Abstract
The protective role of resveratrol in myocardial ischemia/reperfusion is not well understood. The aim of this study was to investigate whether resveratrol modulates inflammation and oxidative stress and the possible role of nuclear factor erythroid 2-related factor 2 (Nrf2)/antioxidant response element (ARE) pathway in an ischemia/reperfusion injured rat heart model. Rats were randomly exposed to sham operation, myocardial ischemia/reperfusion (MI/R) alone, and MI/R + resveratrol. The results demonstrated that compared to MI/R, resveratrol improved cardiac function, reduced myocardial infarction area, myocardial myeloperoxidase (MPO) levels, serum creatinine kinase (CK) and lactate dehydrogenase (LDH) levels. Resveratrol also markedly enhanced the activities of antioxidant enzymes superoxide dismutase (SOD) and glutathione peroxidase (GSH-PX), and reduced the level of malondialdehyde (MDA) in MI/R rats. Resveratrol also enhanced levels of Nrf2 and heme oxygenase-1. In summary, these results demonstrated that resveratrol exerted significant antioxidant and cardioprotective effects following myocardial ischemia, possibly through the activation of the Nrf2/ARE signaling pathway.
Keywords: Resveratrol, myocardial ischemia/reperfusion injury, SOD, MDA, Nrf-2, HO-1
Introduction
Ischemia and reperfusion (IR) injury is a major cause of morbidity and mortality after cardiac operations [1,2] and myocardial infarctions [3]. The inflammatory reaction induced by ischemia/reperfusion is one of the most important links in the myocardial ischemia-reperfusion injury [4]. In the process of inflammation, various cytokines are released, including tumor necrosis factor α (TNF-α), interleukin 6 (IL-6) and IL-8, etc [5]. TNF-α can trigger the inflammatory reaction caused by myocardial ischemia-reperfusion. In addition, vascular endothelial cell injury, and inflammatory cells, such as neutrophils, activated by cytokines and adhesion molecules are also included in inflammation. So TNF-α activity and the amount of neutrophil infiltration can be considered as the indicators of inflammatory reaction. Besides, oxidative stress is also involved in myocardial ischemia/reperfusion injury [6]. The reperfusion leads to the large production of ROS, which aggravates the injury of the myocardium.
Recent studies have demonstrated that nuclear factor erythroid 2-related factor 2 (Nrf2), a key transcription factor, plays an indispensable role in the induction of endogenous antioxidant enzymes against oxidative stress. Under physiological conditions, Nrf2 is localized within the cytoplasm by binding to its negative regulator, Kelch-like ECH associating protein 1 (Keap1), which promotes Nrf2 ubiquitination by the Cul3-Rbk1 complex and subsequent degradation by the proteasome [7]. However, upon exposure to ROS, Nrf2 is liberated from the Keap1-Nrf2 complex and translocates from the cytoplasm to the nucleus, where it sequentially binds to the antioxidant response element (ARE), a regulatory enhancer region within gene promoters. This binding triggers the production of many phase II detoxifying and antioxidant enzyme genes such as heme oxygenase 1 (HO-1) and NAD (P) H: quinone oxidoreductase 1 (NQO1), which protect cells against oxidative stress as well as a wide range of other toxins [8,9].
Recently, many reports have demonstrated that Nrf2 plays a critical role in the induction of endogenous antioxidant enzymes against oxidative damage in a variety of diseases. Nrf2 protects cardiac fibroblasts and cardiomyocytes against oxidative stress by increasing detoxification pathways and antioxidant potentials [10,11]. In addition, Nrf2 protects the lungs from oxidative injury caused by bleomycin and environmental oxidants including hyperoxia, diesel exhaust particles and smoking [12-14].
Resveratrol (Res) is a polyphenolic compound which mainly exists in red grapes and wine. In a previous epidemiological study [15] on the relationship between eating habits and coronary heart disease, an interesting phenomenon suggests that in all developed countries, the French consume the most quantity of wine on the average, but have the lowest morbidity of coronary heart disease. This phenomenon is called the “French paradox”, which has probably benefited from Res in wine. Res has extensive pharmacological effects, including anticancer [16-18], treating diabetic nephropathy [19] and protecting neurons [20,21]. Recent data indicates that the Nrf2/ARE pathway plays a central role in the protective effect against oxidative and apoptotic damage [22,23]. Therefore, the present study aims to investigate the role of Nrf2/ARE signaling pathway in the cardioprotective effect of Res in a rat model of myocardial ischemia-reperfusion (MI/R).
Materials and methods
Reagents
Resveratrol was purchased from Sigma Chemical Co. (St. Louis, MO, USA). MPO assay kit, CK test kit and LDH assay kit were purchased from JianCheng Bioengineering Institute (Nanjing, China). Sodium pentobarbital was purchased from Sigma Chemical Co. (St. Louis, MO, USA). BCA Protein Assay reagent kit was purchased from Novagen (Madison, WI).
