Abstract
Objective
To follow-up on earlier studies of the leukaemogenicity of occupational ionising radiation exposure.
Methods
We conducted a nested case-control analysis of leukaemia mortality in a pooled cohort of US nuclear workers followed through 2005. Each case was matched to four controls on attained age. Exposures were estimated from available records. General relative risk models were used to estimate the excess relative risk (ERR) of leukaemia, excluding chronic lymphocytic (CLL), acute myeloid leukaemia, chronic myeloid leukaemia and CLL while controlling for potential confounders. Preferred exposure lags and time-windows of risks were calculated using joint maximum likelihood. Dose-response was also examined using linear, linear-quadratic, categorical and restricted cubic spline models.
Results
There were 369 leukaemia deaths in 105 245 US nuclear workers. The adjusted ERR for non-CLL leukaemia was 0.09 (95% CI −0.17 to 0.65) per 100 mGy. Elevated non-CLL risks were observed from exposures occurring 6–14 years prior to attained age of cases (ERR per 100 mGy=1.9; 95% CI <0 to 8.0). Lagged models indicated non-linearity of risk at very low (<10 mGy) and high (>100 mGy) doses, which contributed to the imprecision of results in linear models. Similar risk attenuation was not evident in time-windows-based models.
Conclusions
Risk estimates were in reasonable agreement with previous estimates, with the temporality of non-CLL leukaemia risk as a dominant factor in dose-response analyses. Future research should focus on methods that improve evaluations of the dose-response, particularly in the low-dose range.
INTRODUCTION
Most information on the leukaemogenicity of ionising radiation stems from studies of radiation therapy patients and survivors of the atomic bomb blasts in 1945.1,2 In particular, the Life-Span Study (LSS) of the Japanese atomic bomb survivors forms the foundation for current worker protection standards. The LSS has the advantages of large size (~87 000), all ages at exposure, both sexes, long follow-up period and doses over a range of interest (0–4 Gy with 60% less than 0.1 Gy), although questions remain concerning the translation of risks to workers who are intermittently exposed to low doses delivered at low dose rates. A preferred alternative is the direct estimation of risk in working populations; however, most occupational studies lack sufficient statistical power for examining rare outcomes such as leukaemia. As such, recent studies have estimated leukaemia risk in nuclear workers using pooled, or meta-analyses, that offer improved precision.3–7 In particular, Daniels and Schubauer–Berigan conducted5 a meta-analysis using information from occupational and environmental studies to estimate the excess relative risk (ERR) of leukaemia, excluding the chronic lymphocytic subtype (CLL). The summary estimate from 10 studies (ERR at 100 mGy=0.19; (95% CI 0.07 to 0.32) was in reasonable agreement with the LSS. Nevertheless, a meta-analytic approach relies on the quality of data available in the published literature; therefore, these analyses may be vulnerable to biases in the source information. Moreover, studies have typically relied on models that assume risk is linear or linear-quadratic in dose for a heterogeneous group of diseases comprising non-CLL leukaemia. Few studies have examined departures from linearity in the dose-response or risk differences by leukaemia subtype.
This study extends follow-up of workers previously studied by Schubauer–Berigan et al3,4 and expands the cohort to include workers from the Idaho National Laboratory.8 Previous analyses reported estimates of the ERR of mortality per unit dose for non-CLL leukaemia that reasonably agreed with the LSS4; however, there was evidence of risk attenuation at doses above 100 mGy and temporal patterns in risk that varied with time since exposure.4 The purpose of the current study is to: (1) improve upon previous estimates through additional follow-up and cohort expansion; (2) report on the risk by leukaemia subtype and (3) further evaluate the shape and temporal behaviour of the dose-response.
METHODS
Study cohort
This research complied with the requirements of the Federal Policy for Protection of Human Subjects (10CFR745 or, where applicable, 45CFR46), and was reviewed by the National Institute for Occupational Safety and Health (NIOSH) Human Subjects Review Board to ensure that the rights and welfare of study subjects were protected.
The study population consisted of workers who were employed at the Hanford Site (Hanford) near Richland, Washington; the Savannah River Site (SRS) near Aiken, South Carolina; the Oak Ridge National Laboratory (ORNL) in Oak Ridge, Tennessee; the Los Alamos National Laboratory (LANL), including the Zia Company employees, in Los Alamos, New Mexico; the Idaho National Laboratory (INL) near Idaho Falls, Idaho; and the Portsmouth Naval Shipyard (PNS) in Kittery, Maine. Eligible workers were monitored for radiation exposure and employed at one or more of the primary facilities for at least 30 days. Hanford, LANL and ORNL workers were further restricted to those first employed on or after 1 January 1951 to reduce exposure errors believed associated with early dosimetry practices at Manhattan Engineering District Facilities.4 Vital status ascertainment was extended through 31 December 2005, adding 9–15 years of follow-up to the previous study.
Cases were decedents with leukaemia (ICD-9: 204–208; ICD-10: C91–C95, excluding leukaemic reticuloendotheliosis (C91.4)), identified as the underlying cause of death. Distinctions were made by major subtypes, namely: CLL, acute lymphoid leukaemia (ALL), chronic myeloid leukaemia (CML) and acute myeloid leukaemia (AML). Non-CLL cases comprised all-acute, and subacute leukaemias, and myeloid or monocytic leukaemias, excluding CLL-indeterminate cases that were defined as cases lacking sufficient information on cell type and severity to determine non-CLL status (ie, ICD-9 codes: 204.9, 208.1, 208.9; ICD-10 codes: C91.9, C95.9, C95.1). Similarly, AML-indeterminate cases were defined as acute leukaemias of unspecified cell type (ie, ICD-9 code: 208.0; ICD-10 code: C95.0). Four controls were selected for each case using incidence density sampling.9 Controls were matched to their respective case on attained age and by primary study group, whereby, Group I risk sets were formed from the previously studied group of Hanford, LANL/Zia, ORNL, PNS and SRS workers,3,4 and Group II risk sets contained the INL workers.8 This group matching was done to maximise utility of information collected on cases and controls selected for the previous study.
