Abstract
Congenital adrenal hyperplasias (CAH) are a group of autosomal recessive defects in cortisol biosynthesis. Substantial progress has been made since the description of the first report, 150 years ago. This article reviews some of the recent advances in the genetics, diagnosis and treatment of CAH. In addition, we underline the aspects where further progress is required, including, among others, better diagnostic modalities for the mild phenotype and for some of the rare forms of disease, elucidation of epigenetic factors that lead to different phenotypes in patients with identical genotype and expending on treatment options for controlling the adrenal androgen excess.
1. Introduction
In a recent issue of Endocrinology, Luisa Delle Piane and colleagues put the spotlight on the case regarded as the first report of non-salt wasting congenital adrenal hyperplasia (CAH), initially described in 1865 by Luigi de Crecchio1. The article details the autopsy of a prematurely deceased virilized female with enlarged adrenal glands, and the conundrum it presented to the team of pathologists involved2. Much has been learned over the past 150 years about CAH, now recognized as one of the most common inherited diseases. However, contemporary practitioners and researchers still have questions to answer and hypotheses to test even in the modern medical era. In this article, we focus on the recent advances in CAH and on those aspects where progress over the next century is imperative.
2. Congenital adrenal hyperplasia-brief overview
CAH is an umbrella term for inherited enzymatic deficiencies in cortisol synthesis (Figure 1). The defective cortisol production alleviates the negative feedback to the hypothalamus and pituitary gland, resulting in excessive secretion of corticotropin-releasing hormone (CRH) and adrenocorticotropin (ACTH), respectively. The raised ACTH, in turn, cannot overcome the block in cortisol synthesis, but its trophic action leads to enlargement of the adrenal glands. The enzymatic defects in all forms of CAH can be complete or partial, resulting into a broad spectrum of clinical presentations. The most severe forms are conventionally called “classic” CAH and are the easiest to recognize. Conversely, the milder forms or “nonclassic” CAH are often difficult to diagnose or may be overlooked, as their features overlap between each other, as well as with other clinical entities.
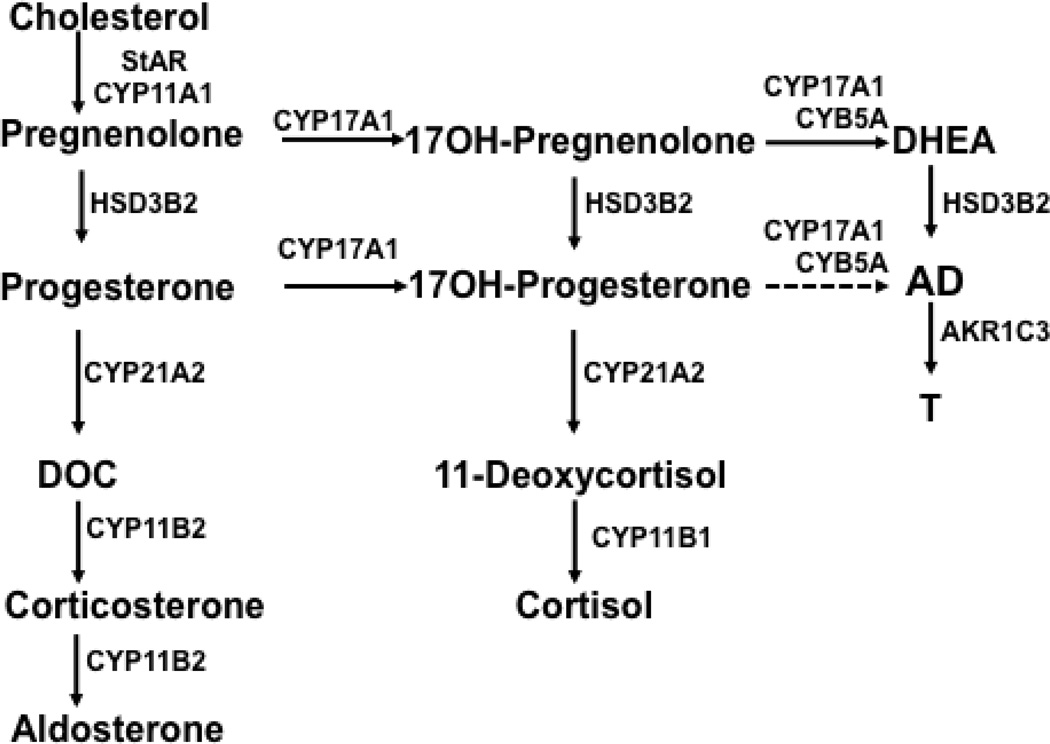
Figure 1.
Pathways of adrenal steroid hormone synthesis. StAR, steroidogenic acute regulatory protein; CYP11A1, cytochrome P450 cholesterol side-chain cleavage; HSD3B2, 3β-hydroxysteroid dehydrogenase type 2; CYP17A1, 17α-hydroxylase/17,20-lyase; CYB5A, cytochrome b5; CYP21A2, 21-hydroxylase; CYP11B1, 11β-hydroxylase; CYP11B2, aldosterone synthase; AKR1C3, 17β-hydroxysteroid dehydrogenase type 5; DOC, 11-deoxycorticosterone; AD, androstenedione; T, testosterone.
The most common form of CAH is 21-hydroxylase (P450c21, CYP21A2) deficiency (21OHD, Table 1), accounting for over 90% of all cases3. Classic 21OHD affects approximately 1 in 16,000 newborns worldwide4. Nonclassic 21OHD occurs in roughly 1 of 1,000 Caucasians and even more frequently in populations of specific ethnicities, such as Ashkenazi Jews (1:27), Hispanics (1:53), Yugoslavs (1:62) and Italians (1:300)5. Complete absence of CYP21A2 activity results in both glucocorticoid and mineralocorticoid deficiencies, as well as in severe adrenal-derived androgen excess. The androgen excess is clinically evident in newborn girls, whose external genitalia are virilized. Patients with nonclassic 21OHD do not have adrenal insufficiency, and they typically present with evidence of androgen excess, such as premature pubarche, hirsutism, acne, and irregular menses, at various ages.
Table 1.
Forms of congenital adrenal hyperplasia
| Defective enzyme |
Gene/Chromoso me |
Incidence & Populations |
Clinical features | Biomarkers |
|---|---|---|---|---|
| 21-hydroxylase (P450c21) |
CYP21A2/6p21.3 | Classic 1:16,000 Nonclassic <1:1,000 More frequent in Ashkenazi Jews, Mediterraneans, Hispanics, and Yupik Eskimos. |
Adrenal insufficiency in classic forms. Variable degrees of virilization. |
17OH- progesterone, AD, T |
| 11β-hydroxylase (P450c11β) |
CYP11B1/8q24.3 | 1:100,000 in Caucasians; 1:7,000 in Moroccan Jews |
Hypertension in most patients; hypokalemia; virilization |
DOC, 11- deoxycortisol, AD, T |
| 3β- hydroxysteroid dehydrogenase type 2 |
HSD3B2/1p13.1 | Rare | Volume depletion, hyponatremia, and hyperkalemia. 46,XX: virilization 46,XY: undervirilization |
Pregnenolone, 17OH- pregnenolone, DHEA, DHEAS |
| 17- hydroxylase/17,2 0-lyase (P450c17) |
CYP17A1/10q21- q22 |
1:50,000 worldwide; More common in Brazil and Asia |
Hypertension, hypokalemia, and hypogonadism; 46,XX: primary amenorrhea and absence of secondary sexual characteristics. 46,XY: undervirilization, abdominal testes. |
Progesterone, DOC, corticosterone; LH & FSH |
| Steroidogenic acute regulatory protein (StAR) |
STAR/8p11.2 | Rare More frequent in Japanese, Palestinians, Koreans |
Adrenal insufficiency; enlarged, lipid- laden adrenal glands. Female phenotype of external genitalia in both sexes |
All steroids decreased |
| Cholesterol side- chain cleavage enzyme (P450scc) |
CYP11A1/15q23 –q24 |
Rare | Adrenal insufficiency; adrenal glands may appear absent. |
All steroids decreased |
| P450- oxidoreductase deficiency (POR) |
POR/7q11.2 | Rare More common in Japan and Korea |
Volume depletion, skeletal malformations (Antley-Bixler); maternal virilization. 46,XX: mild-to- moderate virilization. 46,XY: undervirilization |
Highly variable profiles, multiple partial defects. |
CYP11B1, 11β-hydroxylase; HSD3B2, 3β-hydroxysteroid dehydrogenase type 2; POR, P450-oxidoreductase deficiency; DOC, 11-deoxycorticosterone; AD, androstenedione; T, testosterone; DHEA, dehydroepiandrosterone; DHEAS, DHEA sulfate; LH, luteinizing hormone; FSH, follicle-stimulating hormone.
A second form of CAH is 11β-hydroxylase (CYP11B1) deficiency (11OHD), which represents up to 5–8% of all CAH cases in some series of high-risk populations6,7, although the incidence of this disorder in the general population has never been ascertained. Its incidence is estimated at 1 in 100,000 live births in the general population but is 20 times higher in Moroccan Jews, due to a founder mutation8. CYP11B1 catalyzes the conversion of 11-deoxycorticosterone (DOC) and 11-deoxycortisol into corticosterone and cortisol, respectively, reactions whose substrates are products of CYP21A2. As in 21OHD, patients exhibit decreased cortisol synthesis and adrenal androgen overproduction. In contrast to 21OHD, the distinctive features of classic 11OHD are hypertension and less commonly hypokalemia, owing to the mineralocorticoid action of DOC, which manifests in two-thirds of patients often in mid-childhood and also makes patients with 11OHD less prone to adrenal crisis than those with 21OHD. Nonclassic 11OHD is difficult to distinguish clinically from nonclassic 21OHD but is far less common.
