Abstract
Background
Appropriate and timely screening can significantly reduce breast and cervical cancer morbidity and mortality. Racial/ethnic minorities and immigrant populations have lower screening rates and delays in follow-up after abnormal tests.
Purpose
In this study, we examined the relationship between age, comorbidity, breast and cervical cancer literacy in a sample of African American, Latina, and Arab women (N=371) from Detroit, Michigan.
Methods
Age-adjusted Charlson Comorbidity Index (ACC) was used characterize the impact of age and comorbidity has on breast and cervical cancer literacy; Breast Cancer Literacy Assessment Tool was used to assess breast cancer literacy; Cervical Cancer Literacy Assessment Tool was used to assess cervical cancer literacy. ANOVA was used to assess the relationship between ACC, breast and cervical cancer screening and group differences.
Results
There was a statistically significant difference between breast cancer literacy (Breast-CLAT total scores) scores (F(2,367)= 17.31, p= < 0.01). ACC had a greater impact on breast cancer literacy for African American F(2,214) =11, p = <0.01.
Keywords: Cervical cancer, Breast cancer, Literacy, Age-Adjusted comorbidity, Chronic disease
INTRODUCTION
Appropriate screening and early detection can significantly reduce breast and cervical cancer–associated morbidity and mortality, and the U.S. Preventive Services Task Force,1,2 American Cancer Society, American College of Obstetricians and Gynecologists, and several other national guidelines recommend regular screening.3-5 In this study, we will use the American Cancer Society (ACS) guidelines for breast and cervical cancer screening, which are depicted in Box 1 and Box 2. Many women do not obtain breast and cervical cancer screening at recommended regular intervals and experience delays in diagnostic follow-up after an abnormal mammogram.6-9 Untimely screening and inappropriate follow-up after an abnormal mammogram increase the risk for late-stage diagnosis and larger size tumors.8,9 Late-stage diagnosis negatively impacts treatment, disease course, and survival.10,11 Approximately 33% of eligible women (aged 40 years and older) have not received breast cancer screening within the past two years.12 Breast cancer screening rates are lowest in women that are uninsured (38%) followed by immigrant women who have been in the U. S. less than 10 years (39.9%). In 2013, 11% of women (aged 21 to 65) have not been screened for cervical cancer in the past 3-years.12,13 Cervical cancer screening has been consistently lower in women who are uninsured (61%), recent immigrants (66%), and women with less than a high school education (69%).
Several factors contribute to low breast and cervical cancer screening rates, including, low socioeconomic status, low educational attainment, membership in a minority race/ethnic group, foreign-born or immigrant status, lack of a regular care provider, lack of a doctor's recommendation, lack of healthcare access, inconvenience, cultural beliefs, and lack of social support.14,15 Lower screening rates in immigrant women may be partially attributed to language barriers (English proficiency). Limited English proficiency can lead to decreased access to health care, dissatisfaction with care, decreased quality of care, and limit knowledge about cancer prevention and screening guidelines.14-20 For example, in a study examining mammography screening among “Asian Indian” women, researchers reported that length of stay in the United States, marital status, knowledge of mammogram guidelines, age, having health insurance, physician recommendations, and number of relatives who had a mammogram were positively associated with having a mammogram within the past 2 years.21 These findings suggest that women who have difficulty understanding spoken recommendations about breast and cervical cancer screening may be at risk for non-adherence to screening guidelines.22 Knowledge (health literacy) about breast and cervical cancer and screening benefits are important determinants screening adherence.23,24
Health literacy is a strong predictor of cancer screening rates. Health literacy is defined as a “wide range of skills and competencies that people develop to seek out, comprehend, evaluate, and use health information and concepts to make informed choices, reduce health risks, and increase quality of life.”25 Women with limited health literacy and limited English proficiency may have difficulty understanding screening guidelines, accessing and navigating the healthcare system, and difficulty with clinical decision making.26 Moreover, because provider recommendation is an important factor in obtaining cancer screening, limited health literacy affects patient/provider communication. Low health literacy is associated with limited awareness about cancer screening, lack of desire for screening and limited access to care.27-29
There is also evidence that comorbidities often serve as a barrier to timely and appropriate cancer screening.30,31 Some studies document that specific comorbidities are an independent risk factor for cancer (e.g., diabetes, hypertension).32-34 For example, women with diabetes are at increased risk for development of cancer and are 40% more likely to die from breast cancer than women who do not have diabetes.35,36 In addition, women with diabetes are less likely to obtain breast and cervical cancer screening than women who do not have diabetes.31,37 In one study, women with cardiovascular and lung disease were less likely to be up to date on breast cancer screening and women with diabetes, arthritis, and hypertension were less likely to be up to date with cervical cancer screening compared to women without comorbidities.38
Although there is evidence that regular breast and cervical cancer screening and timely follow-up of abnormal tests lead to decrease mortality;39-42 racial/ethnic minority women, particularly immigrant women, continue to underuse cancer screening. Given the long-standing low screening rates of racial/ethnic minorities and the increasing number of immigrants to the U. S., information that contributes to our understanding of factors that contribute to lower screening rates is important.43-45 Moreover, information obtained from this study may be helpful in understanding screening patterns and facilitation of timely and appropriate screening. The present study addressed this need by examining the association between age, comorbid conditions, and breast and cervical cancer literacy of African American, Latina, and Arab women. These populations were selected for this study because they have lower screening rates, African American and Latina women have higher cancer-related mortality rates than White women, and Latina and Arab women are among the largest group of U. S. immigrants.43 Additionally, Michigan is among the states with the largest percent increase of immigrants and has one of the largest concentrations of Arab Americans.43,46,47 Few studies have addressed the association between age, comorbidity and cancer health literacy in a sample of African American, Latina and Arab women.
Breast and Cervical Cancer
For women in the U. S., breast cancer is the most common cancer and the second leading cause of cancer death. In 2015, 231,840 new cases of invasive breast cancer is expected to be diagnosed and 40,290 women are expected to die from the disease.48 Routine breast cancer screening can reduce morbidity and mortality from late-stage diagnosis and treatment.49,50 Mammography Some women experience a delay in follow-up after an abnormal mammogram.51
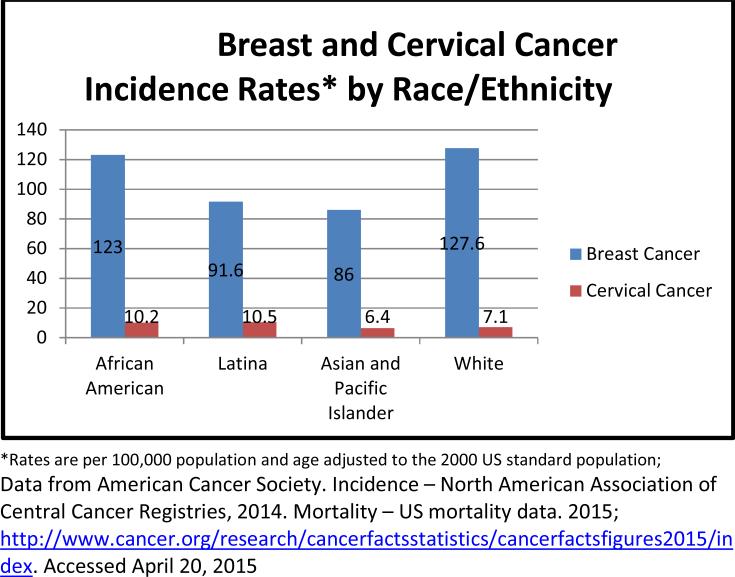
Despite having a lower incidence of breast cancer than White women (Figure 1), African American, Latina, and Arab women are more likely to be diagnosed at a younger age, more advanced stage of disease, and to have more aggressive forms of breast cancer.52-56 Advanced stage of cancer diagnosis has been attributed to lower screening rates, inadequate knowledge about screening guidelines, and delayed followed up for abnormal findings.57,58 Studies have reported that racial/ethnic minority and low income women are more likely to delay follow-up.51,59-61 For example, in a study of African American, Latina, Asian and White women, researchers from that African American race, income, perceived discrimination, not fully understanding the results of the mammogram, and being notified of abnormal finding by letter or phone instead of in-person.61 A 3 to 6 months delay in treatment of breast cancer can reduce survival and delays of over 1 year increases the odds of lymph node metastasis and larger tumors.62,63 Breast cancer is the most commonly diagnosed cancer in Latinas and the leading cause of cancer death.54 African American women are more likely to die from cancer than any other racial/ethnic group.48 Arab American women have similar stage, age, and hormone receptor status as African Americans, but a better survival rate.64,65
Figure 1.
