Abstract
Backgrounds/Aims
International treatment guidelines for branch duct intraductal papillary mucinous neoplasm (BD-IPMN) of the pancreas have been proposed, for features associated with malignancy and invasiveness. We investigated the clinicopathological characteristics that are predictive of malignancy or invasiveness and disease recurrence.
Methods
A review of 324 patients with resected and pathologically confirmed BD-IPMN, between March 1997 and December 2013, was conducted.
Results
There were 144 (44.4%) low grade dysplasia (LGD), 138 (42.6%) intermediate grade dysplasia (IMGD), 17 (5.3%) high grade dysplasia (HGD), and 25 (7.7%) invasive carcinoma (invIPMC) cases. The 5-year survival rates were 98.1% for LGD, 95.3% for IMGD, 100% for HGD, and 71.8% for invIPMC. Through a univariate analysis, the male sex was associated with malignancy, and CA19-9 was related to both malignant and invasive IPMN. The high risk or worrisome features of the international guidelines were associated with both malignant and invasive IPMN: the total bilirubin of the head/uncinate lesion, tumor size, mural nodule, and the size of the main pancreatic duct (MPD). Through a multivariate analysis, the male sex, elevated CA19-9, mural nodule, and dilated MPD diameter were independently correlated with the malignant IPMN. The elevated CA19-9 and dilated MPD diameter were also correlated with invasive carcinoma. The patient age and the initial pathological diagnosis were strongly associated with disease recurrence following surgical resection.
Conclusions
The high risk or worrisome features in the current treatment guidelines for BD-IPMN are confined to the morphological characteristics of the disease. Patient factors and biological features should also be considered in order to develop optimal therapeutic or surveillance strategies.
Keywords: Intraductal papillary mucinous neoplasm, Branch duct, Pancreas, Malignancy
INTRODUCTION
Accompanying the development of novel diagnostic instruments, such as endoscopic ultrasonography or magnetic resonance imaging, asymptomatic cystic tumors of the pancreas have been identified and diagnosed with increasing frequency.1,2,3
Intraductal papillary mucinous neoplasm (IPMN) of the pancreas involves the main duct (MD), and/or side branch ducts (BD), and can be classified histopathologically as one of the three following types: main duct IPMN (MD-IPMN), branch duct IPMN (BD-IPMN), and a mixed duct type that is clinicopathologically included in the category of MD-IPMN. Various authors have reported that MD-IPMN has a higher potential of causing malignant or invasive disease, compared to BD-IPMN, and any invasive carcinoma arising in MD-IPMN is more aggressive. In MD-IPMN versus BD-IPMN, their mean frequencies of malignancy are 61.6% vs. 25.5% and those of invasiveness are 43.1% vs. 17.7%, respectively.1,2,3,4,5,6,7,8,9,10,11,12,13 Classifying these morphological subtypes of ductal involvement is important for deciding upon a treatment strategy.
International consensus guidelines for the management of IPMN were established in 2006, and revised in 2012.12,13 In the 2012 guidelines, surgical resection for MD-IPMN was strongly recommended for all surgically fit patients, with the exception of cases having a main pancreatic duct (MPD) dilation of 5-9 mm; in this instance, the treatment strategies for BD-IPMN are more complicated, and it is these cases for which the concepts of 'high-risk stigmata of malignancy' and 'worrisome features' were adopted. 'High-risk stigmata of malignancy', in BD-IPMN, includes obstructive jaundice in a patient with a cystic lesion of the head of the pancreas, an enhancing solid component within the cyst, and an MPD size of ≥10 mm. 'Worrisome features' include a cyst size of ≥3 cm, thickened/enhancing cyst walls, an MPD size of 5-9 mm, a non-enhancing mural nodule, and an abrupt change in the caliber of the pancreatic duct with distal pancreatic atrophy.13
In this study, we retrospectively reviewed the pathologically confirmed BD-IPMN in patients who underwent surgical resection. The clinicopathological characteristics of BD-IPMN were investigated, by associating the malignant and invasive tendencies with the levels of the total bilirubin, the MPD diameter, the tumor size, and the presence of mural nodules that were classified as 'high-risk stigmata' and/or 'worrisome features,' and we assessed the correlation of these parameters with patient survival and recurrence patterns.