Exposure
The primary exposure was low linear energy transfer (low-LET) ionising radiation from all occupational sources, including work-related medical x-ray examinations (WRX) conducted during routine physical examinations by the employer.10,11 Workplace exposures to neutrons and internally deposited plutonium were also considered as potential cofactors in dose-response analyses. External ionising radiation exposure measurements for each study participant were abstracted from site dosimetry databases and dose registries maintained by the US Department of Energy (DOE) and US Nuclear Regulatory Commission (NRC). All doses were quantified in terms of absorbed dose to haematopoietic tissue using methods described elsewhere.4,11–13 There were five exposure variables describing ionising radiation dose (ie, gamma, tritium, WRX, neutron and plutonium). Low-LET exposure was calculated by summing doses from whole-body gamma, tritium and WRX sources.
Benzene exposures were assessed using a job exposure matrix to assign task-specific benzene ‘scores’ that were based on combinations of qualitative ratings of exposure intensity and frequency over time. Potentially exposed workers were identified using available employment information on job assignment (eg, job title, division and department) and work location in each study facility (eg, area, building and room). Tasks were identified and characterised using site-monitoring records and process information. Task-based scores were calculated for each study subject and period of employment, which were then accumulated over the observation period.
Socioeconomic status
The methods used to assign socioeconomic status (SES) are described elsewhere.4,8 In general, the job title at first hire for each worker was related to an occupational code listed in the 1980 US Bureau of Census. The first job title was chosen because it is thought that initial employment provides a better marker for some lifestyle factors (eg, smoking) that are typically established in adolescence and early adulthood.4,8,14 Each of these codes was then assigned to one of six SES categories, namely (from high to low SES): professional, intermediate, skilled non-manual, skilled manual, partly skilled and unskilled.
STATISTICAL METHODS
All statistical analyses were conducted using SAS software.15 Primary modelling used general relative risk models that represent a linear trend in radiation dose.16 Relative risk models followed the general form
where α is the radiation risk coefficient; βi is the coefficient for the ith covariate of 1, 2 …n covariates indexed by multiplicative factors, Xi; and γj is the radiation weighting factor for the jth dose variable of three linear dose covariates, D. For all models, three radiation sources were considered: (1) low-LET (D1) irradiation; (2) plutonium deposition (D2); and (3) neutron radiation (D3). Setting the low-LET radiation weighting factor to unity (γ1=1) allowed solutions to radiation weights for plutonium and neutron doses that can be compared with standard values. Profile likelihood-based (PL-based) two-sided 95% CIs were calculated for model parameter estimates.
Effect modification was evaluated by likelihood ratio test (LRT) at the 0.05 critical level of the cross-product term of the potential effect modifier and radiation dose. Factors evaluated were: sex, race (White non-Hispanic/all others), birth cohort (tertiles), period of first hire (tertiles), facility of longest employment duration and study group (Group I vs Group II). Potential confounders were incorporated into models when causing >15% relative change in the overall dose parameter estimate. To minimise the potential bias from selection of cutpoints for categories of birth cohort and hire date, continuous variables were constructed from terms calculated by restricted cubic spline (RCS) models, with three knots at the 10th, 50th and 90th percentile of birth date and hire date, respectively.17 Because birth date and hire date were correlated (Pearson coefficient=0.59, p<0.001), models were restricted to include no more than one of these variables at a time. Final selection between birth date and hire date was based on a comparison of observed effect and the statistical significance of the covariates.
Baseline models were constructed for non-CLL leukaemia and three of the four major subtypes: AML, CML and CLL. There were too few cases to adequately examine ALL risks separately. The methods proposed by Richardson et al18 were used to simultaneously estimate the preferred exposure lag by joint maximum likelihood. A preferred time-window of exposure was similarly estimated.
The linearity of the dose-response was examined by: (1) evaluating the fit of a quadratic term added to the model; (2) examining the risk by dose categories; (3) fitting RCS models and (4) fitting piecewise linear spline (PLS) models. Lagged categorical models used categories of 0–<1.0, 1–<10, 10–<50, 50–<100, and ≥100 mGy, with the lowest category as referent. Time-windows-based models used dose categories with cut-points defined by quintiles of the dose distribution of the cases for each outcome. Lagged RCS models set four knots at the 5th, 35th, 65th and 95th percentiles of the distribution of low-LET dose, while windows-based RCS models used three knots defined by the 10th, 50th and 90th percentiles of dose.17 PLS models were restricted to non-CLL (lagged and windows-based); both two- and three-piece solutions were evaluated. Model fits were judged by Akaike Information Criterion (AIC).19 To test improvement in fit over a linear model, LRTs were conducted comparing the adjusted linear-quadratic, RCS and PLS models with the adjusted linear model.
The effects of high-LET radiations were examined in sensitivity analyses using model solutions to radiation weights and by varying weighting schemes for biological effectiveness. Three sets of weights for low-LET, neutron and plutonium were considered: 1, 10 and 20, which is consistent with the protection quantity ‘equivalent dose’20; 1, 11 and 3.6, which are median radiation effectiveness factors suggested by Kocher et al21; and 1, 10 and 1, based on the upper bound suggested by the United States Environmental Protection Agency (US EPA) in 1999 for α emitters deposited on bone surfaces or in bone.22 The current EPA-recommended value of 2 (95% CI 1 to 3) for incorporated α-emitters is within the range analysed.23
RESULTS
Descriptive and univariate
There were 94 517 workers from Hanford, LANL, ORNL, PNS and SRS (ie, Group I) and 63 560 INL workers (ie, Group II). Applying the inclusion criteria, and then combining the two cohorts, resulted in a base cohort of 105 245. There were 30 266 deaths (28%), which included 8721 cancer deaths and 369 leukaemia cases. Among leukaemias, 291 cases were identifiable to one of four subtypes. Non-CLL leukaemia (n=264) comprised AML (n=150), CML (n=52), ALL (n=18), unspecified acute leukaemias (n=36) and myeloid leukaemias (n=2), and other non-CLL subtypes (n=6). There were 74 CLL deaths. (See Web Appendix, tables 1–3 for additional information on the study cohort and case distribution.)