Breast and Cervical Cancer Incidence Rates* by Race/Ethnicity
In 2015, 12,900 new cases of invasive cervical cancer is expected to be diagnosed and 1,400 are expected to die from the disease.48 Over half of the cervical cancer deaths occur in women who have never been screened or women who have not been screened in five years.48 Cervical cancer is preventable with early detection and removal of precancerous cervical lesions.48,66 Routine cervical cancer screening (Papanicolaou [Pap] test) or human papillomavirus test allows for the detection of precancerous lesions that can be treated prior to progression to cancer. In addition, the time interval between cervical cancer screening, diagnosis, and treatment have a significant negative impact on health outcomes.66 A longer interval between diagnostic identification of a precancerous lesion and treatment results in later stage disease, decreased survival, and increased economic cost (individual and society).67,68 In women with a precancerous lesion who receive timely and appropriate evaluation, treatment and follow-up, the probability of survival is almost 100%.12 Racial ethnic minorities are less likely to meet the timeliness diagnostic interval.
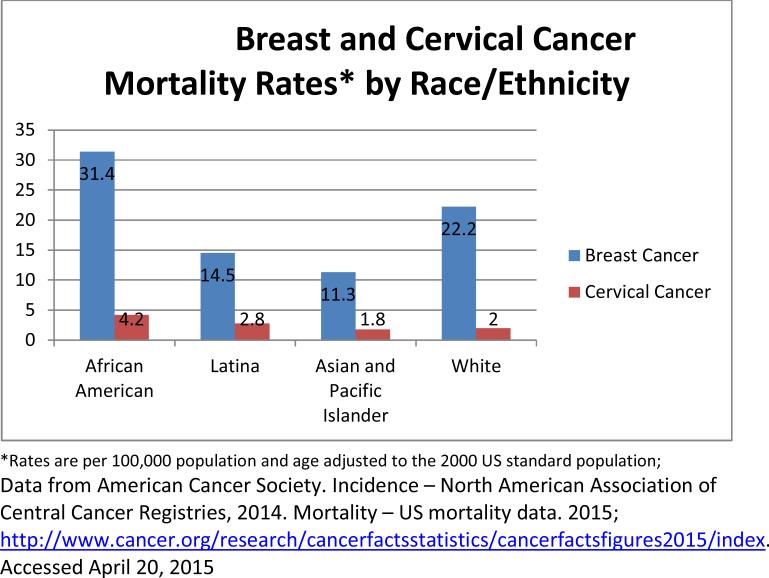
The incidence of cervical cancer in the U. S. is highest in Latinas (10.5), followed by African American women (10.2) (Figure 1).48,54,55 African American women are twice as likely to die from cervical cancer as White women (Figure 2).55 Available information in the literature about cervical cancer in Arab women indicate that in most Arab countries, cervical cancer is the second most common malignancy.69 Studies examining cervical cancer among Arab American women indicate that cervical cancer screening is lower among Arab women than the general population.70 Studies also report that Arab women have a lower level of knowledge about cervical cancer.71,72
Figure 2.
Breast and Cervical Cancer Mortality Rates* by Race/Ethnicity
These findings suggest that early detection and treatment by adherence to established screening guidelines are critical to reducing breast and cervical cancer morbidity and mortality. For example, women who receive cervical cancer screening within 3-36 months prior to cervical cancer diagnosis have a lower mortality rate.39 However, many women do not adhere to these guidelines and many are not aware of these guidelines.73,74 There is thus a clear need to identify factors that impact screening for racial/ ethnic minority women, particularly among immigrants and individuals with limited English proficiency to improve cancer screening rates.
Breast and Cervical Cancer Screening/Literacy/Age/Comorbidity
Symptomatic presentation is often the most common route for a cancer diagnosis. Screening, testing an individual who has no symptoms,12 allows for early detection of disease. In 2013, 65.9% of U. S. women reported having a mammogram within the past two years.12 Race/ethnicity and immigration status play a role in predicting breast and cervical cancer screening. Mammography rates are particularly low for foreign-born individuals who immigrated to the United States more recently or who are less acculturated (living in the US <10 years) (39.9%) and uninsured women (38%).12,75 The Pap test screening rate over the past three years for U. S. women is 80.1%. Similar to mammogram use, Pap test use was lowest in recent immigrants (65.9%) and uninsured women (60.6%).12
Knowledge (health literacy) about screening and its benefits is an important determinant of screening. One in five adults in the U. S., do not have the basic literacy skills to function sufficiently in our society, particularly in healthcare.26 As healthcare consumers, these individuals often do not have the necessary information to make appropriate health care decisions. Underuse of breast and cervical cancer screening is associated with lower health literacy, particularly in ethnic/racial minorities. In a study examining the relationship between health literacy and screening mammography, health literacy had the strongest association with mammography screening.76 Low health literacy contributes to social inequities and poor health outcomes.77 Low health literacy influences decision making about cancer screening and prevention.
In a study assessing cervical cancer screening in underserved African American women and Latinas, screening was low; Latina and older women were less likely to adhere to screening guidelines. Age, knowledge of screening recommendations, and having a regular health care provider were independently associated with both breast and cervical cancer screening in a sample of Latinas.73 Similar results have been reported by researchers in this patient population.78-81 Knowledge about screening recommendations is also low in Arab women and some studies indicate that even in women with adequate knowledge, screening is low.69,82-86
The risk of breast cancer increases with age. Aging is also associated with an increased risk of comorbidities and cancer. There are conflicting reports on the association between age, comorbidity, and cancer screening. Some studies report that comorbidity has little effect on the use of screening mammography and Pap testing. While, other studies indicate that specific comorbidities increase the likelihood of timely cancer screening (e.g., hypertension, digestive disorders).38,87,88 These researchers suggest that individuals with comorbid conditions have more contact with the healthcare system and are more likely to be screened. Other researchers suggest that women with comorbid conditions are less likely to be screened because of competing demands. They suggest that comorbid condition competes with the health care provider time and focus and that it impacts the individual's resources. Diabetes has been consistently related to cancer screening rates.89-91
Low income and African American women are more likely to have two are more comorbidities compared to White women. In this paper, we examined the association between age, comorbid conditions, breast and cervical cancer literacy among medically underserved women (i.e., African American, Latina, and Arab). To meet the needs of women with lower utilization of screening (racial/ethnic minorities, immigrants, low income), will requires that we understand the factors that contribute to lower screening rates.
METHODS
Design
This study used a quantitative, descriptive design. Breast and cervical cancer literacy, age and comorbidity were evaluated. The purpose of this study is to examine the association between age, comorbid conditions, and breast and cervical cancer literacy of African American, Latina, and Arab women.
Participants
This study used a purposive sample of women who participated in the community-based Kin Keeper℠ Cancer Prevention Intervention studies, previously described in detail.92-94 The studies were approved by the Michigan State University Institutional Review Board. Criteria for inclusion in the current study included: (a) female sex, (b) age > 40years, and (c) self-identified as African American, Latina, or Arab. Our inclusion criteria yielded a total sample of 371 women (African American=161; Latina=107; Arab=103). For the original study, inclusion criteria were: female, age 21-70, self-identifies as African American, Latina, or Arab, receiving services from CHWs from one-of-the 3 community-based organizations, biological mother and grandmothers of the same race/ethnicity and willingness to recruit members of her female adult family to participate in a home education visit, and completion of 2 home-based educational sessions on breast and cervical cancer prevention and control.
Procedure
Participants were recruited from community-based organizations affiliated with the Detroit Department of Health and Wellness Promotion including: a) Village Health Worker Program, b) Community Health and Social Services, and c) the Arab Community Center for Economic and Social Services. Briefly, CHWs recruit women (age ≥21) of their respective race (African American, Latina, Arab) from their public health case load for 2 home-based educational sessions. After signing the consent during first home visit, the CHW and family unit completed the Historical Background Questionnaire and the breast cancer assessment (baseline). After completion of pretest to assess breast cancer literacy, the educational intervention was delivered, followed by a posttest. During the second educational session, the second posttest was delivered, followed by the cervical educational session and a posttest. During the second visit, participants also completed a personal-action-plan.
Questionnaire items
Sociodemographic Characteristics
Demographic factors on marital status, income, education, employment status, and age were considered in the current study.
Comorbidities
Data about comorbidities were obtained by the participants’ response to the question, “Have you ever been told by a doctor or health professional that you had?” Based on the comorbidities, all patients were assigned a comorbidity score based on the Age-Adjusted Charlson index score as described by Charlson et al.95,96 The overall score is a weighted summation of medical conditions and age with higher scores indicating a higher medical comorbidity (Box 2).
