SUMMARY
Background
High dietary protein intake is associated with greater survival in maintenance haemodialysis (MHD) patients. High-protein foods may increase dietary phosphorus burden, which is associated with increased mortality in these patients. Hypothesis is: an egg white based diet with low phosphorus to protein ratio (<1.4 mg/g) will lower serum phosphorus without deteriorating the nutritional status in MHD patients.
Objective
We assessed serum phosphorus and albumin levels in MHD patients who agreed to ingest one meal per day with pasteurised liquid egg whites without phosphorus additives, as principal protein source.
Methods
Thirteen otherwise stable MHD patients with serum phosphorus >4.0 mg/dl agreed to consume eight ounces (225 g) of pasteurised liquid egg whites one meal per day for six weeks. Recipes were suggested to improve diet variety.
Results
Thirteen participating patients included seven women, three African Americans and five diabetics. Twelve patients exhibited drop in serum phosphorus. Mean population fall in serum phosphorus was 0.94 mg/dl, i.e. from 5.58 ± 1.34 (mean ± SD) to 4.63 ± 1.18 (p = 0.003). Serum albumin showed an increase by 0.19 g/dl, i.e. from 4.02 ± 0.29 to 4.21 ± 0.36 g/dl (p = 0.014). Changes in phosphorus pill count were not statistically significant (p = 0.88). The egg white diet was well tolerated, and recipe variety appreciated.
Conclusion
Pasteurised liquid egg whites may be an effective diet component lowering serum phosphorus without risking malnutrition. Controlled trials are indicated to examine egg white based dietary interventions in MHD patients at home or during haemodialysis treatment.
Keywords: Dietary intervention, Pasteurised liquid egg whites, Phosphorus, Phosphorus-to-protein ratio, Protein, Protein-energy wasting
INTRODUCTION
In individuals with chronic kidney disease (CKD) who undergo maintenance dialysis treatment, a low dietary protein intake is associated with poor survival (Kopple 2001; Shinaberger et al. 2006). Inadequate protein intake may be caused by anorexia related to inflammation (Kalantar-Zadeh et al. 2004), or as a result of imposed dietary restrictions to control phosphorus (Shinaberger et al. 2008), potassium (Noori et al. 2010a) or fluid intake (Kalantar-Zadeh et al. 2009). Low-protein intake can lead to hypoalbuminaemia and protein-energy wasting (PEW) (Kalantar-Zadeh K et al. 2004; Fouque et al. 2008). The PEW per se may engender or aggravate inflammatory and oxidative processes leading to endothelial dysfunction, cardiovascular and thromboembolic events and death (Ling et al. 2004; Kalantar-Zadeh et al. 2007a; Kovesdy et al. 2009). Malnutrition and inflammation are also related to poor quality of life in dialysis patients (Kalantar-Zadeh et al. 2001; Kalantar-Zadeh & Unruh 2005; Rambod et al. 2009; Noori et al. 2010c). Hence, the so-called malnutrition-inflammation cachexia syndrome has been implicated as a powerful indicator of poor clinical outcome in maintenance dialysis patients (Kalantar-Zadeh et al. 2003a). Dialysis patients with a higher protein and possibly energy intake usually have a higher body mass, larger nutritional reserve, including greater protein mass and higher serum albumin level, and better survival (Kalantar-Zadeh et al. 2003a, 2003b; Kalantar-Zadeh et al. 2005a; Rambod et al. 2008). Hence, dietary interventions to improve nutritional status are urgently needed (Kalantar-Zadeh et al. 2005b).
Foods high in protein are also a main source of dietary phosphorus (Kalantar-Zadeh et al. 2010). Hyperphosphataemia is a common problem in individuals with advanced CKD (Hruska & Teitelbaum 1995; Moe et al. 2006). Gradual decline in renal phosphorus clearance during the progression of CKD leads to increased serum phosphorus concentrations (Levin et al. 2007; Kovesdy et al. 2010a), leading to active vitamin D deficiency, hyperparathyroidism and renal osteodystrophy (Hernandez et al. 2005; Kovesdy & Kalantar-Zadeh 2008). Hyperphosphataemia may also contribute to worsening vascular calcification (Shantouf et al. 2010) and increased risk of cardiovascular morbidity (Block et al. 1998; Block et al. 2004; Achinger & Ayus 2006; Kalantar-Zadeh et al. 2006; Hruska et al. 2007). Hence, correction and prevention of hyperphosphataemia via dietary intervention is a main component of the management of dialysis patients. However, imposing dietary phosphorus restriction is often associated with a reduction in dietary protein intake. The latter can lead to malnutrition and PEW (Kalantar-Zadeh et al. 2003a; Shinaberger et al. 2006). It is thus important to examine sources of dietary protein that are associated with the least phosphorus burden, especially since a recent study showed that higher dietary phosphorus intake or foods with higher phosphorus-to-protein ratio are associated with increased death risk in dialysis patients (Noori et al. 2010b). Egg whites are a good source of biologically high-value protein with a relatively low phosphorus-to-protein ratio (<2 mg/g) and low-cholesterol content (Kalantar-Zadeh et al. 2010; Noori et al. 2010d). Hence, we conducted a pilot project among maintenance haemodialysis (MHD) outpatients of a dialysis clinic and examined the hypothesis that when dialysis patients substituted pasteurised liquid egg white for meat at one meal per day, serum phosphorus level would decrease while serum albumin level would remain constant or increase.
