Abstract
To evaluate the effects of alveolar ridge preservation with Bio-Oss bone substitute (Geistlich Pharma) on delayed implant osseointegration. The 3rd and 4th left and right mandibular premolars were extracted from four adult healthy male and female dogs. For the experimental group, we randomly selected two extraction sockets in each dog to be filled with Bio-Oss bone substitute (Geistlich Pharma). The two remaining extraction sockets remained untreated and served as the control group. Three months after Bio-Oss placement, dental implants were inserted into the alveolar bone of the experimental group and the control group. The osteogenic activity of the bone around the implants was assessed by evaluating the histological morphology and by estimating histomorphometric parameters at 3 and 6 months after delayed implantation. At 3 months, Goldner’s trichrome staining analysis showed that the bone-implant contact rate and mineralised bone area around the implant were significantly higher in the experimental group (75.98% ± 8.97% and 69.52% ± 9.63%, respectively) than in the control group (56.13% ± 8.18% and 52.82% ± 7.25%, respectively; P < 0.05). However, at 6 months, the two groups showed no significant difference. Fluorescence microscopy analysis revealed that the average mineralisation apposition rate of the bone tissue around the dental implant in the experimental group at 3 and 6 months was 6.80 ± 0.43 μm and 8.38 ± 0.84 μm, respectively, which was significantly higher than the rate in the control group (P < 0.05). These data indicated that alveolar ridge preservation by using Bio-Oss placement can promote osseointegration of delayed implantation. This may be a promising option for clinical use.
Keywords: Alveolar ridge, osseointegration, delayed implantation
Introduction
Alveolar bone is a highly malleable tissue that is remodelled constantly in conjunction with tooth development, physiological replacement, and masticatory pressure changes. After tooth extraction, an irreversible progressive absorption process occurs in the alveolar ridge. The hard and soft tissues surrounding the alveolar socket undergo structural changes, and the height and width of the alveolar ridge decrease substantially [1-3]. Mild absorption of the alveolar ridge will affect the function and aesthetics of subsequent denture restoration, whereas severe absorption may even lead to failure of restoration, particularly with regard to implant dentures. According to a report from the New York University Dental Implant Center (New York, NY, USA), 90% of patients referred for implant denture restoration must undergo procedures to resolve alveolar ridge deficiency before the restoration, and a multiple incidence of bone deficiency was similarly evident in patients who received implant restoration in China [4,5]. Wheel et al. [6] reported that 70%-80% of the alveolar ridge absorption observed within 2 years had occurred 3 months after tooth extraction. For the past 20 years, interest has grown in alveolar ridge preservation, which is defined as “any procedure undertaken at the time of or following an extraction that is designed to minimise bone formation within the socket [7]”. Ridge preservation using the guided bone regeneration technique improves the alveolar ridge height and width dimensions, compared to tooth extraction alone. Filling in the extraction sockets with Bio-Oss bone substitute (Geistlich Pharma) can reportedly effectively prevent alveolar ridge absorption and reduce vertical buccal bone loss and ridge width of the alveolar ridge, which is conducive to preserving alveolar ridge morphology and dimensional structure [8-11].
Replacing a tooth with a dental implant is a reliable and effective way to restore edentulous dentition [12]. Formation of a direct structural and functional connection between the implant and the supporting tissues, a process called osseointegration, has emerged as the criterion used to demonstrate initial stability and to evaluate long-term success [13]. Successful osseointegration is closely related to the bone quality and quantity of the alveolar ridge in the implant area. Residual ridge absorption after routine exodontia is an important factor that can potentially affect bone mineral density and bone mass in the implanted area because it influences bone quality and quantity and further osseointegration. The use of Bio-Oss in the extraction socket is indeed effective for preserving the ridge because it reduces the loss of the height and width of the alveolar ridge, and ensures delayed implantation of a dental implant in the ideal region of the alveolar ridge. However, the relationship between Bio-Oss and the dental implant surface remains unclear. Furthermore, the impact of alveolar ridge preservation by using Bio-Oss on delayed implant osseointegration is also unknown.
In the current study, the extraction sockets were first filled with Bio-Oss to preserve the alveolar ridge. What is more important, delayed implantation of dental implants was performed. Osseointegration occurring on the aforementioned preserved alveolar ridge area was assessed via radiography and histological observation with the aim of providing information for further clinical use.
