Abstract
Matrix metalloproteinases (MMPs) have been studied extensively, and MMP inhibitors have been used as dental pretreatment agents prior to dentin bonding because they reduce collagen fiber degradation and improve bonding strength. However, morphologic characteristics of the collagen network after etching and of the post-adhesive dentin hybrid layers (DHL) after MMP inhibitors pretreatment have not been evaluated. Thus, we investigated demineralized dentin pretreated with chlorhexidine (CHX) and minocycline (MI) in an etch- and -rinse adhesive system with field emission scanning electron microscopy (FESEM) and immuno-gold labeling markers to observe the collagen network and DHL. FESEM revealed after CHX and MI, a demineralized dentin surface and improved collagen network formation, reduced collagen degradation, and distinct gold-labeling signals. Applying adhesive after either MMP inhibitor created a better dentin interface as evidenced by immuno-gold staining, better adhesive penetration, and higher DHL quality. With microtensile bond strength tests (µTBS) we estimated bonding strength using µTBS data. Immediate µTBS was enhanced with MMP inhibitor application to the bonding surface, and the CHX group was significantly different than non-treated etched surfaces, but no significant change was detected in the MI group. Surface micromorphology of the fractured dentin resin restoration showed that the CHX group had a better resin and dentin tube combination. Both MMP inhibitors created uniform resin coverage. Thus, morphologic results and µTBS data suggest that CHX and MI can inhibit MMP activity, improve immediate bonding strength, and enhance dentin bonding stability with an etch- and -rinse adhesive system.
Keywords: Dentin collagen fibers, chlorhexidine, minocycline, immuno-gold nanoparticles labeling, FESEM, µTBS
Introduction
Even today, with novel dental bonding adhesive systems, dentin bonding is difficult because of its unique organic nature. The dentin extracellular matrix (ECM) consists of a 3D scaffold of chiefly type I collagen fibrils (90%) [1]. Compared to enamel, dentin is less mineralized and contains abundant organic material (19-20%). Resin-dentin bonds, due to collagen matrix loss in the dentin hybrid layer (DHL) are more complex and can not remain stable over time as resin-enamel bonds [2,3]. Using an acid-etching treatment on the dentin surface, dentin was demineralized, the collagen network was exposed, and the resin monomer penetrated into the tubular and lateral branch to provide mechanical interlocking with dentin to create a DHL [4]. Collagen fiber quality is related to DHL stability and auto-degradation of collagen fibers occurs within the DHL created by contemporary dentin bonding systems via the slow action of matrix metalloproteinases (MMPs) [5]. Thus, MMPs are implicated in degradation of poorly resin-infiltrated DHL and dentin bonding stability [6-8].
MMPs are a family of 30 host-derived proteolytic zinc- and calcium-dependent endopeptidases that, together, degrade practically all ECM components. Thus, they are important to many biological and pathological processes [9]. MMP-1 and -8 (collagenase) [10], -2 and -9 (gelatinase) [11] and -3 (stromelysin) [12] have been found in normal dentin. Under normal circumstances, MMPs are secreted into the ECM as inactive proenzymes which require activation by proteinases, chemicals, or low pH to degrade matrix components [13]. When dentin MMPs are exposed and activated by self-etch or etch- and -rinse adhesives [5,14], they degrade type I collagen fibers [15], as well as influence DHL and bonding strength. With both self-etch or etch- and -rinse adhesives, denuded collagen fibers can be seen at the bottom of the DHL, because of incomplete resin monomer infiltration. Once MMPs are activated, they adversely modify dentin bonding durability.
Recently, MMP inhibitors have been employed to pretreat the demineralized dentin interface or have been incorporated into the bonding component to prevent activation of MMPs. MMP inhibitors are endogenous inhibitors/tissue inhibitors of metalloproteinases (TIMPs) or exogenous inhibitors. A recent review suggested that exogenous MMP inhibitors, such as chlorhexidine (CHX), tetracyclines (TCs) and derivatives, quaternary ammonium compounds, benzalkonium chloride (BAC), natural substances (green tea polyphenol epigallocatechin-3-gallate), and other synthetic MMP inhibitors such as gallardin, and the chelating agent ethylenediaminetetraacetic acid can all inhibit dentin MMPs, preserve the DHL, stabilize the adhesive interface and increase bonding strength [16].
CHX and minocycline (MI) have received increased interest as they compete with MMPs for calcium and zinc ions as metal chelating agents to inhibit their activity. Specifically, MMP-2, -8, -9 activity are reduced and this slows collagen degradation in the DHL and improves bonding strength [17,18]. A 2% CHX solution applied to a demineralized dentin surface and analyzed with enzyme spectrum detection and nanoleakage studies confirmed that CHX could inhibit gelatinase activity and maintain the stability of the bonding interface [19]. TC compounds can also prevent collagen degradation and improve stability in the DHL [20]. Chemical modification of TC class members such as doxycycline and MI can also be used as nonspecific MMP inhibitors for treatment of oral diseases [21-23]. Recent research suggested that the application of aqueous solutions of MI, doxycycline, and semisynthetic TCs as a pretreatment after acid-etching improved immediate bonding performance [18].
