Abstract
Qishen Yiqi formula (QSYQ) has the effect of tonifying Qi and promoting blood circulation, which is widely used to treat the cardiovascular diseases with Qi deficiency and blood stasis syndrome. However, the mechanism of QSYQ to tonify Qi and promote blood circulation is rarely reported at molecular or systems level. This study aimed to elucidate the mechanism of QSYQ based on the protein interaction network (PIN) analysis. The targets' information of the active components was obtained from ChEMBL and STITCH databases and was further used to search against protein-protein interactions by String database. Next, the PINs of QSYQ were constructed by Cytoscape and were analyzed by gene ontology enrichment analysis based on Markov Cluster algorithm. Finally, based on the topological parameters, the properties of scale-free, small world, and modularity of the QSYQ's PINs were analyzed. And based on function modules, the mechanism of QSYQ was elucidated. The results indicated that Qi-tonifying efficacy of QSYQ may be partly attributed to the regulation of amino acid metabolism, carbohydrate metabolism, lipid metabolism, and cAMP metabolism, while QSYQ improves the blood stasis through the regulation of blood coagulation and cardiac muscle contraction. Meanwhile, the “synergy” of formula compatibility was also illuminated.
1. Introduction
Qishen Yiqi formula (QSYQ), consisting of Radix Salvia miltiorrhiza, Panax notoginseng, Dalbergia odorifera, and Astragalus membranaceus, has the effect of tonifying Qi, promoting blood circulation and relieving pain, and hence it has been widely used to treat the cardiovascular diseases with Qi deficiency and blood stasis syndrome [1, 2]. Pharmacological researches have shown that the mechanism of QSYQ is related to improve myocardial function, inhibit platelet aggregation, prevent enlargement of end-diastolic diameter, and slow down ventricular remodeling [3–7]. However, the mechanism of QSYQ was mostly elucidated macroscopically by pharmacology indexes of animal experiments or clinical trials. For example, Tong et al. [6] had reported that the myocardial protection function of QSYQ may relate to the reduction of myocardial cell apoptosis in adriamycin-induced cardiomyopathy animal model. The research of Cui et al. [7] had shown that, by detecting clinical indexes which included the right ventricular end-diastolic volume (RVEDV), end-systolic volume (RVESV), stroke volume (SV), and right ventricular ejection fraction (RVEF), QSYQ could significantly improve the right heart function on patients undergoing valve replacement. The findings of these studies explain the action mechanism of QSYQ to some extent, but the further study of QSYQ is still to be done. Up to now, the mechanism of QSYQ to tonify Qi and promote blood circulation is rarely reported at molecular or systems level. In this study, the mechanism of QSYQ was illuminated by the network analysis approach which has the advantage of evaluating TCM's pharmacological effect as a whole unity at molecular level [8, 9].
Proteins are vital macromolecules, at both cellular and systematic levels, but they rarely act alone. And protein-protein interactions (PPIs) are major bearers of the biological process. So, protein interaction network (PIN) could provide the basis of understanding cellular organization and processes. The GO [10] project is a collaborative effort to construct ontologies which facilitate biologically meaningful annotation of gene products. It provides a collection of well-defined biological terms, spanning biological processes, molecular functions, and cellular components. GO enrichment is a common statistical method used to identify shared associations between proteins and annotations to GO. Module-network and GO analysis may provide an efficient way to illustrate the molecular mechanism of QSYQ.
In this study, a network analysis approach based on functional modules is applied to systematically illuminate the mechanism of QSYQ. The PINs of QSYQ were constructed by Cytoscape, while properties of scale-free, small word, and modularity were analyzed based on topological parameters. Then, the functional modules were identified by gene ontology (GO) enrichment analysis based on Markov Cluster (MCL) algorithm. This study aimed to provide an efficient way to elucidate the mechanism of QSYQ based on functional modules at the molecular level.
2. Materials and Methods
2.1. Targets Mining of Main Active Components of QSYQ
The main active components of QSYQ were used to study the mechanism of QSYQ. By literature retrieval from PubMed and CNKI database, the main active components of QSYQ were obtained based on the principles that components are the main efficacy components, have rich content, and can be absorbed into the blood. The information of the main active components of QSYQ is shown in Table 1.
Table 1.
The information of the main active components of QSYQ.
| Herbs | Active components | Reference |
|---|---|---|
| Salvia miltiorrhiza | Tanshinone IIA, cryptotanshinone, salvianolic acid A, salvianolic acid B, tanshinol, and protocatechuic aldehyde | [46–51] |
| Panax notoginseng | Dencichine, ginsenoside Rb1, ginsenoside Rg1, and notoginsenoside R1 | [52–54] |
| Dalbergia odorifera | Butein, formononetin, isoliquiritigenin, nerolidol | [55–58] |
| Astragalus membranaceus | Calycosin, astragaloside Ι, formononetin, and astragaloside IV | [59, 60] |
The targets' information of main active components of QSYQ was obtained from two parts: pharmacophore virtual screening and the component-protein interaction database including ChEMBL (https://www.ebi.ac.uk/chembl/#) [11] and STITCH 3.1 (http://stitch.embl.de/) [12]. The 27 pharmacophore models which were applied to virtual screen were constructed by our laboratory team [13, 14]. ChEMBL is a manually curated chemical database which contains compound bioactivity data against drug targets. STITCH is a database in which every interaction has a confidence score, and the interactions with a confidence score > 0.7 were selected.
2.2. Network Construction of Single Herb and Formula
The PPIs information of targets was obtained from the online updated database of String 9.1 (http://string-db.org/) which has a confidence score for every protein interaction [15]. PPIs with a confidence score > 0.7 were applied to construct PIN using Cytoscape which is one of the most popular open-source software tools for the visual exploration of biomedical networks composed of protein, gene, and other types of interactions [16]. Every single herb network is formed only by PPIs involving proteins of this herb, and the formula network is formed only by PPIs involving proteins of this formula.
2.3. Network Analysis
The analysis of topological properties based on topological parameters has become very popular for gaining insight into the organization and structure of the resultant large complex networks [17–19]. Therefore, the topological parameters such as degree distribution, average shortest path, and clustering coefficient were analyzed by Network Analyzer [20] in Cytoscape. Properties of scale-free, small word, and modularity of the QSYQ's PIN were also investigated.
Functional modules of the network were explored by the MCL [21] which simulates a flow on the graph by calculating successive powers of the associated adjacency matrix and the value of the inflation parameter strongly influences the number of clusters. Compared to the other algorithms, for example, RNSC [22], MCODE [23], and SPC [24], the MCL is superior with highlighting the robustness to graph alterations [25]. Based on the identified modules, GO enrichment analysis was utilized to predict possible biological roles of the modules by evaluating the involved biological processes, using the BinGO [26] plugin for Cytoscape.
3. Results and Discussion
3.1. The Analysis of the Main Active Components of QSYQ
The main active components of QSYQ are all related to the effect of tonifying Qi or promoting blood stasis.
