Abstract
The generation of potent opioid analgesics that lack the side effects of traditional opioids may be possible by targeting truncated splice variants of the μ-opioid receptor. μ-Opioids act through GPCRs that are generated from the Oprm1 gene, which undergoes extensive alternative splicing. The most abundant set of Oprm1 variants encode classical full-length 7 transmembrane domain (7TM) μ-opioid receptors that mediate the actions of the traditional μ-opioid drugs morphine and methadone. In contrast, 3-iodobenzoyl-6β-naltrexamide (IBNtxA) is a potent analgesic against thermal, inflammatory, and neuropathic pain that acts independently of 7TM μ-opioid receptors but has no activity in mice lacking a set of 6TM truncated μ-opioid receptor splice variants. Unlike traditional opioids, IBNtxA does not depress respiration or result in physical dependence or reward behavior, suggesting it acts through an alternative μ-opioid receptor target. Here we demonstrated that a truncated 6TM splice variant, mMOR-1G, can rescue IBNtxA analgesia in a μ-opioid receptor–deficient mouse that lacks all Oprm1 splice variants, ablating μ-opioid activity in these animals. Intrathecal administration of lentivirus containing the 6TM variant mMOR-1G restored IBNtxA, but not morphine, analgesia in Oprm1-deficient animals. Together, these results confirm that a truncated 6TM GPCR is both necessary and sufficient for IBNtxA analgesia.
Introduction
Traditional opioids are effective against many types of severe pain, but side effects are problematic and opioid efficacy against neuropathic pain is limited. Using biased agonism to develop drugs lacking side effects is one approach to address these limitations (1, 2). An alternative is to pursue novel targets. Recently, a new class of opioid analgesics was described with an unusual pharmacological profile (3–6). IBNtxA (3-iodobenzoyl-6β-naltrexamide), a member of this group, is a potent analgesic against thermal, inflammatory and neuropathic pain, but does not depress respiration or produce physical dependence and shows no reward behavior.
These drugs, exemplified by IBNtxA (4), act through mechanisms distinct from traditional μ drugs. The single-copy μ-opioid receptor Oprm1 creates an array of splice variants through alternative pre-mRNA splicing with patterns conserved from rodents to humans (refs. 7, 8, and Supplemental Figure 1; supplemental material available online with this article; doi:10.1172/JCI81070DS1). The major set of variants are full-length 7 transmembrane domain (7TM) variants associated with exon 1. A second set of variants is characterized by the replacement of the 94 amino acids encoded by exon 1 with 27 amino acids encoded by exon 11 (Supplemental Figure 1). Unlike exon 1, exon 11 lacks a transmembrane domain, resulting in a truncated form of the receptor with only 6TMs. Disruption of exon 1 of the μ-opioid receptor MOR-1 (exon 1 KO) eliminates all full-length 7TM variants and all morphine activity (9). The 6TM variants are still expressed in this mouse, and IBNtxA retains full analgesic activity (4). Conversely, IBNtxA is inactive in an exon 11 KO mouse lacking the truncated 6TM splice variants (4) while morphine retains full analgesic activity (10). Furthermore, IBNtxA labels a binding site in the brain that is lost in exon 11 KO mice and is pharmacologically distinct from the classical opioid receptors.
The 6TM variants were an unanticipated drug target, since they do not conform to the traditional structure of GPCRs. Truncated forms have been reported for over 20 GPCRs, with most acting as dominant-negative mutations (11). In addition to MOR-1, α1A adrenergic, calcitonin, histamine H3, and prostaglandin F2A receptors generate 6TM variants. Unlike the others, the 6TM MOR-1 variants are functionally active. They provide a target for drug development, with the possibility of potent analgesics lacking many of the problems of current medications. We now demonstrate that restoration of a truncated 6TM splice variant through a lentivirus infection in a KO mouse lacking all MOR-1 splice variants can rescue IBNtxA analgesia, confirming that truncated GPCRs are both necessary and sufficient for IBNtxA analgesia.