Materials and methods
Animals and surgical procedures
Four adult healthy male and female dogs were housed and used. After the bilateral 3rd and 4th mandibular premolars of each dog were extracted, two extraction sockets were randomly selected to be filled with Bio-Oss and served as the experimental group, whereas the other two extraction sockets remained untreated and served as the control group. As the sentinel marker, small titanium nails were applied to the buccal root socket in the two groups. Periosteal stealth separation as well as mattress and intermittent tight sutures were subsequently used after sufficient decompression. Three months after the placement of Bio-Oss, delayed implantation of the dental implant was performed. Two animals were euthanized at 3 and 6 months after the delayed implantation surgery. To assess osteogenic activity, two fluorochromic bone markers were sequentially administered subcutaneously to the animals euthanised at 3 and 6 months, as follows [14]: tetracycline (25 mg/kg body weight) was administered 13 days and 14 days before euthanisation, and alizarin red (15 mg/kg body weight) was administered 3 days and 4 days before euthanisation.
Histological examination
The implants and the surrounding bone were immediately excised and excess tissue was removed. The mandible segment specimens with dental implants were promptly fixed in individual containers with 10% buffered formalin and fixed for 72 h at 4°C. They were then dehydrated by increasing concentrations of ethanol, embedded in 15 mm × 15 mm × 10 mm blocks of methyl methacrylate, sliced into sections of approximately 200-μm thickness by using the EXAKT300CP slicing machine (EXAKT Co., Norderstedt, Germany) along the major axis of the implant, and ground into 30-μm sheets by using the EXAKT400 grinding machine (EXAKT Co.). The sections were processed with Goldner’s trichrome stain and observed under a light microscope (Olympus BX51) and a fluorescent microscope (Nikon TS100-F; Nikon, Tokyo, Japan) for histological evaluation and static histomorphometric analysis.
Image-Pro Plus v6.0 software (For Windows) was then used to analyse the images obtained by light and fluorescent microscopy. The static and dynamic parameters (described later) were measured [15,16].
Bone-implant contact was calculated as the ratio between the length of the bone profile in direct contact with the implant surface and the length of the implant profile. The measurements were obtained from along the total perimeter of the implant.
Mineralised bone area was calculated as the ratio between the bone area in the screw and the total area in the screw. This parameter was measured and confirmed by the selected screw-threads in each section of the implants and further confirmation of the areas.
The mineralisation apposition rate was calculated as the ratio between the average thickness of the marking band and the duration of the bone marker administration period.
Statistical analysis
The SPSS Statistics 17.0 package was used to analyse the data. The significance of the difference between the two groups at specified observation points was measured by using the t test for independent samples. A value of P < 0.05 was statistically significant.
Results
Bone morphology analysis
Three months after delayed implantation, a large area of pink osteoid was evident around the implants in both alveolar sockets with orange new bone surrounding the area. A large gap was present between the implant surface and the new bone tissue. Strips of bone lacunae were evident along the edge of new bone near the implant. The new bone contained dense bone lacunae, lamellar bone, and woven bone without an orientation. In particular, the Bio-Oss particles in the experimental group were light blue with irregular edges that appeared serrated, a phenomenon named “silkworm eclipse” (Figure 1). At 6 months after delayed implantation, further observation of bone morphology showed large orange areas, which indicated new bone around the implant in the alveolar sockets of both groups. The area between the implant and the new bone tissues had closed significantly and light blue mineralised bone areas had increased in the experimental group and the control group. Furthermore, dense bone lacuna was evident in the new bone, trachychromatic nuclei were present in some regions of the orange or red lamellar bone, and light blue mineralised bone in some regions with a Haversian system were evident in the cortical bone. Furthermore, small serrated bright blue Bio-Oss particles were detected only in the experimental group (Figure 2).
Figure 1.
