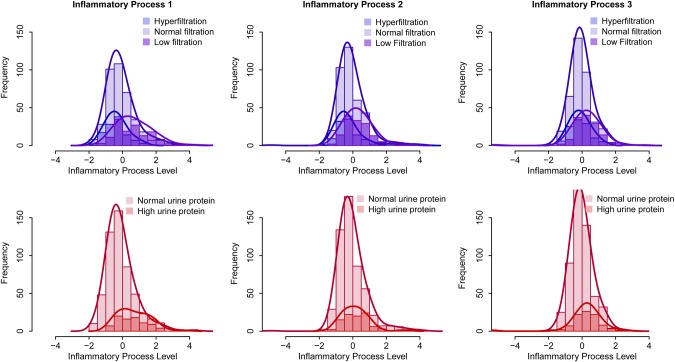
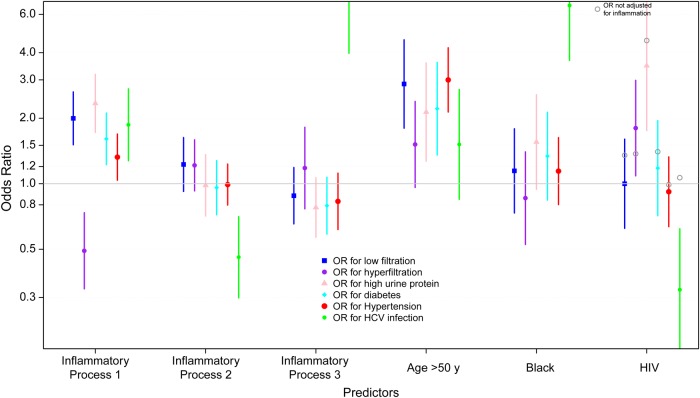
The distributions of the inflammatory processes, stratified by GFR outcome (hyperfiltration, normal filtration, or low filtration) and by proteinuria (normal or high urine protein) are presented in Figure 3. Distributions by HIV serostatus are given in Supplementary Figure A in the Appendix. The results of analyses adjusted for age, race and HIV serostatus, presented in Figure 4, show that inflammatory process 1 levels were significantly related to higher odds of GFR ≤90 mL/min|1.73 m2 (OR, 2.0), urine protein >200 mg/g (OR, 2.3), diabetes (OR, 1.6), hypertension (OR, 1.3), and HCV infection (OR, 1.9) and lower odds of hyperfiltration (OR, 0.5). Inflammatory process 2 levels were significantly related only to lower odds of HCV infection (OR, 0.5). Inflammatory process 3 levels were significantly related to higher odds of HCV infection (OR, 7.4). In analyses stratified by HIV serostatus, associations were similar in the 2 groups, except for a significant relationship between inflammatory process 2 and higher odds of hyperfiltration among HIV-infected men (OR, 1.5), an association which was not present in the HIV-uninfected men. Estimates and 95% confidence intervals are presented in Supplementary Table A in the Appendix.
Figure 3.
Distribution of inflammatory process levels for each of the 3 identified inflammatory processes. Upper panel, distribution stratified by kidney function: low filtration (glomerular filtration rate [GFR] ≤90 mL/min/1.73 m2) hyperfiltration (GFR ≥140 mL/min/1.73 m2 for men aged ≤40 years, subtracting 1 mL/min/1.73 m2 for each year of age >40), or normal filtration (GFR >90 to <140 mL/min/1.73 m2). Lower panel, distribution stratified by normal versus high urine protein levels (urine protein-creatinine ratio, ≤200 vs >200 mg/g).
Figure 4.
Results from adjusted logistic regressions showing the estimated odds ratios (ORs) for the associations between the levels of the 3 inflammatory processes and 6 outcomes: low kidney filtration (glomerular filtration rate [GFR] ≤90 mL/min/1.73 m2), hyperfiltration (GFR ≥140 mL/min/1.73 m2 for men aged ≤40 years, subtracting 1 mL/min/1.73 m2 for each year of age >40), high urine protein-creatinine ratio (>200 mg/g), diabetes, hypertension, and hepatitis C virus (HCV) infection. Models were adjusted for all the predictors noted. Effect estimates associated with human immunodeficiency virus (HIV) serostatus are also presented, without adjustment for inflammatory process levels.
