Abstract
Craniosynostosis, the premature fusion of one or more cranial sutures of the skull, provides a paradigm for investigating the interplay of genetic and environmental factors leading to malformation. Over the past 20 years molecular genetic techniques have provided a new approach to dissect the underlying causes; success has mostly come from investigation of clinical samples, and recent advances in high-throughput DNA sequencing have dramatically enhanced the study of the human as the preferred “model organism.” In parallel, however, we need a pathogenetic classification to describe the pathways and processes that lead to cranial suture fusion. Given the prenatal onset of most craniosynostosis, investigation of mechanisms requires more conventional model organisms; principally the mouse, because of similarities in cranial suture development. We present a framework for classifying genetic causes of craniosynostosis based on current understanding of cranial suture biology and molecular and developmental pathogenesis. Of note, few pathologies result from complete loss of gene function. Instead, biochemical mechanisms involving haploinsufficiency, dominant gain-of-function and recessive hypomorphic mutations, and an unusual X-linked cellular interference process have all been implicated. Although few of the genes involved could have been predicted based on expression patterns alone (because the genes play much wider roles in embryonic development or cellular homeostasis), we argue that they fit into a limited number of functional modules active at different stages of cranial suture development. This provides a useful approach both when defining the potential role of new candidate genes in craniosynostosis and, potentially, for devising pharmacological approaches to therapy.
Main Text
Introduction
Cranial sutures, superficially simple fibrocellular structures separating the rigid plates of the skull bones, are wonderfully subtle in executing their roles. Mature sutures are bridged by fibers1 that unite the bone fronts and resist deformation in both tension and compression. Their principal function is to enable the growth of the skull in coordination with expansion of the developing brain,2 which occurs particularly rapidly in humans and during fetal and infant life; the intracranial pressure produces quasi-static tensile strains, which could act either directly on the suture or indirectly through mechanotransduction by the dura mater, the tough membrane that adheres to the inner surface of the calvaria (skull vault) and separates it from the brain.3 In addition, sutures permit deformation of the skull during birth, absorb cyclic loading during mastication and locomotion, and act as shock absorbers against external impacts.4
Although cranial sutures start off as simple lines of demarcation between developing bones, they become increasingly interdigitated with age, a feature that is more marked on the external (ectocranial) surface.5 These meandering patterns can be described in terms of fractal geometry, with the fractal dimension increasing with age. Mathematical attempts to account for this behavior have employed reaction-diffusion models incorporating diffusible factors, positive and negative feedback loops, mechanical strain, and time-dependent processes.1,5 The explanatory success of these theoretical studies captures the underlying idea that growth at sutures is likely to involve nested sets of cellular signaling pathways controlled by secretion of paracrine factors and responsive to mechanical strain.
Failure of the mechanisms that maintain suture patency leads to craniosynostosis, the premature fusion of one or more of the cranial sutures. This occurs in about 1 in 2,250 children6,7 and usually becomes apparent between the last third of pregnancy and the end of the first year of life. The fusion of a suture abolishes further growth of the abutting bones in a direction perpendicular to the suture. As a consequence, continued enlargement of the brain promotes compensatory overgrowth at other sutures, leading to progressive distortion in the skull shape.8 Multiple complications can arise because of raised intracranial pressure, facial deformities affecting vision, breathing, and dentition, and other features such as hearing loss or intellectual disability that might be caused by the underlying gene defect or alternatively might occur secondary to craniosynostosis. Surgery to remodel the skull and create extra volume for the brain, with the secondary aim to improve psychosocial adjustment, is currently the mainstay of management.9–11 No pharmacological interventions are currently validated for prevention of suture fusion.
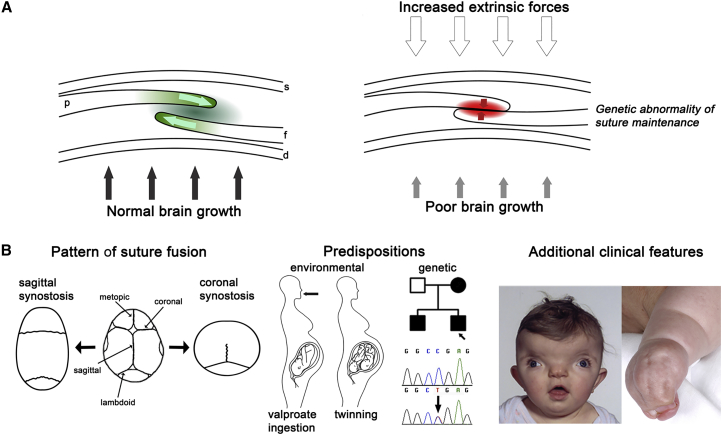
Broadly speaking, three interacting factors predispose to the abnormal fusion of a suture (Figure 1A). Least well understood is the effect of mechanical force (strain) transmitted by the growing brain to maintain suture patency. The relationship between brain growth and failure of suture function is complex, because although microcephaly is a recognized risk factor for craniosynostosis (see GeneReviews in Web Resources), most individuals with microcephaly do not develop this complication. Along similar lines, craniosynostosis has been documented in association with many chromosomal abnormalities,16 but usually only a minority of those with similar chromosome imbalances actually develop the condition, suggesting non-specific mechanisms through poor brain growth and/or transduction of strain.17 The second factor, the intrinsic property of the suture itself, is the focus of this review. Finally, extrinsic forces acting on the skull, especially during fetal life, might frequently precipitate craniosynostosis, especially in the uncomplicated non-syndromic, single-suture-fusion cases. Epidemiological data consistent with a contribution from fetal head constraint include positive associations of craniosynostosis with primiparity, multiple pregnancy, prematurity, and high birth weight.7,18 Compressive strain has been shown experimentally to increase osteogenesis at the suture (reviewed by Herring3). In addition, there is some in vivo support for the role of head constraint in craniosynostosis from experimental manipulations performed in mice.19
Figure 1.
Overview of Clinical Causes and Classification of Craniosynostosis
(A) Cross-section of coronal suture showing the developing parietal bone (p) overlying the frontal bone (f). Internally, the dura mater (d) separates the calvaria from the brain; skin (s) is external. Left, normal growth of the skull vault is regulated by a delicate balance of proliferation and differentiation occurring within the suture (green shading) co-ordinated with enlargement of the underlying brain (black arrows). Right, in craniosynostosis this balance has been disturbed by excessive external force on the skull, usually during pregnancy (unfilled arrows), inefficient transduction of stretch from the growing brain (gray arrows), or intrinsic abnormality of signaling within the suture itself (red shading).
(B) Approaches to clinical classification. Left: view of skull from above (front at top). Normal skull with major vault sutures identified is central. To either side, the examples show skull shapes resulting from sagittal synostosis (left) and bilateral coronal synostosis (right). Note, the metopic suture closes physiologically at the age of 3–9 months.12 Center: the clinical history can reveal possible environmental predispositions such as teratogen exposure (most commonly maternal treatment with the anticonvulsant sodium valproate13) or twinning (intrauterine constraint);14 affected relatives with craniosynostosis (filled symbols in pedigree) suggest a likely genetic cause, which can be confirmed by diagnostic genetic testing. Right: clinical examination can reveal facial dysmorphic features such as hypertelorism (wide spaced eyes) and grooved nasal tip, suggesting craniofrontonasal syndrome (left), or other physical features such as syndactyly characteristic of Apert syndrome (right). Clinical photographs reproduced, with permission, from Johnson and Wilkie9 and Twigg et al.15
As might be anticipated given this plethora of potential causes, craniosynostosis is extremely heterogeneous in its presentation. Three main axes of clinical classification exist (Figure 1B), which enumerate (1) the pattern of suture fusion and consequent skull shape, (2) the occurrence of environmental or genetic predispositions, and (3) the presence of additional clinical features (such as facial dysmorphism, limb anomalies, or learning disability) suggestive of a syndrome.9,10,20 Fusion of multiple sutures, positive family history, and additional syndromic features all suggest an underlying genetic predisposition; in some cases, accompanying dysmorphic features are caused by fusion of sutures separating the facial bones. Over the past two decades our understanding of the molecular processes in craniosynostosis, and hence underlying normal suture development, has been transformed by human genetics studies. To date, mutations in 57 genes have been identified as recurrently causing craniosynostosis, and the number of genes is growing rapidly as high-throughput sequencing of exomes (and, increasingly, whole genomes) is applied to the problem. Although further genes, mutations of which are highly penetrant for craniosynostosis, no doubt await discovery, and despite the lag period before animal modeling can be accomplished, distinct modules of pathogenesis are emerging. It is therefore timely to propose a genetic-pathophysiological framework for classifying craniosynostosis, based on integrating knowledge of clinical genetics, suture biology, biochemical studies of mutant molecules, and cellular and developmental observations of abnormal suture formation in mouse disease models.
Monogenic Causes of Craniosynostosis
Starting with the identification in 1993 of an MSX2 (MIM: 123101) point mutation encoding a specific missense amino acid substitution segregating in a 3-generation family with craniosynostosis (MIM: 604757),21 there are presently 57 human genes for which there is reasonable evidence (based on at least two affected individuals with congruent phenotypes) that mutations are causally related to craniosynostosis. Inspection of the complete list (Table S1) suggests that these genes fall into two broad groups. First, those for which mutations of a particular molecular type (based on the encoded variant protein) are frequently (>50% of independent mutations) associated with craniosynostosis, which might therefore be considered a core characteristic of the particular gene/mutation combination; second, those for which the occurrence of craniosynostosis, although probably causally associated, arises in only a minority of cases with the mutation. The latter grouping includes many (such as filaminopathy, hypophosphatasia, mucopolysaccharidoses, osteosclerosis, and pycnodysostosis) that primarily represent perturbations in osteogenesis (for example affecting the balance of osteoblast and osteoclast activity) rather than in the biology of cranial suture development per se. These general osteogenic genes are not considered further. This review focuses on trying to place the first category, comprising the 20 core genes listed in Table 1, into a pathophysiological context, quoting examples from the wider gene set (Table S1) only where they provide additional support for a salient point.
Table 1.
Core Genes for which Specific Types of Mutation Are Associated with Craniosynostosis in More than Half of Affected Individuals
| Gene (MIM#)a | Inheritance Pattern | Clinical Disorder (MIM#) | Prevalence (%)b | Typical Suture Fusion | Major Phenotypic Features | Ref(s) |
|---|---|---|---|---|---|---|
| ASXL1 (612990) | AD (n) | Bohring-Opitz syndrome (605039) | – | metopic | forehead nevus flammeus, ulnar deviation and flexion of wrists and metacarpalophalangeal joints, severe intellectual disability | 22 |
| CDC45 (603465) | AR | – | – | coronal | thin eyebrows, small ears, variable short stature | 23 |
| COLEC11 (612502) | AR | 3MC syndrome 2 (265050) | – | metopic | hypertelorism, blepharoptosis, arched eyebrows, cleft lip/palate, hearing loss, radio-ulnar synostosis, genital and vesicorenal anomalies | 24 |
| EFNB1 (300035) | XLD (male sparing) | craniofrontonasal syndrome (304110) | 0.8 | coronal | hypertelorism, notched nasal tip, chest anomalies, longitudinal splitting of nails; heterozygous females more severely affected than hemizygous males | 15,25 |
| ERF (611888) | AD | ERF-related craniosynostosis (600775) | 1.1 | multisuture | exorbitism, midface hypoplasia, Chiari type I malformation, postnatal onset of craniosynostosis | 26 |
| FGFR1 (136350) | AD | Pfeiffer syndrome (101600) | – | coronal | mild craniofacial features, broad medially deviated thumbs and halluces, cutaneous syndactyly, specific amino acid substitution p.Pro252Arg | 27 |
| FGFR1 | AD (n) | osteoglophonic dysplasia (166250) | – | multisuture | prominent brow ridges, depressed nasal bridge, rhizomelic dwarfism, localized lytic lesions of metaphyses | 28 |
| FGFR2 (176943) | AD (n) | Apert syndrome (101200) | 3.6 | coronal, multisuture | midface hypoplasia, dilated cerebral ventricles, complex syndactyly of the hands and feet | 29 |
| FGFR2 | AD (n) | Beare-Stevenson syndrome (123790) | ∗ | multisuture | choanal atresia, prominent umbilical stump, furrowed scalp/neck skin, acanthosis nigricans in survivors | 30 |
| FGFR2 | AD | Crouzon syndrome (123500) | 2.4 | multisuture, coronal, sagittal | exorbitism, midface hypoplasia, beaked nose (“crouzonoid” facies), clinically normal hands and feet | 31,32 |
| FGFR2 | AD (n) | Pfeiffer syndrome (101600) | 0.8 | multisuture | broad thumbs and halluces; in severe cases, cloverleaf skull, brain anomalies, tracheal sleeve, fused elbows | 33–35 |
| FGFR2 | AD (n) | bent bone dysplasia (614592) | ∗ | coronal | osteopenia, reduced mineralization of the calvaria, bent long bones; perinatal lethal | 36 |
| FGFR3 (134934) | AD | Muenke syndrome (602849) | 4.0 | coronal | defined by specific amino acid substitution p.Pro250Arg; may include sensorineural hearing loss, mild brachydactyly, cone-shaped epiphyses | 37 |
| AD (n) | Crouzon/acanthosis nigricans (612247) | 0.4 | multisuture | crouzonoid facies, choanal stenosis, hydrocephalus, acanthosis nigricans; specific amino acid substitution p.Ala391Glu | 38 | |
| AD (n) | thanatophoric dysplasia II (187601) | ∗ | multisuture | lethal skeletal dysplasia, micromelic limb shortening, straight femora; specific amino acid substitution p.Lys650Glu | 39 | |
| IHH (600726) | AD | Philadelphia craniosynostosis (185900) | – | sagittal | cutaneous and osseous syndactyly | 40 |
| IL11RA (600939) | AR | craniosynostosis and dental anomalies (614188) | – | multisuture | maxillary hypoplasia, delayed tooth eruption, supernumerary teeth, minor digit abnormalities, conductive hearing loss | 41 |
| MEGF8 (604267) | AR | Carpenter syndrome 2 (614796) | – | metopic | hypertelorism, arched eyebrows, lateralization defects, brachydactyly, syndactyly, preaxial polydactyly | 42 |
| MSX2 (123101) | AD | Boston craniosynostosis (604757) | – | sagittal, coronal, multisuture | none diagnostic; syndrome defined by specific amino acid substitutions p.Pro148His, p.Pro148Leu | 21 |
| POR (124015) | AR | Antley-Bixler syndrome (201750) | – | bicoronal, multisuture | choanal stenosis, radio-humeral synostosis, bowed femora, multiple joint contractures, genital abnormalities; abnormal steroidogenesis | 43 |
| RAB23 (606144) | AR | Carpenter syndrome 1 (201000) | – | multisuture | obesity, cardiac defects, polysyndactyly, brachydactyly, genu valgum, hypogenitalism, umbilical hernia, learning disability | 44 |
| RUNX2 (600211) | AD (n) | – | – | multisuture | none diagnostic; syndrome defined by specific gene duplication | 45,46 |
| SKI (164780) | AD (n) | Shprintzen-Goldberg syndrome (182212) | – | sagittal, multisuture | hypertelorism, micrognathia, high arched palate, arachnodactyly, joint contractures, pectus deformity, aortic root aneurysm, mitral valve prolapse, learning disability | 47 |
| TCF12 (600480) | AD | TCF12-related craniosynostosis (615314) | 1.3 | coronal | resembles mild Saethre-Chotzen syndrome; diagnosis defined by presence of mutations in the gene, ∼50% non-penetrance | 48 |
| TWIST1 (601622) | AD | Saethre-Chotzen syndrome (101400) | 3.6 | coronal | low frontal hairline, hypertelorism, eyelid ptosis, downslanting palpebral fissures, blocked tear ducts, small ears with prominent crus helicis | 49,50 |
| WDR35 (613602) | AR | cranioectodermal dysplasia 2 (613610) | – | sagittal | facial dysmorphism, narrow thorax, short long bones, brachydactyly, sparse hair, hypoplastic teeth, cystic kidneys, hepatic fibrosis | 51 |
| ZIC1 (600470) | AD (n) | ZIC1- craniosynostosis | 0.2 | coronal | severe learning disability | 52 |
Abbreviations are as follows: AD, autosomal dominant; AR, autosomal recessive; XLD, X-linked dominant; (n), usually arises by new mutation; ∗ usually lethal at birth.
