Stimulation by CXCL12 of α4β1-mediated cell adhesion is crucial for lymphocyte trafficking. SLP-76 and ADAP activate the strength of α4β1-VCAM-1 interaction and cell spreading in response to CXCL12, whereas Pyk2 opposes these processes. Rac1 activation mediates SLP-76–, ADAP-, and Pyk2-regulated adhesion involving α4β1.
Abstract
Stimulation by chemokines of integrin α4β1–dependent T-lymphocyte adhesion is a crucial step for lymphocyte trafficking. The adaptor Vav1 is required for chemokine-activated T-cell adhesion mediated by α4β1. Conceivably, proteins associating with Vav1 could potentially modulate this adhesion. Correlating with activation by the chemokine CXCL12 of T-lymphocyte attachment to α4β1 ligands, a transient stimulation in the association of Vav1 with SLP-76, Pyk2, and ADAP was observed. Using T-cells depleted for SLP-76, ADAP, or Pyk2, or expressing Pyk2 kinase–inactive forms, we show that SLP-76 and ADAP stimulate chemokine-activated, α4β1-mediated adhesion, whereas Pyk2 opposes T-cell attachment. While CXCL12-promoted generation of high-affinity α4β1 is independent of SLP-76, ADAP, and Pyk2, the strength of α4β1-VCAM-1 interaction and cell spreading on VCAM-1 are targets of regulation by these three proteins. GTPase assays, expression of activated or dominant-negative Rac1, or combined ADAP and Pyk2 silencing indicated that Rac1 activation by CXCL12 is a common mediator response in SLP-76–, ADAP-, and Pyk2-regulated cell adhesion involving α4β1. Our data strongly suggest that chemokine-stimulated associations between Vav1, SLP-76, and ADAP facilitate Rac1 activation and α4β1-mediated adhesion, whereas Pyk2 opposes this adhesion by limiting Rac1 activation.
INTRODUCTION
Trafficking of T-lymphocytes from blood circulation to lymphoid tissue and to sites of injury and infection depends on rapid and transient activation of αLβ2 and α4β1 integrin function by chemokines located on the endothelium and inside tissues (Luster et al., 2005; Ley et al., 2007). Following the binding of chemokines to their receptors, an inside-out signaling that impinges on the cytoplasmic domains of integrins is generated (Thelen and Stein, 2008). The final consequence of this signaling is the generation of high-affinity extracellular αLβ2- and α4β1-integrin conformations that are competent for binding to their ligands ICAM-1 and VCAM-1, respectively, which are expressed on lymphoid or inflamed endothelium, and also facilitates α4β1 interaction with the CS-1 region of tissue fibronectin.
While talin binding to cytoplasmic motifs on β1- and β2-integrin subunits represents a common and central event for transmission of inside-out signaling from chemokine receptors to generate active α4β1 and αLβ2 (Moser et al., 2009b; Hogg et al., 2011; Kim et al., 2011; Calderwood et al., 2013), important differences exist in the molecular pattern of this signaling to either integrin. αLβ2-dependent leukocyte adhesion stimulated through inside-out signaling shows a crucial requirement for kindlin 3, another important β subunit–binding protein, and to a lesser extent for α4β1 (Malinin et al., 2009; Manevich-Mendelson et al., 2009; Moser et al., 2009a; Svensson et al., 2009). Of note, the RAPL-Rap1 and Rho/Rac-phospholipase D pathways are critical for chemokine-stimulated T-cell adhesion involving αLβ2, with no or lesser roles in α4β1-mediated adhesion (Katagiri et al., 2003; Ghandour et al., 2007; Bolomini-Vittori et al., 2009). Instead, the Vav1-Rac1 pathway is required for T-cell adhesion involving α4β1 upon exposure to chemokines (Garcia-Bernal et al., 2005). Vav1 is a multidomain adaptor protein that functions as a guanine nucleotide exchange factor (GEF) predominantly for the Rac1 GTPase (Bustelo, 2000; Turner and Billadeau, 2002; Tybulewicz, 2005). In T-cells, chemokine-induced, ZAP-70–dependent Vav1 tyrosine phosphorylation and Vav1 dissociation from talin represent an important event involved in α4β1 activation and in adhesion strengthening mediated by this integrin (Garcia-Bernal et al., 2009).
Vav1 binds key molecules involved in T-lymphocyte activation, such as SLP-76 and Pyk2 (Tuosto et al., 1996; Wu et al., 1996; Katagiri et al., 2000; Gismondi et al., 2003). SLP-76 is an adaptor molecule that is essential for T-cell activation and actin reorganization following T-cell receptor (TCR) stimulation (Koretzky et al., 2006). On TCR activation, ZAP-70–mediated phosphorylation of SLP-76 on N-terminal tyrosine residues allows its binding to the SH2 domain of Vav1 (Tuosto et al., 1996; Wu et al., 1996; Raab et al., 1997), whereas the C-terminal SH2 domain of SLP-76 interacts with several tyrosines on ADAP (da Silva et al., 1997; Musci et al., 1997; Geng et al., 1999; Boerth et al., 2000), a key molecule that also regulates T-cell activation (Griffiths and Penninger, 2002).
Pyk2 is a non receptor tyrosine kinase that is homologous to FAK (Avraham et al., 1995; Lev et al., 1995; Sasaki et al., 1995) and is activated following TCR stimulation and cell exposure to chemoattractants (Dikic et al., 1996; Davis et al., 1997; Ganju et al., 1997; Qian et al., 1997). Early data showed that Pyk2 is capable of associating with Vav1 in T and NK cells (Katagiri et al., 2000; Gismondi et al., 2003; Gao and Blystone, 2009). There are three Pyk2 isoforms of 110 kDa, 105 kDa, and a third species called PRNK (Pyk2-related nonkinase) that only contains 238 amino acids from the C-terminus and lacks the catalytic and N-terminal regions (Xiong et al., 1998; Ivankovic-Dikic et al., 2000). A Tyr-402 residue is a target of Pyk2 autophosphorylation, which in turns recruits and activates Src kinases, leading to Src-dependent phosphorylation of several tyrosine residues on Pyk2 (Dikic et al., 1996; Li et al., 1999; Avraham et al., 2000).