In models controlling for inflammatory process levels, positive associations were noted between age >50 years and the following outcomes: GFR ≤ 90 mL/min|1.73 m2, urine protein >200 mg/g, diabetes, and hypertension. Black race was associated only with higher odds of HCV infection. HIV- positivity was significantly associated with higher odds of hyperfiltration, urine protein >200 mg/g, and lower odds of HCV infection. Although HIV seropositivity was not significantly associated with GFR ≤90 mL/min|1.73 m2 in the fully adjusted model (OR, 1.0), the point estimate rose to 1.4 (P = .18) when the inflammatory process levels were not included. Tenofovir disoproxil fumarate use was not significantly associated with any of the kidney outcomes. Interactions between black race and inflammatory process levels were explored, but no significant modifying effects were found. Finally, sensitivity analyses including comorbid risk factors in the kidney outcome models did not change inferences.
DISCUSSION
The degree to which inflammation contributes to the risk of non-AIDS comorbid conditions in HIV-infected populations is an open question. Behavioral factors, such as substance use, along with HAART toxic and metabolic effects, probably explain some of the additional risk. However, a growing body of literature suggests that higher levels of inflammation and immune activation in HIV-infected treated adults are independently associated with a higher risk of age-related chronic disease and mortality [36–40].
Our analysis detected 3 underlying inflammatory processes: inflammatory processes 1 and 2 were found in both our predominantly HIV-infected GFR sample and an all HIV-uninfected sample, and relationships with kidney outcomes were consistent across strata of HIV serostatus, suggesting similar underlying inflammatory processes contributing to CKD risk in HIV-infected and HIV-uninfected men. In contrast, inflammatory process 3 differed between the 2 groups in terms of contributing biomarkers, which could result from measurement variability or suggest different inflammatory process in HIV-infected versus HIV-uninfected men.
The primary associations with kidney outcomes found in the present study were with inflammatory process 1. Because sTNFr2, sCD27, sIL2Rα, sgp130, and sCD14, the primary markers involved in inflammatory process 1, are generally involved in immune activation, inflammatory cell responses, or initiation of apoptosis, our results support a role of immune cell activation in kidney dysfunction and damage. Consistent with other studies, levels of these markers were higher in HIV-infected than in HIV-uninfected men.
One fundamental question is whether HIV infection is a driver of inflammation contributing to kidney damage or whether higher levels of immune activation markers result from decreased clearance due to kidney dysfunction [41, 42]. Supporting the former explanation, we noted that controlling for inflammatory markers attenuated the effect estimate for HIV-positive serostatus on low filtration risk to the null. This could suggest that reduced kidney filtration in HIV-infected men may be partly explained by HIV-associated immune activation, the only significant predictor of this kidney outcome other than older age. However, longitudinal studies are needed to answer this question more definitively.
Comorbid risk factors for CKD were related to higher levels of inflammatory process 1, which could indicate that the primary pathway whereby hypertension, diabetes and HCV contribute to kidney dysfunction is through an inflammatory process. Under this assumption, we did not adjust for these factors in our main analyses of kidney outcomes. This could potentially introduce confounding bias if other important noninflammatory causal pathways exist, although adding these comorbid conditions to models had no effect on estimates.
The mechanism whereby these markers and a process of immune activation may promote kidney damage is speculative. Recent evidence suggests that immune activation may be particularly detrimental in individuals carrying apolipoprotein L1 (APOL1) risk variants; these variants are predominantly found in blacks and are linked to certain kidney diagnoses among HIV-infected individuals [43]. Cytokine-induced overexpression of APOL1 risk variants may be more harmful to cells than overexpression of wild-type APOL1 protein [44, 45]. Consistent with the notion of inflammation-mediated damage to the kidney, up-regulation of proinflammatory cytokines is a prominent feature of HIV-1 infection of renal tubule and endothelial cells [46, 47]. These findings may suggest an inflammatory pathogenesis for some kidney diseases among HIV-infected individuals and or a possible gene-by-inflammation interaction, though in the present study interaction terms between inflammatory factor scores and black race were not significant.