MATERIALS AND METHODS
Among the consecutive patients who underwent pancreatic resection at the Asan Medical Center, between March 1997 and December 2013, 699 patients were pathologically diagnosed as IPMN. The two categories of BD- or MD-mixed type IPMN, and the four pathologic degrees of low- (LGD), intermediate- (IMGD), and high-grade dysplasia (HGD), as well as invasive intraductal papillary mucinous carcinoma (IPMC/invIPMC), were described in the pathological reports. The degree of dysplasia was classified according to the WHO classification, 4th edition,14 and the IPMNs were divided into both non-invasive (LGD, IMGD, and HGD) and invIPMC groups,9 and benign (LGD and IMGD) and malignant (HGD and invIPMC) groups. Among these 699 patients, 337 (48.2%) were proven to be BD-IPMNs, which were defined as being IPMN that did not microscopically involve the MPD. In this study, 324 patients with BD-IPMN were included, and their medical records were reviewed retrospectively; 13 cases were lost during follow-up.
To obtain accurate diagnoses, all patients preoperatively underwent computed tomography (CT) scans, and most also underwent at least one of the following procedures: magnetic resonance cholangiopancreatography (MRCP), endoscopic ultrasonography (EUS), or endoscopic retrograde cholangiopancreatography (ERCP). Although we confirmed and enrolled patients who had pathologically confirmed BD-IPMN, other data (including tumor location, tumor size, the presence of mural nodules, and the MPD diameter) were collected from preoperative imaging studies to investigate the significance of the features of the disease at the time of diagnosis.
At our institution, the surveillance intervals are every 3-6 months for invasive carcinoma and 6-12 months for noninvasive IPMN. All patients underwent CT scans, and some also underwent transcutaneous ultrasonography at each visit. Tumor recurrence and patient survival data were obtained from patient medical records.
The chi-square test or Fisher's exact test were used to compare categorical variables, and Student's t-test was used to compare continuous variables in univariate analyses. An actuarial survival analysis, and comparisons, were performed using the Kaplan-Meier method and the log-rank test. To identify the independent predictive factors, a logistic regression model was employed. A p-value <0.05 was considered to indicate a statistically significant difference. All statistical analyses were performed using SPSS software (version 18; SPSS, Inc, Chicago, IL, USA).