The role of SLP-76, ADAP, and Pyk2 in integrin-mediated T-lymphocyte adhesion has been mostly studied in TCR-activated cells. Thus SLP-76 mediates αLβ2-dependent T-cell adhesion in response to TCR stimulation (Baker et al., 2009; Horn et al., 2009). In addition, it has been demonstrated that ADAP is required for TCR-stimulated cell adhesion mediated by β1 and β2 integrins (Griffiths et al., 2001; Peterson et al., 2001), a process regulated by an ADAP/SKAP-55 signaling module (Wang et al., 2003, 2007; Kliche et al., 2006). As for Pyk2, its tyrosine phosphorylation as a consequence of integrin-dependent outside-in signaling was earlier demonstrated (Li et al., 1996; van Seventer et al., 1998; Zheng et al., 1998), and it was shown that Pyk2 regulates TCR-stimulated, LFA-1–dependent CD8+ cell adhesion (Beinke et al., 2010).
Little is known on the potential involvement of SLP-76 and Pyk2, as well as of ADAP, in chemokine-stimulated T-cell adhesion dependent on integrins. For instance, SLP-76 is required for optimal T-lymphocyte arrest on ICAM-1 under flow conditions (Lee et al., 2012). In addition, it has been recently shown that CCR7-mediated αLβ2 functions in T-cells are regulated by two independent ADAP/SKAP55 modules (Kliche et al., 2012). The integrin α4β1 is especially involved in chemokine-stimulated T-lymphocyte trafficking to sites of inflammation, with lesser roles in cell migration to lymphoid tissues (Berlin-Rufenach et al., 1999), thus highlighting the structural and functional differences between α4β1 and αLβ2. The role of SLP-76, ADAP, and Pyk2 in chemokine-activated T-cell adhesion mediated by α4β1 has not yet been addressed. In the present work, we have studied the potential involvement of these signaling molecules in T-lymphocyte adhesion dependent on α4β1.
RESULTS
Role of SLP-76, ADAP, and Pyk2 on chemokine-activated T-cell adhesion mediated by α4β1
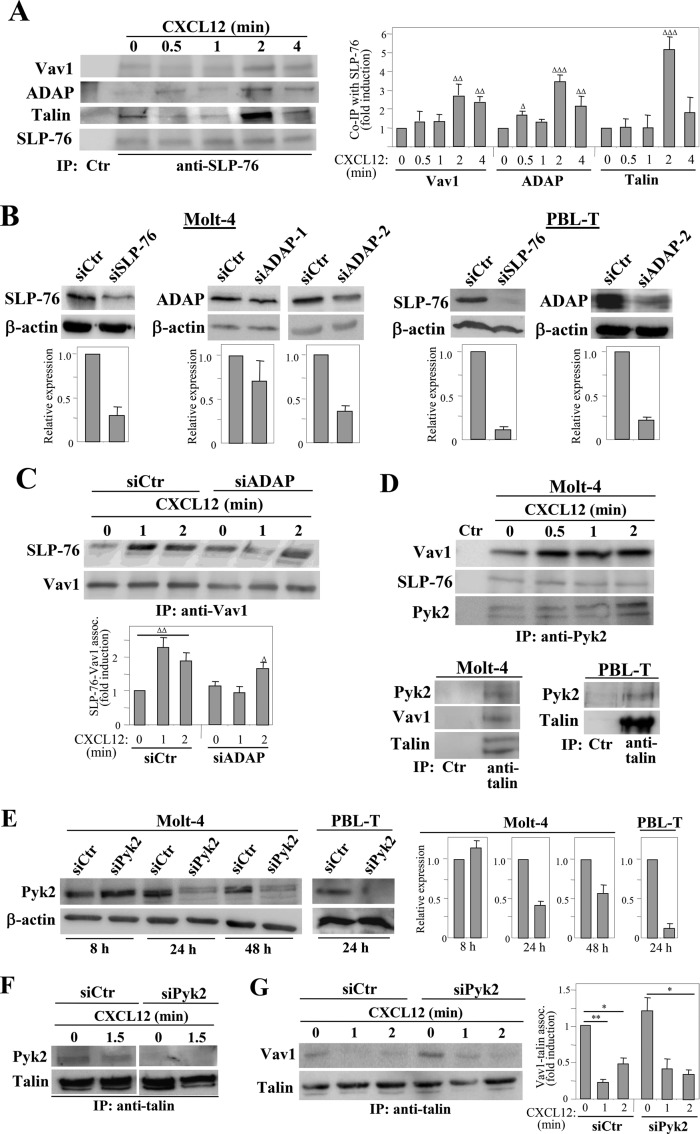
Coimmunoprecipitation analyses using anti–SLP-76 monoclonal antibody (mAb) revealed that CXCL12 stimulates a transient increase in SLP-76 association with Vav1 and ADAP (Figure 1A). In agreement with previous work (Fischer et al., 1998), talin was found in anti–SLP-76 immunoprecipitates, and CXCL12 also up-regulated this association. No β1-integrin subunit was found in anti–SLP-76 immunoprecipitates (unpublished data). Cell fractionation experiments showed that SLP-76 is mainly found on the membrane-associated fraction, and its localization was not altered by CXCL12 (Supplemental Figure S1A). Instead, ADAP mostly displayed a cytosolic localization in nonstimulated cells, but it was rapidly and transiently recruited to the membrane following incubation with CXCL12, coincident with the stimulation by this chemokine of SLP-76-ADAP association.