Tanshinone IIA, cryptotanshinone, salvianolic acid A, salvianolic acid B, tanshinol, and protocatechuic aldehyde are from Salvia miltiorrhiza which is a classical traditional Chinese medicine (TCM) which can promote blood circulation and remove blood stasis with 1000 years of clinical application [27]. It has been demonstrated that Salvia miltiorrhiza can reduce the area of cerebral infarct of ischemia-reperfusion injury rats which results from blood stasis [28]. The chemical components of Salvia miltiorrhiza are divided into water-soluble and liposoluble components. Among the liposoluble components, tanshinone IIA [29] has been reported to improve blood stasis syndrome of patients with coronary heart diseases by inhibiting the circulating inflammatory markers (including IL-6, TNF α, VCAM-1, CD40, sCD40L, MCP-1, and MMP9). Cryptotanshinone [30] has good pharmacological effects on atherosclerosis, while atherosclerosis is one of the diseases resulting from blood stasis. Salvianolic acids, as the main effective components of water-soluble components including salvianolic acid A, salvianolic acid B, tanshinol, and protocatechuic aldehyde, can inhibit thrombosis, thromboxane B2 formation, and platelet aggregation [31]. This indicated that the main active components from Salvia miltiorrhiza are all associated with blood stasis.
Dencichine, ginsenoside Rb1, ginsenoside Rg1, and notoginsenoside R1 are from Panax notoginseng, which is a highly-valued herb and is able to modulate vascular tone such as the activation of blood circulation, removal of blood stasis, and inhibition of platelet aggregation [32]. The main active components of Panax notoginseng include two types of bioactive molecules: one has been reported to have good hemostatic and antithrombotic effects, such as dencichine [33]. In addition, saponins, as the main blood-activating components, which include ginsenoside Rb1, ginsenoside Rg1, and notoginsenoside R1, have showed significant effectiveness on treating cardiovascular diseases [34, 35].
Butein, formononetin, isoliquiritigenin, and nerolidol are from Dalbergia odorifera. Dalbergia odorifera, as blood-activating and stasis-removing TCM, is widely used for promoting blood circulation, relieving pain, and removing blood stasis, which has the effects on antithrombosis, antiplatelet aggregation, antioxidant, antitumor, and anti-inflammation [36]. Volatile oil and flavonoid compounds are two main chemical components of Dalbergia odorifera. According to Guo et al. [37], the ethyl acetate part of Dalbergia odorifera can significantly shorten the bleeding time and clotting time of mice, and it indicated that volatile oil is the material basis of blood-activation in Dalbergia odorifera, while Nerolidol, as a main active component, accounts for 45.23~69.13% of the volatile oil. Butein, formononetin, and isoliquiritigenin, as flavonoid components, show antioxidant activity, antiplatelet aggregation, anti-inflammatory properties, and the capacity for treating cardiovascular diseases [38–40].
Calycosin, astragaloside Ι, formononetin, and astragaloside IV are from Astragalus membranaceus which is a popular Qi-tonifying herb with multiple biological functions, such as antioxidative, antihypertensive, antiaging, and immunomodulatory activities [41]. The main bioactive components including isoflavonoids and triterpene saponins are associated with effects on human health [42]. Isoflavonoids, which are considered “marker components” for the quality control of Astragalus membranaceus including calycosin and formononetin, show strong antioxidant activity, immunoregulation, anti-inflammatory properties, and the capacity for treating cardiovascular diseases [43]. Astragaloside, including astragaloside Ι and astragaloside IV, is the main effective component of astragalus polysaccharides and exerts significant effects on myocardial protection and immunity enhancement [44, 45].
3.2. Targets Information of Active Components of QSYQ
75 targets were obtained from pharmacophore virtual screening. 174 and 65 targets were, respectively, extracted from the ChEMBL and STITCH 3.1. The targets' number of each active component is listed in Table 2, and the additional targets' information is shown in Table S1 in Supplementary Material available online at http://dx.doi.org/10.1155/2015/497314.
Table 2.
The targets' number of each active component from QSYQ.
| Active components | total |
|---|---|
| Tanshinone IIA | 45 |
| Cryptotanshinone | 34 |
| Salvianolic acid A | 17 |
| Salvianolic acid B | 21 |
| Tanshinol | 8 |
| Protocatechuic aldehyde | 9 |
| Dencichine | 7 |
| Ginsenoside Rb1 | 15 |
| Ginsenoside Rg1 | 18 |
| Notoginsenoside R1 | 11 |
| Butein | 23 |
| Formononetin | 27 |
| Isoliquiritigenin | 51 |
| Nerolidol | 3 |
| Calycosin | 8 |
| Astragaloside Ι | 7 |
| Astragaloside IV | 10 |
3.3. Construction of Network
PPIs information of the targets from String 9.1 with their confidence score > 0.7 was imported in Cytoscape 2.8.3, and then union calculation was carried out, followed by the removal of duplicated edges of PPIs using Advanced Network Merge [20] of Plugins. The structural information of constructed networks was listed in Table 3.
Table 3.
The structural information of networks on herbs and formula.
| Networks | Nodes | Edges |
|---|---|---|
| Salvia miltiorrhiza | 604 | 2362 |
| Panax notoginseng | 264 | 963 |
| Dalbergia odorifera | 588 | 2379 |
| Astragalus membranaceus | 399 | 1294 |
| QSYQ | 993 | 4215 |
3.4. Network Analysis
3.4.1. Topological Analysis
All the topological parameters of QSYQ were calculated and they are shown in Table 4.
Table 4.
The simple parameters of protein interaction network of QSYQ.
| Parameters | PIN of QSYQ |
|---|---|
| Clustering coefficient | 0.673 |
| Network diameter (radius) | 11 (1) |
| Network centralization | 0.104 |
| Shortest path | 804676 |
| Mean path length | 4.455 |
| Network heterogeneity | 0.955 |
Notes. The network diameter is the longest distance between any pair of vertices and the radius of a graph is the minimum eccentricity of any vertex. Network centralization is a network index that measures the degree of dispersion of all node centrality scores in a network. And network heterogeneity quantifies the degree of uneven distribution of the network.
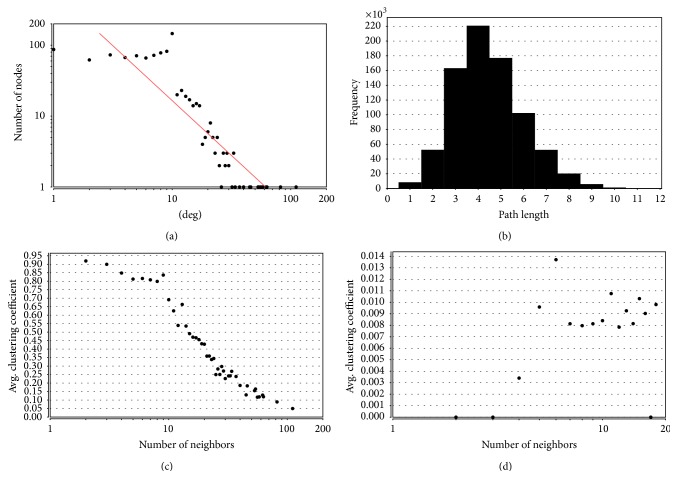
Biological networks have been proposed to have scale-free topology whose degree distribution follows a power law distribution P(k) ~ k −γ (γ < 3) [61]. As shown in Figure 1(a), the degree distribution of the PIN of QSYQ followed the power law distribution and the equation is y = 582.55x −1.547. So, the PIN of QSYQ was a scale-free network.
Figure 1.