A third form of CAH is 3β-hydroxysteroid dehydrogenase/isomerase type 2 (3βHSD2) deficiency and is characterized by both mineralo- and glucocorticoid deficiency. This enzyme catalyzes the reactions preceding those of CYP21A2, being responsible for the C3 dehydrogenation and simultaneous Δ5 to Δ4 transfer of the double bond into the A ring of the core steroid structure. Humans have 2 homologous 3βHSD enzymes: type 1, expressed in placenta and peripheral tissues (skin, prostate and breast), and type 2, expressed in the adrenal glands and gonads9–11. Severe impairment of 3βHSD2 results in both mineralocorticoid and glucocorticoid deficiencies and limits steroid flux to dehydroepiandrosterone (DHEA). In peripheral tissues containing 3βHSD1 and downstream enzymes, DHEA is metabolized to androgens, which causes mild virilization in newborn girls, such as slight clitoral enlargement without the labioscrotal fusion found in 21OHD and 11OHD. In contrast with the latter two, boys are undervirilized, due to the impaired androgen synthesis in the testes, which also requires 3βHSD2. In women, nonclassic 3βHSD deficiency was thought to be common but is actually extremely rare, and most children with premature adrenarche do not have a mutation in the HSD3B2 gene (see section 3.1 and 3.2.2). Nonclassic 3βHSD deficiency is difficult to distinguish from nonclassic 21OHD and 11OHD or PCOS clinically, all presenting with hirsutism and oligomenorrhea.
Deficiency of 17α-hydroxylase/17,20-lyase (CYP17A1) is rare, with most cases described coming from Brazil and Asia. CYP17A1 is required for both cortisol and androgen synthesis. The only unaffected pathway is that to progesterone, aldosterone and other mineralocorticoids, and the weak glucocorticoid corticosterone, similar to the rodent adrenal that lacks this enzyme. In order to produce enough corticosterone to substitute for cortisol, DOC rises markedly, and patients present with hypertension, hypokalemia and hypogonadism. Both 46,XY and 46,XX individuals have feminine external genitalia and primarily present during pubertal age, with amenorrhea and absence of secondary sexual characteristics. Occasionally, 46,XY cases of 17OHD are diagnosed in infancy due to inguinal hernias. Partial defects of CYP17A1 activities have been found in women with poor breast development and/or menstrual abnormalities12, 13 and in males with ambiguous external genitalia, absence of male secondary sexual characteristics, and gynecomastia at puberty14, but a “nonclassic” form has not been described. Conceivably, patients with very mild or nonclassic 17OHD might be dismissed as having low-renin hypertension and primary hypogonadism and never evaluated for 17OHD. Isolated 17,20-lyase deficiency has been described in patients with mutations in the CYP17A115,16, POR17 or CYB5A18,19 genes, but these patients do not have CAH because cortisol synthesis is normal. Isolated 17,20-lyase deficiency can be difficult to distinguish from partial androgen insensitivity or from rare steroidogenic defects such as AKR1C2+AKR1C420.
Lipoid CAH (LCAH), the most severe defect in steroidogenesis, is named for the massively enlarged, lipid-laden adrenals characteristic of this disease. LCAH is caused by a defect in the steroidogenic acute regulatory protein (StAR), which prevents the mobilization of cholesterol into steroidogenic pathways and results in negligible production of all steroids. Cholesterol side-chain cleavage enzyme (P450scc) deficiency yields a similar severe global defect in steroidogenesis as LCAH but without the enlarged adrenals found with StAR deficiency21,22. Affected children typically present with life-threatening adrenal insufficiency in early infancy, and males appear phenotypically female due to impaired testicular androgen synthesis in utero. Nonclassic forms of both lipoid CAH and P450scc deficiency have been described, presenting with late-onset isolated glucocorticoid insufficiency and normal external genitalia23–26.
Finally, P450-oxidoreductase (POR) deficiency combines features of 21OHD, 17OHD, and aromatase (CYP19A1) deficiencies, in various combinations and severities27. In addition, these patients may present skeletal malformations, such as craniosynostosis, radiohumeral or radioulnar synostosis, and femoral bowing, as part of the Antley–Bixler syndrome28. In contrast, one of the initial cases described was a phenotypically normal woman with infertility, illustrating the wide spectrum of this disease and suggesting “nonclassic” disease could be defined by the absence of skeletal and/or genital anomalies.
3. Diagnosis of CAH
3.1. Hormonal testing
For each enzymatic defect, the precursor to product ratio is the mainstay of diagnosis. Cosyntropin stimulation maximizes these ratios and is particularly important for all cases with indeterminate baseline results29.
Newborn screening for 21OHD was first implemented in 197830,31, and is currently available throughout the United States and many other countries. Screening reduces the time to diagnosis in infants, particularly for boys who are often not diagnosed at birth and suffer crises several days later32,33. The most important role of early diagnosis is to reduce morbidity and mortality for severely affected babies, although this aspect remains controversial and might also depend on the economic and healthcare status of each country34–36. First-tier testing measures 17OHP in dried blood spots by an immunoflurometric assay. A random 17OHP >20,000 ng/dL is suggestive of 21OHD; however, false-positive results are commonly seen in premature and severely ill infants37,38. Factors contributing to false-positive screening include activation of the hypothalamic-pituitary-adrenal axis in response to perinatal stress, the relative immaturity of adrenal CYP11B1 activity in preterm infants39, which also elevates 17OHP39,40, and interfering steroids in the immunoassays. False-negative rates of up to 22% have been reported in infant screening41,42, particularly when mothers had been exposed to glucocorticoids prenatally. Although higher false-negative rates have been reported in girls42, it is possible that boys not identified with newborn screening remained undiagnosed for several years. Weight- and gestational age-adjusted cutoffs for 17OHP have been implemented to improve the positive predictive value of screening,43–45 and more specific second-tier screening procedures to adjudicate abnormal first-tier screens have been successfully implemented in some states46. Beyond neonatal screening, children with clinical evidence of androgen excess—such as pubic and axillary hair growth, oily skin, rapid somatic growth, and advanced skeletal maturation— initially undergo testing of a morning baseline 17OHP47. Intermediate screening values (200–1000 ng/dl) are followed by retesting after cosyntropin stimulation. The diagnosis of classic 21OHD is based on stimulated 17OHP levels above 10,000 ng/dl, while nonclassic 21OHD requires a 17OHP >1,000 ng/dl47.
Classic 11OHD and 3βHSD2 deficiency overlap clinically with classic 21OHD, and in all 3 conditions, 17OHP can be elevated. Due to their low prevalence, the diagnosis of 11OHD is considered when hypertension and/or hypokalemia are present, or when the 17OHP is lower than expected for the degree of androgen excess in a 21OHD patient. 3βHSD2 deficiency is considered if virilization is mild relative to the degree of salt wasting. Nonclassic 21OHD is vastly more common than other forms of nonclassic CAH, but these conditions are even more difficult to distinguish clinically than their classic forms. A diagnosis of nonclassic 11OHD or 3βHSD2 deficiencies should be pursued in patients with androgen excess during childhood only after more common causes (such as exposure to exogenous androgens, PCOS, 21OHD, adrenal or gonadal tumors, chorionic gonadotropin-secreting tumor) have been excluded. Nonclassic 11OHD is diagnosed when 11-deoxycortisol rises above 1,800 ng/dl and cortisol is >18 µg/dl after cosyntropin7. The diagnosis of nonclassic 3βHSD deficiency requires a 17-hydroxypregnenolone >3,000 ng/dl and a cortisol >18 µg/dl after cosyntropin, with a 17-hydroxypregnenolone/cortisol ratio >10 standard deviations above normal48.
In most centers, steroid measurements are performed by immunoassays, which are limited by cross-reactivity and can yield unreliable results, particularly when ordinarily minor steroids are markedly elevated. More accurate testing can be attained by using liquid chromatography/tandem mass spectrometry (LC-MS/MS)49–51. In addition to increased specificity and sensitivity, LC-MS/MS also affords quantitation of multiple steroids in a single measurement. Elevated 21-deoxycortisol by LC-MS/MS has been shown to increase the sensitivity of newborn screening50 and to discriminate heterozygote carriers of CYP21A2 mutations from nonclassic 21OHD better than 17OHP52. Simultaneous measurement of 11-deoxycortisol can distinguish 11OHD from 21OHD in second-tier screening. Other novel biomarkers for diagnosis, such as 16α-hydroxyprogesterone and 11β-hydroxyprogesterone, have been recently proposed (Fig. 2)53. Multi-steroid panels simultaneously testing all forms of CAH clinically in question from a small volume serum sample will likely dominate in the future. For now, however, the widespread use of LC-MS/MS is constrained by its limited availability, technical demands and high cost. Alternatively, comprehensive analysis of urinary steroid metabolites using gas chromatography-mass spectrometry (GC/MS) affords patterns characteristic for each form of CAH54. While GC/MS has been used for decades, the availability of this methodology is limited, and sample throughput is slow and tedious.
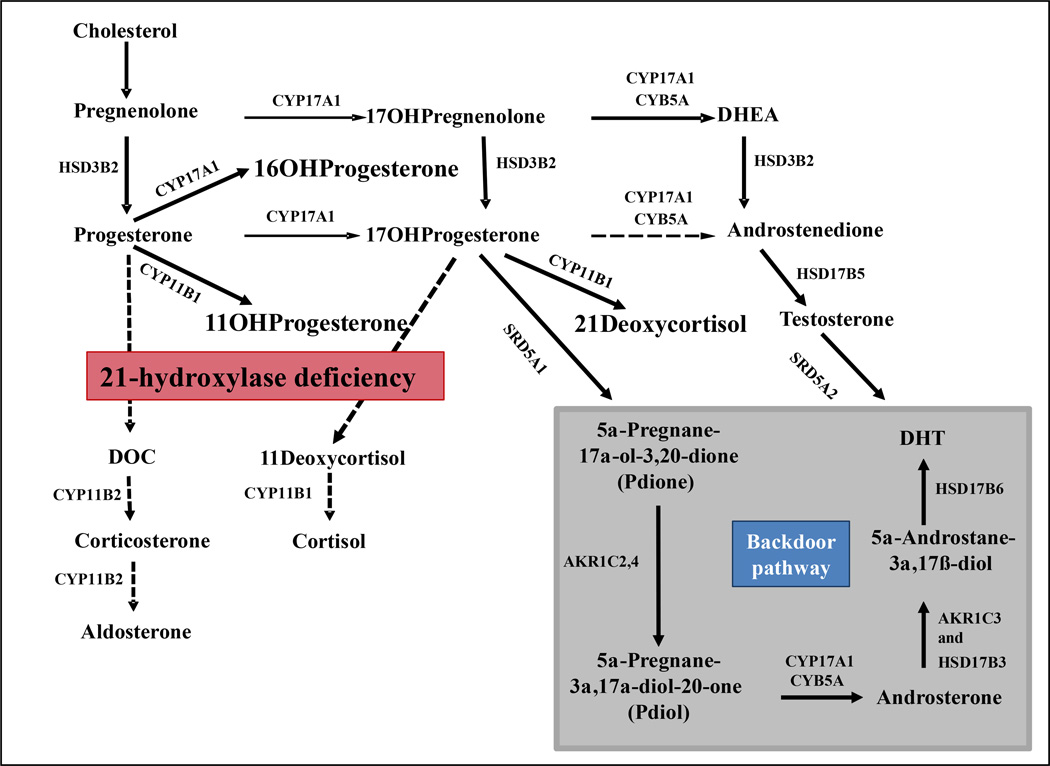
Figure 2.