Therefore, we analyzed and evaluated the effect of CHX and MI pretreatment on the morphology of dentin collagen fibers and the structure of the DHL under field emission scanning electron microscopy (FESEM). Together with the structural analysis, we also measured immediate microtensile bonding strength created by an etch- and -rinse adhesive system.
Material and methods
Forty-five freshly extracted caries-free human (18-30 years-of-age) third molars were selected with informed donor consent. All extracted teeth, removed tissue remnants, and debris were stored in refrigerated distilled water at 4°C, and were used within 2 weeks post-extraction.
Initial specimen preparation
The tooth occlusal enamel was removed perpendicular to the long axis of the tooth to expose flat dentin and then roots were removed at the cementum-enamel junction. A dentin block (6.0 mm × 6.0 mm × 2.0 mm) was obtained from the mid-coronal portion of each tooth using a low-speed precision diamond saw (Model SYJ-150A, Shenyang Kejing Equipment Manufacturing Co., Ltd) with water-cooling. Confirmation of lack of enamel residue was made with a stereomicroscope (12 × SZ51/61, Olympus, Tokyo, Japan). Next, the dentin block surface was polished with a #600-grit wet silicon carbide (SiC) abrasive paper on a grinder (UNIPOL-820, Shenyang Kejing Equipment Manufacturing Co., Ltd) for 60 sec to standardize the smear layer. Then, the sample was rinsed thoroughly with water.
Etching and bonding procedures and treatment groups
All dentin blocks (n = 45) were experimentally pretreated for 60 s with either 0.01 M phosphate buffered saline (PBS, pH 7.2), 2% CHX solution, or 2% MI solution after etching with phosphoric acid and before applying dentin adhesive. Dentin blocks were randomly divided into nine equal groups (n = 5). Three groups were acid etched and examined without further application of adhesive or resin: (etching-PBS (PE) group, etching-CHX (CE) group and etching-MI (ME) group). Three groups had adhesive applied after etching and experimental pre-treatment, and these were adhesive-PBS (PA) group, adhesive-CHX (CA) group and adhesive-MI (MA) group. Finally, three groups had composite resin applied after etching, experimental pre-treatment, and adhesive application (Filtek Z250, 3M ESPE, St. Paul, MN), and these were the resin-PBS (PR) group, the resin-CHX (CR) group and the resin-MI (MR) group. The corresponding etch-and-rinse adhesive systems (Adper Single Bond 2) were applied in accordance with the manufacturer’s instructions (See Table 1). For PR, CR, MR groups, to each adhesive surface a composite resin in three increments was applied, and each increment was compressed firmly (~2 mm high) and light cured for 20 s. Completed samples were rinsed in distilled water and then incubated for 24 h at 37°C.
Table 1.
Adhesive system (batch number), composition, manufacturer and application technique according to manufacturer’s instructions
| Adhesive system (batch number) | Compositions | Manufacturer | Application Technique |
|---|---|---|---|
| Adper Single Bond 2 (479674) | 1. Etchant: 35% phosphoric acid (GelEtch, SCI-PHARM, America) | 3M ESPE, St. Paul, MN | 1. Apply etchant for 15 s |
| 2. Adhesive: Bis-GMA, HEMA, dimethacrylates, ethanol, water, photoinitiator system, methacrylate functional copolymer of polyacrylic and polyitaconic acids, 10% by weight of 5 nm-diameter spherical silica nanopaticles | 2. Rinse for 15 s | ||
| 3. Blot excess water using a cotton pellet without air-drying | |||
| 4. Apply one coat of adhesive for 10 s with gentle agitation | |||
| 5. Gently air-dry (10 s at 20 cm) | |||
| 6. Apply one coat of adhesive for 10 s with gentle agitation | |||
| 7. Gently air-dry (10 s at 20 cm) | |||
| 8. Light curing for 10 s |
Bis-GMA: bisphenol-A-diglycidylmethacrylate; HEMA: hydroxyethylmethacrylate.
Immunolabeling specimen preparation
For the PE, PA, CE, CA, ME, and MA groups, immuno-gold nanoparticle labeling was performed using an IgG anti-type I collagen (rabbit polyclonal, Bioss Antibodies) and goat anti-rabbit-IgG antibodies conjugated to 25-nm gold nanoparticles (Yikang Bio-engineering Co.) Briefly, specimens were rinsed in PBS and non-specific protein binding sites were blocked with normal goat serum (Yikang Bio-engineering Co.) for 30 min at room temperature. Overnight incubation was performed using the primary antibodies (1:50 diluted in 0.01 M PBS at pH 7.2) at 4°C. After incubation, specimens were rinsed with Triton X-100. Next, immunolabeling was performed with secondary antibodies (1:20 diluted in 0.01 M PBS at pH 7.2) for type I collagen visualization. The secondary antibodies were incubated with the specimens for 90 min at room temperature then rinsed in Triton X-100. After labeling of the collagen, dentin plates (1.0 ± 0.02 mm thick and 2.0 mm × 4.0 mm2) were then obtained from the first piece of dentin near the enamel-dentinal junction sectioned perpendicular to the treated side, using a slow-speed diamond saw with cold water. Dentin plates surfaces were then treated with 37% phosphoric acid solution for 3 s, and cutting debris were removed for observation of the collagen fibers and the DHL. Then, samples were saline-flushed for 15 min [24].