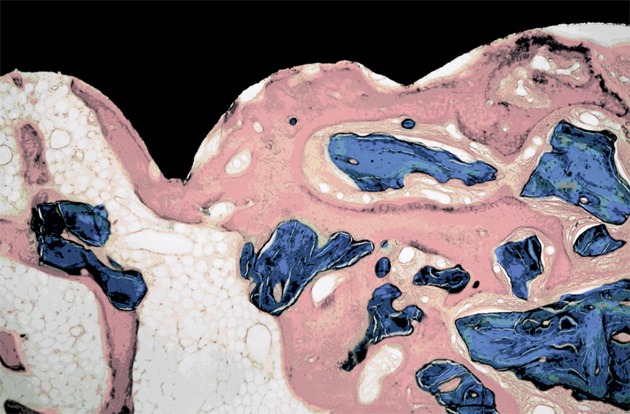
At 3 months after implantation, osseointegration is evident between the implant and the bone in the undecalcified section of the experimental group (Goldner’s trichrome stain; magnification, 40 ×).
Figure 2.
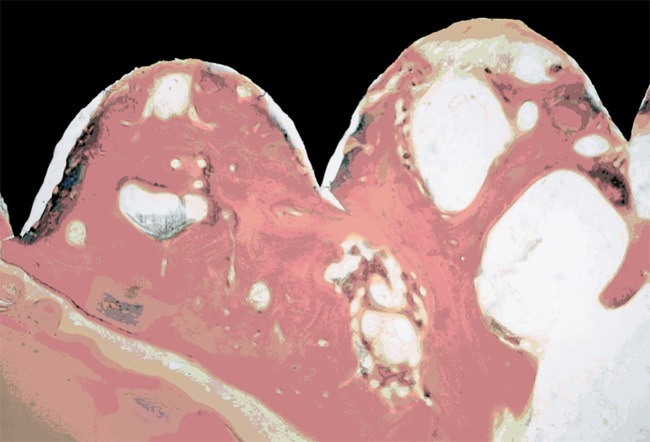
Histological morphology of the bone-implant section in the experimental group at 6 months after implantation (Goldner’s trichrome stain; magnification, 40 ×).
Fluorescence microscopy analysis
In both treatment groups, tetracycline and alizarin red fluorescence were evident in each section under fluorescence microscopy. The contour of the bone-implant interface in the marked area was clear and was composed of a band, a mesh, or flaky markers. At 3 months after implantation, the distribution of fluorescent banding was uneven in both groups. The highlighted band was narrow with a reticular distribution, which was primarily concentrated in the new edges and at the bone-implant interface (Figure 3). At 6 months, the distribution of fluorescence banding was relatively uniform and wide bright bands with a planar distribution were evident. There was a distinct interval between tetracycline fluorescence near the implant side and alizarin red fluorescence away from the implant side, which corresponded to new bone deposition that had occurred within 10 days (Figure 4).
Figure 3.
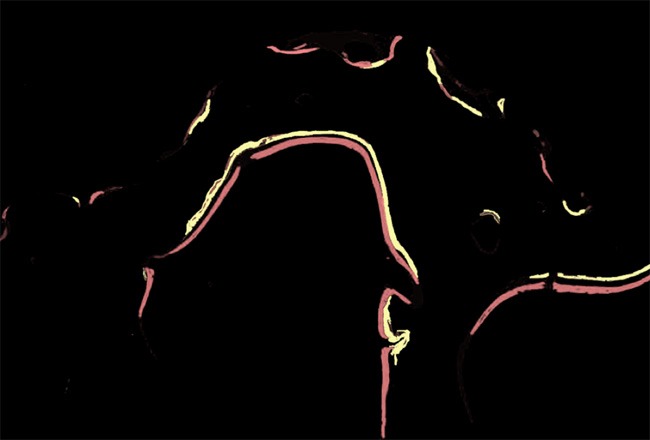
Double fluorescent strips of tetracycline and alizarin red in the experimental group at 3 months after implantation (magnification, 100 ×).
Figure 4.
