Abstract
Osteopontin (OPN) is a 33.7 kDa intrinsically disordered protein and a member of the SIBLING family of proteins. OPN is bearing a signal peptide for secretion into the extracellular space, where it exerts its main physiological function, the control of calcium biomineralization. It is often involved in tumorigenic processes influencing proliferation, migration and survival, as well as the adhesive properties of cancer cells via CD44 and integrin signaling pathways. Here we report the nearly complete NMR chemical shift assignment of recombinant human osteopontin.
Keywords: Osteopontin, Intrinsically disordered protein, Extracellular matrix, Biomineralization
Biological context
Osteopontin (OPN) is an intrinsically disordered protein (IDP) with a molecular mass of the recombinant version around 33.7 kDa. It is highly negatively charged (25 % of the protein are glutamic or aspartic acid residues) and target of several post translational modifications including phosphorylation and glycosylation (Christensen et al. 2005). OPN is involved in a multitude of physiological and pathophysiological processes and a wide variety of cell types express OPN (Kunii et al. 2009). In bone, both matrix-synthesizing osteoblasts and bone-resorbing osteoclasts secrete OPN into the extracellular space where the protein controls calcium mineralization and the attachment of osteogenic cells to the bone matrix. OPN can also add physical strength to the extracellular matrix (ECM) since it is being cross-linked by transglutaminase to other ECM-proteins, for example collagen (Kaartinen et al. 1999). OPN is also expressed by activated immune cells such as macrophages, T-cells and B-cells, acting as a cytokine and homing these cells to sites of inflammation or injury (Ashkar et al. 2000). Epithelial cells (breast, urinary tract, gall bladder..) secrete OPN into biological fluids like blood, urine and milk. OPN is also localized to the luminal surfaces of these cells suggesting a protective role against unwanted interactions with the environment (Brown et al. 1992). Aside it’s physiological role, OPN is overexpressed in a variety of malignant tissues exerting its tumorigenic function by interacting with CD44 receptors and several integrin variants through RGD-dependent (Arginine-Glycine-Aspartic Acid) and independent mechanisms altering cell signaling events that ultimately lead to the formation of metastases (Rangaswami et al. 2006).
Methods and results
Protein expression and purification
The coding region of human Osteopontin (hOPN), excluding the first 16 N-terminal amino acids composing the signal peptide sequence, was amplified by PCR from a mammalian expression vector pDNR-LIB (Thermo Scientific Bio-Clone ID: 3828885) introducing a 5′ NotI and a 3′ NcoI restriction site. The obtained fragment was inserted in-frame into the bacterial expression vector pet-M11 (Pinotsis et al. 2006) yielding pet-M11-hopn coding for human hOPN fused to an N-terminal His6-Tag with a TEV-cleveage site separating the coding region of hOPN from the tag. 15N/13C labeled protein was expressed in the E. Coli strain BL21(DE3) (New England Biolabs) in isotopically labeled minimal media following the protocol of Marley et al. (2001). Protein expression was induced at an A600 nm of 0.8 by addition of a final concentration of 1 mM IPTG. Cells were harvested after 16 h of expression at 28 °C by centrifugation at 4,500 rpm for 20 min. The bacterial pellet was resuspended 20 ml of 1xPBS low Imidazole buffer (140 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.8 mM KH2PO4 and 20 mM Imidazole) per liter of bacterial culture. Bacterial cells were lysed by sonication and subsequently subjected to boiling (10 min at 95 °C) removing all heat-unstable impurities including proteases. The obtained cell lysate was cleared by centrifugation at 18,000 rpm for 20 min. The supernatant containing the soluble protein fraction was loaded onto a 6 ml HisTrap FF Crude (GE Healthcare) affinity column pre-loaded wit Ni2+. After loading, the column was washed with 10 volumes of 1xPBS low Imidazole buffer before elution with 1xPBS high Imidazole buffer (140 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.8 mM KH2PO4 and 500 mM Imidazole) using a step gradient. The collected fraction containing H6-hOPN was then loaded onto a ResourceQ 6 ml ion exchange column (GE Healthcare). After a wash step with 10 column volumes of 1xPBS buffer (140 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.8 mM KH2PO4), the protein was eluted using 1xPBS high salt buffer (1 M NaCl, 2.7 mM KCl, 10 mM Na2HPO4 and 1.8 mM KH2PO4) with a linear gradient over 10 column volumes. Fractions containing the protein were pooled and concentrated to 500 μl using a 15 ml Amicon Centrifugal Filter Unit 10000MWCO (Millipore). TEV cleavage was performed in 1xPBS cleavage buffer (140 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.8 mM KH2PO4, 1 mM DTT and 1 mM EDTA) by incubating H6-hOPN with 1 mg of TEV protease for every 50 mg of protein for 16 h at 4 °C. A final ion-exchange step was used to remove the cleaved HisTag and TEV protease. hOPN was concentrated to a final concentration of 0,55 mM in phosphate buffer (50 mM NaCl and 50 mM sodium phosphate, pH 6.5) for subsequent NMR analysis.