Breast and Cervical Cancer Literacy
Breast cancer literacy was assessed with the Breast Cancer Literacy Assessment Tool (Breast-CLAT), a 35-item assessment instrument, that measures functional breast cancer literacy in three domains: (a) awareness (items 1-6), (b) knowledge and screening (items 7-19), and (c) prevention and control (items 20-35).97 The Breast-CLAT uses a multiple choice and true/false format and is scored as a binary variable (0=Incorrect, 1=Correct). Scores range from 0-35, with higher scores indicating higher level of functional breast cancer literacy. The instrument has been validated in English, Spanish and Arabic with a total scale Cronbach α=.73.97 The total scale reliability was highest in African Americans and lowest in Latina (.81 and .61 [respectively]).98,99 Breast-CLAT total and subscale scores were assessed.
Cervical cancer literacy was assessed with the Cervical Cancer Literacy Assessment Tool (C-CLAT).98,100 The 16-item instrument contains three domains: (a) Awareness, (b) Knowledge and Screening, and (c) Prevention and Control.101 The items are scored as a binary variable (0=incorrect, 1=correct). Scores range from 0-16, with higher scores indicating better literacy. The internal consistency of the cervical cancer literacy assessment tool was high (0.72). The CCLAT reliabilities in African American, Latina, and Arab women were 0.73, 0.76, and 0.60, respectively. The C-CLAT scores were assessed by subscale and total scores.
Data Analysis
Data were analyzed using Stata (version 12.1) software.102 Descriptive statistics (means, standard deviations [SD], frequencies, and proportions as appropriate) were used to describe the sample, including sociodemographic characteristics and comorbidities by racial/ethnic group. Analysis of variance (ANOVA), multivariate analysis of variance (MANOVA) and Tukey Honestly Significant Difference (HSD) post hoc tests were conducted to evaluate the effect of age-adjusted comorbidity on breast and cervical cancer literacy, plus measure the combined effect of age-adjusted comorbidity and race on breast and cervical cancer literacy. The main factors of interest were age-adjusted comorbidity and race; both categorical variables. This analysis was used, given the likelihood that the dependent variables (i.e., breast and cervical cancer literacy) are related to one another. Also, a two-way MANOVA allowed for not only tests of the main effects of the independent variables (i.e., age-adjusted comorbidity and race) but also for possible interaction effects between these variables, which is important, given the relatively high co-occurrence of low breast and cervical cancer literacy in these populations. The use of MANOVA also reduces the risk of Type I errors, which are more common with the use of repeated analyses of variance (ANOVA). Box's test of equality was used to test the assumption of homogeneity of variance–covariance matrices, and Levene's test of equality was used to test the assumption of equality of variances. No significant violations to these assumptions were noted. Statistical significance was based on Wilks’ λ statistic and partial η2 statistics were reported to illustrate effect size. The Tukey HSD tests were used to test all pairwise comparisons. The statistical significance of each result was evaluated according to its P-value (P<0.05 being significant) (Polit and Beck, 2012).
Results
The sample included 371 women ranging in age from 41 to 101 years. African American women were more likely to be unmarried (Table 1). Participants were also dichotomized into three groups based on age-adjusted comorbidity scores: (a) low= 0-1 (N=153); (b) mild=2-3 (N=144); and (c) severe=>3 (N=74).103 The two most common comorbidities were hypertension and diabetes.
Table 1.
Sociodemographic Characteristics and Comorbidities by Racial/Ethnic Group (N=371)
| African American (N=161) | Latina (N=107) | Arab American (N=103) | |
|---|---|---|---|
| Variables | |||
| Age, years (Mean ± SD) | 53 ± 9 | 51 ± 9 | 53 ± 11 |
| Marital status | |||
| Married, N(%) | 42(27) | 69(66) | 80(78) |
| Education, N(%) | |||
| < High school | 14(9) | 79(74) | 43(42) |
| High school/GED | 53(33) | 17(16) | 37(36) |
| >High school | 93(58) | 10(9) | 22(21) |
| Incomea N(%) | |||
| < $9,999 | 28(18) | 62(60) | 43(43) |
| $10,000-$19,999 | 38(24) | 20(19) | 32(32) |
| $20,000-$39,999 | 49(31) | 18(17) | 15(15) |
| ≥ $40,000 | 43(27) | 4(4) | 10(10) |
| Employment statusa N(%) | |||
| Employed | 99(61) | 47(45) | 19(19) |
| Unemployed | 62(39) | 57(55) | 83(81) |
| Age-Adjusted Charlson | |||
| Comorbidity (AAC), N(%) | |||
| Low (AAC 0-1) | 56(35) | 47(44) | 50(49) |
| Mild (AAC 2-3) | 70(43) | 44(41) | 30(29) |
| Severe (AAC >3) | 35(22) | 16(15) | 23(22) |
| Total no. of comorbid conditions | |||
| Median | 2 | 2 | 2 |
| Range | 0-6 | 0-5 | 0-5 |
≠100 because of missing data; SD= Standard deviation
The distribution of age-adjusted comorbidity, breast and cervical cancer literacy scores (total and subscale scores) are summarized in Table 2. Analysis of variance was conducted to compare age-adjusted comorbidity, breast and cervical cancer literacy scores. Results of the ANOVA for breast cancer literacy (Breast-CLAT total scores) indicated that literacy was significant different between groups (F(2,367) = 17.31, p= < 0.01). Similarly, age-adjusted comorbidity scores indicated that there was a significant difference between groups (F(2, 367)=3.08, p=<0.05). Given the statistical omnibus of the ANOVA F test, MANOVA and Tukey HSD tests were conducted to examine pairwise contrasts.104 For breast cancer literacy (total score), Tukey post hoc comparisons indicated that Latinas scores (mean =19, SE=.43) were significantly different than African American (mean=22, SE=.34) and Arab (mean=22, SE= .43) women. Age-adjusted comorbidity scores were significantly different for African American women (mean=1.9, SE=0.06) compared to Latina women (mean=1.7, SE=0.06). Subscale scores were only significant for breast cancer awareness. For breast cancer awareness, Arab women scores (mean=2, SE=0.2) were significantly different than African American (mean= 4, SE=0.1) and Latina women (mean=4, SE=0.2). Cervical cancer literacy (total scores and subscale scores) comparisons were not significant.
Table 2.
Means and Standard Deviations of Age-adjusted Comorbidity, Breast and Cervical Cancer Literacy by Racial/Ethnic Group (N=371)
| Racial/Ethnic Group | Total Sample | |||
|---|---|---|---|---|
| African American (N=161) | Latina (N=107) | Arab (N=103) | ||
| Measure | ||||
| Age-Adjusted Comorbidity (Mean ± SD) | 3 ± 2 | 2 ± 2 | 2 ± 2 | 2 ± 2 |
| Breast Cancer literacy (Mean ± SD) Total score | 22±4.8 | 19±4.3 | 22 ± 3.8 | 21±4.6 |
| Awareness | 4±1.0 | 4±3 | 3 ± 1 | 4 ± 1 |
| Prevention | 10±3 | 12±4 | 12±2 | 11±2 |
| Screening | 7±2 | 7±2 | 8±2 | 7±2 |
| Cervical Cancer Screening (Mean ± SD) | ||||
| Total Score | 10±3 | 10±2 | 10±2 | 10±3 |
| Awareness | 1±1 | 2±1 | 1±1 | 1±1 |
| Prevention | 6±2 | 6±2 | 6±2 | 6±2 |
| Screening | 3±1 | 3±2 | 3±2 | 3±2 |
SD= Standard deviation
Effect of Age-adjusted Comorbidity and Race on Breast Cancer Literacy
A factorial ANOVA was conducted to determine if breast cancer literacy differed based on age-adjusted comorbidity and race (African American, Latina, and Arab). The two-way factor analysis showed no significant main effect for age-adjusted comorbidity, F(2,12) = 0.65 p = >0.05. There was a significant main effect for race, F(2,214) =11, p = <0.01; indicating that breast cancer literacy was influenced by race. The interaction between age-adjusted comorbidity and race was also significant, F(2,70) = 3, p = <0.01; indicating that differences in breast cancer literacy and age-adjusted comorbidity depends on race.