Topological properties of network. (a) The degree distribution of QSYQ network; (b) shortest path length distribution of QSYQ network; (c) average clustering coefficient of QSYQ network; (d) average clustering coefficient of random network.
Small world networks have a property that mean path length is short [62]. The shortest path length between any two proteins was calculated, and it turned out to be 4.455. As shown in Figure 1(b), network path length was mostly concentrated in 3–5 steps, which meant that most proteins were closely linked and the PIN of QSYQ was a small world network.
In graph theory, a clustering coefficient is a measure of the degree to which nodes in a graph tend to cluster together. As shown in Figures 1(c) and 1(d), compared with random network whose numbers of nodes and edges are the same as PIN of QSYQ, the clustering coefficient of PIN was higher. It meant the PIN of QSYQ was more modular. These results suggested that the network exhibited the properties of scale-free, small word, and modularity.
3.4.2. Clustering and GO Enrichment Analysis
With the MCL algorithm, 57, 24, 49, 29, and 85 modules were, respectively, identified from salvia miltiorrhiza, Panax notoginseng, Dalbergia odorifera, Astragalus membranaceus, and QSYQ. The modules of QSYQ are shown in Figure 2, and the others are shown in Figures S1–S4.
Figure 2.
Modules in the PIN of QSYQ. With the MCL algorithm, 85 modules are extracted from the network.
The results of functional enrichment analysis of QSYQ using BinGO are shown in Table 5, and they show that QSYQ played a pharmacodynamics with the biological processes, such as DNA metabolic process, regulation of cAMP metabolic process, lipid metabolic process, and the regulation of blood coagulation. The results of functional enrichment analysis of salvia miltiorrhiza, Panax notoginseng, Dalbergia odorifera, and Astragalus membranaceus are shown in Tables S2–S5.
Table 5.
GO biological process terms of the modules of QSYQ.
| Modules | P value | GO terms |
|---|---|---|
| 1 | 1.97E − 14 | Regulation of protein metabolic process |
| 2 | 1.32E − 32 | DNA metabolic process |
| 3 | 2.01E − 28 | Regulation of cAMP metabolic process |
| 4 | 4.16E − 24 | G-protein coupled receptor signaling pathway |
| 5 | 5.28E − 23 | DNA-dependent transcription, initiation |
| 6 | 3.03E − 25 | Transmembrane receptor protein tyrosine kinase signaling pathway |
| 7 | 4.74E − 23 | Cellular lipid metabolic process |
| 8 | 6.43E − 15 | Apoptotic process |
| 9 | 1.80E − 18 | Tricarboxylic acid cycle |
| 10 | 9.96E − 27 | G-protein coupled receptor signaling pathway |
| 11 | 1.32E − 32 | Xenobiotic metabolic process |
| 12 | 2.74E − 15 | Toll-like receptor signaling pathway |
| 13 | 5.96E − 32 | Potassium ion transport |
| 14 | 1.50E − 20 | Lipid metabolic process |
| 15 | 2.57E − 20 | Xenobiotic metabolic process |
| 16 | 8.66E − 12 | Positive regulation of RNA metabolic process |
| 17 | 2.78E − 27 | Regulation of blood coagulation |
| 18 | 3.33E − 14 | Inflammatory response |
| 19 | 1.94E − 16 | Immune response-activating signal transduction |
| 20 | 3.65E − 19 | Apoptotic process |
| 21 | 1.04E − 11 | Regulation of blood coagulation |
| 22 | 2.64E − 16 | Nucleotide metabolic process |
| 23 | 4.14E − 13 | Transmembrane receptor protein tyrosine kinase signaling pathway |
| 24 | 3.85E − 12 | Interferon-gamma-mediated signaling pathway |
| 25 | 5.59E − 07 | Regulation of cellular protein metabolic process |
| 26 | 4.34E − 06 | RNA processing |
| 27 | 1.19E − 22 | Cell cycle phase transition |
| 28 | 2.71E − 06 | Regulation of RNA metabolic process |
| 29 | 9.03E − 14 | Regulation of systemic arterial blood pressure by renin-angiotensin |
| 30 | 5.05E − 18 | Cardiac muscle contraction |
| 31 | 2.53E − 06 | Regulation of RNA splicing |
| 32 | 1.13E − 09 | Carbohydrate metabolic process |
| 33 | 9.21E − 14 | Insulin receptor signaling pathway |
| 34 | 3.77E − 08 | Lipid catabolic process |
| 35 | 2.50E − 22 | Cellular amino acid catabolic process |
| 36 | 1.93E − 17 | Regulation of cell cycle |
| 37 | 2.42E − 14 | Lipid metabolic process |
| 38 | 3.52E − 17 | mRNA metabolic process |
| 39 | 2.31E − 06 | Execution phase of apoptosis |
| 40 | 1.16E − 15 | Toll-like receptor signaling pathway |
| 41 | 3.70E − 11 | RNA biosynthetic process |
| 42 | 3.65E − 12 | Cell cycle phase |
| 43 | 2.80E − 12 | Cellular amino acid metabolic process |
| 44 | 6.84E − 05 | Regulation of cell proliferation |
| 45 | 1.17E − 07 | Inflammatory response |
| 46 | 4.51E − 09 | RNA biosynthetic process |
| 47 | 3.74E − 11 | Regulation of interleukin-1 secretion |
| 48 | 1.49E − 13 | DNA repair |
| 49 | 1.67E − 18 | Insulin receptor signaling pathway |
| 50 | 3.18E − 06 | Negative regulation of RNA metabolic process |
| 51 | 1.24E − 04 | Transport |
| 52 | 8.34E − 14 | Regulation of transforming growth factor beta receptor signaling pathway |
| 53 | 4.47E − 06 | Xenobiotic metabolic process |
| 54 | 1.16E − 08 | Negative regulation of inflammatory response |
| 55 | 1.37E − 07 | Positive regulation of RNA metabolic process |
| 56 | 5.56E − 07 | Transmission of nerve impulse |
| 57 | 5.79E − 12 | Mitotic cell cycle |
| 58 | 6.69E − 06 | Negative regulation of protein metabolic process |
| 59 | 2.66E − 12 | Regulation of apoptotic signaling pathway |
| 60 | 2.00E − 09 | Regulation of apoptotic signaling pathway |
| 61 | 2.47E − 11 | NIK/NF-kappaB cascade |
| 62 | 4.69E − 15 | Vascular endothelial growth factor receptor signaling pathway |
| 63 | 1.98E − 06 | DNA metabolic process |
| 64 | 2.04E − 04 | Chromatin organization |
| 65 | 1.11E − 05 | DNA packaging |
| 66 | 9.39E − 07 | Inflammatory response |
| 67 | 3.22E − 09 | Xenobiotic metabolic process |
| 68 | 8.09E − 12 | Insulin receptor signaling pathway |
| 69 | 8.52E − 04 | DNA-dependent transcription, initiation |
| 70 | 1.56E − 05 | DNA replication |
| 71 | 5.51E − 07 | Toll-like receptor signaling pathway |
| 72 | 2.16E − 08 | Copper ion import |
| 73 | 2.90E − 08 | Transmission of nerve impulse |
| 74 | 4.63E − 07 | Regulation of blood coagulation |
| 76 | 6.90E − 04 | Alanine catabolic process |
| 77 | 6.76E − 05 | RNA biosynthetic process |
| 78 | 7.60E − 04 | Lipid metabolic process |
| 79 | 2.86E − 05 | TRIF-dependent toll-like receptor signaling pathway |
| 81 | 4.18E − 05 | Regulation of type I interferon production |
| 83 | 5.21E − 03 | Cellular lipid metabolic process |
| 84 | 1.27E − 11 | Neural crest cell migration |
| 85 | 2.10E − 03 | AMP catabolic process |
Notes. P value is the probability of obtaining the observed effect under the null hypothesis; a very small P value indicates that the observed effect is very unlikely to have arisen purely by chance.