Results and Discussion
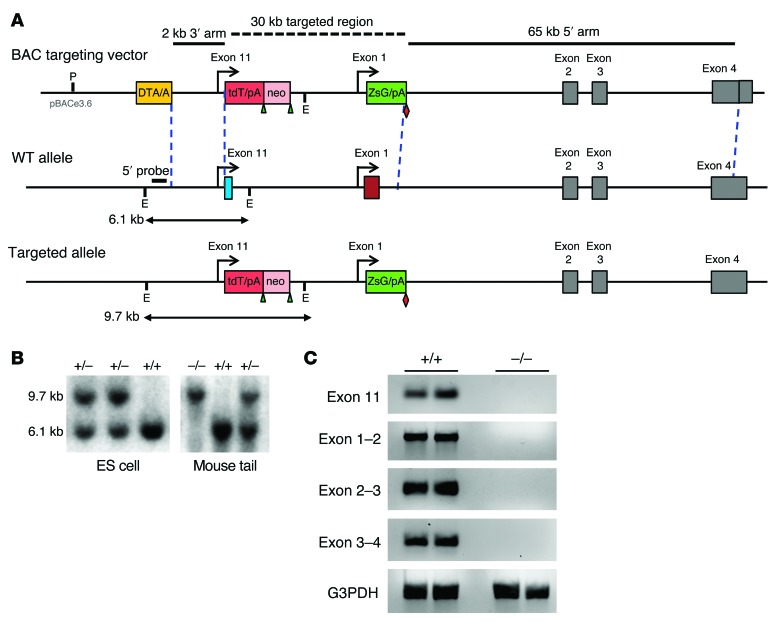
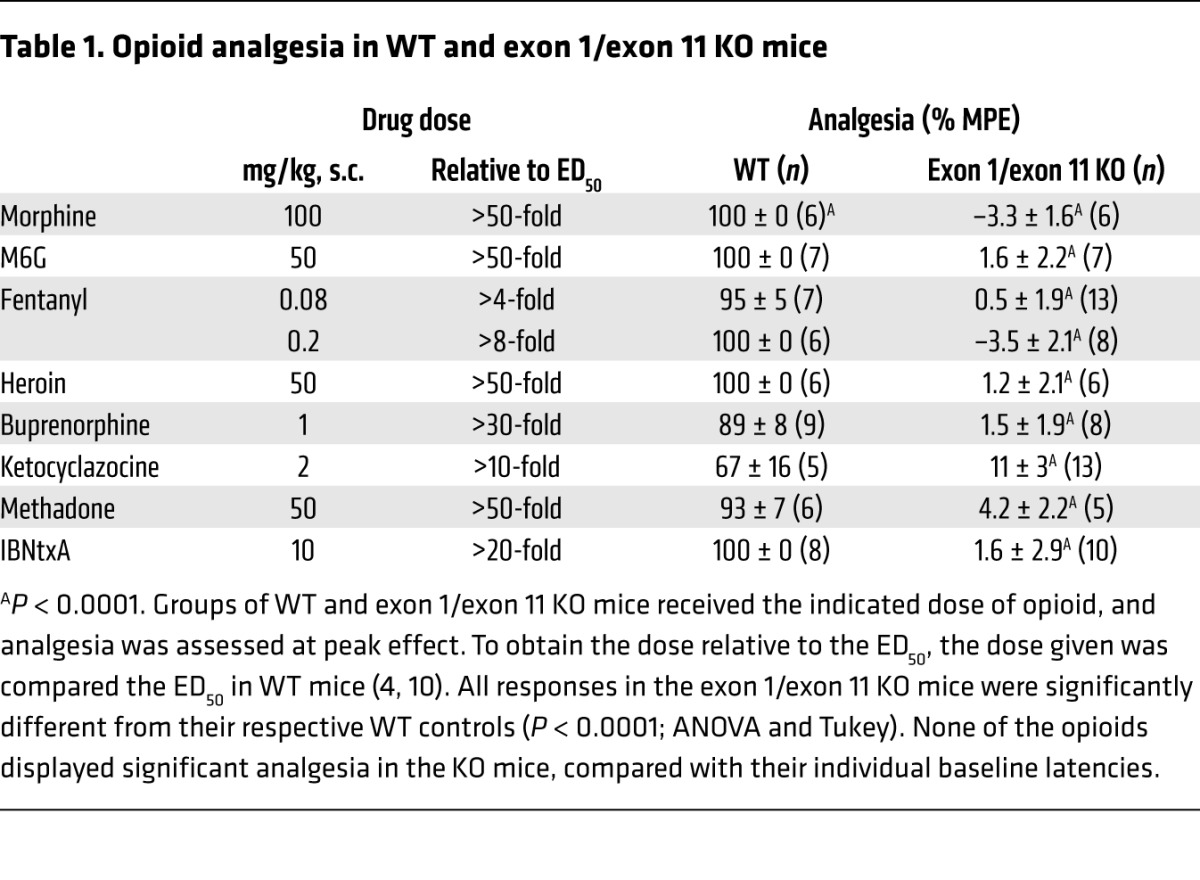
The μ-opioid receptor gene Oprm1 contains 2 independent promoters associated with either exon 1 or exon 11, which is located approximately 30 kb upstream of exon 1 (Supplemental Figure 1 and refs. 7, 8). A number of μ-receptor KO mice eliminate morphine actions by targeting different regions of the gene (9, 12–15), but individual mouse models may not completely eliminate all Oprm1 transcripts. Examples exist in which disruption of exon 1 does not impair the expression of the exon 11 variants (9) and in which elimination of exon 11 does not impact the expression of exon 1 variants (4, 10). We therefore generated a complete MOR-1 KO mouse (exon 1/exon 11 KO) lacking all Oprm1 splice variants by targeting both exon 1 and exon 11 coding and adjacent intron regions (Figure 1, A and B), providing a valuable model to assess the functional role of individual splice variants. Homozygous mice were produced through heterozygous mating, yielding 27.6% WT, 49.4% heterozygous, and 23.0% homozygous KO mice (Figure 1B, n = 87), consistent with the lack of embryonic or postnatal lethality. Homozygous mice were healthy and fertile, with no obvious morphological abnormalities. Reverse transcriptase PCR (RT-PCR) confirmed the absence of both exon 1– and exon 11–containing transcripts in the homozygous mice (Figure 1C). No μ opioids tested, including IBNtxA, were active in the exon 1/exon 11 KO mice, even at high doses (P < 0.001; Table 1). None of the mice showed other typical opioid behaviors, such as hyperlocomotion or Straub tail.