Information has been extracted from Table S1, which contains further details on mutational category and phenotype and additional references.
The prevalence figures are for percent total craniosynostosis cases with specified mutation, from the cohort attending the Craniofacial Unit, Oxford, born between 1998 and 2008 (n = 531), and surgically treated before end of 2013 (updated from Wilkie et al.17).
Processes in Suture Formation
The cranial vault bones are relatively unusual in arising by intramembranous ossification, without a cartilaginous intermediate,4 and the cranial sutures are key to understanding their development. To formulate our pathophysiological framework, we distinguish five processes in suture formation. These are stem cell specification and migration, lineage commitment, boundary formation and integrity, osteogenic proliferation/differentiation, and resorption/homeostasis. We can use this classification to distinguish events both on a temporal basis (for example, early versus late events) and on an anatomical basis (for example, events primarily affecting undifferentiated cells located centrally in the suture versus more mature peripheral osteogenic cells). These processes are exquisitely coordinated and overlapping in both time and space, so that the distinctions are to some extent artificial, but we think a useful perspective emerges from this analysis. This overview focuses primarily on the coronal suture, which has the most complex biogenesis and is the suture most commonly fused in genetic forms of craniosynostosis.17 The other major vault sutures (metopic, sagittal, lambdoid; Figure 1B) are anatomically more simple, being formed when initially distant bone fronts approximate each other later in embryonic development.4,53
Stem Cell Specification and Migration
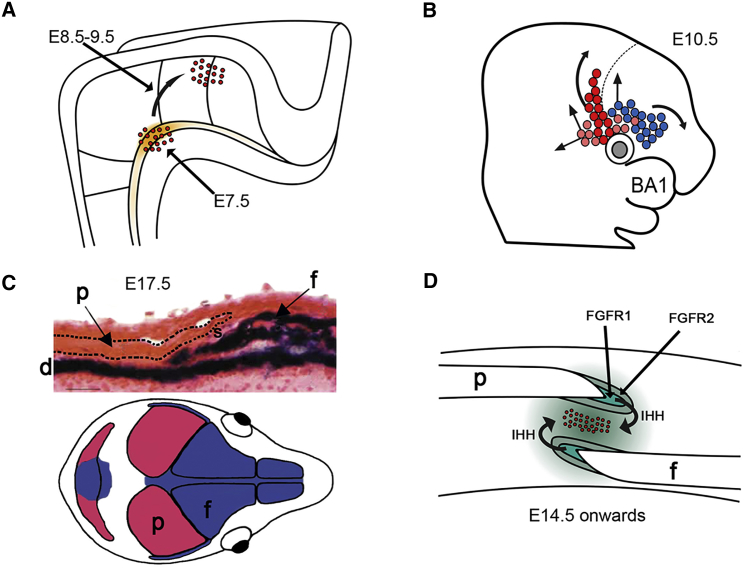
It has long been presumed that sutures contain a population of undifferentiated osteogenic cells with stem cell-like properties,54,55 and this has recently received further experimental support with the demonstration that expression of Gli1 (a classical marker of hedgehog [HH] signaling) probably defines sutural stem cells at postnatal stages; cells emanating from the suture populate the periosteum on both surfaces of the growing bones, and genetic ablation of Gli1-expressing cells in 1-month-old mice caused coronal synostosis within a further month.56 An elegant study (which also exploited Gli1 as the key marker of cell identity) pinpoints that the precursors of these cells originate from cephalic paraxial mesoderm, in the region of the rostral mesencephalon/caudal diencephalon and located immediately adjacent to the neural tube, at embryonic day (E)7.5 in response to sonic hedgehog (SHH) accumulation in the adjacent notochord (Figure 2A).57 Lineage tracing using genetically marked cells shows that this population migrates laterally during E8.5–E9.5 to locate above the developing eye.57 Of note, expression of Gli1 is transient because calvarial structures do not label after E8, indicating that the influence of SHH on these cells is rapidly lost.
Figure 2.
Key Embryological Processes in Coronal Suture Formation
(A) At E7.5, sonic hedgehog secreted by the notochord (orange shading) induces Gli1 expression in adjacent cells of cephalic paraxial mesoderm (red dots). Over the next 48 hr, these cells migrate laterally (curved arrow) to a position above the developing eye.
(B) Supraorbital regulatory center at E10.5, showing cells of mesodermal and neural crest origin (pink/red and blue, respectively). These cells migrate to populate the future coronal suture (red), parietal bone (mesodermal cells, pink), and frontal bone (neural crest, with small contribution from mesoderm). Dashed line indicates the diencephalic-telencephalic boundary. BA1, first branchial arch.
(C) Top: cross-section of coronal suture at E17.5, β-galactosidase staining of Wnt1-Cre/R26R mice to demonstrate neural-crest-derived tissues (dark blue). Note that the frontal bone (f) and underlying dura mater (d) are of neural crest origin, whereas the parietal bone (p; dashed outline) and sutural gap (s) show no blue staining, indicating a mesodermal origin. Bottom: dual origin of the skull bones from neural crest (blue) and cephalic mesoderm (red).
(D) Simplified view of an established coronal suture (E14 onward). For continued patency, a population of undifferentiated stem cells (red dots) must be maintained in the mid-sutural mesenchyme. The proliferation-differentiation balance between these cells and those in the growing margins of the bones is maintained by a hierarchy of paracrine signaling feedback loops, such as those provided by IHH and FGF receptor signaling.
Figure redrawn from original data presented elsewhere.56–61 Part C adapted from Jiang et al.58 with permission.
Lineage Commitment
During a critical period from E9.5 to E11.5, the cells above the developing eye pattern the future coronal suture. Deckelbaum et al.57 have proposed the term “supraorbital regulatory center” for this region, in recognition of its role as an organizing center. Put simply, groups of cells with osteogenic potential, originating from neural crest (future frontal bone) and mesoderm (future parietal bone), are separated by the undifferentiated stem cell population originating from a localized section of paraxial mesoderm (Figure 2B). During E11.5–E13.5, and co-ordinated with growth of the underlying brain, cells from the supraorbital region extend apically, centered above the diencephalic-telencephalic boundary, to overlay the surface of the rapidly growing brain.57,62,63 Descendants of these cells can still be identified in the mid-sutural mesenchyme of the definitive coronal suture at birth (P0), demonstrating that they make a permanent contribution to the population of undifferentiated cells.57 A separate population of descendants becomes integrated into the parietal bone mesenchyme and adopts an osteogenic fate; the future frontal bone originates mostly from cells of neural crest origin (next section).
Key transcription factors involved in orchestrating this early lineage commitment are EN1 (engrailed 1), MSX2, and TWIST1, all of which are present in the supraorbital regulatory center at E11.57 Recently, individuals with severe bilateral coronal synostosis were described with heterozygous truncations or missense substitution of another transcription factor, ZIC1.23,52 Constructs containing these ZIC1 mutations caused altered or enhanced expression of a target gene, engrailed-2, in a Xenopus embryo assay, consistent with a gain of function.52 Given the epistatic relationship of ZIC homologs (odd-paired in Drosophila) upstream of engrailed in both Drosophila64 and Xenopus,65,66 and the expression of murine Zic1 in the territory corresponding to supraorbital regulatory center,52ZIC1 (MIM: 600470) mutations are likely to disrupt early lineage commitment in the coronal suture.
A complex and incompletely understood set of positive and negative feedback loops links these transcription factors to the major signaling pathways required to commit cells to an osteogenic fate, including bone morphogenetic proteins (BMPs), wingless-related family members (WNTs), and fibroblast growth factors (FGFs); there is evidence for activity of each of these pathways at this time (reviewed in Mishina and Snider67). However, expression studies do not enable disentanglement of the exact sequence of events or signaling hierarchies involved; neither do classical knockout approaches answer the question, because the multiple roles of these pathways in organogenesis at earlier stages of embryonic development tend to lead to lethality before the cranial sutures are established.67 The availability of suitable drivers for suture expression, together with temporal cell labeling and sorting, should enable considerable progress in teasing apart these processes over the coming decade. Within the next 24–48 hr of development (E12.0–E13.5), the expression of master regulators of osteogenic differentiation Runx2 and Sp7 is initiated.
Boundary Formation and Integrity
Coordinated with the processes described above, guidance cues are required to ensure that cells migrate along the correct path. Of particular significance for the coronal suture is that it lies at the boundary between tissues with distinct embryological origins: trigeminal neural crest (frontal bone) and cephalic mesoderm (parietal bone) (reviewed in Morriss-Kay and Wilkie53). Lineage tracing using the Wnt1-Cre/R26R system to label cells of neural crest origin and their progeny shows that the coronal suture itself arises from mesoderm, and this was confirmed using a reciprocal mesodermal driver, Mesp1-Cre.58,62 The neural crest/mesoderm boundary can be identified from E9.5, and so includes the critical period during which the definitive coronal sutures are forming. The characteristic overlapping of the frontal bone by the parietal bone, forming the oblique cross-section of the coronal suture (Figure 2C), can be understood from the expansion of the underlying cerebral hemispheres toward the hindbrain, taking the neural crest underneath the mesoderm.58 The dura mater underlying both bones is of neural crest origin (Figure 2C). Interestingly, the other major sutures have different tissue origins and relationships: the metopic suture uniquely forms within neural crest and the sagittal and lambdoid sutures, like the coronal, have dual neural crest/mesodermal origins, but unlike the coronal, this is not the case along their entire length (Figure 2C).53 For this reason, in the subset of cases of metopic synostosis that are associated with neurocognitive abnormalities and/or dysmorphic features, a disturbance of neural crest development is the likely mechanism.68
Although it was originally thought that the coronal suture boundary did not permit migration of sutural cells either into or from the neural crest, evidence has emerged that the barrier is in fact unidirectional, in that cells of neural crest origin cannot normally cross into the suture, but the reverse does not apply.57 This latter conclusion is consistent with the evidence from Zhao et al.56 that the coronal suture contains stem cells; the progeny of these cells can be expected to fuel the growth of both the adjacent bones, parietal and frontal (Figures 2B and 2D). Molecules thought to be important in the maintenance of the suture boundary include the transcription factors EN1, TWIST1, and MSX2 (previous section) and members of the EPHRIN/EPH receptor and JAGGED/NOTCH families; all these molecules have been implicated in tissue boundary formation in multiple other contexts.63,69,70
Osteogenic Proliferation and Differentiation
The importance of SHH in the earliest stages of coronal suture development was described above. Gli1, a general marker of HH signaling, is also expressed in the undifferentiated mesenchyme of established postnatal sutures;56 however, in this context the paralogous molecule Indian hedgehog (IHH) is the instructive ligand. Mice lacking IHH (Ihh−/−) have reduced sizes of the developing frontal and parietal bones, reduced Bmp2/Bmp4 expression, and correspondingly widened midline sutures at E15.5; this phenotype is thought to result from a differentiation defect, because proliferation was found to be unaffected.56,71 Secretion of IHH by differentiating cells is proposed to maintain the recruitment of undifferentiated osteoprogenitors from mid-sutural mesenchyme56 (Figure 2D). For calvarial expansion to occur, this differentiation process must be exquisitely tuned to ongoing cell proliferation.55 Notably, the observation that craniosynostosis is frequently associated with defects in certain key cell division genes (discussed later) points to the need for rapid mitotic turnover in the cranial sutures. Twist1, expressed in mid-sutural mesenchyme in the established coronal suture (E16),59 is directly antagonistic to RUNX2, potentially providing an important mechanism to prevent the mid-sutural cells from undergoing osteogenesis.72 TWIST1 also has inhibitory activity on levels of bone sialoprotein and osteocalcin, two of the downstream targets of RUNX2: this requires heterodimerization with a type I basic-helix-loop-helix partner.73 In vivo, the strong epistatic genetic interaction between Twist1 and Tcf12 in the murine coronal suture shows that TCF12/HEB is the key partner protein.48
Apart from the importance of maintaining the undifferentiated fate of mid-sutural stem cells, a second key element of cranial vault growth is provided by positive differentiation signals emanating from osteoid, the collagenous unmineralized matrix produced by osteoblasts along the expanding osteogenic fronts (reviewed in Morriss-Kay and Wilkie53). Redundant signaling by several fibroblast growth factors (there is evidence to implicate particularly FGF2, FGF9, FGF10, and FGF18) is crucial to orchestrating this process.60,74–80 The receptor Fgfr2 is expressed in the rapidly proliferating osteoprogenitor cells, whereas Fgfr1 is associated with a more differentiated state (Figure 2D); increased FGF-signaling flux drives a switch from Fgfr2 to Fgfr1 expression, associated with the onset of osteogenic differentiation.60,61 The dura mater underlying the sutures also provides a source of growth factors including FGF2, BMP4, and TGFβ15 (reviewed in Levi et al.81).