FESEM evaluation
Dentin plates were selected from each group after storage for 24 h and each sample was irrigated thoroughly with distilled water. Specimens were fixed in 2.5% glutaraldehyde in 0.1 M PBS (pH 7.2) for 4 h at room temperature, rinsed in 0.01 M PBS, dehydrated in an ascending ethanol series (25, 50, 75, 90, 95 and 100%). Then, samples were air-dried at room temperature for 24 h and sputter-coated with gold nanoparticles (nanoparticles < 5 nm, thickness < 10 nm), using an ion-sputter coater (Hitachi E-1010) for 10 s, Observations were performed under an FESEM (Hitachi S-4800 Nissei Sangyo Co. Ltd., Tokyo, Japan) at 7 kV.
Microtensile bond strength (µTBS) specimen preparation
Samples from the resin-treated teeth (groups PR, CR, and MR) were embedded with medicinal plaster to prevent micro-cracks during the cutting process. Then, samples were sectioned with a low-speed diamond saw in the buccal-lingual direction perpendicular to the adhesive interface to obtain bonding sheets. Sheets were embedded again, and dentin-resin composite sticks were produced by cutting the sheets in the mesial-distal direction, each stick had an adhesive area of ~1 mm2, and this was confirmed with a stereomicroscope. Defective sticks with micro-cracks were excluded. Fifteen sticks were used for each group (n = 15) and were incubated at 37°C for 24 h prior to µTBS testing.
µTBS testing
The adhesive area of each stick was measured with a micrometer, and sticks were placed on a paper towel to absorb excess moisture. Individual sticks were bonded onto a test block of the microtensile testing machine (Bisco Inc, Schaumburg, IL). The block was composed of two test jaws with sticks testing surfaces, linkage and slide keys, the jaws had a 2-mm gap between them and the edges were cut back at an angle to allow the testing sticks to be glued to the block in place without glue migrating and dripping between the jaws. We ensured that no tension was placed on the stick, and we placed a drop of glue on each jaw in the middle of the test stick mount area. We handled sticks with tweezers at the interface to place them on the jaws. All sticks were parallel to the direction of the linkage to prevent sideways vectors for tension, the stick bond interface was over the gap and no excess glue had dripped into the gap. The test block was run at a speed of 1 mm/min until the sample was broken.
Then μTBS was calculated by dividing the load at failure by the cross-sectional bonding area.
Statistical analysis
One-way ANOVA was used to compare differences of immediate bonding strengths, and to determine significance among groups (a = 0.05).
Failure analysis
After µTBS testing, failure sticks surfaces were dried with a gradient alcohol dehydration and gold-sputter coated nanoparticles, and examined under FESEM to determine the mode of failure. The failure mode was classified as cohesive (failure within resin or dentin composite), adhesive (failure at resin-dentin interface) or mixed (failure included both cohesive and adhesive types). Differences between groups were evaluated for significance by Fishers exact test.
Results
Immunolabeling and FESEM
FESEM images of the demineralized collagen fibers in the etching-treatment groups with different pretreatments are shown in Figure 1. PE, CE, and ME groups all had exposed collagen fibers but the CE group had more exposed collagen fibers than the rest and dentin is less mineralized. Major collagen fibers were visible and gold nanoparticles were clearly visible in secondary fibers (Figure 1D). In the ME group, there was also gold labeling of type I collagen fibers and fibers were exposed (Figure 1F). Adhesive-treatment groups with different pretreatments are shown in Figure 2. PA, CA, and MA groups all had a uniform and dense DHL with high density regions visible, at the bottom of the DHL exposed collagen fibers indicated by a layer of colloidal gold (Figure 2A, 2B). The CA group had the most uniform and dense DHL (Figure 2C) and exposed collagen fibers at the bottom of the DHL were seen (Figure 2D). For the MA group, a boundary between the bonding agent layer and the hybrid layer was evident (Figure 2E) and high density regions of labeled collagen were minimally exposed at the bottom of the hybrid layer (Figure 2F).
Figure 1.