RESULTS
Demographic features and univariate analysis associated with malignant and/or invasive IPMN
Among the 324 patients with BD-IPMN, there were LGD in 144 (44.4%), IMGD in 138 (42.6%), HGD in 17 (5.3%), and invIPMC in 25 (7.7%). Additionally, 282 patients had benign IPMN (LGD and IMGD), and 42 had malignant IPMN (HGD and invIPMC). They could also be categorized as non-invasive IPMN (LGD, IMGD, and HGD) in 299, and invasive IPMC in 25 cases (Table 1).
Table 1. Demographic and pathologic characteristics of all patients who underwent surgical resection for branch duct type intraductal papillary mucinous neoplasm of the pancreas (n=324).
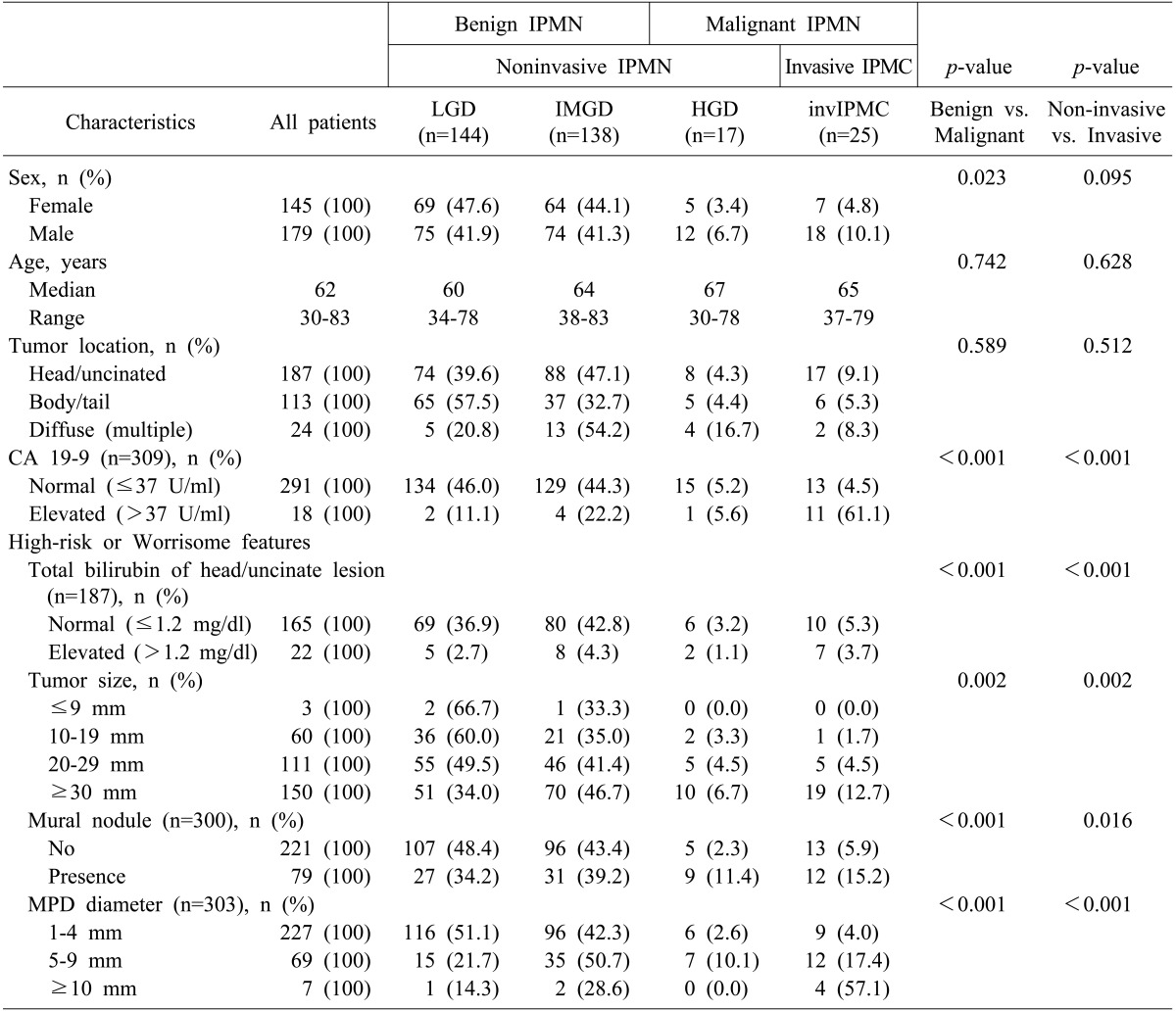
IPMN, intraductal papillary mucinous neoplasm; IPMC, intraductal papillary mucinous carcinoma; LGD, low grade dysplasia; IMGD, intermediate grade dysplasia; HGD, high grade dysplasia; MPD, main pancreatic duct
Table 1 also shows the univariate analyses that detected the correlations between the clinical characteristics and the malignant and invasive diseases. Male patients were more likely to have malignant IPMN (p=0.023), but there was no significant difference in the non-invasive and invasive subgroups according to the sex (p=0.095). The median ages were higher in HGD and invasive carcinoma (67 and 65, respectively) than in benign IPMN, although this difference was not statistically significant. The location of the tumor was not associated with malignancy or invasiveness (p=0.589 and p=0.512, respectively). Elevated levels of CA19-9 (>37 U/ml) were associated with both malignant and invasive disease (for both, p<0.001). All four factors that were included in the high-risk stigmata and/or worrisome features of the 2012 international consensus were significantly correlated with both malignant and invasive diseases. These factors included the total bilirubin of the head/uncinate process lesion (for both, p<0.001), the tumor size (for both, p=0.002), the presence of a mural nodule (p<0.001 or p=0.016, respectively), and the MPD diameter (for both, p<0.001).
Patient survival
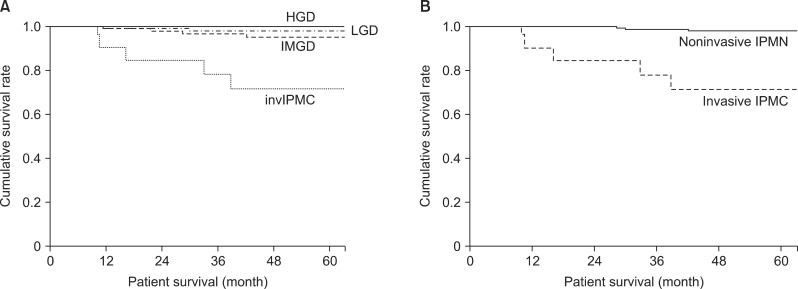
Patient survival curves are shown in Fig. 1A and 1B. The 5-year survival rates were 98.1% in LGD, 95.3% in IMGD, 100% in HGD, and 71.8% in invIPMC cases (Fig. 1A). Although HGD was included in malignant IPMN, the three survival curves for LGD, IMGD, and HGD were not significantly different. We did detect a significant difference between noninvasive IPMN and invasive IPMC (p<0.001; Fig. 1B).