METHODS
PARTICIPANTS
All MHD patients treated at DaVita Carroll County Dialysis Centre, Westminster, MD, who had undergone haemodialysis treatment for at least three months were invited to participate in this project. In order to be eligible, serum phosphorus of the MHD patient had to be ≥ 4.0 mg/dl. The main exclusion criterion was egg allergy. Participants were required to adhere to the intervention guidelines and agreed to the addition of pasteurised liquid egg white as a substitution for meat during one meal per day for at least six weeks. Fourteen MHD patients, who met the inclusion and exclusion criteria, agreed to participate in this project, out of whom, 13 subjects completed the six weeks of the intervention. One candidate was excluded from analyses due to a prolonged hospitalisation at the initial stages of the project and unrelated to the project.
DATA COLLECTION
The project included a baseline phase of up to four weeks for data collection and a diet intervention phase of six weeks. During the baseline phase, serum concentrations of phosphorus and albumin were recorded. Each participant maintained daily records of food and phosphorus binder intake, received weekly blood draws throughout the six weeks of diet supplementation.
Final blood work was collected at the conclusion of the study.
DIETARY INTERVENTION
All participants were instructed to consume eight ounces of pasteurised liquid egg whites as a meat replacement at one meal each day. Recipe suggestions were provided for smoothies and also for cooking. Participants continued to record consumption in daily food diaries.
The pasteurised liquid egg white product used in this study was AllWhites™ manufactured and distributed by Crystal Farms (Lake Mills, WI, USA), an off-shelf marketed product available in the dairy section of grocery stores. According to the manufacturer information on its publicly accessed website (http://www.betterneggs.com), it constitutes 100% liquid egg whites without preservatives or additives. Each eight oz serving contained 120 kcal, 380 mg sodium, 360 mg potassium, 4 g carbohydrates, 24 g protein and 34 mg phosphorus (data based on United States Department of Agriculture SR-21 nutrient database).
SERUM PHOSPHORUS AND ALBUMIN, AND PHOSPHORUS BINDER
For each participant, the mean of all serum phosphorus and albumin levels during the preintervention period was calculated as the baseline levels. The same biochemical levels were also recorded at the end of the dietary intervention phase, and the change in the levels for each patient was calculated. The change in the daily intake of phosphorus binders, averaged over the entire week, was calculated during the six weeks of intervention and three weeks of post-intervention.
STATISTICAL ANALYSIS
A paired t-test was used to assess the meaningfulness of the change in serum phosphorus and albumin levels. The Wilcoxon Signed-Ranks test was used to confirm the t-test. Similar analyses were performed for the changes in the averaged daily phosphorus binder intake. A p value <0.05 was considered statistically significant.
RESULTS
The 13 participating patients included seven women (54%), three African Americans (23%) and five diabetics (38%). Patients’ mean age was 62 years ranging from 35 to 85 years. Table 1 shows baseline and post-intervention serum concentrations of phosphorus and albumin as well as the averaged counts of the phosphorus binder pills in each of the 13 participating patients. As shown in Figure 1, all but one patient exhibited a drop in serum phosphorus by the end of intervention. Figure 2 shows the change in serum albumin. In 10 patients, serum albumin increased, whereas in one patient, it decreased and in two patients, no change occurred. Table 2 and Figure 3 show that longitudinal changes in daily phosphorus binder pill counts over the nine weeks of observation. In general, no major changes in the pill count were observed. Table 3 shows the results of the statistical analyses. Serum phosphorus dropped by 0.94 mg/dl, i.e. from 5.58 to 4.63 (p = 0.003). Serum albumin showed an increase by 0.19 g/dl, i.e. from 4.02 to 4.21 g/dl (p = 0.014). Changes in phosphorus pill count were not statistically significant (p = 0.88). The egg white diet was well tolerated and patients reported that they enjoyed the variety of both smoothie and cooked options provided.
Table 1.
Baseline and postintervention serum concentrations of phosphorus and albumin as well as the averaged counts of the phosphorus binder pills in each of the 13 participating patients.
| Baseline | Posttrial | |||||
|---|---|---|---|---|---|---|
| Patient ID | Phosphorus (mg/dl) | Albumin (g/dl) | Daily binder pill count | Phosphorus (mg/dl) | Albumin (g/dl) | Daily binder pill count |
| A | 5 | 4.1 | 3.0 | 4.7 | 4.4 | 2.3 |
| B | 3.5 | 3.9 | 9.0 | 3.3 | 4 | 9.0 |
| C | 4.7 | 4.0 | 6.0 | 3.7 | 4.1 | 6.0 |
| D | 4.6 | 3.4 | 7.7 | 3.9 | 3.4 | 8.6 |
| E | 5.4 | 4.1 | 12.0 | 3.6 | 4.3 | 14.9 |
| F | 6.7 | 4.0 | 2.9 | 6.5 | 4.2 | 0.0 |
| G | 8.4 | 3.9 | 11.7 | 5.3 | 4.1 | 12.4 |
| H | 6.5 | 4.5 | 6.6 | 5.7 | 4.6 | 5.7 |
| I | 5.5 | 3.7 | 4.3 | 4.8 | 4.6 | 6.0 |
| J | 7.5 | 4.2 | 7.7 | 6.3 | 4.4 | 2.6 |
| K | 5.3 | 4.4 | 6.9 | 5.4 | 4.7 | 7.7 |
| L | 4.6 | 4.3 | 5.1 | 4.3 | 4.2 | 8.1 |
| M | 4.8 | 3.8 | 5.0 | 2.7 | 3.8 | 5.7 |
Phosphorus binder pill data are from week nine of the project.
