Abstract
Background:
Tumoural infiltration of T lymphocytes is determined by local patterns of specific chemokine expression. In this report, we examined the roles of CCL4 and CCL20 in the accumulation of CD8+ cytotoxic T lymphocytes (CTLs) and regulatory T (Treg) lymphocytes in oesophageal squamous cell carcinoma (ESCC), and determined the correlations between chemokine expressions and ESCC patients' survival.
Methods:
Reverse transcriptase–PCR and immunohistochemistry (IHC) staining were performed to examine the expressions of interested genes. Flow cytometry was adopted to check the expressions of CCL4- and CCL6-specific receptors, CCR5 and CCR6, on CTLs and Treg cells. In addition, transwell assay was carried on.
Results:
The CCL4 expression was significantly correlated with the expression of CTL markers (CD8 and Granzyme B), whereas CCL20 was positively correlated with Treg markers (FoxP3 and IL-10). Consistently, CCR5 was found to be mainly expressed on CD8+ T lymphocytes, while CCR6 showed prevalence on Treg lymphocytes and the frequencies of CCR5+CD8+ CTLs and CCR6+ Treg cells were higher in TIL compared with PBMC. Respectively, CCL4 and CCL20 recruited CD8+ and regulatory T cells in vitro. Importantly, high levels of CCL4 in the lesions of ESCC patients predicted prolonged survival. Furthermore, CCL4high/CCL20low group demonstrated better overall survival, whereas CCL4low/CCL20low and CCL4low/CCL20high groups showed the worst overall survival.
Conclusions:
Our data showed that CCL4 and CCL20 recruit functionally different T lymphocyte subsets into oesophageal carcinoma, indicating CCL4 and CCL20 are potential predictors of ESCC patients' survival.
Keywords: CCL4, CCL20, oesophageal squamous cell carcinoma, T lymphocyte, chemotaxis, prognosis
With ∼482 300 new cases and 406 800 deaths each year, oesophageal carcinoma is the ninth most frequent cancer in the world and the fifth most frequent cancer in developed countries (Mariette et al, 2007; Sudo et al, 2014). Although a lot of new therapeutic modalities have been developed to treat oesophageal carcinoma, surgery is still used most frequently (Mariette et al, 2007). As surgical resection is only possible in <20% of patients and the relapse occurs in >60% of patients receiving chemoradiotherapy, the overall survival of patients with oesophageal carcinoma is <20% (Mariette et al, 2007; Sudo et al, 2014). Among the patients with oesophageal carcinoma, squamous cell carcinoma accounts for 90% of cases (Rustgi and El-Serag, 2014). Therefore, it's necessary to develop more promising therapeutic strategies for oesophageal carcinoma, especially oesophageal squamous cell carcinoma (ESCC). Although several key molecular targets have been demonstrated to be useful for prognosis prediction and novel therapeutic interventions, patients benefiting from such approaches remains challenging (Izzo et al, 2007; Yue et al, 2014).
It is well established that the communications between cancer cells and tumour microenvironment modulate cancer progression. The properties of tumour microenvironments have a strong impact on the disease progression, response to therapy and prognosis (Junttila and de Sauvage, 2013). Within tumour microenvironment, chemokines play central roles (Roussos et al, 2011). Apart from their impacts on tumour cells, chemokines decide the constitutions of tumour immune microenvironment (Roussos et al, 2011; Bindea et al, 2013), especially the types and intensities of infiltrated T lymphocytes (Harlin et al, 2009; Chew et al, 2012). Different T lymphocytes lead to opposite outcomes. Infiltration of CD8+ T lymphocytes usually favours better prognosis and prolonged survival, whereas tumoural presence of regulatory T (Treg) lymphocytes means poor prognosis (Zhang et al, 2003; Curiel et al, 2004; Pages et al, 2005; Sato et al, 2005; Galon et al, 2006). Various chemokines are expressed in tumours. However, T lymphocyte subsets are selectively recruited into malignant tissues by specific chemokines (Franciszkiewicz et al, 2012; Gajewski et al, 2013). Therefore, exploring the chemokines deciding the tumoural accumulations of T lymphocyte subsets may provide new target for therapy and help to predict patients' prognosis.
In the past decades, a lot of efforts have been made to study the molecular signals and genetic mutations related with carcinogenesis and progression of oesophageal carcinoma (Hamano et al, 2011; Wu et al, 2014). However, the tumour microenvironmental factors and their relations with oesophageal carcinoma have been very limited debated. Particularly, the effects of chemokines on T lymphocyte recruitment and patients' prognosis are rarely reported. In this report, we explored the impact of CCL4 and CCL20, the respective ligands for CCR5 and CCR6, on migrations of CD8+ and Treg lymphocytes towards ESCC tissues, and on survival of ESCC patients.
Materials and methods
Clinical subjects
A total of 225 patients with ESCC were recruited, following the approval by the Ethics Committee Board of the First Affiliated Hospital of Zhengzhou University. Signed informed consent forms were obtained from all subjects. The detailed information of subjects is listed in Supplementary Table 1. In all, 225 tumour samples and paired 68 peripheral blood samples were collected during surgery in the First Affiliated Hospital of Zhengzhou University between January 2008 and July 2014. In addition, 75 adjacent tissues were sampled.
Isolation of tumour-infiltrating lymphocytes (TILs) and peripheral blood lymphocytes (PBLs)
Freshly sampled tumour tissues were washed with RPMI-1640 (Invitrogen Life Technologies, Carlsbad, CA, USA) 3 times to remove traces of blood. Then, the tissues were cut into small pieces and incubated with 300 μg ml−1 collagenase (Roche, Indianapolis, IN, USA) and 50 μg ml−1 deoxyribonuclease I (Sigma-Aldrich, St Louis, MO, USA) for 2 h at 37 °C. Following this, the samples were mechanically disaggregated, supernatants were collected and TILs were separated by centrifugation at 2000 r.p.m. for 20 min on Ficoll-Paque Plus (Sigma-Aldrich). Peripheral blood lymphocytes were isolated from heparinised blood samples by Ficoll-Paque Plus density centrifugation.
Flow cytometry
Tumour-infiltrating lymphocytes and PBLs were suspended in flow buffer (PBS containing 2% fetal bovine serum), and incubated with anti-CD3, anti-CD8, anti-CD4, anti-CCR5 and anti-CCR6 antibodies (Biolegend, San Diego, CA, USA) against surface antigens for 30 min at 4 °C in the dark. Following that, samples were rinsed in flow buffer 2 times. Then, cells were fixed, permeabilised and stained with anti-FoxP3 antibody (Biolegend). After staining, cells were washed twice and analysed using a BD CantoII flow cytometer (Becton Dickinson, San Jose, CA, USA). To detect nonspecific signals, concentration- and isotype-matched nonspecific antibodies were used.