Fig. 1. Survival curve analysis. (A) The overall survival of patients with each type of dysplasia (low grade dysplasia [LGD], intermediate grade dysplasia [IMGD], high grade dysplasia [HGD] and invasive carcinoma [invIPMC]). The 5-year survival rate was 98.1% for LGD, 95.3% for IMGD, 100% for HGD, and 71.8% for invIPMC cases. (B) The overall survival of patients with non-invasive or invasive IPMN of the pancreas; the 5-year survival rate was 98.2% and 71.8%, respectively (p<0.001).
Multivariate analysis of factors associated with malignant or invasive IPMN
Among the factors associated with malignant IPMN described in Table 1, we identified four statistically significant factors, via a multivariate analysis (Table 2), that included the following: male sex (odds ratio: OR 2.928), the presence of a mural nodule (OR 3.093), the dilated MPD diameter (OR 7.158 in 5-9 mm and 20.872 in more than 10 mm), and an elevated level of CA19-9 (OR 29.019). The Multivariate analysis identified two factors as being predictive of invasive carcinoma (Table 2) that included the following: a dilated MPD diameter (OR 6.512 in 5-9 mm and 32.674 in more than 10 mm) and an elevated level of CA19-9 (OR 36.774).
Table 2. Multivariate analysis of factors predictive of malignant or invasive IPMN.
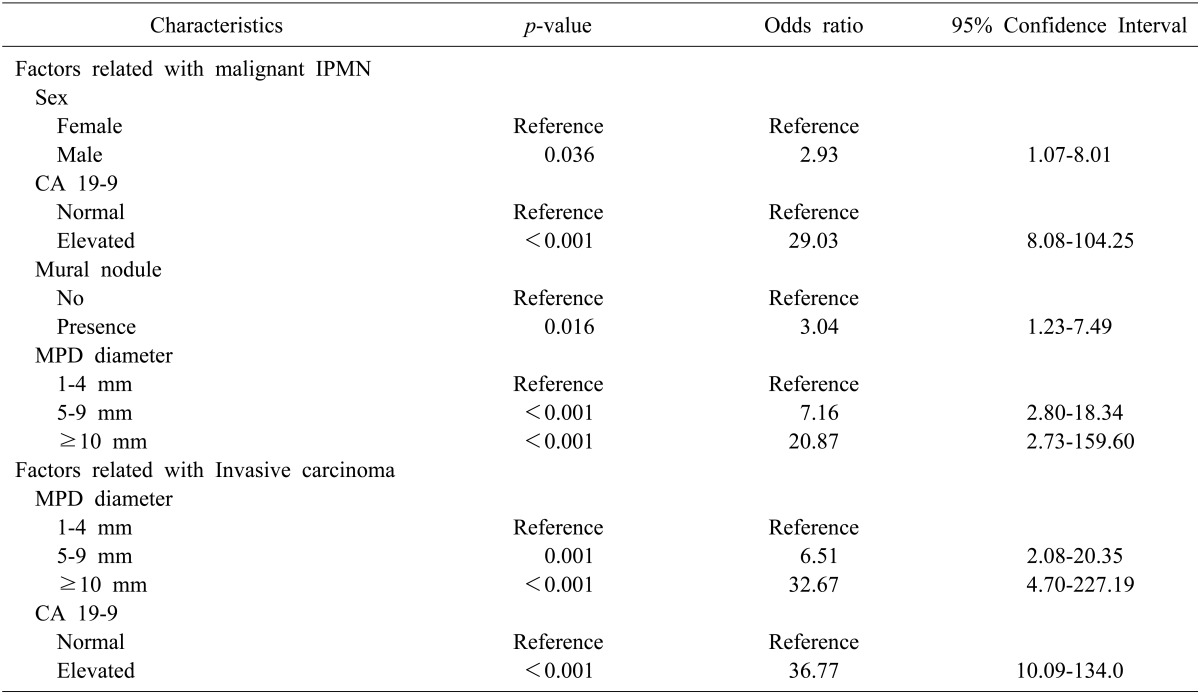
IPMN, intraductal papillary mucinous neoplasm; MPD, main pancreatic duct
Disease recurrence following surgical resection
There were 16 patients who experienced disease recurrence following surgical resection; their clinicopathological characteristics are described in Table 3. The affected patients included 9 invasive carcinoma, 4 IMGD, and 3 LGD cases at the initial diagnosis. Of the 7 patients with initially noninvasive IPMN, 5 had recurrences or a progression of disease in the remnant pancreas (No. 7, 8, 12, 14, and 16; Table 3). 2 of the initially noninvasive IPMN patients experienced carcinomatous recurrences (peritoneal seeding and liver metastasis) at 22.5 and 24.9 months following surgical resection, and died as a consequence of disease progression (No. 9 and 11; Table 3).