Animals
Thirty adult male Sprague-Dawley rats (250-300 g) were purchased from the Center of Experimental Animal in the Fourth Military Medical University, China. All animals used in this study were cared for in accordance with the Guide for the Care and Use of Laboratory Animals published by the United States National Institute of Health (NIH publication no. 85-23, revised 1996), and all procedures were approved by the Committee of Experimental Animals of the Fourth Military Medical University.
Myocardial ischemia-reperfusion model and experimental protocol
Male Sprague-Dawley rats (250-300 g) were anesthetized i.p. with sodium pentobarbital (Sigma, St. Louis, USA, 40 mg/kg). Myocardial ischemia was produced by exteriorizing the heart with a left thoracic incision followed by a slipknot (5-0 silk) around the left anterior descending coronary artery (LAD). After 30 min of ischemia, the slipknot was released and the animal received 120 min of reperfusion.
Rats were randomly assigned to three experimental groups. There were 10 rats in each group: (1) sham group: silk was drilled underneath the LAD but the LAD was not ligated; (2) MI/R group: LAD was ligated for 30 min and then allowed 120 min reperfusion with receiving vehicle (0.9% NaCl i.v.); (3) MI/R + Res group: Res (100 μmol/L, i.v.) was administered 5 min before reperfusion.
Detection of cardiac function
Rats were anesthetized i.p. with sodium pentobarbital (Sigma, St. Louis, USA, 40 mg/kg), and a catheter was inserted into the left ventricle through the right common carotid artery for measurement of left ventricular function, which included left ventricular ejection fraction (LVEF), left ventricular end-diastolic pressure (LVEDP) and the maximal rate of rise and decline of ventricular pressure (± dp/dt [max]).
Assay of myocardial infarct area
After reperfusion, myocardial infarct size was determined by means of a double-staining technique and a digital imaging system (infarct area/area at risk × 100%) [24]. After reperfusing, coronary blood flow was again blocked and Evans blue (2%, 4 ml) was injected by the rapid distribution of the right ventricle into the body. The heart was quickly removed to a -20°C refrigerator for cryopreservation. The heart was cut into 1 mm slices, placed in 1% 2,3,5-triphenyltetrazolium chloride (TTC) solution, incubated for 15 min, and then placed in 4% formaldehyde solution overnight. Evans blue stained area (blue staining, non-ischemic area), TTC stained area (red staining, ischemic area) and non-TTC stained area (white, infarct area) was analyzed with a digital imaging system by computer. Myocardial infarct area (infarct area/area at risk%, INF/AAR%) were calculated.
Detection of creatine kinase (CK) activity
After reperfusion, blood was taken from the carotid artery and was placed at room temperature for 30 min. Then, the serum was collected by centrifugation and placed at -70°C for preservation. According to manufacturer’s instruction, the CK test kit was utilized to detect the serum CK activity.
Determination of lactate dehydrogenase (LDH) level
After reperfusion, blood was taken from the carotid artery and was placed at room temperature for 30 min. Then, the serum was collected by centrifugation and placed at -70°C for preservation. The extent of cell injury was monitored by measuring LDH leakage. According to the manufacturer’s instruction, the CK test kit was utilized to detect the serum LDH level.
Determination of myeloperoxidase (MPO) level
After reperfusion, the myocardial tissue was placed at -70°C for preservation. MPO test kit was used to detect level of MPO in the myocardial tissue according to manufacturer’s instruction.
Detection of SOD, GSH-PX activity and MDA content
After reperfusion, myocardial tissue was homogenized in ice cold phosphate buffer to make a 10% homogenate. Then the homogenate was centrifuged at 3000 rpm for 15 min. Superoxide dismutase (SOD), glutathione peroxidase (GSH-PX), and malondialdehyde (MDA) in the supernatant were measured by using commercially available kits. All assays were conducted according to the manufacturer’s instructions.
Western blot analysis
Protein extracts were prepared from ischemic myocardium or heart in sham group by homogenization in a RIPA buffer. Protein concentration was assayed using the BCA Protein Assay reagent kit (Novagen, Madison, WI), and equal amounts (50 μg) of protein per sample were separated by electrophoresis on a 10% polyacrylamide gel and electrotransferred to a nitrocellulose membrane. The nonspecific binding of antibodies was blocked using TBS with 5% nonfat milk and with 0.1% Tween-20 for 2 h at room temperature. Membranes were then incubated overnight (4°C) with the primary antibody at the appropriate dilution (rabbit anti-Nrf2, 1:200, Santa Cruz Biotechnology; rabbit anti-HO-1, 1:2000, Santa Cruz Biotechnology). Protein loading was normalized by Western blotting to histone 1 (H1, 1:1000, Santa Cruz Biotechnology) or anti-β-actin (1:2000; Santa Cruz Biotechnology) in blocking buffer overnight at 4°C. Subsequently, the membranes were incubated for 1 h at room temperature with the corresponding horseradish peroxidase-conjugated secondary antibodies (1:3000, goat anti-rabbit; 1:5000, goat anti-mouse, both from Santa Cruz Biotechnology). Finally, immunoreactive bands were detected by chemiluminescence using the Amersham ECL Plus Western Blotting Detection kit (GE Healthcare Bio-Sciences, Piscataway, NJ), according to the manufacturer’s instructions. Nrf2 expression levels were normalized to H1, while HO-1 expression level was normalized to β-actin.