NMR experiments
All spectra were acquired at 293 K on an Agilent Direct Drive 700 MHz spectrometer using the standard 5 mm 1H–13C–15N triple-resonance probe head. The backbone 1H, 13C and 15N resonances were assigned using sparse random sampling of indirectly detected time domains, in order to increase resolution. A 3D HNCO experiment was used as a base spectrum for SMFT (Sparse Multidimensional Fourier Transform) processing of higher dimensionality experiments (Kazimierczuk et al. 2009). Backbone assignment was achieved using 5D HN(CA)CONH (Kazimierczuk et al. 2010), (HACA)CON(CA)CONH (Zawadzka-Kazimierczuk et al. 2012b), (H)NCO(NCA)CONH (Zawadzka-Kazimierczuk et al. 2012b) and HNCOCACB (Zawadzka-Kazimierczuk et al. 2012b) experiments. Side-chain assignments were obtained using 5D HabCabCONH (Kazimierczuk et al. 2010) and HC(CC-tocsy)CONH (Kazimierczuk et al. 2009; Hiller et al. 2008) experiments.
All NMR data sets were processed by multidimensional Fourier transformation using the home written software package (http://nmr.cent3.uw.edu.pl/software). The resonance assignment was performed using the TSAR program (Zawadzka-Kazimierczuk et al. 2012a). The input data for TSAR was prepared using Sparky (Goddard and Kneller 2002).
Extent of assignment and data deposition
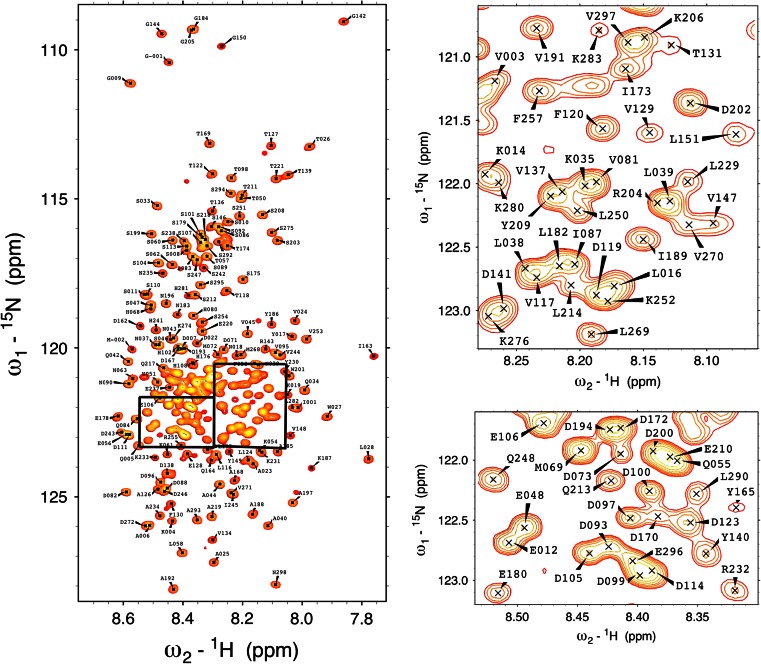
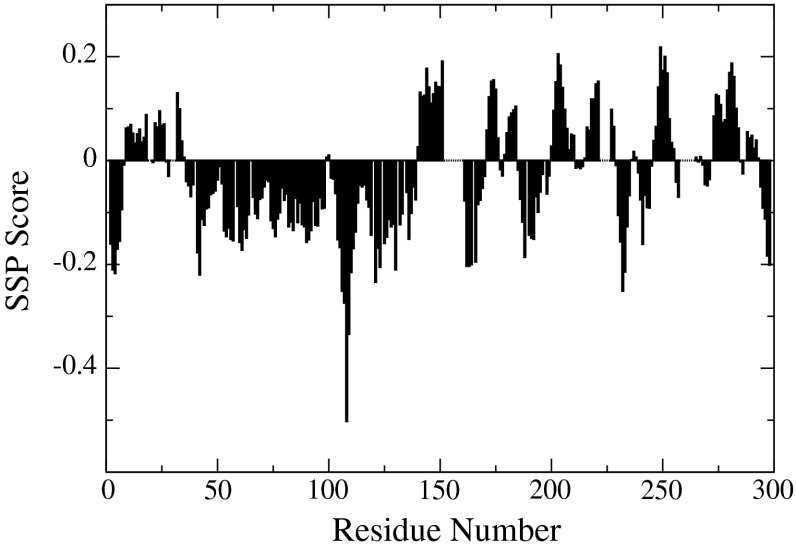
As expected for an intrinsically disordered protein the proton chemical shift dispersion of the 1H–15N HSQC spectrum (Fig. 1) shows a narrow profile with chemical shift values close to random coil values. Despite being classified as an IDP, a thorough characterization of a quail OPN homologue shows that the protein partially occupies stably folded substructures (Platzer et al. 2011; Kurzbach et al. 2013). Extensive signal overlap in conventional 2D & 3D spectra could be overcome by using the aforementioned 5D experiments. Additionally, signal assignment was supported by 3D HNN and HN(C)N experiments (Panchal et al. 2001). Notably, several segments of the protein remained undetectable. Presumably, the high percentage of basic residues in the missing regions (152–160, 222–226, 258–265) gives rise to an increased hydrogen exchange rate for the backbone amides leading to exchange broadening below the limit of detection (Molday et al. 1972). In total 90 % of backbone 15N, 90 % of 1HN, 88 % of 13Cα, 87 % of 13Cβ and 83 % of 13C′ resonances have been assigned. Additionally, HC(CC-tocsy)CONH spectra allowed the unambiguous assignment of 46 % side-chain carbons and 12 % side-chain protons. The SSP (secondary structure propensity) score (Marsh et al. 2006) only shows small deviations from random coil values (Fig. 2) with a long conformationally extended stretch (negative index values) in the N-terminus of the protein.
Fig. 1.
1H–15N HSQC spectrum of hOPN at pH6.5 at 293 K. For clarity reasons, the labeling in the highly crowded aspartic and glutamic acid rich region has been omitted in this figure
Fig. 2.
Secondary structure propensity (SSP) scores for hOPN using NH, HN, Cα, Cβ and CO chemical shifts. Positive values represent α-helical propensities and negative values represent extended or β-strand propensities