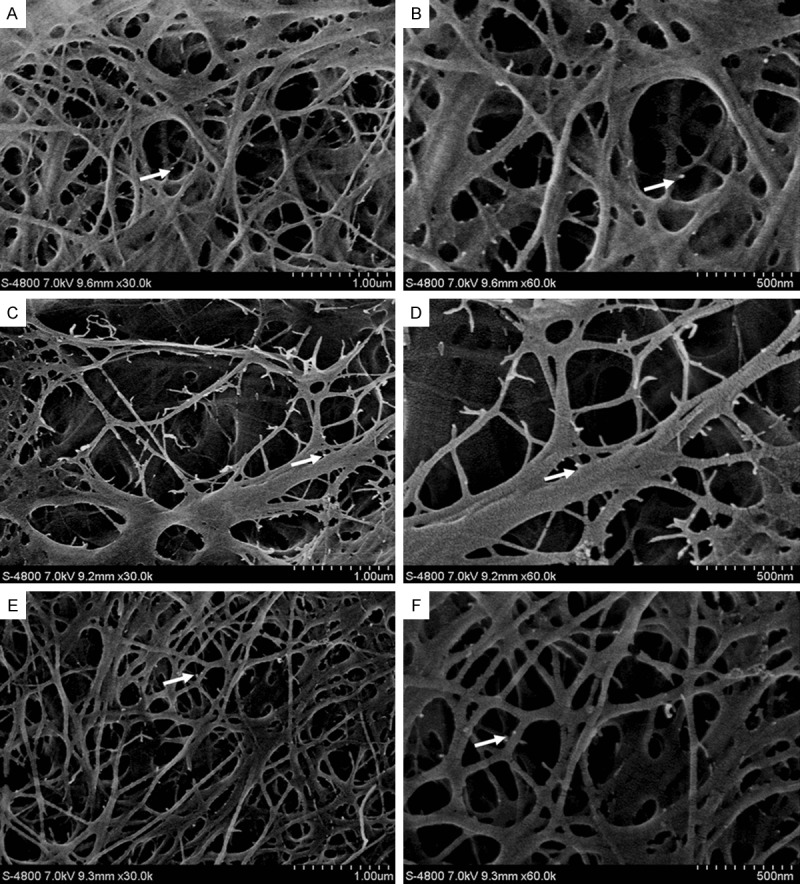
Image of etched surface (FESEM images of the pretreatments on etching groups): A. PE: collagen fibers were visibly exposed with a 3D reticular structure, collagen fibers staggered in a mesh formation, and gold was a dense white dot. Gold labeling is absent in areas without demineralization (30,000×); B. amplification of PE local collagen fibrils, the 3D structure is clearly visible, gold nanoparticles are evident (60,000×); C. CE: collagen fibers exposed, collagen fibers staggered in a mesh formation, visible major collagen fibers, gold increased in secondary fibers (30,000×); D. amplification of CE local collagen fibrils, 3D structure is more clearly visible, gold nanoparticles are evident (60,000×); E. ME: collagen fibers exposed, collagen fibers staggered in a mesh formation, visible major collagen fibers, gold nanoparticles clear in secondary fibers (30,000×); F. amplification of ME local collagen fibrils, 3D structure is more visible, gold nanoparticles are clear (60,000×) (white arrows indicate gold nanopaticles).
Figure 2.
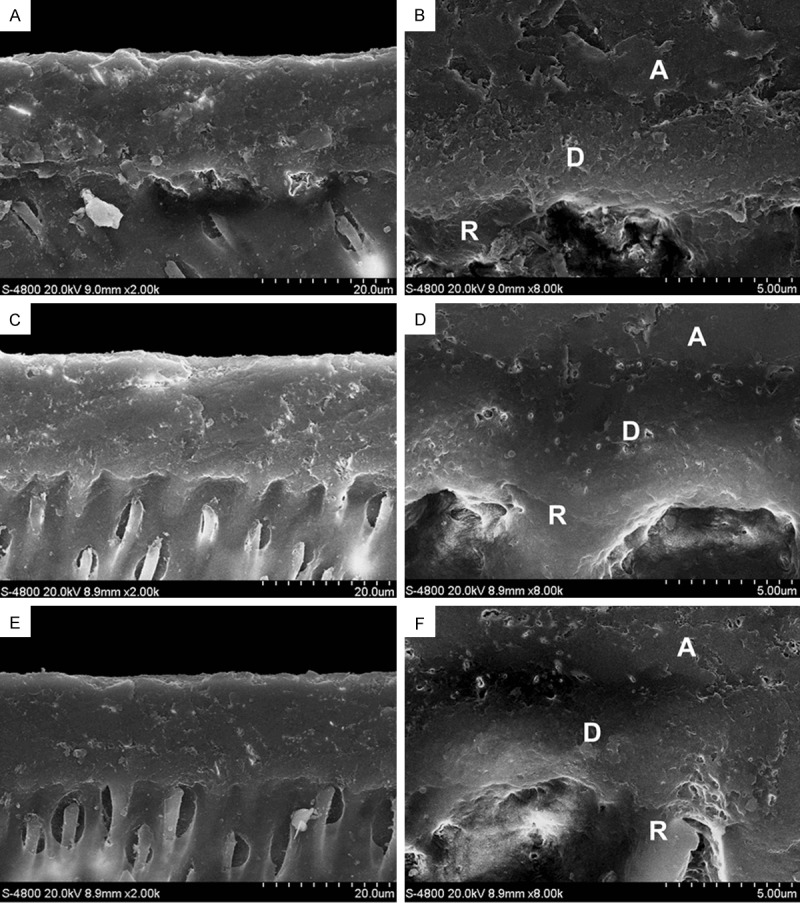
Cross-sectional image of the DHL (FESEM images of pretreatments on adhesive treatment groups): A. PA: DHL is uniform and dense, high density regions are visible at the bottom of the DHL where exposed collagen fibers marked by gold nanoparticles (2,000×); B. part of the exposed collagen fibers at the bottom of the DHL can be clearly seen (8,000×); C. CA: DHL is more uniform and dense, high density regions are visible at the bottom of DHL where exposed collagen fibers are indicated by gold nanoparticles (2,000×); D. few exposed collagen fibers at the bottom of the DHL can be seen (8,000×); E. MA: DHL is more uniform and dense, with no obvious boundary between the bonding agent layer and the hybrid layer, high density regions are less visible at the bottom of the hybrid layer where exposed collagen fibers are indicated by gold nanoparticles (2,000×); F. few exposed collagen fibers at the bottom of the DHL can be seen (8,000×) (A: adhesive D: dentin hybrid layer R: resin tag).