HIV seropositivity was significantly associated with risk of both high urine protein and hyperfiltration, the latter of which was associated with lower levels of inflammatory process 1. Prior work in the MACS indicated that hyperfiltration in HIV-infected men may result from cumulative antiretroviral exposure and blood glucose dysregulation [24]; thus, immune activation may play a lesser role in this process. Alternatively, hyperfiltration could result in greater clearance of these markers and lower serum levels. Both HIV seropositivity and higher levels of inflammatory process 1 were independently associated with higher urine protein; HIV may contribute to glomerular damage through inflammatory and noninflammatory pathways.
One strength of the present study was the direct assessments of GFR, resulting in less measurement error than estimated GFR, which can be affected by diet, health, and age. Moreover, estimating formulas are less accurate in persons with near-normal kidney function, as in this study. Although our marker measurements reflected systemic, rather than renal, inflammation, higher systemic inflammatory levels among HIV-infected individuals have been shown to reflect higher renal interstitial and glomerular levels [48]. Most marker measurements were made within 2 years of the GFR measurement, but we used measurements from up to 5 years prior. Inflammatory markers have high correlations across this span of time in the MACS, but there is the potential for changes in therapy or health to have occurred in the intervening period. In our sample, 2 men initiated HAART and 1 seroconverted in the period between biomarker sampling and GFR measurement, which probably affected inflammatory biomarker levels for these individuals. However, earlier measurements could be more relevant for influencing current levels of kidney damage and dysfunction. Finally, we note that the MACS includes only men, and the findings may not apply to women. In summary, results of the present study suggest that an inflammatory process composed of markers of immune activation could be an important contributor to the higher burden of kidney disease in virally suppressed, HIV-infected persons.
Supplementary Data
Supplementary materials are available at The Journal of Infectious Diseases online (http://jid.oxfordjournals.org). Supplementary materials consist of data provided by the author that are published to benefit the reader. The posted materials are not copyedited. The contents of all supplementary data are the sole responsibility of the authors. Questions or messages regarding errors should be addressed to the author.
Notes
Acknowledgments. We would like to thank all the collaborators, staff, and participants of the Multicenter AIDS Cohort Study (MACS). Data in this manuscript were collected by the MACS at the following centers: Baltimore (U01-AI35042; The Johns Hopkins University [JHU] Bloomberg School of Public Health): Joseph B. Margolick (principal investigator [PI]), Jay Bream, Todd Brown, Barbara Crain, Adrian Dobs, Richard Elion, Richard Elion, Michelle Estrella, Lisette Johnson-Hill, Sean Leng, Anne Monroe, Cynthia Munro, Michael W. Plankey, Wendy Post, Ned Sacktor, Jennifer Schrack, and Chloe Thio; Chicago (U01-AI35039; Feinberg School of Medicine, Northwestern University, and Cook County Bureau of Health Services): Steven M. Wolinsky (PI), John P. Phair, Sheila Badri, Dana Gabuzda, Frank J. Palella, Jr, Sudhir Penugonda, Susheel Reddy, Matthew Stephens, and Linda Teplin; Los Angeles (U01-AI35040; University of California, UCLA Schools of Public Health and Medicine): Roger Detels (PI), Otoniel Martínez-Maza (co-PI), Aaron Aronow, Peter Anton, Robert Bolan, Elizabeth Breen, Anthony Butch, Shehnaz Hussain, Beth Jamieson, Eric N. Miller, John Oishi, Harry Vinters, Dorothy Wiley, Mallory Witt, Otto Yang, Stephen Young, and Zuo Feng Zhang; Pittsburgh (U01-AI35041l University of Pittsburgh, Graduate School of Public Health): Charles R. Rinaldo (PI), Lawrence A. Kingsley (co-PI), James T. Becker, Phalguni Gupta, Kenneth Ho, Susan Koletar, Jeremy J. Martinson, John W. Mellors, Anthony J. Silvestre, and Ronald D. Stall; Data Coordinating Center (UM1-AI35043; JHU Bloomberg School of Public Health): Lisa P. Jacobson (PI), Gypsyamber D'Souza (co-PI), Alison, Abraham, Keri Althoff, Jennifer Deal, Priya Duggal, Sabina Haberlen, Alvaro Muoz, Derek Ng, Janet Schollenberger, Eric C. Seaberg, Sol Su, and Pamela Surkan; Institute of Allergy and Infectious Diseases: Robin E. Huebner; and National Cancer Institute: Geraldina Dominguez. The MACS Web site is located at http://www.statepi.jhsph.edu/macs/macs.html.