Discussion
The purpose of this study was to assess the association between age-adjusted comorbidity, breast and cervical and literacy for African American, Latina, and Arab women. In this study, medical comorbidity as measured by the validated Age-Adjusted Charlson Comorbidity Index indicated that it had a significant effect on health literacy. Medical comorbidities had a greater impact on African American women breast cancer literacy than Latina or Arab women. There are a limited number of studies that have used the Age-Adjusted Charlson Comorbidity index to assess the relationship between age, comorbidity and breast and cervical cancer in a racial/ethnic diverse sample of women. Most studies used the Age-Adjusted Charlson Comorbidity index as a predictor of mortality, survival prediction, and cancer treatment options.105-107
Studies examining the relationship between comorbidities and screening report that comorbidities are associated with adherence to breast and cervical cancer screening guidelines and later stage at cancer diagnosis.38,108,109 Vaeth et al.,110 evaluated comorbid conditions in women newly diagnosed with breast cancer and reported that women with two or more comorbid conditions were more likely to be diagnosed at an advanced stage of disease. Similar results were reported by Kiefe et al.,111 who examined the role of chronic disease as a barrier to screening for breast and cervical cancer. The researchers reported that selected chronic diseases (e.g., heart disease, gastrointestinal disorders) contribute to lower screening rates. Studies also report the underuse of cancer screening by women with diabetes.112,113 These findings suggest that age and comorbid conditions may contribute to lower literacy levels for African American women and delay obtaining screening test. This is particularly important for African Americans because they are more likely to have chronic diseases.114 For example, African Americans have a higher rate of stroke (31%) and heart disease (23%) than Whites.115
The women in this study demonstrated a moderate to relatively low level of breast cancer literacy (African American and Arab: mean=22, SD= 4; Latina: mean=19, SD=4) and cervical cancer literacy (mean 10, SD=2 for all groups). Similar results were reported by Dumenci et al.,116 who reported limited cancer health literacy. In the study, 44% of African Americans believed that exposing a tumor to air during surgery cause the tumor to spread and that 23% of African Americans believe that rather than taking a pill twice a day as prescribed, taking it three times a day will help them get better faster. This inadequate knowledge can impact an individual's decision to seek screening. Research by Garger et al.,117 indicates that Spanish speaking women with inadequate health literacy are 16.7 times less likely to obtain cervical cancer screening.
There are limitations to this study that should be acknowledged. First, our findings may not generalize to other states, as our data come from only one state. However, Michigan has one of the largest Arab populations outside of the Middle East. The Age-Adjusted Charlson Index to is a valid and reliable instrument;118 however, it does not represent functional impairment, which could influence cancer screening, particularly in older adults.119-121 Future studies should also include functional impairment.
Summary
The results of this study support previous studies of racial/ethnic minorities and immigrant populations, indicating overall low breast and cervical literacy (awareness, screening, and prevention). The influence of comorbidity on stage at diagnosis and screening varies. However, our study supports the link between age-adjusted comorbidity in breast cancer literacy for African American women. This may be related to the fact that African Americans have higher number of comorbidities. Breast cancer screening among African American women may be better targeted by considering comorbidities in addition to race. Increased breast and cervical cancer knowledge could potentially lead to decreased stage at diagnosis and decreased mortality rates. Strategies to increase cancer screening at the primary and secondary level are essential to the reduction of advanced stage of cancer diagnosis. These strategies should include: cancer literacy and assessing comorbid conditions that may delay screening in medically underserved and immigrant populations.
KEY POINTS.
Cancer literacy and cancer screening rates are lower among medically underserved populations
Comorbidities (chronic medical conditions) may serve as a barrier to timely and appropriate cancer screening, particularly for African American women.
Rates of screening are particularly low for foreign-born individuals who immigrated to the United States recently or who are less acculturated
Health literacy about breast and cervical cancer can improve screening, reduce burden, and improve health outcomes
Health care providers should consider age and comorbidity when designing screening interventions for underserved populations.
BOX 1.
Comparison of USPSTF and ACS Screening Guidelines for Breast Cancer for Women at Average Risk
| U.S. Preventive Services Task Force (USPSTF) | American Cancer Society (ACS) |
|---|---|
| Biennial screening mammography beginning at age 50. | Annual screening mammography beginning at age 40. |
| Not enough evidence to support assessing the additional benefits of screening mammography in women past age 74. | Annual screening mammography for as long as a woman is in good health. |
| Recommends against health care providers teaching women how to perform breast self-examination | Breast self-examination is optional. Beginning in their early 20s' women should be told about the benefits and limitations of breast self-examination. Instructions should be given to women who choose to do breast self-examination by their health provider. |
| Evidence is insufficient for assessing the additional benefits of clinical breast examination beyond screening mammography in women 40 years or older. | Recommends clinical breast examination every three years for women in their 20s and 30s, and annually for women aged 40 and older. |
| There is insufficient evidence to support the additional benefits and harms of MRI as a screening method for breast cancer | In addition to screening mammography, annual MRI screening is recommended for women with greater than 20% lifetime risk of breast cancer. |
BOX 2.
Comparison of USPSTF and ACS Screening Guidelines for Cervical Cancer for Women at Average Risk
| U.S. Preventive Services Task Force (USPSTF) | American Cancer Society (ACS) |
|---|---|
| Cervical cancer screening should begin at age 21 years, regardless of the age of sexual initiation or other risk factors. | Cervical cancer screening should begin at age 21 years, regardless of the age of sexual initiation or other risk factors. |
| Screening recommended for women age 21 to 65 years with Pap test every 3 years or, for women age 30 to 65 years who want to lengthen the screening interval, screening with a combination of Pap test and HPV testing every 5 years. | Screening recommendations by age group: Aged 21 to 29 years, screening with Pap test alone every 3 years, no testing for HPV unless abnormal Pap test; Aged 30 to 65 years should be screened with Pap test and HPV testing every 5 years (preferred) or Pap test alone every 3 years (acceptable) |
| Recommends Against screening in women > age 65 years who have had adequate prior screening and are not otherwise at high risk for cervical cancer | Women over age 65 who have had regular screenings with normal results should not be screened |
| Recommends against screening for cervical cancer in women who have had a hysterectomy with removal of the cervix and who do not have a history of a high-grade precancerous lesion or cervical cancer | Women who have had their uterus and cervix removed in a hysterectomy and have no history of cervical cancer or pre-cancer should not be screened |
Data from Moyer VA. Screening for Cervical Cancer: U.S. Preventive Services Task Force Recommendation Statement. Annals of Internal Medicine. 2012;156(12):880-891 and American Cancer Society. New Screening Guidelines for Cervical Cancer. 2012; http://www.cancer.org/cancer/news/new-screening-guidelines-for-cervical-cancer. Accessed January 10, 2015.
Box 3.
Age-Adjusted Charlson Comorbidity Index Scoring
| Score | Comorbid Condition |
|---|---|
| 1 | ○ Myocardial infarction (MI) |
| ○ Congestive heart failure (CHF) | |
| ○ Cerebral vascular disease | |
| ○ Peripheral vascular disease | |
| ○ Dementia | |
| ○ Chronic obstructive pulmonary disease (COPD) | |
| ○ Connective tissue disease | |
| ○ Peptic ulcer disease (PUD) | |
| ○ Mild liver disease | |
| 2 | ○ Diabetes |
| ○ Hemiplegia | |
| ○ Moderate/severe renal disease | |
| ○ Diabetes with end-organ damage | |
| ○ Any solid tumor | |
| ○ Leukemia | |
| ○ Lymphoma | |
| 3 | ○ Moderate/severe liver disease |
| 6 | ○ Metastatic solid tumor |
| ○ Acquired immunodeficiency syndrome (AIDS) | |
| Age | |
| 41-50 | ○ 1 point |
| 51-60 | ○ 2 points |
| 61-70 | ○ 3 points |
| 71 or older | ○ 4 points |
Acknowledgements
This work has been supported by the National Institutes of Health National Institute for Nursing Research (R01NR011323) and (R21NR010366). The authors would like to thank their community partners at the Detroit Department of Health and Wellness Promotion and the Arab Community Center for Economic and Social Services.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosures: None
Contributor Information
Costellia H. Talley, College of Nursing Michigan State University 1355 Bogue St Rm C-247 East Lansing, Michigan 48824.
Karen Patricia Williams, Department of Obstetrics, Gynecology & Reproductive Biology Michigan State University East Lansing, Michigan 48824 Phone: 517-432-4790 Fax: 517-353-1663 Karen.Williams@ht.msu.edu.
References
- 1.U.S. Preventive Services Task Force Screening for Breast Cancer: U.S. Preventive Services Task Force Recommendation Statement. Annals of Internal Medicine. 2009;151(10):716–726. doi: 10.7326/0003-4819-151-10-200911170-00008. [DOI] [PubMed] [Google Scholar]
- 2.U. S. Preventive Services Task Force Screening for Cervical Cancer: U.S. Preventive Services Task Force Recommendation Statement. Annals of Internal Medicine. 2012 [Google Scholar]
- 3.American Cancer Society [January 10, 2015];Breast Cancer Prevention and Early Detection. 2014 http://www.cancer.org/cancer/breastcancer/moreinformation/breastcancerearlydetection/breast-cancer-early-detection-toc.