Figure 1.
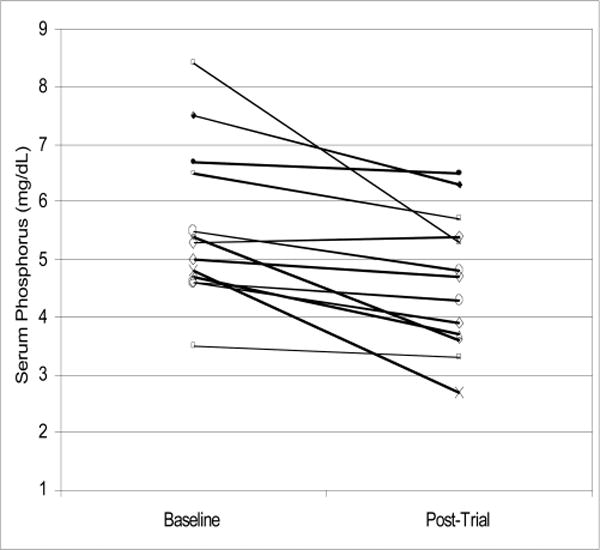
Change in serum phosphorus concentrations from baseline to postintervention in each of the 13 participating patients.
Figure 2.
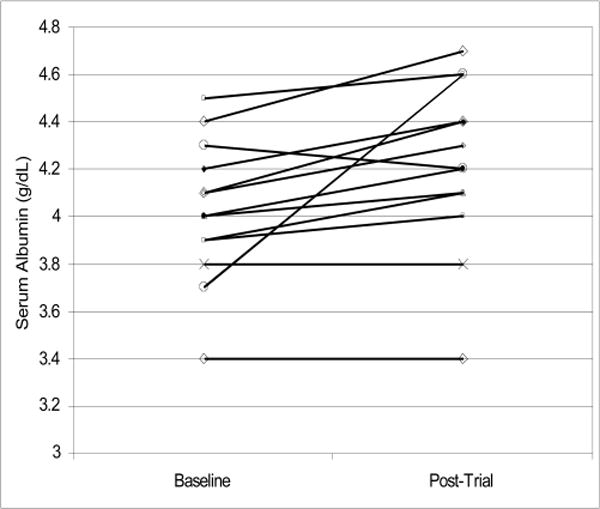
Change in serum albumin concentrations from baseline to postintervention in each of the 13 participating patients.
Table 2.
Changes in phosphorus binder pill count over the nine weeks of the study.
| ID | Physician order | Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9 |
|---|---|---|---|---|---|---|---|---|---|---|
| A | 1 TM w meals | 3.0 | 2.1 | 2.6 | 2.3 | 2.9 | 2.1 | 2.1 | 2.0 | 2.3 |
| B | 3 PL w meals | 9.0 | 9.0 | 9.0 | 9.0 | 9.0 | 9.0 | 9.0 | 9.0 | 9.0 |
| C | 2 TM w meals | 6.0 | 4.0 | 5.1 | 4.3 | 3.4 | 4.0 | 5.7 | 4.0 | 6.0 |
| D | 3 TM w meals | 7.7 | 7.7 | 8.1 | 7.3 | 9.0 | 8.6 | 8.1 | 8.6 | 8.6 |
| E | 4 RG w meals | 12.0 | 13.7 | 13.7 | 13.7 | 13.1 | 15.4 | 12.9 | 14.9 | 14.9 |
| F | 2 RG w meals | 2.9 | 2.3 | 2.0 | 4.0 | 0.6 | 2.6 | 0.0 | 0.0 | 0.0 |
| G | 4 PL w meals | 11.7 | 6.9 | 13.7 | 12.0 | 7.4 | 10.3 | 12.0 | 9.0 | 12.4 |
| H | 2 RG w meals | 6.6 | 5.7 | 6.0 | 5.4 | 5.7 | 4.0 | 6.3 | 5.7 | 5.7 |
| I | 2 PL w meals | 4.3 | 6.0 | 6.0 | 6.0 | 4.3 | 6.0 | 6.0 | 6.0 | 6.0 |
| J | 3 PL w meals | 7.7 | 7.0 | 7.4 | 7.6 | 7.0 | 4.4 | 4.6 | 3.0 | 2.6 |
| K | 3 PL w meals | 6.9 | 4.7 | 5.6 | 4.9 | 7.3 | 8.1 | 8.1 | 6.4 | 7.7 |
| L | 3 RG w meals | 5.1 | 5.1 | 4.9 | 5.4 | 4.3 | 4.7 | 3.4 | 7.0 | 8.1 |
| M | 3 PL w meals | 5.0 | 6.6 | 5.7 | 4.4 | 4.7 | 5.6 | 5.4 | 6.3 | 5.7 |
| Average daily pills (over one week) | 6.8 | 6.2 | 6.9 | 6.6 | 6.1 | 6.5 | 6.4 | 6.3 | 6.8 | |
| Average daily pills (over three weeks) | 6.6 | 6.4 | 6.5 | |||||||
TM: tums (calcium carbonate), PL: phoslo (calcium acetate), RG: renagel (sevelamer).