As is the case for cartilaginous bones, the transcription factor Runx2 represents a well-established positive regulator of commitment to terminal osteogenic differentiation. However, for reasons that remain poorly understood, bones undergoing intramembraneous ossification (such as the skull vault) are dependent on a higher RUNX2 dosage than those undergoing endochondral ossification, so that Runx2/RUNX2 haploinsufficiency is associated with pathologically widened cranial sutures in both mice and humans.82 Conversely, craniosynostosis occurs in most individuals with RUNX2 duplication.45,46
Genetic studies have illuminated the role of another molecule in osteoblast differentiation, retinoic acid. Recessive mutations in POR (MIM: 124015), the flavoprotein that donates electrons to all microsomal P450 enzymes, cause Antley-Bixler syndrome (MIM: 201750), in which craniosynostosis is associated with abnormal steroidogenesis.43 A genotype-phenotype correlation is apparent, such that individuals with skeletal malformations are usually compound heterozygous for variants encoding a combination of a missense and a null allele; moreover, no subjects harboring homozygous null mutations have been encountered, supporting the conclusion from mouse studies that complete loss of function is lethal.83,84 Residual POR activity could differentially affect the function of the many P450 enzymes, raising the question as to which aspect of steroidogenesis is related to craniosynostosis. Homozygous mutations of CYP26B1 (MIM: 605207), which encodes one of the P450 enzymes required for degradation of retinoic acid, lead to mineralization defects of the skull and/or craniosynostosis (MIM: 614416), related to increased differentiation of osteoblasts to terminal osteocytes.85 This evidence, together with the observation of elevated retinoic acid levels in blood or tissues of POR mutants in both mice86,87 and humans,88 suggests that disturbed retinoic acid metabolism is a key contributor to the pathology.89 Whether this manifests with craniosynostosis or mineralization defects might depend on the precise extent of osteoblast/osteocyte imbalance.85
How mechanical forces are integrated into the proliferation/differentiation response is uncertain, but primary cilia are likely to play a key role.90 In calvarial cultures, stretched sutures released FGF2 and demonstrated an immediate increase in permeability to Ca2+ ions.91 Potentially linked to these observations, mice with a neural crest-driven knockout of Pkd2, encoding a calcium channel that is a key component of the ciliary mechanotransduction system, exhibited postnatal fusions of sutures in the facial bones (causing a bent snout) and features such as fractured molar roots that were suggestive of increased trauma, attributed to failure of normal feedback linking tissue strength to mechanical loading.92 Furthermore, mice with a mutation in Fuz, a key regulator of ciliogenesis, have coronal synostosis.93 Cilia are also involved in integrating HH and WNT signaling, both of which are implicated in suture biogenesis, and mice with a Wnt1-driven neural crest conditional mutation of Kif3a have metopic synostosis.68,94 Given the proposed role of the IHH-Gli1 feedback loop in suture maintenance (Figure 2D), it is tempting to speculate that mechanotransduction in the suture could feed directly into this pathway; evidence is lacking at present but this would be a fruitful field for future investigation.
Resorption/Homeostasis
The steady state in the mature suture represents a balance between osteogenesis and resorption, the latter of which is mediated by osteoclasts of hematopoietic origin. Many of the genes associated with generalized skeletal dysplasias but with only low frequency of craniosynostosis (Table S1) are likely to affect this balance; for example, it is not surprising that osteopetroses, characterized by excessively dense bones and deficient osteoclast function, sometimes feature craniosynostosis in the clinical presentation. A more suture-specific mechanism is likely in the case of interleukin 11 (IL11) signaling, because loss-of-function mutations in the co-receptor IL11RA are predictably associated with craniosynostosis (MIM: 614188) in humans (and fusion of facial sutures in the case of mouse Il11ra−/− mutants).41 However, interpretation is complicated because of evidence that both osteoblast and osteoclast function are compromised. Although IL11 has a stimulatory role in osteoblasts,95 predicting that deficient signaling would be associated with osteopenia, the opposite turns out to be the case; trabecular bone volume was increased in knockout mice and osteoclast precursors showed a cell-autonomous defect in differentiation.96 This suggests that the osteoclast defect might predominate in the context of loss of IL11 function. Circumstantial support is provided by the observation that individuals homozygous for IL11RA (MIM: 600939) mutations often have delayed eruption of the secondary dentition, because localized resorption of the jaw bone by osteoclasts is a prerequisite for tooth eruption.41
The role and importance of cartilage intermediates in craniosynostosis is unclear. Although the skull bones are classically described as forming by intramembranous ossification, transient cartilage intermediates are well known to occur.60 Behr et al.97,98 have identified cartilage both in the normally fusing posterior frontal suture of wild-type mice (equivalent to the human metopic suture) and in the fusing coronal sutures of Twist1+/− mice. Of note, Twist1 inhibits chondrogenesis and is regulated by β-catenin (canonical WNT signaling).99 Consistent with this, the appearance of cartilage is accompanied by downregulation of canonical WNT signaling (revealed, for example, by loss of AXIN2 protein). However, these events were described during postnatal stages, long after the initiating processes leading to craniosynostosis, consistent with the possibility that these represent secondary, downstream consequences of altered signaling arising from earlier events.
Genetic and Biochemical Mechanisms in Craniosynostosis
Moving on from defining the developmental role of genes, another key element toward constructing a pathophysiological framework is to delineate the activity of mutations at a genetic and biochemical level. Here it is important to appreciate that the constellation of gene mutations that cause craniosynostosis is highly idiosyncratic. Unlike the situation, for example, with the BBSome,100 there are few cases where craniosynostosis results from simple loss of gene function (RAB23 [MIM: 606144] and IL11RA mutations [Table 2] are exceptions). One explanation could be that skull growth and cranial suture development occur relatively late during mammalian embryogenesis; loss of function of many key genes in suture development would probably already cause lethality owing to a separate and earlier requirement in organogenesis. Rather, craniosynostosis often seems to result as an “accidental” phenotypic consequence of particular genotypes, which emerges when mutations evade multiple earlier developmental sieves. A striking example is provided by the seemingly cytotoxic gain-of-function mechanisms associated with FGF receptor mutations, which—given the near-universal importance of FGF-mediated signaling in development—might be expected to have widespread adverse effects on many other aspects of organogenesis. Presumably in most other tissues and time points, the toxic effects of increased signal (Table 2) and associated defects in cellular processing of mutant protein117–119 are efficiently mitigated by tight feedback regulation. At present, however, this is speculation, because systems biology description of the cellular processes is too crude to provide explanatory power.
Table 2.
Biochemical Mechanisms in Craniosynostosis and Relationship to Pattern of Mendelian Inheritance
| Biochemical Mechanism (and Pattern of Mendelian Inheritance) | Example Genes | Example Alleles | Comments | References |
|---|---|---|---|---|
| Complete loss of function (recessive) | RAB23, IL11RA | homozygous nonsense or frameshifting mutations; also aa substitutions | consanguinity leading to autozygosity frequent | 41,44 |
| Partial loss of function (recessive; hypomorphic mutation) | POR, MEGF8, CDC45 | missense, weak splice site mutations (outside invariant ag/ and /gt sequences) | consanguinity leading to autozygosity occasional. Leaky mutations in essential genes; combinations of compound heterozygous alleles give more opportunity for combined output to fall within narrow window of activity for phenotypic effect | 23,42,83,84,89 |
| Partial loss of function (dominant; haploinsufficiency) | TWIST1, TCF12, ERF | complete and partial deletions; nonsense, frameshift; some aa substitutions | pathogenic aa substitutions localized to key DNA binding/dimerization regions. Biochemical and genetic evidence of interaction between Twist1 and Tcf12 | 26,48,73 |
| Partial loss of function (dominant negative) | STAT3 | aa substitutions | aa substitutions cluster in SH2 or DNA binding domains | 101 |
| Gain of function: increased ligand affinity/broadened specificity (dominant) | FGFR1, FGFR2, FGFR3 | equivalent Pro>Arg substitutions in IgII-IgIII linker of FGFR1, -2, and -3 | substitution to bulky arginine residue introduces additional contacts to FGFs, causing increased binding affinity and illegitimate binding by FGF10 | 102–106 |
| MSX2 | Pro>His and Pro>Leu substitutions at 7th position of MSX2 homeodomain | contacts minor groove of DNA, associated with altered DNA-binding specificity | 107 | |
| Gain of function: constitutive activation (dominant) | FGFR2, FGFR3 | aa substitutions to or from cysteine | results in covalent cross-linking of receptor monomers | 108,109 |
| FGFR2, FGFR3 | aa substitutions in kinase domain | abrogates requirement for dimerization | 110 | |
| FGFR3 | aa substitutions near membrane-spanning region | enhances transient interactions between monomers | 111 | |
| ZIC1 | loss of inhibitory motif | enhanced transcriptional activation | 52,65,66 | |
| Gain of function: increased dosage (dominant) | MSX2, IHH, RUNX2 | complete gene or regulatory element duplications | opposite phenotype (reduced ossification) associated with deletions in MSX2 and RUNX2 further illustrating dosage sensitivity | 40,46,112 |
| Gain of function: ectopic isoform expression (dominant) | FGFR2 | deletions or insertions involving exon encoding FGFR2c spliceform | drives illegitimate expression of FGFR2b in usually non-expressing tissues | 74,113,114 |
| Cellular interference (X-linked dominant with paradoxical male sparing) | EFNB1 | partial and complete heterozygous deletions and missense substitutions associated with similar phenotypes in females | occurs in females owing to random X-inactivation of X-linked EFNB1; secondary abnormality of receptor levels (EPHB2, EPHA4) demonstrated in mice | 115,116 |
Abbreviation is as follows: aa, amino acid.
Emphasizing the unpredictability of these phenotypes, species differences between human and mouse are frequent for identical mutations in orthologous genes. For example, mice with relevant mutations in Fgfr3 and Efnb1 lack the coronal synostosis associated with the equivalent human mutations,120,121 and mice with exact mutations mimicking Apert syndrome usually lack syndactyly.122–124 There are many examples of phenomena that rely on particular levels of wild-type or mutant protein selectively affecting suture biology, including the particular sensitivity of the sutures to haploinsufficiency (TWIST1, TCF12, ERF) and increased dosage states (IHH, MSX2, RUNX2), the role of X-inactivation interacting with heterozygous EFNB1 (MIM: 300035) mutations to disrupt tissue boundaries in craniofrontonasal syndrome, and the particular sensitivity of the cranial sutures to hypomorphic mutations in essential genes involved in cell division (CDC45 [MIM: 603465], ESCO2 [MIM: 609353], RECQL4 [MIM: 603780]). Selected examples are summarized, with references, in Table 2.
One observation of particular note is that equivalent amino acid substitutions in the extracellular IgII-IgIII linker of three FGFR paralogs (p.Pro252Arg in FGFR1, p.Pro253Arg in FGFR2, and p.Pro250Arg in FGFR3) each causes a distinct craniosynostosis syndrome (Pfeiffer [MIM: 101600], Apert [MIM: 101200], and Muenke [MIM: 602849] syndromes, respectively).27,29,37 Moreover, each of these syndromes is characteristically associated with coronal synostosis, whereas allelic mutations in each gene more typically present with multisuture fusion (Table 1). The likely common factor is that all three mutant receptors illegitimately bind FGF10,102–104 creating an abnormal paracrine signaling loop (see next section). Despite being caused by such very specific mutations, Muenke and Apert syndromes are two of the most common genetic diagnoses in craniosynostosis (Table 1); remarkably, new mutations at these particular nucleotides arise in the male germline at a frequency 500- to 1,000-fold higher than the background rate, because of a selective advantage of mutant cells in the testis (termed selfish spermatogonial selection).125
Pathophysiology
Given the specific temporal sequence of developmental processes taking place in the cranial sutures, the mouse provides the most accurate animal model for investigating the details of pathophysiology in craniosynostosis. However, as noted above, even accurate genetic models do not always have true craniosynostosis involving the cranial vault, although synostosis of facial bones is frequently identified in these mutants.41,120,121 Currently, the models that most accurately phenocopy the onset of vault craniosynostosis include several different gain-of-function mutations of Fgfr2 (Apert-Fgfr2Ser252Trp/+,123 Apert-Fgfr2Pro253Arg/+,124 Apert splicing Fgfr2ΔIIIc/+,113 Crouzon-Fgfr2Trp290Arg/+,126 Crouzon-Fgfr2Cys342Tyr/+,127 Beare-Stevenson-Fgfr2Tyr394Cys/+,128), Pfeiffer- Fgfr1Pro250Arg/+,129Twist1+/− haploinsufficiency,49,130 and ErfloxP/− dosage reduction.26 Additional mouse models are well reviewed in Holmes.122 In documenting pathogenesis in these models, it is particularly important to devote efforts to identifying the earliest developmental abnormality, ideally before any anatomical change is evident; once craniosynostosis has become manifest, it becomes extremely challenging to distinguish primary effects from secondary consequences of earlier abnormal processes in development. In this section we focus on studies that fulfil this requirement to illuminate the underlying pathogenic processes.