- 4.Saslow D, Solomon D, Lawson HW, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA: a cancer journal for clinicians. 2012 May-Jun;62(3):147–172. doi: 10.3322/caac.21139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.American College of Obstetricians and Gynecologists (ACOG) Practice Bulletin No. 131: Screening for Cervical Cancer. Obstetrics & Gynecology. 2012;120(5):1222–1238. doi: 10.1097/aog.0b013e318277c92a. [DOI] [PubMed] [Google Scholar]
- 6.Gierisch JM, Earp JA, Brewer NT, Rimer BK. Longitudinal predictors of nonadherence to maintenance of mammography. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2010 Apr;19(4):1103–1111. doi: 10.1158/1055-9965.EPI-09-1120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hahn KM, Bondy ML, Selvan M, et al. Factors associated with advanced disease stage at diagnosis in a population-based study of patients with newly diagnosed breast cancer. American journal of epidemiology. 2007 Nov 1;166(9):1035–1044. doi: 10.1093/aje/kwm177. [DOI] [PubMed] [Google Scholar]
- 8.Taplin SH, Ichikawa L, Buist DS, Seger D, White E. Evaluating organized breast cancer screening implementation: the prevention of late-stage disease? Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2004 Feb;13(2):225–234. doi: 10.1158/1055-9965.epi-03-0206. [DOI] [PubMed] [Google Scholar]
- 9.Zapka J, Taplin SH, Price RA, Cranos C, Yabroff R. Factors in quality care--the case of follow-up to abnormal cancer screening tests--problems in the steps and interfaces of care. Journal of the National Cancer Institute. Monographs. 2010;2010(40):58–71. doi: 10.1093/jncimonographs/lgq009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Braithwaite D, Tammemagi CM, Moore DH, et al. Hypertension is an independent predictor of survival disparity between African-American and white breast cancer patients. International journal of cancer. Journal international du cancer. 2009 Mar 1;124(5):1213–1219. doi: 10.1002/ijc.24054. [DOI] [PubMed] [Google Scholar]
- 11.Louwman WJ, Janssen-Heijnen ML, Houterman S, et al. Less extensive treatment and inferior prognosis for breast cancer patient with comorbidity: a population-based study. European journal of cancer. 2005 Mar;41(5):779–785. doi: 10.1016/j.ejca.2004.12.025. [DOI] [PubMed] [Google Scholar]
- 12.American Cancer Society . Cancer Prevention and Early Detection: Facts and figures. Atlanta: 2015-2016. p. GA2015. [Google Scholar]
- 13.Centers for Disease Control [January 13, 2015];Millions of US women are not getting screened for cervical cancer. 2014 http://www.cdc.gov/media/releases/2014/p1105-vs-cervical-cancer.html. 2014.
- 14.Shelton RC, Jandorf L, King S, Thelemaque L, Erwin DO. Cervical cancer screening among immigrant Hispanics: an analysis by country of origin. Journal of immigrant and minority health / Center for Minority Public Health. 2012 Aug;14(4):715–720. doi: 10.1007/s10903-011-9541-y. [DOI] [PubMed] [Google Scholar]
- 15.Shirazi M, Bloom J, Shirazi A, Popal R. Afghan immigrant women's knowledge and behaviors around breast cancer screening. Psycho-oncology. 2013 Aug;22(8):1705–1717. doi: 10.1002/pon.3216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chavez LR, McMullin JM, Mishra SI, Hubbell FA. Beliefs Matter: Cultural Beliefs and the Use of Cervical Cancer-Screening Tests. American Anthropologist. 2001;103(4):1114–1129. [Google Scholar]
- 17.De Alba I, Sweningson JM, Chandy C, Hubbell FA. Impact of English language proficiency on receipt of pap smears among Hispanics. Journal of general internal medicine. 2004 Sep;19(9):967–970. doi: 10.1007/s11606-004-0009-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Goel MS, Wee CC, McCarthy EP, Davis RB, Ngo-Metzger Q, Phillips RS. Racial and ethnic disparities in cancer screening: the importance of foreign birth as a barrier to care. Journal of general internal medicine. 2003 Dec;18(12):1028–1035. doi: 10.1111/j.1525-1497.2003.20807.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Breen N, Rao SR, Meissner HI. Immigration, Health Care Access, and Recent Cancer Tests Among Mexican-Americans in California. Journal of Immigrant & Minority Health. 2010;12(4):433–444. doi: 10.1007/s10903-008-9198-3. [DOI] [PubMed] [Google Scholar]
- 20.Seid M, Stevens GD, Varni JW. Parents' perceptions of pediatric primary care quality: effects of race/ethnicity, language, and access. Health services research. 2003 Aug;38(4):1009–1031. doi: 10.1111/1475-6773.00160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Somanchi M, Juon HS, Rimal R. Predictors of screening mammography among Asian Indian American women: a cross-sectional study in the Baltimore-Washington metropolitan area. Journal of women's health. 2010 Mar;19(3):433–441. doi: 10.1089/jwh.2008.0873. [DOI] [PubMed] [Google Scholar]
- 22.Mazor KM, Williams AE, Roblin DW, et al. Health Literacy and Pap Testing in Insured Women. Journal of Cancer Education. 2014;29(4):698–701. doi: 10.1007/s13187-014-0629-7. 2014/12/01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bener A, Honein G, Carter AO, Da'ar Z, Miller C, Dunn EV. The determinants of breast cancer screening behavior: a focus group study of women in the United Arab Emirates. Oncology nursing forum. 2002 Oct;29(9):E91–98. doi: 10.1188/02.ONF.E91-E98. [DOI] [PubMed] [Google Scholar]
- 24.Soskolne V, Marie S, Manor O. Beliefs, recommendations and intentions are important explanatory factors of mammography screening behavior among Muslim Arab women in Israel. Health education research. 2007 Oct;22(5):665–676. doi: 10.1093/her/cyl132. [DOI] [PubMed] [Google Scholar]
- 25.Zarcadoolas C, Pleasant A, Greer DS. Understanding health literacy: an expanded model. Health Promotion International. 2005 Jun 1;20(2):195–203. doi: 10.1093/heapro/dah609. 2005. [DOI] [PubMed] [Google Scholar]
- 26.Davis TC, Williams MV, Marin E, Parker RM, Glass J. Health literacy and cancer communication. CA: a cancer journal for clinicians. 2002 May-Jun;52(3):134–149. doi: 10.3322/canjclin.52.3.134. [DOI] [PubMed] [Google Scholar]
- 27.Pagán JA, Brown CJ, Asch DA, Armstrong K, Bastida E, Guerra C. Health Literacy and Breast Cancer Screening among Mexican American Women in South Texas. Journal of Cancer Education. 2012 Mar;27(1):132–137. doi: 10.1007/s13187-011-0239-6. 2012 2014-08-30. [DOI] [PubMed] [Google Scholar]
- 28.Lindau ST, Basu A, Leitsch SA. Health literacy as a predictor of follow-up after an abnormal Pap smear: a prospective study. Journal of general internal medicine. 2006 Aug;21(8):829–834. doi: 10.1111/j.1525-1497.2006.00534.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lindau ST, Tomori C, Lyons T, Langseth L, Bennett CL, Garcia P. The association of health literacy with cervical cancer prevention knowledge and health behaviors in a multiethnic cohort of women. American journal of obstetrics and gynecology. 2002 May;186(5):938–943. doi: 10.1067/mob.2002.122091. [DOI] [PubMed] [Google Scholar]
- 30.Lipscombe LL, Hux JE, Booth GL. Reduced Screening Mammography Among Women With Diabetes. Arch Intern Med. 2005;165(18):20902095. doi: 10.1001/archinte.165.18.2090. [DOI] [PubMed] [Google Scholar]
- 31.Jiménez-Garcia RPHD, Hernandez-Barrera VMPH, Carrasco-Garrido PPHD, Gil APHD. Prevalence and Predictors of Breast and Cervical Cancer Screening Among Spanish Women With Diabetes. Diabetes care. 2009 Aug;32(8):1470–1472. doi: 10.2337/dc09-0479. 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Larsson SC, Mantzoros CS, Wolk A. Diabetes mellitus and risk of breast cancer: a meta-analysis. International journal of cancer. Journal international du cancer. 2007 Aug 15;121(4):856–862. doi: 10.1002/ijc.22717. [DOI] [PubMed] [Google Scholar]
- 33.Michels KB, Solomon CG, Hu FB, et al. Type 2 diabetes and subsequent incidence of breast cancer in the Nurses' Health Study. Diabetes care. 2003 Jun;26(6):1752–1758. doi: 10.2337/diacare.26.6.1752. [DOI] [PubMed] [Google Scholar]