Figure 3.
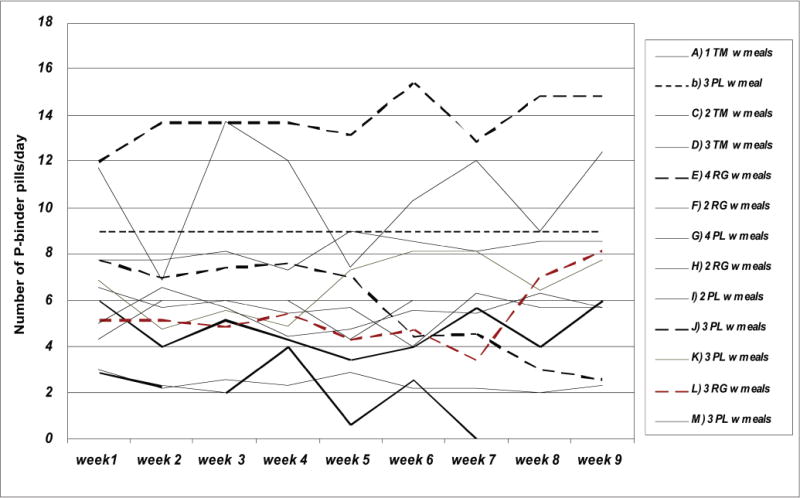
Longitudinal changes in phosphorus binder pill counts, over the entire week, in 13 participating patients.
Table 3.
Baseline and posttrial serum concentrations of phosphorus and albumin and averaged counts of the phosphorus binder pills in each of the 13 participating patients.
| Baseline | Posttrial | Difference | Paired t-test p-value | |
|---|---|---|---|---|
| Serum phosphorus (mg/dl) | 5.58 ± 1.34 | 4.63 ± 1.18 | −0.94 ± 0.91 | 0.003 |
| Serum albumin (g/dl) | 4.02 ± 0.29 | 4.21 ± 0.36 | +0.19 ± −0.24 | 0.014 |
| Daily phosphorus binder pill count | 6.8 ± 2.9 | 6.8 ± 4.0 | +0.0 ± 2.2 | 0.883 |
Phosphorus binder pill data are from week nine of the project.
DISCUSSION
We conducted a pilot dietary intervention in a small group of MHD patients in an outpatient dialysis clinic and found that when MHD patients substituted pasteurised liquid egg whites for meat at one meal per day, serum phosphorus level decreased significantly by 0.9 mg/dl, whereas serum albumin level tended to increase. To our knowledge, this is the first study that examines the effect of an egg white based diet on improving phosphorus control and nutritional status in dialysis patients, and our findings may have important clinical and nutritional implications in the care of CKD patients.
In CKD patients, both the PEW (Kalantar-Zadeh et al. 2003a; Fouque et al. 2008) and the ‘Mineral and Bone Disorders’ (MBD) (Levin et al. 2007; Moe et al. 2007) are common and related to high mortality (Block et al. 2004; Kalantar-Zadeh et al. 2005a; Melamed et al. 2006; Shinaberger et al. 2006; Kalantar-Zadeh et al. 2006). The MBD develops with worsening hyperphosphataemia as a result of inadequate renal phosphorus clearance, leading to increased activation of fibroblast growth factor, FGF-23, and subsequent inhibition of 1-alpha hydroxylation of 25-hydroxy vitamin D, secondary hyperparathyroidism and renal osteodystrophy (Hruska & Teitelbaum 1995; Gupta et al. 2004; Gutierrez et al. 2005). On the other hand, PEW is believed to result from inadequate protein intake (Ikizler 2004) due to anorexia from the uraemic state (Kalantar-Zadeh et al. 2004) and other conditions that restrict oral food ingestion or metabolism in MHD patients, and is usually associated with hypoalbuminaemia, chronic inflammation, sarcopenia and weight loss (Kalantar-Zadeh et al. 2003a; Ikizler 2005). Hence, the restriction of dietary phosphorus intake while increasing dietary protein intake is recommended to MHD patients (Kalantar-Zadeh et al. 2010; Noori et al. 2010d). Nonetheless, dietary prevention of MBD may be at the expense of worsening PEW, and vice versa, since dietary phosphorus restriction may lead to malnutrition, while higher protein intake to improve nutritional status may lead to hyperphosphataemia. This therapeutic conundrum, which is encountered frequently during the medical care of dialysis patients, has confused both patients and healthcare providers (Martin & Reams 2003). Many nephrologists and dietitians are not sure whether they should reinforce dietary restrictions in their MHD patients (which often includes significant protein restriction) in order to achieve a lowered serum phosphorus within the recommended target zone (Martin & Reams 2003) or whether they should liberalise or encourage protein intake in order to improve nutritional status and prevent hypoalbuminaemia and PEW (which is associated with elevated death risk). Indeed, the lower mortality in African American dialysis patients may be related to their higher protein intake at the expense of worsening hyperphosphatemia (Kalantar-Zadeh et al. 2007b). Indeed, a recent study showed that the risk of controlling serum phosphorus by imposing dietary protein restriction may outweigh its benefit in MHD patients leading to increased death risk (Shinaberger et al. 2008). Hence, our finding may suggest that an egg white based diet is an appropriate reconciliation between high-protein diet to improve PEW and low-phosphorus diet to improve MBD.