Apert-Fgfr2Ser252Trp/+ Mutant and Encroachment by Osteogenic Fronts
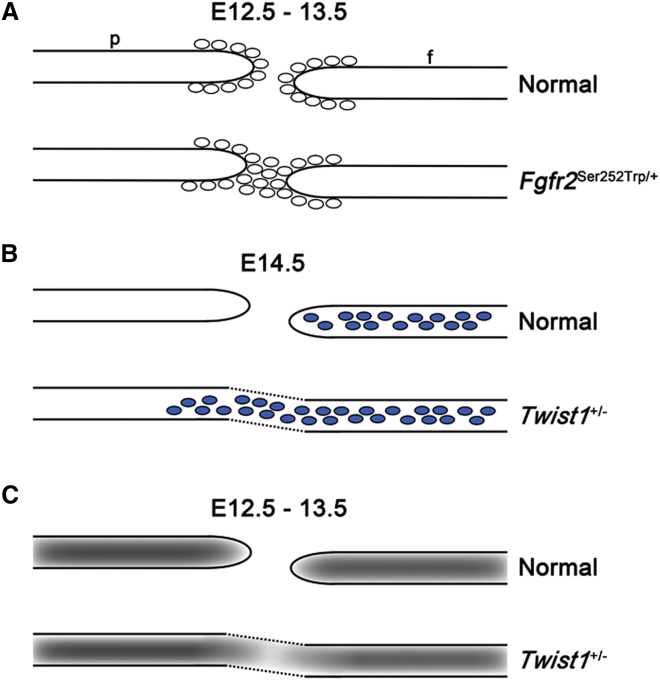
An exemplar for a carefully conducted disease model study is that on the Apert-Fgfr2Ser252Trp/+ mutant.131 Although no abnormality could be detected in mutant embryos at E12.5, by E13.5 multiple changes in expression of osteogenic genes were apparent, associated with narrowing of the nascent gap at the base of the coronal suture. Although more apical parts of the coronal suture remained patent for several further embryonic days, effectively its fate was already sealed because the bones could not grow separately at their bases. Having identified this critical E13.5 time point, studies of sutures from 1 day earlier (E12.5) were conducted in an attempt to identify even earlier markers for these events. A small but consistent increase in proliferation (assessed by bromodeoxyuridine incorporation and detection of Ki67 antigen) was found, demonstrating a biological abnormality at E12.5, corresponding to the later stages in organization of the supraorbital regulatory center. By further analysis of osteoblast cultures (postnatal day 1), which showed both accelerated proliferation and differentiation, it was proposed that the primary defect involved failure of the osteogenic fronts to halt their progress across the undifferentiated mesenchyme at the base of the cranial suture. In other words, the delicate balance that maintains the existence of a population of undifferentiated cells in the mid-sutural mesenchyme in the face of rapid proliferation and differentiation to each side is disturbed, and the stem cells are overwhelmed (Figure 3A).
Figure 3.
Early Pathological Processes in Coronal Sutures of Mutant Mice
(A) Apert-Fgfr2Ser252Trp/+ mutant. Mid-sutural region containing undifferentiated stem cells becomes populated by osteogenic cells (ovals; evidence supports both increased proliferation and failure of mechanisms preventing differentiation) at E12.5–E13.5. Abbreviations are as follows: f, frontal bone; p, parietal bone.
(B) Neural crest cell migration defect in Twist1+/− mutant. Normally, the neural crest cells (blue) are restricted to the future frontal bone territory. In mutant mice, cells do not observe the normal boundary and additionally populate the coronal suture and developing parietal bone.
(C) Primary failure to maintain a population of undifferentiated sutural stem cells. This is reflected in abnormal NOTCH1 protein levels (gray shading) at E12.5–E13.5.
Figure based on original data presented elsewhere.63,69,70,131
How do these findings relate to knowledge of craniosynostosis in humans? Embryonic day 13.0 in the mouse is equivalent to 6 weeks post-conception in terms of human skull development (8 weeks’ gestational age).4 At this stage it would be technically challenging to identify a causative mutation and, if a decision was made to terminate an affected pregnancy, study the embryonic material. The earliest available human pathological fetal material is from around 19 weeks,132 illustrating the critical role of the mouse studies in understanding the earliest stages of pathogenesis of craniosynostosis. However, Mathijssen et al.,133 by calibration against the separation of the frontal and parietal bone ossification centers in a series of human fetal skulls, found that the distance between ossification centers in two skulls of neonates who had Apert syndrome corresponded to 15 weeks’ gestation, not long after coronal sutures are first apparent. This corroborates the mouse study described above,131 that in Apert syndrome there is a primary failure in the function of the coronal suture around the time of its formation. Unfortunately, most functional studies of clinical material from craniosynostosis cases utilize cells obtained months or years after disease onset, and from poorly controlled tissue sources; hence, their pathogenic interpretation should be treated with particular caution.
Further analysis of the Apert-Fgfr2Ser252Trp/+ mutant was undertaken using tissue-specific drivers to enable selective expression of the mutation in cells of mesodermal or neural crest origin.134 This demonstrated that craniosynostosis still occurs in the mesoderm-driven Fgfr2Ser252Trp/+ mutant, typically with bony bridging from the frontal bone edge to a point behind the parietal bone edge. This suggests that Fgfr2Ser252Trp/+ expressed in early mesodermal osteoprogenitors is sufficiently sensitive to allow paracrine osteogenic induction by FGFs secreted from neural crest-derived tissue.134 By contrast, the reciprocal neural-crest-driven Fgfr2Ser252Trp/+ mutant does not have craniosynostosis, eliminating a deterministic role in pathology for either the nascent frontal bone or the underlying dura mater. However, the onset of the earliest abnormalities in the mesoderm-conditional mutant was 2–3 days later (E15.5) than in the germline mutant, indicating that the presence of mutant cells of neural crest origin does have a synergistic effect.
The mechanism outlined above falls into the general category of altered proliferation/differentiation balance, which at this early time point severely compromises coronal suture function. As indicated in Table 2, Apert substitutions result in both increased ligand affinity and broadened specificity of mutant FGFR2 receptors. Illegitimate binding to FGF10 is strongly implicated in the coronal (and facial) suture pathology, because reduction in Fgf10 dosage in the Apert-Fgfr2ΔIIIc/+ mouse model prevents the occurrence of craniosynostosis.74 The selective early coronal closure, combined with rapid expansion of the brain, might act to keep open the other major sutures even in a pro-osteogenic environment, leading to the widely open metopic and sagittal sutures seen in most individuals with Apert syndrome.135 A similar FGF10-mediated mechanism is likely to explain the selective coronal synostosis that occurs in Muenke syndrome (FGFR3-p.Pro250Arg heterozygote). Although the equivalent Fgfr3Pro244Arg/+ mouse model does not develop craniosynostosis, impeding a direct test of this idea, characteristic hearing loss in this model is rescued by reduced Fgf10 expression.105
Unlike in Apert syndrome, the activation mechanisms occurring for the FGFR2 mutations associated with Crouzon (MIM: 123500) and Pfeiffer syndromes are typically constitutive rather than ligand dependent (Table 2). Perhaps reflecting this distinct biochemical abnormality, these syndromes show a different pattern of craniosynostosis with less marked predilection for the coronal suture; instead, sagittal or multisuture synostoses are also common presentations. In severe Pfeiffer syndrome, all sutures either fail to form or fuse before birth, so the skull can grow only by remodelling; this leads to cloverleaf skull, a very severe disorder.136,137 A milder manifestation occurs in some Crouzon syndrome and also ERF-related craniosynostosis, whereby delayed closure of all sutures is associated with a relatively normal skull shape but high risk of raised intracranial pressure.26,138,139 Interestingly, some of the mouse mutants initially show paradoxically delayed markers of early osteogenesis, followed by later craniosynostosis,26,126 or differences in ossification potential between bones of differing origins,140 illustrating a shift of the proliferation-differentiation balance occurring over time. Neben et al.141 propose that this shift might reflect differential utilization of non-canonical nucleolar FGF receptor signaling.
The key effector pathway perturbed by FGF receptor mutations is the RAS-ERK pathway, as demonstrated by the marked reduction in abnormal phenotypes associated with genetic inhibition of FRS2-mediated signal transduction,142 in vivo inhibition of MEK1/2,143 and the craniosynostosis associated with Erf mutations.26 However, the complex relationship of RAS-ERK activation with craniosynostosis is highlighted by the fact that only a minority of individuals harboring RASopathy mutations develop craniosynostosis.144
Twist1+/− Mutant, Neural Crest Migration, and Boundary Integrity
The second process that has been subject to the most rigorous investigation in mouse mutants has been that of boundary formation at the coronal suture. The starting point for this work was the observation that in Saethre-Chotzen syndrome, caused by TWIST1 halpoinsufficiency, the coronal sutures are typically fused. By use of Wnt1-Cre/R26R labeling of cells of neural crest origin, Merrill et al.70 showed that in Twist1+/− mouse mutants, abnormal migration of cells derived from neural crest could be detected within the coronal suture and extending into the parietal bone territory, first observed at E14.5 (Figure 3B). Importantly, this cell-mixing process was shown to correlate with craniosynostosis. Given the frequent role of EPHRIN-EPH interactions in tissue boundary formation, protein levels of family members were examined and this revealed disturbed distribution of EPHRINs-A2 and -A4 and their receptor EPHA4 at E14.5.70 Consistent with this, EphA4−/− mutant mice were found to have craniosynostosis and Twist1+/−EphA4+/− embryos exhibited abnormal partitioning of DiI-labeled osteogenic cells, which occupied the coronal suture territory.63 Postnatally, the number of Gli1-expressing, presumptive stem cells was reduced in the Twist1+/− mutants.56
Mutations in another ligand, the X-chromosome-encoded EPHRIN-B1, cause craniofrontonasal syndrome (CFNS), which is characteristically associated with coronal synostosis; orthologous Efnb1 is expressed in neural crest but not cephalic mesoderm in mice.15 Based on studies of mouse limb and craniofacial development,115,116 there is strong evidence to implicate the female-restricted process of X inactivation in disrupting the normal coronal suture boundary, although this could not be directly proved because Efnb1+/− mice do not develop craniosynostosis.121 This unusual mechanism has been termed cellular interference (Table 2).25
Twist1+/− Mutant and Mesoderm Specification
NOTCH signaling also contributes to boundary formation, although no genes from this pathway have been identified as being frequently mutated in clinical craniosynostosis. In mice, the earliest abnormality identified in the sutures of Twist1+/− mutants, observed at E12.5 (the identical time point as for the Fgfr2Ser252Trp/+ mutant reviewed above, but 48 hr prior to observation of abnormal cell mixing), was spreading of NOTCH2 protein to include the mid-sutural mesenchyme (Figure 3C).69 Also of note, crossing of a conditional transgenic allele of Spry1 (encoding an inhibitor of FGF receptor signaling) with Twist1+/− mutant mice largely prevented the craniosynostosis from occurring.145 Together, these observations show that TWIST1 lies primarily upstream of FGF receptor signaling (as in nematode worms and fruit flies)146,147 and implicate TWIST1 in maintaining the identity of sutural cells, which might be important to prevent later cell mixing. Importantly, however, the conditional Spry1 experiment demonstrates that these abnormalities can be overridden in an environment that reduces the overall osteogenic drive. Hence the relative contributions that loss of boundary integrity versus increased osteogenic differentiation make to the craniosynostosis associated with TWIST1 mutation are difficult to separate.
Synthesis: A Genetic-Pathophysiological Framework
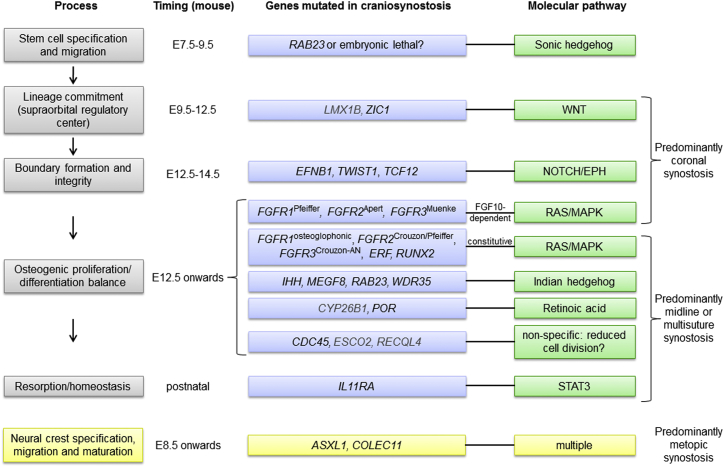
By combining information on (1) human gene mutations and their genetic/biochemical characteristics (Tables 1 and 2), (2) processes in suture formation and gene expression patterns (previous sections and Figure 2), and (3) detailed studies of pathophysiology (section above and Figure 3), we propose a disease framework for craniosynostosis built around the key processes of tissue boundary formation and proliferation/differentiation balance (Figure 4). This model includes earlier developmental stages (stem cell specification and migration, lineage commitment, and organization of supraorbital regulatory center) for which the link between genes and pathogenetic mechanisms is less certain, either because the mouse model does not phenocopy the human disorder (for example, murine Rab23 loss of function causes a severe neural tube defect)148,149 or because relevant studies have not yet been performed. The emerging importance of signaling through the IL11RA-STAT3 pathway, which probably affects remodelling, is also highlighted and provides an unexpected link between craniosynostosis and immunity. Although there is experimental evidence for TGFβ signaling providing an instructive role for the dura mater in suture homeostasis (reviewed in Levi et al.81), to our knowledge no genetic mutants have been shown to act primarily by disturbing dura mater-suture interactions. Analysis of Ski mutants targeted to contain the localized mutations found in Shprintzen-Goldberg syndrome would be interesting, because among TGFβ or BMP-related disorders, this is the one most consistently associated with craniosynostosis.47
Figure 4.
A Genetic-Pathophysiological Framework for Craniosynostosis
Processes in suture formation described in the text are displayed on the left (gray boxes). Other panels show relative timing of events in mouse, genes mutated (blue boxes; black type for core genes and gray type for additional genes), and pathways proposed to be affected (green boxes). Patterning of the supraorbital regulatory center and boundary formation are events particular to coronal suture development; correspondingly, mutations disrupting these processes lead predominantly to coronal craniosynostosis. Later developmental processes (proliferation-differentiation balance, homeostasis) apply to all sutures and correspondingly, pathological suture involvement tends to be more generalized. Mutations involving BMP signaling (MSX2, SKI) are not placed in this framework, pending further information from specific mouse models. Note that the origin of the metopic suture within the neural crest suggests that abnormal maturation of this tissue (through disturbed dynamics of cell identity or migration) might be a common factor predisposing to metopic synostosis, which is not reviewed here (yellow boxes, bottom).
Therapeutic Approaches?