Statistical analysis
Data is presented as means ± SD. The significance of differences among groups was evaluated by a Student’s t-test for unpaired data or Dunnett’s t-test for multiple comparisons preceded by one-way analysis of variance (ANOVA). For all test, P < 0.05 was considered as statistically significant.
Results
Resveratrol ameliorated cardiac dysfunction induced by MI/R
Compared with the MI/R group, resveratrol significantly elevated LVEF, +dp/dt max and -dp/dt max and decreased LVEDP (P < 0.05) (Figure 1).
Figure 1.
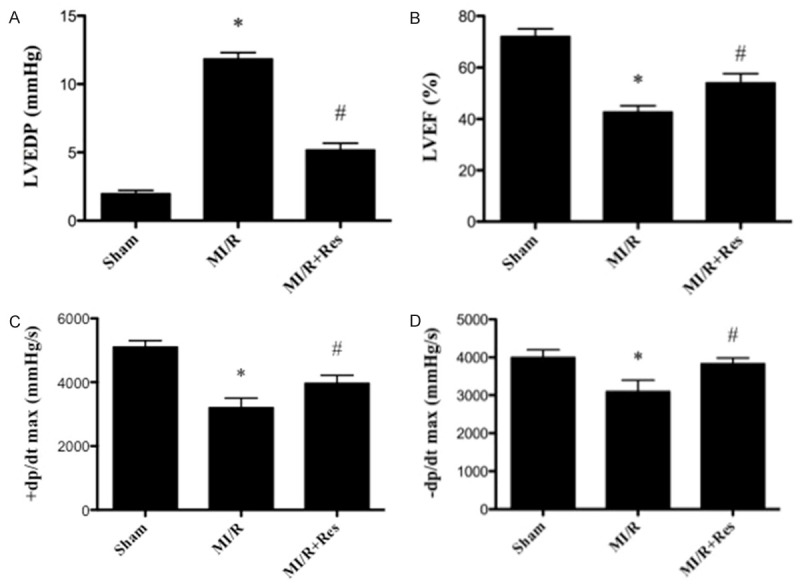
Effect of resveratrol on cardiac function. A. The effect of resveratrol on LVEDP. B. The effect of resveratrol on LVEF. C. The effect of resveratrol on +dp/dx max. D. The effect of resveratrol on -dp/dx max. LVEF, left ventricle ejection fraction; LVEDP, left ventricle end-diastolic pressure. Data were expressed as mean ± SD (n = 10 in each group). *P < 0.05 versus the sham group, #P < 0.05 versus MI/R group.
Resveratrol reduced the myocardial infarction area induced by MI/R
MI/R induced a significant infarction area. Compared with the MI/R group, resveratrol reduced myocardial infarction area significantly (P < 0.05) (Figure 2).
Figure 2.
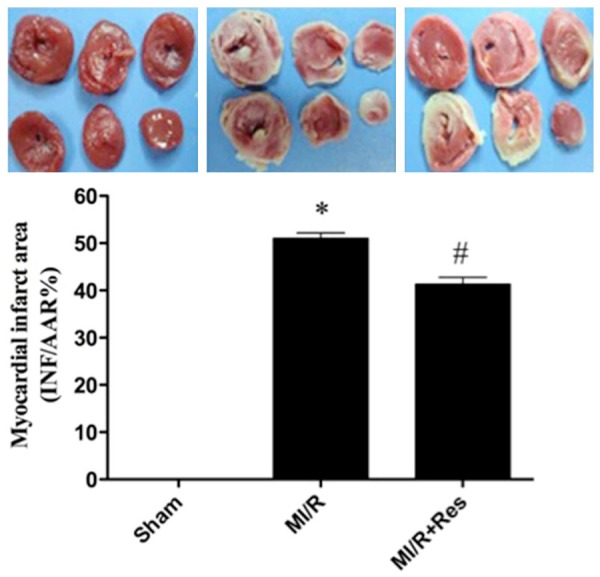
Effect of resveratrol on the myocardial infarction area induced by MI/R. Data were expressed as mean ± SD (n = 10 in each group). *P < 0.05 versus the sham group, #P < 0.05 versus MI/R group.
Resveratrol reduced the activity of serum CK in MI/R rats
As shown in Figure 3, the activity of CK increased significantly in the MI/R group compared with the sham group (P < 0.05). CK activity decreased significantly in the MI/R + Res group compared with the MI/R group (P < 0.05).
Figure 3.