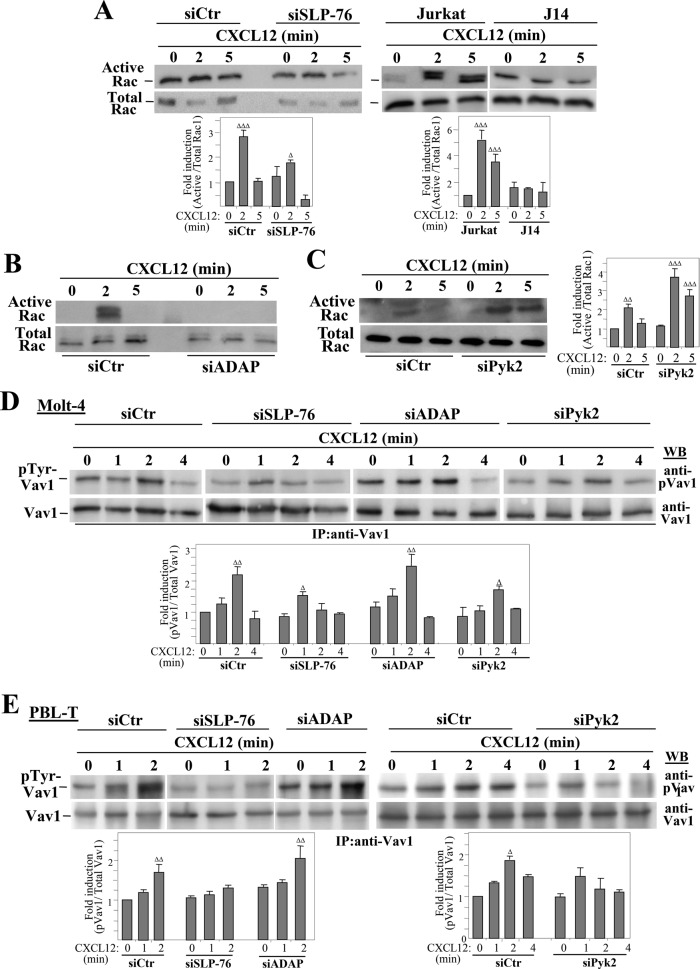
FIGURE 1:
Chemokine-stimulated associations between Vav1, SLP-76, ADAP, and Pyk2. (A) Left, Molt-4 T-cells incubated for the indicated times with CXCL12 were subjected to immunoprecipitation with anti–SLP-76 followed by immunoblotting with antibodies to the indicated proteins. Right, bars represent densitometric quantification of gel bands showing the mean ± SD of six independent experiments (ΔΔΔ, p < 0.001; ΔΔ, p < 0.01; Δ, p < 0.05). (B) Top, cells were transfected with SLP-76, ADAP, or control siRNA, and expression of SLP-76 and ADAP was analyzed by immunoblotting. Control loading is shown by blotting with anti–β-actin antibodies. Bottom, densitometric quantification of gel bands showing the mean ± SD of four (Molt-4) or three (PBL-T) independent experiments. (C) CXCL12-incubated control or ADAP siRNA Molt-4 transfectants were assayed by immunoprecipitation with anti-Vav1 antibodies, followed by immunoblotting with antibodies to the proteins shown (ΔΔ, p < 0.01; Δ, p <0.05). (D) Cells were incubated in the absence or presence of CXCL12, and subsequently subjected to immunoprecipitation and Western blotting. (E) Left, Cells were transfected with Pyk2 or control siRNA, and transfectants were assayed by Western blotting at the indicated times. Right, densitometric analyses of gel bands showing the mean ± SD of three independent experiments. (F and G) Control or Pyk2 siRNA transfectants were subjected to immunoprecipitation with anti-talin antibodies, followed by immunoblotting with antibodies to the shown proteins. Talin-Vav1 coprecipitation was significantly diminished (**, p < 0.001; *, p < 0.05; n = 4).
To study potential connections between SLP-76 and ADAP in chemokine-activated T-cell adhesion involving α4β1, we knocked them down using RNA interference in Molt-4 and peripheral blood T-lymphocytes (PBL-T). SLP-76 was depleted with a pool of SLP-76 small interfering RNA (siRNA; see Materials and Methods), whereas ADAP was found to be more efficiently silenced by ADAP-2 than ADAP-1 siRNA (Figure 1B), and we therefore used ADAP-2 siRNA to carry out the subsequent functional assays. Control experiments indicated that SLP-76 or ADAP depletion did not affect cell viability or the expression of CXCR4, α4 or β1 (unpublished data). ADAP knocking down did not alter the coprecipitation between Vav1 and SLP-76 in nonstimulated cells (t = 0), but their increased association in CXCL12-incubated cells was delayed and of smaller magnitude (Figure 1C), suggesting that a critical level of ADAP expression and/or its localization was needed for enhanced Vav1-SLP-76 association.
Previous data showed that the kinase Pyk2 binds to the SH3 domain of Vav1 in Jurkat T-cells (Katagiri et al., 2000). Likewise, Vav1 associated with Pyk2 in Molt-4 cells, and CXCL12 triggered a transient increase in Vav1-Pyk2 binding (Figure 1D, top, and Supplemental Figure S1B), in line with previous findings (Okabe et al., 2005). In addition, low amounts of SLP-76 were also detected in anti-Pyk2 immunoprecipitates. ADAP was minimally detected only in Pyk2 immunoprecipitates from cells not exposed to CXCL12 (unpublished data). Furthermore, we found that Vav1-Pyk2 coimmunoprecipitation was independent of SLP-76 or ADAP, as this association was similarly detected in control, SLP-76 (Supplemental Figure S1C, left), or ADAP siRNA transfectants (unpublished data). Interestingly, antibodies to talin, an additional Vav1 binding partner (Fischer et al., 1998; Garcia-Bernal et al., 2009), coprecipitated Pyk2 in addition to Vav1 in Molt-4 and PBL-T (Figure 1D, bottom). Supporting the specificity of talin-Pyk2 assembly, talin-Pyk2 coprecipitation was diminished in Pyk2 knockdown cells (Figure 1, E and F). Whether Pyk2 directly binds to talin, as in FAK-talin interaction (Mitra et al., 2005), or indirectly through its association with Vav1 has not been addressed in the present study. In addition, Pyk2 knocking down did not significantly alter Vav1-talin association or CXCL12-promoted Vav1-talin dissociation (Figure 1G), an event contributing to the initial steps of α4β1-mediated T-cell adhesion (Garcia-Bernal et al., 2009). Moreover, Vav1 and SLP-76 (Supplemental Figure S1C, right) and SLP-76-talin (unpublished data) interactions were not altered following Pyk2 silencing. We also observed that Pyk2-depleted cells had similar Erk1/2 MAP kinase activation at early time points following stimulation with CXCL12, but this activation was defective at the longest times tested (Supplemental Figure S1D). Instead, CXCL12-promoted Akt stimulation was not altered in Pyk2-knockdown transfectants.