Figure 1. Gene-targeting exons 1 and 11 in the Oprm1 gene.
(A) Schematic of the targeting strategy. The coding and its adjacent intron regions of exons 1 and 11 were replaced with the tdTomato/BGHpolyA (tdT/pA)/PGK-Neo (neo) and ZsGree/SVpolyA (ZsG/pA) cassettes, respectively. The expected EcoRV-digested fragment lengths for WT and targeted alleles are indicated by arrows. The 5′ probe is indicated by a short line. LoxP and Flp sites are shown by triangles and diamonds, respectively. P, PI-SecI; E, EcoRV. (B) Southern blot analysis with the 5′ probe. (C) RT-PCR using RNAs from brain and appropriate primers. Each line represents data from 1 mouse. +/+, WT; +/–, heterozygous; –/–, homozygous.
Table 1. Opioid analgesia in WT and exon 1/exon 11 KO mice.
Loss of IBNtxA actions in exon 11 KO mice implicated 6TM variants in its actions. To determine if a truncated 6TM exon 11 MOR-1 variant could restore IBNtxA analgesia in the exon 1/exon 11 KO mice, we generated a lentivirus expressing the 6TM variant mMOR-1G, the predominant 6TM variant in the spinal cord (16), along with a coexpressed EGFP marker (Supplemental Figure 2). A lentivirus expressing the EGFP marker but lacking the mMOR-1G variant served as a control. Since IBNtxA analgesia is mediated predominantly at the spinal cord, the virus was administered intrathecally on days 1, 3, and 5 (Supplemental Figure 3A).