Dose distributions were right-skewed; the average, median and maximum values of low-LET dose lagged 2 years were 26.5 and 7.2 mGy, and 634 mGy, respectively. There were 293 (15.9%) cases and controls exposed to tritium, 237 (12.8%) with neutron dose, 130 (7.0%) with plutonium dose and 360 (19.5%) who were assigned benzene scores (see Web Appendix, table 4 for additional information on exposures).
Univariable analysis of potential confounders revealed similar results across non-CLL leukaemia and subtypes. The most pronounced effect was observed across categories of birth year and year of first hire, where less risk was consistently observed in persons born earlier and/or hired earlier. Decreased risk was observed in women and non-Hispanic Whites compared with men and other races/ethnicities, respectively; however, among these factors, only CML in other races/ethnicities compared with non-Hispanic Whites was statistically significant (HR=4.1; 95% CI 1.3 to 12.7). More information on univariate analyses is available in the Web Appendix, table 5.
Multivariable analyses
Of variables tested for interaction with low-LET radiation, only race approached statistical significance (eg, non-CLL: Χ2=3.07, df=1, p=0.080). As observed previously,4 restricting analyses to Group I showed evidence of risk heterogeneity across facilities (Χ2=18.07, df=4, p=0.003). Positive dose-responses were observed in PNS workers (ERR per 100 mGy=3.7; 95% CI 0.76 to 20.0) and SRS (ERR per 100 mGy=2.5; 95% CI 0.44 to 8.3), which influenced the overall estimate for the group (ERR per 100 mGy=0.12; 95% CI −0.16 to 0.66). The estimate of non-CLL leukaemia risk for INL was also positive (ERR per 100 mGy=0.03; 95% CI −0.23 to 0.81), although markedly less than Group I. There was no evidence of significant risk differences between Group I and Group II (Χ2=0.086, df=1, p=0.769).
Including sex in the model reduced risk estimates for non-CLL leukaemia by over 27%. Adjusting for race alone increased the estimate by nearly 40%. Including hire year by tertiles resulted in a 40% increase in the estimate compared with just under a 15% increase when controlling for hire year using RCS. Adjustment for benzene, SES or birth cohort resulted in less than 15% relative change. Similar results were obtained for CML, where final models were adjusted for race, sex and hire year. The model of AML risk was not influenced by sex or race, but was increased by 28% when including RCS terms for birth cohort, and 17% when including hire date. Given the strong association between birth cohort and AML compared with hire date and AML, subsequent models were controlled for birth date using spline terms. CLL risk estimates were influenced by sex (fourfold decrease), race (twofold increase), and birth cohort (10-fold increase using RCS).
Solving for radiation weights resulted in relatively large values for both high-LET components and a slight reduction in the risk per unit dose (table 1). Fixing weights in sensitivity analyses moderately increased ERR estimates of non-CLL leukaemia per unit dose. These patterns were consistent in lagged models; however, weight solutions were substantially reduced in windows-based models. For example, non-CLL risk from exposures 6–14 years before the attained age of the case had solutions for plutonium and neutron weights of 0 (fixed bound) and 16, respectively, and the relative change in the ERR estimate was about 3%. Given evidence of only moderate high-LET effects and small percentages of exposed workers, we excluded plutonium and neutron doses from final models.
Table 1.
Sensitivity analysis of non-chronic lymphocytic leukaemia risk from high and low linear energy transfer radiations combined (lagged 2 years, fixed), adjusted for sex, race and hire year
| Radiation weights*
|
Linear ERR per 100 mGy (95% CI) | Relative change in ERR (%) | |
|---|---|---|---|
| Plutonium | Neutron | ||
| 0 | 0 | 0.09 (−0.14 to 0.51) | NA |
| 171 (solved) | 406 (solved) | 0.07 (<0 to 0.49) | −22 |
| 20 | 10 | 0.15 (−0.06 to 0.57) | 67 |
| 1 | 10 | 0.11 (−0.13 to 0.54) | 22 |
| 3.6 | 11 | 0.13 (−0.12 to 0.56) | 33 |
Radiation weights used to construct weighted absorbed dose to the red bone marrow.
ERR, excess relative risk; NA, not applicable.
In general, positive, but non-statistically significant estimates of excess radiation-associated risk were observed in all lagged models except for CLL (table 2). There was no evidence of CLL radiogenicity in any model tested. The overall estimate of non-CLL risk was 0.09 (95% CI −0.17 to 0.65) per 100 mGy (lagged 2 year), excluding CLL-indeterminate cases, and adjusting for sex, race and hire year. Including CLL-indeterminate cases in the model reduced the observed non-CLL risk (ERR per 100 mGy=0.03; 95% CI −1.5 to 0.38). Among leukaemia subtypes, the highest risk in lagged models was observed in CML (ERR per 100 mGy=0.29; 95% CI <0 to 1.8; 6.3 year lag). Temporal risk patterns were clearly evident in time-windows-based models, and the most pronounced effect was observed for AML in the 6–14-years window (ERR per 100 mGy=7.0; 95% CI 0.079 to 32.0), although CIs were wide. AML cases in the 6–14-years window had mean and maximum cumulative doses of 3.8 and 71.2 mSv, respectively, while control mean and maximum doses were 2.1 and 137.4 mSv, respectively. Results from alternate analyses of exposure lags and time since exposure using methods comparable with the previous study are reported in tables 6–8 of the Web Appendix.
Table 2.
Linear dose*-response modelling results by preferred lag and exposure window
| Outcome | No. cases | Adjusted for | Best fit exposure lag ERR per 100 mGy (95% CI)
|
Best fit exposure window ERR per 100 mGy (95% CI)
|
||
|---|---|---|---|---|---|---|
| Lag (years) | Lag model | Window (years) | Window model | |||
| Non-CLL† | 264 | Sex, race, hire year | 2 | 0.09 (−0.17 to 0.65) | 6 to 14 | 1.9 (<0 to 8.0) |
| AML | 150 | Birth cohort | 1.1 | 0.22 (−0.19 to 1.2) | 6 to 14 | 7.0 (0.079 to 32.0) |
| CML | 52 | Sex, race, hire year | 6.3 | 0.29 (<0 to 2.6) | 6.3 to 14 | 3.8 (−1.2 to 26.0) |
| CLL | 74 | Sex, race, birth cohort | 24 | −0.032 (−0.16 to 0.74) | 11 to 14 | −2.8 (NC to NC) |
‘Dose’ was defined as absorbed dose to red bone marrow from low linear energy transfer radiation exposure.