Disclaimer. The contents of this publication are solely the responsibility of the authors and do not represent the official views of the National Institutes of Health (NIH), the JHU Institute for Clinical and Translational Research, or the National Center for Advancing Translational Sciences.
Financial support. This work was supported by the National Institute of Allergy and Infectious Diseases (grants U01-AI35039, U01-AI35040, U01-AI35041, U01-AI35042, and UM1-AI35043), with additional cofunding from the National Cancer Institute, the National Institute on Drug Abuse, and the National Institute of Mental Health. Targeted supplemental funding for specific projects in the MACS was provided by the National Heart, Lung, and Blood Institute and the National Institute on Deafness and Communication Disorders. MACS data collection is also supported by the National Center for Advancing Translational Sciences, NIH (grant UL1-TR001079 to the JHU Institute for Clinical and Translational Research) and the NIH Roadmap for Medical Research.
Potential conflicts of interest. All authors: No potential conflicts of interest.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Breen EC, Rezai AR, Nakajima K, et al. Infection with HIV is associated with elevated IL-6 levels and production. J Immunol 1990; 144:480–4. [PubMed] [Google Scholar]
- 2.Wright SC, Jewett A, Mitsuyasu R, Bonavida B. Spontaneous cytotoxicity and tumor necrosis factor production by peripheral blood monocytes from AIDS patients. J Immunol 1988; 141:99–104. [PubMed] [Google Scholar]
- 3.Gage JR, Breen EC, Echeverri A, et al. Human herpesvirus 8-encoded interleukin 6 activates HIV-1 in the U1 monocytic cell line. AIDS 1999; 13:1851–5. [DOI] [PubMed] [Google Scholar]
- 4.Henderson AJ, Zou X, Calame KL. C/EBP proteins activate transcription from the human immunodeficiency virus type 1 long terminal repeat in macrophages/monocytes. J Virol 1995; 69:5337–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ito M, Baba M, Sato A, et al. Tumor necrosis factor enhances replication of human immunodeficiency virus (HIV) in vitro. Biochem Biophys Res Commun 1989; 158:307–12. [DOI] [PubMed] [Google Scholar]
- 6.Rodger AJ, Fox Z, Lundgren JD, et al. Activation and coagulation biomarkers are independent predictors of the development of opportunistic disease in patients with HIV infection. J Infect Dis 2009; 200:973–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Descamps-Latscha B, Herbelin A, Nguyen AT, et al. Balance between IL-1 beta, TNF-alpha, and their specific inhibitors in chronic renal failure and maintenance dialysis. Relationships with activation markers of T cells, B cells, and monocytes. J Immunol 1995; 154:882–92. [PubMed] [Google Scholar]
- 8.Spoto B, Leonardis D, Parlongo RM, et al. Plasma cytokines, glomerular filtration rate and adipose tissue cytokines gene expression in chronic kidney disease (CKD) patients. Nutr Metab Cardiovasc Dis 2012; 22:981–8. [DOI] [PubMed] [Google Scholar]
- 9.Stenvinkel P. New insights on inflammation in chronic kidney disease-genetic and non-genetic factors. Nephrol Ther 2006; 2:111–9. [DOI] [PubMed] [Google Scholar]
- 10.Shankar A, Sun L, Klein BE, et al. Markers of inflammation predict the long-term risk of developing chronic kidney disease: a population-based cohort study. Kidney Int 2011; 80:1231–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stinghen AE, Bucharles S, Riella MC, Pecoits-Filho R. Immune mechanisms involved in cardiovascular complications of chronic kidney disease. Blood Purif 2010; 29:114–20. [DOI] [PubMed] [Google Scholar]
- 12.Yilmaz MI, Solak Y, Covic A, Goldsmith D, Kanbay M. Renal anemia of inflammation: the name is self-explanatory. Blood Purif 2011; 32:220–5. [DOI] [PubMed] [Google Scholar]