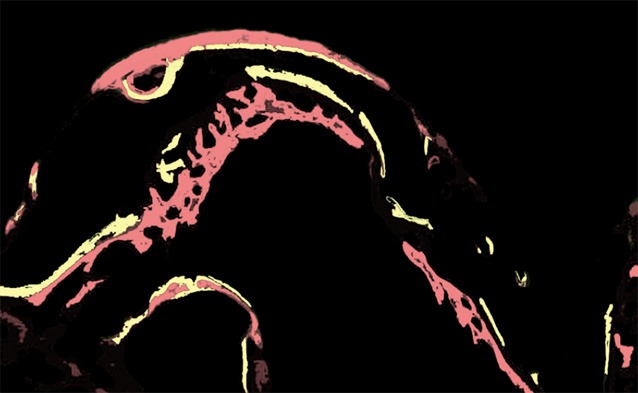
Double fluorescent strips of tetracycline and alizarin red in the experimental group at 6 months after implantation (magnification, 100 ×).
Bone histomorphometric analysis
At 3 months, the bone-implant contact rate, the mineralised bone area, and the mineralisation apposition rate were 75.98% ± 8.97%, 69.52% ± 9.63%, and 6.80 ± 0.43 µm, respectively, in the experimental group. These values were significantly higher than those of the control group at 56.13% ± 8.18%, 52.82% ± 7.25% and 5.23 ± 0.37 µm, respectively (P < 0.05). At 6 months, the mineralisation apposition rate was 8.38 ± 0.84 µm in the experimental group, which was significantly higher than the rate of 6.79 ± 0.54 µm in the control group (P < 0.05). However, the bone-implant contact rate and the mineralised bone area were slightly higher in the experimental group, but these differences were not statistically significant (P > 0.05; Table 1).
Table 1.
Bone-implant contact, mineralised bone area, and mineralisation apposition rate of bone around implant in six teeth
| Group | BIC (%) | BA (%) | MAR (μm) |
|---|---|---|---|
| Experimental (3 mo.) | 75.98 ± 8.97* | 69.52 ± 9.63* | 6.80 ± 0.43* |
| Control (3 mo.) | 56.13 ± 8.18 | 52.82 ± 7.25 | 5.23 ± 0.37 |
| Experimental (6 mo.) | 81.02 ± 6.73 | 77.47 ± 9.88 | 8.38 ± 0.85# |
| Control (6 mo.) | 74.66 ± 8.57 | 70.62 ± 8.92 | 6.79 ± 0.54 |
BA, bone area; BIC, bone-implant contact; MAR, mineralisation apposition rate. The data are presented as the mean ± the standard deviation.
P < 0.05, compared to the control group at 3 months.
P < 0.05 compared to the control group at 6 months.
Discussion
Osseointegration is a dynamic process that involves mechanical and biologic fixation. Numerous efforts have been devoted to facilitating osseointegration such as improving surgical procedures and postsurgical care, developing favourable implant design and surface properties, and incorporating bioactive factors. Four basic factors affect the osseointegration of an implant: (1) the biocompatibility, design, and surface state of the implant, (2) the quality and quantity of the alveolar ridge, (3) the technique of surgical implantation, and (4) the load state [17,18]. In our study, pure spiral titanium implants with a 3.0-mm diameter and 10.0-mm length were used in the delayed implant operation and non-loaded was selected. Our focus was on the effects of the alveolar ridge preservation on osseointegration.
The comparative advantage of Goldner’s trichrome method was used at various stages of bone mineralisation in our study. Based on the morphological changes of bone tissue surrounding the implant, we inferred that the bone tissue surrounding the dental implants in the two groups underwent the histological changes of osseointegration. These changes were in accordance with the bone formation process from reconstruction to maturation during an extended period. Hence, Bio-Oss did not affect the remodelling and maturation of the bone tissue surrounding the dental implant. Furthermore, at 3 months after implantation, it is notable that the bone-contact ratio and the mineralised bone area between the implant surface and bone tissue were significantly higher in the experimental group than in the control group, as demonstrated by histological observation and statistical analysis. This result indicated that filling the extraction sockets with Bio-Oss was beneficial for post extraction ridge preservation and had the exceptional ability to increase the bone-implant ratio and mineralised the bone area to improve osseointegration effectively under the condition of delayed dental implantation. It is difficult to explain the correlation between the residual quantity of the Bio-Oss and osseointegration; however, we hypothesise that it is related to the mechanism of bone remodelling and bone maturation. At 3 months, new bone formation was in an active state, and the osteoinductive effect of Bio-Oss promoted new bone generation. However, at 6 months, the bone was in a mature state with bone mineralisation, and the quantity of recently formed bone had decreased. In addition, the amount of Bio-Oss was significantly decreased at 6 months, compared to at 3 months, which suggested that new bone formation induced by Bio-Oss had decreased.