It is important to note that a reduced dietary protein intake may also be the result of poor appetite that happens commonly in MHD patients, for instance as a result of chronic inflammation, independent of restricting or liberalising dietary intake (Kalantar-Zadeh et al. 2004; Carrero et al. 2007). Indeed, very low serum phosphorus (<3.5 mg/dl) is a strong correlate of death risk (Kalantar-Zadeh et al. 2006), which may be due to the exceptionally strong effect of PEW, since very low phosphorus levels are usually observed in patients with inadequate food intake. Nevertheless, a rise in serum phosphorus over time is consistently associated with increased mortality (Kalantar-Zadeh et al. 2006), whereas higher protein intake is associated with lower mortality (Shinaberger et al. 2006), probably by virtue of improving nutritional status (Kovesdy & Kalantar-Zadeh 2009). In a recent study, higher dietary phosphorus intake or frequent ingestion of food with a higher phosphorus to protein ratio was associated with increased death risk (Noori et al. 2010b). Hence, our study may have major clinical implications because we showed that meals that include egg whites can improve serum albumin and nutritional status without dietary phosphorus burden.
In a non-vegetarian western diet, over one-half of the dietary phosphorus load originates from animal proteins (Pecoits-Filho 2007). The main food sources of phosphorus are meat, poultry, fish, eggs and dairy products (National Research Council, Food and Nutrition Board 1989). Digestibility of phosphorus from animal-derived foods is higher than that of plant-based proteins (Noori et al. 2010d). Different sources of animal proteins contain different proportion of phosphorus. One large whole egg contains 6 g of protein and 86 mg of phosphorus, i.e. a phosphorus-to-protein ratio of 14.3 mg/g, whereas egg white from one large egg (4 g of protein) contains only 5 mg of phosphorus (phosphorus protein ratio: 1.2 mg/g), indicating that the bulk of egg phosphorus is in the egg yolk (Kovesdy et al. 2010b). Poultry (such as chicken and turkey) contains less phosphorus than red meat (such as beef and veal) and fish. Each 100 g of salmon contains 21 g of protein and 282 mg phosphorus (phosphorus-protein ratio of 13.4 mg/g) (Kovesdy et al. 2010b). Moreover, meat and dairy products are frequently ‘enhanced’ by the addition of phosphate additives (Sullivan et al. 2009; Kalantar-Zadeh et al. 2010), which may markedly increase the total phosphorus content. Similarly, different types of cheese may contain from <100 mg to almost 1,000 mg per serving of combined organic and inorganic phosphorus based on the type of the cheese and its method of processing (Murphy-Gutekunst 2007; Kalantar-Zadeh et al. 2010).
Egg white based diets including the pasteurised liquid egg whites are high in protein and low in phosphorus with a phosphorus-to-protein ratio of <2 mg/g. This ratio is the lowest among virtually all natural sources of high-value dietary protein (Kalantar-Zadeh et al. 2010). Furthermore, the liquid egg whites have no phosphorus additives, which are usually easily (80–100%) absorbable inorganic phosphorus as compared to lower phosphorus absorption in protein from animal sources such as meat, fish and poultry (40–60%) and plants (<40%) (Noori et al. 2010d). Egg white based products are appropriate for dialysis patients and offer a large variety of cooking and preparation styles including smoothies, veggie casseroles and egg salads. In particular, the smoothie recipes may be more nutritionally compatible with the renal diet than commercial supplements. In our opinion, liquid egg whites are an improvement over sports protein bars which can be used as protein and energy supplements but which are problematic for patients with dentures and which do not allow alternate preparation methods (Meade 2007). In another study, egg albumin-powder supplementation successfully increased serum albumin in peritoneal dialysis patients but unlike liquid egg whites, the egg albumin powder did not decrease serum phosphorus levels that may be related to phosphorus-based additives (Gonzalez-Espinoza et al. 2005).
Our pilot project should be qualified for its small sample size, which prevents conclusive statements about the improved phosphorus control with egg whites, even though patient adherence and positive responses were promising. The intervention assignment was not random, there was no control arm, the dietary intervention was rather short (six weeks) and patients and providers were not blinded. A more ideal trial format would include parallel groups with wash-out period followed by cross-over. Nevertheless, the trends in albumin, phosphorus and phosphorus binder pills were observed closely. Changes in serum albumin were likely minimal given the average well-nourished status of the study population with eight of 13 patients having a baseline serum albumin ≥4.0 g/dl. Furthermore, it is likely that at least some drop in serum phosphorus is attributable to the reduction in dietary meat intake during the intervention. Despite all the aforementioned limitations, we believe that our findings are encouraging and should be further examined in larger studies.