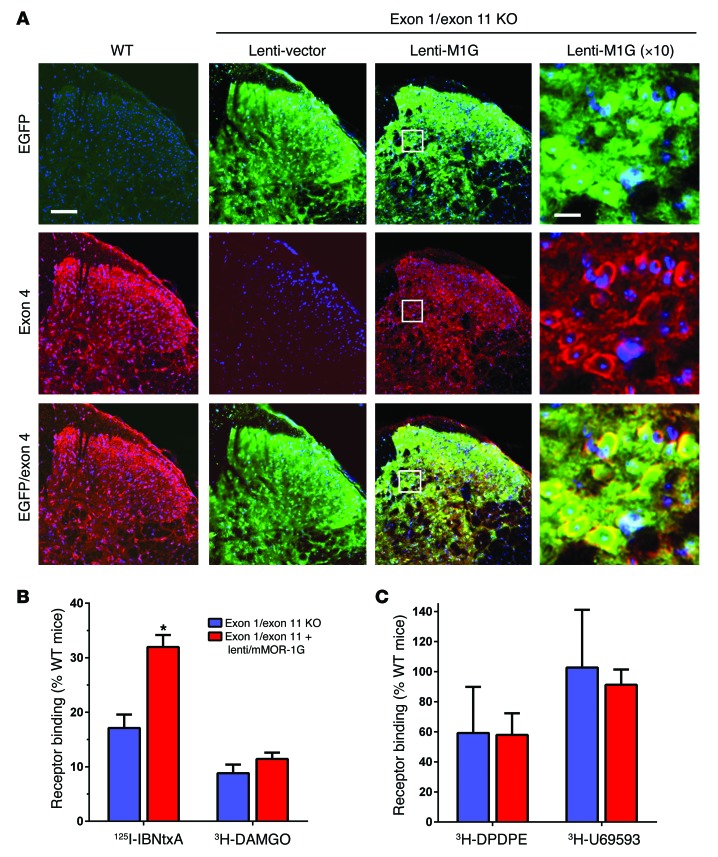
Viral expression was seen as early as 1 week; it gradually increased over 4 weeks and then remained stable for at least 14 weeks (Supplemental Figure 3B). EGFP labeling was highest in the lumbar/sacral levels, near the site of injection; was colocalized with the neuronal nuclear marker NeuN; and was prominent in the dorsal horns, a region important in pain modulation (Supplemental Figure 3C). WT mice that did not receive virus showed no EGFP staining but showed exon 4 antibody labeling in the superficial laminae of the dorsal horn, reflecting the endogenous expression of a number of exon 4–containing variants (Figure 2A, first column, and Supplemental Figure 1) Administration of the control lentivirus expressing only EGFP (i.e., no mMOR-1G) to exon 1/exon 11 KO mice revealed robust EGFP staining, but no exon 4 staining (Figure 2A, second column), confirming the absence of those variants. Both antibodies revealed staining following the lentivirus expressing both EGFP and mMOR-1G, which indicated the coexpression of both proteins in lentiviral-transduced cells (Figure 2A, third column). Higher power showed EGFP labeling throughout the cell, whereas exon 4 staining was localized to the surface (Figure 2A, fourth column), suggesting cell surface expression of the mMOR-1G.
Figure 2. Lentivirus expression in the spinal cord following intrathecal administration.
(A) Distribution of lentiviral-expressed mMOR-1G (lenti–mMOR-1G) and EGFP. An exon 4 antibody was used for staining mMOR-1G. Nuclei were stained with DAPI. Scale bars: 100 μm in first three columns and 10 μm in last column. (B and C) Receptor binding in the spinal cord of exon 1/exon 11 KO mice without and following lenti–mMOR-1G (percent of WT; mean ± SEM from 3–4 pooled samples. Each pool contains the spinal cord from 2–3 mice). Significant differences between WT, exon 1/exon 11 KO, and lenti–mMOR-1G groups were seen with ANOVA for 125I-IBNtxA and 3H-DAMGO (*P < 0.001) (A) but not 3H-DPDPE and 3H-U69,593 (B). For 125I-IBNtxA, the exon 1/exon 11 KO and the lenti–mMOR-1G groups differed from WT (P < 0.001) and from each other (P < 0.05; Tukey). For 3H-DAMGO, the exon 1/exon 11 KO and the lenti–mMOR-1G groups differed from WT (P < 0.001) but not from each other (P > 0.05).
Lentivirus infection restored 125I-IBNtxA binding to exon 11 targets. In WT mice, 125I‑IBNtxA labels traditional μ binding sites, as well as the exon 11 target. This contrasts with 3H‑DAMGO ([d-Ala2,MePhe4,Gly(ol)5]enkephalin), which only labels traditional μ sites. In the spinal cord of the exon 1/exon 11 KO mice, 125I‑IBNtxA binding was decreased by more than 80% and 3H‑DAMGO by more than 90%, compared with WT controls (Figure 2B). These decreases reflect the loss of traditional full-length 7TM μ receptors as well as exon 11–associated targets; the residual 125I-IBNtxA binding likely reflects cross-labeling of δ and/or κ sites. Administration of the lentivirus with the mMOR-1G insert significantly increased 125I‑IBNtxA binding but not 3H-DAMGO binding (Figure 2B). Similarly, neither the δ ligand 3H-DPDPE nor the κ1 ligand 3H-U69,593 showed an increase in binding following virus administration (Figure 2C). The absence of any increase in traditional μ, δ, or κ1 binding (Figure 2B) indicated that expression of mMOR-1G restored 125I-IBNtxA binding to an exon 11–associated target.