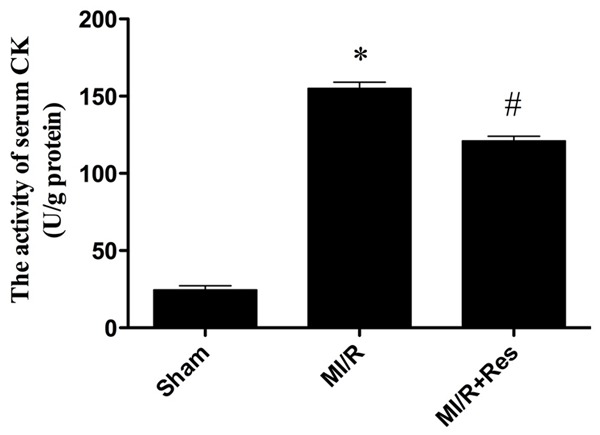
The comparison of CK activity in each group. The CK activity in the sham group was relatively lower, while the CK activity in MI/R group was significantly increased, which was inhibited by resveratrol. Data were expressed as mean ± SD (n = 10 in each group). *P < 0.05 versus the sham group, #P < 0.05 versus MI/R group.
Resveratrol reduced LDH level in MI/R rats
As shown in Figure 4, the activity of LDH increased significantly in the MI/R group compared with the sham group (P < 0.05). LDH activity decreased significantly in the MI/R + Res group compared with the MI/R group (P < 0.05).
Figure 4.
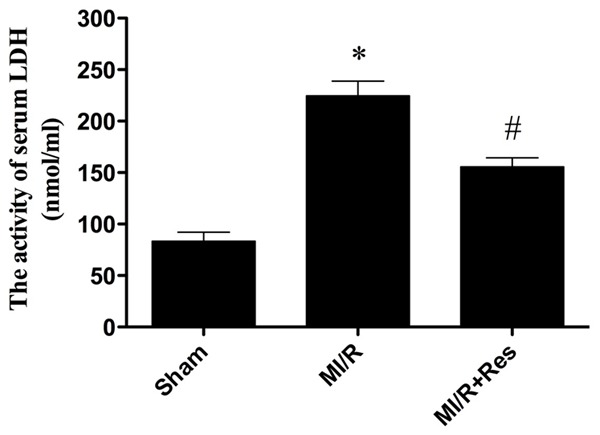
The comparison of LDH activity in each group. The LDH activity in the sham group was relatively lower, while the LDH activity in MI/R group was significantly increased, which was inhibited by resveratrol. Data were expressed as mean ± SD (n = 10 in each group). *P < 0.05 versus the sham group, #P < 0.05 versus MI/R group.
Resveratrol inhibited neutrophil infiltration in MI/R tissue
Neutrophil contains a certain amount of myeloperoxidase (MPO), accounting for 5% of dry cell weight. As shown in Figure 5, the MPO activity in the sham group was relatively lower, while the MPO activity in MI/R group was significantly increased (P < 0.05). Resveratrol significantly decreased myocardial MPO activity (P < 0.05) (Figure 5).
Figure 5.
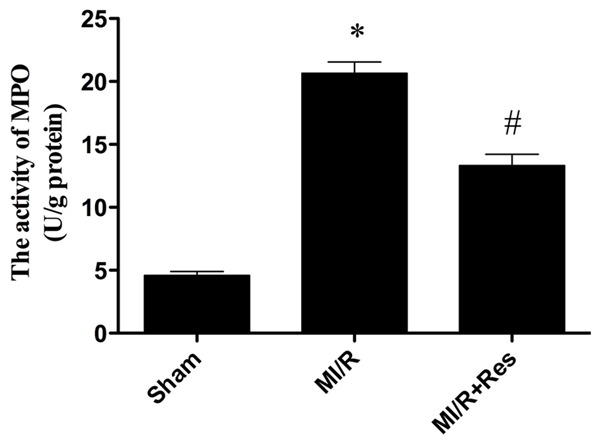
The comparison of MPO activity in each group. The MPO activity in the sham group was relatively lower, while the MPO activity in MI/R group was significantly increased, which was blocked by resveratrol. Data were expressed as mean ± SD (n = 10 in each group). *P < 0.05 versus the sham group, #P < 0.05 versus MI/R group.
Resveratrol elevated antioxidant enzymes activities and decreased MDA content
To investigate whether Resveratrol affects oxidative stress damage, we evaluated the activities of antioxidant enzyme and the content of MDA. The activities of GSH-PX and SOD were decreased significantly in the MI/R group as compared with the sham group (P < 0.05). However, Resveratrol treatment induced significant elevation of GSH-PX and SOD activities compared with the MI/R group (P < 0.05). The content of MDA is an index of lipid peroxidation, which increased significantly after myocardial I/R injury (P < 0.05). Resveratrol treatment significantly decreased the MDA content compared with the MI/R group (P < 0.05) (Figure 6).
Figure 6.