- 34.Salinas-Martínez AM, Flores-Cortés LI, Cardona-Chavarría JM, et al. Prediabetes, Diabetes, and Risk of Breast Cancer: A Case-Control Study. Archives of Medical Research. 2014;45(5):432–438. doi: 10.1016/j.arcmed.2014.06.004. 7// [DOI] [PubMed] [Google Scholar]
- 35.Barone BB, Yeh HC, Snyder CF, et al. Long-term all-cause mortality in cancer patients with preexisting diabetes mellitus: a systematic review and meta-analysis. Jama. 2008 Dec 17;300(23):2754–2764. doi: 10.1001/jama.2008.824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lipscombe LL, Goodwin PJ, Zinman B, McLaughlin JR, Hux JE. The impact of diabetes on survival following breast cancer. Breast cancer research and treatment. 2008 May;109(2):389–395. doi: 10.1007/s10549-007-9654-0. [DOI] [PubMed] [Google Scholar]
- 37.Lipscombe L, Fischer H, Austin P, et al. The association between diabetes and breast cancer stage at diagnosis: a population-based study. Breast cancer research and treatment. 2015;150(3):613–620. doi: 10.1007/s10549-015-3323-5. 2015/04/01. [DOI] [PubMed] [Google Scholar]
- 38.Liu BY, O'Malley J, Mori M, et al. The Association of Type and Number of Chronic Diseases with Breast, Cervical, and Colorectal Cancer Screening. The Journal of the American Board of Family Medicine. 2014 Sep 1;27(5):669–681. doi: 10.3122/jabfm.2014.05.140005. 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Vicus D, Sutradhar R, Lu Y, Elit L, Kupets R, Paszat L. The association between cervical cancer screening and mortality from cervical cancer: A population based case–control study. Gynecologic Oncology. 2014;133(2):167–171. doi: 10.1016/j.ygyno.2014.02.037. 5// [DOI] [PubMed] [Google Scholar]
- 40.Waterhouse H. National cancer screening programmes: Risks, benefits and concerns. Practice Nurse. 2013;43(7):44–48. [Google Scholar]
- 41.Gemignani ML. Breast Cancer Screening: Why, When, and How Many? Clinical Obstetrics & Gynecology. 2011;54(1):125–132. doi: 10.1097/GRF.0b013e318208020d. [DOI] [PubMed] [Google Scholar]
- 42.Onitilo AA, Engel JM, Liang H, et al. Mammography Utilization: Patient Characteristics and Breast Cancer Stage at Diagnosis. American Journal of Roentgenology. 2013;201(5):1057–1063. doi: 10.2214/AJR.13.10733. 2013/11/01. [DOI] [PubMed] [Google Scholar]
- 43.Camarota SA, Zeigler K. U.S. Immigrant Population Record 41.3 Million in 2013. Washington, D. C.: 2014. [Google Scholar]
- 44.Zong J, Batalova J. [April 18, 2015];Frequently Requested Statistics on Immigrants and Immigration in the United States. 2015 http://www.migrationpolicy.org/article/frequently-requested-statistics-immigrants-and-immigration-united-states.
- 45.Brown W, Consedine N, Magai C. Time spent in the united states and breast cancer screening behaviors among ethnically diverse immigrant women: Evidence for acculturation? Journal of Immigrant and Minority Health. 2006;8(4):347–358. doi: 10.1007/s10903-006-9005-y. 2006/10/01. [DOI] [PubMed] [Google Scholar]
- 46.Auclair G, Batalova J. Middle Eastern and North African Immigrants in the United States. Washington, D C: 2013. [Google Scholar]
- 47.Schwartz K, Fakhouri M, Bartoces M, Monsur J, Younis A. Mammography screening among Arab American women in metropolitan Detroit. Journal of immigrant and minority health / Center for Minority Public Health. 2008 Dec;10(6):541–549. doi: 10.1007/s10903-008-9140-8. [DOI] [PubMed] [Google Scholar]
- 48.American Cancer Society . Cancer Facts & Figures. Atlanta: 2015. p. GA2015. [Google Scholar]
- 49.Malmgren JA, Parikh J, Atwood MK, Kaplan HG. Impact of Mammography Detection on the Course of Breast Cancer in Women Aged 40–49 Years. Radiology. 2012;262(3):797–806. doi: 10.1148/radiol.11111734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Helvie MA, Chang JT, Hendrick RE, Banerjee M. Reduction in late-stage breast cancer incidence in the mammography era: Implications for overdiagnosis of invasive cancer. Cancer. 2014;120(17):2649–2656. doi: 10.1002/cncr.28784. [DOI] [PubMed] [Google Scholar]
- 51.Press R, Carrasquillo O, Sciacca RR, Giardina EG. Racial/ethnic disparities in time to follow-up after an abnormal mammogram. Journal of women's health. 2008 Jul-Aug;17(6):923–930. doi: 10.1089/jwh.2007.0402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hensley Alford S, Schwartz K, Soliman A, Johnson CC, Gruber SB, Merajver SD. Breast cancer characteristics at diagnosis and survival among Arab-American women compared to European- and African-American women. Breast cancer research and treatment. 2009 Mar;114(2):339–346. doi: 10.1007/s10549-008-9999-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Iqbal J, Ginsburg O, Rochon PA, Sun P, Narod SA. DIfferences in breast cancer stage at diagnosis and cancer-specific survival by race and ethnicity in the united states. Jama. 2015;313(2):165–173. doi: 10.1001/jama.2014.17322. [DOI] [PubMed] [Google Scholar]
- 54.American Cancer Society . Cancer Facts & Figures for Hispanics/Latinos: 2012-2014. Atlanta: p. GA2012. [Google Scholar]
- 55.American Cancer Society Incidence – North American Association of Central Cancer Registries, 2014. [April 20, 2015];Mortality – US mortality data. 2015 http://www.cancer.org/research/cancerfactsstatistics/cancerfactsfigures2015/index.
- 56.Jandorf L, Fatone A, Borker PV, et al. Creating alliances to improve cancer prevention and detection among urban medically underserved minority groups. The East Harlem Partnership for Cancer Awareness. Cancer. 2006 Oct 15;107(8 Suppl):2043–2051. doi: 10.1002/cncr.22153. [DOI] [PubMed] [Google Scholar]
- 57.Arshad S, Williams KP, Mabiso A, Dey S, Soliman AS. Evaluating the Knowledge of Breast Cancer Screening and Prevention among Arab-American Women in Michigan. Journal of Cancer Education. 2011 Mar;26(1):135–138. doi: 10.1007/s13187-010-0130-x. 2014-10-13 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.McLaughlin JM, Anderson RT, Ferketich AK, Seiber EE, Balkrishnan R, Paskett ED. Effect on survival of longer intervals between confirmed diagnosis and treatment initiation among low-income women with breast cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2012 Dec 20;30(36):4493–4500. doi: 10.1200/JCO.2012.39.7695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Jones BA, Dailey A, Calvocoressi L, et al. Inadequate follow-up of abnormal screening mammograms: findings from the race differences in screening mammography process study (United States). Cancer causes & control : CCC. 2005 Sep;16(7):809–821. doi: 10.1007/s10552-005-2905-7. [DOI] [PubMed] [Google Scholar]
- 60.Wujcik D, Shyr Y, Li M, et al. Delay in diagnostic testing after abnormal mammography in low-income women. Oncology nursing forum. 2009 Nov;36(6):709–715. doi: 10.1188/09.ONF.709-715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Pérez-Stable EJ, Afable-Munsuz A, Kaplan CP, et al. Factors Influencing Time to Diagnosis After Abnormal Mammography in Diverse Women. Journal of women's health. 2013;22(2):159–166. doi: 10.1089/jwh.2012.3646. 2013/02/01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Olivotto IA, Gomi A, Bancej C, et al. Influence of delay to diagnosis on prognostic indicators of screen-detected breast carcinoma. Cancer. 2002 Apr 15;94(8):2143–2150. doi: 10.1002/cncr.10453. [DOI] [PubMed] [Google Scholar]
- 63.Richards MA, Westcombe AM, Love SB, Littlejohns P, Ramirez AJ. Influence of delay on survival in patients with breast cancer: a systematic review. Lancet. 1999 Apr 3;353(9159):1119–1126. doi: 10.1016/s0140-6736(99)02143-1. [DOI] [PubMed] [Google Scholar]
- 64.Hensley Alford S, Schwartz K, Soliman A, Johnson CC, Gruber SB, Merajver SD. Breast cancer characteristics at diagnosis and survival among Arab-American women compared to European- and African-American women. Breast cancer research and treatment. 2009 Mar;114(2):339–346. doi: 10.1007/s10549-008-9999-z. 2014-09-10 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chouchane L, Boussen H, Sastry KSR. Breast cancer in Arab populations: molecular characteristics and disease management implications. The Lancet Oncology. 14(10):e417–e424. doi: 10.1016/S1470-2045(13)70165-7. [DOI] [PubMed] [Google Scholar]