Our study design included a histomorphometric analysis of the average mineralisation apposition rate of the bone around the dental implants. This mineralisation apposition was analysed by injecting fluorescent materials such as tetracycline and alizarin red into the animals. During bone tissue mineralisation, the fluorescent materials combined with calcium and were absorbed into the hydroxyapatite crystallization surface or within the crystal lattice, which could mark the process of new bone formation [19]. Fluorescence microscopy of the implant-bone sections of both groups showed that the highlighted fluorescent belt occurred around the bone-implant interface. This belt was narrow with a meshy distribution at 3 months and was wide with a planar distribution at 6 months. The interval between the green tetracycline fluorescence band close to the implant side and the alizarin red fluorescence band far from the implant side was measured and calculated. The results showed that the average mineralisation apposition rate was significantly higher in the experimental group at 3 months after delayed implantation, which was consistent with the histomorphometric results analysed by Goldner’s trichrome staining. In addition, more osteogenesis was observed on the surface of the Bio-Oss particles around the implants with better mineralisation, which was consistent with the results of Artzi et al [20-22]. Therefore, these results indicated that Bio-Oss served as a scaffold to promote bone tissue regeneration and promoted osseointegration of the delayed implant.
In summary, this study confirms that alveolar ridge preservation with Bio-Oss promoted osseointegration of a delayed implant. This factor could enlarge the suitable range of implant denture restoration and improve the stability and success of implantation.
Acknowledgements
The work was supported by the National Natural Science Foundation of China (No. 81402298).
Disclosure of conflict of interest
None.
References
- 1.Tan WL, Wong TLT, Wong MCM, Lang NP. A systematic review of post-extractional alveolar hard and soft tissue dimensional changes in humans. Clin Oral Implants Res. 2012;23:1–21. doi: 10.1111/j.1600-0501.2011.02375.x. [DOI] [PubMed] [Google Scholar]
- 2.Araújo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32:212–218. doi: 10.1111/j.1600-051X.2005.00642.x. [DOI] [PubMed] [Google Scholar]
- 3.Weijden FV, Acqua FD, Slot DE. Alveolar bone dimensional changes of post-extraction sockets in humans: a systematic review. J Clin Periodontol. 2009;36:1048–1058. doi: 10.1111/j.1600-051X.2009.01482.x. [DOI] [PubMed] [Google Scholar]
- 4.Ashman A. Postextraction ridge preservation using a synthetic alloplast. Implant Dent. 2000;9:168–176. doi: 10.1097/00008505-200009020-00011. [DOI] [PubMed] [Google Scholar]
- 5.Weng YL, Wang M, Liu W, Hu XJ, Chai G, Yan QM, Zhu L, Cui L, Cao YL. Repair of experimental alveolar bone defects by tissue-engineered bone. Tissue Engineering. 2006;12:1503–1513. doi: 10.1089/ten.2006.12.1503. [DOI] [PubMed] [Google Scholar]
- 6.Wheeler SL, Vogel RE, Casellini R. Tissue preservation and maintenance of optimum aesthetics: a clinical report. Int J Oral Maxillofac Implants. 2000;15:265–271. [PubMed] [Google Scholar]
- 7.Darby I, Chen S, De Poi R. Ridge preservation: What is it and when should it be considered. Aust Den J. 2008;53:11–21. doi: 10.1111/j.1834-7819.2007.00008.x. [DOI] [PubMed] [Google Scholar]
- 8.Mladenović Z, Sahlin-Platt A, Andersson B, Johansson A, Björn E, Ransjö M. In vitro study of the biological interface of Bio-Oss: implications of the experimental setup. Clin Oral Implants Res. 2013;24:329–335. doi: 10.1111/j.1600-0501.2011.02334.x. [DOI] [PubMed] [Google Scholar]