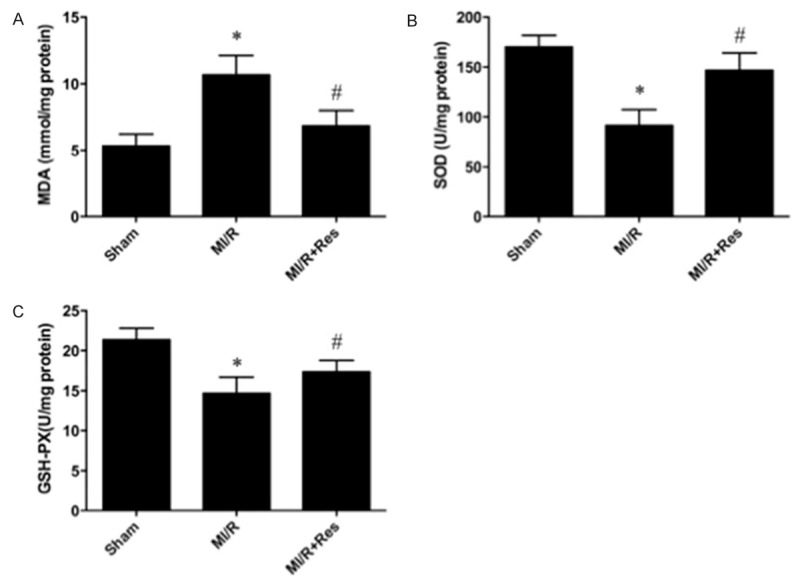
Resveratrol ameliorates oxidative stress after myocardial ischemia/reperfusion injury. Resveratrol significantly decreased (A) the content of malondialdehyde (MDA) and (B) enhanced superoxide dismutase (SOD); and (C) glutathione peroxidase (GSH-PX) activities in ischemia reperfusion rats’ heart. Data were expressed as mean ± SD (n = 10 in each group). *P < 0.05 versus the sham group, #P < 0.05 versus MI/R group.
Effects of resveratrol on Nrf2 and HO-1 Expressions in heart subjected to ischemia-reperfusion
Western blot analysis showed weak signals for Nrf2 and HO-1 in the sham group. In contrast, significant increases in protein expression for Nrf2 and HO-1 were found in the MI/R group (P < 0.05). Compared with the MI/R group, the Western blot signals were intensified in MI/R + Res group (P < 0.05) (Figure 7).
Figure 7.
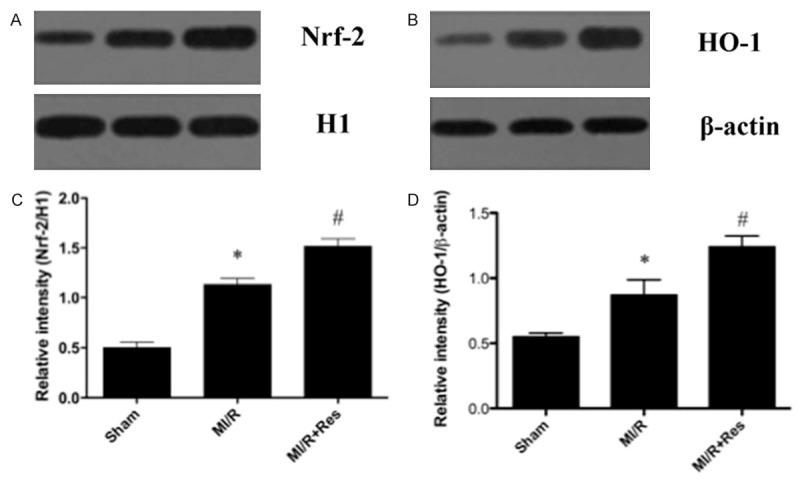
Western blot analysis for Nrf2 and HO-1 expression. A, B. Representative photographs of the Western blot show Nrf2 and HO-1 protein levels in sham, MI/R and MI/R + Res respectively. C, D. The graphs show that Nrf2 and HO-1, and protein levels were increased after MI/R and more significantly induced by treatment with resveratrol. Data were expressed as mean ± SD (n = 10 in each group). *P < 0.05 versus the sham group, #P < 0.05 versus MI/R group.
Discussion
In the present study, we demonstrated that resveratrol pretreatment has protective effects on myocardial I/R injury in a rat model of ischemia reperfusion demonstrated by improved cardiac function, reduced infarct area, reduced CK, LDH and MPO activity, increased GSH-PX and SOD biosynthesis and decreased MDA content. The mechanism was possibly attributed to activating Nrf2 expression.
Inflammatory reaction plays an important role in myocardial ischemia/reperfusion injury [4]. The release of inflammatory cytokines and the aggregation and infiltration of inflammatory cells are the key steps in inflammation [25]. MI/R injury seems to be induced in part by neutrophil activation. The underlying mechanisms include: (1) Cell damage caused by the release of oxygen free radicals, proteolytic enzymes, and cytotoxic substances. (2) The released inflammatory mediators cause vascular endothelial cell damage, increased vascular permeability, and edema. (3) Further activation of inflammatory cells increase further the inflammatory response [26]. (4) Neutrophil adhesion to vascular endothelium and small blood vessels occlusion result in no-reflow phenomenon.