Pathways of steroid hormone synthesis in 21-hydroxylase deficiency. HSD3B2, 3β-hydroxysteroid dehydrogenase type 2; CYP17A1, 17α-hydroxylase/17,20-lyase; CYB5A, cytochrome b5; CYP11B1, 11β-hydroxylase; CYP11B2, aldosterone synthase; DOC, 11-deoxycorticosterone; DHT, dehydrotestosterone; AKR1C3, 17β-hydroxysteroid dehydrogenase type 5; AKR1C2,4, aldo-keto reductase types 1C2 and 1C4; HSD17B6, 17β-hydroxysteroid dehydrogenase type 6 (an oxidative 3α-HSD); SRD5A1 and SRD5A2, 5α-reductase, types 1 and / 2, respectively.
3.2. Genetics of CAH and role of genetic testing
3.2.1. Genetics of CAH
All CAH forms are monogenic, autosomal-recessive disorders. The gene encoding human CYP21A2 is located on chromosome 6p21.3, within the human leucocyte antigen (HLA) major histocompatibility complex and adjacent to the genes for the fourth component of complement55–57. In addition to this active gene, humans also have a 98% homologous pseudogene (CYP21A1P), situated within 30 kb, which encodes a truncated, inactive enzyme. Most of the mutant 21OHD alleles occur by intergenic recombinations and gene conversion between the two CYP21A genes58. Mutations that result in complete or nearly complete compromise of 21-hydroxylase activity (such as complete deletions, large gene conversions, and non-sense or frame-shift mutations), typically result in classic 21OHD. Nonclassic 21OHD alleles preserve up to 20–30% of the enzyme activity, which is sufficient for adequate cortisol and aldosterone production. A strong genotype-phenotype correlation exists for classic 21OHD; however, the clinical manifestations of nonclassic disease are quite variable, suggesting that other factors (genetic, epigenetic or environmental) may affect the phenotypic expression58. The elucidation of these additional factors is an important priority for future CAH research. The human 11-hydroxylase (CYP11B1) gene comprises nine exons and encodes a protein of 503 amino acids.
The CYP11B1 gene is located on chromosome 8q21-22, approximately 40 kb apart from the highly homologous aldosterone synthase gene (CYP11B2)59. Over 80 mutations have been described to date, the majority of which are associated with classic 11OHD60–72. Relatively few mutations associated with nonclassic 11OHD have been identified72–76, most over the recent years. Earlier studies on women with androgen excess failed to identify mutations in the CYP11B1 gene, even in the presence of elevated precursor 11-deoxycortisol76. The relationship between genotype and phenotype remains unclear; for example, blood pressure correlates poorly with serum DOC concentrations.
The HSD3B2 gene, located on chromosome 1p13.1, is expressed almost exclusively in the adrenal and gonads10. The highly homologous type I 3βHSD gene (HSD3B1) is located in vicinity on the same chromosome, but it is expressed in placenta and peripheral tissues, such as skin, breast and prostate77. The HSD3B2 gene consists of four exons, of which exons 2–4 are translated into a protein of 371 amino acids77. In proximity reside 5 pseudogenes (HSD3Bψ1–5); two of these pseudogenes (ψ1 and ψ2) separate the two expressed HSD3B1 and HSD3B2 genes, preventing them from sharing common promoter elements78. Thus, HSD3B1 is usually intact in patients with 3βHSD2 deficiency, explaining why serum concentrations of some Δ4 steroids can be normal or even elevated in these patients79. A strong genotype-phenotype correlation exists. Nonsense and frame-shift mutations that ablate enzyme transcription or function result in salt-wasting forms of 3βHSD2 deficiency. Conversely, single amino-acid substitutions that moderately decrease the affinity of the enzyme for substrate or cofactors lead to non-salt-wasting forms of 3βHSD2 deficiency80–84. However, some mutations, like A82T or T259M, have been associated with phenotypic heterogeneity77.
Although human CYP17A1 catalyzes two separate reactions, it is encoded by a single gene located on chromosome 10q24.3, which consists of 8 exons and encodes a 508-amino-acid protein85–87. Over 90 CYP17A1 mutations have been described throughout the entire gene. Populations in which 17OHD is more prevalent, such as Brazilians88, Canadian Mennonites, Dutch Frieslanders89, Japanese90, and patients from East Asia91, however, have specific reoccurring mutations, due to founder effects. Rare mutations located in the redox-partner interaction site or active site can lead to isolated 17,20-lyase deficiency15,16,92,93.
3.2.2. Clinical use of genetic testing
The role of genetic testing is best established in cases that remain equivocal after cosyntropin stimulation or for prenatal genetic counseling. Up to 70% of patients with nonclassic 21OHD carry a classic 21OHD mutation on one of their alleles, rendering them a carrier for classic 21OHD94, 95. Children of women with nonclassic 21OHD have an approximately 2.5% risk of being born with classic 21OHD and a 15% risk for having nonclassic 21OHD96. The same increased risk of 21OHD theoretically applies equally to fathers with nonclassic 21OHD, but similar data for men are lacking. The risk increases significantly in ethnic groups known to have a higher prevalence of the disease96. Thus, particularly in cases of a known affected prospective parent, preconception genetic testing of the second parent might help families to understand the risk of an affected offspring and to receive appropriate genetic counseling.
Another role for genetic testing evolved from efforts to prevent virilization of female fetuses affected with classic 21OHD. Prenatal treatment of the mother carrying an affected child with dexamethasone can reduce genital virilization compared to sisters who were not treated in utero. Prenatal diagnosis of 21OHD has been accomplished by chorionic villus sampling at approximately 14 weeks of gestation or by amniocentesis at approximately 20 weeks97. Both approaches are invasive and increase the risk of miscarriages98–100. To be successful, however, dexamethasone treatment must be started by 8 weeks’ gestation, when genital anatomy is sensitive to dihydrotestosterone action101, 102. Thus, presumptive treatment must be started before the prenatal diagnosis can be established. Statistically, only 1 in 8 fetuses is an affected girl, so up to 88% of pregnancies are treated unnecessarily. Prenatal dexamethasone was associated with impaired verbal working memory and related cognitive metrics in one study,103 and more recent data have suggested that exposure to prenatal synthetic glucocorticoids interferes with the normal brain development104,105. Thus, prenatal therapy is currently regarded as experimental by academic societies47,106.
In recent years, early noninvasive genetic testing has been proposed. Lo and colleagues first documented the presence of cell-free fetal DNA (cff-DNA) in the maternal circulation in 1997107. Quantitative polymerase chain reaction analysis of the SRY gene in maternal plasma was introduced in 2001, as a sensitive method for fetal gender determination in women with CAH108. Technological advances in genetic testing have recently made the diagnosis of 21OHD possible as early as 6 weeks of gestation, by targeted massively parallel sequencing performed on cff-DNA circulating in maternal plasma109. Rarely, prenatal DNA sequencing can lead to erroneous diagnosis of CAH, as in duplication of the CYP21A2 gene110. Moreover, massively parallel sequencing remains technically challenging, costly and only available in selected centers.
As genetic testing becomes cheaper and more widely available, one can anticipate that targeted sequencing of several genes of interest could clarify the diagnosis in patients with atypical clinical presentations and borderline biochemical workup. Diagnosis of all forms of CAH currently relies almost exclusively on hormonal testing, which is fraught with pitfalls. For example, earlier studies of hirsute women over-diagnosed nonclassic 3βHSD2 deficiency, when relying on measurement of Δ5 steroids111–114. Genetic testing uncovered mutations in the HSD3B2 gene in only a vanishingly small number of such women48, 115. As genetic databases continue to grow, more robust links between genotype and phenotype will develop. In the interim however, the clinical and biochemical context remain critical for the diagnosis of this heterogeneous group of diseases.
4. Treatment of CAH
4.1. Conventional and experimental glucocorticoid and mineralocorticoid therapy
The two general goals of treatment in all forms of CAH are to replace the deficient hormones on one hand, and to offset the undesirable effects of excessive hormonal production on the other. With a variety of available formulations, corticosteroid replacement has become relatively facile, with some limitations. For example, the aldosterone substitute fludrocortisone acetate is not available in mainland China; hydrocortisone tablets are not sold in many countries including Brazil; and liquid forms of hydrocortisone must be obtained from compounding pharmacies without quality control in the United States. Conversely, management of adrenal androgen excess in both classic and nonclassic CAH remains challenging. Several strategies to counteract the excessive adrenal androgen synthesis have been explored, as outlined below.