Table 3. Characteristics of the patients who had recurrent disease following surgical resection.
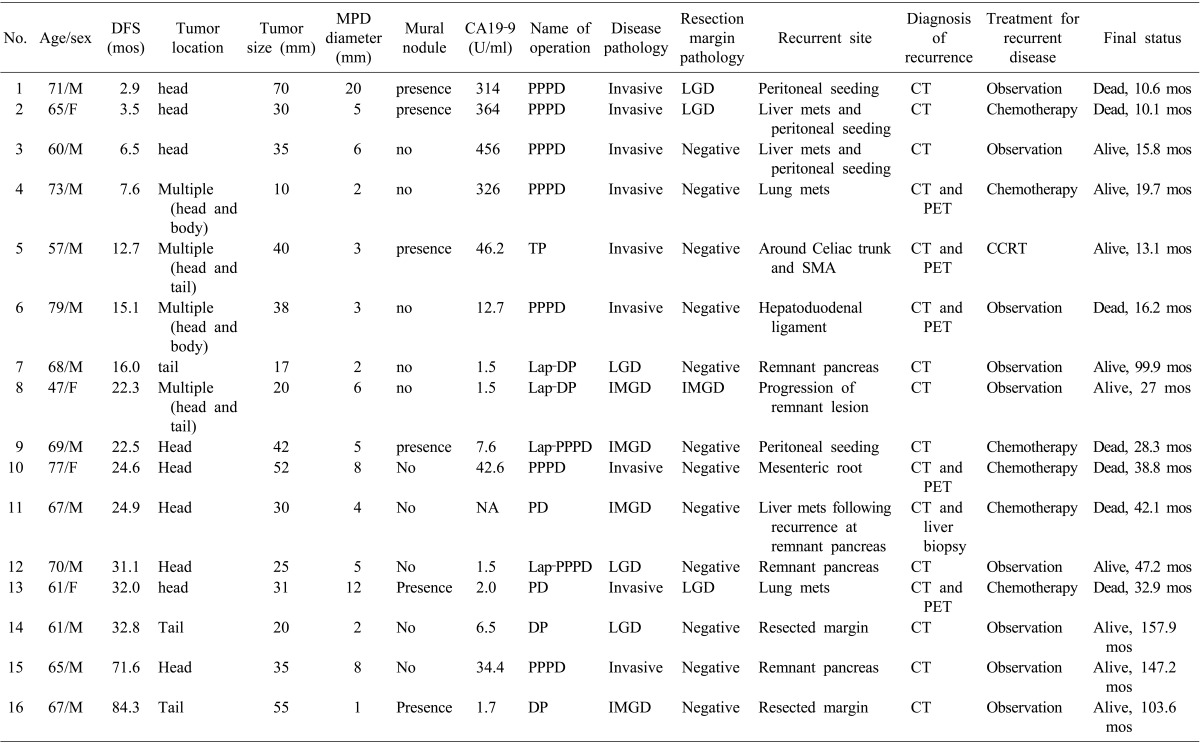
DFS, disease-free survival; MPD, main pancreatic duct; PPPD, pylorus-preserving pancreaticoduodenectomy; NA, not available; LGD, low grade dysplasia; TP, total pancreatectomy; SMA, superior mesenteric artery; CCRT, concurrent chemo-radiotherapy; Lap-DP, laparoscopic distal pancreatectomy; IMGD, intermediate grade dysplasia; Lap-PPPD, laparoscopic pylorus-preserving pancreaticoduodenectomy; PD, pancreaticoduodenectomy; DP, distal pancreatectomy; mets, metastasis; CT, computed tomography; PET, positron emission tomography
Old age, an elevated level of CA19-9 (>37 U/ml), a dilated MPD diameter (≥10 mm), and invasive pathology were associated with disease recurrence via a univariate analysis (p=0.036, p<0.001, p=0.006, and p<0.001, respectively; Table 4). According to the multivariate analysis, old age (OR 1.092) and invasive IPMC (OR 32.840) patients were independently significant, in regard to disease recurrence following surgical resection.
Table 4. Univariate and multivariate analyses for disease recurrence in branch duct IPMN following surgical resection.