Under physiological conditions ROS are generated at low levels and play important roles in signaling and metabolic pathways [27], however, under pathologic conditions such as MI/R, their overproduction leads to oxidative stress, causing cell damage to nervous tissue, which may lead to DNA oxidation, promoting chain reactions of membrane lipid peroxidation, and alterations in membrane fluidity [28]. ROS produces malondialdehyde (MDA), an end product of lipid peroxidation. Therefore in the present study the level of MDA was estimated to estimate extent of ROS. Our result showed that the elevated level of MDA was markedly decreased by treatment with resveratrol, indicating that the cardioprotection conferred by resveratrol may be attributed to attenuating lipid peroxidation following myocardial ischemia/reperfusion. The overproduction of ROS can be detoxified by endogenous antioxidants, causing their cellular stores to be depleted [29]. Superoxide dismutase (SOD) and glutathione peroxidase (GSH-PX) are thought to be two dominant enzymes acting as free radical scavengers that could prevent ROS generation [30]. SOD scavenges the superoxide anion radical (O− 2) by catalyzing its dismutation to H2O2, which is scavenged to water by GSH-PX at the expense of glutathione [31]. In the present study, resveratrol was suggested to be effective in stimulating the activities of SOD and GSH-PX.
Ischemia-reperfusion enhances Nrf2 dissociation from Keap1, translocation to the nucleus, binding to the ARE, and activation of phase 2 detoxifying and antioxidant genes [32,33]. The Nrf2/ARE pathway affects cell survival through a variety of substrates, including apoptotic proteins such as Bcl-2 and Bax [34,35] and phase 2 enzymes such as HO-1 [36,37]. HO-1, which is considered a stress protein, is regarded as a sensitive and reliable indicator of cellular oxidative stress [38]. Our results suggest that resveratrol attenuates inflammation and oxidative stress induced by myocardial ischemia reperfusion injury by activating the expression of Nrf-2, which upregulating HO-1 expression.
Taken together, the results of this study suggest that resveratrol has robust protective effects and can potently protect against the myocardial I/R injury through activation of Nrf2/ARE pathway. Further studies in models of myocardial I/R injury are thus warranted to further identify other molecules involved in the protective effect and to clarify potential cross talk between upstream and downstream signaling molecules.
Disclosure of conflict of interest
None.
References
- 1.Basgut B, Kayki G, Bartosova L, Ozakca I, Seymen A, Kandilci HB, Ugur M, Turan B, Ozcelikay AT. Cardioprotective effects of 44Bu, a newly synthesized compound, in rat heart subjected to ischemia/reperfusion injury. Eur J Pharmacol. 2010;640:117–123. doi: 10.1016/j.ejphar.2010.04.045. [DOI] [PubMed] [Google Scholar]
- 2.Ramzy D, Rao V, Weisel RD. Clinical applicability of preconditioning and postconditioning: the cardiothoracic surgeons’s view. Cardiovasc Res. 2006;70:174–180. doi: 10.1016/j.cardiores.2006.01.020. [DOI] [PubMed] [Google Scholar]
- 3.Heusch G, Musiolik J, Gedik N, Skyschally A. Mitochondrial STAT3 activation and cardioprotection by ischemic postconditioning in pigs with regional myocardial ischemia/reperfusion. Circ Res. 2011;109:1302–1308. doi: 10.1161/CIRCRESAHA.111.255604. [DOI] [PubMed] [Google Scholar]
- 4.Xiong J, Xue FS, Yuan YJ, Wang Q, Liao X, Wang WL. Cholinergic anti-inflammatory pathway: a possible approach to protect against myocardial ischemia reperfusion injury. Chin Med J (Engl) 2010;123:2720–2726. [PubMed] [Google Scholar]
- 5.Naidu BV, Farivar AS, Woolley SM, Grainger D, Verrier ED, Mulligan MS. Novel broad-spectrum chemokine inhibitor protects against lung ischemia-reperfusion injury. J Heart Lung Transplant. 2004;23:128–134. doi: 10.1016/s1053-2498(03)00102-5. [DOI] [PubMed] [Google Scholar]