Excluding leukaemias of indeterminate subtype.
AML, acute myeloid leukaemia; CML, chronic myeloid leukaemia; CLL, chronic lymphocytic leukaemia; ERR, excess relative risk; NC, not calculable.
Dose-response shape analyses
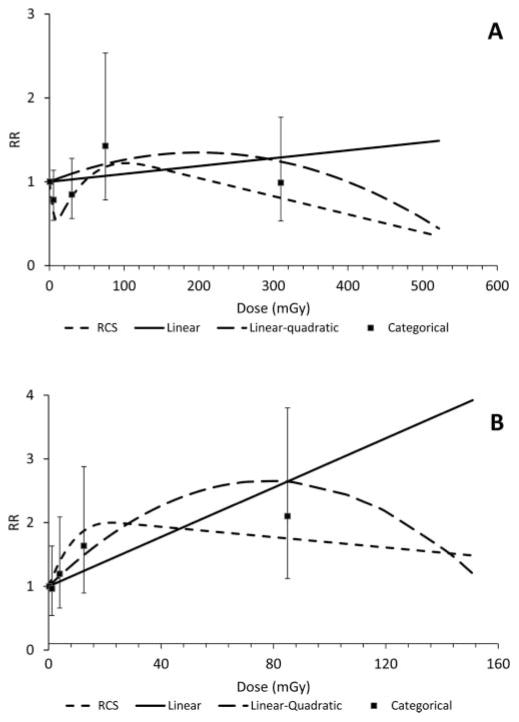
The results of fitting linear, linear-quadratic, categorical and RCS dose-response models of low-LET dose and non-CLL leukaemia are shown in figure 1. Figures for AML and CML dose-response modelling are available as figures 1 and 2 in the Web Appendix. Panel A shows the results of lagged models while Panel B shows results using the best-fit exposure window. Model convergence was not achieved for some time-windows-based models (ie, AML and CML linear-quadratic forms and CML RCS, see Web Appendix). The best fitting non-CLL and AML-lagged models were obtained from RCS (dashed lines) based on AIC and the LRT (table 3). The shape of the dose-response for AML was similar, whereby the lagged RCS and categorical plots show risk attenuation at cumulative doses below 10 mGy, followed by a sharp increase to 100 mGy before decreasing again. CML dose-response under a 5-year lag was best-fit to a linear model; however, inspection of RCS and categorical results suggested some attenuation at low and high doses, albeit diminished relative to AML. By contrast, there is little evidence of a departure from linearity in low-dose regions of windows-based analyses of all outcomes, although non-linear models suggested some attenuation at higher doses. Alternate analyses examining the non-linearity of the dose-response at high doses are reported in the table 9 of the Web Appendix.
Figure 1.
Mortality from leukaemia, excluding the chronic lymphocytic subtype, and cumulative absorbed dose to bone marrow. (A) Two-year lag; (B) 6–14-year window. Solid line: linear; long dashed line: linear-quadratic; short dashed line: restricted cubic spline (RCS); filled squares: category midpoints (with 95% CI shown by error bars).
Table 3.
Model fit characteristics by outcome, exposure (preferred lag or window), and model type
| Outcome Adjust for | Non-CLL (n=264) race, sex and hire date
|
AML (n=150) birth date
|
CML (n=52) race, sex and hire date
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Model Fit statistic* | Lag (2 years)
|
Windows (6–14 years)
|
Lag (1.1 years)
|
Windows (6–14 years)
|
Lag (6.3 years)
|
Windows (6.3–14 years)
|
||||||
| AIC | LRT p value | AIC | LRT p value | AIC | LRT p value | AIC | LRT p value | AIC | LRT p value | AIC | LRT p value | |
| Linear | 850.9 | NA | 847.6 | NA | 480.9 | NA | 473.7 | NA | 168.0 | NA | 166.2 | NA |
| Linear-quadratic | 852.1 | 0.382 | 846.9 | 0.104 | 481.7 | 0.281 | NC | NC | 170.0 | 0.962 | NC | NC |
| RCS | 844.7 | 0.006 | 845.6 | 0.046 | 479.4 | 0.065 | 473.5 | 0.136 | 170.6 | 0.488 | NC | NC |
| PLS† | 846.2 | 0.013 | 847.6 | 0.135 | NM | NM | NM | NM | ||||
| Categorical | 852.2 | NA | 849.8 | NA | 486.1 | NA | 477.7 | NA | 170.9 | NA | 169.6 | NA |
LRT degrees of freedom (df): Linear-quadratic all=1; RCS lag=2, window=1; and PLS lag=4, window=2.
Best fitting PLS models were obtained using two knots (8.3 and 46.9 mGy) in the 2-year-lagged model and one knot (20.1 mGy) in the windows-based model.
AIC, Akaike Information Criterion; AML, acute myeloid leukaemia; CML, chronic myeloid leukaemia; CLL, chronic lymphocytic leukaemia; LRT, likelihood ratio test; NA, not applicable; NC, not calculable; NM, not modelled; PLS, piecewise linear spline; RCS, restricted cubic spline.
Two knot solutions (8.3 and 46.9 mGy) in the non-CLL PLS-lagged model indicated the points of departure from linearity in the low- and high-dose ranges. The slope estimates (ERR per 100 mGy) in each segment were: −6.6 (95% CI −9.8 to −0.92) for doses ≤8.3 mGy; 2.0 (95% CI 0.30 to 4.2), for doses >8.3 and <46.9 mGy; and −0.16 (95% CI: <−0.22 to 0.39) for doses ≥46.9 mGy. By contrast, the best fit of time-windows-based data was achieved in models using two segments with the knot at 20.1 mGy; however, the two segments were not statistically significantly different (p=0.86). The first segment was positive and statistically significant (ERR per 100 mGy=6.4; 95% CI 0.31 to 17.0). The second segment was negative and lacked statistical significance (ERR per 100 mGy= −0.82; 95% CI −1.7 to 4.4).