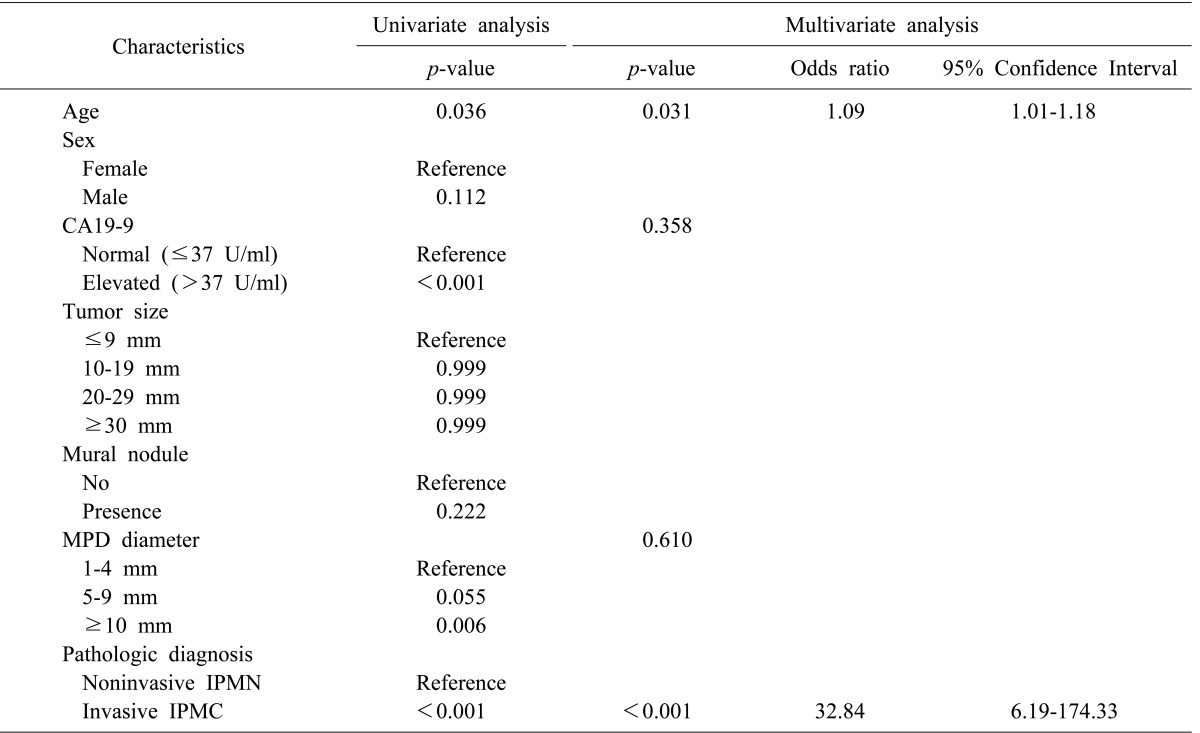
MPD, main pancreatic duct; IPMN, intraductal papillary mucinous neoplasm; IPMC, intraductal papillary mucinous carcinoma
DISCUSSION
IPMN is a disease entity that has a wide range of histological subtypes, and a natural history that follows an adenoma-to-carcinoma sequence. Knowledge of the natural history and disease traits of each subtype of IPMN is important for devising treatment strategies that avail patients of optimal treatment opportunities. International consensus guidelines for the management of IPMN were generated to assist in devising treatment strategies, by the working group of the International Association of Pancreatology. The 2012 guidelines adopted new concepts, 'high-risk stigmata of malignancy' and 'worrisome features,' so we stratified patients into these two categories.13 The recommendation of these guidelines was to let the stratified patients follow different therapeutic flows. In this retrospective single-institutional study, we investigated preoperative clinical predictors of malignant and invasive diseases of BD-IPMN of the pancreas, and validated the flows recommended by the 2012 international consensus guidelines. To the best of our knowledge, this is the largest series- to date- of patients with BD-IPMNs that were resected and confirmed pathologically.
The prevalence of malignant IPMN was reported to range from 10% to 32% in patients with BD-IPMN.5,10,15,16 In the series presented here, malignant IPMN (HGD and invIPMC) was 13.0%, and invasive carcinoma (invIPMC) was 7.7% of the entire resected BD-IPMN cohort. Therefore, the proportion of malignant or invasive diseases of BD-IPMN would be reduced if non-resected non-invasive BD-IPMNs under observation had been included.