µTBS
The mean µTBS and standard deviations for each group are shown in Table 2. One way ANOVA test revealed a significant difference in the bonding strength among pretreatments (P < 0.0001). CHX and control groups were significantly different (P < 0.01) as were the CHX and MI groups (P < 0.01). μTBS of the MI group exceeded that of control but not significantly (Table 2). Within failure models, there were no significant differences (P > 0.05) among each group according to Fisher’s exact test. Almost all failures were of the adhesive mode (Table 2). Data from micromorphology for the resin treatment groups with different pretreatments are shown in Figure 3.
Table 2.
Mean µTBS and modes of failure model for each group
| Group | N | Mean (MPa) ± SD | F-value | P-value | Failure Mode % (Adhesive/Cohesive/Mixed) |
|---|---|---|---|---|---|
| PBS | 15 | 32.25 ± 4.12A | - | - | (100/0/0)a |
| CHX | 15 | 43.47 ± 3.94B | 32.24 | 0.0000 | (86.7/6.7/6.7)a |
| MI | 15 | 34.16 ± 4.52A | - | - | (100/0/0)a |
Groups identified by different uppercase letters are significantly different (p < 0.05). Groups identified by the same lowercase letters are not significantly different (p > 0.05).
Figure 3.

Image of the resin fracture surface (FESEM images of failed interface of the resin treatment groups after force fracture): A. PR: binder covers the surface homogeneously at the bottom of the DHL (2,000×); B. PR local amplification; fractures between resin and tubular wall exist, remaining tubular resin is short, no exposed collagen fibers observed (10,000×); C. CR: binder connects to the DHL completely, much better resin infiltration into dentin tubules can be seen (2,000×); D. CR local amplification; resin is obviously thicker and longer and combined with dentin tubule, no collagen fiber exposure (10,000×); E. MR: homogeneous binder coverage, most of the tubes have resin infiltration (2,000×); F. MR local amplification; resin is thick and within tubules, with small cracks between the resin and tube wall, no exposed collagen fiber can be seen (10,000×).
Discussion
Commercial adhesive systems can be classified into two categories: self-etch and etch- and -rinse adhesive systems. In spite of the simplification of adhesive systems, three-step etch- and -rinse adhesives are still gold standard methods in terms of durability and bond strength [25-27]. Etch- and -rinse adhesives use phosphoric acidic to demineralize the dental substrate and to remove the smear layer, leaving behind a collagen-rich mesh-work. Demineralized dentin has two types of porosity: tubules and collagen networks. Characterization of this open network porosity is crucial to predict bonding efficiency [28]. To obtain stable bonding, effective impregnation of the collagen network porosity is necessary [29]. Previous work suggests that 35% phosphoric acid (pH 0.7 to -1) to acid-etching dentin in an etch- and -rinse adhesive system approach initially inactivated pro-MMPs trapped in the mineralized dentin due to low pH which was believed to denature MMPs [30]. However, recent work suggests that dentin treatment with 37% phosphoric acid does not denature endogenous proteases of the dentin matrix [31]. When acid-etching dentin, the acidic environment can activate MMPs trapped in the peripheral dentin. Thus, we selected etch- and -rinse adhesive system to study the morphology of collagen networks and to learn how the DHL is influenced by MMP inhibitors.
Many studies have confirmed that MMP inhibitors can reduce collagen fibers degradation in the DHL, but few studies have examined the potential effect of CHX and MI and compared them to determine the best pretreatment for the formation of a collagen network and a DHL for better immediate bond strength. Also, there are little morphological study data about the effect of MMP inhibitors on collagen fibers and the DHL. Immunolabeling methods with FESEM along with antigen-specific probes labeled with gold nanoparticles allow observation of target antigens at high resolution [32].