Currently the mainstay of treatment of craniosynostosis is surgery, allied to support from specialties such as speech therapy and clinical psychology. Although it is attractive to envisage the surgeons losing business to novel medical therapies, serious challenges will need to be overcome. The majority of severe craniosynostosis is unanticipated at birth (many arise by new mutation), yet the associated suture fusion(s) have already initiated prenatally. Moreover, the idea that medical therapy could provide an adjunct to surgery (for example, by maintaining the patency of sutures post-operatively) is weakened by the counterintuitive fact that surgery frequently involves the destruction of patent sutures; rather, surgery aims to create an expanded but rigid skull vault (the separate bones being wired together),10,11 with further increase in brain growth accommodated within the remaining space, plus remodelling of the inner surface of the calvaria. The creation of artificial sutures as simple gaps between the bones could potentially lead to progressive skull deformity, as illustrated by occurrence of vertex bulge arising from strip craniectomy,150 unless the mechanical properties of normal sutures were accurately replicated.
Even with these caveats, the experiment of Shukla et al.,143 in which they virtually cured progeny mice with the Apert Fgfr2Ser252Trp/+ mutation by intraperitoneal injection of the pregnant mother with U0126, an inhibitor of MEK1/2, was undoubtedly spectacular.151 This treatment was able to reverse the coronal synostosis, runting, and infertility in some heterozygous mutant mice. However, the independent replication of these findings has not yet been reported and subsequent progress has been relatively slow, with the most notable development being the discovery that an inhibitor of p38 MAP kinase ameliorated the associated skin disorder (but not craniosynostosis) in the murine Beare-Stevenson-Fgfr2Y394C/+ model;128 the accessibility of skin makes this a therapeutically tractable target. Clearly the intelligent design of therapies requires detailed knowledge of the mechanisms of craniosynostosis and pathways affected, as surveyed above; recent reviews81,152 provide further details. In parallel with the development of potential therapeutics, careful natural history studies need to be conducted so that potential benefits and drawbacks can be assessed realistically in relation to outcomes based on current care.
Conclusions
The study of craniosynostosis has been transformed over the past two decades by the identification of pathogenic mutations in the most common classical syndromes. This has had many direct benefits, both for affected individuals and their families (improved diagnostic testing, genetic counselling, risk estimation, reproductive advice, and prognostic guidance) and for identifying key molecules and pathways in cranial suture development. Since 2010, the availability of whole exome and genome approaches to molecular genetics research has accelerated the rate of discovery of genes enriched for mutations in subjects with craniosynostosis.23,26,42,48,51 Here we have illustrated how these human genetics discoveries can be understood by integrating the information with multiple complementary approaches from biochemistry, structural studies, cell and developmental biology, and mouse genetics. Maintenance of cranial sutures requires an ongoing, exquisitely delicate balance to sustain a contiguous population of stem cells correctly located in the mid-sutural mesenchyme, while allowing for the rapid proliferation and differentiation of closely adjacent cells to enable skull growth. As further genes are discovered and reported, we anticipate that the pathophysiological framework described here (Figure 4) will prove useful when trying to incorporate new molecular components into the picture. In this review we have indicated areas needing further research; we think the analysis of mouse models using conditional drivers of expression combined with tracing of genetically marked cells, and understanding how genetic and biomechanical signals integrate through primary cilia (and other mechanisms) to effect coordinated brain/suture growth, are particularly exciting topics for the next decade. Given the importance of environmental factors in craniosynostosis and observation of clinical variability, including frequent asymmetry in suture fusion and non-penetrance, the contribution of genetic and epigenetic influences on allelic expression, somatic mosaicism, and mutation in regulatory motifs are all lines of inquiry that should be explored further and are likely to yield diagnostic answers in individual cases.
An alternative methodology in human genetics, more suited to identifying common susceptibility alleles conferring modest relative risk, is to use genome wide association studies (GWASs). The first GWAS for sagittal synostosis, reported in 2012, identified two significantly associated loci; one located 120 kb downstream of BMP2 (encoding a ligand in BMP signaling) and the other within an intron of BBS9 (component of the BBSome, involved in moving cargo molecules in and out of cilia).153 Although it should not be assumed that genetic susceptibility factors will occupy the same pathophysiological landscape as monogenic ones, both these components affect processes that overlap with those disrupted by previously identified single gene mutations.
Acknowledgments
We thank Aimee Fenwick, Anne Goriely, Deborah Lloyd, and anonymous referees for helpful comments on the manuscript, Simeon Boyd and Wanda Lattanzi for suggesting genes to include in Table S1, and Ryan McAllister (http://www.handjazz.com) for artwork. We are grateful to Steven Wall and David Johnson, lead surgeons at the Oxford Craniofacial Unit, for facilitating genetic research in their patients, and to the Wellcome Trust for sustained funding over a period of more than 20 years, which has made this work possible. The laboratory is currently supported by the Wellcome Trust (Senior Investigator Award to A.O.M.W., ref. 102731), the NIHR Oxford Biomedical Research Centre, and the NIH.
Footnotes
Supplemental Data include one table and can be found with this article online at http://dx.doi.org/10.1016/j.ajhg.2015.07.006.
Web Resources
The URLs for data presented herein are as follows:
GeneReviews, Verloes, A., Drunat, S., Gressens, P., et al. (2013). Primary autosomal recessive microcephalies and Seckel syndrome spectrum disorders, http://www.ncbi.nlm.nih.gov/books/NBK9587/
OMIM, http://www.omim.org/
Supplemental Data
References
- 1.Khonsari R.H., Olivier J., Vigneaux P., Sanchez S., Tafforeau P., Ahlberg P.E., Di Rocco F., Bresch D., Corre P., Ohazama A. A mathematical model for mechanotransduction at the early steps of suture formation. Proc. Biol. Sci. 2013;280:20122670. doi: 10.1098/rspb.2012.2670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nieman B.J., Blank M.C., Roman B.B., Henkelman R.M., Millen K.J. If the skull fits: magnetic resonance imaging and microcomputed tomography for combined analysis of brain and skull phenotypes in the mouse. Physiol. Genomics. 2012;44:992–1002. doi: 10.1152/physiolgenomics.00093.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Herring S.W. Mechanical influences on suture development and patency. Front. Oral Biol. 2008;12:41–56. doi: 10.1159/0000115031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rice D.P. Developmental anatomy of craniofacial sutures. Front. Oral Biol. 2008;12:1–21. doi: 10.1159/000115028. [DOI] [PubMed] [Google Scholar]
- 5.Miura T., Perlyn C.A., Kinboshi M., Ogihara N., Kobayashi-Miura M., Morriss-Kay G.M., Shiota K. Mechanism of skull suture maintenance and interdigitation. J. Anat. 2009;215:642–655. doi: 10.1111/j.1469-7580.2009.01148.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lajeunie E., Le Merrer M., Bonaïti-Pellie C., Marchac D., Renier D. Genetic study of nonsyndromic coronal craniosynostosis. Am. J. Med. Genet. 1995;55:500–504. doi: 10.1002/ajmg.1320550422. [DOI] [PubMed] [Google Scholar]
- 7.Boulet S.L., Rasmussen S.A., Honein M.A. A population-based study of craniosynostosis in metropolitan Atlanta, 1989-2003. Am. J. Med. Genet. A. 2008;146A:984–991. doi: 10.1002/ajmg.a.32208. [DOI] [PubMed] [Google Scholar]
- 8.Badve C.A., K M.M., Iyer R.S., Ishak G.E., Khanna P.C. Craniosynostosis: imaging review and primer on computed tomography. Pediatr. Radiol. 2013;43:728–742. doi: 10.1007/s00247-013-2673-6. quiz 725–727. [DOI] [PubMed] [Google Scholar]
- 9.Johnson D., Wilkie A.O.M. Craniosynostosis. Eur. J. Hum. Genet. 2011;19:369–376. doi: 10.1038/ejhg.2010.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Forrest C.R., Hopper R.A. Craniofacial syndromes and surgery. Plast. Reconstr. Surg. 2013;131:86e–109e. doi: 10.1097/PRS.0b013e318272c12b. [DOI] [PubMed] [Google Scholar]
- 11.Kirmi O., Lo S.J., Johnson D., Anslow P. Craniosynostosis: a radiological and surgical perspective. Semin. Ultrasound CT MR. 2009;30:492–512. doi: 10.1053/j.sult.2009.08.002. [DOI] [PubMed] [Google Scholar]
- 12.Vu H.L., Panchal J., Parker E.E., Levine N.S., Francel P. The timing of physiologic closure of the metopic suture: a review of 159 patients using reconstructed 3D CT scans of the craniofacial region. J. Craniofac. Surg. 2001;12:527–532. doi: 10.1097/00001665-200111000-00005. [DOI] [PubMed] [Google Scholar]
- 13.Lajeunie E., Barcik U., Thorne J.A., El Ghouzzi V., Bourgeois M., Renier D. Craniosynostosis and fetal exposure to sodium valproate. J. Neurosurg. 2001;95:778–782. doi: 10.3171/jns.2001.95.5.0778. [DOI] [PubMed] [Google Scholar]
- 14.Lajeunie E., Crimmins D.W., Arnaud E., Renier D. Genetic considerations in nonsyndromic midline craniosynostoses: a study of twins and their families. J. Neurosurg. 2005;103(4, Suppl):353–356. doi: 10.3171/ped.2005.103.4.0353. [DOI] [PubMed] [Google Scholar]
- 15.Twigg S.R.F., Kan R., Babbs C., Bochukova E.G., Robertson S.P., Wall S.A., Morriss-Kay G.M., Wilkie A.O.M. Mutations of ephrin-B1 (EFNB1), a marker of tissue boundary formation, cause craniofrontonasal syndrome. Proc. Natl. Acad. Sci. USA. 2004;101:8652–8657. doi: 10.1073/pnas.0402819101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Passos-Bueno M.R., Fanganiello R.D., Jehee F.S. Craniosynostosis and chromosomal alterations. In: Muenke M., Kress W., Collmann H., Solomon B., editors. Craniosynostoses: Molecular Genetics, Principles of Diagnosis and Treatment. Karger; Basel: 2011. pp. 152–164. [Google Scholar]
- 17.Wilkie A.O.M., Byren J.C., Hurst J.A., Jayamohan J., Johnson D., Knight S.J.L., Lester T., Richards P.G., Twigg S.R.F., Wall S.A. Prevalence and complications of single-gene and chromosomal disorders in craniosynostosis. Pediatrics. 2010;126:e391–e400. doi: 10.1542/peds.2009-3491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sanchez-Lara P.A., Carmichael S.L., Graham J.M., Jr., Lammer E.J., Shaw G.M., Ma C., Rasmussen S.A., National Birth Defects Prevention Study Fetal constraint as a potential risk factor for craniosynostosis. Am. J. Med. Genet. A. 2010;152A:394–400. doi: 10.1002/ajmg.a.33246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jacob S., Wu C., Freeman T.A., Koyama E., Kirschner R.E. Expression of Indian Hedgehog, BMP-4 and Noggin in craniosynostosis induced by fetal constraint. Ann. Plast. Surg. 2007;58:215–221. doi: 10.1097/01.sap.0000232833.41739.a5. [DOI] [PubMed] [Google Scholar]
- 20.Cohen M.M., Jr., MacLean R.E. Oxford University Press; Oxford: 2000. Craniosynostosis: Diagnosis, Evaluation, and Management. [Google Scholar]
- 21.Jabs E.W., Müller U., Li X., Ma L., Luo W., Haworth I.S., Klisak I., Sparkes R., Warman M.L., Mulliken J.B. A mutation in the homeodomain of the human MSX2 gene in a family affected with autosomal dominant craniosynostosis. Cell. 1993;75:443–450. doi: 10.1016/0092-8674(93)90379-5. [DOI] [PubMed] [Google Scholar]
- 22.Hoischen A., van Bon B.W., Rodríguez-Santiago B., Gilissen C., Vissers L.E., de Vries P., Janssen I., van Lier B., Hastings R., Smithson S.F. De novo nonsense mutations in ASXL1 cause Bohring-Opitz syndrome. Nat. Genet. 2011;43:729–731. doi: 10.1038/ng.868. [DOI] [PubMed] [Google Scholar]