- 6.Cabigas EB, Somasuntharam I, Brown ME, Che PL, Pendergrass KD, Chiang B, Taylor WR, Davis ME. Over-expression of catalase in myeloid cells confers acute protection following myocardial infarction. Int J Mol Sci. 2014;15:9036–9050. doi: 10.3390/ijms15059036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sekhar KR, Yan XX, Freeman ML. Nrf2 degradation by the ubiquitin proteasome pathway is inhibited by KIAA0132, the human homolog to INrf2. Oncogene. 2002;21:6829–6834. doi: 10.1038/sj.onc.1205905. [DOI] [PubMed] [Google Scholar]
- 8.Radjendirane V, Joseph P, Lee YH, Kimura S, Klein-Szanto AJ, Gonzalez FJ, Jaiswal AK. Disruption of the DT diaphorase (NQO1) gene in mice leads to increased menadione toxicity. J Biol Chem. 1998;273:7382–7389. doi: 10.1074/jbc.273.13.7382. [DOI] [PubMed] [Google Scholar]
- 9.Ku BM, Joo Y, Mun J, Roh GS, Kang SS, Cho GJ, Choi WS, Kim HJ. Heme oxygenase protects hippocampal neurons from ethanol-induced neurotoxicity. Neurosci Lett. 2006;405:168–171. doi: 10.1016/j.neulet.2006.06.052. [DOI] [PubMed] [Google Scholar]
- 10.Zhu H, Itoh K, Yamamoto M, Zweier JL, Li Y. Role of Nrf2 signaling in regulation of antioxidants and phase 2 enzymes in cardiac fibroblasts: protection against reactive oxygen and nitrogen species-induced cell injury. FEBS Lett. 2005;579:3029–3036. doi: 10.1016/j.febslet.2005.04.058. [DOI] [PubMed] [Google Scholar]
- 11.Purdom-Dickinson SE, Lin Y, Dedek M, Morrissy S, Johnson J, Chen QM. Induction of antioxidant and detoxification response by oxidants in cardiomyocytes: evidence from gene expression profiling and activation of Nrf2 transcription factor. J Mol Cell Cardiol. 2007;42:159–176. doi: 10.1016/j.yjmcc.2006.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cho HY, Jedlicka AE, Reddy SP, Kensler TW, Yamamoto M, Zhang LY, Kleeberger SR. Role of NRF2 in protection against hyperoxic lung injury in mice. Am J Respir Cell Mol Biol. 2002;26:175–182. doi: 10.1165/ajrcmb.26.2.4501. [DOI] [PubMed] [Google Scholar]
- 13.Rangasamy T, Cho CY, Thimmulappa RK, Zhen L, Srisuma SS, Kensler TW, Yamamoto M, Petrache I, Tuder RM, Biswal S. Genetic ablation of Nrf2 enhances susceptibility to cigarette smoke-induced emphysema in mice. J Clin Invest. 2004;114:1248–1259. doi: 10.1172/JCI21146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li YJ, Takizawa H, Azuma A, Kohyama T, Yamauchi Y, Takahashi S, Yamamoto M, Kawada T, Kudoh S, Sugawara I. Disruption of Nrf2 enhances susceptibility to airway inflammatory responses induced by low-dose diesel exhaust particles in mice. Clin Immunol. 2008;128:366–373. doi: 10.1016/j.clim.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 15.Renaud S, de Lorgeril M. Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet. 1992;339:1523–1526. doi: 10.1016/0140-6736(92)91277-f. [DOI] [PubMed] [Google Scholar]
- 16.Athar M, Back JH, Tang X, Kim KH, Kopelovich L, Bickers DR, Kim AL. Resveratrol: a review of preclinical studies for human cancer prevention. Toxicol Appl Pharmacol. 2007;224:274–283. doi: 10.1016/j.taap.2006.12.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Aluyen JK, Ton QN, Tran T, Yang AE, Gottlieb HB, Bellanger RA. Resveratrol: potential as anticancer agent. J Diet Suppl. 2012;9:45–56. doi: 10.3109/19390211.2011.650842. [DOI] [PubMed] [Google Scholar]
- 18.Piotrowska H, Myszkowski K, Ziolkowska A, Kulcenty K, Wierzchowski M, Kaczmarek M, Murias M, Kwiatkowska-Borowczyk E, Jodynis-Liebert J. Resveratrol analogue 3,4,4’,5-tetramethoxystilbene inhibits growth, arrests cell cycle and induces apoptosis in ovarian SKOV-3 and A-2780 cancer cells. Toxicol Appl Pharmacol. 2012;263:53–60. doi: 10.1016/j.taap.2012.05.023. [DOI] [PubMed] [Google Scholar]
- 19.Xu Y, Nie L, Yin YG, Tang JL, Zhou JY, Li DD, Zhou SW. Resveratrol protects against hyperglycemia-induced oxidative damage to mitochondria by activating SIRT1 in rat mesangial cells. Toxicol Appl Pharmacol. 2012;259:395–401. doi: 10.1016/j.taap.2011.09.028. [DOI] [PubMed] [Google Scholar]
- 20.Li J, Feng L, Xing Y, Wang Y, Du L, Xu C, Cao J, Wang Q, Fan S, Liu Q, Fan F. Radioprotective and antioxidant effect of resveratrol in hippocampus by activating Sirt1. Int J Mol Sci. 2014;15:5928–5939. doi: 10.3390/ijms15045928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Li F, Gong Q, Dong H, Shi J. Resveratrol, a neuroprotective supplement for Alzheimer’s disease. Curr Pharm Des. 2012;18:27–33. doi: 10.2174/138161212798919075. [DOI] [PubMed] [Google Scholar]