Thus, we measured gold immunolabeling of type I collagen fibers and the corresponding DHL and compared treatments to controls. The CHX group had more uniform and exposed collagen networks and offered better fibers protection, assisting adhesives to penetrate into the collagen network, enhancing adhesiveness between the fibers and the resin. The quality of the DHL and the bonding strength was significantly improved as well, suggesting that CHX can prevent MMP activity that degrades collagen fibers in the DHL [33]. The MI group also had a higher density DHL, and bonding strength was improved. The MI groups also had a denser DHL and bonding strength was improved. Although these data were not statistically significant, the fracture mode was relatively uniform, resin combined with the dentin tubule closely. There were smaller cracks between resin and dentin tube wall, but longer resin tags penetrated to the demineralized dentin surface was still able to maintain the stability. MI can be used as a nonspecific MMPs inhibitor in the treatment on oral diseases in many aspects, the effect of chemical modification of tetracyclines still need further study.
This work supported previous studies which suggested that a 2% aqueous solution of CHX and MI did not impair resin-dentin bond strength or the DHL formation for etch- and -rinse adhesives [18]. Thus, CHX and MI pretreatments of demineralized dentin offered better collagen networks and increased interfibrillar volume, which enhanced adhesive resin penetration and produced a higher quality DHL with greater immediate bonding strengths.
Over time, studies have confirmed that MMPs are critical for degrading collagen fibers, damaging the DHL and decreasing bonding strength. MMP inhibitors thus have an enormous potential for inhibiting MMPs activity by protecting collagen fibers from degradation and increasing bonding strengths. Additional approaches have been attempted to obtain an effective bond to the dentin substrate such as remineralization [34], ethanol wet-bonding [35], and the use of collagen-cross-linkers, such as proanthocyanidin, glutaraldehyde, genipin, riboflavin, carbodiimide, and ultraviolet radiation [36]. Although CHX and MI as MMP inhibitors have been studied recently, they still have many aspects to research. Even CHX and MI cooperate with these additional approaches have not been studied.
In our hands, CHX and MI as MMP inhibitors can be used to pretreat the dentin bonding interface. Optimal treatment conditions are the application of CHX or MI for 60 sec after etching to stabilize the DHL and improve immediate bonding strength via inactivation of endogenous proteases. This treatment also prevented disintegration of DHL collagen fibrils. Thus, collagen-associated MMP inhibitors can prevent collagen fibers degradation during and after acid-etching but the long-term efficacy of different MMP inhibitor pretreatments warrant further study.
Acknowledgements
We acknowledgement the work of all authors, especially Professor Dr. Zhimin Zhang of Hospital of Stomatology, Jinlin University for his extensive technical support.
Disclosure of conflict of interest
None.
References
- 1.Opsahl Vital S, Gaucher C, Bardet C, Rowe PS, George A, Linglart A, Chaussain C. Tooth dentin defects reflect genetic disorders affecting bone mineralization. Bone. 2012;50:989–997. doi: 10.1016/j.bone.2012.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Loguercio AD, Moura SK, Pellizzaro A, Dal-Bianco K, Patzlaff RT, Grande RHM, Reis A. Durability of enamel bonding using two-step self-etch systems on ground and unground enamel. Operative Dentistry. 2008;33:79–88. doi: 10.2341/07-42. [DOI] [PubMed] [Google Scholar]
- 3.Hashimoto M. A Review-Micromorphological Evidence of Degradation in Resin-Dentin Bonds and Potential Preventional Solutions. J Biomed Mater Res B Appl Biomater. 2010;92B:268–280. doi: 10.1002/jbm.b.31535. [DOI] [PubMed] [Google Scholar]
- 4.Nakabayashi N, Kojima K, Masuhara E. The promotion of adhesion by the infiltration of monomers into tooth substrates. J Biomed Mater Res. 1982;16:265–273. doi: 10.1002/jbm.820160307. [DOI] [PubMed] [Google Scholar]