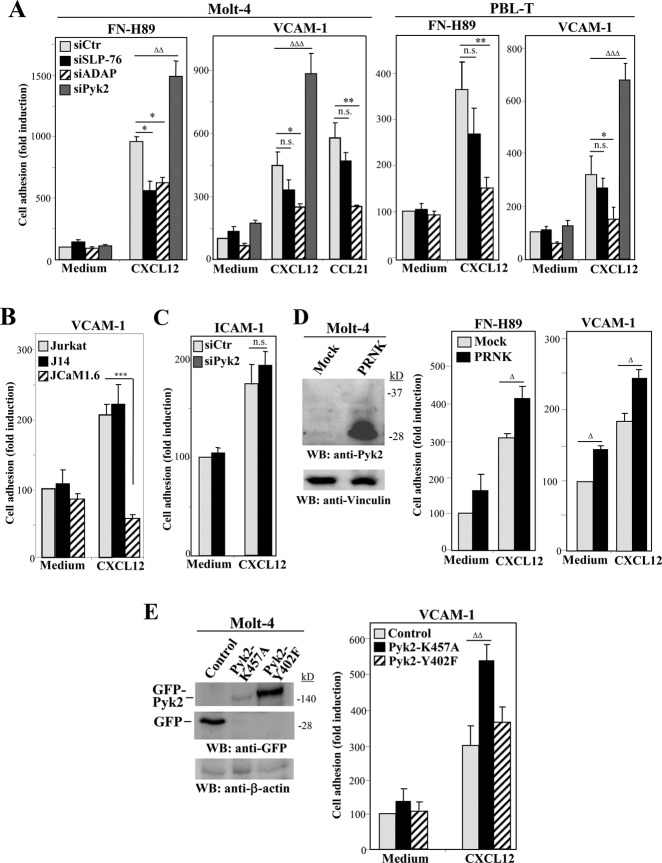
We next tested SLP-76–, ADAP-, and Pyk2-depleted T-cells in α4β1-mediated static cell adhesion assays. As chemokine–up-regulated T-cell attachment is rapid and transient (Grabovsky et al., 2000), the adhesion assays to α4β1 ligands coimmobilized with chemokines were performed using short incubation times (2 min) upon a brief cell centrifugation (15 s) to place cells immediately in contact with ligands. Under these conditions, we observed that SLP-76–depleted Molt-4 and PBL-T had modest reductions in adhesion to FN-H89 (CS-1/FN), whereas their attachment to VCAM-1 displayed small but nonsignificant decreases compared with control siRNA transfectants (Figure 2A). Similarly, SLP-76–deficient J14 cells displayed chemokine-promoted adhesion to VCAM-1 comparable with parental Jurkat cells, whereas activated adhesiveness of Lck-deficient JCaM1.6 cells was fully abrogated (Figure 2B), consistent with previous data (Feigelson et al., 2001). ADAP depletion generally affected to a higher degree the stimulation of α4β1-dependent T-cell attachment compared with SLP-76 knocking down (Figure 2A). In addition, ADAP-depleted Molt-4 cells exhibited a substantial inhibition of CXCL12-triggered, α4β1-mediated migration across tumor necrosis factor α–treated HUVEC monolayers under static conditions (Supplemental Figure S2), whereas transfectants knocked down for SLP-76 (Supplemental Figure S2) and J14 cells (unpublished data) displayed partial impairments in migration.
FIGURE 2:
Chemokine-stimulated static adhesion of SLP-76, ADAP, or Pyk2 transfectants to α4β1 ligands. (A) Control, SLP-76–, ADAP-, or Pyk2-knockdown Molt-4 or PBL-T transfectants were subjected to adhesion assays to FN-H89 or VCAM-1 in the absence (Medium) or presence of coimmobilized CXCL12 or CCL21 (n = 3–5). (B) Parental Jurkat, J14, and JCaM1.6 cells were tested in adhesion assays to VCAM-1 as in A (n = 2). (C) Molt-4 cells were transfected with control or Pyk2 siRNA and transfectants tested in adhesion assays to ICAM-1 coimmobilized with or without CXCL12 (n = 3). (D) Cells were transfected with empty (Mock) or PRNK vectors, and transfectants were tested by Western blotting for PRNK expression (left) or in adhesion assays (middle and right) (n = 4). (E) Cells were transfected with control GFP vector or with the indicated GFP-fused Pyk2 mutants, and transfectants were subjected to immunoblotting or to adhesion assays (n = 4). Adhesions were significantly inhibited (***, p < 0.001; **, p < 0.01; *, p < 0.05) or significantly stimulated (ΔΔΔ, p < 0.001; ΔΔ, p < 0.01; Δ, p <0.05) (n.s., nonsignificant).
Of note, Pyk2 knocking down resulted in significant increases in chemokine-triggered T-cell adhesion to both FN-H89 and VCAM-1 relative to control siRNA transfectants (Figure 2A). Instead, we were unable to detect alterations in attachment to ICAM-1 with CXCL12-incubated, Pyk2-silenced cells (Figure 2C), in line with previous results using Pyk2−/− T-cells exposed to standard doses of anti-CD3 antibodies (Beinke et al., 2010). Moreover, overexpression of PRNK, a Pyk2 C-terminal region isoform that lacks the kinase domain (Xiong et al., 1998) and inhibits the endogenous Pyk2 kinase activity, led to up-regulation of α4β1-dependent attachment of nonstimulated T-cells, which achieved a further enhancement in the presence of CXCL12 (Figure 2D). In support of an involvement of Pyk2 kinase activity in the regulation of α4β1-mediated T-cell adhesion, we found that the expression of the Pyk2-K457A kinase–inactive mutant (Katagiri et al., 2000) resulted in a remarkable increase in cell attachment to VCAM-1 (Figure 2E). Instead, mutation at the Tyr-402 Pyk2 autophosphorylation site (Pyk2-Y402F) did not alter this adhesion. We were unable to use transfectants overexpressing wild-type Pyk2 in the adhesion assays, as they displayed a high degree of cell death (unpublished data), consistent with previously reported data (Xiong and Parsons, 1997).
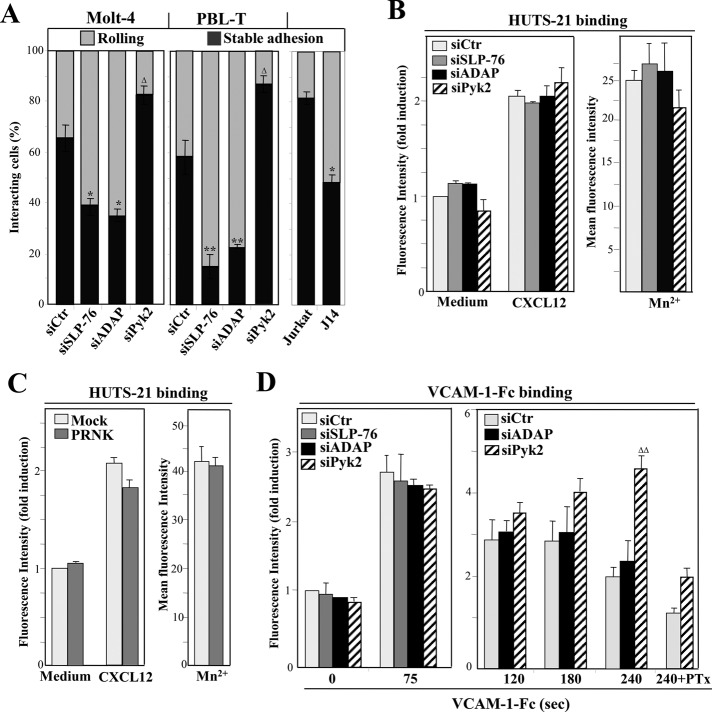
Next we carried out adhesion assays under flow conditions using SLP-76–, ADAP-, or Pyk2-depleted T-cells, measuring the cells that rapidly (< 20 s) and stably adhere under a shear stress of 1 dyne/cm2 to VCAM-1 coimmobilized with CXCL12. Results revealed that Molt-4 and PBL-T knocked down for SLP-76 or ADAP have a decrease (40–75%) in stable cell arrest on VCAM-1/CXCL12 compared with control siRNA transfectants (Figure 3A). Confirming the involvement of SLP-76, J14 cells also displayed a significant reduction in cell arrest relative to parental Jurkat cells (Figure 3A). Therefore the contribution of SLP-76 in chemokine-stimulated lymphocyte adhesion to VCAM-1 is especially relevant under shear stress but hardly seen in static conditions, similar to what has been reported in CXCL12–up-regulated, αLβ2-dependent adhesion to ICAM-1 of J14 and SLP-76–depleted cells (Horn et al., 2009; Lee et al., 2012). Contrary to SLP-76 and ADAP silencing, Pyk2 knockdown transfectants showed stronger cell arrest than control siRNA transfectants (Figure 3A). Control experiments indicated that adhesion was abolished by pretreating cells with pertussis toxin (PTx; unpublished data).