Reverse transcriptase–PCR
Tumour or marginal tissues were cut into 20 mm of pieces and mechanically grinded. Then, total RNA was extracted using Trizol solution (Invitrogen Life Technologies). Independently, RNA from each sample was reverse-transcribed using PrimeScript RT reagent Kit (Takara Bio, Otsu, Shiga, Japan). Subsequently, the expressions of interested genes were tested. Listed primers were used: 5′-GGAGCCAAAAGGGTCATCATCTC-3′ sense primer and 5′-GAGGGGCCATCCACAGTCTTCT-3′ antisense primer for GAPDH, 5′-CGCTGTCAGATCCCC TTTGT-3′ sense primer and 5′-GAGGAAGGACCCTCTCCCTT-3′ antisense primer for CD8, 5′-GCAGGAAGATCGAAAGTGCG-3′ sense primer and 5′-TACAGCGGGGGCTTAGTTTG-3′ antisense primer for Granzyme B, 5′-GCTTCCTCGCAACT TTGTGG-3′ sense primer and 5′-GCGGAGAGGAGTCCTGAGTA-3′ antisense primer for CCL4, 5′-ATTGTGCGTCTCCTCAGTAAAAA-3′ sense primer and 5′-TGTGATGCTTAAACAAAGCAAAC-3′ antisense primer for CCL20, 5′-TCCAGGACAGGCCACATTTC-3′ sense primer and 5′-GGGATTTGGGAAGGTGCAGA-3′ antisense primer for Foxp3, 5′-ACATCAAGGCGCATGTGAAC-3′ sense primer and 5′-GCCACCCTGATGTCTCAGTT-3′ antisense primer for interleukin-10 (IL-10). The initial step was performed at 94 °C for 5 min, the amplification was performed for 30 cycles of denaturation at 95 °C for 30 s, annealing at 58 °C for 30 s and elongation at 72 °C for 30 s. Following the last cycle, a terminal elongation step (5 min at 72 °C) was added and then the samples were kept at 4 °C. The PCR products were separated on 1.5% agarose gel and recorded. Targeted bands were analysed in ImageJ software (National Institute of Health, Bethesda, MD, USA) and optical densities were calculated. Then, relative expressions of genes were determined. The house-keeping gene GAPDH was used as reference.
Immunohistochemistry
Formalin-fixed, paraffin-embedded sections (3 μm) were deparaffinised in xylene, then rehydrated through graded alcohol and washed briefly in tap water. Endogenous peroxidase was blocked with methanol containing 0.3% hydrogen peroxide for 30 min. To retrieve antigenicity, sections were boiled in 10 mmol l−1 citrate buffer (pH 5.8) for 30 min in microwave (800 W). Following that, sections were incubated with goat serum diluted in PBS (pH 7.4) for 30 min at room temperature. Subsequently, sections were incubated at 4 °C overnight with the primary antibodies specific for CCL4 (dilution 1 : 200) or CCL20 (dilution 1 : 200) (Abcam, Cambridge, UK). Then, sections were rinsed with fresh PBS and incubated with horseradish peroxidase-linked secondary antibodies at room temperature for 30 min. Finally, sections were stained with 3, 3′-diaminobenzidine (DAB) substrate (Dako, Carpinteria, CA, USA) and counterstained with Mayer's haematoxylin. Photos were recorded under microscopy (Leica, Wetzlar, Germany).
Evaluation of immunohistochemical (IHC) staining
All sections were assessed at × 20 magnification by two experienced observers. The staining was evaluated based on the intensity (0=none, weak=1, moderate=2 and high=3) of chemokines staining and the density (0%=0, 1–40%=1, 41–75%=2 and >76%=3) of positive tumour cells. The scores of sections were multiplied intensity and density. If the evaluations did not agree the sections were reevaluated and then classified according to the assessment given most frequently by the observers. The variances within sample were taken into consideration and the average scores were adopted. To determine the cutoff value, we checked the distribution of the frequencies of IHC scores (Supplementary Figure 4). Then, the cutoff value was assigned according to the value at peak. For CCL4, score ⩽1 was considered low expression. For CCL20, score ⩽1.5 was considered low expression.
Purification of T lymphocyte
Tumour-infiltrating lymphocytes were isolated from fresh tumour tissues of ESCC patients as the protocols described above. T lymphocyte were suspended in flow buffer, stained with ant-CD4, anti-CD8, anti-CCR5 and anti-CCR6 antibodies in dark and rinsed. Then, T lymphocyte subsets were independently isolated using MoFlo XDP cytometer (Beckman Coulter Inc., Indianapolis, IN, USA).
Transwell assay
Directional migration of T lymphocytes was evaluated in 24-well plates with 5-μm pore size polycarbonate filters (Corning Inc., Corning, NY, USA). Purified T lymphocytes from TILs of ESCC patients were counted. Then, 4 × 105 of T lymphocytes were added to the upper chambers with 100 μl RPMI-1640 media with 1% serum. The lower chambers were then filled with 600 μl RPMI-1640 media containing 1% serum or supplemented with recombinant human chemokine (CCL4 or CCL20) (Pepro-Tech, Rocky Hill, NJ, USA). The plates were incubated at 37 °C in a 5% CO2 atmosphere for 2.5 h. After that, cells in the bottom chambers were collected. The CD8+ T lymphocytes were counted by a limited 60-s run on cytometer. For analysis of Treg cells, migrated cells were stained with anti-CD4 and anti-FoxP3 antibodies, and then the numbers of interested cells were calculated by flow cytometry.
Statistical analysis
Analyses were performed using the SPSS for Windows version 19.0 (IBM, Armonk, NY, USA). Data were expressed as mean±s.d. Student's t-test and one-way ANOVA were conducted to compare the differences between variables. Spearman's test was adopted to determine the correlation between chemokine genes and T cell-associated markers. Kaplan–Meier survival curves and log-rank statistics were used to evaluate overall survival. Multivariate regression analysis was performed using the Cox proportional hazards model. Values with P <0.05 (two tailed) were considered significant.
Results
The expression levels of CCL4 and CCL20 in ESCC tissues correlate with local expression of, respectively, CTL and Treg lymphocytes markers
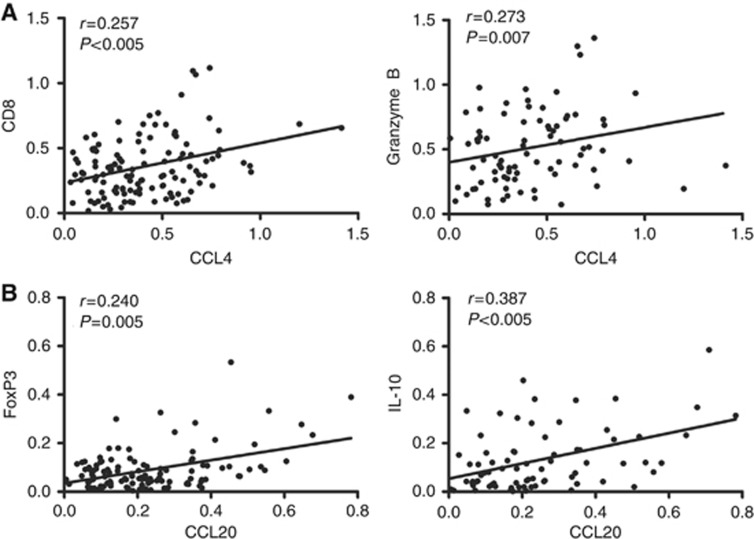
As reported, CCL4 is involved in the migration of not only CD8+ T lymphocytes but also Treg lymphocytes (Harlin et al, 2009; Schlecker et al, 2012). CCL20 mainly dictates the movement of Treg lymphocytes (Chen et al, 2011; Cook et al, 2014). However, the impacts of CCL4 and CCL20 on T lymphocytes in ESCC have not been reported. To elucidate how the tumoural accumulation of T lymphocytes was affected by CCL4 and CCL20 in ESCC, we performed RT–PCR assay to detect the associations between chemokines and T lymphocyte markers in malignant sites. As shown in Figure 1A, the expressions of CCL4 were positively related with cytotoxic T lymphocyte (CTL) markers, CD8 and Granzyme B (rCD8=0.257, P<0.005; rGranB=0.273, P<0.01). Meanwhile, the intensity of CCL20 was clearly correlated with Treg lymphocyte markers: FoxP3 and IL-10 (rFoxP3=0.240, P<0.01; rIL-10=0.387, P<0.005; Figure 1B). Furthermore, the mRNA levels of CTL markers were not statistically associated with that of CCL20 (rCD8=−0.024, P>0.05; rGranB=0.107, P>0.05; Supplementary Figure 1B), whereas the expressions of FoxP3 and IL-10 were independent on CCL4 levels (rFoxP3=0.103, P>0.05; rIL-10=0.159, P>0.05; Figure 1B). These data suggest that CCL4 and CCL20 are probably implicated in the migration and retention of CD8+ CTLs and Treg lymphocytes, respectively, into ESCC lesions.