(1) Modules Related to Qi Deficiency. In TCM, Qi refers to the energy which flows within our body, to support a variety of biological functions such as movement, digesting food, and fight against diseases [43]. Qi deficiency is reflected in the lack of energy. Therefore, the regulation of energy metabolism would improve the Qi deficiency. As shown in Table 5, QSYQ participated in the amino acid metabolic process, carbohydrate metabolic process, lipid metabolic process, and cAMP metabolic process which are related to the energy metabolism and also have been demonstrated to play critical roles in cardiovascular diseases [63–66]. Among them, amino acid metabolism, carbohydrate metabolism, and lipid metabolism are the main energy source of the body.
Amino acid metabolism (module 35) contained proteins such as GLUD2, GLUD1, and GLS. Glutamate dehydrogenase (GLUD) is an enzyme central to the glutamate and energy metabolism of the cell [67]. GLUD activity is raised in order to increase the amount of α-ketoglutarate produced, which can be used to provide energy by being used in the citric acid cycle to ultimately produce ATP. GLUD2 and GLUD1 are the GLUD's isozymes that differ in amino acid sequence but catalyze the same chemical reaction. Glutaminase (GLS) is a multifunctional enzyme involved in energy metabolism [68]. And GLS is the GLS2's isozyme, which regulates cellular energy metabolism by increasing production of glutamate and alpha-ketoglutarate and in turn results in enhanced mitochondrial respiration and ATP generation [69]. This shows that proteins in amino acid metabolism are all involved in the energy metabolism, and QSYQ can improve the Qi deficiency by regulating the amino acid metabolism.
Carbohydrate metabolism is the basis of the body to produce energy. Carbohydrate metabolism (module 32) contained proteins such as GALK1, SORD, and DCXR. Galactokinase 1 (GALK1) is an enzyme (phosphotransferase) that facilitates the phosphorylation of α-D-galactose to galactose 1-phosphate at the expense of one molecule of ATP. Sorbitol dehydrogenase (SORD) is an enzyme in carbohydrate metabolism converting sorbitol, the sugar alcohol form of glucose, into fructose [70]. Dicarbonyl/L-xylulose reductase (DCXR) is involved in carbohydrate metabolism and glucose metabolism which is a highly conserved and phylogenetically widespread enzyme converting L-xylulose into xylitol [71]. This shows that proteins in carbohydrate metabolism make contribution to provide energy for the body by participating in carbohydrate metabolism.
Lipid metabolism (module 83) contained proteins such as ACOT8, AACS. Acyl-coenzyme A thioesterase 8 (ACOT8) is a peroxisomal thioesterase involved more in the oxidation of fatty acids which are in order to generate acetyl-CoA, the entry molecule for the citric acid cycle, the main energy supply of animals [72]. Acetoacetyl-CoA synthetase (AACS) can directly activate ketone bodies for the synthesis of physiologically important lipidic substances such as cholesterol and fatty acid [73]. So, AACS can provide basic substances for energy metabolism. This shows that proteins in lipid metabolism are all related to energy metabolism, and QSYQ can improve the Qi deficiency by regulating the lipid metabolism.
cAMP metabolism (module 3) contained proteins such as GCG, ADCY7, and ADCYAP1. Glucagon (GCG) is a peptide hormone of cAMP metabolic process, which generally elevates the concentration of glucose in the blood by promoting gluconeogenesis and glycogenolysis [74]. Adenylate cyclase type 7 (ADCY7) is a membrane-bound adenylate cyclase that catalyses the formation of cyclic AMP from ATP [75]. ADCYAP1 is also known as pituitary adenylate cyclase-activating polypeptide (PACAP), which stimulates adenylate cyclase and subsequently increases the cAMP level and plays crucial roles in energy metabolism, including lipid metabolism [76].
This indicated that the QSYQ reinforced Qi efficacy by the regulation of the cAMP metabolism, amino acid metabolism, carbohydrate metabolism, and lipid metabolism. And Qi deficiency may be associate with the modules including amino acid metabolism, carbohydrate metabolism, lipid metabolism, and the cAMP metabolism.
(2) Modules Related to Blood Stasis. Blood stasis is caused by disturbance of blood circulation and is reflected in microcirculation relating to vessel and cell function, such as blood viscosity and blood cell adhesion [77]. As shown in Table 5, QSYQ took part in the regulation of blood coagulation and cardiac muscle contraction which can promote blood circulation.
The regulation of blood coagulation (module 17) contained proteins such as GGCX, F2, and SERPIND1. Gamma-glutamyl carboxylase (GGCX) catalyzes the posttranslational modification of vitamin K-dependent proteins which are involved in coagulation [78]. F2 is also known as thrombin (IIa) acts as a serine protease that converts soluble fibrinogen into insoluble strands of fibrin and activation of thrombin is crucial in physiological and pathological coagulation [79]. SERPIND1, known as heparin cofactor II, is a coagulation factor which rapidly inhibits thrombin in the presence of dermatan sulfate or heparin. SERPIND1 deficiency can lead to increased thrombin generation and a hypercoagulable state [80]. This shows that proteins in this module are all involved in the blood coagulation, and QSYQ can improve the blood stasis by the regulation of blood coagulation.
The cardiac muscle contraction (module 30) contained proteins such as MYL2, TNNC1, and TNNI3. MYL2 is also known as myosin regulatory light chain 2, ventricular/cardiac muscle isoform (MLC-2v) which plays a key role in the regulation of cardiac muscle contraction, through its interactions with myosin [81]. TNNC1 is also known as troponin C which is a protein that resides in the troponin complex on actin thin filaments of striated muscle (cardiac) and is responsible for binding calcium to activate muscle contraction [82]. Troponin I (TNNI3) has been shown to interact with TNNC1 [83] and has been reported to have a special role in the control of cardiac contractility [84]. This shows that proteins in this module are all participated in the cardiac muscle contraction. The mechanism of QSYQ has been reported to be related to improve myocardial function [7]. So, QSYQ can promote blood circulation and hence can improve the blood stasis by regulating the cardiac muscle contraction.
This indicated that QSYQ improved the blood stasis through the regulation of blood coagulation and cardiac muscle contraction. And blood stasis may be associated with the modules including the regulation of blood coagulation and cardiac muscle contraction.