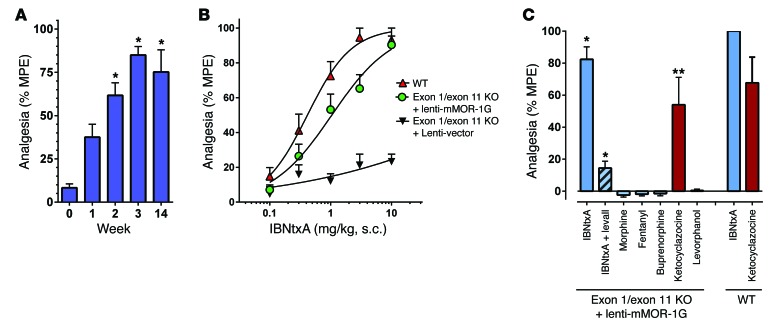
The expressed mMOR-1G was functionally active. IBNtxA analgesia returned as early as 1 week following lentivirus infection, gradually increasing until it reached a peak response after 3 weeks (Figure 3A) and remained constant for at least 14 weeks, a time course similar to that of EGFP expression (Supplemental Figure 3B). Mice infected with a control lentivirus that did not contain mMOR‑1G failed to display an analgesic response (Figure 3B). IBNtxA was fully efficacious in the mice infected with the lentivirus containing mMOR-1G with an ED50 (1.1 mg/kg, s.c.) similar to that of WT mice (0.42 mg/kg, s.c.; Figure 3B).
Figure 3. Lentivirus rescue of IBNtxA analgesia.
(A) Opioid analgesia. Analgesia was determined in groups of mice (n = 6–13) at the stated time. (*P < 0.0001 compared with week 0; ANOVA followed by Tukey). (B) IBNtxA cumulative dose–response curves were carried out in exon 1/exon 11 KO mice with lentivirus vector alone (n = 4), lenti–mMOR-1G (ED50 1.1 mg/kg [95% CI, 0.72–1.53], n = 18) and WT mice (ED50 0.42 mg/kg [95% CI, 0.29–0.58], n = 16). (C) Single doses of IBNtxA (2.5 mg/kg, s.c. n = 5), morphine (10 mg/kg, s.c. n = 7), fentanyl (0.08 mg/kg, s.c. n = 7), buprenorphine (1 mg/kg, s.c. n = 7), ketocyclazocine (2 mg/kg, s.c. n = 7), or levorphanol (0.8 mg/kg, s.c. n = 7) were administered to groups of either WT or exon 1/exon 11 KO mice infected with lenti–mMOR-1G. The mice were assessed for analgesia. Another group of mice received IBNtxA with levallorphan (2.5 mg/kg, s.c. n = 7). ANOVA shows that the IBNtxA group and the ketocyclazocine group were significantly different (*P < 0.001 and **P < 0.006 by ANOVA, respectively). The IBNtxA WT and lenti–mMOR-1G animals were not significantly different from each other but were different from both the exon 1/exon 11 KO alone and the lenti–mMOR‑1G/levallorphan groups (Tukey). Ketocyclazocine in WT and lenti–mMOR-1G were not significantly different from each other but were different from exon 1/exon 11 alone (Tukey).
IBNtxA analgesia was readily reversed by levallorphan, confirming an opioid mechanism (Figure 3C). Ketocyclazocine is a nonselective opioid that produces an exon 11–dependent analgesia (4) that was lost in the exon 1/exon 11 KO (Table 1). As with IBNtxA, ketocyclazocine analgesia returned in the lentivirus-infected mice, with a response similar to that seen in WT mice (Figure 3C). Although expression of mMOR-1G rescued IBNtxA and ketocyclazocine analgesia, it failed to rescue high doses of the other opiates. Morphine analgesia is independent of exon 11 variants (4), so this result was anticipated. Although the diminished activity of the other drugs in exon 11 KO mice in earlier studies implicated exon 11 variants in their actions, the inability to rescue their response in the exon 1/exon 11 KO indicated that a 6TM variant was necessary but not sufficient. Other factors may be involved.