FIGURE 3:
Flow-chamber adhesion assays and determination of high-affinity α4β1 expression in SLP-76–, ADAP-, or Pyk2-depleted T-cells. (A) Control, SLP-76, ADAP, or Pyk2 siRNA transfectants or Jurkat or J14 cells were perfused in flow chambers coated with VCAM-1 coimmobilized with CXCL12 and analyzed for rolling and stable cell arrest (n = 3–4). Data are presented as mean ± SD of cell percentages from the total cell population. Adhesions were significantly inhibited or stimulated in comparison with those of control siRNA transfectants or parental Jurkat cells, *p < 0.05 or Δp < 0.05, respectively. (B and C) The indicated siRNA Molt-4 transfectants or cells transfected with PRNK or empty vector were tested by flow cytometry for HUTS-21 mAb binding after stimulation with CXCL12 or Mn2+. (D) Following exposure to CXCL12 for 20 s, transfectants were analyzed by flow cytometry for VCAM-1-Fc binding after the indicated times. PTx denotes cells preincubated with pertussis toxin.
Generation of α4β1 high-affinity conformations upon CXCL12 stimulation is independent of SLP-76, ADAP, or Pyk2 activities
Changes in adhesion to VCAM-1 following SLP-76, ADAP, or Pyk2 depletion could arise from altered acquisition of integrin high-affinity conformations. We used HUTS-21, a reporter mAb that recognizes a β1-integrin activation epitope, to analyze whether knockdown of these proteins affects the affinity of α4β1. Chemokine-incubated SLP-76–, ADAP-, or Pyk2-depleted cells, as well as PRNK transfectants, showed no gross alterations in HUTS-21 binding (Figure 3, B and C). Control experiments revealed that all transfectants retained similar degrees of HUTS-21 mAb binding upon exposure to Mn2+ (Figure 3, B and C), a positive control for integrin-affinity regulation. These results indicate that chemokine-stimulated high-affinity α4β1 does not require SLP-76, ADAP, or Pyk2 functions.
To further examine a potential involvement of these three proteins in early steps of CXCL12-stimulated, α4β1-dependent cell adhesion, we used VCAM-1-Fc in soluble binding assays to determine the efficiency of α4β1-VCAM-1 interaction without the subsequent cell spreading step. Using short-incubation conditions to obtain a two- to threefold induction in VCAM-1-Fc binding (20-s stimulation with CXCL12 followed by 75-s incubation with VCAM-1-Fc; Garcia-Bernal et al., 2005), we observed similar VCAM-1-Fc binding to SLP-76–, ADAP-, or Pyk2-depleted cells relative to control siRNA transfectants (Figure 3D, left), which was inhibited by 80% by PTx (unpublished data). When we tested longer incubations with VCAM-1-Fc, we observed that while control and ADAP siRNA transfectants (Figure 3D, right) and SLP-76 knockdown cells (unpublished data) have maximal interaction with VCAM-1 between 2 and 3 min, after which binding decreases, Pyk2-depleted cells displayed a consistent sustainment of α4β1-VCAM-1-Fc interaction that was inhibited by PTx. All transfectants displayed comparable VCAM-1-Fc binding upon exposure to Mn2+ (unpublished data). These results suggest that Pyk2 silencing allows a Gαi-dependent sustained signaling to regulate α4β1 avidity, which likely contributes to maintaining α4β1-VCAM-1 interaction.
Role of SLP-76, ADAP, and Pyk2 in adhesion strengthening and spreading mediated by α4β1
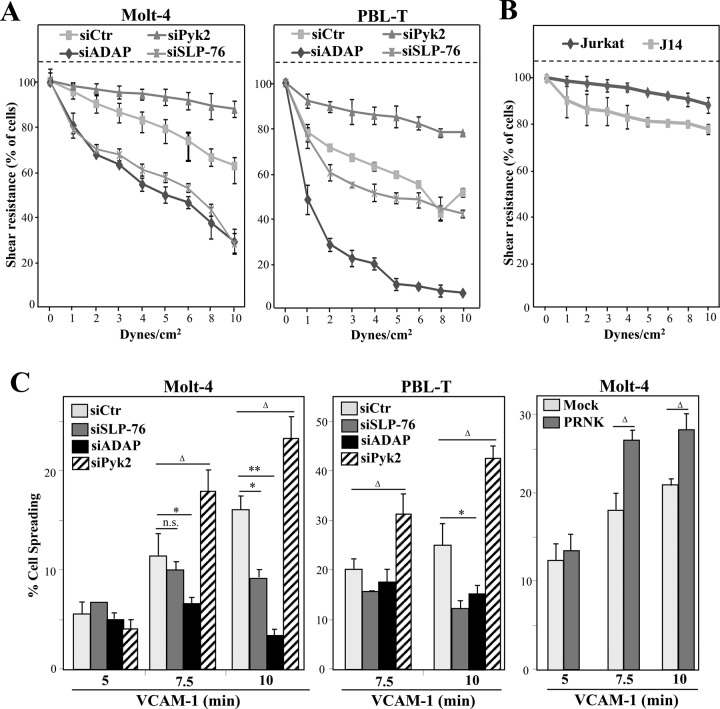
To analyze the α4β1-dependent adhesion strength of transfectants depleted for SLP-76, ADAP, or Pyk2, we subjected cells that had been attached to VCAM-1/CXCL12 at 1 dyne/cm2 to detachment at sequential increases of shear stress. ADAP- and SLP-76–depleted PBL-T and Molt-4 transfectants, as well as J14 cells, were found to develop lower resistance to detachment at high shear stress than control siRNA transfectants, although ADAP silencing consistently led to higher detachment rates than SLP-76 knockdown (Figure 4, A and B). Instead, Pyk2-depleted T-cells exhibited higher resistance to detachment than control siRNA transfectants (Figure 4A).