3.4.3. The Synergetic Effects of QSYQ
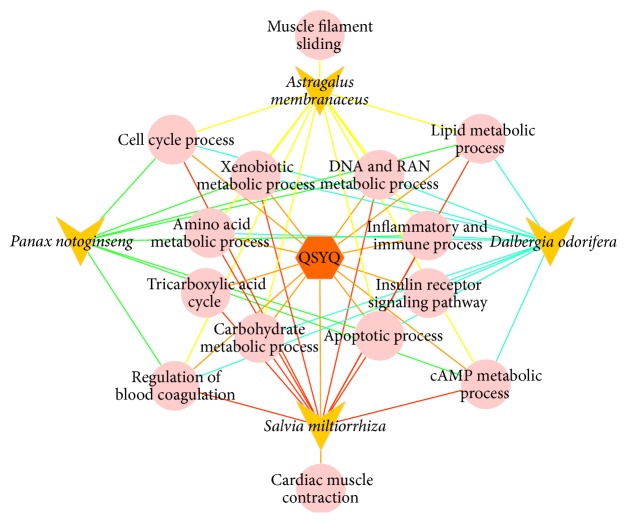
Synergetic effects occur when the efficacy of herbs are combined. The scientific interpretation of these properties is a benefit to the explanation of the compatibility rule and it is further beneficial to the action mechanism of formulae. Synergy refers to the efficacy of combinations of herbs that is greater than the summed responses of each individual herb. As shown in Figure 3, Salvia miltiorrhiza, Panax notoginseng, Dalbergia odorifera, and Astragalus membranaceus all participate in the energy metabolism process, including cAMP metabolic process, carbohydrate metabolic process, and lipid metabolic process, and they hence have the synergetic effect on enhancing the Qi efficacy of QSYQ. The regulation of blood coagulation is involved by four herbs which reinforce the efficacy of promoting the blood circulation of QSYQ. This indicated that the synergy of formula can be illustrated based on the functional modules.
Figure 3.
The schematic diagram of biological processes QSYQ and its herbs involved in. The hexagon represents formula. The triangle represents the herbs of QSYQ. The circle represents the biological processes.
4. Conclusion
In this paper, the PIN of QSYQ exhibited the properties of scale-free, small world, and modularity based on the analysis of topological parameters. A module-based network analysis approach was proposed to expound the mechanism of QSYQ. Qi-tonifying efficacy of QSYQ may be partly attributed to the regulation of amino acid metabolic process, carbohydrate metabolic process, lipid metabolic process, and the cAMP metabolic process, while QSYQ improves the blood stasis through the regulation of blood coagulation and cardiac muscle contraction. A systematic exploration of mechanism of QSYQ based on module-network analysis may bring out the best between research on drug molecules and TCM phenotypic information, so as to facilitate the therapy for the disease.
Further experiments are needed to confirm the conclusions. However, despite the lack of validation of wet experiments, this study provides an efficient way to understand the mechanisms of QSYQ faster and better considering the complexity of TCM analogous formulae. What is more, the scientific intension of “synergy” of TCM can be also illustrated based on the functional modules at the molecular level.
Supplementary Material
Table S1 is about the additional targets' information of each active components from QSYQ. Tables S2–S5 are respective about the results of functional enrichment analysis of salvia miltiorrhiza, Panax notoginseng, Dalbergia odorifera,and Astragalus membranaceus. Figures S1–S4 are respective about the Modules in the PIN of salvia miltiorrhiza, panax notoginseng, dalbergia odorifera, astragalus membranaceus.
Acknowledgments
This research is financially supported by the National Natural Science Foundation of China (nos. 81430094 and 81173522) and the National Key Technology R&D Program (2008BAI51B01) in Beijing University of Chinese Medicine.
Conflict of Interests
The authors declared that there is no conflict of interests.
References
- 1.Ge Y. B., Mao J. Y. Study on the distribution characteristics of the TCM syndromes of 7512 coronary artery disease patients. Shandong Journal of Traditional Chinese Medicine. 2011;4:227–229. [Google Scholar]
- 2.Hou Y. Z., Wang S., Zhao Z. Q., et al. Clinical assessment of complementary treatment with Qishen Yiqi dripping pills on ischemic heart failure: Study protocol for a randomized, double-blind, multicenter, placebo-controlled trial (CACT-IHF) Trials. 2013;14, article 138 doi: 10.1186/1745-6215-14-138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xie D. X., Huang X. Z., Mao B. Y. Mechanisms of Qishen Yiqi dropping pills on ventricular remodeling after myocardial infarction of rat model. Chinese Journal of Experimental Traditional Medical Formulae. 2011;(6):180–183. [Google Scholar]
- 4.Song Y. Z., Guo L. P., Shang H., Wang J. Effects of Qishenyiqi dripping pills on lipid metabolism in experimental hypercholesterolemia rabbits. Jilin Journal of Traditional Chinese Medicine. 2011;1(31):71–73. [Google Scholar]
- 5.Wang Y., Wang J., Guo L., Gao X. Antiplatelet effects of Qishen Yiqi Dropping Pill in platelets aggregation in hyperlipidemic rabbits. Evidence-Based Complementary and Alternative Medicine. 2012;2012:5. doi: 10.1155/2012/205451.205451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tong J.-Y., Xu Y.-J., Bian Y.-P., Shen X.-B., Yan L., Zhu X.-Y. Effect and mechanism of Qishen Yiqi Pills on adriamycin-induced cardiomyopathy in mice. Chinese Journal of Natural Medicines. 2013;11(5):514–518. doi: 10.1016/s1875-5364(13)60093-x. [DOI] [PubMed] [Google Scholar]
- 7.Cui Z.-T., Wei W.-L., Liu M., Wang W.-J. Effect of pretreatment with Qishen Yiqi dropping pills on right cardiac function of patients undergoing valve replacement. Zhongguo Zhongyao Zazhi. 2014;39(5):916–919. doi: 10.4268/cjcmm20140531. [DOI] [PubMed] [Google Scholar]
- 8.Barabási A.-L. Scale-free networks: a decade and beyond. Science. 2009;325(5939):412–413. doi: 10.1126/science.1173299. [DOI] [PubMed] [Google Scholar]
- 9.Janga S. C., Tzakos A. Structure and organization of drug-target networks: insights from genomic approaches for drug discovery. Molecular BioSystems. 2009;5(12):1536–1548. doi: 10.1039/b908147j. [DOI] [PubMed] [Google Scholar]
- 10.Pal D. On gene ontology and function annotation. Bioinformation. 2006;1(3):97–98. doi: 10.6026/97320630001097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bento A. P., Gaulton A., Hersey A., et al. The ChEMBL bioactivity database: an update. Nucleic Acids Research. 2014;42(1):D1083–D1090. doi: 10.1093/nar/gkt1031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kuhn M., Szklarczyk D., Pletscher-Frankild S., et al. STITCH 4: integration of protein-chemical interactions with user data. Nucleic Acids Research. 2014;42(1):D401–D407. doi: 10.1093/nar/gkt1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang X., Xiang Y., Ren Z., Zhang Y., Qiao Y. Rational questing for inhibitors of endothelin converting enzyme-1 from Salvia miltiorrhiza by combining ligand-and structure-based virtual screening. Canadian Journal of Chemistry. 2013;91(6):448–456. doi: 10.1139/cjc-2012-0523. [DOI] [Google Scholar]