The most commonly used approach has been that of suppressing ACTH by strategic dosing of glucocorticoids. The typical short- or intermediate-acting glucocorticoid regimens, which are sufficient for replacement in non-CAH related adrenal insufficiency, often fail to blunt the early morning rise of ACTH, which is the principal drive for adrenal androgen overproduction. Physicians have resorted to supraphysiologic or non-physiologic (nocturnal) doses of glucocorticoids in an attempt to counteract the excessive androgen synthesis, thus promoting bone loss, obesity and features of metabolic syndrome in these patients116,117. Sustained-released hydrocortisone preparations have been recently developed, attempting to mimic the cortisol circadian rhythm and to suppress the early morning ACTH elevation118,119. In a phase two clinical trial, a modified-release formulation of hydrocortisone administered once daily mimicked the normal diurnal cortisol rhythm more closely than conventional hydrocortisone dosing; however, androstenedione rose higher than with conventional hydrocortisone in the afternoon. After reformulation, a second phase two trial with twice-daily dosing achieved lower hydrocortisone dose equivalent, as well as lower 17OHP and adrostenedione levels, compared to conventional therapies118. Continuous subcutaneous hydrocortisone infusion via a pump, mimicking a circadian secretory profile, has been used experimentally in young patients with increased cortisol clearance, and this approach was shown to decrease 17OHP and adrenal androgen production with a lower total daily dose120,121.
4.2. Experimental therapies beyond glucocorticoids
Another alternative for decreasing ACTH was undertaken in a recent proof-of-concept single-blind, placebo-controlled, single center, fixed-sequence, single-dose trial study, using the corticotropin-releasing factor receptor type 1 antagonist NBI77860122. At 300 and 600 mg, a nocturnal dose lowered morning ACTH and 17OHP by >50% from baseline in 4 of 8 participants, and the action correlated with drug exposure. Further studies are needed to determine the effects of NBI77860 on key parameters of disease control such as serum androgens when given in repeated doses at steady state.
A second strategy is that of directly inhibiting androgen synthesis or antagonizing androgen action. The combination of flutamide, an antiandrogen, and testolactone, an aromatase inhibitor, permitted the use of lower doses of hydrocortisone and fludrocortisone acetate and normalized linear growth and bone maturation in children followed for 2 years123,124. Long-term results from this trial are anticipated soon; however, latter-generation anti-androgens and aromatase inhibitors have supplanted both flutamide and testolactone for the treatment of prostate and breast cancers, respectively. A GnRH antagonist has been successful in improving height in children with 21OHD and precocious puberty125. Abiraterone acetate, a potent CYP17A1 inhibitor approved for use in castration-resistant prostate cancer, normalized androstenedione in adult women with classic 21OHD and elevated androgens when added to physiologic hydrocortisone and fludrocortisone acetate replacement in one short-term study126.
Monitoring and titrating treatment remains a major clinical challenge, and no consensus exists among practitioners. Although 17OHP has long been used to guide the management of 21OHD, its serum concentration correlates poorly with DHEA and androstenedione127. DHEAS, the major C19 adrenal product, can be paradoxically low in 21OHD patients with inadequate control128–130. Moreover, there is no good correlation between the routinely measured androgens (androstenedione and, in women, testosterone) and clinical evidence of androgen excess131,132. Research conducted in the recent years has brought into consideration non-conventional androgens and precursors, such as steroids derived from the so-called “back-door pathway”133 (Figure 2). This pathway starts with two consecutive 5α-, then 3α- reductions of 17OHP and leads via androsterone and 5α-androstane-3α,17β-diol to dihydrotestosterone, the most potent endogenous androgen, while bypassing androstenedione and testosterone. Kamrath and colleagues found increased excretion of 5α-reduced products and intermediates of the backdoor pathway in 142 children and young adults with CAH, compared with 138 similarly aged controls, particularly during the first year of life127. Consequently, this pathway has been proposed to contribute to the virilization of female fetuses with 21OHD,134 as has been shown in POR deficiency135. In addition, the adrenal glands also produce other active androgens, such as 11β-hydoxytestosterone. Substantial amounts of 11-oxygenated C19 steroids were documented in adrenal vein samples obtained from normal adrenals, with 11β-hydroxyandrostenedione being particularly abundant136. In a cell-based androgen transactivation assay, 11β-hydroxytestosterone and 11-ketotestostereone activated the androgen receptors at concentration approximately ten times higher than testosterone, but 30 times lower than androstenedione and 11β-hydroxyandrostenedione 136. The contribution of these steroids to the androgen excess of 21OHD remains to be further investigated. As these 11-oxygenated C19 steroids are products of CYP11B1, an adrenal-specific enzyme, these compounds might serve as specific biomarkers of adrenal-derived androgen excess and facilitate titration of therapy.
5. Summary
We have reviewed the state of knowledge of CAH 150 years after the initial description and highlighted some recent advances in the field. Based on this review, we propose a research agenda and related questions for improved understanding and management of CAH (Box 1).
Box 1. Areas of future research in congenital adrenal hyperplasia (CAH).
Research agenda
- Diagnosis
- Biomarker panels for improved diagnosis of all nonclassic CAH forms
- Early prenatal genetic diagnosis
- Treatment
- Characterization of biomarkers for treatment monitoring and titration
- Improved treatment modalities, to allow suppression of androgen excess while avoiding supraphysiologic doses of glucocorticoids.
Elucidation of modifier genes/epigenetic contributors to phenotypes
1. The mild phenotype
What would be the phenotype of a patient with nonclassic 17OHD? How many cases of nonclassic 11OHD are we missing? What is the minimum enzyme activity that distinguishes unaffected and nonclassic CAH patients?
2. Modifier genes
How can siblings with the same CAH genotype show very different phenotypes and disease control? What genes control drug metabolism, response to androgens, and peripheral metabolism of C19-adrenal products? Why does salt wasting lessen in many but not all adults with classic 21OHD? Why do not all women with nonclassic 21OHD suffer from oligomenorrhea and subfertility?
3. Biomarkers for diagnosis and treatment
Can a panel of adrenal-derived 21-deoxysteroids be used to diagnose nonclassic 21OHD without cosyntropin stimulation and to eliminate false-positive newborn screens for classic 21OHD? Can similar panels be developed for 11OHD, 17OHD, POR, and 3βHSD2 deficiencies? What are the best steroids to use to monitor and titrate treatment?
4. Better treatments
What are the optimal glucocorticoid regimens? How do we position hydrocortisone as continuous subcutaneous infusion or modified-release oral preparations in treatment algorithms? Can we eliminate Cushingoid side effects and maintain control of androgen excess with the addition of abiraterone acetate, NBI77860, or other novel therapeutics to physiologic replacement doses of hydrocortisone?
5. Genetic diagnosis
Can cff-DNA in the maternal circulation be used to diagnose CAH and to determine sex chromosome complement for fetuses at risk for having CAH before 8 weeks’ gestation? What is the value of whole-exome sequencing in children with suspected CAH?
This incomplete list represents a substantial amount of work. To answer these questions and accomplish these goals will take research funding, collaboration among and creativity from investigators, participation of patients and their families, and partnerships with the pharmaceutical industry. With advances in technology, biochemistry, and genetics, the time is right to make major advances in CAH, which will have important implications for other related and often more common diseases. These patients need better options today, and we cannot take another 150 years to bring these advances to fruition.
Highlights.
Better steroid biomarkes might improve diagnosis and management of CAH
Improved treatments might allow good control of CAH with low glucocorticoid doses
The spectrum of mild or nonclassic CAH is known for most enzyme defects
Important modifier genes for CAH remain to be discovered
Acknowledgement