DISCUSSION
With 369 leukaemia cases in a population of 105 245 US nuclear workers, the current study is among the largest examining the leukaemogenicity of protracted ionising radiation exposures. The large size improved power compared with the previous analyses,3,4 and enabled analyses by leukaemia subtype. Estimates of risk per unit dose were consistent with results from previous studies (table 4), as were temporal patterns observed in populations exposed to known leukaemogens, such as benzene,24,25 ionising radiation4,26–28 and smoking,29 whereby leukaemia risks lessen with time since exposure. Nevertheless, statistical power remains limited, leading to results which, in many cases, could be explained by the role of chance.
Table 4.
Leukaemia ERR at 100 mGy exposure by subtype and by study
| Study | Non-CLL | AML | CML | CLL |
|---|---|---|---|---|
| Current study | 0.09 (95% CI −0.17 to 0.65) | 0.22 (95% CI −0.19 to 1.2) | 0.29 (95% CI <0 to 2.6) | −0.03 (95% CI −0.16 to 0.74) |
| 264 decedents | 150 decedents | 52 decedents | 74 decedents | |
| LCCS3,4 | 0.26 (95% CI <−0.10 to 1.03) | NR | NR | −0.20 (95% CI <0 to 1.4) |
| 184 decedents | 43 decedents | |||
| 15-country study*7,30 | 0.196 (90% CI −0.026 to 0.59) | −0.41 (90% CI −0.76 to 0.17) | 1.0 (90% CI −0.086 to 4.02) | −0.17 (95% CI −0.89 to 0.40) |
| 196 decedents | 81 decedents | 45 decedents | 47 decedents | |
| Three-country study31 | 0.22 (90% CI 0.013 to 0.57) | 0.34 (90% CI <0 to 1.49) | 1.1 (90% CI 0.29 to 3.09) | −0.09 (90% CI <0 to 0.73) |
| 119 decedents | 32 decedents | 28 decedents | 27 decedents | |
| NRRW6 | 0.17 (90% CI 0.006 to 0.43) | 0.12 (90% CI −0.12 to 0.65) | 0.33 (90% CI 0.04 to 0.93) | <−0.19 (90% CI <−0.19 to 0.12) |
| 198 decedents | 102 decedents | 44 decedents | 69 decedents | |
| LSS†26 | 0.47 (90% CI 0.35 to 0.64) | 0.43 (90% CI 0.27 to 0.66) | 0.64 (90% CI 0.30 to 1.37) | NR |
| 310 decedents‡ | 124 decedents | 58 decedents |
AML and CLL results were calculated using a log-linear relative risk model of the form RR(d)=exp(βd) and ERR=RR−1.
Based on a linear ERR model without effect modification of the form ERR(d)=βd.
Also included seven CLL cases and 121 leukaemias not specified as AML or CML.
AML, acute myeloid leukaemia; CML, chronic myeloid leukaemia; CLL, chronic lymphocytic leukaemia; ERR, excess relative risk; LCCS, NIOSH Leukaemia Mortality Case control Study; LSS, Life-Span Study of Japanese Atomic Bomb Survivors; NC, not calculable; NR, not reported; NRRW, UK National Registry of Radiation Workers.
A non-linear dose-response was most evident in lagged models, whereby risk attenuation was observed at very low doses (<10 mGy) and at doses greater than 100 mGy. By contrast, strong departure from linearity was not evident in time-windows-based models, where risks appear greater than in lagged models. The elevated risk in windows-based models suggested temporal patterns in dose-response with time since exposure that were similar to, but more pronounced than, that observed in Japanese atomic bomb survivors26 and in other nuclear worker cohorts.4,28 AML risk appears particularly susceptible to temporal effects, where a 30-fold change in risk was observed in the windows-based model compared with a lagged model. Without accounting for these temporal effects in studies of protracted exposures with long follow-up since periods of highest exposure, dose-response analyses may provide distorted results, as evident in the lagged models.
We examined the combined effects from low- and high-LET radiations and determined that there was little evidence of high-LET effects in this study. Solutions to weights in time-windows-based models appeared in fair agreement with expectation.22,32,33 By contrast, weights in lagged models were larger than expected, although CIs were wide. Exposure misclassification is a possible cause of spurious results given the sparseness and poor quality of available plutonium and neutron exposure measurements. It is also conceivable that other characteristics unique to the few plutonium workers or persons with measurable neutron dose may have led to inflated weight estimates. Last, the cohort was assembled from nuclear facilities selected to minimise high-LET dose contributions4; therefore, only a small number of study participants were significantly exposed to plutonium or neutrons.
Risk attenuation in the low-dose range was not evident in previous studies of workers employed at these facilities.4,8 The addition of low-exposed cases through extended follow-up may be a source of the non-linear dose-response. Restricting the current study to non-CLL cases with follow-up through 1990 (n=112 cases) reduced the non-linear low-dose effect and the best fit was obtained from a linear model (ERR per 100 mGy=0.6; 95% CI −0.06 to 2.0). Diminishing leukaemia risk with continued follow-up has been observed in populations exposed to benzene24,34 and ionising radiation.35,36 Rinsky et al34 reported a decline in the leukaemia standardised mortality ratio from 3.37 (95% CI 1.54 to 6.41) to 2.56 (95% CI 1.43 to 4.22) after adding 15 years of follow-up to a cohort of white male benzene-exposed rubber hydrochloride workers. Silver et al24 also examined these workers using yearly follow-up information and found a fivefold decrease in the risk estimates from 1961 to 1996. In studies of ionising radiation, Boice et al35 reported decreased non-CLL leukaemia mortality after adding 9 years of follow-up to the study of Rocketdyne radiation workers,37 as did Krestinina et al36 after adding 6 years of follow-up to the Techa River cohort.38 Exposure profiles for these populations are temporally similar to the current study, where exposures occurred earlier in the follow-up period, and continued follow-up resulted in negligible additional dose. Given that leukaemia risk diminishes with time since exposure, it is less likely that new cases, who have not accrued additional dose in the extended follow-up period, are a consequence of ionising radiation exposure.