- 14.Wang X., Ren Z., He Y., Xiang Y., Zhang Y., Qiao Y. A combination of pharmacophore modeling, molecular docking and virtual screening for iNOS inhibitors from Chinese herbs. Bio-Medical Materials and Engineering. 2014;24(1):1315–1322. doi: 10.3233/bme-130934. [DOI] [PubMed] [Google Scholar]
- 15.Franceschini A., Szklarczyk D., Frankild S., et al. STRING v9.1: protein-protein interaction networks, with increased coverage and integration. Nucleic Acids Research. 2013;41(1):D808–D815. doi: 10.1093/nar/gks1094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Su G., Morris J. H., Demchak B., Bader G. D. Biological network exploration with cytoscape 3. Current Protocols in Bioinformatics. 2014;47:8.13.1–8.13.24. doi: 10.1002/0471250953.bi0813s47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Barabási A.-L., Oltvai Z. N. Network biology: understanding the cell's functional organization. Nature Reviews Genetics. 2004;5(2):101–113. doi: 10.1038/nrg1272. [DOI] [PubMed] [Google Scholar]
- 18.Almaas E. Biological impacts and context of network theory. The Journal of Experimental Biology. 2007;210(part 9):1548–1558. doi: 10.1242/jeb.003731. [DOI] [PubMed] [Google Scholar]
- 19.Zhu X., Gerstein M., Snyder M. Getting connected: analysis and principles of biological networks. Genes and Development. 2007;21(9):1010–1024. doi: 10.1101/gad.1528707. [DOI] [PubMed] [Google Scholar]
- 20.Assenov Y., Ramírez F., Schelhorn S.-E. S.-E., Lengauer T., Albrecht M. Computing topological parameters of biological networks. Bioinformatics. 2008;24(2):282–284. doi: 10.1093/bioinformatics/btm554. [DOI] [PubMed] [Google Scholar]
- 21.Enright A. J., Van Dongen S., Ouzounis C. A. An efficient algorithm for large-scale detection of protein families. Nucleic Acids Research. 2002;30(7):1575–1584. doi: 10.1093/nar/30.7.1575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.King A. D., Pržulj N., Jurisica I. Protein complex prediction via cost-based clustering. Bioinformatics. 2004;20(17):3013–3020. doi: 10.1093/bioinformatics/bth351. [DOI] [PubMed] [Google Scholar]
- 23.Bader G. D., Hogue C. W. V. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinformatics. 2003;4(1, article 2) doi: 10.1186/1471-2105-4-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Blatt M., Wiseman S., Domany E. Superparamagnetic clustering of data. Physical Review Letters. 1996;76(18):3251–3254. doi: 10.1103/PhysRevLett.76.3251. [DOI] [PubMed] [Google Scholar]
- 25.Brohée S., van Helden J. Evaluation of clustering algorithms for protein-protein interaction networks. BMC Bioinformatics. 2006;7, article 488 doi: 10.1186/1471-2105-7-488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Maere S., Heymans K., Kuiper M. BiNGO: a Cytoscape plugin to assess overrepresentation of gene ontology categories in biological networks. Bioinformatics. 2005;21(16):3448–3449. doi: 10.1093/bioinformatics/bti551. [DOI] [PubMed] [Google Scholar]
- 27.Chen X., Guo J., Bao J., Lu J., Wang Y. The anticancer properties of Salvia miltiorrhiza Bunge (Danshen): a systematic review. Medicinal Research Reviews. 2014;34(4):768–794. doi: 10.1002/med.21304. [DOI] [PubMed] [Google Scholar]
- 28.Lao C.-J., Lin J.-G., Kuo J.-S., et al. Effect of salvia miltiorrhiza bunge on cerebral infarct in ischemia-reperfusion injured rats. The American Journal of Chinese Medicine. 2003;31(2):191–200. doi: 10.1142/s0192415x03000916. [DOI] [PubMed] [Google Scholar]
- 29.Shang Q., Wang H., Li S., Xu H. The effect of sodium tanshinone IIA sulfate and simvastatin on elevated serum levels of inflammatory markers in patients with coronary heart disease: a study protocol for a randomized controlled trial. Evidence-based Complementary and Alternative Medicine. 2013;2013:8. doi: 10.1155/2013/756519.756519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu Z., Xu S., Huang X., et al. Cryptotanshinone, an orally bioactive herbal compound from Danshen, attenuates atherosclerosis in apolipoprotein E-deficient mice: role of LOX-1. British Journal of Pharmacology. 2015 doi: 10.1111/bph.13068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jiang R.-W., Lau K.-M., Hon P.-M., Mak T. C. W., Woo K.-S., Fung K.-P. Chemistry and biological activities of caffeic acid derivatives from Salvia miltiorrhiza . Current Medicinal Chemistry. 2005;12(2):237–246. doi: 10.2174/0929867053363397. [DOI] [PubMed] [Google Scholar]
- 32.Ma L. Y., Xiao P. G. Effects of Panax notoginseng saponins on platelet aggregation in rats with middle cerebral artery occlusion or in vitro and on lipid fluidity of platelet membrane. Phytotherapy Research. 1998;12(2):138–140. doi: 10.1002/(SICI)1099-1573(199803)12:2x003C;138::AID-PTR200x0003e;3.0.CO;2-C. [DOI] [Google Scholar]
- 33.Formica J. V., Regelson W. Review of the biology of Quercetin and related bioflavonoids. Food and Chemical Toxicology. 1995;33(12):1061–1080. doi: 10.1016/0278-6915(95)00077-1. [DOI] [PubMed] [Google Scholar]
- 34.Zhang X. J., He C., Tian K., Li P., Su H., Wan J. Ginsenoside Rb1 attenuates angiotensin II-induced abdominal aortic aneurysm through inactivation of the JNK and p38 signaling pathways. Vascular Pharmacology. 2015 doi: 10.1016/j.vph.2015.04.003. [DOI] [PubMed] [Google Scholar]
- 35.Jia C., Xiong M., Wang P., et al. Notoginsenoside R1 attenuates atherosclerotic lesions in ApoE deficient mouse model. PLoS ONE. 2014;9(6) doi: 10.1371/journal.pone.0099849.e99849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yang Z.-H., Mei C., He X.-H., Sun X.-B. Advance in studies on chemical constitutions, pharmacological mechanism and pharmacokinetic profile of dalbergiae odoriferae lignum. Zhongguo Zhong Yao Za Zhi. 2013;38(11):1679–1683. doi: 10.4268/cjcmm20131107. [DOI] [PubMed] [Google Scholar]
- 37.Guo L. B., Huang L. R., Zhao L. H., Tang C. P. Pharmacological screening of effective parts on Xingqi Zhitong and Huoxue Zhixue in Dalbergia odorifera . Journal of Chinese Medicinal Materials. 2007;30(6):696–698. [Google Scholar]
- 38.Cheng Z.-J., Kuo S.-C., Chan S.-C., Ko F.-N., Teng C.-M. Antioxidant properties of butein isolated from Dalbergia odorifera. Biochimica et Biophysica Acta. 1998;1392(2-3):291–299. doi: 10.1016/s0005-2760(98)00043-5. [DOI] [PubMed] [Google Scholar]