This work was supported by F32DK 103461 to A.F.T.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Delle Piane L, Rinaudo PF, Miller WL. 150 Years of Congenital Adrenal Hyperplasia: Translation and Commentary of De Crecchio’s Classic Paper from 1865. Endocrinology. 2015;156:1210–1217. doi: 10.1210/en.2014-1879. [DOI] [PubMed] [Google Scholar]
- 2.De Crecchio L. Sopra un caso di apparenze virili in una donna. Il Morgagni. 1865:151–189. [Google Scholar]
- 3.Speiser PW, White PC. Congenital adrenal hyperplasia. N Engl J Med. 2003;349:776–788. doi: 10.1056/NEJMra021561. [DOI] [PubMed] [Google Scholar]
- 4.Therrell BL, Jr., Berenbaum SA, Manter-Kapanke V, et al. Results of screening 1.9 million Texas newborns for 21-hydroxylase-deficient congenital adrenal hyperplasia. Pediatrics. 1998;101:583–590. doi: 10.1542/peds.101.4.583. [DOI] [PubMed] [Google Scholar]
- 5.Speiser PW, Dupont B, Rubinstein P, Piazza A, Kastelan A, New MI. High frequency of nonclassical steroid 21-hydroxylase deficiency. Am J Hum Genet. 1985;37:650–667. [PMC free article] [PubMed] [Google Scholar]
- 6.White PC, New MI, Dupont B. Congenital adrenal hyperplasia. (1) N Engl J Med. 1987;316:1519–1524. doi: 10.1056/NEJM198706113162406. [DOI] [PubMed] [Google Scholar]
- 7.White PC, Curnow KM, Pascoe L. Disorders of steroid 11β-hydroxylase isozymes. Endocr Rev. 1994;15:421–438. doi: 10.1210/edrv-15-4-421. [DOI] [PubMed] [Google Scholar]
- 8.Rosler A, Leiberman E, Cohen T. High frequency of congenital adrenal hyperplasia (classic 11 β-hydroxylase deficiency) among Jews from Morocco. Am J Med Genet. 1992;42:827–834. doi: 10.1002/ajmg.1320420617. [DOI] [PubMed] [Google Scholar]
- 9.Simard J, Durocher F, Mebarki F, et al. Molecular biology and genetics of the 3β-hydroxysteroid dehydrogenase/Δ5-Δ4 isomerase gene family. J Endocrinol. 1996;150(Suppl):S189–S207. [PubMed] [Google Scholar]
- 10.Rheaume E, Lachance Y, Zhao HF, et al. Structure and expression of a new complementary DNA encoding the almost exclusive 3β-hydroxysteroid dehydrogenase/Δ5-Δ4-isomerase in human adrenals and gonads. Mol Endocrinol. 1991;5:1147–1157. doi: 10.1210/mend-5-8-1147. [DOI] [PubMed] [Google Scholar]
- 11.Gingras S, Moriggl R, Groner B, Simard J. Induction of 3β-hydroxysteroid dehydrogenase/Δ5-Δ4 isomerase type 1 gene transcription in human breast cancer cell lines and in normal mammary epithelial cells by interleukin-4 and interleukin-13. Mol Endocrinol. 1999;13:66–81. doi: 10.1210/mend.13.1.0221. [DOI] [PubMed] [Google Scholar]
- 12.Singhellakis PN, Panidis D, Papadimas J, et al. Spontaneous sexual development and menarche in a female with 17α-hydroxylase deficiency. J Endocrinol Invest. 1986;9:177–183. doi: 10.1007/BF03348093. [DOI] [PubMed] [Google Scholar]
- 13.Katayama Y, Kado S, Wada S, et al. A case of 17α-hydroxylase deficiency with retained menstruation. Endocr J. 1994;41:213–218. doi: 10.1507/endocrj.41.213. [DOI] [PubMed] [Google Scholar]
- 14.New MI. Male pseudohermaphroditism due to 17α-hydroxylase deficiency. J Clin Invest. 1970;49:1930–1941. doi: 10.1172/JCI106412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Geller DH, Auchus RJ, Mendonca BB, Miller WL. The genetic and functional basis of isolated 17,20-lyase deficiency. Nat Genet. 1997;17:201–205. doi: 10.1038/ng1097-201. [DOI] [PubMed] [Google Scholar]
- 16.Sherbet DP, Tiosano D, Kwist KM, Hochberg Z, Auchus RJ. CYP17 mutation E305G causes isolated 17,20-lyase deficiency by selectively altering substrate binding. J Biol Chem. 2003;278:48563–48569. doi: 10.1074/jbc.M307586200. [DOI] [PubMed] [Google Scholar]
- 17.Hershkovitz E, Parvari R, Wudy SA, et al. Homozygous mutation G539R in the gene for P450 oxidoreductase in a family previously diagnosed as having 17,20-lyase deficiency. J Clin Endocrinol Metab. 2008;93:3584–3588. doi: 10.1210/jc.2008-0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kok RC, Timmerman MA, Wolffenbuttel KP, Drop SL, de Jong FH. Isolated 17,20-lyase deficiency due to the cytochrome b5 mutation W27X. J Clin Endocrinol Metab. 2010;95:994–999. doi: 10.1210/jc.2008-1745. [DOI] [PubMed] [Google Scholar]
- 19.Idkowiak J, Randell T, Dhir V, et al. A missense mutation in the human cytochrome b5 gene causes 46,XY disorder of sex development due to true isolated 17,20 lyase deficiency. J Clin Endocrinol Metab. 2012;97:E465–E475. doi: 10.1210/jc.2011-2413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fluck CE, Meyer-Boni M, Pandey AV, et al. Why boys will be boys: two pathways of fetal testicular androgen biosynthesis are needed for male sexual differentiation. Am J Hum Genet. 2011;89:201–218. doi: 10.1016/j.ajhg.2011.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bose HS, Sugawara T, Strauss JF, 3rd, Miller WL. International Congenital Lipoid Adrenal Hyperplasia C. The pathophysiology and genetics of congenital lipoid adrenal hyperplasia. N Engl J Med. 1996;335:1870–1878. doi: 10.1056/NEJM199612193352503. [DOI] [PubMed] [Google Scholar]
- 22.Tee MK, Abramsohn M, Loewenthal N, et al. Varied clinical presentations of seven patients with mutations in CYP11A1 encoding the cholesterol side-chain cleavage enzyme, P450scc. J Clin Endocrinol Metab. 2013;98:713–720. doi: 10.1210/jc.2012-2828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Baker BY, Lin L, Kim CJ, et al. Nonclassic congenital lipoid adrenal hyperplasia: a new disorder of the steroidogenic acute regulatory protein with very late presentation and normal male genitalia. J Clin Endocrinol Metab. 2006;91:4781–4785. doi: 10.1210/jc.2006-1565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rubtsov P, Karmanov M, Sverdlova P, Spirin P, Tiulpakov A. A novel homozygous mutation in CYP11A1 gene is associated with late-onset adrenal insufficiency and hypospadias in a 46,XY patient. J Clin Endocrinol Metab. 2009;94:936–939. doi: 10.1210/jc.2008-1118. [DOI] [PubMed] [Google Scholar]
- 25.Sahakitrungruang T, Tee MK, Blackett PR, Miller WL. Partial defect in the cholesterol side-chain cleavage enzyme P450scc (CYP11A1) resembling nonclassic congenital lipoid adrenal hyperplasia. J Clin Endocrinol Metab. 2011;96:792–798. doi: 10.1210/jc.2010-1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Parajes S, Kamrath C, Rose IT, et al. A novel entity of clinically isolated adrenal insufficiency caused by a partially inactivating mutation of the gene encoding for P450 side chain cleavage enzyme (CYP11A1) J Clin Endocrinol Metab. 2011;96:E1798–E1806. doi: 10.1210/jc.2011-1277. [DOI] [PubMed] [Google Scholar]
- 27.Huang N, Pandey AV, Agrawal V, et al. Diversity and function of mutations in P450 oxidoreductase in patients with Antley-Bixler syndrome and disordered steroidogenesis. Am J Hum Genet. 2005;76:729–749. doi: 10.1086/429417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fluck CE, Tajima T, Pandey AV, et al. Mutant P450 oxidoreductase causes disordered steroidogenesis with and without Antley-Bixler syndrome. Nat Genet. 2004;36:228–230. doi: 10.1038/ng1300. [DOI] [PubMed] [Google Scholar]
- 29.New MI, Lorenzen F, Lerner AJ, et al. Genotyping steroid 21-hydroxylase deficiency: hormonal reference data. J Clin Endocrinol Metab. 1983;57:320–326. doi: 10.1210/jcem-57-2-320. [DOI] [PubMed] [Google Scholar]
- 30.Pang S, Hotchkiss J, Drash AL, Levine LS, New MI. Microfilter paper method for 17α-hydroxyprogesterone radioimmunoassay: its application for rapid screening for congenital adrenal hyperplasia. J Clin Endocrinol Metab. 1977;45:1003–1008. doi: 10.1210/jcem-45-5-1003. [DOI] [PubMed] [Google Scholar]
- 31.Pang SY, Wallace MA, Hofman L, et al. Worldwide experience in newborn screening for classical congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Pediatrics. 1988;81:866–874. [PubMed] [Google Scholar]
- 32.Therrell BL. Newborn screening for congenital adrenal hyperplasia. Endocrinol Metab Clin North Am. 2001;30:15–30. doi: 10.1016/s0889-8529(08)70017-3. [DOI] [PubMed] [Google Scholar]
- 33.Popov A, Beloev I. [Early diagnosis and prevention of internal diseases] Vutr Boles. 1977;16:1–10. [PubMed] [Google Scholar]
- 34.Strnadova KA, Votava F, Lebl J, et al. Prevalence of congenital adrenal hyperplasia among sudden infant death in the Czech Republic and Austria. Eur J Pediatr. 2007;166:1–4. doi: 10.1007/s00431-006-0154-8. [DOI] [PubMed] [Google Scholar]
- 35.Grosse SD, Van Vliet G. How many deaths can be prevented by newborn screening for congenital adrenal hyperplasia? Horm Res. 2007;67:284–291. doi: 10.1159/000098400. [DOI] [PubMed] [Google Scholar]
- 36.Hird BE, Tetlow L, Tobi S, Patel L, Clayton PE. No evidence of an increase in early infant mortality from congenital adrenal hyperplasia in the absence of screening. Arch Dis Child. 2014;99:158–164. doi: 10.1136/archdischild-2013-304473. [DOI] [PubMed] [Google Scholar]
- 37.Cavarzere P, Samara-Boustani D, Flechtner I, et al. Transient hyper-17-hydroxyprogesteronemia: a clinical subgroup of patients diagnosed at neonatal screening for congenital adrenal hyperplasia. Eur J Endocrinol. 2009;161:285–292. doi: 10.1530/EJE-09-0145. [DOI] [PubMed] [Google Scholar]