Risk attenuation at higher exposures is often observed in occupational studies, and may result from a number of causes, including a depletion of susceptible persons, biologic saturation, exposure misclassification or a healthy worker survivor bias.39 A healthy worker effect is less likely because leukaemia is not strongly related to lifestyle factors. Biologic saturation is also unlikely given that ‘high doses’ in this study are well below known thresholds for deterministic effects, and below doses that appear within the linear range in higher-dose studies. Exposure misclassification, although inevitable, is believed to be reduced by the availability of individual measurement data and the exclusion of early workers who were prone to highly uncertain dose estimates. Nevertheless, this source of uncertainty cannot be completely ruled out as cause for the observed attenuation. Some suggest that variations in risk in an exposed group may be attributable to small numbers of individuals who are predisposed to radiation-induced leukaemia.40,41 In chronically exposed populations, those most susceptible to the disease present early after relevant exposure, and persons who are resistant remain leukaemia-free and continue to accumulate dose, which may explain the observed risk attenuation at higher doses and the dilution of risk with increased follow-up.
Of course, results from low-dose studies are highly susceptible to distortion by confounding and exposure measurement errors. Benzene, a known leukaemogen and a confounder in the previous study, was not associated with leukaemia risk in this study. A possible explanation is the exclusion of early workers who were at greatest risk of benzene exposure in the previous study. SES was used as a surrogate for lifestyle factors; therefore, a potential for incomplete control of smoking effects cannot be ruled out, although smoking is only weakly related to leukaemia. Regarding exposure, extensive searches for exposure records were conducted to estimate career doses; nevertheless, some exposure misclassification from incomplete or erroneous exposure data is likely. For example, we found that cases with exposures of 1.0 mGy, or less, were typically professionals who were hired later, were older at hire, and worked fewer years prior to termination compared with cases in the higher-dose categories. These patterns suggested a potential for previous dose during employment elsewhere and the lack of this information in dose-response analyses may have led to biased estimates. To examine this potential, non-CLL cases in the lowest-dose category (n=74) were matched to exposure data maintained on claimants under the Energy Employee Occupational Illness Compensation Programme Act. There were eight claimants identified, and four had additional exposure information in claimant files. Had these data been available for our study, a change in exposure category would have resulted in every case.
Although we have offered several possible explanations, the cause for the non-linear dose-response remains unknown. In the absence of known biases, one could interpret the attenuated risk at low doses as evidence of a threshold or protective effect. There has been considerable debate regarding the validity of the linear-no-threshold (LNT) dose-response model because the carcinogenicity of low- and very low-dose radiation exposure is largely uncertain.42–48 Although threshold or protective effects at low doses cannot be ruled out by our findings, we did not observe attenuated risk in time-windows-based models that very likely represent the most relevant period of risk following exposure.
CONCLUSIONS
Positive, but imprecise estimates of excess relative risk were observed for all leukaemia outcomes except the CLL subtype. Non-CLL leukaemia risk was time-dependent, where peak excess risk per unit dose was observed at select times prior to disease onset. Marked improvements in estimates were observed in time-windows analyses, where linear dose-response models prevailed over other forms tested. Overall, the point estimates and temporal patterns observed in this study were in reasonable agreement with previous results; however, some dilution from extended follow-up without concomitant increases in dose was apparent.
Risk models using exposure lags exhibited attenuated risk in the low-dose (<10 mGy) and high-dose (>100 mGy) regions. The low-dose attenuation disappeared in models based on dose accrued in time-windows associated with maximum leukaemia risk prior to attained age of the case. Risk attenuation in the low-dose region is particularly difficult to explain given that similar results were not observed in previous studies; however, there is some evidence that extended follow-up and exposure misclassification in the low-dose range may be partially at fault. Low-dose effects, which are most important for risk assessment, are much more difficult to discern compared with the effects at high doses given limited statistical power and results that are highly susceptible to bias. Future research should focus on methods that improve evaluations of the dose-response, particularly in the low-dose range. In particular, better collection of career dose information may greatly reduce the potential for exposure misclassification. For example, future studies could be markedly improved by a consolidation of exposure information from NRC, DOE and military sources into a national dose registry.
Supplementary Material
What this study adds.
Questions remain concerning the translation of risks observed in Japanese atomic bomb survivors to working populations who are exposed to low doses of ionising radiation delivered at low rates of exposure.
With 369 leukaemia deaths identified in a pooled cohort of over 100 000 nuclear workers from six major US facilities, this study is among the largest leukaemia studies ever conducted with low-dose radiation. The large study size enabled analysis by leukaemia subtype and detailed examination of dose-response characteristics that have not been completed prior.
Positive but imprecise estimates of leukemia risk, which increased with dose, were observed for myeloid leukaemias but not for chronic lymphocytic leukaemia (CLL). These findings were consistent with previous studies that suggest a weak leukaemogenicity of ionising radiation for non-CLL subtypes.
Acknowledgments
The study also served to partially satisfy the requirements for doctoral dissertation research by the corresponding author, who is grateful to the dissertation committee: Dr S Pinney, Dr M Schubauer-Berigan, Dr C R Buncher, Dr R Hornung, and Dr H Spitz, for their encouragement and valuable input.
Funding This research was funded by the National Institute for Occupational Safety and Health (NIOSH) following competitive award as an intramural research project under the National Occupational Research Agenda (NORA), and made possible through the cooperation and support of the DOE and their employees and contractors.
Footnotes
Contributors All coauthors had full access to the data used in the study, and take full responsibility for the integrity of the data and data analyses.
Disclaimer The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institute for Occupational Safety and Health.
Competing interests None.
Ethics approval The NIOSH Institutional Review Board (IRB).