Figure 1.
Expression levels of CCL4 and CCL20 correlate with, respectively, local expressions of (A) CTL markers (CD8 and Granzyme B) and (B) regulatory T lymphocytes markers (FoxP3 and IL-10). A total of 123 fresh tumour tissues from ESCC patients were collected and processed. RNA was extracted. Reverse transcriptase–PCR assay of interested genes was performed. Spearman's test was performed to determine the correlations among gene expressions.
CCL4- and CCL20-specific receptors, CCR5 and CCR6, are differently expressed on T lymphocyte subsets in ESCC
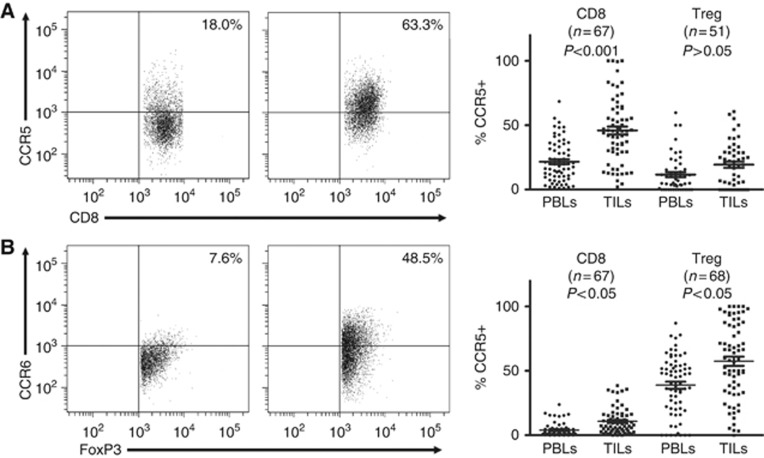
As specific receptors are critical for T lymphocytes moving towards tumour tissues in response to the recruitment of chemokines, monitoring the expression differences of chemokine receptors is helpful for elucidating the roles of specific chemokines. First, we detected the frequencies of CCR5 (CCL4-specific receptor) and CCR6 (CCL20-specific receptor) on peripheral CD8+ T and Treg lymphocytes. As shown in Figure 2A, the proportions of CCR5-positive cells in CD8+ T lymphocytes were significantly greater than in CD4+FoxP3+ Treg lymphocytes (21.60%±15.83% vs 11.61%±14.34% P<0.01). The percentages of CCR6-positive cells in regulatory T lymphocyte groups were 38.83%±22.76%, and the counterparts in CD8+ T lymphocytes were 3.95%±5.03% (P<0.001; Figure 2B). These observations indicate that CCR5 preferentially expresses on CD8+ T lymphocytes, whereas CCR6 shows biased expression on Treg lymphocytes. Following that, we examined tumour-infiltrating T lymphocytes. Compared with 34.38%±13.35% in peripheral blood, the percentages of CD8+ T lymphocytes were obviously increased to 42.84%±15.24% (t=3.156, P<0.005; Supplementary Figure 2). Similarly, the portions of Treg lymphocytes were sharply elevated to 16.65%±9.77% in TILs from 6.67%±6.71% in PBLs (Supplementary Figure 2). The difference is statistically significant (t=6.42, P<0.0001). These observations indicated that CD8+ T and Treg lymphocytes are efficiently recruited into ESCC sites. In tumour-infiltrating CD8+ T lymphocytes, CCR5 expressed on greater portions of cells (Figure 2A). The difference between CCR5-expressing CD8+ T lymphocytes in TILs and PBLs is statistically significant (45.90%±25.28% vs 21.60%±15.82%, P<0.001; Figure 2A). In Treg lymphocytes in tumour sites, CCR5 expression was also observed. However, the intensities of CCR5+ Treg lymphocytes in TILs were not obviously enhanced, compared with the counterparts in PBLs (19.32%±17.41% vs 11.61%±14.34%, P>0.05; Figure 2A). Moreover, CCR5+ Treg lymphocytes were less frequent than CCR5+CD8+ T lymphocytes in tumours (P<0.0001). Conversely, CCR6 expression was robustly increased in tumour-infiltrating Treg lymphocytes (Figure 2B). The portions of CCR6+ Treg lymphocytes in TILs were much greater than that in PBLs (57.49%±29.58% vs 38.83%±22.76%, P<0.05), or CCR6+CD8+ T lymphocytes in TILs (57.49%±29.58% vs 10.77%±10.04%, P<0.0001; Figure 2B). Together with the RT–PCR data, these results indicate that CCL4 plays major roles in recruiting CD8+ T lymphocytes into malignancies, whereas CCL20 mainly directs the tumoural movement of Treg lymphocytes.
Figure 2.
CCR5 and CCR6 show differential expressions on CD8+ and regulatory T lymphocytes in PBLs and matched TILS. Paired PBL and TIL samples were stained with antibodies specific for CD3, CD4, CD8, FoxP3, CCR4 and CCR6. The CD8+ T lymphocytes (CD3+CD8+) and Treg lymphocytes (CD3+CD4+FoxP3+) were separately gated, and then the expressions of CCR5 and CCR6 were determined with multicolour flow cytometry. (A) Representative plots of CCR5 staining on CD8+ T lymphocytes in matched PBLs and TILs (left panel). Paired analysis of CCR5 expressions on CD8+ T and Treg lymphocytes in paired PBLs and TILs (right panel). (B) Representative plots of CCR6 staining on Treg lymphocytes in matched PBLs and TILs (left panel). Paired analysis of CCR6 expressions on CD8+ T and Treg lymphocytes in paired PBLs and TILs (right panel).
CCL4 and CCL20 promote migrations of ESCC patients' CD8+ T and Treg lymphocytes, respectively, in vitro
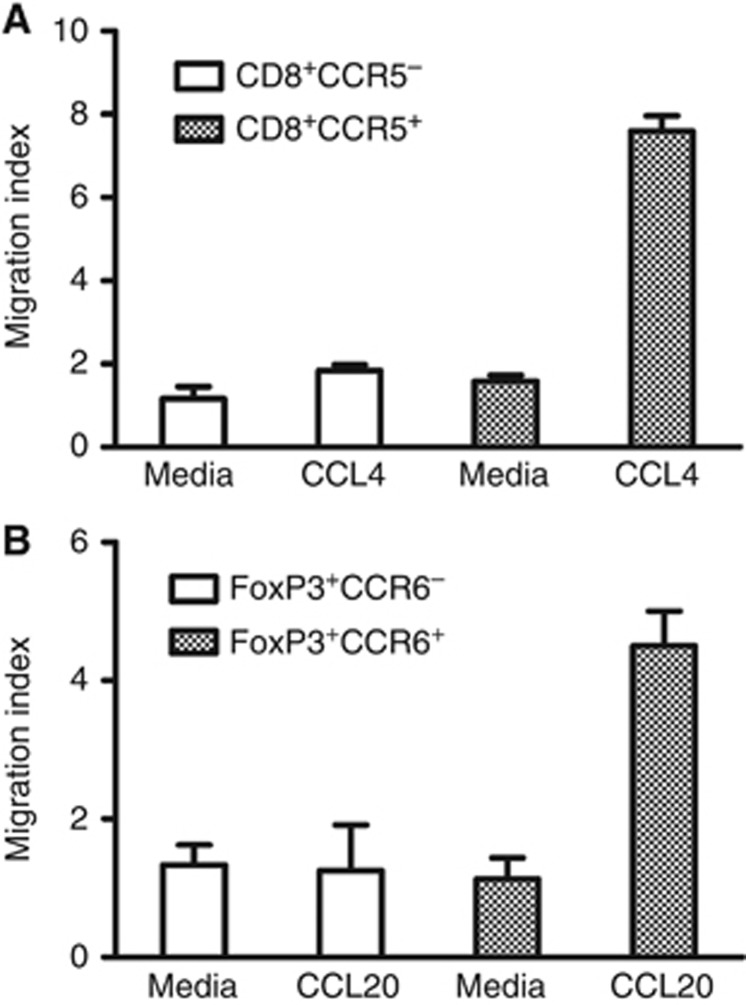
To further validate the roles of CCL4 and CCL20 in T lymphocyte chemotaxis, we performed transwell experiments. First, CD8+CCR5−, CD8+CCR5+, CD4+CCR6− and CD4+CCR6+ T lymphocytes (the purities were over 90%) were purified from TILs. Then, the purified T lymphocytes were separately added into transwell chambers, whose bottom wells were filled of media containing 1% FBS alone or with corresponding chemokines (recombinant human CCL4 or CCL20). The migrated cells in bottom wells were collected, stained and calculated. As shown in Figure 3, CCL4 obviously enhanced the migrating ability of the CCR5-expressing subset in CD8+ T lymphocytes, whereas media alone had little impact on the movement of CD8+ T lymphocytes. Similarly, the directional movement of Treg cells was relying on CCL20 and CCR6 (Figure 3). These observations verify that CCL4–CCR5 and CCL20–CCR6 axes are critical for the migrations of CD8+ T and Treg lymphocytes, respectively.