- 23.Taylor J.C., Martin H.C., Lise S., Broxholme J., Cazier J.-B., Rimmer A., Kanapin A., Lunter G., Fiddy S., Allan C. Factors influencing success of clinical genome sequencing across a broad spectrum of disorders. Nat. Genet. 2015;47:717–726. doi: 10.1038/ng.3304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rooryck C., Diaz-Font A., Osborn D.P., Chabchoub E., Hernandez-Hernandez V., Shamseldin H., Kenny J., Waters A., Jenkins D., Kaissi A.A. Mutations in lectin complement pathway genes COLEC11 and MASP1 cause 3MC syndrome. Nat. Genet. 2011;43:197–203. doi: 10.1038/ng.757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wieland I., Jakubiczka S., Muschke P., Cohen M., Thiele H., Gerlach K.L., Adams R.H., Wieacker P. Mutations of the ephrin-B1 gene cause craniofrontonasal syndrome. Am. J. Hum. Genet. 2004;74:1209–1215. doi: 10.1086/421532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Twigg S.R.F., Vorgia E., McGowan S.J., Peraki I., Fenwick A.L., Sharma V.P., Allegra M., Zaragkoulias A., Sadighi Akha E., Knight S.J. Reduced dosage of ERF causes complex craniosynostosis in humans and mice and links ERK1/2 signaling to regulation of osteogenesis. Nat. Genet. 2013;45:308–313. doi: 10.1038/ng.2539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Muenke M., Schell U., Hehr A., Robin N.H., Losken H.W., Schinzel A., Pulleyn L.J., Rutland P., Reardon W., Malcolm S. A common mutation in the fibroblast growth factor receptor 1 gene in Pfeiffer syndrome. Nat. Genet. 1994;8:269–274. doi: 10.1038/ng1194-269. [DOI] [PubMed] [Google Scholar]
- 28.White K.E., Cabral J.M., Davis S.I., Fishburn T., Evans W.E., Ichikawa S., Fields J., Yu X., Shaw N.J., McLellan N.J. Mutations that cause osteoglophonic dysplasia define novel roles for FGFR1 in bone elongation. Am. J. Hum. Genet. 2005;76:361–367. doi: 10.1086/427956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wilkie A.O.M., Slaney S.F., Oldridge M., Poole M.D., Ashworth G.J., Hockley A.D., Hayward R.D., David D.J., Pulleyn L.J., Rutland P. Apert syndrome results from localized mutations of FGFR2 and is allelic with Crouzon syndrome. Nat. Genet. 1995;9:165–172. doi: 10.1038/ng0295-165. [DOI] [PubMed] [Google Scholar]
- 30.Przylepa K.A., Paznekas W., Zhang M., Golabi M., Bias W., Bamshad M.J., Carey J.C., Hall B.D., Stevenson R., Orlow S. Fibroblast growth factor receptor 2 mutations in Beare-Stevenson cutis gyrata syndrome. Nat. Genet. 1996;13:492–494. doi: 10.1038/ng0896-492. [DOI] [PubMed] [Google Scholar]
- 31.Reardon W., Winter R.M., Rutland P., Pulleyn L.J., Jones B.M., Malcolm S. Mutations in the fibroblast growth factor receptor 2 gene cause Crouzon syndrome. Nat. Genet. 1994;8:98–103. doi: 10.1038/ng0994-98. [DOI] [PubMed] [Google Scholar]
- 32.Jabs E.W., Li X., Scott A.F., Meyers G., Chen W., Eccles M., Mao J.I., Charnas L.R., Jackson C.E., Jaye M. Jackson-Weiss and Crouzon syndromes are allelic with mutations in fibroblast growth factor receptor 2. Nat. Genet. 1994;8:275–279. doi: 10.1038/ng1194-275. [DOI] [PubMed] [Google Scholar]
- 33.Rutland P., Pulleyn L.J., Reardon W., Baraitser M., Hayward R., Jones B., Malcolm S., Winter R.M., Oldridge M., Slaney S.F. Identical mutations in the FGFR2 gene cause both Pfeiffer and Crouzon syndrome phenotypes. Nat. Genet. 1995;9:173–176. doi: 10.1038/ng0295-173. [DOI] [PubMed] [Google Scholar]
- 34.Lajeunie E., Ma H.W., Bonaventure J., Munnich A., Le Merrer M., Renier D. FGFR2 mutations in Pfeiffer syndrome. Nat. Genet. 1995;9:108. doi: 10.1038/ng0295-108. [DOI] [PubMed] [Google Scholar]
- 35.Schell U., Hehr A., Feldman G.J., Robin N.H., Zackai E.H., de Die-Smulders C., Viskochil D.H., Stewart J.M., Wolff G., Ohashi H. Mutations in FGFR1 and FGFR2 cause familial and sporadic Pfeiffer syndrome. Hum. Mol. Genet. 1995;4:323–328. doi: 10.1093/hmg/4.3.323. [DOI] [PubMed] [Google Scholar]
- 36.Merrill A.E., Sarukhanov A., Krejci P., Idoni B., Camacho N., Estrada K.D., Lyons K.M., Deixler H., Robinson H., Chitayat D. Bent bone dysplasia-FGFR2 type, a distinct skeletal disorder, has deficient canonical FGF signaling. Am. J. Hum. Genet. 2012;90:550–557. doi: 10.1016/j.ajhg.2012.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bellus G.A., Gaudenz K., Zackai E.H., Clarke L.A., Szabo J., Francomano C.A., Muenke M. Identical mutations in three different fibroblast growth factor receptor genes in autosomal dominant craniosynostosis syndromes. Nat. Genet. 1996;14:174–176. doi: 10.1038/ng1096-174. [DOI] [PubMed] [Google Scholar]
- 38.Meyers G.A., Orlow S.J., Munro I.R., Przylepa K.A., Jabs E.W. Fibroblast growth factor receptor 3 (FGFR3) transmembrane mutation in Crouzon syndrome with acanthosis nigricans. Nat. Genet. 1995;11:462–464. doi: 10.1038/ng1295-462. [DOI] [PubMed] [Google Scholar]
- 39.Tavormina P.L., Shiang R., Thompson L.M., Zhu Y.-Z., Wilkin D.J., Lachman R.S., Wilcox W.R., Rimoin D.L., Cohn D.H., Wasmuth J.J. Thanatophoric dysplasia (types I and II) caused by distinct mutations in fibroblast growth factor receptor 3. Nat. Genet. 1995;9:321–328. doi: 10.1038/ng0395-321. [DOI] [PubMed] [Google Scholar]
- 40.Klopocki E., Lohan S., Brancati F., Koll R., Brehm A., Seemann P., Dathe K., Stricker S., Hecht J., Bosse K. Copy-number variations involving the IHH locus are associated with syndactyly and craniosynostosis. Am. J. Hum. Genet. 2011;88:70–75. doi: 10.1016/j.ajhg.2010.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nieminen P., Morgan N.V., Fenwick A.L., Parmanen S., Veistinen L., Mikkola M.L., van der Spek P.J., Giraud A., Judd L., Arte S. Inactivation of IL11 signaling causes craniosynostosis, delayed tooth eruption, and supernumerary teeth. Am. J. Hum. Genet. 2011;89:67–81. doi: 10.1016/j.ajhg.2011.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Twigg S.R.F., Lloyd D., Jenkins D., Elçioglu N.E., Cooper C.D., Al-Sannaa N., Annagür A., Gillessen-Kaesbach G., Hüning I., Knight S.J. Mutations in multidomain protein MEGF8 identify a Carpenter syndrome subtype associated with defective lateralization. Am. J. Hum. Genet. 2012;91:897–905. doi: 10.1016/j.ajhg.2012.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Flück C.E., Tajima T., Pandey A.V., Arlt W., Okuhara K., Verge C.F., Jabs E.W., Mendonça B.B., Fujieda K., Miller W.L. Mutant P450 oxidoreductase causes disordered steroidogenesis with and without Antley-Bixler syndrome. Nat. Genet. 2004;36:228–230. doi: 10.1038/ng1300. [DOI] [PubMed] [Google Scholar]
- 44.Jenkins D., Seelow D., Jehee F.S., Perlyn C.A., Alonso L.G., Bueno D.F., Donnai D., Josifova D., Mathijssen I.M., Morton J.E. RAB23 mutations in Carpenter syndrome imply an unexpected role for hedgehog signaling in cranial-suture development and obesity. Am. J. Hum. Genet. 2007;80:1162–1170. doi: 10.1086/518047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mefford H.C., Shafer N., Antonacci F., Tsai J.M., Park S.S., Hing A.V., Rieder M.J., Smyth M.D., Speltz M.L., Eichler E.E., Cunningham M.L. Copy number variation analysis in single-suture craniosynostosis: multiple rare variants including RUNX2 duplication in two cousins with metopic craniosynostosis. Am. J. Med. Genet. A. 2010;152A:2203–2210. doi: 10.1002/ajmg.a.33557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Varvagiannis K., Stefanidou A., Gyftodimou Y., Lord H., Williams L., Sarri C., Pandelia E., Bazopoulou-Kyrkanidou E., Noakes C., Lester T. Pure de novo partial trisomy 6p in a girl with craniosynostosis. Am. J. Med. Genet. A. 2013;161A:343–351. doi: 10.1002/ajmg.a.35727. [DOI] [PubMed] [Google Scholar]
- 47.Doyle A.J., Doyle J.J., Bessling S.L., Maragh S., Lindsay M.E., Schepers D., Gillis E., Mortier G., Homfray T., Sauls K. Mutations in the TGF-β repressor SKI cause Shprintzen-Goldberg syndrome with aortic aneurysm. Nat. Genet. 2012;44:1249–1254. doi: 10.1038/ng.2421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sharma V.P., Fenwick A.L., Brockop M.S., McGowan S.J., Goos J.A., Hoogeboom A.J., Brady A.F., Jeelani N.O., Lynch S.A., Mulliken J.B., 500 Whole-Genome Sequences (WGS500) Consortium Mutations in TCF12, encoding a basic helix-loop-helix partner of TWIST1, are a frequent cause of coronal craniosynostosis. Nat. Genet. 2013;45:304–307. doi: 10.1038/ng.2531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.el Ghouzzi V., Le Merrer M., Perrin-Schmitt F., Lajeunie E., Benit P., Renier D., Bourgeois P., Bolcato-Bellemin A.-L., Munnich A., Bonaventure J. Mutations of the TWIST gene in the Saethre-Chotzen syndrome. Nat. Genet. 1997;15:42–46. doi: 10.1038/ng0197-42. [DOI] [PubMed] [Google Scholar]
- 50.Howard T.D., Paznekas W.A., Green E.D., Chiang L.C., Ma N., Ortiz de Luna R.I., Garcia Delgado C., Gonzalez-Ramos M., Kline A.D., Jabs E.W. Mutations in TWIST, a basic helix-loop-helix transcription factor, in Saethre-Chotzen syndrome. Nat. Genet. 1997;15:36–41. doi: 10.1038/ng0197-36. [DOI] [PubMed] [Google Scholar]
- 51.Gilissen C., Arts H.H., Hoischen A., Spruijt L., Mans D.A., Arts P., van Lier B., Steehouwer M., van Reeuwijk J., Kant S.G. Exome sequencing identifies WDR35 variants involved in Sensenbrenner syndrome. Am. J. Hum. Genet. 2010;87:418–423. doi: 10.1016/j.ajhg.2010.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Twigg S.R.F., Forecki J., Goos J.A.C., Richardson I.C.A., Hoogeboom A.J.M., Van den Ouweland A.M.W., Swagemakers S.M.A., Lequin M.H., Van Antwerp D., McGowan S.J. Gain-of-function mutations in ZIC1 are associated with coronal craniosynostosis and learning disability. Am. J. Hum. Genet. 2015;97:378–388. doi: 10.1016/j.ajhg.2015.07.007. this issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Morriss-Kay G.M., Wilkie A.O.M. Growth of the normal skull vault and its alteration in craniosynostosis: insights from human genetics and experimental studies. J. Anat. 2005;207:637–653. doi: 10.1111/j.1469-7580.2005.00475.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wilkie A.O.M. Craniosynostosis: genes and mechanisms. Hum. Mol. Genet. 1997;6:1647–1656. doi: 10.1093/hmg/6.10.1647. [DOI] [PubMed] [Google Scholar]
- 55.Lana-Elola E., Rice R., Grigoriadis A.E., Rice D.P. Cell fate specification during calvarial bone and suture development. Dev. Biol. 2007;311:335–346. doi: 10.1016/j.ydbio.2007.08.028. [DOI] [PubMed] [Google Scholar]
- 56.Zhao H., Feng J., Ho T.V., Grimes W., Urata M., Chai Y. The suture provides a niche for mesenchymal stem cells of craniofacial bones. Nat. Cell Biol. 2015;17:386–396. doi: 10.1038/ncb3139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Deckelbaum R.A., Holmes G., Zhao Z., Tong C., Basilico C., Loomis C.A. Regulation of cranial morphogenesis and cell fate at the neural crest-mesoderm boundary by engrailed 1. Development. 2012;139:1346–1358. doi: 10.1242/dev.076729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Jiang X., Iseki S., Maxson R.E., Sucov H.M., Morriss-Kay G.M. Tissue origins and interactions in the mammalian skull vault. Dev. Biol. 2002;241:106–116. doi: 10.1006/dbio.2001.0487. [DOI] [PubMed] [Google Scholar]
- 59.Johnson D., Iseki S., Wilkie A.O.M., Morriss-Kay G.M. Expression patterns of Twist and Fgfr1, -2 and -3 in the developing mouse coronal suture suggest a key role for twist in suture initiation and biogenesis. Mech. Dev. 2000;91:341–345. doi: 10.1016/s0925-4773(99)00278-6. [DOI] [PubMed] [Google Scholar]
- 60.Iseki S., Wilkie A.O.M., Morriss-Kay G.M. Fgfr1 and Fgfr2 have distinct differentiation- and proliferation-related roles in the developing mouse skull vault. Development. 1999;126:5611–5620. doi: 10.1242/dev.126.24.5611. [DOI] [PubMed] [Google Scholar]
- 61.Iseki S., Wilkie A.O.M., Heath J.K., Ishimaru T., Eto K., Morriss-Kay G.M. Fgfr2 and osteopontin domains in the developing skull vault are mutually exclusive and can be altered by locally applied FGF2. Development. 1997;124:3375–3384. doi: 10.1242/dev.124.17.3375. [DOI] [PubMed] [Google Scholar]
- 62.Yoshida T., Vivatbutsiri P., Morriss-Kay G., Saga Y., Iseki S. Cell lineage in mammalian craniofacial mesenchyme. Mech. Dev. 2008;125:797–808. doi: 10.1016/j.mod.2008.06.007. [DOI] [PubMed] [Google Scholar]
- 63.Ting M.C., Wu N.L., Roybal P.G., Sun J., Liu L., Yen Y., Maxson R.E., Jr. EphA4 as an effector of Twist1 in the guidance of osteogenic precursor cells during calvarial bone growth and in craniosynostosis. Development. 2009;136:855–864. doi: 10.1242/dev.028605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Benedyk M.J., Mullen J.R., DiNardo S. odd-paired: a zinc finger pair-rule protein required for the timely activation of engrailed and wingless in Drosophila embryos. Genes Dev. 1994;8:105–117. doi: 10.1101/gad.8.1.105. [DOI] [PubMed] [Google Scholar]
- 65.Kuo J.S., Patel M., Gamse J., Merzdorf C., Liu X., Apekin V., Sive H. Opl: a zinc finger protein that regulates neural determination and patterning in Xenopus. Development. 1998;125:2867–2882. doi: 10.1242/dev.125.15.2867. [DOI] [PubMed] [Google Scholar]