- 66.Benard VB, Howe W, Royalty J, Helsel W, Kammerer W, Richardson LC. Timeliness of Cervical Cancer Diagnosis and Initiation of Treatment in the National Breast and Cervical Cancer Early Detection Program. Journal of Women's Health (15409996) 2012;21(7):776–782. doi: 10.1089/jwh.2011.3224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ashing-Giwa KT, Tejero JS, Kim J, et al. Cervical cancer survivorship in a population based sample. Gynecol Oncol. 2009 Feb;112(2):358–364. doi: 10.1016/j.ygyno.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 68.Subramanian S, Trogdon J, Ekwueme DU, Gardner JG, Whitmire JT, Rao C. Cost of cervical cancer treatment: implications for providing coverage to low-income women under the Medicaid expansion for cancer care. Women's health issues : official publication of the Jacobs Institute of Women's Health. 2010 Nov-Dec;20(6):400–405. doi: 10.1016/j.whi.2010.07.002. [DOI] [PubMed] [Google Scholar]
- 69.Ghazal-Aswad S, Hassan T, Badrinath P, Chandrasekhar Nair S. Screening for cervical cancer – the experience of the United Arab Emirates. Hamdan Medical Journal. 2013;6(1):105–110. [Google Scholar]
- 70.Darwish-Yassine M, Wing D. Cancer epidemiology in Arab Americans and Arabs outside the Middle East. Ethnicity & disease. 2005;15(1 Suppl 1):S1–5-8. Winter. [PubMed] [Google Scholar]
- 71.Al-Omran H. Measurement of the knowledge, attitudes, and beliefs of Arab-American adults toward cancer screening and early detection: development of a survey instrument. Ethnicity & disease. 2005;15(1 Suppl 1):S1–15-16. Winter. [PubMed] [Google Scholar]
- 72.Shah SM, Ayash C, Pharaon NA, Gany FM. Arab American Immigrants in New York: Health Care and Cancer Knowledge, Attitudes, and Beliefs. Journal of Immigrant and Minority Health. 2008 Oct;10(5):429–436. doi: 10.1007/s10903-007-9106-2. 2008. [DOI] [PubMed] [Google Scholar]
- 73.Martinez-Donate AP, Vera-Cala LM, Zhang X, Vedro R, Angulo R, Atkinson T. Prevalence and correlates of breast and cervical cancer screening among a Midwest community sample of low-acculturated Latinas. Journal of health care for the poor and underserved. 2013 Nov;24(4):1717–1738. doi: 10.1353/hpu.2013.0165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lawsin C, Erwin D, Bursac Z, Jandorf L. Heterogeneity in Breast and Cervical Cancer Screening Practices Among Female Hispanic Immigrants in the United States. Journal of Immigrant and Minority Health. 2011;13(5):834–841. doi: 10.1007/s10903-010-9378-9. 2011/10/01. [DOI] [PubMed] [Google Scholar]
- 75.Jacobs EA, Karavolos K, Rathouz PJ, Ferris TG, Powell LH. Limited English proficiency and breast and cervical cancer screening in a multiethnic population. American journal of public health. 2005;95(8):1410–1416. doi: 10.2105/AJPH.2004.041418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Komenaka IK, Nodora JN, Hsu CH, et al. Association of health literacy with adherence to screening mammography guidelines. Obstetrics and gynecology. 2015 Apr;125(4):852–859. doi: 10.1097/AOG.0000000000000708. [DOI] [PubMed] [Google Scholar]
- 77.Zarcadoolas C, Greer DS, Pleasant A. Advancing Health Literacy: A Framework for Understanding and Action. Wiley, John & Sons, Inc; San Francisco, CA: 2006. [Google Scholar]
- 78.Pagán J, Brown C, Asch D, Armstrong K, Bastida E, Guerra C. Health Literacy and Breast Cancer Screening among Mexican American Women in South Texas. Journal of Cancer Education. 2012;27(1):132–137. doi: 10.1007/s13187-011-0239-6. [DOI] [PubMed] [Google Scholar]
- 79.Molina Y, Hohl S, Ko L, Rodriguez E, Thompson B, Beresford SA. Understanding the Patient-Provider Communication Needs and Experiences of Latina and Non-Latina White Women Following an Abnormal Mammogram. Journal of Cancer Education. 2014;29(4):781–789. doi: 10.1007/s13187-014-0654-6. 2014/12/01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Armin J, Torres CH, Vivian J, Vergara C, Shaw SJ. Breast self-examination beliefs and practices, ethnicity, and health literacy: Implications for health education to reduce disparities. Health Education Journal. 2014 May 1;73(3):274–284. doi: 10.1177/0017896912471048. 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Garbers S, Chiasson MA. Inadequate Functional Health Literacy in Spanish as a Barrier to Cervical Cancer Screening Among Immigrant Latinas in New York City. Preventing chronic disease. 2004;1(4):A07. 09/15. [PMC free article] [PubMed] [Google Scholar]
- 82.Alkhasawneh IM, Akhu-Zaheya LM, Suleiman SM. Jordanian nurses' knowledge and practice of breast self-examination. Journal of advanced nursing. 2009 Feb;65(2):412–416. doi: 10.1111/j.1365-2648.2008.04891.x. [DOI] [PubMed] [Google Scholar]
- 83.Bener A, El Ayoubi HR, Moore MA, Basha B, Joseph S, Chouchane L. Do we need to maximise the breast cancer screening awareness? Experience with an endogamous society with high fertility. Asian Pacific journal of cancer prevention : APJCP. 2009 Oct-Dec;10(4):599–604. [PubMed] [Google Scholar]
- 84.Montazeri A, Vahdaninia M, Harirchi I, et al. Breast cancer in Iran: need for greater women awareness of warning signs and effective screening methods. Asia Pacific family medicine. 2008;7(1):6. doi: 10.1186/1447-056X-7-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Taha H, Halabi Y, Berggren V, et al. Educational intervention to improve breast health knowledge among women in Jordan. Asian Pacific journal of cancer prevention : APJCP. 2010;11(5):1167–1173. [PubMed] [Google Scholar]
- 86.Arshad S, Williams K, Mabiso A, Dey S, Soliman A. Evaluating the Knowledge of Breast Cancer Screening and Prevention among Arab-American Women in Michigan. Journal of Cancer Education. 2011;26(1):135–138. doi: 10.1007/s13187-010-0130-x. 2011/03/01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Heflin MT, Oddone EZ, Pieper CF, Burchett BM, Cohen HJ. The Effect of Comorbid Illness on Receipt of Cancer Screening by Older People. Journal of the American Geriatrics Society. 2002;50(10):1651–1658. doi: 10.1046/j.1532-5415.2002.50456.x. [DOI] [PubMed] [Google Scholar]
- 88.Yasmeen S, Xing G, Morris C, Chlebowski RT, Romano PS. Comorbidities and mammography use interact to explain racial/ethnic disparities in breast cancer stage at diagnosis. Cancer. 2011;117(14):3252–3261. doi: 10.1002/cncr.25857. [DOI] [PubMed] [Google Scholar]
- 89.Chan W, Yun L, Austin PC, et al. Impact of socio-economic status on breast cancer screening in women with diabetes: a population-based study. Diabetic medicine : a journal of the British Diabetic Association. 2014 Jul;31(7):806–812. doi: 10.1111/dme.12422. [DOI] [PubMed] [Google Scholar]
- 90.Fleming ST, Love MM, Bennett K. Diabetes and cancer screening rates among Appalachian and non-Appalachian residents of Kentucky. Journal of the American Board of Family Medicine : JABFM. 2011 Nov-Dec;24(6):682–692. doi: 10.3122/jabfm.2011.06.110094. [DOI] [PubMed] [Google Scholar]
- 91.Martinez-Huedo MA, Lopez de Andres A, Hernandez-Barrera V, Carrasco-Garrido P, Martinez Hernandez D, Jimenez-Garcia R. Adherence to breast and cervical cancer screening in Spanish women with diabetes: associated factors and trend between 2006 and 2010. Diabetes & metabolism. 2012 Apr;38(2):142–148. doi: 10.1016/j.diabet.2011.09.007. [DOI] [PubMed] [Google Scholar]
- 92.Williams KP, Mabiso A, Todem D, et al. Differences in knowledge of breast cancer screening among African American, Arab American, and Latina women. Prev Chronic Dis. 2011 Jan;8(1):A20. [PMC free article] [PubMed] [Google Scholar]