In conclusion, we found that dietary egg white in the form of eight ounces of liquid pasteurised egg white product with 24 g of protein was well tolerated as a protein substitute for one meal a day in MHD patients and effectively lowered serum phosphorus by 0.9 mg/dl over six weeks while serum albumin increased. We also felt encouraged by the observation that the egg white product was pleasing and palatable for most patients. Since it is plausible that the risk of controlling serum phosphorus by imposing dietary protein restriction may outweigh its benefit in MHD patients, foods with extremely low phosphorus to protein ratio such as egg whites may provide the most appropriate diet for CKD patients. The persistent association between low-protein intake and worse survival may indicate that methods other than restricting protein intake should be sought to restrict dietary phosphorus intake. Egg white products may be a means to that end, while more attention to nonprotein sources of phosphorus such as food additives or highly processed convenience foods is warranted (Uribarri 2007). Because increased protein intake with a concurrent decline in serum phosphorus appears to be associated with the lowest mortality, egg white based diet may be helpful. In any event, our results underscore the need for well-designed controlled trials to examine the role of egg whites and meals during haemodialysis treatment to control phosphorus and improve nutritional status in MHD patients.
Acknowledgments
This study was supported by research grants from DaVita Clinical Research, a General Clinical Research Centre (GCRC) grant number M01-RR00425 from the National Centres for Research Resources, National Institutes of Health and a philanthropist grant from Mr. Harold Simmons.
The authors would like to thank DaVita Clinical Research for funding this research and the team and patients of DaVita Carroll County Dialysis who assisted in this study. Special thanks to Dr. Robert Levy for his encouragement and intellectual support for this research. These data were presented in part as a poster at the Spring Clinical Meeting of the National Kidney Foundation, 2008.
Biographies

Lynn Taylor is a Registered Dietitian with Davita Carroll County Dialysis in Westminster Maryland. She is a member of The American Dietetic Association and The Renal Practice Group. She has coauthored in perspectives: The Journal of the Council of Nephrology Social Workers. She supports chronic kidney disease education by recipe and article submission for newsletters and websites.

Kamyar Kalantar-Zadeh is the Associate Professor of Medicine, Pediatrics & Epidemiology at UCLA David Geffen Schools of Medicine and Public Health. He has received numerous awards including those from National Institutes of Health and National Kidney Foundation. He is a member of the editorial board of several renal journals including AJKD, CJASN, AJN, ACKD, IUN, CN, JREN. He has written more than 150 research papers and lectures frequently on nontraditional cardiovascular risk factors in patients with CKD such as malnutrition, inflammation-cachexia syndrome, iron and anaemia and bone and mineral disorders.
Footnotes
CONFLICT OF INTEREST
None declared.
References
- Achinger SG, Ayus JC. Left ventricular hypertrophy: is hyperphosphatemia among dialysis patients a risk factor? Journal of American Society of Nephrology. 2006;17:S255–S261. doi: 10.1681/ASN.2006080923. [DOI] [PubMed] [Google Scholar]
- Block GA, Hulbert-Shearon TE, Levin NW, et al. Association of serum phosphorus and calcium x phosphate product with mortality risk in chronic hemodialysis patients: a national study. American Journal of Kidney Diseases. 1998;31:607–617. doi: 10.1053/ajkd.1998.v31.pm9531176. [DOI] [PubMed] [Google Scholar]
- Block GA, Klassen PS, Lazarus JM, et al. Mineral metabolism, mortality, and morbidity in maintenance hemodialysis. Journal of American Society of Nephrology. 2004;15:2208–2218. doi: 10.1097/01.ASN.0000133041.27682.A2. [DOI] [PubMed] [Google Scholar]
- Carrero JJ, Qureshi AR, Axelsson J, et al. Comparison of nutritional and inflammatory markers in dialysis patients with reduced appetite. The American Journal of Clinical Nutrition. 2007;85:695–701. doi: 10.1093/ajcn/85.3.695. [DOI] [PubMed] [Google Scholar]
- Fouque D, Kalantar-Zadeh K, Kopple J, et al. A proposed nomenclature and diagnostic criteria for protein-energy wasting in acute and chronic kidney disease. Kidney International. 2008;73:391–398. doi: 10.1038/sj.ki.5002585. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Espinoza L, Gutierrez-Chavez J, del Campo FM, et al. Randomized, open label, controlled clinical trial of oral administration of an egg albumin-based protein supplement to patients on continuous ambulatory peritoneal dialysis. Peritoneal Dialysis International. 2005;25:173–180. [PubMed] [Google Scholar]
- Gupta A, Winer K, Econs MJ, et al. FGF-23 is elevated by chronic hyperphosphatemia. The Journal of Clinical Endocrinology and Metabolism. 2004;89:4489–4492. doi: 10.1210/jc.2004-0724. [DOI] [PubMed] [Google Scholar]
- Gutierrez O, Isakova T, Rhee E, et al. Fibroblast growth factor-23 mitigates hyperphosphatemia but accentuates calcitriol deficiency in chronic kidney disease. Journal of American Society of Nephrology. 2005;16:2205–2215. doi: 10.1681/ASN.2005010052. [DOI] [PubMed] [Google Scholar]