- 66.Merzdorf C.S., Sive H.L. The zic1 gene is an activator of Wnt signaling. Int. J. Dev. Biol. 2006;50:611–617. doi: 10.1387/ijdb.052110cm. [DOI] [PubMed] [Google Scholar]
- 67.Mishina Y., Snider T.N. Neural crest cell signaling pathways critical to cranial bone development and pathology. Exp. Cell Res. 2014;325:138–147. doi: 10.1016/j.yexcr.2014.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Liu B., Chen S., Johnson C., Helms J.A. A ciliopathy with hydrocephalus, isolated craniosynostosis, hypertelorism, and clefting caused by deletion of Kif3a. Reprod. Toxicol. 2014;48:88–97. doi: 10.1016/j.reprotox.2014.05.009. [DOI] [PubMed] [Google Scholar]
- 69.Yen H.Y., Ting M.C., Maxson R.E. Jagged1 functions downstream of Twist1 in the specification of the coronal suture and the formation of a boundary between osteogenic and non-osteogenic cells. Dev. Biol. 2010;347:258–270. doi: 10.1016/j.ydbio.2010.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Merrill A.E., Bochukova E.G., Brugger S.M., Ishii M., Pilz D.T., Wall S.A., Lyons K.M., Wilkie A.O.M., Maxson R.E., Jr. Cell mixing at a neural crest-mesoderm boundary and deficient ephrin-Eph signaling in the pathogenesis of craniosynostosis. Hum. Mol. Genet. 2006;15:1319–1328. doi: 10.1093/hmg/ddl052. [DOI] [PubMed] [Google Scholar]
- 71.Lenton K., James A.W., Manu A., Brugmann S.A., Birker D., Nelson E.R., Leucht P., Helms J.A., Longaker M.T. Indian hedgehog positively regulates calvarial ossification and modulates bone morphogenetic protein signaling. Genesis. 2011;49:784–796. doi: 10.1002/dvg.20768. [DOI] [PubMed] [Google Scholar]
- 72.Bialek P., Kern B., Yang X., Schrock M., Sosic D., Hong N., Wu H., Yu K., Ornitz D.M., Olson E.N. A twist code determines the onset of osteoblast differentiation. Dev. Cell. 2004;6:423–435. doi: 10.1016/s1534-5807(04)00058-9. [DOI] [PubMed] [Google Scholar]
- 73.Connerney J., Andreeva V., Leshem Y., Muentener C., Mercado M.A., Spicer D.B. Twist1 dimer selection regulates cranial suture patterning and fusion. Dev. Dyn. 2006;235:1345–1357. doi: 10.1002/dvdy.20717. [DOI] [PubMed] [Google Scholar]
- 74.Hajihosseini M.K., Duarte R., Pegrum J., Donjacour A., Lana-Elola E., Rice D.P., Sharpe J., Dickson C. Evidence that Fgf10 contributes to the skeletal and visceral defects of an Apert syndrome mouse model. Dev. Dyn. 2009;238:376–385. doi: 10.1002/dvdy.21648. [DOI] [PubMed] [Google Scholar]
- 75.Ornitz D.M., Marie P.J. FGF signaling pathways in endochondral and intramembranous bone development and human genetic disease. Genes Dev. 2002;16:1446–1465. doi: 10.1101/gad.990702. [DOI] [PubMed] [Google Scholar]
- 76.Liu Z., Xu J., Colvin J.S., Ornitz D.M. Coordination of chondrogenesis and osteogenesis by fibroblast growth factor 18. Genes Dev. 2002;16:859–869. doi: 10.1101/gad.965602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ohbayashi N., Shibayama M., Kurotaki Y., Imanishi M., Fujimori T., Itoh N., Takada S. FGF18 is required for normal cell proliferation and differentiation during osteogenesis and chondrogenesis. Genes Dev. 2002;16:870–879. doi: 10.1101/gad.965702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Harada M., Murakami H., Okawa A., Okimoto N., Hiraoka S., Nakahara T., Akasaka R., Shiraishi Y., Futatsugi N., Mizutani-Koseki Y. FGF9 monomer-dimer equilibrium regulates extracellular matrix affinity and tissue diffusion. Nat. Genet. 2009;41:289–298. doi: 10.1038/ng.316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yokota M., Kobayashi Y., Morita J., Suzuki H., Hashimoto Y., Sasaki Y., Akiyoshi K., Moriyama K. Therapeutic effect of nanogel-based delivery of soluble FGFR2 with S252W mutation on craniosynostosis. PLoS ONE. 2014;9:e101693. doi: 10.1371/journal.pone.0101693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hajihosseini M.K., Heath J.K. Expression patterns of fibroblast growth factors-18 and -20 in mouse embryos is suggestive of novel roles in calvarial and limb development. Mech. Dev. 2002;113:79–83. doi: 10.1016/s0925-4773(01)00656-6. [DOI] [PubMed] [Google Scholar]
- 81.Levi B., Wan D.C., Wong V.W., Nelson E., Hyun J., Longaker M.T. Cranial suture biology: from pathways to patient care. J. Craniofac. Surg. 2012;23:13–19. doi: 10.1097/SCS.0b013e318240c6c0. [DOI] [PubMed] [Google Scholar]
- 82.Mundlos S., Otto F., Mundlos C., Mulliken J.B., Aylsworth A.S., Albright S., Lindhout D., Cole W.G., Henn W., Knoll J.H.M. Mutations involving the transcription factor CBFA1 cause cleidocranial dysplasia. Cell. 1997;89:773–779. doi: 10.1016/s0092-8674(00)80260-3. [DOI] [PubMed] [Google Scholar]
- 83.Fukami M., Nishimura G., Homma K., Nagai T., Hanaki K., Uematsu A., Ishii T., Numakura C., Sawada H., Nakacho M. Cytochrome P450 oxidoreductase deficiency: identification and characterization of biallelic mutations and genotype-phenotype correlations in 35 Japanese patients. J. Clin. Endocrinol. Metab. 2009;94:1723–1731. doi: 10.1210/jc.2008-2816. [DOI] [PubMed] [Google Scholar]
- 84.Krone N., Reisch N., Idkowiak J., Dhir V., Ivison H.E., Hughes B.A., Rose I.T., O’Neil D.M., Vijzelaar R., Smith M.J. Genotype-phenotype analysis in congenital adrenal hyperplasia due to P450 oxidoreductase deficiency. J. Clin. Endocrinol. Metab. 2012;97:E257–E267. doi: 10.1210/jc.2011-0640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Laue K., Pogoda H.M., Daniel P.B., van Haeringen A., Alanay Y., von Ameln S., Rachwalski M., Morgan T., Gray M.J., Breuning M.H. Craniosynostosis and multiple skeletal anomalies in humans and zebrafish result from a defect in the localized degradation of retinoic acid. Am. J. Hum. Genet. 2011;89:595–606. doi: 10.1016/j.ajhg.2011.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ribes V., Otto D.M., Dickmann L., Schmidt K., Schuhbaur B., Henderson C., Blomhoff R., Wolf C.R., Tickle C., Dollé P. Rescue of cytochrome P450 oxidoreductase (Por) mouse mutants reveals functions in vasculogenesis, brain and limb patterning linked to retinoic acid homeostasis. Dev. Biol. 2007;303:66–81. doi: 10.1016/j.ydbio.2006.10.032. [DOI] [PubMed] [Google Scholar]
- 87.Schmidt K., Hughes C., Chudek J.A., Goodyear S.R., Aspden R.M., Talbot R., Gundersen T.E., Blomhoff R., Henderson C., Wolf C.R., Tickle C. Cholesterol metabolism: the main pathway acting downstream of cytochrome P450 oxidoreductase in skeletal development of the limb. Mol. Cell. Biol. 2009;29:2716–2729. doi: 10.1128/MCB.01638-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Fukami M., Nagai T., Mochizuki H., Muroya K., Yamada G., Takitani K., Ogata T. Anorectal and urinary anomalies and aberrant retinoic acid metabolism in cytochrome P450 oxidoreductase deficiency. Mol. Genet. Metab. 2010;100:269–273. doi: 10.1016/j.ymgme.2010.03.023. [DOI] [PubMed] [Google Scholar]
- 89.Pandey A.V., Flück C.E. NADPH P450 oxidoreductase: structure, function, and pathology of diseases. Pharmacol. Ther. 2013;138:229–254. doi: 10.1016/j.pharmthera.2013.01.010. [DOI] [PubMed] [Google Scholar]
- 90.Mathieu P.S., Bodle J.C., Loboa E.G. Primary cilium mechanotransduction of tensile strain in 3D culture: Finite element analyses of strain amplification caused by tensile strain applied to a primary cilium embedded in a collagen matrix. J. Biomech. 2014;47:2211–2217. doi: 10.1016/j.jbiomech.2014.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Yu J.C., Lucas J.H., Fryberg K., Borke J.L. Extrinsic tension results in FGF-2 release, membrane permeability change, and intracellular Ca++ increase in immature cranial sutures. J. Craniofac. Surg. 2001;12:391–398. doi: 10.1097/00001665-200107000-00018. [DOI] [PubMed] [Google Scholar]
- 92.Khonsari R.H., Ohazama A., Raouf R., Kawasaki M., Kawasaki K., Porntaveetus T., Ghafoor S., Hammond P., Suttie M., Odri G.A. Multiple postnatal craniofacial anomalies are characterized by conditional loss of polycystic kidney disease 2 (Pkd2) Hum. Mol. Genet. 2013;22:1873–1885. doi: 10.1093/hmg/ddt041. [DOI] [PubMed] [Google Scholar]
- 93.Tabler J.M., Barrell W.B., Szabo-Rogers H.L., Healy C., Yeung Y., Perdiguero E.G., Schulz C., Yannakoudakis B.Z., Mesbahi A., Wlodarczyk B. Fuz mutant mice reveal shared mechanisms between ciliopathies and FGF-related syndromes. Dev. Cell. 2013;25:623–635. doi: 10.1016/j.devcel.2013.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Liu B., Chen S., Cheng D., Jing W., Helms J.A. Primary cilia integrate hedgehog and Wnt signaling during tooth development. J. Dent. Res. 2014;93:475–482. doi: 10.1177/0022034514528211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kido S., Kuriwaka-Kido R., Imamura T., Ito Y., Inoue D., Matsumoto T. Mechanical stress induces Interleukin-11 expression to stimulate osteoblast differentiation. Bone. 2009;45:1125–1132. doi: 10.1016/j.bone.2009.07.087. [DOI] [PubMed] [Google Scholar]
- 96.Sims N.A., Jenkins B.J., Nakamura A., Quinn J.M., Li R., Gillespie M.T., Ernst M., Robb L., Martin T.J. Interleukin-11 receptor signaling is required for normal bone remodeling. J. Bone Miner. Res. 2005;20:1093–1102. doi: 10.1359/JBMR.050209. [DOI] [PubMed] [Google Scholar]
- 97.Behr B., Longaker M.T., Quarto N. Differential activation of canonical Wnt signaling determines cranial sutures fate: a novel mechanism for sagittal suture craniosynostosis. Dev. Biol. 2010;344:922–940. doi: 10.1016/j.ydbio.2010.06.009. [DOI] [PubMed] [Google Scholar]
- 98.Behr B., Longaker M.T., Quarto N. Craniosynostosis of coronal suture in twist1 mice occurs through endochondral ossification recapitulating the physiological closure of posterior frontal suture. Front. Physiol. 2011;2:37. doi: 10.3389/fphys.2011.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Goodnough L.H., Chang A.T., Treloar C., Yang J., Scacheri P.C., Atit R.P. Twist1 mediates repression of chondrogenesis by β-catenin to promote cranial bone progenitor specification. Development. 2012;139:4428–4438. doi: 10.1242/dev.081679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Jin H., Nachury M.V. The BBSome. Curr. Biol. 2009;19:R472–R473. doi: 10.1016/j.cub.2009.04.015. [DOI] [PubMed] [Google Scholar]
- 101.Minegishi Y., Karasuyama H. Hyperimmunoglobulin E syndrome and tyrosine kinase 2 deficiency. Curr. Opin. Allergy Clin. Immunol. 2007;7:506–509. doi: 10.1097/ACI.0b013e3282f1baea. [DOI] [PubMed] [Google Scholar]
- 102.Ibrahimi O.A., Zhang F., Eliseenkova A.V., Linhardt R.J., Mohammadi M. Proline to arginine mutations in FGF receptors 1 and 3 result in Pfeiffer and Muenke craniosynostosis syndromes through enhancement of FGF binding affinity. Hum. Mol. Genet. 2004;13:69–78. doi: 10.1093/hmg/ddh011. [DOI] [PubMed] [Google Scholar]
- 103.Yu K., Ornitz D.M. Uncoupling fibroblast growth factor receptor 2 ligand binding specificity leads to Apert syndrome-like phenotypes. Proc. Natl. Acad. Sci. USA. 2001;98:3641–3643. doi: 10.1073/pnas.081082498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ibrahimi O.A., Zhang F., Eliseenkova A.V., Itoh N., Linhardt R.J., Mohammadi M. Biochemical analysis of pathogenic ligand-dependent FGFR2 mutations suggests distinct pathophysiological mechanisms for craniofacial and limb abnormalities. Hum. Mol. Genet. 2004;13:2313–2324. doi: 10.1093/hmg/ddh235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Mansour S.L., Li C., Urness L.D. Genetic rescue of Muenke syndrome model hearing loss reveals prolonged FGF-dependent plasticity in cochlear supporting cell fates. Genes Dev. 2013;27:2320–2331. doi: 10.1101/gad.228957.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ibrahimi O.A., Eliseenkova A.V., Plotnikov A.N., Yu K., Ornitz D.M., Mohammadi M. Structural basis for fibroblast growth factor receptor 2 activation in Apert syndrome. Proc. Natl. Acad. Sci. USA. 2001;98:7182–7187. doi: 10.1073/pnas.121183798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Ma L., Golden S., Wu L., Maxson R. The molecular basis of Boston-type craniosynostosis: the Pro148-->His mutation in the N-terminal arm of the MSX2 homeodomain stabilizes DNA binding without altering nucleotide sequence preferences. Hum. Mol. Genet. 1996;5:1915–1920. doi: 10.1093/hmg/5.12.1915. [DOI] [PubMed] [Google Scholar]
- 108.Naski M.C., Wang Q., Xu J., Ornitz D.M. Graded activation of fibroblast growth factor receptor 3 by mutations causing achondroplasia and thanatophoric dysplasia. Nat. Genet. 1996;13:233–237. doi: 10.1038/ng0696-233. [DOI] [PubMed] [Google Scholar]