- 39.Zhan C., Yang J. Protective effects of isoliquiritigenin in transient middle cerebral artery occlusion-induced focal cerebral ischemia in rats. Pharmacological Research. 2006;53(3):303–309. doi: 10.1016/j.phrs.2005.12.008. [DOI] [PubMed] [Google Scholar]
- 40.Sung J., Lee J. Anti-inflammatory activity of butein and luteolin through suppression of NFκB activation and induction of heme oxygenase-1. Journal of Medicinal Food. 2015;18(5):557–564. doi: 10.1089/jmf.2014.3262. [DOI] [PubMed] [Google Scholar]
- 41.Yeh T.-S., Chuang H.-L., Huang W.-C., Chen Y.-M., Huang C.-C., Hsu M.-C. Astragalus membranaceus improves exercise performance and ameliorates exercise-induced fatigue in trained mice. Molecules. 2014;19(3):2793–2807. doi: 10.3390/molecules19032793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lin L.-Z., He X.-G., Lindenmaier M., et al. Liquid chromatography-electrospray ionization mass spectrometry study of the flavonoids of the roots of Astragalus mongholicus and A. membranaceus . Journal of Chromatography A. 2000;876(1-2):87–95. doi: 10.1016/s0021-9673(00)00149-7. [DOI] [PubMed] [Google Scholar]
- 43.Zhou W., Wang Y. A network-based analysis of the types of coronary artery disease from traditional Chinese medicine perspective: potential for therapeutics and drug discovery. Journal of Ethnopharmacology. 2014;151(1):66–77. doi: 10.1016/j.jep.2013.11.007. [DOI] [PubMed] [Google Scholar]
- 44.Ren S., Zhang H., Mu Y., Sun M., Liu P. Pharmacological effects of Astragaloside IV: a literature review. Journal of Traditional Chinese Medicine. 2013;33(3):413–416. doi: 10.1016/s0254-6272(13)60189-2. [DOI] [PubMed] [Google Scholar]
- 45.Lu M., Tang F., Zhang J., et al. Astragaloside IV attenuates injury caused by myocardial ischemia/reperfusion in rats via regulation of toll-like receptor 4/nuclear factor-kappaB signaling pathway. Phytotherapy Research. 2015;29(4):599–606. doi: 10.1002/ptr.5297. [DOI] [PubMed] [Google Scholar]
- 46.Ye G., Wang C.-S., Li Y.-Y., Ren H., Guo D.-A. Simultaneous determination and pharmacokinetic studies on (3,4-dihydroxyphenyl)-lactic acid and protocatechuic aldehyde in rat serum after oral administration of Radix Salviae miltiorrhizae extract. Journal of Chromatographic Science. 2003;41(6):327–330. doi: 10.1093/chromsci/41.6.327. [DOI] [PubMed] [Google Scholar]
- 47.Lu T., Yang J., Gao X., et al. Plasma and urinary tanshinol from Salvia miltiorrhiza (Danshen) can be used as pharmacokinetic markers for cardiotonic pills, a cardiovascular herbal medicine. Drug Metabolism and Disposition. 2008;36(8):1578–1586. doi: 10.1124/dmd.108.021592. [DOI] [PubMed] [Google Scholar]
- 48.Mahesh R., Jung H. W., Kim G. W., Kim Y. S., Park Y.-K. Cryptotanshinone from Salviae miltiorrhizae radix inhibits sodium-nitroprusside-induced apoptosis in neuro-2 cells. Phytotherapy Research. 2012;26(8):1211–1219. doi: 10.1002/ptr.3705. [DOI] [PubMed] [Google Scholar]
- 49.Chiu S.-C., Huang S.-Y., Chang S.-F., et al. Potential therapeutic roles of tanshinone IIA in human bladder cancer cells. International Journal of Molecular Sciences. 2014;15(9):15622–15637. doi: 10.3390/ijms150915622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang T., Xu J., Li D., et al. Salvianolic acid A, a matrix metalloproteinase-9 inhibitor of Salvia miltiorrhiza, attenuates aortic aneurysm formation in apolipoprotein E-deficient mice. Phytomedicine. 2014;21(10):1137–1145. doi: 10.1016/j.phymed.2014.05.003. [DOI] [PubMed] [Google Scholar]
- 51.Fu J., Fan H.-B., Guo Z., et al. Salvianolic acid B attenuates spinal cord ischemia-reperfusion-induced neuronal injury and oxidative stress by activating the extracellular signal-regulated kinase pathway in rats. Journal of Surgical Research. 2014;188(1):222–230. doi: 10.1016/j.jss.2013.11.1118. [DOI] [PubMed] [Google Scholar]
- 52.Wei Y., Li P., Fan H., et al. Metabolism study of notoginsenoside R1, ginsenoside Rg1 and ginsenoside Rb1 of radix panax notoginseng in zebrafish. Molecules. 2011;16(8):6621–6633. doi: 10.3390/molecules16086621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Huang J., Ding L., Shi D., et al. Transient receptor potential vanilloid-1 participates in the inhibitory effect of ginsenoside Rg1 on capsaicin-induced interleukin-8 and prostaglandin E2 production in HaCaT cells. Journal of Pharmacy and Pharmacology. 2012;64(2):252–258. doi: 10.1111/j.2042-7158.2011.01392.x. [DOI] [PubMed] [Google Scholar]
- 54.Qiao C.-F., Liu X.-M., Cui X.-M., et al. High-performance anion-exchange chromatography coupled with diode array detection for the determination of dencichine in Panax notoginseng and related species. Journal of Separation Science. 2013;36(15):2401–2406. doi: 10.1002/jssc.201300334. [DOI] [PubMed] [Google Scholar]
- 55.Cheng Z.-J., Kuo S.-C., Chan S.-C., Ko F.-N., Teng C.-M. Antioxidant properties of butein isolated from Dalbergia odorifera . Biochimica et Biophysica Acta. 1998;1392(2-3):291–299. doi: 10.1016/s0005-2760(98)00043-5. [DOI] [PubMed] [Google Scholar]
- 56.Lee S. H., Kim J. Y., Seo G. S., Kim Y.-C., Sohn D. H. Isoliquiritigenin, from Dalbergia odorifera, up-regulates anti-inflammatory heme oxygenase-1 expression in RAW264.7 macrophages. Inflammation Research. 2009;58(5):257–262. doi: 10.1007/s00011-008-8183-6. [DOI] [PubMed] [Google Scholar]
- 57.Xu L., Shi H., Liang T., et al. Selective separation of flavonoid glycosides in Dalbergia odorifera by matrix solid-phase dispersion using titania. Journal of Separation Science. 2011;34(11):1347–1354. doi: 10.1002/jssc.201100024. [DOI] [PubMed] [Google Scholar]
- 58.Silva M. P. N., Oliveira G. L. S., De Carvalho R. B. F., et al. Antischistosomal activity of the terpene nerolidol. Molecules. 2014;19(3):3793–3803. doi: 10.3390/molecules19033793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kwon H.-J., Hwang J., Lee S.-K., Park Y.-D. Astragaloside content in the periderm, cortex, and xylem of Astragalus membranaceus root. Journal of Natural Medicines. 2013;67(4):850–855. doi: 10.1007/s11418-013-0741-8. [DOI] [PubMed] [Google Scholar]
- 60.Li W., Sun Y. N., Yan X. T., et al. Flavonoids from Astragalus membranaceus and their inhibitory effects on LPS-stimulated pro-inflammatory cytokine production in bone marrow-derived dendritic cells. Archives of Pharmacal Research. 2014;37(2):186–192. doi: 10.1007/s12272-013-0174-7. [DOI] [PubMed] [Google Scholar]