Figure 3.
CCL6 and CCL20 individually enhance the migrations of CD8+ and regulatory T lymphocytes in vitro. (A) CD8+CCR5− and CD8+CCR5+ T lymphocytes were isolated from TILs of ESCC patients using MoFlo XDP cell sorter (Purity >90%), counted and suspended in media containing 1% FBS. Then, cells were placed in transwell inserts (5 μm pore size) with media containing 1% FBS alone or supplemented with recombinant CCL4 (20 ng ml−1) in the bottom chambers. Migration index was calculated by dividing the number of cells that migrated in each group by the number migrating in CD8+CCR5−/media alone group. (B) Purified CD4+CCR6− and CD4+CCR6+ T lymphocytes from TILs of ESCC patients were counted, suspended and placed in transwell inserts (5 μm pore size) with media containing 1% FBS alone or added with CCL20 (50 ng ml−1) in the bottom chambers as indicated. After incubation, FoxP3+ Treg cells that migrated through the transwell were counted as described in the Materials and Methods. Migration index was calculated by dividing the number of cells that migrated in each group by the number migrating in FoxP3+CCR6−/media alone group. Data shown represent mean±s.d.
Elevated production of both CCL4 and CCL20 in ESCC tissues compared with marginal sites
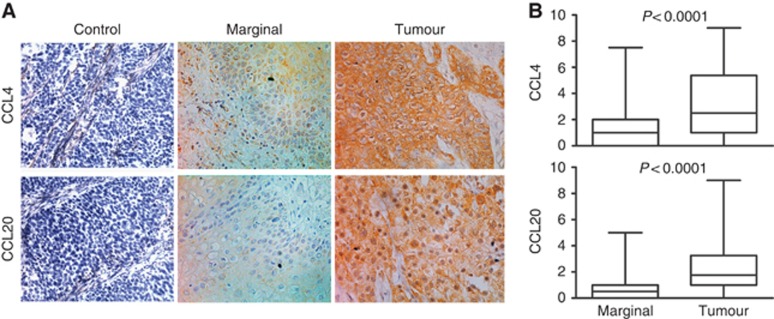
Next, we checked the expressions of CCL4 and CCL20 in tumour and marginal tissues. The IHC analysis showed that only 68.0% of marginal tissues (51 out of 75) were stained positively for CCL4, compared with 93.75% of tumour tissues (120 out of 128) (Figure 4A). The average scores of CCL4 were different between marginal and tumour tissues (1.26±1.50 vs 3.22±2.61; t=5.914, P<0.0001; Figure 4B). For CCL20 staining, 89.84% of tumour samples (115 out of 128) were CCL20 positive in contrast with 54.67% of marginal tissues (41 out of 75) (Figure 4A). The average IHC scores of tumour tissues were obviously higher than marginal tissues (2.28±1.89 vs 0.89±1.25; t=5.648, P<0.0001; Figure 4B). This result was also reflected in the RT–PCR analysis. In paired samples, mRNA intensities of CCL4 and CCL20 were significantly increased in tumour tissues (tCCL4=2.268, P<0.05; tCCL20=2765, P<0.01; Supplementary Figure 3).
Figure 4.
Expressions of CCL4 and CCL20 are elevated in tumour tissues. Both marginal and tumour tissues were checked by immunohistochemistry for the expressions of CCL4 and CCL20. Then, the immunohistochemical scores were calculated as described in the Materials and Methods. (A) Representative photographs of marginal and tumour tissue sections stained with specific primary antibodies or non-immune rabbit IgG, × 400. (B) Scores of CCL4 and CCL20 were significantly higher in malignant tissues (n=128) than that in marginal tissues (n=75).
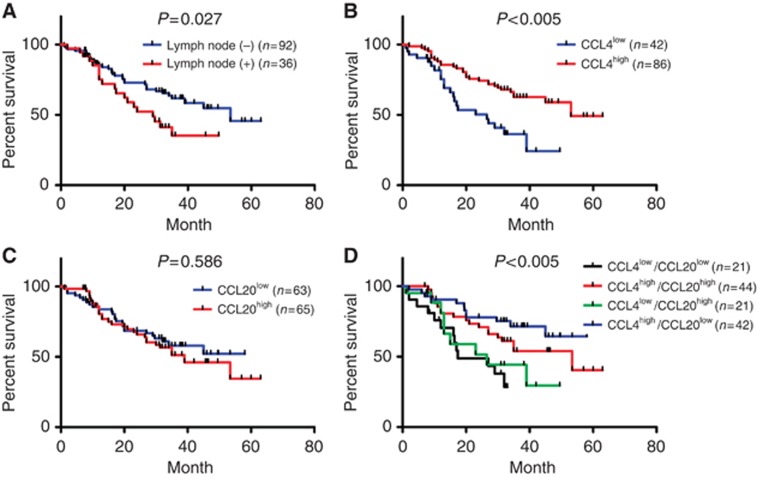
Intratumoural expression of CCL4 and CCL20 predicts overall survival of ESCC patients
First, we checked the associations of the patterns of chemokine expression with clinical and pathologic parameters (Supplementary Table 2). The expressions of CCL4 were not associated with patients' gender, age, clinical stage, T stage, lymph node metastasis and tumour length. Similarly, the CCL20 levels were not associated with these factors, except for lymph node invasion (Supplementary Table 2). Increased expressions of CCL20 were observed in patients with lymph node metastases (P<0.05). The lymph node invasion was significantly associated with patients' survival (P<0.05; Figure 5A and Table 1). Prompted by these data, we investigated the effects of CCL4 and CCL20 on clinical outcomes. For CCL4, increased expression showed a significant association with prolonged overall survival (Figure 5B and Table 1). The medium survival time of CCL4high group was 53 months, whereas it was only 26.5 months in CCL4low group (P<0.001). Between low and high CCL20-expressing patients, the difference in survival was not significant (P>0.05; Figure 5C and Table 1). Nevertheless, further classification of patients into four groups, according to both expressions of CCL4 and CCL20, revealed a prognostic role of CCL20. As shown in Figure 5D, the CCL4high/CCL20low group demonstrated best overall survival compared with other three groups (P<0.005), whereas the CCL4low/CCL20high and CCL4low/CCL20low groups showed poor survival. To determine whether chemokines were predicting markers for patients' survival, multivariate analysis was performed. The results revealed that CCL4 expression is a strong predictive factor for increased survival (P<0.005; Table 1). Lymph node invasion was an independent predictive factor for survival as well (P<0.05; Table 1).
Figure 5.