- 5.Mazzoni A, Pashley DH, Nishitani Y, Breschi L, Marinello F, Tjäderhane L, Toledano M, Pashley EL, Tay FR. Reactivation of inactivated endogenous proteolytic activities in phosphoric acid-etched dentine by etch-and-rinseadhesives. Biomaterials. 2006;27:4470–4476. doi: 10.1016/j.biomaterials.2006.01.040. [DOI] [PubMed] [Google Scholar]
- 6.De Munck J, Van den Steen PE, Mine A, Van Landuyt KL, Poitevin A, Opdenakker G, Van Meerbeek B. Inhibition of enzymatic degradation of adhesive-dentin interfaces. J Dent Res. 2009;88:1101–1106. doi: 10.1177/0022034509346952. [DOI] [PubMed] [Google Scholar]
- 7.Breschi L, Martin P, Mazzoni A, Nato F, Carrilho M, Tjaderhane L, Visintini E, Cadenaro M, Tay FR, Dorigo EDS, Pashley DH. Use of a specific MMP-inhibitor (galardin) for preservation of hybrid layer. Dent Mater. 2010;26:571–578. doi: 10.1016/j.dental.2010.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pashley DH, Tay FR, Breschi L, Tjäderhane L, Carvalho RM, Carrilho M, Tezvergil-Mutluay A. State of the art etch-and-rinse adhesives. Dent Mater. 2011;27:1–16. doi: 10.1016/j.dental.2010.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jacobsen JA, Fullagar JL, Miller MT, Cohen SM. Identifying Chelators for Metalloprotein Inhibitors Using a Fragment-Based Approach. J Med Chem. 2011;54:591–602. doi: 10.1021/jm101266s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sulkala M, Tervahartiala T, Sorsa T, Larmas M, Salo T, Tjäderhane L. Matrix metalloproteinase-8 (MMP-8) is the major collagenase in human dentin. Archives of Oral Biology. 2007;52:121–127. doi: 10.1016/j.archoralbio.2006.08.009. [DOI] [PubMed] [Google Scholar]
- 11.Mazzoni A, Mannello F, Tay FR, Tonti GAM, Papa S, Mazzotti G, Di Lenarda R, Pashley DH, Breschi L. Zymographic analysis and characterization of MMP-2 and-9 forms in human sound dentin. J Dent Res. 2007;86:436–440. doi: 10.1177/154405910708600509. [DOI] [PubMed] [Google Scholar]
- 12.Boukpessi T, Menashi S, Camoin L, TenCate JM, Goldberg M, Chaussain-Miller C. The effect of stromelysin-1 (MMP-3) on non-collagenous extracellular matrix proteins of demineralized dentin and the adhesive properties of restorative resins. Biomaterials. 2008;29:4367–4373. doi: 10.1016/j.biomaterials.2008.07.035. [DOI] [PubMed] [Google Scholar]
- 13.Chaussain C, Boukpessi T, Khaddam M, Tjäderhane L, George A, Menashi S. Dentin matrix degradation by host matrix metalloproteinases: inhibition and clinical perspectives toward regeneration. Front Physiol. 2013;4:308. doi: 10.3389/fphys.2013.00308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nishitani Y, Yoshiyama M, Wadgaonkar B, Breschi L, Mannello F, Mazzoni A, Carvalho RM, Tjäderhane L, Tay FR, Pashley DH. Activation of gelatinolytic/collagenolytic activity in dentin by self-etching adhesives. Eur J Oral Sci. 2006;114:160–166. doi: 10.1111/j.1600-0722.2006.00342.x. [DOI] [PubMed] [Google Scholar]
- 15.Carrilho MR, Tay FR, Donnelly AM, Agee KA, Tjäderhane L, Mazzoni A, Breschi L, Foulger S, Pashley DH. Host-derived loss of dentin matrix stiffness associated with solubilization of collagen. J Biomed Mater Res B Appl Biomater. 2009;90:373–380. doi: 10.1002/jbm.b.31295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Perdigao J, Reis A, Loguercio AD. Dentin adhesion and MMPs: a comprehensive review. J Esthet Restor Dent. 2013;25:219–241. doi: 10.1111/jerd.12016. [DOI] [PubMed] [Google Scholar]
- 17.Scaffa PMC, Vidal CM, Barros N, Gesteira TF, Carmona AK, Breschi L, Pashley DH, Tjäderhane L, Tersariol IL, Nascimento FD, Carrilho MR. Chlorhexidine inhibits the activity of dental cysteine cathepsins. J Dent Res. 2012;91:420–425. doi: 10.1177/0022034511435329. [DOI] [PubMed] [Google Scholar]
- 18.Stanislawczuk R, Costa JA, Polli LG, Reis A, Loguercio AD. Effect of tetracycline on the bond performance of etch-and-rinse adhesives to dentin. Braz Oral Res. 2011;25:459–465. doi: 10.1590/s1806-83242011000500014. [DOI] [PubMed] [Google Scholar]
- 19.Carrilho MR, Geraldeli S, Tay F, de Goes MF, Carvalho RM, Tjäderhane L, Reis AF, Hebling J, Mazzoni A, Breschi L, Pashley D. In vivo preservation of the hybrid layer by chlorhexidine. J Dent Res. 2007;86:529–533. doi: 10.1177/154405910708600608. [DOI] [PubMed] [Google Scholar]