The 1H, 13C and 15N chemical shifts have been deposited in the BioMagResBank (http://www.bmrb.wisc.edu/) under the BMRB accession number 19999.
Acknowledgments
All NMR experiments were carried out in the Structural Research Laboratory at the Faculty of Chemistry, University of Warsaw. This work was supported by the EAST-NMR project as part of a transnational access program (proposal acronym: HOPNASSIGNMENT) and by the grant W-1221-B03 by the Austrian Science Foundation FWF. S. Ż, S. S and W. K. thank the Foundation for Polish Science for support with the TEAM Programme. Co-financed by the EU European Regional Development.
Contributor Information
Gerald Platzer, Email: gerald.platzer@univie.ac.at.
Robert Konrat, Email: robert.konrat@univie.ac.at.
References
- Ashkar S, Weber GF, Panoutsakopoulou V, Sanchirico ME, Jansson M, Zawaideh S, Rittling SR, Denhardt DT, Glimcher MJ, Cantor H. Eta-1 (osteopontin): an early component of type-1 (cell-mediated) immunity. Science. 2000;287(5454):860–864. doi: 10.1126/science.287.5454.860. [DOI] [PubMed] [Google Scholar]
- Brown LF, Berse B, Van de Water L, Papadopoulos-Sergiou A, Perruzzi CA, Manseau EJ, Dvorak HF, Senger DR. Expression and distribution of osteopontin in human tissues: widespread association with luminal epithelial surfaces. Mol Biol Cell. 1992;3(10):1169–1180. doi: 10.1091/mbc.3.10.1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christensen B, Nielsen MS, Haselmann KF, Petersen TE, Sorensen ES. Post-translationally modified residues of native human osteopontin are located in clusters: identification of 36 phosphorylation and five O-glycosylation sites and their biological implications. Biochem J. 2005;390(Pt 1):285–292. doi: 10.1042/BJ20050341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goddard TD, Kneller DG. Sparky 3. San Francisco: University of California; 2002. [Google Scholar]
- Hiller S, Joss R, Wider G (2008) Automated NMR assignment of protein side chain resonances using automated projection spectroscopy (APSY) experiments. JACS 130(36):12073–12079 [DOI] [PubMed]
- Kaartinen MT, Pirhonen A, Linnala-Kankkunen A, Maenpaa PH. Cross-linking of osteopontin by tissue transglutaminase increases its collagen binding properties. J Biol Chem. 1999;274(3):1729–1735. doi: 10.1074/jbc.274.3.1729. [DOI] [PubMed] [Google Scholar]
- Kazimierczuk K, Zawadzka A, Koźmiński W. Narrow peaks and high dimensionalities: exploiting the advantages of random sampling. J Magn Reson. 2009;197(2):219–228. doi: 10.1016/j.jmr.2009.01.003. [DOI] [PubMed] [Google Scholar]
- Kazimierczuk K, Zawadzka-Kazimierczuk A, Koźmiński W. Non-uniform frequency domain for optimal exploitation of non-uniform sampling. J Magn Reson. 2010;205(2):286–292. doi: 10.1016/j.jmr.2010.05.012. [DOI] [PubMed] [Google Scholar]
- Kunii Y, Niwa S, Hagiwara Y, Maeda M, Seitoh T, Suzuki T. The immunohistochemical expression profile of osteopontin in normal human tissues using two site-specific antibodies reveals a wide distribution of positive cells and extensive expression in the central and peripheral nervous systems. Med Mol Morphol. 2009;42(3):155–161. doi: 10.1007/s00795-009-0459-6. [DOI] [PubMed] [Google Scholar]