Intratumoural expression levels of CCL4 and CCL20 predict patients' survival. (A) Kaplan–Meier survival curve comparing the survival of patients with or without lymph node metastasis. (B and C) Kaplan–Meier survival curve comparing the high and the low CCL4 (B) or CCL20 (C) expression groups. (D) Kaplan–Meier survival curve according to the expressions of both CCL4 and CCL20.
Table 1. Univariate and multivariate analysis of overall survival.
|
Univariate analysis |
Multivariate analysis |
|||
|---|---|---|---|---|
| Clinical factors | Deaths/no. of patients | Log-rankP | Risk ratio (95% confidence interval) | P |
|
Gender | ||||
| Male | 40/93 | 0.542 | 0.816 (0.430–1.548) | 0.534 |
| Female | 13/35 | |||
|
Age, years | ||||
| ⩽60 | 21/53 | 0.810 | 1.169 (0.659–2.071) | 0.594 |
| >60 | 32/75 | |||
|
Clinical stage | ||||
| I–IIa | 33/87 | 0.082 | 0.550 (0.067–4.535) | 0.579 |
| IIb–IV | 20/41 | |||
|
Tumour invasion | ||||
| T1 | 4/16 | 0.226 | 1.150 (0.811–1.633) | 0.433 |
| T2 | 21/49 | |||
| T3 | 25/53 | |||
| T4 | 3/10 | |||
|
Lymph node metastasis | ||||
| Negative | 34/92 | 0.027 | 1.946 (1.109–3.479) | 0.021 |
| Positive | 19/36 | |||
|
Tumour length, cm | ||||
| ⩽3 | 28/64 | 0.999 | 0.961 (0.639–1.447) | 0.849 |
| 3–5 | 18/44 | |||
| >5 | 7/20 | |||
|
CCL4 expression | ||||
| Low | 22/42 | 0.001 | 0.386 (0.220–0.678) | 0.001 |
| High | 31/86 | |||
|
CCL20 expression | ||||
| Low | 25/63 | 0.586 | 0.653 (0.098–4.337) | 0.659 |
| High | 28/65 | |||
|
CCL4/CCL20 | ||||
| Low/low | 13/22 | 0.003 | 1.139 (0.646–2.007) | 0.653 |
| Low/high | 9/20 | |||
| High/low | 11/41 | |||
| High/high | 20/45 | |||
The bold value indicates the difference reaches statistical significance.
Discussion
The infiltration of T lymphocytes into tumour tissues is a common phenomenon, but its magnification and character are widely different (Wehler et al, 2011; Salerno et al, 2013; Sherwood et al, 2013; Sugiyama et al, 2013). T lymphocyte accumulation in tumour tissues has significant impacts on tumour differentiation, metastasis, disease progress and patients' survival (Zhang et al, 2006; Chen et al, 2011; Liu et al, 2011; Winkler et al, 2011; Gu-Trantien et al, 2013). The subtypes and intensities of T lymphocytes infiltrated into special sites are tightly related with specific chemokines (Oldham et al, 2012; Oo et al, 2012; Parsonage et al, 2012). Although several studies have examined the expressions of chemokines and chemokine receptors by oesophageal carcinoma cells (Verbeke et al, 2012; Tachezy et al, 2013), the relationship between oesophageal carcinoma-associated chemokine and T lymphocyte accumulation is rarely reported. More importantly, the clinical consequence of chemokine-directed tumoural recruitment of T lymphocytes has not been noticed in oesophageal carcinoma. In this report, we conducted the first study on the effects of CCL4 and CCL20 on T lymphocyte subgroup migration and patients' survival in ESCC. In ESCC, CCL4 expression was correlated with the infiltrations of CD8+ T lymphocytes. The tumoural expression of CCL20 was associated with Treg lymphocyte accumulation. Meanwhile, CCR5 (CCL4-specific receptor) was mainly expressed in oesophageal cancer-associated CD8+ T lymphocytes, and CCR6 (CCL20-specific receptor) was preferentially expressed by oesophageal cancer-associated regulatory T lymphocytes. Most importantly, the patterns of intratumoural expressions of CCL4 and CCL20 were strongly predictive of patients' survival.
Although CCL4 has not been extensively studied, its importance in the recruitment of CD8+ T lymphocytes has been highlighted (Harlin et al, 2009). Conversely, CCL20 plays critical roles in the migration of Treg lymphocytes (Yamazaki et al, 2008; Cook et al, 2014). Consistently, our data showed that CCL4 was dominantly involved in the trafficking of CD8+ T lymphocytes and CCL20 mainly regulated the infiltration of Treg lymphocytes in ESCC. The difference in chemotaxis of CD8+ T and Treg lymphocytes is related with the expressing patterns of specific chemokine receptors. As the only receptor for CCL4, CCR5 shows preferred expression on CD8+ T lymphocytes in cancer patients (Musha et al, 2005; Parsonage et al, 2012). In contrast, CCR6, the specific receptor of CCL20, is highly expressed on Treg lymphocytes (Chen et al, 2011). Consistent with these reports, we found that CCR5 and CCR6 were primarily expressed on CD8+ T and Treg lymphocytes, respectively, in ESCC patients (Figure 2). In ESCC tissues, the portions of CCR5+CD8+ T and CCR6+ Treg lymphocytes were significantly greater than that of CCR6+CD8+ T and CCR5+ Treg lymphocytes, separately (Figure 2). These observations suggested that the specific expressions of receptors are related with the selective recruitments of CD8+ T and Treg lymphocytes by CCL4 and CCL20 into ESCC lesions.
Chemokines have been reported to impact the survival of cancer patients (Popple et al, 2012; Zeng et al, 2013). Apart from their effects on tumour cells themselves, the impacts of chemokines on patient survival are, at least partially, associated with T lymphocyte recruitment (Wehler et al, 2011; Chew et al, 2012; Mulligan et al, 2013). T lymphocyte infiltration is intimately associated with prognosis (Correale et al, 2012; Katz et al, 2013). Intense infiltration of CD8+ T lymphocytes usually indicates favourable prognosis and longer survival times (Seo et al, 2013; Hermans et al, 2014), whereas the accumulation of Treg lymphocytes is related with poor survival (Chen et al, 2011; Suzuki et al, 2013). Our data showed CCL4 and CCL20 were related with tumoural infiltration of CD8+ T and Treg lymphocytes, respectively, in ESCC. Upregulation of CD8+ T lymphocyte-associated CCL4 predicted favoured overall survival (Figure 5B). However, CCL20 was not significantly related with patients' survival (Figure 5C). CCL20 is also involved in the recruitment of T helper 17 (Th17) lymphocytes (Zhao et al, 2014). The Th17 lymphocytes could promote the activation of CD8+ T lymphocytes by recruiting dendritic cells (Martin-Orozco et al, 2009). Chen et al (2012) reported that CCL20 attracted Th17 lymphocytes in oesophageal carcinoma. In our study, CCL20 was correlated with Th17 lymphocyte-related molecules, IL-17 and IL-23 (rIL-17=0.196, P<0.05; rIL-23=0.519, P<0.001; Supplementary Figure 1C), although the coefficient between CCL20 and IL-17 was relative weak. This observation indicated that CCL20 in ESCC probably recruits Treg and Th17 lymphocytes that have opposite functions. The dual effects of CCL20 on tumour immunity may help to explain why CCL20 lacks the prognostic value in ESCC patients. Interestingly, patients' survival was further affected when CCL4 and CCL20 were together analysed. For patients with high levels of CCL4, high expression of CCL20 decreased survival (Figure 5D). This phenomenon is probably related with the interaction between CD8+ T and Treg lymphocytes. Katz et al (2013) reported that the ratio between Treg and CD8+ T lymphocytes was the key to determine survival when both cell types were taken into consideration, and enhanced Treg lymphocyte accumulation hampered the function of CD8+ CTLs. Our data imply that chemokine-directed T lymphocyte infiltration exerts great impacts on ESCC patients' survival.