- 13.Bruggeman LA, Bark C, Kalayjian RC. HIV and the Kidney. Curr Infect Dis Rep 2009; 11:479–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gardner LI, Holmberg SD, Williamson JM, et al. Development of proteinuria or elevated serum creatinine and mortality in HIV-infected women. J Acquir Immune Defic Syndr 2003; 32:203–9. [DOI] [PubMed] [Google Scholar]
- 15.Gupta SK, Mamlin BW, Johnson CS, Dollins MD, Topf JM, Dube MP. Prevalence of proteinuria and the development of chronic kidney disease in HIV-infected patients. Clin Nephrol 2004; 61:1–6. [DOI] [PubMed] [Google Scholar]
- 16.Gupta SK, Eustace JA, Winston JA, et al. Guidelines for the management of chronic kidney disease in HIV-infected patients: recommendations of the HIV Medicine Association of the Infectious Diseases Society of America. Clin Infect Dis 2005; 40:1559–85. [DOI] [PubMed] [Google Scholar]
- 17.Kimmel PL, Barisoni L, Kopp JB. Pathogenesis and treatment of HIV-associated renal diseases: lessons from clinical and animal studies, molecular pathologic correlations, and genetic investigations. Ann Intern Med 2003; 139:214–26. [PubMed] [Google Scholar]
- 18.Leventhal JS, Ross MJ. Pathogenesis of HIV-associated nephropathy. Semin Nephrol 2008; 28:523–34. [DOI] [PubMed] [Google Scholar]
- 19.Lucas GM, Mehta SH, Atta MG, et al. End-stage renal disease and chronic kidney disease in a cohort of African-American HIV-infected and at-risk HIV-seronegative participants followed between 1988 and 2004. AIDS 2007; 21:2435–43. [DOI] [PubMed] [Google Scholar]
- 20.Naicker S, Fabian J. Risk factors for the development of chronic kidney disease with HIV/AIDS. Clin Nephrol 2010; 74(suppl 1):S51–6. [DOI] [PubMed] [Google Scholar]
- 21.Szczech LA, Gange SJ, van der Horst C, et al. Predictors of proteinuria and renal failure among women with HIV infection. Kidney Int 2002; 61:195–202. [DOI] [PubMed] [Google Scholar]
- 22.Szczech LA, Hoover DR, Feldman JG, et al. Association between renal disease and outcomes among HIV-infected women receiving or not receiving antiretroviral therapy. Clin Infect Dis 2004; 39:1199–206. [DOI] [PubMed] [Google Scholar]
- 23.Margolick JB, Jacobson LP, Schwartz GJ, et al. Factors affecting glomerular filtration rate, as measured by iohexol disappearance, in men with or at risk for HIV infection. PLoS One 2014; 9:e86311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ng DK, Jacobson LP, Brown TT, et al. HIV therapy, metabolic and cardiovascular health are associated with glomerular hyperfiltration among men with and without HIV infection. AIDS 2014; 28:377–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kaslow RA, Ostrow DG, Detels R, Phair JP, Polk BF, Rinaldo CR., Jr The Multicenter AIDS Cohort Study: rationale, organization, and selected characteristics of the participants. Am J Epidemiol 1987; 126:310–8. [DOI] [PubMed] [Google Scholar]
- 26.Caliendo AM, Valsamakis A, Zhou Y, et al. Multilaboratory comparison of hepatitis C virus viral load assays. J Clin Microbiol 2006; 44:1726–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schwartz GJ, Munoz A, Schneider MF, et al. New equations to estimate GFR in children with CKD. J Am Soc Nephrol 2009; 20:629–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schwartz GJ, Furth S, Cole SR, Warady B, Munoz A. Glomerular filtration rate via plasma iohexol disappearance: pilot study for chronic kidney disease in children. Kidney Int 2006; 69:2070–7. [DOI] [PubMed] [Google Scholar]