- 109.Robertson S.C., Meyer A.N., Hart K.C., Galvin B.D., Webster M.K., Donoghue D.J. Activating mutations in the extracellular domain of the fibroblast growth factor receptor 2 function by disruption of the disulfide bond in the third immunoglobulin-like domain. Proc. Natl. Acad. Sci. USA. 1998;95:4567–4572. doi: 10.1073/pnas.95.8.4567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Chen H., Ma J., Li W., Eliseenkova A.V., Xu C., Neubert T.A., Miller W.T., Mohammadi M. A molecular brake in the kinase hinge region regulates the activity of receptor tyrosine kinases. Mol. Cell. 2007;27:717–730. doi: 10.1016/j.molcel.2007.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Placone J., Hristova K. Direct assessment of the effect of the Gly380Arg achondroplasia mutation on FGFR3 dimerization using quantitative imaging FRET. PLoS ONE. 2012;7:e46678. doi: 10.1371/journal.pone.0046678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Plaisancié J., Collet C., Pelletier V., Perdomo Y., Studer F., Fradin M., Schaefer E., Speeg-Schatz C., Bloch-Zupan A., Flori E., Dollfus H. MSX2 gene duplication in a patient with eye development defects. Ophthalmic Genet. 2014 doi: 10.3109/13816810.2014.886270. Published online March 25, 2014. [DOI] [PubMed] [Google Scholar]
- 113.Hajihosseini M.K., Wilson S., De Moerlooze L., Dickson C. A splicing switch and gain-of-function mutation in FgfR2-IIIc hemizygotes causes Apert/Pfeiffer-syndrome-like phenotypes. Proc. Natl. Acad. Sci. USA. 2001;98:3855–3860. doi: 10.1073/pnas.071586898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Oldridge M., Zackai E.H., McDonald-McGinn D.M., Iseki S., Morriss-Kay G.M., Twigg S.R.F., Johnson D., Wall S.A., Jiang W., Theda C. De novo alu-element insertions in FGFR2 identify a distinct pathological basis for Apert syndrome. Am. J. Hum. Genet. 1999;64:446–461. doi: 10.1086/302245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Davy A., Aubin J., Soriano P. Ephrin-B1 forward and reverse signaling are required during mouse development. Genes Dev. 2004;18:572–583. doi: 10.1101/gad.1171704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Compagni A., Logan M., Klein R., Adams R.H. Control of skeletal patterning by ephrinB1-EphB interactions. Dev. Cell. 2003;5:217–230. doi: 10.1016/s1534-5807(03)00198-9. [DOI] [PubMed] [Google Scholar]
- 117.Hatch N.E., Hudson M., Seto M.L., Cunningham M.L., Bothwell M. Intracellular retention, degradation, and signaling of glycosylation-deficient FGFR2 and craniosynostosis syndrome-associated FGFR2C278F. J. Biol. Chem. 2006;281:27292–27305. doi: 10.1074/jbc.M600448200. [DOI] [PubMed] [Google Scholar]
- 118.Bonaventure J., Horne W.C., Baron R. The localization of FGFR3 mutations causing thanatophoric dysplasia type I differentially affects phosphorylation, processing and ubiquitylation of the receptor. FEBS J. 2007;274:3078–3093. doi: 10.1111/j.1742-4658.2007.05835.x. [DOI] [PubMed] [Google Scholar]
- 119.Lievens P.M.-J., Mutinelli C., Baynes D., Liboi E. The kinase activity of fibroblast growth factor receptor 3 with activation loop mutations affects receptor trafficking and signaling. J. Biol. Chem. 2004;279:43254–43260. doi: 10.1074/jbc.M405247200. [DOI] [PubMed] [Google Scholar]
- 120.Twigg S.R.F., Healy C., Babbs C., Sharpe J.A., Wood W.G., Sharpe P.T., Morriss-Kay G.M., Wilkie A.O.M. Skeletal analysis of the Fgfr3(P244R) mouse, a genetic model for the Muenke craniosynostosis syndrome. Dev. Dyn. 2009;238:331–342. doi: 10.1002/dvdy.21790. [DOI] [PubMed] [Google Scholar]
- 121.Babbs C., Stewart H.S., Williams L.J., Connell L., Goriely A., Twigg S.R.F., Smith K., Lester T., Wilkie A.O.M. Duplication of the EFNB1 gene in familial hypertelorism: imbalance in ephrin-B1 expression and abnormal phenotypes in humans and mice. Hum. Mutat. 2011;32:930–938. doi: 10.1002/humu.21521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Holmes G. The role of vertebrate models in understanding craniosynostosis. Childs Nerv. Syst. 2012;28:1471–1481. doi: 10.1007/s00381-012-1844-3. [DOI] [PubMed] [Google Scholar]
- 123.Chen L., Li D., Li C., Engel A., Deng C.X. A Ser252Trp [corrected] substitution in mouse fibroblast growth factor receptor 2 (Fgfr2) results in craniosynostosis. Bone. 2003;33:169–178. doi: 10.1016/s8756-3282(03)00222-9. [DOI] [PubMed] [Google Scholar]
- 124.Yin L., Du X., Li C., Xu X., Chen Z., Su N., Zhao L., Qi H., Li F., Xue J. A Pro253Arg mutation in fibroblast growth factor receptor 2 (Fgfr2) causes skeleton malformation mimicking human Apert syndrome by affecting both chondrogenesis and osteogenesis. Bone. 2008;42:631–643. doi: 10.1016/j.bone.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 125.Goriely A., Wilkie A.O.M. Paternal age effect mutations and selfish spermatogonial selection: causes and consequences for human disease. Am. J. Hum. Genet. 2012;90:175–200. doi: 10.1016/j.ajhg.2011.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Gong S.G. The Fgfr2 W290R mouse model of Crouzon syndrome. Childs Nerv. Syst. 2012;28:1495–1503. doi: 10.1007/s00381-012-1792-y. [DOI] [PubMed] [Google Scholar]
- 127.Eswarakumar V.P., Horowitz M.C., Locklin R., Morriss-Kay G.M., Lonai P. A gain-of-function mutation of Fgfr2c demonstrates the roles of this receptor variant in osteogenesis. Proc. Natl. Acad. Sci. USA. 2004;101:12555–12560. doi: 10.1073/pnas.0405031101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Wang Y., Zhou X., Oberoi K., Phelps R., Couwenhoven R., Sun M., Rezza A., Holmes G., Percival C.J., Friedenthal J. p38 Inhibition ameliorates skin and skull abnormalities in Fgfr2 Beare-Stevenson mice. J. Clin. Invest. 2012;122:2153–2164. doi: 10.1172/JCI62644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zhou Y.-X., Xu X., Chen L., Li C., Brodie S.G., Deng C.-X. A Pro250Arg substitution in mouse Fgfr1 causes increased expression of Cbfa1 and premature fusion of calvarial sutures. Hum. Mol. Genet. 2000;9:2001–2008. doi: 10.1093/hmg/9.13.2001. [DOI] [PubMed] [Google Scholar]
- 130.Carver E.A., Oram K.F., Gridley T. Craniosynostosis in Twist heterozygous mice: a model for Saethre-Chotzen syndrome. Anat. Rec. 2002;268:90–92. doi: 10.1002/ar.10124. [DOI] [PubMed] [Google Scholar]
- 131.Holmes G., Rothschild G., Roy U.B., Deng C.X., Mansukhani A., Basilico C. Early onset of craniosynostosis in an Apert mouse model reveals critical features of this pathology. Dev. Biol. 2009;328:273–284. doi: 10.1016/j.ydbio.2009.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Lomri A., Lemonnier J., Hott M., de Parseval N., Lajeunie E., Munnich A., Renier D., Marie P.J. Increased calvaria cell differentiation and bone matrix formation induced by fibroblast growth factor receptor 2 mutations in Apert syndrome. J. Clin. Invest. 1998;101:1310–1317. [PMC free article] [PubMed] [Google Scholar]
- 133.Mathijssen I.M.J., van Splunder J., Vermeij-Keers C., Pieterman H., de Jong T.H.R., Mooney M.P., Vaandrager J.M. Tracing craniosynostosis to its developmental stage through bone center displacement. J. Craniofac. Genet. Dev. Biol. 1999;19:57–63. [PubMed] [Google Scholar]
- 134.Holmes G., Basilico C. Mesodermal expression of Fgfr2S252W is necessary and sufficient to induce craniosynostosis in a mouse model of Apert syndrome. Dev. Biol. 2012;368:283–293. doi: 10.1016/j.ydbio.2012.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Cohen M.M., Jr., Kreiborg S. Suture formation, premature sutural fusion, and suture default zones in Apert syndrome. Am. J. Med. Genet. 1996;62:339–344. doi: 10.1002/(SICI)1096-8628(19960424)62:4<339::AID-AJMG3>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- 136.Khonsari R.H., Delezoide A.L., Kang W., Hébert J.M., Bessières B., Bodiguel V., Collet C., Legeai-Mallet L., Sharpe P.T., Fallet-Bianco C. Central nervous system malformations and deformations in FGFR2-related craniosynostosis. Am. J. Med. Genet. A. 2012;158A:2797–2806. doi: 10.1002/ajmg.a.35598. [DOI] [PubMed] [Google Scholar]
- 137.Lajeunie E., Heuertz S., El Ghouzzi V., Martinovic J., Renier D., Le Merrer M., Bonaventure J. Mutation screening in patients with syndromic craniosynostoses indicates that a limited number of recurrent FGFR2 mutations accounts for severe forms of Pfeiffer syndrome. Eur. J. Hum. Genet. 2006;14:289–298. doi: 10.1038/sj.ejhg.5201558. [DOI] [PubMed] [Google Scholar]
- 138.Hoefkens M.F., Vermeij-Keers C., Vaandrager J.M. Crouzon syndrome: phenotypic signs and symptoms of the postnatally expressed subtype. J. Craniofac. Surg. 2004;15:233–240. doi: 10.1097/00001665-200403000-00013. discussion 241–242. [DOI] [PubMed] [Google Scholar]
- 139.Connolly J.P., Gruss J., Seto M.L., Whelan M.F., Ellenbogen R., Weiss A., Buchman S.R., Cunningham M.L. Progressive postnatal craniosynostosis and increased intracranial pressure. Plast. Reconstr. Surg. 2004;113:1313–1323. doi: 10.1097/01.prs.0000111593.96440.30. [DOI] [PubMed] [Google Scholar]
- 140.Liu J., Nam H.K., Wang E., Hatch N.E. Further analysis of the Crouzon mouse: effects of the FGFR2(C342Y) mutation are cranial bone-dependent. Calcif. Tissue Int. 2013;92:451–466. doi: 10.1007/s00223-013-9701-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Neben C.L., Idoni B., Salva J.E., Tuzon C.T., Rice J.C., Krakow D., Merrill A.E. Bent bone dysplasia syndrome reveals nucleolar activity for FGFR2 in ribosomal DNA transcription. Hum. Mol. Genet. 2014;23:5659–5671. doi: 10.1093/hmg/ddu282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Eswarakumar V.P., Özcan F., Lew E.D., Bae J.H., Tomé F., Booth C.J., Adams D.J., Lax I., Schlessinger J. Attenuation of signaling pathways stimulated by pathologically activated FGF-receptor 2 mutants prevents craniosynostosis. Proc. Natl. Acad. Sci. USA. 2006;103:18603–18608. doi: 10.1073/pnas.0609157103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Shukla V., Coumoul X., Wang R.-H., Kim H.-S., Deng C.-X. RNA interference and inhibition of MEK-ERK signaling prevent abnormal skeletal phenotypes in a mouse model of craniosynostosis. Nat. Genet. 2007;39:1145–1150. doi: 10.1038/ng2096. [DOI] [PubMed] [Google Scholar]
- 144.Takenouchi T., Sakamoto Y., Miwa T., Torii C., Kosaki R., Kishi K., Takahashi T., Kosaki K. Severe craniosynostosis with Noonan syndrome phenotype associated with SHOC2 mutation: clinical evidence of crosslink between FGFR and RAS signaling pathways. Am. J. Med. Genet. A. 2014;164A:2869–2872. doi: 10.1002/ajmg.a.36705. [DOI] [PubMed] [Google Scholar]
- 145.Connerney J., Andreeva V., Leshem Y., Mercado M.A., Dowell K., Yang X., Lindner V., Friesel R.E., Spicer D.B. Twist1 homodimers enhance FGF responsiveness of the cranial sutures and promote suture closure. Dev. Biol. 2008;318:323–334. doi: 10.1016/j.ydbio.2008.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Shishido E., Higashijima S., Emori Y., Saigo K. Two FGF-receptor homologues of Drosophila: one is expressed in mesodermal primordium in early embryos. Development. 1993;117:751–761. doi: 10.1242/dev.117.2.751. [DOI] [PubMed] [Google Scholar]
- 147.Harfe B.D., Vaz Gomes A., Kenyon C., Liu J., Krause M., Fire A. Analysis of a Caenorhabditis elegans Twist homolog identifies conserved and divergent aspects of mesodermal patterning. Genes Dev. 1998;12:2623–2635. doi: 10.1101/gad.12.16.2623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Eggenschwiler J.T., Bulgakov O.V., Qin J., Li T., Anderson K.V. Mouse Rab23 regulates hedgehog signaling from smoothened to Gli proteins. Dev. Biol. 2006;290:1–12. doi: 10.1016/j.ydbio.2005.09.022. [DOI] [PubMed] [Google Scholar]
- 149.Eggenschwiler J.T., Espinoza E., Anderson K.V. Rab23 is an essential negative regulator of the mouse Sonic hedgehog signalling pathway. Nature. 2001;412:194–198. doi: 10.1038/35084089. [DOI] [PubMed] [Google Scholar]
- 150.Marucci D.D., Johnston C.P., Anslow P., Jayamohan J., Richards P.G., Wilkie A.O.M., Wall S.A. Implications of a vertex bulge following modified strip craniectomy for sagittal synostosis. Plast. Reconstr. Surg. 2008;122:217–224. doi: 10.1097/PRS.0b013e3181774240. [DOI] [PubMed] [Google Scholar]
- 151.Wilkie A.O.M. Cancer drugs to treat birth defects. Nat. Genet. 2007;39:1057–1059. doi: 10.1038/ng0907-1057. [DOI] [PubMed] [Google Scholar]
- 152.Melville H., Wang Y., Taub P.J., Jabs E.W. Genetic basis of potential therapeutic strategies for craniosynostosis. Am. J. Med. Genet. A. 2010;152A:3007–3015. doi: 10.1002/ajmg.a.33703. [DOI] [PubMed] [Google Scholar]
- 153.Justice C.M., Yagnik G., Kim Y., Peter I., Jabs E.W., Erazo M., Ye X., Ainehsazan E., Shi L., Cunningham M.L. A genome-wide association study identifies susceptibility loci for nonsyndromic sagittal craniosynostosis near BMP2 and within BBS9. Nat. Genet. 2012;44:1360–1364. doi: 10.1038/ng.2463. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.