Our survival analysis showed that the 5-year survival rate of non-invasive IPMN was 98.2%. Although HGD was included in the malignant category, its survival curve resembled that of benign IPMNs. The postoperative survival rate of HGD of BD-IPMN was already verified by a previous study.2 However, Winner et al.17 showed that carcinoma in situ (HGD) of IPMN showed a higher recurrence rate after a postoperative period of 5 years. Tamura et al also demonstrated that HGD of MD-IPMN showed a high recurrence rate and poor survival, when long-term surveillance of more than 5 years was carried out.11 Therefore, although HGD shows favorable survival outcomes for the initial 5 years after surgical resection, it should be followed as a malignant disease using long-term surveillance.
The strategic factors that were considered to be 'high risk stigmata' and/or 'worrisome features', from the 2012 international consensus guidelines, were validated in this study. Although we did not investigate the presence of a thickened cystic wall or cytological pathology, additional factors that were associated with malignant and/or invasive disease were identified. We also revealed other prognostic factors, including sex and CA19-9, which were correlated with malignant and/or invasive IPMN in the multivariate analysis. Pedrazzoli et al.18 reported that positron emission tomography was more accurate than the guidelines, in distinguishing benign from malignant IPMN. Additionally, more predictive factors will likely be identified in future studies. When more prognostic and diagnostic data are accumulated, these guidelines should then be revised. Therefore, various trials, including those with retrospective and prospective designs, should be repeated to identify more accurate factors. The present algorithm for management is quite complicated for application in an outpatient clinical setting, and it only focuses on the morphological features, without regard to patient factors or biomarkers that are capable of reflecting the propensity of the disease.
Previously, patients with invasive IPMC were reported to be more likely to recur with a probability of hazard ratio of 5.2, but the original pathology did not predict disease severity on recurrence.17 Here, we observed recurrences in 16 (4.9%) of the 324 patients with resected BD-IPMN, including 36% of the invasive IPMC and 2.5% of the non-invasive IPMN cases. We determined that the initial disease pathology was the most powerful predictor of recurrence (OR 32.84: 95% CI 6.19-174.33), and that the initial pathology was related to the recurrence pattern (or severity) following resection. There were 8 of 9 patients with initially invasive IPMN who had distant metastases. or peritoneal seeding, while 5 of 7 patients with originally non-invasive IPMN had recurrences on the remnant pancreas or resected margin (Table 3). The latter 5 patients have remained alive and are under periods of regular observation that have ranged from 27.0 to 157.9 months. We experienced 2 cases of malignant recurrences of IMGD in the two year follow-up period after surgical resection (No. 9 and 11 in Table 3). Those cases initially had a single lesion in the pancreas, and the resected margins were clear from disease. Therefore, there must still be undisclosed biological characteristics of IPMN, and molecular or large scale clinical studies to prevent the deaths of patients with initially benign disease should be considered.
In conclusion, in a high-volume single-institutional study, we investigated the clinicopathological characteristics of BD-IPMN of the pancreas. Although the current treatment guidelines for BD-IPMN reflect malignant and invasive features appropriately, they are confined to the morphological characteristics of the disease. Patient factors and biological features should be considered when devising optimal therapeutic or surveillance strategies.
References
- 1.Conlon KC. Intraductal papillary mucinous tumors of the pancreas. J Clin Oncol. 2005;23:4518–4523. doi: 10.1200/JCO.2005.22.517. [DOI] [PubMed] [Google Scholar]