- 61.Barabási A.-L., Albert R. Emergence of scaling in random networks. Science. 1999;286(5439):509–512. doi: 10.1126/science.286.5439.509. [DOI] [PubMed] [Google Scholar]
- 62.Strogatz S. H. Exploring complex networks. Nature. 2001;410(6825):268–276. doi: 10.1038/35065725. [DOI] [PubMed] [Google Scholar]
- 63.Wang Z., Li F. Effects of ilexonin A on cAMP metabolism in platelets. Chinese Medical Sciences Journal. 1993;8(4):215–217. [PubMed] [Google Scholar]
- 64.Gaziano J. M., Sesso H. D., Breslow J. L., Hennekens C. H., Buring J. E. Relation between systemic hypertension and blood lipids on the risk of myocardial infarction. American Journal of Cardiology. 1999;84(7):768–773. doi: 10.1016/s0002-9149(99)00435-x. [DOI] [PubMed] [Google Scholar]
- 65.Mann J. Dietary carbohydrate: relationship to cardiovascular disease and disorders of carbohydrate metabolism. European Journal of Clinical Nutrition. 2007;61(supplement 1):S100–S111. doi: 10.1038/sj.ejcn.1602940. [DOI] [PubMed] [Google Scholar]
- 66.Yun K. U., Ryu C. S., Oh J. M., et al. Plasma homocysteine level and hepatic sulfur amino acid metabolism in mice fed a high-fat diet. European Journal of Nutrition. 2013;52(1):127–134. doi: 10.1007/s00394-011-0294-0. [DOI] [PubMed] [Google Scholar]
- 67.Rosso L., Marques A. C., Reichert A. S., Kaessmann H. Mitochondrial targeting adaptation of the hominoid-specific glutamate dehydrogenase driven by positive darwinian selection. PLoS Genetics. 2008;4(8) doi: 10.1371/journal.pgen.1000150.e1000150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bae N., Wang Y., Li L., Rayport S., Lubec G. Network of brain protein level changes in glutaminase deficient fetal mice. Journal of Proteomics. 2013;80:236–249. doi: 10.1016/j.jprot.2013.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hu W., Zhang C., Wu R., Sun Y., Levine A., Feng Z. Glutaminase 2, a novel p53 target gene regulating energy metabolism and antioxidant function. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(16):7455–7460. doi: 10.1073/pnas.1001006107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.El-Kabbani O., Darmanin C., Chung R. P.-T. Sorbitol dehydrogenase: structure, function and ligand design. Current Medicinal Chemistry. 2004;11(4):465–476. doi: 10.2174/0929867043455927. [DOI] [PubMed] [Google Scholar]
- 71.Lee S.-K., Son L. T., Choi H.-J., Ahnn J. Dicarbonyl/l-xylulose reductase (DCXR): the multifunctional pentosuria enzyme. International Journal of Biochemistry and Cell Biology. 2013;45(11):2563–2567. doi: 10.1016/j.biocel.2013.08.010. [DOI] [PubMed] [Google Scholar]
- 72.Hunt M. C., Siponen M. I., Alexson S. E. H. The emerging role of acyl-CoA thioesterases and acyltransferases in regulating peroxisomal lipid metabolism. Biochimica et Biophysica Acta. 2012;1822(9):1397–1410. doi: 10.1016/j.bbadis.2012.03.009. [DOI] [PubMed] [Google Scholar]
- 73.Hasegawa S., Ikeda Y., Yamasaki M., Fukui T. The role of acetoacetyl-CoA synthetase, a ketone body-utilizing enzyme, in 3T3-L1 adipocyte differentiation. Biological and Pharmaceutical Bulletin. 2012;35(11):1980–1985. doi: 10.1248/bpb.b12-00435. [DOI] [PubMed] [Google Scholar]
- 74.Ahrén B. Glucagon—early breakthroughs and recent discoveries. Peptides. 2015;67:74–81. doi: 10.1016/j.peptides.2015.03.011. [DOI] [PubMed] [Google Scholar]
- 75.Hellevuo K., Berry R., Sikela J. M., Tabakoff B. Localization of the gene for a novel human adenylyl cyclase (ADCY7) to chromosome 16. Human Genetics. 1995;95(2):197–200. doi: 10.1007/BF00209401. [DOI] [PubMed] [Google Scholar]
- 76.Tomimoto S., Ojika T., Shintani N., et al. Markedly reduced white adipose tissue and increased insulin sensitivity in Adcyap1-deficient mice. Journal of Pharmacological Sciences. 2008;107(1):41–48. doi: 10.1254/jphs.fp0072173. [DOI] [PubMed] [Google Scholar]
- 77.Hou J., Wang J., Lin C., et al. Circulating microRNA profiles differ between Qi-stagnation and Qi-deficiency in coronary heart disease patients with blood stasis syndrome. Evidence-Based Complementary and Alternative Medicine. 2014;2014:9. doi: 10.1155/2014/926962.926962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.King C. R., Deych E., Milligan P., et al. Gamma-glutamyl carboxylase and its influence on warfarin dose. Thrombosis and Haemostasis. 2010;104(4):750–754. doi: 10.1160/TH09-11-0763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Guria K., Guria G. T. Spatial aspects of blood coagulation: two decades of research on the self-sustained traveling wave of thrombin. Thrombosis Research. 2015;135(3):423–433. doi: 10.1016/j.thromres.2014.12.014. [DOI] [PubMed] [Google Scholar]
- 80.Parker K. A., Tollefsen D. M. The protease specificity of heparin cofactor II. Inhibition of thrombin generated during coagulation. The Journal of Biological Chemistry. 1985;260(6):3501–3505. [PubMed] [Google Scholar]
- 81.Sheikh F., Lyon R. C., Chen J. Getting the skinny on thick filament regulation in cardiac muscle biology and disease. Trends in Cardiovascular Medicine. 2014;24(4):133–141. doi: 10.1016/j.tcm.2013.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Townsend P. J., Yacoub M. H., Barton P. J. R. Assignment of the human cardiac/slow skeletal muscle troponin C gene (TNNC1) between D3S3118 and GCT4B10 on the short arm of chromosome 3 by somatic cell hybrid analysis. Annals of Human Genetics. 1997;61(4):375–377. doi: 10.1046/j.1469-1809.1997.6140375.x. [DOI] [PubMed] [Google Scholar]
- 83.Ward D. G., Brewer S. M., Gallon C. E., Gao Y., Levine B. A., Trayer I. P. NMR and mutagenesis studies on the phosphorylation region of human cardiac troponin I. Biochemistry. 2004;43(19):5772–5781. doi: 10.1021/bi036310m. [DOI] [PubMed] [Google Scholar]
- 84.Solaro R. J., Rosevear P., Kobayashi T. The unique functions of cardiac troponin I in the control of cardiac muscle contraction and relaxation. Biochemical and Biophysical Research Communications. 2008;369(1):82–87. doi: 10.1016/j.bbrc.2007.12.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1 is about the additional targets' information of each active components from QSYQ. Tables S2–S5 are respective about the results of functional enrichment analysis of salvia miltiorrhiza, Panax notoginseng, Dalbergia odorifera,and Astragalus membranaceus. Figures S1–S4 are respective about the Modules in the PIN of salvia miltiorrhiza, panax notoginseng, dalbergia odorifera, astragalus membranaceus.