- Hernandez JD, Wesseling K, Salusky IB. Role of parathyroid hormone and therapy with active vitamin D sterols in renal osteodystrophy. Seminars in Dialysis. 2005;18:290–295. doi: 10.1111/j.1525-139X.2005.18404.x. [DOI] [PubMed] [Google Scholar]
- Hruska KA, Teitelbaum SL. Renal osteodystrophy. The New England Journal of Medicine. 1995;333:166–174. doi: 10.1056/NEJM199507203330307. [DOI] [PubMed] [Google Scholar]
- Hruska KA, Saab G, Mathew S, et al. Renal osteodystrophy, phosphate homeostasis, and vascular calcification. Seminars in Dialysis. 2007;20:309–315. doi: 10.1111/j.1525-139X.2007.00300.x. [DOI] [PubMed] [Google Scholar]
- Ikizler TA. Protein and energy: recommended intake and nutrient supplementation in chronic dialysis patients. Seminars in Dialysis. 2004;17:471–478. doi: 10.1111/j.0894-0959.2004.17608.x. [DOI] [PubMed] [Google Scholar]
- Ikizler TA. Effects of hemodialysis on protein metabolism. Journal of Renal Nutrition. 2005;15:39–43. doi: 10.1053/j.jrn.2004.09.019. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Kopple JD, Block G, et al. Association among SF36 quality of life measures and nutrition, hospitalization, and mortality in hemodialysis. Journal of American Society of Nephrology. 2001;12:2797–2806. doi: 10.1681/ASN.V12122797. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Ikizler TA, Block G, et al. Malnutrition-inflammation complex syndrome in dialysis patients: causes and consequences. American Journal of Kidney Diseases. 2003a;42:864–881. doi: 10.1016/j.ajkd.2003.07.016. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Block G, Humphreys MH, et al. Reverse epidemiology of cardiovascular risk factors in maintenance dialysis patients. Kidney International. 2003b;63:793–808. doi: 10.1046/j.1523-1755.2003.00803.x. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Block G, McAllister CJ, et al. Appetite and inflammation, nutrition, anemia and clinical outcome in hemodialysis patients. The American Journal of Clinical Nutrition. 2004;80:299–307. doi: 10.1093/ajcn/80.2.299. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Kilpatrick RD, Kuwae N, et al. Revisiting mortality predictability of serum albumin in the dialysis population: time dependency, longitudinal changes and population-attributable fraction. Nephrology, Dialysis, Transplantation. 2005a;20:1880–1888. doi: 10.1093/ndt/gfh941. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Braglia A, Chow J, et al. An anti-inflammatory and antioxidant nutritional supplement for hypoalbuminemic hemodialysis patients: a pilot/feasibility study. Journal of Renal Nutrition 1. 2005b;5:318–331. doi: 10.1016/j.jrn.2005.04.004. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Kuwae N, Regidor DL, et al. Survival predictability of time-varying indicators of bone disease in maintenance hemodialysis patients. Kidney International. 2006b;70:771–780. doi: 10.1038/sj.ki.5001514. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Horwich TB, Oreopoulos A, et al. Risk factor paradox in wasting diseases. Current Opinion in Clinical Nutrition and Metabolic Care. 2007a;10:433–442. doi: 10.1097/MCO.0b013e3281a30594. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Kovesdy CP, Derose SF, et al. Racial and survival paradoxes in chronic kidney disease. Nature Clinical Practice. Nephrology. 2007b;3:493–506. doi: 10.1038/ncpneph0570. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Regidor DL, Kovesdy CP, et al. Fluid retention is associated with cardiovascular mortality in patients undergoing long-term hemodialysis. Circulation. 2009;119:671–679. doi: 10.1161/CIRCULATIONAHA.108.807362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Gutekunst L, Mehrotra R, et al. Understanding sources of dietary phosphorus in the treatment of patients with chronic kidney disease. Clinical Journal of the American Society of Nephrology. 2010;5:519–530. doi: 10.2215/CJN.06080809. [DOI] [PubMed] [Google Scholar]
- Kalantar-Zadeh K, Unruh M. Health related quality of life in patients with chronic kidney disease. International Urology and Nephrology. 2005;37:367–378. doi: 10.1007/s11255-004-0012-4. [DOI] [PubMed] [Google Scholar]
- Kopple JD. The National Kidney Foundation K/DOQI clinical practice guidelines for dietary protein intake for chronic dialysis patients. American Journal of Kidney Diseases. 2001;38:S68–S73. doi: 10.1053/ajkd.2001.27578. [DOI] [PubMed] [Google Scholar]
- Kovesdy CP, Kalantar-Zadeh K. Bone and mineral disorders in pre-dialysis CKD. International Urology and Nephrology. 2008;40:427–440. doi: 10.1007/s11255-008-9346-7. [DOI] [PubMed] [Google Scholar]
- Kovesdy CP, Kalantar-Zadeh K. Why is protein-energy wasting associated with mortality in chronic kidney disease? Seminars in Nephrology. 2009;29:3–14. doi: 10.1016/j.semnephrol.2008.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovesdy CP, George SM, Anderson JE, et al. Outcome predictability of biomarkers of protein-energy wasting and inflammation in moderate and advanced chronic kidney disease. The American Journal of Clinical Nutrition. 2009;90:407–414. doi: 10.3945/ajcn.2008.27390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovesdy CP, Anderson JE, Kalantar-Zadeh K. Outcomes associated with serum phosphorus level in males with non-dialysis dependent chronic kidney disease. Clinical Nephrology. 2010a;73:268–275. doi: 10.5414/cnp73268. [DOI] [PubMed] [Google Scholar]