FIGURE 4:
Analyses of T-cell adhesion strengthening and spreading on VCAM-1. The indicated siRNA transfectants (A) or Jurkat and J14 cells (B), preattached onto coimmobilized VCAM-1 and CXCL12 in flow chambers, were subjected to cell detachment after sequential increases of shear stress. Data show mean ± SD of cell percentages from the initial number of bound cells remaining attached at the indicated shear stresses (A, n = 4; B, n = 3). (C) Molt-4 and PBL-T were transfected with the indicated siRNA or vectors, and transfectants were allowed to attach to VCAM-1 immobilized with CXCL12. Spreading was evaluated from Nomarski images at the indicated times. Percentage of cell spreading was determined from cells (2500–3000) from different fields of view. Spreading was significantly inhibited (**, p < 0.01; *, p < 0.05) or stimulated (Δ, p < 0.05) compared with control siRNA or mock transfectants (n = 3). n.s., nonsignificant.
In the adhesion process, cells undergo spreading after the strengthening of the adhesion. Both SLP-76– and ADAP-knockdown Molt-4 and PBL-T transfectants developed a time-dependent progressive reduction in the spreading on VCAM-1/CXCL12 compared with control counterparts (Figure 4C, left and middle). On the contrary, Pyk2 knockdown and PRNK transfectants displayed higher spreading than control cells (Figure 4C). These data indicate that ADAP and Pyk2 regulate the chemokine-stimulated T-cell adhesion strengthening and spreading steps mediated by α4β1, whereas SLP-76 mainly contributes to cell spreading, with lesser roles in adhesion strengthening.
Rac1 involvement in SLP-76, ADAP, and Pyk2 regulation of CXCL12-activated T-cell adhesion dependent on α4β1
The strength of α4β1-mediated adhesion and the subsequent spreading process in chemokine-stimulated T-cells depends on the activation of the GTPase Rac1 (Garcia-Bernal et al., 2005). Compared with control cells, knocking down SLP-76 led to a less efficient Rac1 activation in Molt-4 cells in response to CXCL12, which was rapidly lost at the longest incubation time tested (Figure 5A, top). Moreover, CXCL12-exposed J14 cells displayed no stimulation of Rac activation relative to parental Jurkat cells (Figure 5A, bottom). In addition, ADAP-depleted transfectants displayed a blockade of CXCL12-stimulated Rac activation (Figure 5B). Furthermore, inhibition of Rac activation in SLP-76– and ADAP-knockdown transfectants was associated with impaired stimulation by CXCL12 of F-actin polymerization (Supplemental Figure S3A). In contrast, Pyk2 silencing led to a more sustained Rac1 activation and F-actin polymerization than control transfectants (Figure 5C and Supplemental Figure S3B).
FIGURE 5:
Analyses of Rac1 activation and Vav1 tyrosine phosphorylation in SLP-76–, ADAP-, or Pyk2-deficient T-cells. Control, SLP-76, ADAP, or Pyk2 siRNA transfectants in Molt-4 cells, or Jurkat and J14 cells (A–C) were stimulated with CXCL12 and subjected to GTPase assays to detect Rac1 activation. Activation was significantly increased compared with untreated cells (ΔΔΔ, p < 0.001; ΔΔ, p < 0.01; Δ, p < 0.05; A, n = 7; B and C, n = 3). The indicated Molt-4 (D) or PBL-T (E) siRNA transfectants incubated with or without CXCL12 for the indicated times were analyzed by immunoprecipitation and Western blotting. Vav1 tyrosine phosphorylation was significantly up-regulated compared with untreated cells (ΔΔ, p < 0.01; Δ, p < 0.05; D, n = 6; E, left, n = 2; E, right, n = 3).
Chemokine-stimulated Rac1 activation in T-cells was previously shown to involve Vav1 tyrosine phosphorylation (Garcia-Bernal et al., 2005). In SLP-76–silenced cells, impaired Rac1 activation was associated with a lower degree and less maintained chemokine-promoted Vav1 tyrosine phosphorylation compared with control siRNA transfectants, whereas no gross alterations in Vav1 phosphorylation were detected in ADAP-depleted cells (Figure 5D), similar to the unaltered Vav1 phosphorylation in ADAP knockout mice (Reynolds et al., 2002). Furthermore, the CXCL12-triggered enhancement in Rac1 activation seen in Pyk2-silenced transfectants did not correlate with higher Vav1 tyrosine phosphorylation than control counterparts, and this phosphorylation generally displayed lower intensity compared with control transfectants (Figure 5D). Similar to Molt-4 cells, SLP-76 knockdown in PBL-T resulted in inefficient Vav1 tyrosine phosphorylation in response to CXCL12, whereas phosphorylation was not affected in ADAP-depleted cells (Figure 5E). Again, Pyk2-silenced PBL-T exhibited CXCL12-stimulated Vav1 tyrosine phosphorylation but of lower magnitude and less maintained than control siRNA transfectants (Figure 5E).