The current studies describe a new exon 1/exon 11 KO mouse with disruptions of both exon 1 and exon 11, yielding a full μ receptor–null mouse lacking all Oprm1 splice variants (Supplemental Figure 1) that is insensitive to μ opioids. The absence of all the variants provides an excellent model to assess the functional significance of individual variants. The 6TM variant mMOR‑1G restored both IBNtxA and ketocyclazocine analgesia in the exon 1/exon 11 KO mouse, indicating that a 6TM variant is both necessary and sufficient for their actions. The failure of mMOR-1G to restore morphine analgesia is consistent with a mechanism independent of exon 11 variants. The lack of rescue of the others has several potential explanations. Since the mouse generates 5 different 6TM variants, one of the other 6TM variants may be needed. The spinal injection site may be important, since the other drugs may act supraspinally. Finally, some drugs may require expression of both 6TM and 7TM MOR-1 variants for activity. For example, buprenorphine analgesia is lost in the exon 11 KO mouse (4) and is also markedly reduced in an exon 1 KO mouse lacking only 7TM variants, implying a need for both sets of variants.
The 6TM MOR-1 splice variant mMOR-1G is both necessary and sufficient for IBNtxA analgesia. With its broad analgesic activity in thermal, inflammatory, and neuropathic pain models (5), as well as its lack of respiratory depression, physical dependence, and reward, IBNtxA may represent a new class of analgesic distinct from those associated with traditional opiates and 6TM variants may prove valuable targets for future analgesic drug development. These results also raise the broader question of the significance of truncated GPCRs (8, 11, 17).
Methods
Detailed information is described in the Supplemental Methods.
Exon 1/exon 11 KO mice.
KO mice with disruptions of exons 1 and 11 were generated using a targeting vector in W4 ES cells (a gift from Alexandra Joyner, Memorial Sloan-Kettering Cancer Center, New York, NY, USA; Figure 1 and Supplemental Methods). Heterozygotes were crossbred to obtain WT and homozygous mice.
Lentivirus.
Lentivirus (a gift from Didler Trono, Ecole Polytechnique Federale de Lausanne, Lausanne, Switzerland) was generated using the constructs with and without the mMOR-1G sequence in HEK293T cells (Supplemental Figure 3). The 2 μl of concentrated lentiviral particles expressing mMOR-1G or vector alone without insertion (1.5 × 109 transducing units/ml) were administrated intrathecally on days 1, 3, and 5, as previously described (18, 19).
Statistics.
Analysis of the EGFP qPCR and analgesia utilized one-way ANOVA with post hoc Tukey’s multiple comparisons test. Opioid binding was assessed using 2-tailed Student’s t test. Data are represented as the mean ± SEM.
Study approval.
Animal studies were approved by the Institutional Animal Care and Use Committee (IACUC) of Memorial Sloan-Kettering Cancer Center.
Supplementary Material
Acknowledgments
This work was supported by the NIH with grants from NIDA (DA013997 and DA02944) to Y.-X. Pan and (DA06241 and DA07242) to G.W. Pasternak, a Harrington Discovery Institute Scholar-Innovator Grant to G.W. Pasternak and a core grant from the NCI to MSKCC (CA08748). We thank Didler Trono for providing the lentiviral vectors, Chingwen Yang of Rockefeller University for help with ES cells, and Ke Xu, Margaret Leversha, and Willie Mark from Memorial Sloan-Kettering Cancer Center for help with imaging, ES clone karyotyping, and generating the targeted mice, respectively.
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Reference information:J Clin Invest. 2015;125(7):2626–2630. doi:10.1172/JCI81070.
See the related Commentary beginning on page 2558.