In addition, we noticed that lymph node metastasis and CCL4 expression were independent predictors for ESCC patients' survival (Figure 5). Relapse occurs in great numbers of patients with oesophageal carcinoma and leads to death (Sudo et al, 2013; Mariette et al, 2014; Sudo et al, 2014). However, the efficient prognosis predictors would help guide treatment and management of oesophageal carcinoma patients according to individual risk, finally improving patients' survival. Lymph node metastasis is the only important prognostic factor in ESCC patients (Sehitogullari et al, 2011; Cho et al, 2014). Our data confirmed the predictive ability of lymph node invasion in ESCC patients (Figure 5A). CCL4 was an independent predictor for survival as well (Figure 5B). Although not significantly associated with survival, CCL20 helped further classify patients into high-, middle- and low-risk groups (Figure 5D). Therefore, CCL4 and CCL20 are valuable predictive factors and helpful for the improvement of personalised therapy.
In summary, our data demonstrate that the chemokine character of ESCC tumour microenvironment determines local recruitments of functionally distinct T lymphocyte subsets and predicts patients' survival. T lymphocyte subgroups, CD8+ CTLs and Treg lymphocytes, were separately recruited towards malignant lesions by CCL4 and CCL20. CCL4 proved to be a strong independent predictive marker of ESCC patients' survival, with particularly strong prognostic value when analysed jointly with CCL20. CCL4 and CCL20 might prove valuable not only as survival predictors, but also as targets for ESCC therapy. Our data highlight the importance of chemokines in immune surveillance and survival of ESCC patients.
Acknowledgments
This study was supported by grants from the China-US (NFSC-NIH) Program for Biomedical Collaborative Research (Grant Nos. 812111102 and P01CA132714-04S1) and the National Natural Science Foundation of China (Grant Nos. 81171986 and 81271815).
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on British Journal of Cancer website (http://www.nature.com/bjc)
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Material
References
- Bindea G, Mlecnik B, Tosolini M, Kirilovsky A, Waldner M, Obenauf AC, Angell H, Fredriksen T, Lafontaine L, Berger A, Bruneval P, Fridman WH, Becker C, Pages F, Speicher MR, Trajanoski Z, Galon J. Spatiotemporal dynamics of intratumoral immune cells reveal the immune landscape in human cancer. Immunity. 2013;39 (4:782–795. doi: 10.1016/j.immuni.2013.10.003. [DOI] [PubMed] [Google Scholar]
- Chen D, Hu Q, Mao C, Jiao Z, Wang S, Yu L, Xu Y, Dai D, Yin L, Xu H. Increased IL-17-producing CD4(+) T cells in patients with esophageal cancer. Cell Immunol. 2012;272 (2:166–174. doi: 10.1016/j.cellimm.2011.10.015. [DOI] [PubMed] [Google Scholar]
- Chen KJ, Lin SZ, Zhou L, Xie HY, Zhou WH, Taki-Eldin A, Zheng SS. Selective recruitment of regulatory T cell through CCR6-CCL20 in hepatocellular carcinoma fosters tumor progression and predicts poor prognosis. PLoS One. 2011;6 (9:e24671. doi: 10.1371/journal.pone.0024671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chew V, Chen J, Lee D, Loh E, Lee J, Lim KH, Weber A, Slankamenac K, Poon RT, Yang H, Ooi LL, Toh HC, Heikenwalder M, Ng IO, Nardin A, Abastado JP. Chemokine-driven lymphocyte infiltration: an early intratumoural event determining long-term survival in resectable hepatocellular carcinoma. Gut. 2012;61 (3:427–438. doi: 10.1136/gutjnl-2011-300509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho JW, Choi SC, Jang JY, Shin SK, Choi KD, Lee JH, Kim SG, Sung JK, Jeon SW, Choi IJ, Kim GH, Jee SR, Lee WS, Jung HY, Korean ESDSG Lymph node metastases in esophageal carcinoma: an endoscopist's view. Clin Endosc. 2014;47 (6:523–529. doi: 10.5946/ce.2014.47.6.523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook KW, Letley DP, Ingram RJ, Staples E, Skjoldmose H, Atherton JC, Robinson K. CCL20/CCR6-mediated migration of regulatory T cells to the Helicobacter pylori-infected human gastric mucosa. Gut. 2014;63 (10:1550–1559. doi: 10.1136/gutjnl-2013-306253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Correale P, Rotundo MS, Botta C, Del Vecchio MT, Ginanneschi C, Licchetta A, Conca R, Apollinari S, De Luca F, Tassone P, Tagliaferri P. Tumor infiltration by T lymphocytes expressing chemokine receptor 7 (CCR7) is predictive of favorable outcome in patients with advanced colorectal carcinoma. Clin Cancer Res. 2012;18 (3:850–857. doi: 10.1158/1078-0432.CCR-10-3186. [DOI] [PubMed] [Google Scholar]
- Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P, Evdemon-Hogan M, Conejo-Garcia JR, Zhang L, Burow M, Zhu Y, Wei S, Kryczek I, Daniel B, Gordon A, Myers L, Lackner A, Disis ML, Knutson KL, Chen L, Zou W. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004;10 (9:942–949. doi: 10.1038/nm1093. [DOI] [PubMed] [Google Scholar]
- Franciszkiewicz K, Boissonnas A, Boutet M, Combadiere C, Mami-Chouaib F. Role of chemokines and chemokine receptors in shaping the effector phase of the antitumor immune response. Cancer Res. 2012;72 (24:6325–6332. doi: 10.1158/0008-5472.CAN-12-2027. [DOI] [PubMed] [Google Scholar]
- Gajewski TF, Schreiber H, Fu YX. Innate and adaptive immune cells in the tumor microenvironment. Nat Immunol. 2013;14 (10:1014–1022. doi: 10.1038/ni.2703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoue F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pages F. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science. 2006;313 (5795:1960–1964. doi: 10.1126/science.1129139. [DOI] [PubMed] [Google Scholar]
- Gu-Trantien C, Loi S, Garaud S, Equeter C, Libin M, de Wind A, Ravoet M, Le Buanec H, Sibille C, Manfouo-Foutsop G, Veys I, Haibe-Kains B, Singhal SK, Michiels S, Rothe F, Salgado R, Duvillier H, Ignatiadis M, Desmedt C, Bron D, Larsimont D, Piccart M, Sotiriou C, Willard-Gallo K. CD4+ follicular helper T cell infiltration predicts breast cancer survival. J Clin Invest. 2013;123 (7:2873–2892. doi: 10.1172/JCI67428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamano R, Miyata H, Yamasaki M, Kurokawa Y, Hara J, Moon JH, Nakajima K, Takiguchi S, Fujiwara Y, Mori M, Doki Y. Overexpression of miR-200c induces chemoresistance in esophageal cancers mediated through activation of the Akt signaling pathway. Clin Cancer Res. 2011;17 (9:3029–3038. doi: 10.1158/1078-0432.CCR-10-2532. [DOI] [PubMed] [Google Scholar]