- 38.Coulm B, Coste J, Tardy V, et al. Efficiency of neonatal screening for congenital adrenal hyperplasia due to 21-hydroxylase deficiency in children born in mainland France between 1996 and 2003. Arch Pediatr Adolesc Med. 2012;166:113–120. doi: 10.1001/archpediatrics.2011.774. [DOI] [PubMed] [Google Scholar]
- 39.Lee MM, Rajagopalan L, Berg GJ, Moshang T., Jr. Serum adrenal steroid concentrations in premature infants. J Clin Endocrinol Metab. 1989;69:1133–1136. doi: 10.1210/jcem-69-6-1133. [DOI] [PubMed] [Google Scholar]
- 40.Hingre RV, Gross SJ, Hingre KS, Mayes DM, Richman RA. Adrenal steroidogenesis in very low birth weight preterm infants. J Clin Endocrinol Metab. 1994;78:266–270. doi: 10.1210/jcem.78.2.8106610. [DOI] [PubMed] [Google Scholar]
- 41.Sarafoglou K, Banks K, Kyllo J, Pittock S, Thomas W. Cases of congenital adrenal hyperplasia missed by newborn screening in Minnesota. JAMA. 2012;307:2371–2374. doi: 10.1001/jama.2012.5281. [DOI] [PubMed] [Google Scholar]
- 42.Varness TS, Allen DB, Hoffman GL. Newborn screening for congenital adrenal hyperplasia has reduced sensitivity in girls. J Pediatr. 2005;147:493–498. doi: 10.1016/j.jpeds.2005.04.035. [DOI] [PubMed] [Google Scholar]
- 43.Steigert M, Schoenle EJ, Biason-Lauber A, Torresani T. High reliability of neonatal screening for congenital adrenal hyperplasia in Switzerland. J Clin Endocrinol Metab. 2002;87:4106–4110. doi: 10.1210/jc.2002-012093. [DOI] [PubMed] [Google Scholar]
- 44.Olgemoller B, Roscher AA, Liebl B, Fingerhut R. Screening for congenital adrenal hyperplasia: adjustment of 17-hydroxyprogesterone cut-off values to both age and birth weight markedly improves the predictive value. J Clin Endocrinol Metab. 2003;88:5790–5794. doi: 10.1210/jc.2002-021732. [DOI] [PubMed] [Google Scholar]
- 45.van der Kamp HJ, Oudshoorn CG, Elvers BH, et al. Cutoff levels of 17α-hydroxyprogesterone in neonatal screening for congenital adrenal hyperplasia should be based on gestational age rather than on birth weight. J Clin Endocrinol Metab. 2005;90:3904–3907. doi: 10.1210/jc.2004-2136. [DOI] [PubMed] [Google Scholar]
- 46.Chan CL, McFann K, Taylor L, Wright D, Zeitler PS, Barker JM. Congenital adrenal hyperplasia and the second newborn screen. J Pediatr. 2013;163:109–113. doi: 10.1016/j.jpeds.2013.01.002. e1. [DOI] [PubMed] [Google Scholar]
- 47.Speiser PW, Azziz R, Baskin LS, et al. Congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2010;95:4133–4160. doi: 10.1210/jc.2009-2631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mermejo LM, Elias LL, Marui S, Moreira AC, Mendonca BB, de Castro M. Refining hormonal diagnosis of type II 3β-hydroxysteroid dehydrogenase deficiency in patients with premature pubarche and hirsutism based on HSD3B2 genotyping. J Clin Endocrinol Metab. 2005;90:1287–1293. doi: 10.1210/jc.2004-1552. [DOI] [PubMed] [Google Scholar]
- 49.Schwarz E, Liu A, Randall H, et al. Use of steroid profiling by UPLC-MS/MS as a second tier test in newborn screening for congenital adrenal hyperplasia: the Utah experience. Pediatr Res. 2009;66:230–235. doi: 10.1203/PDR.0b013e3181aa3777. [DOI] [PubMed] [Google Scholar]
- 50.Janzen N, Peter M, Sander S, et al. Newborn screening for congenital adrenal hyperplasia: additional steroid profile using liquid chromatography-tandem mass spectrometry. J Clin Endocrinol Metab. 2007;92:2581–2589. doi: 10.1210/jc.2006-2890. [DOI] [PubMed] [Google Scholar]
- 51.Janzen N, Sander S, Terhardt M, Peter M, Sander J. Fast and direct quantification of adrenal steroids by tandem mass spectrometry in serum and dried blood spots. J Chromatogr B Analyt Technol Biomed Life Sci. 2008;861:117–122. doi: 10.1016/j.jchromb.2007.11.006. [DOI] [PubMed] [Google Scholar]
- 52.Costa-Barbosa FA, Tonetto-Fernandes VF, Carvalho VM, et al. Superior discriminating value of ACTH-stimulated serum 21-deoxycortisol in identifying heterozygote carriers for 21-hydroxylase deficiency. Clin Endocrinol (Oxf) 2010;73:700–706. doi: 10.1111/j.1365-2265.2010.03871.x. [DOI] [PubMed] [Google Scholar]
- 53.Turcu AF, Rege J, Chomic R, Liu J, Nishimoto HK, Else T, Moraitis AG, Palapattu GS, Rainey WE, Auchus RJ. J. Clin. Endocrinol. Metab. 2015;100:2283–2290. doi: 10.1210/jc.2015-1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Krone N, Hughes BA, Lavery GG, Stewart PM, Arlt W, Shackleton CH. Gas chromatography/mass spectrometry (GC/MS) remains a pre-eminent discovery tool in clinical steroid investigations even in the era of fast liquid chromatography tandem mass spectrometry (LC/MS/MS) J Steroid Biochem Mol Biol. 2010;121:496–504. doi: 10.1016/j.jsbmb.2010.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Carroll MC, Campbell RD, Porter RR. Mapping of steroid 21-hydroxylase genes adjacent to complement component C4 genes in HLA, the major histocompatibility complex in man. Proc Natl Acad Sci U S A. 1985;82:521–525. doi: 10.1073/pnas.82.2.521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.White PC, Grossberger D, Onufer BJ, et al. Two genes encoding steroid 21-hydroxylase are located near the genes encoding the fourth component of complement in man. Proc Natl Acad Sci U S A. 1985;82:1089–1093. doi: 10.1073/pnas.82.4.1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.White PC, New MI, Dupont B. Structure of human steroid 21-hydroxylase genes. Proc Natl Acad Sci U S A. 1986;83:5111–5115. doi: 10.1073/pnas.83.14.5111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.White PC, Speiser PW. Congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Endocr Rev. 2000;21:245–291. doi: 10.1210/edrv.21.3.0398. [DOI] [PubMed] [Google Scholar]
- 59.Mornet E, Dupont J, Vitek A, White PC. Characterization of two genes encoding human steroid 11β-hydroxylase (P-450(11) β) J Biol Chem. 1989;264:20961–20967. [PubMed] [Google Scholar]
- 60.White PC, Dupont J, New MI, Leiberman E, Hochberg Z, Rosler A. A mutation in CYP11B1 (Arg-448----His) associated with steroid 11β-hydroxylase deficiency in Jews of Moroccan origin. J Clin Invest. 1991;87:1664–1667. doi: 10.1172/JCI115182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Curnow KM, Slutsker L, Vitek J, et al. Mutations in the CYP11B1 gene causing congenital adrenal hyperplasia and hypertension cluster in exons 6, 7, and 8. Proc Natl Acad Sci U S A. 1993;90:4552–4556. doi: 10.1073/pnas.90.10.4552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Helmberg A, Ausserer B, Kofler R. Frame shift by insertion of 2 basepairs in codon 394 of CYP11B1 causes congenital adrenal hyperplasia due to steroid 11β-hydroxylase deficiency. J Clin Endocrinol Metab. 1992;75:1278–1281. doi: 10.1210/jcem.75.5.1430088. [DOI] [PubMed] [Google Scholar]
- 63.Naiki Y, Kawamoto T, Mitsuuchi Y, et al. A nonsense mutation (TGG [Trp116]-->TAG [Stop]) in CYP11B1 causes steroid 11β-hydroxylase deficiency. J Clin Endocrinol Metab. 1993;77:1677–1682. doi: 10.1210/jcem.77.6.7903314. [DOI] [PubMed] [Google Scholar]
- 64.Skinner CA, Rumsby G. Steroid 11β-hydroxylase deficiency caused by a five base pair duplication in the CYP11B1 gene. Hum Mol Genet. 1994;3:377–378. doi: 10.1093/hmg/3.2.377. [DOI] [PubMed] [Google Scholar]
- 65.Yang LX, Toda K, Miyahara K, et al. Classic steroid 11β-hydroxylase deficiency caused by a C-->G transversion in exon 7 of CYP11B1. Biochem Biophys Res Commun. 1995;216:723–728. doi: 10.1006/bbrc.1995.2681. [DOI] [PubMed] [Google Scholar]
- 66.Nakagawa Y, Yamada M, Ogawa H, Igarashi Y. Missense mutation in CYP11B1 (CGA[Arg-384]-->GGA[Gly]) causes steroid 11β-hydroxylase deficiency. Eur J Endocrinol. 1995;132:286–289. doi: 10.1530/eje.0.1320286. [DOI] [PubMed] [Google Scholar]
- 67.Geley S, Kapelari K, Johrer K, et al. CYP11B1 mutations causing congenital adrenal hyperplasia due to 11β-hydroxylase deficiency. J Clin Endocrinol Metab. 1996;81:2896–2901. doi: 10.1210/jcem.81.8.8768848. [DOI] [PubMed] [Google Scholar]
- 68.Merke DP, Tajima T, Chhabra A, et al. Novel CYP11B1 mutations in congenital adrenal hyperplasia due to steroid 11β-hydroxylase deficiency. J Clin Endocrinol Metab. 1998;83:270–273. doi: 10.1210/jcem.83.1.4513. [DOI] [PubMed] [Google Scholar]
- 69.Zhu YS, Cordero JJ, Can S, et al. Mutations in CYP11B1 gene: phenotype-genotype correlations. Am J Med Genet A. 2003;122A:193–200. doi: 10.1002/ajmg.a.20108. [DOI] [PubMed] [Google Scholar]
- 70.Krone N, Riepe FG, Gotze D, et al. Congenital adrenal hyperplasia due to 11-hydroxylase deficiency: functional characterization of two novel point mutations and a three-base pair deletion in the CYP11B1 gene. J Clin Endocrinol Metab. 2005;90:3724–3730. doi: 10.1210/jc.2005-0089. [DOI] [PubMed] [Google Scholar]
- 71.Krone N, Grischuk Y, Muller M, et al. Analyzing the functional and structural consequences of two point mutations (P94L and A368D) in the CYP11B1 gene causing congenital adrenal hyperplasia resulting from 11-hydroxylase deficiency. J Clin Endocrinol Metab. 2006;91:2682–2688. doi: 10.1210/jc.2006-0209. [DOI] [PubMed] [Google Scholar]