Provenance and peer review Not commissioned; externally peer reviewed
References
- 1.Committee to Assess Health Risks from Exposure to Low Levels of Ionizing Radiation, National Research Council. Health Risks from Exposure to Low Levels of Ionizing Radiation: BEIR VII—Phase 2. Washington, DC: National Academies Press; 2006. p. 406. [PubMed] [Google Scholar]
- 2.United Nations Scientific Committee on the Effects of Atomic Radiation. UNSCEAR 2006 Report: Effects of Ionizing Radiation, Annex A: Epidemiological Studies of Radiation and Cancer. New York, NY: 2009. p. 310. [Google Scholar]
- 3.Schubauer-Berigan MK, Daniels RD, Fleming DA, et al. Chronic lymphocytic leukaemia and radiation: findings among workers at five US nuclear facilities and a review of the recent literature. Br J Haematol. 2007;139:799–808. doi: 10.1111/j.1365-2141.2007.06843.x. [DOI] [PubMed] [Google Scholar]
- 4.Schubauer-Berigan MK, Daniels RD, Fleming DA, et al. Risk of chronic myeloid and acute leukemia mortality after exposure to ionizing radiation among workers at four U.S. nuclear weapons facilities and a nuclear naval shipyard. Radiat Res. 2007;167:222–32. doi: 10.1667/rr0724.1. [DOI] [PubMed] [Google Scholar]
- 5.Daniels RD, Schubauer-Berigan MK. A meta-analysis of leukaemia risk from protracted exposure to low-dose gamma radiation. Occup Environ Med. 2011;68:457–64. doi: 10.1136/oem.2009.054684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Muirhead CR, O’Hagan JA, Haylock RG, et al. Mortality and cancer incidence following occupational radiation exposure: third analysis of the national registry for radiation workers. Br J Cancer. 2009;100:206–12. doi: 10.1038/sj.bjc.6604825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cardis E, Vrijheid M, Blettner M, et al. The 15-country collaborative study of cancer risk among radiation workers in the nuclear industry: estimates of radiation-related cancer risks. Radiat Res. 2007;167:396–416. doi: 10.1667/RR0553.1. [DOI] [PubMed] [Google Scholar]
- 8.Schubauer-Berigan MK, Macievic GV, Utterback DF, et al. An epidemiologic study of mortality and radiation-related risk of cancer among workers at the Idaho National Engineering and Environmental Laboratory, a US Department of Energy facility. Cincinnati, OH: National Institute for Occupational Safety and Health; 2005. p. 220. [Google Scholar]
- 9.Beaumont JJ, Steenland K, Minton A, et al. A computer program for incidence density sampling of controls in case-control studies nested within occupational cohort studies. Am J Epidemiol. 1989;129:212–19. doi: 10.1093/oxfordjournals.aje.a115111. [DOI] [PubMed] [Google Scholar]
- 10.Daniels RD, Kubale TL, Spitz HB. Radiation exposure from work-related medical X-rays at the Portsmouth Naval Shipyard. Am J Ind Med. 2005;47:206–16. doi: 10.1002/ajim.20141. [DOI] [PubMed] [Google Scholar]
- 11.Anderson JL, Daniels RD. Bone marrow dose estimates from work-related medical x-ray examinations given between 1943 and 1966 for personnel from five U.S. nuclear facilities. Health Phys. 2006;90:544–53. doi: 10.1097/01.HP.0000194230.29763.0c. [DOI] [PubMed] [Google Scholar]
- 12.Daniels RD, Lodwick CJ, Schubauer-Berigan MK, et al. Assessment of plutonium exposures for an epidemiological study of US nuclear workers. Radiat Prot Dosimetry. 2006;118:43–55. doi: 10.1093/rpd/nci330. [DOI] [PubMed] [Google Scholar]
- 13.Daniels RD, Schubauer-Berigan MK. Bias and uncertainty of penetrating photon dose measured by film dosemeters in an epidemiological study of US nuclear workers. Radiat Prot Dosimetry. 2005;113:275–89. doi: 10.1093/rpd/nch470. [DOI] [PubMed] [Google Scholar]
- 14.Yiin JH, Schubauer-Berigan MK, Silver SR, et al. Risk of lung cancer and leukemia from exposure to ionizing radiation and potential confounders among workers at the Portsmouth Naval Shipyard. Radiat Res. 2005;163:603–13. doi: 10.1667/rr3373. [DOI] [PubMed] [Google Scholar]
- 15.SAS Software, Version 9.2, Copyright (2002–2003) SAS and all other SAS Institute Inc. product or service names are registered trademarks or trademarks of SAS Institute. Cary, NC, USA: SAS Institute Inc; [Google Scholar]
- 16.Langholz B, Richardson DB. Fitting general relative risk models for survival time and matched case-control analysis. Am J Epidemiol. 2010;171:377–83. doi: 10.1093/aje/kwp403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Harrell FE. Regression modeling strategies with applications in linear models, logistic regression, and survival analysis. New York, NY: Springer-Verlag New York Inc; 2001. [Google Scholar]
- 18.Richardson DB, Cole SR, Chu H, et al. Lagging exposure information in cumulative exposure-response analyses. Am J Epidemiol. 2011;174:1416–22. doi: 10.1093/aje/kwr260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Akaike H. Information theory and an extension of the maximum likelihood principle. In: Petrov BNCF, editor. Second International Symposium on Information Theory. Budapest: 1973. pp. 267–81. [Google Scholar]
- 20.International Commission on Radiological Protection. Conversion coefficients for use in radiological protection against external radiation. Adopted by the ICRP and ICRU in September 1995. Ann ICRP. 1996;26:1–205. [PubMed] [Google Scholar]
- 21.Kocher DC, Apostoaei AI, Hoffman FO. Radiation effectiveness factors for use in calculating probability of causation of radiogenic cancers. Health Phys. 2005;89:3–32. doi: 10.1097/01.hp.0000154172.48895.45. [DOI] [PubMed] [Google Scholar]
- 22.Puskin JS, Nelson CB, Pawel DJ. Estimating radiological cancer risks, addendum: uncertainty analysis. Washington, DC: U.S. Environmental Protection Agency; 1999. p. 37. [Google Scholar]