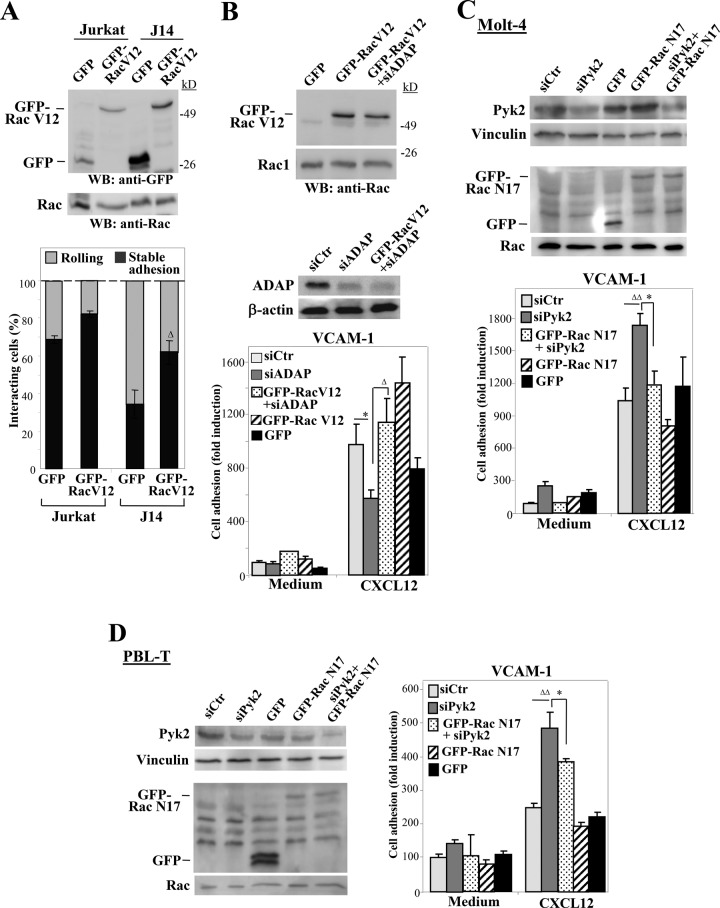
Next we tested the relevance of the deficient Rac activation shown by SLP-76– and ADAP-deficient T-cells on their impaired adhesion. For this, we transfected green fluorescent protein (GFP)-fused, constitutively active forms of Rac (GFP-Rac V12) in J14 and parental Jurkat cells or expressed GFP-Rac V12 together with ADAP siRNA, and then we tested the transfectants in adhesion assays to VCAM-1 coimmobilized with CXCL12. Flow-chamber adhesion assays revealed that the reduction in stable adhesion shown by J14-GFP cells relative to parental Jurkat-GFP was significantly rescued upon expression of GFP-Rac V12 (Figure 6A). Likewise, GFP-Rac V12 expression reversed the inhibitory effects of ADAP siRNA transfection on the stimulation of cell adhesion to VCAM-1/CXCL12 (Figure 6B). Moreover, the up-regulated adhesion to VCAM-1/CXCL12 shown by Pyk2-knockdown Molt-4 and PBL-T transfectants was partially reversed when dominant-negative, GFP-fused Rac1 (GFP-Rac N17) was transfected together with Pyk2 siRNA (Figure 6, C and D). These results strongly suggest that altered Rac1 activation in SLP-76–, ADAP-, or Pyk2-knockdown T-cells represents an important mechanism mediating their changes in chemokine-stimulated, α4β1-dependent cell adhesion.
FIGURE 6:
Rac1 involvement in SLP-76, ADAP, and Pyk2 regulation of CXCL12-activated T-cell adhesion dependent on α4β1. (A) Cells were transfected with GFP or GFP-Rac V12 vectors and were analyzed by immunoblotting (top) or were subjected to flow-chamber adhesion assays to VCAM-1 coimmobilized with CXCL12 (bottom). Adhesion was significantly rescued in comparison with J14-GFP transfectants (Δ, p < 0.05; n = 3). (B) Molt-4 cells were transfected with the indicated siRNA and GFP vector combinations and were analyzed by Western blotting (top) or in static adhesion assays to VCAM-1 immobilized with or without CXCL12 (bottom). Adhesion was significantly inhibited (*, p < 0.05) or recovered (Δ, p < 0.05). (C and D) The indicated Molt-4 or PBL-T transfectants were tested by immunoblotting or were subjected to adhesion assays to VCAM-1 immobilized with or without CXCL12. Adhesion was significantly stimulated (ΔΔ, p < 0.01) or inhibited (*, p < 0.05) (B–D, n = 3).
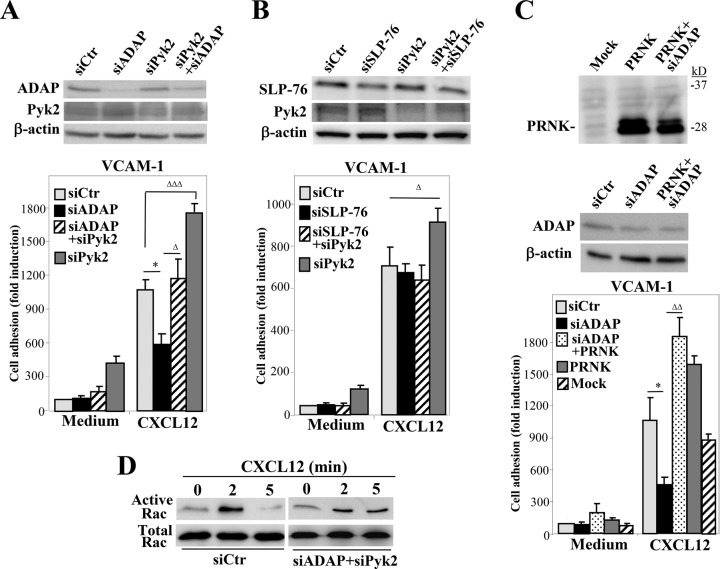
As the adhesion data indicate that T-cell attachment involving α4β1 is especially altered upon knocking down ADAP or Pyk2, we next analyzed the effect of their combined depletion on cell adhesion to VCAM-1. Results revealed that the inefficient stimulation of adhesion displayed by ADAP-silenced cells could be rescued by Pyk2 siRNA cotransfection, although the double-knockdown transfectants did not reach the adhesion values exhibited by cells transfected with Pyk2 siRNA alone (Figure 7A). In addition, the up-regulated cell attachment following Pyk2 silencing was not detected when both SLP-76 and Pyk2 were knocked down (Figure 7B), suggesting that cooperative responses between ADAP and SLP-76 exist during their positive regulation of α4β1-dependent T-cell adhesion. Similar to ADAP-Pyk2 siRNA cotransfection, PRNK transfection together with ADAP siRNA led to a recovery of adhesion to VCAM-1/CXCL12 (Figure 7C). Notably, the rescue in adhesion shown by cells knocked down for both ADAP and Pyk2 correlated with longer, sustained Rac1 activation in these transfectants (Figure 7D). The small decrease in activation of Rac at early time points might reflect initial inhibition due to ADAP depletion that is later recovered by siPyk2 silencing. These results suggest that the stimulatory actions of ADAP and the inhibitory activity of Pyk2 during CXCL12-activated adhesion mediated by α4β1 are connected through Rac1.