- 72.Polat S, Kulle A, Karaca Z, et al. Characterisation of three novel CYP11B1 mutations in classic and non-classic 11β-hydroxylase deficiency. Eur J Endocrinol. 2014;170:697–706. doi: 10.1530/EJE-13-0737. [DOI] [PubMed] [Google Scholar]
- 73.Joehrer K, Geley S, Strasser-Wozak EM, et al. CYP11B1 mutations causing non-classic adrenal hyperplasia due to 11β-hydroxylase deficiency. Hum Mol Genet. 1997;6:1829–1834. doi: 10.1093/hmg/6.11.1829. [DOI] [PubMed] [Google Scholar]
- 74.Reisch N, Hogler W, Parajes S, et al. A diagnosis not to be missed: nonclassic steroid 11β-hydroxylase deficiency presenting with premature adrenarche and hirsutism. J Clin Endocrinol Metab. 2013;98:E1620–E1625. doi: 10.1210/jc.2013-1306. [DOI] [PubMed] [Google Scholar]
- 75.Menabo S, Polat S, Baldazzi L, et al. Congenital adrenal hyperplasia due to 11β-hydroxylase deficiency: functional consequences of four CYP11B1 mutations. Eur J Hum Genet. 2014;22:610–616. doi: 10.1038/ejhg.2013.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Parajes S, Loidi L, Reisch N, et al. Functional consequences of seven novel mutations in the CYP11B1 gene: four mutations associated with nonclassic and three mutations causing classic 11β-hydroxylase deficiency. J Clin Endocrinol Metab. 2010;95:779–788. doi: 10.1210/jc.2009-0651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Simard J, Ricketts ML, Gingras S, Soucy P, Feltus FA, Melner MH. Molecular biology of the 3β-hydroxysteroid dehydrogenase/Δ5-Δ4 isomerase gene family. Endocr Rev. 2005;26:525–582. doi: 10.1210/er.2002-0050. [DOI] [PubMed] [Google Scholar]
- 78.McBride MW, McVie AJ, Burridge SM, et al. Cloning, expression, and physical mapping of the 3β-hydroxysteroid dehydrogenase gene cluster (HSD3BP1-HSD3BP5) in human. Genomics. 1999;61:277–284. doi: 10.1006/geno.1999.5459. [DOI] [PubMed] [Google Scholar]
- 79.Cara JF, Moshang T, Jr., Bongiovanni AM, Marx BS. Elevated 17-hydroxyprogesterone and testosterone in a newborn with 3β-hydroxysteroid dehydrogenase deficiency. N Engl J Med. 1985;313:618–621. doi: 10.1056/NEJM198509053131007. [DOI] [PubMed] [Google Scholar]
- 80.Zhang L, Mason JI, Naiki Y, et al. Characterization of two novel homozygous missense mutations involving codon 6 and 259 of type II 3β-hydroxysteroid dehydrogenase (3βHSD) gene causing, respectively, nonsalt-wasting and salt-wasting 3βHSD deficiency disorder. J Clin Endocrinol Metab. 2000;85:1678–1685. doi: 10.1210/jcem.85.4.6539. [DOI] [PubMed] [Google Scholar]
- 81.Rheaume E, Sanchez R, Simard J, et al. Molecular basis of congenital adrenal hyperplasia in two siblings with classical nonsalt-losing 3β-hydroxysteroid dehydrogenase deficiency. J Clin Endocrinol Metab. 1994;79:1012–1018. doi: 10.1210/jcem.79.4.7962268. [DOI] [PubMed] [Google Scholar]
- 82.Russell AJ, Wallace AM, Forest MG, Donaldson MD, Edwards CR, Sutcliffe RG. Mutation in the human gene for 3β-hydroxysteroid dehydrogenase type II leading to male pseudohermaphroditism without salt loss. J Mol Endocrinol. 1994;12:225–237. doi: 10.1677/jme.0.0120225. [DOI] [PubMed] [Google Scholar]
- 83.Sanchez R, Rheaume E, Laflamme N, Rosenfield RL, Labrie F, Simard J. Detection and functional characterization of the novel missense mutation Y254D in type II 3β-hydroxysteroid dehydrogenase (3β HSD) gene of a female patient with nonsalt-losing 3β HSD deficiency. J Clin Endocrinol Metab. 1994;78:561–567. doi: 10.1210/jcem.78.3.8126127. [DOI] [PubMed] [Google Scholar]
- 84.Mendonca BB, Russell AJ, Vasconcelos-Leite M, et al. Mutation in 3β-hydroxysteroid dehydrogenase type II associated with pseudohermaphroditism in males and premature pubarche or cryptic expression in females. J Mol Endocrinol. 1994;12:119–122. doi: 10.1677/jme.0.0120119. [DOI] [PubMed] [Google Scholar]
- 85.Matteson KJ, Picado-Leonard J, Chung BC, Mohandas TK, Miller WL. Assignment of the gene for adrenal P450c17 (steroid 17α-hydroxylase/17,20 lyase) to human chromosome 10. J Clin Endocrinol Metab. 1986;63:789–791. doi: 10.1210/jcem-63-3-789. [DOI] [PubMed] [Google Scholar]
- 86.Picado-Leonard J, Miller WL. Cloning and sequence of the human gene for P450c17 (steroid 17α-hydroxylase/17,20 lyase): similarity with the gene for P450c21. DNA. 1987;6:439–448. doi: 10.1089/dna.1987.6.439. [DOI] [PubMed] [Google Scholar]
- 87.Chung BC, Picado-Leonard J, Haniu M, et al. Cytochrome P450c17 (steroid 17α-hydroxylase/17,20 lyase): cloning of human adrenal and testis cDNAs indicates the same gene is expressed in both tissues. Proc Natl Acad Sci U S A. 1987;84:407–411. doi: 10.1073/pnas.84.2.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Costa-Santos M, Kater CE, Auchus RJ. Brazilian Congenital Adrenal Hyperplasia Multicenter Study G. Two prevalent CYP17 mutations and genotype-phenotype correlations in 24 Brazilian patients with 17-hydroxylase deficiency. J Clin Endocrinol Metab. 2004;89:49–60. doi: 10.1210/jc.2003-031021. [DOI] [PubMed] [Google Scholar]
- 89.Imai T, Yanase T, Waterman MR, Simpson ER, Pratt JJ. Canadian Mennonites and individuals residing in the Friesland region of The Netherlands share the same molecular basis of 17α-hydroxylase deficiency. Hum Genet. 1992;89:95–96. doi: 10.1007/BF00207050. [DOI] [PubMed] [Google Scholar]
- 90.Miura K, Yasuda K, Yanase T, et al. Mutation of cytochrome P-45017α gene (CYP17) in a Japanese patient previously reported as having glucocorticoid-responsive hyperaldosteronism: with a review of Japanese patients with mutations of CYP17. J Clin Endocrinol Metab. 1996;81:3797–3801. doi: 10.1210/jcem.81.10.8855840. [DOI] [PubMed] [Google Scholar]
- 91.Fardella CE, Zhang LH, Mahachoklertwattana P, Lin D, Miller WL. Deletion of amino acids Asp487-Ser488-Phe489 in human cytochrome P450c17 causes severe 17α-hydroxylase deficiency. J Clin Endocrinol Metab. 1993;77:489–493. doi: 10.1210/jcem.77.2.8345056. [DOI] [PubMed] [Google Scholar]
- 92.Auchus RJ, Miller WL. Molecular modeling of human P450c17 (17α-hydroxylase/17,20-lyase): insights into reaction mechanisms and effects of mutations. Mol Endocrinol. 1999;13:1169–1182. doi: 10.1210/mend.13.7.0326. [DOI] [PubMed] [Google Scholar]
- 93.Van Den Akker EL, Koper JW, Boehmer AL, et al. Differential inhibition of 17α-hydroxylase and 17,20-lyase activities by three novel missense CYP17 mutations identified in patients with P450c17 deficiency. J Clin Endocrinol Metab. 2002;87:5714–5721. doi: 10.1210/jc.2001-011880. [DOI] [PubMed] [Google Scholar]
- 94.Bidet M, Bellanne-Chantelot C, Galand-Portier MB, et al. Clinical and molecular characterization of a cohort of 161 unrelated women with nonclassical congenital adrenal hyperplasia due to 21-hydroxylase deficiency and 330 family members. J Clin Endocrinol Metab. 2009;94:1570–1578. doi: 10.1210/jc.2008-1582. [DOI] [PubMed] [Google Scholar]
- 95.Finkielstain GP, Chen W, Mehta SP, et al. Comprehensive genetic analysis of 182 unrelated families with congenital adrenal hyperplasia due to 21-hydroxylase deficiency. J Clin Endocrinol Metab. 2011;96:E161–E172. doi: 10.1210/jc.2010-0319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Moran C, Azziz R, Weintrob N, et al. Reproductive outcome of women with 21-hydroxylase-deficient nonclassic adrenal hyperplasia. J Clin Endocrinol Metab. 2006;91:3451–3456. doi: 10.1210/jc.2006-0062. [DOI] [PubMed] [Google Scholar]
- 97.Nimkarn S, New MI. Prenatal diagnosis and treatment of congenital adrenal hyperplasia owing to 21-hydroxylase deficiency. Nat Clin Pract Endocrinol Metab. 2007;3:405–413. doi: 10.1038/ncpendmet0481. [DOI] [PubMed] [Google Scholar]
- 98.Sundberg K, Bang J, Smidt-Jensen S, et al. Randomised study of risk of fetal loss related to early amniocentesis versus chorionic villus sampling. Lancet. 1997;350:697–703. doi: 10.1016/S0140-6736(97)02449-5. [DOI] [PubMed] [Google Scholar]
- 99.Mujezinovic F, Alfirevic Z. Procedure-related complications of amniocentesis and chorionic villous sampling: a systematic review. Obstet Gynecol. 2007;110:687–694. doi: 10.1097/01.AOG.0000278820.54029.e3. [DOI] [PubMed] [Google Scholar]
- 100.Kollmann M, Haeusler M, Haas J, Csapo B, Lang U, Klaritsch P. Procedure-related complications after genetic amniocentesis and chorionic villus sampling. Ultraschall Med. 2013;34:345–348. doi: 10.1055/s-0032-1312939. [DOI] [PubMed] [Google Scholar]
- 101.Witchel SF, Miller WL. Prenatal treatment of congenital adrenal hyperplasia-not standard of care. J Genet Couns. 2012;21:615–624. doi: 10.1007/s10897-012-9508-8. [DOI] [PubMed] [Google Scholar]
- 102.New MI, Carlson A, Obeid J, et al. Prenatal diagnosis for congenital adrenal hyperplasia in 532 pregnancies. J Clin Endocrinol Metab. 2001;86:5651–5657. doi: 10.1210/jcem.86.12.8072. [DOI] [PubMed] [Google Scholar]
- 103.Hirvikoski T, Nordenstrom A, Lindholm T, et al. Cognitive functions in children at risk for congenital adrenal hyperplasia treated prenatally with dexamethasone. J Clin Endocrinol Metab. 2007;92:542–548. doi: 10.1210/jc.2006-1340. [DOI] [PubMed] [Google Scholar]