- 23.Environmental Protection Agency. EPA Radiogenic cancer risk models and projections for the US population. Washington, DC: U.S. Environmental Protection Agency; 2011. p. 175. [Google Scholar]
- 24.Silver SR, Rinsky RA, Cooper SP, et al. Effect of follow-up time on risk estimates: a longitudinal examination of the relative risks of leukemia and multiple myeloma in a rubber hydrochloride cohort. Am J Ind Med. 2002;42:481–9. doi: 10.1002/ajim.10139. [DOI] [PubMed] [Google Scholar]
- 25.Finkelstein MM. Leukemia after exposure to benzene: temporal trends and implications for standards. Am J Ind Med. 2000;38:1–7. doi: 10.1002/1097-0274(200007)38:1<1::aid-ajim1>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]
- 26.Richardson D, Sugiyama H, Nishi N, et al. Ionizing radiation and leukemia mortality among Japanese atomic bomb survivors, 1950–2000. Radiat Res. 2009;172:368–82. doi: 10.1667/RR1801.1. [DOI] [PubMed] [Google Scholar]
- 27.Richardson DB, Wing S. Leukemia mortality among workers at the Savannah River Site. Am J Epidemiol. 2007;166:1015–22. doi: 10.1093/aje/kwm176. [DOI] [PubMed] [Google Scholar]
- 28.Shilnikova NS, Preston DL, Ron E, et al. Cancer mortality risk among workers at the Mayak nuclear complex. Radiat Res. 2003;159:787–98. doi: 10.1667/0033-7587(2003)159[0787:cmrawa]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 29.Richardson DB, Terschuren C, Pohlabeln H, et al. Temporal patterns of association between cigarette smoking and leukemia risk. Cancer Causes Control. 2008;19:43–50. doi: 10.1007/s10552-007-9068-7. [DOI] [PubMed] [Google Scholar]
- 30.Vrijheid M, Cardis E, Ashmore P, et al. Ionizing radiation and risk of chronic lymphocytic leukemia in the 15-country study of nuclear industry workers. Radiat Res. 2008;170:661–5. doi: 10.1667/RR1443.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cardis E, Gilbert ES, Carpenter L, et al. Effects of low doses and low dose rates of external ionizing radiation: cancer mortality among nuclear industry workers in three countries. Radiat Res. 1995;142:117–32. [PubMed] [Google Scholar]
- 32.Grogan HA, Sinclair WK, Voilleque PG. Risks of fatal cancer from inhalation of 239,240 plutonium by humans: a combined four-method approach with uncertainty evaluation. Health Phys. 2001;80:447–61. doi: 10.1097/00004032-200105000-00005. [DOI] [PubMed] [Google Scholar]
- 33.Spiers FW, Vaughan J. The toxicity of the bone seeking radionuclides. Leuk Res. 1989;13:347–50. doi: 10.1016/0145-2126(89)90073-8. [DOI] [PubMed] [Google Scholar]
- 34.Rinsky RA, Hornung RW, Silver SR, et al. Benzene exposure and hematopoietic mortality: a long-term epidemiologic risk assessment. Am J Ind Med. 2002;42:474–80. doi: 10.1002/ajim.10138. [DOI] [PubMed] [Google Scholar]
- 35.Boice JD, Cohen SS, Mumma MT, et al. Updated mortality analysis of radiation workers at rocketdyne (atomics international), 1948–2008. Radiat Res. 2011;176:244–58. doi: 10.1667/RR2487.1. [DOI] [PubMed] [Google Scholar]
- 36.Krestinina L, Preston DL, Davis FG, et al. Leukemia incidence among people exposed to chronic radiation from the contaminated Techa River, 1953–2005. Radiat Environ Biophys. 2010;49:195–201. doi: 10.1007/s00411-009-0257-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Boice JD, Cohen SS, Mumma MT, et al. Mortality among radiation workers at Rocketdyne (atomics international), 1948–1999. Radiat Res. 2006;166:98–115. doi: 10.1667/RR3582.1. [DOI] [PubMed] [Google Scholar]
- 38.Krestinina LY, Preston DL, Ostroumova EV, et al. Protracted radiation exposure and cancer mortality in the Techa River Cohort. Radiat Res. 2005;164:602–11. doi: 10.1667/rr3452.1. [DOI] [PubMed] [Google Scholar]
- 39.Stayner L, Steenland K, Dosemeci M, et al. Attenuation of exposure-response curves in occupational cohort studies at high exposure levels. Scand J Work Environ Health. 2003;29:317–24. doi: 10.5271/sjweh.737. [DOI] [PubMed] [Google Scholar]
- 40.Nakamura N. A hypothesis: radiation-related leukemia is mainly attributable to the small number of people who carry pre-existing clonally expanded preleukemic cells. Radiat Res. 2005;163:258–65. doi: 10.1667/rr3311. [DOI] [PubMed] [Google Scholar]
- 41.Hall EJ, Brenner DJ, Worgul B, et al. Genetic susceptibility to radiation. Adv Space Res. 2005;35:249–53. doi: 10.1016/j.asr.2004.12.032. [DOI] [PubMed] [Google Scholar]
- 42.Brenner DJ, Sachs RK. Estimating radiation-induced cancer risks at very low doses: rationale for using a linear no-threshold approach. Radiat Environ Biophys. 2006;44:253–6. doi: 10.1007/s00411-006-0029-4. [DOI] [PubMed] [Google Scholar]
- 43.Tubiana M, Aurengo A, Averbeck D, et al. The debate on the use of linear no threshold for assessing the effects of low doses. J Radiol Prot. 2006;26:317–24. doi: 10.1088/0952-4746/26/3/N01. [DOI] [PubMed] [Google Scholar]
- 44.Scott BR. Low-dose radiation risk extrapolation fallacy associated with the linear-no-threshold model. Hum Exp Toxicol. 2008;27:163–8. doi: 10.1177/0960327107083410. [DOI] [PubMed] [Google Scholar]
- 45.Breckow J. Linear-no-threshold is a radiation-protection standard rather than a mechanistic effect model. Radiat Environ Biophys. 2006;44:257–60. doi: 10.1007/s00411-006-0030-y. [DOI] [PubMed] [Google Scholar]
- 46.Wall BF, Kendall GM, Edwards AA, et al. What are the risks from medical X-rays and other low dose radiation? Br J Radiol. 2006;79:285–94. doi: 10.1259/bjr/55733882. [DOI] [PubMed] [Google Scholar]
- 47.Charles MW. LNT—an apparent rather than a real controversy? J Radiol Prot. 2006;26:325–9. doi: 10.1088/0952-4746/26/3/N02. [DOI] [PubMed] [Google Scholar]
- 48.Chadwick KH, Leenhouts HP. Radiation risk is linear with dose at low doses. Br J Radiol. 2005;78:8–10. doi: 10.1259/bjr/51173413. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.