- Harlin H, Meng Y, Peterson AC, Zha Y, Tretiakova M, Slingluff C, McKee M, Gajewski TF. Chemokine expression in melanoma metastases associated with CD8+ T-cell recruitment. Cancer Res. 2009;69 (7:3077–3085. doi: 10.1158/0008-5472.CAN-08-2281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hermans C, Anz D, Engel J, Kirchner T, Endres S, Mayr D. Analysis of FoxP3+ T-regulatory cells and CD8+ T-cells in ovarian carcinoma: location and tumor infiltration patterns are key prognostic markers. PLoS One. 2014;9 (11:e111757. doi: 10.1371/journal.pone.0111757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Izzo JG, Luthra R, Sims-Mourtada J, Chao KS, Lee JH, Wu TT, Correa AM, Luthra M, Aggarwal B, Hung MC, Ajani JA. Emerging molecular targets in esophageal cancers. Gastrointest Cancer Res. 2007;1 (4 Suppl 2:S3–S6. [PMC free article] [PubMed] [Google Scholar]
- Junttila MR, de Sauvage FJ. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature. 2013;501 (7467:346–354. doi: 10.1038/nature12626. [DOI] [PubMed] [Google Scholar]
- Katz SC, Bamboat ZM, Maker AV, Shia J, Pillarisetty VG, Yopp AC, Hedvat CV, Gonen M, Jarnagin WR, Fong Y, D'Angelica MI, DeMatteo RP. Regulatory T cell infiltration predicts outcome following resection of colorectal cancer liver metastases. Ann Surg Oncol. 2013;20 (3:946–955. doi: 10.1245/s10434-012-2668-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Zhang N, Li Q, Zhang W, Ke F, Leng Q, Wang H, Chen J. Tumor-associated macrophages recruit CCR6+ regulatory T cells and promote the development of colorectal cancer via enhancing CCL20 production in mice. PLoS One. 2011;6 (4:e19495. doi: 10.1371/journal.pone.0019495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mariette C, Dahan L, Mornex F, Maillard E, Thomas PA, Meunier B, Boige V, Pezet D, Robb WB, Le Brun-Ly V, Bosset JF, Mabrut JY, Triboulet JP, Bedenne L, Seitz JF. Surgery alone versus chemoradiotherapy followed by surgery for stage I and II esophageal cancer: final analysis of randomized controlled phase III trial FFCD 9901. J Clin Oncol. 2014;32 (23:2416–2422. doi: 10.1200/JCO.2013.53.6532. [DOI] [PubMed] [Google Scholar]
- Mariette C, Piessen G, Triboulet JP. Therapeutic strategies in oesophageal carcinoma: role of surgery and other modalities. Lancet Oncol. 2007;8 (6:545–553. doi: 10.1016/S1470-2045(07)70172-9. [DOI] [PubMed] [Google Scholar]
- Martin-Orozco N, Muranski P, Chung Y, Yang XO, Yamazaki T, Lu S, Hwu P, Restifo NP, Overwijk WW, Dong C. T helper 17 cells promote cytotoxic T cell activation in tumor immunity. Immunity. 2009;31 (5:787–798. doi: 10.1016/j.immuni.2009.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulligan AM, Raitman I, Feeley L, Pinnaduwage D, Nguyen LT, O'Malley FP, Ohashi PS, Andrulis IL. Tumoral lymphocytic infiltration and expression of the chemokine CXCL10 in breast cancers from the Ontario Familial Breast Cancer Registry. Clin Cancer Res. 2013;19 (2:336–346. doi: 10.1158/1078-0432.CCR-11-3314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Musha H, Ohtani H, Mizoi T, Kinouchi M, Nakayama T, Shiiba K, Miyagawa K, Nagura H, Yoshie O, Sasaki I. Selective infiltration of CCR5(+)CXCR3(+) T lymphocytes in human colorectal carcinoma. Int J Cancer. 2005;116 (6:949–956. doi: 10.1002/ijc.21135. [DOI] [PubMed] [Google Scholar]
- Oldham KA, Parsonage G, Bhatt RI, Wallace DM, Deshmukh N, Chaudhri S, Adams DH, Lee SP. T lymphocyte recruitment into renal cell carcinoma tissue: a role for chemokine receptors CXCR3, CXCR6, CCR5, and CCR6. Eur Urol. 2012;61 (2:385–394. doi: 10.1016/j.eururo.2011.10.035. [DOI] [PubMed] [Google Scholar]
- Oo YH, Banz V, Kavanagh D, Liaskou E, Withers DR, Humphreys E, Reynolds GM, Lee-Turner L, Kalia N, Hubscher SG, Klenerman P, Eksteen B, Adams DH. CXCR3-dependent recruitment and CCR6-mediated positioning of Th-17 cells in the inflamed liver. J Hepatol. 2012;57 (5:1044–1051. doi: 10.1016/j.jhep.2012.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pages F, Berger A, Camus M, Sanchez-Cabo F, Costes A, Molidor R, Mlecnik B, Kirilovsky A, Nilsson M, Damotte D, Meatchi T, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Galon J. Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med. 2005;353 (25:2654–2666. doi: 10.1056/NEJMoa051424. [DOI] [PubMed] [Google Scholar]
- Parsonage G, Machado LR, Hui JW, McLarnon A, Schmaler T, Balasothy M, To KF, Vlantis AC, van Hasselt CA, Lo KW, Wong WL, Hui EP, Chan AT, Lee SP. CXCR6 and CCR5 localize T lymphocyte subsets in nasopharyngeal carcinoma. Am J Pathol. 2012;180 (3:1215–1222. doi: 10.1016/j.ajpath.2011.11.032. [DOI] [PubMed] [Google Scholar]
- Popple A, Durrant LG, Spendlove I, Rolland P, Scott IV, Deen S, Ramage JM. The chemokine, CXCL12, is an independent predictor of poor survival in ovarian cancer. Br J Cancer. 2012;106 (7:1306–1313. doi: 10.1038/bjc.2012.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roussos ET, Condeelis JS, Patsialou A. Chemotaxis in cancer. Nat Rev Cancer. 2011;11 (8:573–587. doi: 10.1038/nrc3078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rustgi AK, El-Serag HB. Esophageal carcinoma. N Engl J Med. 2014;371 (26:2499–2509. doi: 10.1056/NEJMra1314530. [DOI] [PubMed] [Google Scholar]
- Salerno EP, Olson WC, McSkimming C, Shea S, Slingluff CL., Jr T cells in the human metastatic melanoma microenvironment express site-specific homing receptors and retention integrins. Int J Cancer. 2013;134 (3:563–574. doi: 10.1002/ijc.28391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato E, Olson SH, Ahn J, Bundy B, Nishikawa H, Qian F, Jungbluth AA, Frosina D, Gnjatic S, Ambrosone C, Kepner J, Odunsi T, Ritter G, Lele S, Chen YT, Ohtani H, Old LJ, Odunsi K. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc Natl Acad Sci USA. 2005;102 (51:18538–18543. doi: 10.1073/pnas.0509182102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlecker E, Stojanovic A, Eisen C, Quack C, Falk CS, Umansky V, Cerwenka A. Tumor-infiltrating monocytic myeloid-derived suppressor cells mediate CCR5-dependent recruitment of regulatory T cells favoring tumor growth. J Immunol. 2012;189 (12:5602–5611. doi: 10.4049/jimmunol.1201018. [DOI] [PubMed] [Google Scholar]
- Sehitogullari A, Cikman O, Sayir F, Cobanoglu U, Demir C, Demir H. Impact of lymph node metastases in esophageal carcinoma patients is independent of patient age. Asian Pac J Cancer Prev. 2011;12 (3:599–603. [PubMed] [Google Scholar]