References
- 1.Dewire SM, et al. A G protein-biased ligand at the μ-opioid receptor is potently analgesic with reduced gastrointestinal and respiratory dysfunction compared with morphine. J Pharmacol Exp Ther. 2013;344(3):708–717. doi: 10.1124/jpet.112.201616. [DOI] [PubMed] [Google Scholar]
- 2.Violin JD, Lefkowitz RJ. β-Arrestin-biased ligands at seven-transmembrane receptors. Trends Pharmacol Sci. 2007;28(8):416–422. doi: 10.1016/j.tips.2007.06.006. [DOI] [PubMed] [Google Scholar]
- 3.Grinnell SG, et al. Pharmacologic characterization in the rat of a potent analgesic lacking respiratory depression, IBNtxA. J Pharmacol Exp Ther. 2014;350(3):710–718. doi: 10.1124/jpet.114.213199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Majumdar S, et al. Truncated G protein-coupled mu opioid receptor MOR-1 splice variants are targets for highly potent opioid analgesics lacking side effects. Proc Natl Acad Sci U S A. 2011;108(49):19776–19783. doi: 10.1073/pnas.1115231108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wieskopf JS, et al. Broad-spectrum analgesic efficacy of IBNtxA is mediated by exon 11-associated splice variants of the mu-opioid receptor gene. Pain. 2014;155(10):2063–2070. doi: 10.1016/j.pain.2014.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Majumdar S, et al. Synthesis and evaluation of aryl-naloxamide opiate analgesics targeting truncated exon 11-associated mu opioid receptor (MOR-1) splice variants. J Med Chem. 2012;55(14):6352–6362. doi: 10.1021/jm300305c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Pan Y-X, Pasternak GW. Molecular biology of mu opioid receptors. In: Pasternak GW, ed. The Opiate Receptors. 2nd ed. New York, New York, USA: Springer; 2011:121–160. [Google Scholar]
- 8.Pasternak GW, Pan Y-X. Mu opioids and their receptors: Evolution of a concept. Pharmacol Rev. 2013;65(4):1257–1317. doi: 10.1124/pr.112.007138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schuller AG, et al. Retention of heroin and morphine-6 β-glucuronide analgesia in a new line of mice lacking exon 1 of MOR-1. Nat Neurosci. 1999;2(2):151–156. doi: 10.1038/5706. [DOI] [PubMed] [Google Scholar]
- 10.Pan YX, Xu J, Xu M, Rossi GC, Matulonis JE, Pasternak GW. Involvement of exon 11-associated variants of the mu opioid receptor MOR-1 in heroin, but not morphine, actions. Proc Natl Acad Sci U S A. 2009;106(12):4917–4922. doi: 10.1073/pnas.0811586106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wise H. The roles played by highly truncated splice variants of G protein-coupled receptors. J Mol Signal. 2012;7(1):13. doi: 10.1186/1750-2187-7-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tian M, et al. Altered hematopoiesis, behavior, and sexual function in μ opioid receptor-deficient mice. J Exp Med. 1997;185(8):1517–1522. doi: 10.1084/jem.185.8.1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sora I, et al. Opiate receptor knockout mice define μ receptor roles in endogenous nociceptive responses and morphine-induced analgesia. Proc Natl Acad Sci U S A. 1997;94(4):1544–1549. doi: 10.1073/pnas.94.4.1544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Loh HH, Liu HC, Cavalli A, Yang WL, Chen YF, Wei LN. μ Opioid receptor knockout in mice: effects on ligand-induced analgesia and morphine lethality. Mol Brain Res. 1998;54(2):321–326. doi: 10.1016/S0169-328X(97)00353-7. [DOI] [PubMed] [Google Scholar]
- 15.Matthes HW, et al. Loss of morphine-induced analgesia, reward effect and withdrawal symptoms in mice lacking the μ-opioid-receptor gene. Nature. 1996;383(6603):819–823. doi: 10.1038/383819a0. [DOI] [PubMed] [Google Scholar]
- 16.Xu J, et al. Stabilization of morphine tolerance with long-term dosing: Association with selective upregulation of mu-opioid receptor splice variant mRNAs. Proc Natl Acad Sci U S A. 2014;112(1):279–284. doi: 10.1073/pnas.1419183112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Xu J, et al. Stabilization of the mu opioid receptor by truncated single transmembrane splice variants through a chaperone-like action. J Biol Chem. 2013;288(29):21211–21227. doi: 10.1074/jbc.M113.458687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hylden JL, Wilcox GL. Intrathecal morphine in mice: a new technique. Eur J Pharmacol. 1980;67(2–3):313–316. doi: 10.1016/0014-2999(80)90515-4. [DOI] [PubMed] [Google Scholar]
- 19.Rossi GC, Pan Y-X, Brown GP, Pasternak GW. Antisense mapping the MOR-1 opioid receptor: Evidence for alternative splicing and a novel morphine-6β-glucuronide receptor. FEBS Lett. 1995;369(2–3):192–196. doi: 10.1016/0014-5793(95)00757-z. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.