- Kovesdy CP, Shinaberger CS, Kalantar-Zadeh K. Epidemiology of dietary nutrient intake in ESRD. Seminars in Dialysis. 2010b;23(4):353–358. doi: 10.1111/j.1525-139X.2010.00745.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ling PR, Smith RJ, Kie S, et al. Effects of protein malnutrition on IL-6-mediated signaling in the liver and the systemic acutephase response in rats. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 2004;287:R801–R808. doi: 10.1152/ajpregu.00715.2003. [DOI] [PubMed] [Google Scholar]
- Levin A, Bakris GL, Molitch M, et al. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney International. 2007;71:31–38. doi: 10.1038/sj.ki.5002009. [DOI] [PubMed] [Google Scholar]
- Martin CJ, Reams SM. The renal dietitian’s role in managing hyperphosphatemia and secondary hyperparathyroidism in dialysis patients: a national survey. Journal of Renal Nutrition. 2003;13:133–136. doi: 10.1053/jren.2003.50015. [DOI] [PubMed] [Google Scholar]
- Meade A. Protein supplementation with sports protein bars in renal patients. Journal of Renal Nutrition. 2007;17:214–217. doi: 10.1053/j.jrn.2006.12.006. [DOI] [PubMed] [Google Scholar]
- Melamed ML, Eustace JA, Plantinga L, et al. Changes in serum calcium, phosphate, and PTH and the risk of death in incident dialysis patients: a longitudinal study. Kidney International. 2006;70:351–357. doi: 10.1038/sj.ki.5001542. [DOI] [PubMed] [Google Scholar]
- Moe S, Drueke T, Cunningham J, et al. Definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO) Kidney International. 2006;69:1945–1953. doi: 10.1038/sj.ki.5000414. [DOI] [PubMed] [Google Scholar]
- Moe SM, Drueke T, Lameire N, et al. Chronic kidney disease-mineral-bone disorder: a new paradigm. Advances in Chronic Kidney Disease. 2007;14:3–12. doi: 10.1053/j.ackd.2006.10.005. [DOI] [PubMed] [Google Scholar]
- Murphy-Gutekunst L. Hidden phosphorus: where do we go from here? Journal of Renal Nutrition. 2007;17:e31–e6. [Google Scholar]
- National Research Council, Food and Nutrition Board. Recommended Dietary Allowances. 10th. Washington, DC: National Academy Press; 1989. pp. 184–187. [Google Scholar]
- Noori N, Kalantar-Zadeh K, Kovesdy CP, et al. Dietary potassium intake and mortality in long-term hemodialysis patients. American Journal of Kidney Diseases. 2010a;56(2):338–347. doi: 10.1053/j.ajkd.2010.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noori N, Kalantar-Zadeh K, Kovesdy CP, et al. Association of dietary phosphorus intake and phosphorus to protein ratio with mortality in hemodialysis patients. Clinical Journal of the American Society of Nephrology. 2010b;5:683–692. doi: 10.2215/CJN.08601209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noori N, Kopple JD, Kovesdy CP, et al. Mid-arm muscle circumference and quality of life and survival in maintenance hemodialysis patients. Clin J Am Soc Nephrol. 2010c doi: 10.2215/CJN.02080310. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noori N, Sims JJ, Kopple JD, et al. Organic and inorganic dietary phosphorus and its management in chronic kidney disease. Iranian Journal of Kidney Diseases. 2010d;4:89–100. [PubMed] [Google Scholar]
- Pecoits-Filho R. Dietary protein intake and kidney disease in Western diet. Contributions to Nephrology. 2007;155:102–112. doi: 10.1159/000101003. [DOI] [PubMed] [Google Scholar]
- Rambod M, Kovesdy CP, Bross R, et al. Association of serum prealbumin and its changes over time with clinical outcomes and survival in patients receiving hemodialysis. The American Journal of Clinical Nutrition. 2008;88:1485–1494. doi: 10.3945/ajcn.2008.25906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rambod M, Bross R, Zitterkoph J, et al. Association of Malnutrition-Inflammation Score with quality of life and mortality in hemodialysis patients: a 5-year prospective cohort study. American Journal of Kidney Diseases. 2009;53:298–309. doi: 10.1053/j.ajkd.2008.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shantouf RS, Budoff MJ, Ahmadi N, et al. Total and individual coronary artery calcium scores as independent predictors of mortality in hemodialysis patients. American Journal of Nephrology. 2010;31:419–425. doi: 10.1159/000294405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinaberger CS, Kilpatrick RD, Regidor DL, et al. Longitudinal associations between dietary protein intake and survival in hemodialysis patients. American Journal of Kidney Diseases. 2006;48:37–49. doi: 10.1053/j.ajkd.2006.03.049. [DOI] [PubMed] [Google Scholar]
- Shinaberger CS, Greenland S, Kopple JD, et al. Is controlling phosphorus by decreasing dietary protein intake beneficial or harmful in persons with chronic kidney disease? The American Journal of Clinical Nutrition. 2008;88:1511–1518. doi: 10.3945/ajcn.2008.26665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan C, Sayre SS, Leon JB, et al. Effect of food additives on hyperphosphatemia among patients with end-stage renal disease: a randomized controlled trial. JAMA. 2009;301:629–635. doi: 10.1001/jama.2009.96. [DOI] [PubMed] [Google Scholar]
- Uribarri J. Phosphorus homeostasis in normal health and in chronic kidney disease patients with special emphasis on dietary phosphorus intake. Seminars in Dialysis. 2007;20:295–301. doi: 10.1111/j.1525-139X.2007.00309.x. [DOI] [PubMed] [Google Scholar]