- Seo AN, Lee HJ, Kim EJ, Kim HJ, Jang MH, Lee HE, Kim YJ, Kim JH, Park SY. Tumour-infiltrating CD8+ lymphocytes as an independent predictive factor for pathological complete response to primary systemic therapy in breast cancer. Br J Cancer. 2013;109 (10:2705–2713. doi: 10.1038/bjc.2013.634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherwood AM, Emerson RO, Scherer D, Habermann N, Buck K, Staffa J, Desmarais C, Halama N, Jaeger D, Schirmacher P, Herpel E, Kloor M, Ulrich A, Schneider M, Ulrich CM, Robins H. Tumor-infiltrating lymphocytes in colorectal tumors display a diversity of T cell receptor sequences that differ from the T cells in adjacent mucosal tissue. Cancer Immunol Immunother. 2013;62 (9:1453–1461. doi: 10.1007/s00262-013-1446-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sudo K, Taketa T, Correa AM, Campagna MC, Wadhwa R, Blum MA, Komaki R, Lee JH, Bhutani MS, Weston B, Skinner HD, Maru DM, Rice DC, Swisher SG, Hofstetter WL, Ajani JA. Locoregional failure rate after preoperative chemoradiation of esophageal adenocarcinoma and the outcomes of salvage strategies. J Clin Oncol. 2013;31 (34:4306–4310. doi: 10.1200/JCO.2013.51.7250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sudo K, Xiao L, Wadhwa R, Shiozaki H, Elimova E, Taketa T, Blum MA, Lee JH, Bhutani MS, Weston B, Ross WA, Komaki R, Rice DC, Swisher SG, Hofstetter WL, Maru DM, Skinner HD, Ajani JA. Importance of surveillance and success of salvage strategies after definitive chemoradiation in patients with esophageal cancer. J Clin Oncol. 2014;32 (30:3400–3405. doi: 10.1200/JCO.2014.56.7156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiyama D, Nishikawa H, Maeda Y, Nishioka M, Tanemura A, Katayama I, Ezoe S, Kanakura Y, Sato E, Fukumori Y, Karbach J, Jager E, Sakaguchi S. Anti-CCR4 mAb selectively depletes effector-type FoxP3+CD4+ regulatory T cells, evoking antitumor immune responses in humans. Proc Natl Acad Sci USA. 2013;110 (44:17945–17950. doi: 10.1073/pnas.1316796110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki H, Onishi H, Morisaki T, Tanaka M, Katano M. Intratumoral FOXP3+VEGFR2+ regulatory T cells are predictive markers for recurrence and survival in patients with colorectal cancer. Clin Immunol. 2013;146 (1:26–33. doi: 10.1016/j.clim.2012.10.007. [DOI] [PubMed] [Google Scholar]
- Tachezy M, Zander H, Gebauer F, von Loga K, Pantel K, Izbicki JR, Bockhorn M. CXCR7 expression in esophageal cancer. J Transl Med. 2013;11:238. doi: 10.1186/1479-5876-11-238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verbeke H, Geboes K, Van Damme J, Struyf S. The role of CXC chemokines in the transition of chronic inflammation to esophageal and gastric cancer. Biochim Biophys Acta. 2012;1825 (1:117–129. doi: 10.1016/j.bbcan.2011.10.008. [DOI] [PubMed] [Google Scholar]
- Wehler TC, Graf C, Altherr K, Zimmermann T, Brenner W, Thuroff JW, Biesterfeld S, Gockel I, Theobald M, Galle PR, Schimanski CC. SDF1beta expression in renal cell carcinoma correlates with grading and infiltration by CD8+ T-cells. Anticancer Res. 2011;31 (9:2797–2803. [PubMed] [Google Scholar]
- Winkler AE, Brotman JJ, Pittman ME, Judd NP, Lewis JS, Jr, Schreiber RD, Uppaluri R. CXCR3 enhances a T-cell-dependent epidermal proliferative response and promotes skin tumorigenesis. Cancer Res. 2011;71 (17:5707–5716. doi: 10.1158/0008-5472.CAN-11-0907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu C, Wang Z, Song X, Feng XS, Abnet CC, He J, Hu N, Zuo XB, Tan W, Zhan Q, Hu Z, He Z, Jia W, Zhou Y, Yu K, Shu XO, Yuan JM, Zheng W, Zhao XK, Gao SG, Yuan ZQ, Zhou FY, Fan ZM, Cui JL, Lin HL, Han XN, Li B, Chen X, Dawsey SM, Liao L, Lee MP, Ding T, Qiao YL, Liu Z, Liu Y, Yu D, Chang J, Wei L, Gao YT, Koh WP, Xiang YB, Tang ZZ, Fan JH, Han JJ, Zhou SL, Zhang P, Zhang DY, Yuan Y, Huang Y, Liu C, Zhai K, Qiao Y, Jin G, Guo C, Fu J, Miao X, Lu C, Yang H, Wang C, Wheeler WA, Gail M, Yeager M, Yuenger J, Guo ET, Li AL, Zhang W, Li XM, Sun LD, Ma BG, Li Y, Tang S, Peng XQ, Liu J, Hutchinson A, Jacobs K, Giffen C, Burdette L, Fraumeni JF, Jr, Shen H, Ke Y, Zeng Y, Wu T, Kraft P, Chung CC, Tucker MA, Hou ZC, Liu YL, Hu YL, Liu Y, Wang L, Yuan G, Chen LS, Liu X, Ma T, Meng H, Sun L, Li XM, Li XM, Ku JW, Zhou YF, Yang LQ, Wang Z, Li Y, Qige Q, Yang WJ, Lei GY, Chen LQ, Li EM, Yuan L, Yue WB, Wang R, Wang LW, Fan XP, Zhu FH, Zhao WX, Mao YM, Zhang M, Xing GL, Li JL, Han M, Ren JL, Liu B, Ren SW, Kong QP, Li F, Sheyhidin I, Wei W, Zhang YR, Feng CW, Wang J, Yang YH, Hao HZ, Bao QD, Liu BC, Wu AQ, Xie D, Yang WC, Wang L, Zhao XH, Chen SQ, Hong JY, Zhang XJ, Freedman ND, Goldstein AM, Lin D, Taylor PR, Wang LD, Chanock SJ. Joint analysis of three genome-wide association studies of esophageal squamous cell carcinoma in Chinese populations. Nat Genet. 2014;46 (9:1001–1006. doi: 10.1038/ng.3064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamazaki T, Yang XO, Chung Y, Fukunaga A, Nurieva R, Pappu B, Martin-Orozco N, Kang HS, Ma L, Panopoulos AD, Craig S, Watowich SS, Jetten AM, Tian Q, Dong C. CCR6 regulates the migration of inflammatory and regulatory T cells. J Immunol. 2008;181 (12:8391–8401. doi: 10.4049/jimmunol.181.12.8391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yue D, Fan Q, Chen X, Li F, Wang L, Huang L, Dong W, Chen X, Zhang Z, Liu J, Wang F, Wang M, Zhang B, Zhang Y. Epigenetic inactivation of SPINT2 is associated with tumor suppressive function in esophageal squamous cell carcinoma. Exp Cell Res. 2014;322 (1:149–158. doi: 10.1016/j.yexcr.2013.11.009. [DOI] [PubMed] [Google Scholar]
- Zeng J, Yang X, Cheng L, Liu R, Lei Y, Dong D, Li F, Lau QC, Deng L, Nice EC, Xie K, Huang C. Chemokine CXCL14 is associated with prognosis in patients with colorectal carcinoma after curative resection. J Transl Med. 2013;11:6. doi: 10.1186/1479-5876-11-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Conejo-Garcia JR, Katsaros D, Gimotty PA, Massobrio M, Regnani G, Makrigiannakis A, Gray H, Schlienger K, Liebman MN, Rubin SC, Coukos G. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med. 2003;348 (3:203–213. doi: 10.1056/NEJMoa020177. [DOI] [PubMed] [Google Scholar]
- Zhang T, Somasundaram R, Berking C, Caputo L, Van Belle P, Elder D, Czerniecki B, Hotz S, Schuchter L, Spitz FR, Berencsi K, Rani P, Marincola F, Qiu R, Herlyn D. Preferential involvement of CX chemokine receptor 4 and CX chemokine ligand 12 in T-cell migration toward melanoma cells. Cancer Biol Ther. 2006;5 (10:1304–1312. doi: 10.4161/cbt.5.10.3153. [DOI] [PubMed] [Google Scholar]
- Zhao J, Zhang Z, Luan Y, Zou Z, Sun Y, Li Y, Jin L, Zhou C, Fu J, Gao B, Fu Y, Wang FS. Pathological functions of interleukin-22 in chronic liver inflammation and fibrosis with hepatitis B virus infection by promoting T helper 17 cell recruitment. Hepatology. 2014;59 (4:1331–1342. doi: 10.1002/hep.26916. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.