- 2.Rodriguez JR, Salvia R, Crippa S, Warshaw AL, Bassi C, Falconi M, et al. Branch-duct intraductal papillary mucinous neoplasms: observations in 145 patients who underwent resection. Gastroenterology. 2007;133:72–79. doi: 10.1053/j.gastro.2007.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sohn TA, Yeo CJ, Cameron JL, Iacobuzio-Donahue CA, Hruban RH, Lillemoe KD. Intraductal papillary mucinous neoplasms of the pancreas: an increasingly recognized clinicopathologic entity. Ann Surg. 2001;234:313–321. doi: 10.1097/00000658-200109000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Crippa S, Fernández-Del Castillo C, Salvia R, Finkelstein D, Bassi C, Domínguez I, et al. Mucin-producing neoplasms of the pancreas: an analysis of distinguishing clinical and epidemiologic characteristics. Clin Gastroenterol Hepatol. 2010;8:213–219. doi: 10.1016/j.cgh.2009.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mimura T, Masuda A, Matsumoto I, Shiomi H, Yoshida S, Sugimoto M, et al. Predictors of malignant intraductal papillary mucinous neoplasm of the pancreas. J Clin Gastroenterol. 2010;44:e224–e229. doi: 10.1097/MCG.0b013e3181d8fb91. [DOI] [PubMed] [Google Scholar]
- 6.Salvia R, Fernández-del Castillo C, Bassi C, Thayer SP, Falconi M, Mantovani W, et al. Main-duct intraductal papillary mucinous neoplasms of the pancreas: clinical predictors of malignancy and long-term survival following resection. Ann Surg. 2004;239:678–685. doi: 10.1097/01.sla.0000124386.54496.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schmidt CM, White PB, Waters JA, Yiannoutsos CT, Cummings OW, Baker M, et al. Intraductal papillary mucinous neoplasms: predictors of malignant and invasive pathology. Ann Surg. 2007;246:644–651. doi: 10.1097/SLA.0b013e318155a9e5. [DOI] [PubMed] [Google Scholar]
- 8.Shin SH, Han DJ, Park KT, Kim YH, Park JB, Kim SC. Validating a simple scoring system to predict malignancy and invasiveness of intraductal papillary mucinous neoplasms of the pancreas. World J Surg. 2010;34:776–783. doi: 10.1007/s00268-010-0416-5. [DOI] [PubMed] [Google Scholar]
- 9.Sohn TA, Yeo CJ, Cameron JL, Hruban RH, Fukushima N, Campbell KA, et al. Intraductal papillary mucinous neoplasms of the pancreas: an updated experience. Ann Surg. 2004;239:788–797. doi: 10.1097/01.sla.0000128306.90650.aa. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sugiyama M, Izumisato Y, Abe N, Masaki T, Mori T, Atomi Y. Predictive factors for malignancy in intraductal papillary-mucinous tumours of the pancreas. Br J Surg. 2003;90:1244–1249. doi: 10.1002/bjs.4265. [DOI] [PubMed] [Google Scholar]
- 11.Tamura K, Ohtsuka T, Ideno N, Aso T, Shindo K, Aishima S, et al. Treatment strategy for main duct intraductal papillary mucinous neoplasms of the pancreas based on the assessment of recurrence in the remnant pancreas after resection: a retrospective review. Ann Surg. 2014;259:360–368. doi: 10.1097/SLA.0b013e3182a690ff. [DOI] [PubMed] [Google Scholar]
- 12.Tanaka M, Chari S, Adsay V, Fernandez-del Castillo C, Falconi M, Shimizu M, et al. International Association of Pancreatology. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology. 2006;6:17–32. doi: 10.1159/000090023. [DOI] [PubMed] [Google Scholar]
- 13.Tanaka M, Fernández-del Castillo C, Adsay V, Chari S, Falconi M, Jang JY, et al. International Association of Pancreatology. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology. 2012;12:183–197. doi: 10.1016/j.pan.2012.04.004. [DOI] [PubMed] [Google Scholar]
- 14.Bosman FT, Carneiro F, Hruban RH, Theise ND. WHO classification of tumours of the digestive system. Lyon, France: IARC Press; 2010. [Google Scholar]
- 15.Lee SY, Lee KT, Lee JK, Jeon YH, Choi D, Lim JH, et al. Long-term follow up results of intraductal papillary mucinous tumors of pancreas. J Gastroenterol Hepatol. 2005;20:1379–1384. doi: 10.1111/j.1440-1746.2005.04014.x. [DOI] [PubMed] [Google Scholar]
- 16.Nagai K, Doi R, Kida A, Kami K, Kawaguchi Y, Ito T, et al. Intraductal papillary mucinous neoplasms of the pancreas: clinicopathologic characteristics and long-term follow-up after resection. World J Surg. 2008;32:271–278. doi: 10.1007/s00268-007-9281-2. [DOI] [PubMed] [Google Scholar]
- 17.Winner M, Epelboym I, Remotti H, Lee JL, Schrope BA, Chabot JA, et al. Predictors of recurrence in intraductal papillary mucinous neoplasm: experience with 183 pancreatic resections. J Gastrointest Surg. 2013;17:1618–1626. doi: 10.1007/s11605-013-2242-1. [DOI] [PubMed] [Google Scholar]
- 18.Pedrazzoli S, Sperti C, Pasquali C, Bissoli S, Chierichetti F. Comparison of International Consensus Guidelines versus 18-FDG PET in detecting malignancy of intraductal papillary mucinous neoplasms of the pancreas. Ann Surg. 2011;254:971–976. doi: 10.1097/SLA.0b013e3182383137. [DOI] [PubMed] [Google Scholar]