- 20.Tezvergil-Mutluay A, Agee KA, Hoshika T, Tay FR, Pashley DH. The inhibitory effect of polyvinylphosphonic acid on functional matrix metalloproteinase activities in human demineralized dentin. Acta Biomaterialia. 2010;6:4136–4142. doi: 10.1016/j.actbio.2010.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tjäderhane L, Hotakainen T, Kinnunen S, Ahonen M, Salo T. The effect of chemical inhibition of matrix metalloproteinases on the size of experimentally induced apical periodontitis. Int Endod J. 2007;40:491–491. doi: 10.1111/j.0143-2885.2007.01223.x. [DOI] [PubMed] [Google Scholar]
- 22.Osorio R, Yamauti M, Osorio E, Ruiz-Requena ME, Pashley DH, Tay FR, Toledano M. Zinc reduces collagen degradation in demineralized human dentin explants. J Dent. 2011;39:148–153. doi: 10.1016/j.jdent.2010.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tallant C, Marrero A, Gomis-Ruth FX. Matrix metalloproteinases: Fold and function of their catalytic domains. Biochim Biophys Acta. 2010;1803:20–28. doi: 10.1016/j.bbamcr.2009.04.003. [DOI] [PubMed] [Google Scholar]
- 24.Breschi L, Prati C, Gobbi P, Pashley D, Mazzotti G, Teti G, Perdigao J. Immunohistochemical analysis of collagen fibrils within the hybrid layer: A FEISEM study. Oper Dent. 2004;29:538–546. [PubMed] [Google Scholar]
- 25.Van Meerbeek B, van Landuyt K, de Munck J, Hashimoto M, Peumans M, Lambrechts P, Yoshida Y, Inoue S, Suzuki K. Technique-sensitivity of contemporary adhesives. Dent Mater J. 2005;24:1–13. doi: 10.4012/dmj.24.1. [DOI] [PubMed] [Google Scholar]
- 26.De Munck J, Van Landuyt K, Peumans M, Poitevin A, Lambrechts P, Braem M, Van Meerbeek B. A critical review of the durability of adhesion to tooth tissue: methods and results. J Dent Res. 2005;84:118–132. doi: 10.1177/154405910508400204. [DOI] [PubMed] [Google Scholar]
- 27.Hamouda IM, Samra NR, Badawi MF. Microtensile bond strength of etch and rinse versus self-etch adhesive systems. J Mech Behav Biomed Mater. 2011;4:461–466. doi: 10.1016/j.jmbbm.2010.12.007. [DOI] [PubMed] [Google Scholar]
- 28.Vennat E, Bogicevic C, Fleureau JM, Degrange M. Demineralized dentin 3D porosity and pore size distribution using mercury porosimetry. Dent Mater. 2009;25:729–735. doi: 10.1016/j.dental.2008.12.002. [DOI] [PubMed] [Google Scholar]
- 29.Breschi L, Mazzoni A, Ruggeri A, Cadenaro M, Di Lenarda R, De Stefano Dorigo E. Dental adhesion review: aging and stability of the bonded interface. Dent Mater. 2008;24:90–101. doi: 10.1016/j.dental.2007.02.009. [DOI] [PubMed] [Google Scholar]
- 30.Pashley DH, Tay FR, Yiu C, Hashimoto M, Breschi L, Carvalho RM, Ito S. Collagen degradation by host-derived enzymes during aging. J Dent Res. 2004;83:216–221. doi: 10.1177/154405910408300306. [DOI] [PubMed] [Google Scholar]
- 31.Tezvergil-Mutluay A, Mutluay M, Seseogullari-Dirihan R, Agee KA, Key WO, Scheffel DLS, Breschi L, Mazzoni A, Tjäderhane L, Nishitani Y, Tay FR, Pashley DH. Effect of Phosphoric Acid on the Degradation of Human Dentin Matrix. J Dent Res. 2013;92:87–91. doi: 10.1177/0022034512466264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kandela IK, Bleher R, Albrecht RM. Multiple correlative immunolabeling microscopy using fluorophores and for light and electron colloidal metal particles. J Histochem Cytochem. 2007;55:983–990. doi: 10.1369/jhc.6A7124.2007. [DOI] [PubMed] [Google Scholar]
- 33.Breschi L, Mazzoni A, Nato F, Carrilho M, Visintini E, Tjäderhane L, Ruggeri A, Tay FR, Dorigo ED, Pashley DH. Chlorhexidine stabilizes the adhesive interface: A 2-year in vitro study. Dental Materials. 2010;26:320–325. doi: 10.1016/j.dental.2009.11.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tay FR, Pashley DH. Biomimetic Remineralization of Resin-bonded Acid-etched Dentin. J Dent Res. 2009;88:719–724. doi: 10.1177/0022034509341826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Costa AR, Naves LZ, Raposo L, Correr AB, Sinhoreti MA, Correr-Sobrinho L, Garcia-Godoy F, Puppin-Rontani RM. Long-term evaluation of ethanol wet-bonding to different dentin substrates. Dent Mater. 2013;29:e69. [Google Scholar]
- 36.Tjäderhane L, Nascimento FD, Breschi L, Mazzoni A, Tersariol IL, Geraldeli S, Tezvergil-Mutluay A, Carrilho MR, Carvalho RM, Tay FR, Pashley DH. Optimizing dentin bond durability: Control of collagen degradation by matrix metalloproteinases and cysteine cathepsins. Dent Mater. 2013;29:116–135. doi: 10.1016/j.dental.2012.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]