- 22.Taguchi K, Motohashi H, Yamamoto M. Molecular mechanisms of the Keap1-Nrf2 pathway in stress response and cancer evolution. Genes Cells. 2011;16:123–140. doi: 10.1111/j.1365-2443.2010.01473.x. [DOI] [PubMed] [Google Scholar]
- 23.Tkachev VO, Menshchikova EB, Zenkov NK. Mechanism of the Nrf2/Keap1/ARE signaling system. Biochemistry (Mosc) 2011;76:407–422. doi: 10.1134/s0006297911040031. [DOI] [PubMed] [Google Scholar]
- 24.Black SC, Rodger IW. Methods for studying experimental myocardial ischemic and reperfusion injury. J Pharmacol Toxicol Methods. 1996;35:179–190. doi: 10.1016/1056-8719(96)00051-2. [DOI] [PubMed] [Google Scholar]
- 25.Speyer CL, Ward PA. Role of endothelial chemokines and their receptors during inflammation. J Invest Surg. 2011;24:18–27. doi: 10.3109/08941939.2010.521232. [DOI] [PubMed] [Google Scholar]
- 26.Lefer AM, Ma XL, Weyrich A, Lefer DJ. Endothelial dysfunction and neutrophil adherence as critical events in the development of reperfusion injury. Agents Actions Suppl. 1993;41:127–135. [PubMed] [Google Scholar]
- 27.Loh KP, Huang SH, De Silva R, Tan BK, Zhu YZ. Oxidative stress: apoptosis in neuronal injury. Curr Alzheimer Res. 2006;3:327–337. doi: 10.2174/156720506778249515. [DOI] [PubMed] [Google Scholar]
- 28.Manzanero S, Santro T, Arumugam TV. Neuronal oxidative stress in acute ischemic stroke: sources and contribution to cell injury. Neurochem Int. 2013;62:712–718. doi: 10.1016/j.neuint.2012.11.009. [DOI] [PubMed] [Google Scholar]
- 29.Candelario-Jalil E, Mhadu NH, Al-Dalain SM, Martinez G, Leon OS. Time course of oxidative damage in different brain regions following transient cerebral ischemia in gerbils. Neurosci Res. 2001;41:233–241. doi: 10.1016/s0168-0102(01)00282-6. [DOI] [PubMed] [Google Scholar]
- 30.Niizuma K, Yoshioka H, Chen H, Kim GS, Jung JE, Katsu M, Okami N, Chan PH. Mitochondrial and apoptotic neuronal death signaling pathways in cerebral ischemia. Biochim Biophys Acta. 2010;1802:92–99. doi: 10.1016/j.bbadis.2009.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chan PH. Reactive oxygen radicals in signaling and damage in the ischemic brain. J Cereb Blood Flow Metab. 2001;21:2–14. doi: 10.1097/00004647-200101000-00002. [DOI] [PubMed] [Google Scholar]
- 32.Shah ZA, Li RC, Thimmulappa RK, Kensler TW, Yamamoto M, Biswal S, Dore S. Role of reactive oxygen species in modulation of Nrf2 following ischemic reperfusion injury. Neuroscience. 2007;147:53–59. doi: 10.1016/j.neuroscience.2007.02.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kaspar JW, Niture SK, Jaiswal AK. Nrf2:INrf2 (Keap1) signaling in oxidative stress. Free Radic Biol Med. 2009;47:1304–1309. doi: 10.1016/j.freeradbiomed.2009.07.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Niture SK, Jaiswal AK. INrf2 (Keap1) targets Bcl-2 degradation and controls cellular apoptosis. Cell Death Differ. 2011;18:439–451. doi: 10.1038/cdd.2010.114. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 35.Hseu YC, Chou CW, Senthil Kumar KJ, Fu KT, Wang HM, Hsu LS, Kuo YH, Wu CR, Chen SC, Yang HL. Ellagic acid protects human keratinocyte (HaCaT) cells against UVA-induced oxidative stress and apoptosis through the upregulation of the HO-1 and Nrf-2 antioxidant genes. Food Chem Toxicol. 2012;50:1245–1255. doi: 10.1016/j.fct.2012.02.020. [DOI] [PubMed] [Google Scholar]
- 36.Srisook K, Kim C, Cha YN. Molecular mechanisms involved in enhancing HO-1 expression: de-repression by heme and activation by Nrf2, the “one-two” punch. Antioxid Redox Signal. 2005;7:1674–1687. doi: 10.1089/ars.2005.7.1674. [DOI] [PubMed] [Google Scholar]
- 37.Surh YJ, Kundu JK, Li MH, Na HK, Cha YN. Role of Nrf2-mediated heme oxygenase-1 upregulation in adaptive survival response to nitrosative stress. Arch Pharm Res. 2009;32:1163–1176. doi: 10.1007/s12272-009-1807-8. [DOI] [PubMed] [Google Scholar]
- 38.Kim YM, Pae HO, Park JE, Lee YC, Woo JM, Kim NH, Choi YK, Lee BS, Kim SR, Chung HT. Heme oxygenase in the regulation of vascular biology: from molecular mechanisms to therapeutic opportunities. Antioxid Redox Signal. 2011;14:137–167. doi: 10.1089/ars.2010.3153. [DOI] [PMC free article] [PubMed] [Google Scholar]