- 93.Williams KP, Roman L, Meghea CI, Penner L, Hammad A, Gardiner J. Kin KeeperSM: design and baseline characteristics of a community-based randomized controlled trial promoting cancer screening in Black, Latina, and Arab women. Contemporary clinical trials. 2013 Mar;34(2):312–319. doi: 10.1016/j.cct.2012.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Williams KP, Mullan PB, Todem D. Moving from theory to practice: implementing the Kin Keeper Cancer Prevention Model. Health education research. 2009 Apr;24(2):343–356. doi: 10.1093/her/cyn026. [DOI] [PubMed] [Google Scholar]
- 95.Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. Journal of Chronic Diseases. 1987;40(5):373–383. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 96.Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. Journal of Clinical Epidemiology. 1994;47(11):1245–1251. doi: 10.1016/0895-4356(94)90129-5. 11// [DOI] [PubMed] [Google Scholar]
- 97.Williams KP, Templin TN, Hines RD. Answering the call: a tool that measures functional breast cancer literacy. Journal of health communication. 2013;18(11):1310–1325. doi: 10.1080/10810730.2013.778367. [DOI] [PubMed] [Google Scholar]
- 98.Rivera-Vasquez O, Mabiso A, Hammad A, Williams KP. A community-based approach to translating and testing cancer literacy assessment tools. Journal of cancer education : the official journal of the American Association for Cancer Education. 2009;24(4):319–325. doi: 10.1080/08858190902997373. [DOI] [PubMed] [Google Scholar]
- 99.Williams KP, Templin TN, Hines RD. Answering the Call: A Tool That Measures Functional Breast Cancer Literacy. Journal of health communication. 2013 Aug 1; doi: 10.1080/10810730.2013.778367. [DOI] [PubMed] [Google Scholar]
- 100.Williams KP, Templin TN. Bringing the real world to psychometric evaluation of cervical cancer literacy assessments with Black, Latina, and Arab women in real-world settings. Journal of cancer education : the official journal of the American Association for Cancer Education. 2013 Dec;28(4):738–743. doi: 10.1007/s13187-013-0549-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Williams KP, Reckase M, Rivera-Vasquez O. Toward the development of Cancer Literacy Assessment Tools. Michigan J of Public Health. 2008;2(1):21–31. [Google Scholar]
- 102.Stata Data Analysis and Statistical Software [computer program] StataCorp LP; College Station, Texas: 2013. [Google Scholar]
- 103.Robbins JR, Gayar OH, Zaki M, Mahan M, Buekers T, Elshaikh MA. Impact of age-adjusted Charlson comorbidity score on outcomes for patients with early-stage endometrial cancer. Gynecol Oncol. 2013 Dec;131(3):593–597. doi: 10.1016/j.ygyno.2013.10.007. [DOI] [PubMed] [Google Scholar]
- 104.Lomax RG, Hahs-Vaughn DL. An Introduction to Statistical Concepts. Third ed. Taylor & Francis Group, LLC; New York: 2012. [Google Scholar]
- 105.Koppie TM, Serio AM, Vickers AJ, et al. Age-adjusted Charlson comorbidity score is associated with treatment decisions and clinical outcomes for patients undergoing radical cystectomy for bladder cancer. Cancer. 2008;112(11):2384–2392. doi: 10.1002/cncr.23462. [DOI] [PubMed] [Google Scholar]
- 106.Ouellette JR, Small DG, Termuhlen PM. Evaluation of Charlson-Age Comorbidity Index as predictor of morbidity and mortality in patients with colorectal carcinoma. Journal of Gastrointestinal Surgery. 2004;8(8):1061–1067. doi: 10.1016/j.gassur.2004.09.045. 12/1/ [DOI] [PubMed] [Google Scholar]
- 107.Robbins JR, Gayar OH, Zaki M, Mahan M, Buekers T, Elshaikh MA. Impact of Age-Adjusted Charlson Comorbidity score on outcomes for patients with early-stage endometrial cancer. Gynecologic Oncology. 2013;131(3):593–597. doi: 10.1016/j.ygyno.2013.10.007. 12// [DOI] [PubMed] [Google Scholar]
- 108.Liu BY, O'Malley J, Mori M, et al. The Association of Type and Number of Chronic Diseases with Breast, Cervical and Colorectal Cancer Screening in Rural Primary Care Practices. Journal of the American Board of Family Medicine : JABFM. 2014 Sep-Oct;27(5):669–681. doi: 10.3122/jabfm.2014.05.140005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Kiefe CI, Funkhouser E, Fouad MN, May DS. Chronic Disease as a Barrier to Breast and Cervical Cancer Screening. Journal of general internal medicine. 1998;13(6):357–365. doi: 10.1046/j.1525-1497.1998.00115.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Vaeth PA, Satariano WA, Ragland DR. Limiting comorbid conditions and breast cancer stage at diagnosis. The journals of gerontology. Series A, Biological sciences and medical sciences. 2000 Oct;55(10):M593–600. doi: 10.1093/gerona/55.10.m593. [DOI] [PubMed] [Google Scholar]
- 111.Kiefe CI, Funkhouser E, Fouad MN, May DS. Chronic disease as a barrier to breast and cervical cancer screening. Journal of general internal medicine. 1998 Jun;13(6):357–365. doi: 10.1046/j.1525-1497.1998.00115.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Beckman TJ, Cuddihy RM, Scheitel SM, Naessens JM, Killian JM, Pankratz VS. Screening mammogram utilization in women with diabetes. Diabetes care. 2001 Dec;24(12):2049–2053. doi: 10.2337/diacare.24.12.2049. [DOI] [PubMed] [Google Scholar]
- 113.McBean AM, Yu X. The underuse of screening services among elderly women with diabetes. Diabetes care. 2007 Jun;30(6):1466–1472. doi: 10.2337/dc06-2233. [DOI] [PubMed] [Google Scholar]
- 114.Zonderman AB, Ejiogu N, Norbeck J, Evans MK. The Influence of Health Disparities on Targeting Cancer Prevention Efforts. American journal of preventive medicine. 2014;46(3, Supplement 1):S87–S97. doi: 10.1016/j.amepre.2013.10.026. 3// [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kung HC, Hoyert DL, Xu JQ, Murphy SL. Deaths: final data for 2005. National Vital Statistics Reports. 2008;56(10) http://www.cdc.gov/nchs/data/nvsr/nvsr56/nvsr56_10.pdf. [PubMed] [Google Scholar]
- 116.Dumenci L, Matsuyama R, Riddle DL, et al. Measurement of cancer health literacy and identification of patients with limited cancer health literacy. Journal of health communication. 2014;19(Suppl 2):205–224. doi: 10.1080/10810730.2014.943377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Garbers S, Chiasson MA. Inadequate functional health literacy in Spanish as a barrier to cervical cancer screening among immigrant Latinas in New York City. Preventing chronic disease. 2004 Oct;1(4):A07. [PMC free article] [PubMed] [Google Scholar]
- 118.de Groot V, Beckerman H, Lankhorst GJ, Bouter LM. How to measure comorbidity. a critical review of available methods. J Clin Epidemiol. 2003 Mar;56(3):221–229. doi: 10.1016/s0895-4356(02)00585-1. [DOI] [PubMed] [Google Scholar]
- 119.Extermann M, Overcash J, Lyman GH, Parr J, Balducci L. Comorbidity and functional status are independent in older cancer patients. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 1998 Apr;16(4):1582–1587. doi: 10.1200/JCO.1998.16.4.1582. [DOI] [PubMed] [Google Scholar]
- 120.Wedding U, Rohrig B, Klippstein A, Pientka L, Hoffken K. Age, severe comorbidity and functional impairment independently contribute to poor survival in cancer patients. Journal of cancer research and clinical oncology. 2007 Dec;133(12):945–950. doi: 10.1007/s00432-007-0233-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Wu CC, Hsu TW, Chang CM, Yu CH, Lee CC. Age-adjusted Charlson comorbidity index scores as predictor of survival in colorectal cancer patients who underwent surgical resection and chemoradiation. Medicine. 2015 Jan;94(2):e431. doi: 10.1097/MD.0000000000000431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.American Cancer Society [January 10, 2015];Breast Cancer Prevention and Early Detection. 2003 [Google Scholar]
- 123.Moyer VA. Screening for Cervical Cancer: U.S. Preventive Services Task Force Recommendation Statement. Annals of Internal Medicine. 2012;156(12):880–891. doi: 10.7326/0003-4819-156-12-201206190-00424. [DOI] [PubMed] [Google Scholar]
- 124.American Cancer Society [January 10, 2015];New Screening Guidelines for Cervical Cancer. 2012 http://www.cancer.org/cancer/news/new-screening-guidelines-for-cervical-cancer.