- 9.Fickl S, Zuhr O, Wachtel H, Stappert CF, Stein JM, Hürzeler MB. Dimensional changes of the alveolar ridge contour after different socket preservation techniques. J Clin Periodontol. 2008;35:906–913. doi: 10.1111/j.1600-051X.2008.01305.x. [DOI] [PubMed] [Google Scholar]
- 10.Araújo MG, Lindhe J. Ridge preservation with the use of Bio-Oss collagen: A 6-month study in the dog. Clin Oral Implants Res. 2009;20:433–440. doi: 10.1111/j.1600-0501.2009.01705.x. [DOI] [PubMed] [Google Scholar]
- 11.Mardas N, Chadha V, Donos N. Alveolar ridge preservation with guided bone regeneration and a synthetic bone substitute or a bovine derived xenograft: a randomized, controlled clinical trial. Clin Oral Implants Res. 2010;21:688–698. doi: 10.1111/j.1600-0501.2010.01918.x. [DOI] [PubMed] [Google Scholar]
- 12.Wittneben JG, Buser D, Salvi GE, Bürgin W, Hicklin S, Brägger U. Complication and failure rates with implant-supported fixed dental prostheses and single crowns: a 10-year retrospective study. Clin Implant Dent Relat Res. 2014;16:356–364. doi: 10.1111/cid.12066. [DOI] [PubMed] [Google Scholar]
- 13.Khan SN, Ramachandran M, Kumar SS, Krishnan V, Sundaram R. Osseointegration and more-A review of literature. Indian J Dent. 2012;3:72–76. [Google Scholar]
- 14.Pautke C, Vogt S, Tischer T, Wexel G, Deppe H, Milz S, Schieker M, Kolk A. Polychrome labeling of bone with seven different fluorochromes: enhancing fluorochrome discrimination by spectral image analysis. Bone. 2005;37:441–445. doi: 10.1016/j.bone.2005.05.008. [DOI] [PubMed] [Google Scholar]
- 15.Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Mallluche H, Meunier PJ, Ott SM, Recker RR. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMB Histomorphometry Nomenclature Committee. J Bone Miner Res. 1987;2:595–610. doi: 10.1002/jbmr.5650020617. [DOI] [PubMed] [Google Scholar]
- 16.Giavaresi G, Fini M, Cigada A, Chiesa R, Rondelli G, Rimondini L, Torricelli P, Aldini NN, Giardino R. Mechanical and histomorphometric evalutions of titanium implants with different surface treatments inserts in sheep cortical bone. Biomaterials. 2003;24:1583–1594. doi: 10.1016/s0142-9612(02)00548-3. [DOI] [PubMed] [Google Scholar]
- 17.Sykaras N, Iacopino AM, Marker VA, Triplett RG, Woody RD. Implant materials, designs, and surface topographies: their effect on osseointegration. A literature review. Int J Oral Maxillofac Implants. 2000;15:675–690. [PubMed] [Google Scholar]
- 18.Chang PC, Giannobile WV. Functional Assessment of Dental Implant Osseointegration. Int J Periodontics Restorative Dent. 2012;32:e147–153. [PubMed] [Google Scholar]
- 19.Lee TC, Mohsin S, Taylor D, Parkesh R, Gunnlaugsson T, O’Brien FJ, Giehl M, Gowin W. Detecting microdamage in bone. J Anat. 2003;203:161–172. doi: 10.1046/j.1469-7580.2003.00211.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Artzi Z, Tal H, Dayan D. Porous Bovine Bone Mineral in Healing of Human Extraction Sockets. Part 1: Histomorphometric Evaluations at 9 Months. J Periodontol. 2000;71:1015–1023. doi: 10.1902/jop.2000.71.6.1015. [DOI] [PubMed] [Google Scholar]
- 21.Artzi Z, Tal H, Dayan D. Porous bovine bone mineral in healing of human extraction sockets: 2. Histochemical observations at 9 months. J Periodontol. 2001;72:152–159. doi: 10.1902/jop.2001.72.2.152. [DOI] [PubMed] [Google Scholar]
- 22.Fickl S, Zuhr O, Wachtel H, Bolz W, Hürzeler MB. Hard tissue alterations after socket preservation: An experimental study in the beagle dog. Clin Oral Implants Res. 2008;19:1111–1118. doi: 10.1111/j.1600-0501.2008.01575.x. [DOI] [PubMed] [Google Scholar]