- 104.Davis EP, Sandman CA, Buss C, Wing DA, Head K. Fetal glucocorticoid exposure is associated with preadolescent brain development. Biol Psychiatry. 2013;74:647–655. doi: 10.1016/j.biopsych.2013.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Miller WL, Witchel SF. Prenatal treatment of congenital adrenal hyperplasia: risks outweigh benefits. Am J Obstet Gynecol. 2013;208:354–359. doi: 10.1016/j.ajog.2012.10.885. [DOI] [PubMed] [Google Scholar]
- 106.Hirvikoski T, Nordenstrom A, Wedell A, Ritzen M, Lajic S. Prenatal dexamethasone treatment of children at risk for congenital adrenal hyperplasia: the Swedish experience and standpoint. J Clin Endocrinol Metab. 2012;97:1881–1883. doi: 10.1210/jc.2012-1222. [DOI] [PubMed] [Google Scholar]
- 107.Lo YM, Corbetta N, Chamberlain PF, et al. Presence of fetal DNA in maternal plasma and serum. Lancet. 1997;350:485–487. doi: 10.1016/S0140-6736(97)02174-0. [DOI] [PubMed] [Google Scholar]
- 108.Rijnders RJ, van der Schoot CE, Bossers B, de Vroede MA, Christiaens GC. Fetal sex determination from maternal plasma in pregnancies at risk for congenital adrenal hyperplasia. Obstet Gynecol. 2001;98:374–378. doi: 10.1016/s0029-7844(01)01480-6. [DOI] [PubMed] [Google Scholar]
- 109.New MI, Tong YK, Yuen T, et al. Noninvasive prenatal diagnosis of congenital adrenal hyperplasia using cell-free fetal DNA in maternal plasma. J Clin Endocrinol Metab. 2014;99:E1022–E1030. doi: 10.1210/jc.2014-1118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lekarev O, Tafuri K, Lane AH, et al. Erroneous prenatal diagnosis of congenital adrenal hyperplasia owing to a duplication of the CYP21A2 gene. J Perinatol. 2013;33:76–78. doi: 10.1038/jp.2012.5. [DOI] [PubMed] [Google Scholar]
- 111.Lobo RA, Goebelsmann U. Evidence for reduced 3β-ol-hydroxysteroid dehydrogenase activity in some hirsute women thought to have polycystic ovary syndrome. J Clin Endocrinol Metab. 1981;53:394–400. doi: 10.1210/jcem-53-2-394. [DOI] [PubMed] [Google Scholar]
- 112.Pang SY, Lerner AJ, Stoner E, et al. Late-onset adrenal steroid 3β-hydroxysteroid dehydrogenase deficiency. I. A cause of hirsutism in pubertal and postpubertal women. J Clin Endocrinol Metab. 1985;60:428–439. doi: 10.1210/jcem-60-3-428. [DOI] [PubMed] [Google Scholar]
- 113.Siegel SF, Finegold DN, Lanes R, Lee PA. ACTH stimulation tests and plasma dehydroepiandrosterone sulfate levels in women with hirsutism. N Engl J Med. 1990;323:849–854. doi: 10.1056/NEJM199009273231301. [DOI] [PubMed] [Google Scholar]
- 114.Eldar-Geva T, Hurwitz A, Vecsei P, Palti Z, Milwidsky A, Rosler A. Secondary biosynthetic defects in women with late-onset congenital adrenal hyperplasia. N Engl J Med. 1990;323:855–863. doi: 10.1056/NEJM199009273231302. [DOI] [PubMed] [Google Scholar]
- 115.Lutfallah C, Wang W, Mason JI, et al. Newly proposed hormonal criteria via genotypic proof for type II 3β-hydroxysteroid dehydrogenase deficiency. J Clin Endocrinol Metab. 2002;87:2611–2622. doi: 10.1210/jcem.87.6.8615. [DOI] [PubMed] [Google Scholar]
- 116.Arlt W, Willis DS, Wild SH, et al. Health status of adults with congenital adrenal hyperplasia: a cohort study of 203 patients. J Clin Endocrinol Metab. 2010;95:5110–5121. doi: 10.1210/jc.2010-0917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Finkielstain GP, Kim MS, Sinaii N, et al. Clinical characteristics of a cohort of 244 patients with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2012;97:4429–4438. doi: 10.1210/jc.2012-2102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Mallappa A, Sinaii N, Kumar P, et al. A phase 2 study of chronocort, a modified-release formulation of hydrocortisone, in the treatment of adults with classic congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2015;100:1137–1145. doi: 10.1210/jc.2014-3809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Johannsson G, Bergthorsdottir R, Nilsson AG, Lennernas H, Hedner T, Skrtic S. Improving glucocorticoid replacement therapy using a novel modified-release hydrocortisone tablet: a pharmacokinetic study. Eur J Endocrinol. 2009;161:119–130. doi: 10.1530/EJE-09-0170. [DOI] [PubMed] [Google Scholar]
- 120.Bryan SM, Honour JW, Hindmarsh PC. Management of altered hydrocortisone pharmacokinetics in a boy with congenital adrenal hyperplasia using a continuous subcutaneous hydrocortisone infusion. J Clin Endocrinol Metab. 2009;94:3477–3480. doi: 10.1210/jc.2009-0630. [DOI] [PubMed] [Google Scholar]
- 121.Hindmarsh PC. The child with difficult to control Congenital Adrenal Hyperplasia: is there a place for continuous subcutaneous hydrocortisone therapy. Clin Endocrinol (Oxf) 2014;81:15–18. doi: 10.1111/cen.12453. [DOI] [PubMed] [Google Scholar]
- 122.Auchus RJ, Turcu AF, Spencer-Segal JL, et al. A Pharmacokinetic and Biomarker Study of the Corticotropin-Releasing Factor Receptor Antagonist NBI-77860 in Adult Females with Classic 21-Hydroxylase Deficiency. Endocr. Rev. 2015;36:OR06-6. [Google Scholar]
- 123.Laue L, Merke DP, Jones JV, Barnes KM, Hill S, Cutler GB., Jr. A preliminary study of flutamide, testolactone, and reduced hydrocortisone dose in the treatment of congenital adrenal hyperplasia. J Clin Endocrinol Metab. 1996;81:3535–3539. doi: 10.1210/jcem.81.10.8855797. [DOI] [PubMed] [Google Scholar]
- 124.Merke DP, Keil MF, Jones JV, Fields J, Hill S, Cutler GB., Jr. Flutamide, testolactone, and reduced hydrocortisone dose maintain normal growth velocity and bone maturation despite elevated androgen levels in children with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2000;85:1114–1120. doi: 10.1210/jcem.85.3.6462. [DOI] [PubMed] [Google Scholar]
- 125.Lin-Su K, Vogiatzi MG, Marshall I, et al. Treatment with growth hormone and luteinizing hormone releasing hormone analog improves final adult height in children with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2005;90:3318–3325. doi: 10.1210/jc.2004-2128. [DOI] [PubMed] [Google Scholar]
- 126.Auchus RJ, Buschur EO, Chang AY, et al. Abiraterone acetate to lower androgens in women with classic 21-hydroxylase deficiency. J Clin Endocrinol Metab. 2014;99:2763–2770. doi: 10.1210/jc.2014-1258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kamrath C, Hochberg Z, Hartmann MF, Remer T, Wudy SA. Increased activation of the alternative “backdoor” pathway in patients with 21-hydroxylase deficiency: evidence from urinary steroid hormone analysis. J Clin Endocrinol Metab. 2012;97:E367–E375. doi: 10.1210/jc.2011-1997. [DOI] [PubMed] [Google Scholar]
- 128.Ferreira F, Martins JM, do Vale S, Esteves R, Nunes G, Carmo I. Rare and severe complications of congenital adrenal hyperplasia due to 21-hydroxylase deficiency: a case report. J Med Case Rep. 2013;7:39. doi: 10.1186/1752-1947-7-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Nermoen I, Folling I, Vegge K, et al. Two adults with adrenal myelolipoma and 21-hydroxylase deficiency. Case Rep Med. 2009;2009:916891. doi: 10.1155/2009/916891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Mermejo LM, Elias Junior J, Saggioro FP, et al. Giant adrenal myelolipoma associated with 21-hydroxylase deficiency: unusual association mimicking an androgen-secreting adrenocortical carcinoma. Arq Bras Endocrinol Metabol. 2010;54:419–424. doi: 10.1590/s0004-27302010000400012. [DOI] [PubMed] [Google Scholar]
- 131.Speiser PW, Dupont J, Zhu D, et al. Disease expression and molecular genotype in congenital adrenal hyperplasia due to 21-hydroxylase deficiency. J Clin Invest. 1992;90:584–595. doi: 10.1172/JCI115897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Krone N, Braun A, Roscher AA, Knorr D, Schwarz HP. Predicting phenotype in steroid 21-hydroxylase deficiency? Comprehensive genotyping in 155 unrelated, well defined patients from southern Germany. J Clin Endocrinol Metab. 2000;85:1059–1065. doi: 10.1210/jcem.85.3.6441. [DOI] [PubMed] [Google Scholar]
- 133.Wilson JD, Auchus RJ, Leihy MW, et al. 5α-androstane-3α,17β-diol is formed in tammar wallaby pouch young testes by a pathway involving 5αa-pregnane-3α,17α-diol-20-one as a key intermediate. Endocrinology. 2003;144:575–580. doi: 10.1210/en.2002-220721. [DOI] [PubMed] [Google Scholar]
- 134.Auchus RJ. The backdoor pathway to dihydrotestosterone. Trends Endocrinol Metab. 2004;15:432–438. doi: 10.1016/j.tem.2004.09.004. [DOI] [PubMed] [Google Scholar]
- 135.Homma K, Hasegawa T, Nagai T, et al. Urine steroid hormone profile analysis in cytochrome P450 oxidoreductase deficiency: implication for the backdoor pathway to dihydrotestosterone. J Clin Endocrinol Metab. 2006;91:2643–2649. doi: 10.1210/jc.2005-2460. [DOI] [PubMed] [Google Scholar]
- 136.Rege J, Nakamura Y, Satoh F, et al. Liquid chromatography-tandem mass spectrometry analysis of human adrenal vein 19-carbon steroids before and after ACTH stimulation. J Clin Endocrinol Metab. 2013;98:1182–1188. doi: 10.1210/jc.2012-2912. [DOI] [PMC free article] [PubMed] [Google Scholar]