- 29.Ng DK, Schwartz GJ, Jacobson LP, et al. Universal GFR determination based on two time points during plasma iohexol disappearance. Kidney Int 2011; 80:423–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Abraham AG, Munoz A, Furth SL, Warady B, Schwartz GJ. Extracellular volume and glomerular filtration rate in children with chronic kidney disease. Clin J Am Soc Nephrol 2011; 6:741–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Haycock GB, Schwartz GJ, Wisotsky DH. Geometric method for measuring body surface area: a height-weight formula validated in infants, children, and adults. J Pediatr 1978; 93:62–6. [DOI] [PubMed] [Google Scholar]
- 32.Osberg IM, Hammond KB. A solution to the problem of bilirubin interference with the kinetic Jaffe method for serum creatinine. Clin Chem 1978; 24:1196–7. [PubMed] [Google Scholar]
- 33.Wada NI, Jacobson LP, Margolick JB, et al. The effect of HAART-induced HIV suppression on circulating markers of inflammation and immune activation. AIDS 2015; 29:463–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hultin LE, Menendez FA, Hultin PM, et al. Assessing immunophenotyping performance: proficiency-validation for adopting improved flow cytometry methods. Cytometry B Clin Cytom 2007; 72:249–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guidelines for the evaluation and management of chronic kidney disease. Kidney Int Suppl 2013; 3:1–150. [Google Scholar]
- 36.Kuller LH, Tracy R, Belloso W, et al. Inflammatory and coagulation biomarkers and mortality in patients with HIV infection. PLoS Med 2008; 5:e203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sandler NG, Wand H, Roque A, et al. Plasma levels of soluble CD14 independently predict mortality in HIV infection. J Infect Dis 2011; 203:780–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Burdo TH, Lo J, Abbara S, et al. Soluble CD163, a novel marker of activated macrophages, is elevated and associated with noncalcified coronary plaque in HIV-infected patients. J Infect Dis 2011; 204:1227–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Baker JV, Hullsiek KH, Singh A, et al. Immunologic predictors of coronary artery calcium progression in a contemporary HIV cohort. AIDS 2014; 28:831–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Deeks SG, Lewin SR, Havlir DV. The end of AIDS: HIV infection as a chronic disease. Lancet 2013; 382:1525–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bemelmans MH, van Tits LJ, Buurman WA. Tumor necrosis factor: function, release and clearance. Crit Rev Immunol 1996; 16:1–11. [DOI] [PubMed] [Google Scholar]
- 42.Bemelmans MH, Gouma DJ, Buurman WA. Influence of nephrectomy on tumor necrosis factor clearance in a murine model. J Immunol 1993; 150:2007–17. [PubMed] [Google Scholar]
- 43.Kopp JB, Nelson GW, Sampath K, et al. APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol 2011; 22:2129–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nichols B, Jog P, Lee JH, et al. Innate immunity pathways regulate the nephropathy gene apolipoprotein L1. Kidney Int 2015; 87:332–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Taylor HE, Khatua AK, Popik W. The innate immune factor apolipoprotein L1 restricts HIV-1 infection. J Virol 2014; 88:592–603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ross MJ, Fan C, Ross MD, et al. HIV-1 infection initiates an inflammatory cascade in human renal tubular epithelial cells. J Acquir Immune Defic Syndr 2006; 42:1–11. [DOI] [PubMed] [Google Scholar]
- 47.Segerer S, Nelson PJ, Schlondorff D. Chemokines, chemokine receptors, and renal disease: from basic science to pathophysiologic and therapeutic studies. J Am Soc Nephrol 2000; 11:152–76. [DOI] [PubMed] [Google Scholar]
- 48.Kimmel PL, Cohen DJ, Abraham AA, Bodi I, Schwartz AM, Phillips TM. Upregulation of MHC class II, interferon-alpha and interferon-gamma receptor protein expression in HIV-associated nephropathy. Nephrol Dial Transplant 2003; 18:285–92. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.