- Kurzbach D, Platzer G, Schwarz TC, Henen MA, Konrat R, Hinderberger D. Cooperative unfolding of compact conformations of the intrinsically disordered protein osteopontin. Biochemistry. 2013;52:5167–5175. doi: 10.1021/bi400502c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marley J, Lu M, Bracken C. A method for efficient isotopic labeling of recombinant proteins. J Biomol NMR. 2001;20(1):71–75. doi: 10.1023/A:1011254402785. [DOI] [PubMed] [Google Scholar]
- Marsh JA, Singh VK, Jia Z, Forman-Kay JD. Sensitivity of secondary structure propensities to sequence differences between alpha- and gamma-synuclein: implications for fibrillation. Protein Sci. 2006;15(12):2795–2804. doi: 10.1110/ps.062465306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molday RS, Englander SW, Kallen RG. Primary structure effects on peptide group hydrogen exchange. Biochemistry. 1972;11(2):150–158. doi: 10.1021/bi00752a003. [DOI] [PubMed] [Google Scholar]
- Panchal SC, Bhavesh NS, Hosur RV. Improved 3D triple resonance experiments, HNN and HN(C)N, for HN and 15N sequential correlations in (13C, 15N) labeled proteins: application to unfolded proteins. J Biomol NMR. 2001;20(2):135–147. doi: 10.1023/A:1011239023422. [DOI] [PubMed] [Google Scholar]
- Pinotsis N, Petoukhov M, Lange S, Svergun D, Zou P, Gautel M, Wilmanns M. Evidence for a dimeric assembly of two titin/telethonin complexes induced by the telethonin C-terminus. J Struct Biol. 2006;155(2):239–250. doi: 10.1016/j.jsb.2006.03.028. [DOI] [PubMed] [Google Scholar]
- Platzer G, Schedlbauer A, Chemelli A, Ozdowy P, Coudevylle N, Auer R, Kontaxis G, Hartl M, Miles AJ, Wallace BA, Glatter O, Bister K, Konrat R. The metastasis-associated extracellular matrix protein osteopontin forms transient structure in ligand interaction sites. Biochemistry. 2011;50(27):6113–6124. doi: 10.1021/bi200291e. [DOI] [PubMed] [Google Scholar]
- Rangaswami H, Bulbule A, Kundu GC. Osteopontin: role in cell signaling and cancer progression. Trends Cell Biol. 2006;16(2):79–87. doi: 10.1016/j.tcb.2005.12.005. [DOI] [PubMed] [Google Scholar]
- Zawadzka-Kazimierczuk A, Kazimierczuk K, Koźmiński W. A set of 4D NMR experiments of enhanced resolution for easy resonance assignment in proteins. J Magn Reson. 2010;202(1):109–116. doi: 10.1016/j.jmr.2009.10.006. [DOI] [PubMed] [Google Scholar]
- Zawadzka-Kazimierczuk A, Koźmiński W, Billeter M. TSAR: a program for automatic resonance assignment using 2D cross-sections of high dimensionality, high-resolution spectra. J Biomol NMR. 2012;54(1):81–95. doi: 10.1007/s10858-012-9652-3. [DOI] [PubMed] [Google Scholar]
- Zawadzka-Kazimierczuk A, Koźmiński W, Sanderova H, Krasny L. High dimensional and high resolution pulse sequences for backbone resonance assignment of intrinsically disordered proteins. J Biomol NMR. 2012;52(4):329–337. doi: 10.1007/s10858-012-9613-x. [DOI] [PMC free article] [PubMed] [Google Scholar]