FIGURE 7:
Analysis of functional connections between ADAP and Pyk2 in CXCL12-stimulated adhesion mediated by α4β1. (A–C) Molt-4 cells were transfected with the indicated individual or combined siRNA and/or vectors, and transfectants were analyzed by immunoblotting or in adhesion assays to VCAM-1 immobilized with or without CXCL12. Adhesions were significantly inhibited (*p < 0.05) or significantly stimulated or rescued (ΔΔΔ, p < 0.001; ΔΔ, p < 0.01; or Δ, p < 0.05) (A, n = 4; B and C, n = 3). (D) The indicated transfectants were tested in GTPase assays to detect active Rac.
DISCUSSION
Vav1 is needed for chemokine-stimulated T-cell adhesion involving α4β1 (Garcia-Bernal et al., 2005, 2009), and thus it is conceivable that Vav1-binding proteins such as SLP-76 and Pyk2 could modulate this process. Using Molt-4 and primary human PBL-T depleted for SLP-76 or for ADAP, an SLP-76–interacting partner, as well as SLP-76–deficient J14 cells, we show here that these two proteins contribute to optimal stimulation by CXCL12 of α4β1-mediated T-cell attachment under flow conditions. Instead, Pyk2 negatively regulates this adhesion, as its depletion or expression of kinase-inactive Pyk2 forms led to increased cell attachment. Of note, modulation of cell adhesion by SLP-76, ADAP, and Pyk2 correlated with a chemokine-stimulated transient increase in association of these proteins with Vav1, strongly suggesting that the dynamics of this protein module can regulate the extent of α4β1-mediated T-cell attachment.
Alterations in adhesion to α4β1 ligands exhibited by SLP-76–, ADAP-, or Pyk2-depleted cells were independent of changes in α4β1 affinity following CXCL12 stimulation. Rather, flow-chamber adhesion assays revealed that the strengthening of α4β1-dependent attachment was regulated by SLP-76 and ADAP, though the strength of ADAP-knockdown transfectant attachment was consistently lower than SLP-76–depleted counterparts. Moreover, we show that spreading on VCAM-1/CXCL12 was similarly decreased in T-cells silenced for SLP-76 and ADAP. Compared with the absolute requirement of talin and Vav1 in the control of α4β1 affinity and strength of chemokine-activated T-cell attachment (Garcia-Bernal et al., 2006, 2009; Manevich et al., 2007), the contribution of SLP-76 and ADAP in these processes appears to be of lesser magnitude. However, both adaptors are clearly needed to achieve optimal stability of this adhesion.
VCAM-1-Fc soluble binding to Pyk2-silenced cells was maintained longer than with control transfectants, suggesting that Pyk2 can regulate the avidity of α4β1. Sustained VCAM-1 binding likely represents one of the mechanisms accounting for the stronger cell arrest and resistance to detachment under shear stress shown by Pyk2-knockdown cells relative to control counterparts. Associated with increased strength of attachment to VCAM-1/CXCL12 exhibited by Pyk2-depleted cells, they displayed enhanced spreading, an observation in line with previous results showing high spreading correlating with defective cell detachment of Pyk2−/− macrophages (Okigaki et al., 2003). Therefore our data indicate that Pyk2 negatively regulates the strength and spreading phases of chemokine-activated T-cell attachment involving α4β1.
Our previous results revealed that Vav1-mediated Rac1 activation in response to CXCL12 in T-cells represents an important event for strengthening of α4β1-VCAM-1 interaction, as well as for cell spreading (Garcia-Bernal et al., 2005). The present data indicate that Rac1 is a common mediator of SLP-76–, ADAP-, and Pyk2-modulated T-cell adhesion to α4β1 ligands. Thus SLP-76 and ADAP likely function as adaptors with Vav1 to optimize chemokine-promoted Rac activation and stimulation of α4β1-dependent cell adhesion. Instead, Pyk2 restricts this attachment by limiting Rac activation. As knocking down any of these three proteins influences the outcome of Rac activation, the data strongly suggest that depletion-mediated changes in the composition of the complex formed by Vav1, SLP-76, ADAP, and Pyk2 could account for alterations in Rac activation. In this regard, we show that knocking down ADAP delays Vav1-SLP-76 association and impairs Rac activation in response to CXCL12, suggesting that the Vav1-SLP-76-ADAP module facilitates Rac1 activation upon chemokine stimulation and optimizes the strength and spreading of α4β1-mediated T-cell adhesion. On this point, SLP-76 assembly with Vav1 and Nck contributes to TCR-stimulated Rac1 activation (Bubeck Wardenburg et al., 1998; Barda-Saad et al., 2010), and importantly, ADAP associates with these complexes together with WASP (Krause et al., 2000; Coppolino et al., 2001) and cooperates with Nck in the recruitment of SLP-76 for regulation of Rho GTPase–mediated actin rearrangement (Pauker et al., 2011). Earlier results reported that SLP-76 depletion in Jurkat cells did not affect the CXCL12-stimulated Rac1 activation (Horn et al., 2009). The basis for the discrepancies between these results and our observation of inefficient Rac1 activation by CXCL12 in SLP-76–deficient cells is not known at present.
Characterization of the mechanics of Pyk2-regulated, α4β1-mediated cell attachment reveals that the Pyk2 kinase domain negatively controls this adhesion. Thus expression of PRNK, which lacks the Pyk2 kinase domain, as well as of a point mutant, kinase-inactive Pyk2 form, mimicked the effects of Pyk2 depletion on the increase in α4β1-mediated attachment, strongly suggesting that Pyk2-dependent tyrosine phosphorylation activity in T-cells contributes to keep Rac1 activation levels under control. Of note, negative regulation of Rac activation and cell adhesion to VCAM-1 by Pyk2 directly opposed the stimulatory activity of ADAP in these processes, as detected in cells silenced for both proteins. In this experimental setting, the absence of Pyk2 might rescue the activation of Rac1 concurrent with the inhibitory effects of ADAP knockdown, leading to stimulation of adhesion to VCAM-1, but we cannot exclude that recovery of these activities occurs subsequent to the negative actions of ADAP depletion. Our data are in line with early work proposing that Pyk2 regulates Rac1 activation (Tokiwa et al., 1996) and with results showing that macrophages from mice knocked out for the Pyk2 closely related kinase FAK have elevated Rac activation (Owen et al., 2007). However, the level of activation of this GTPase based on Pyk2 activity might be cell-context dependent (Chapman and Houtman, 2014), as NK cells expressing a kinase-dead Pyk2 displayed reduced Rac activation (Gismondi et al., 2003).
CXCL12 promotes tyrosine phosphorylation of Vav1 associated with Rac1 activation and stimulation of α4β1-mediated T-cell adhesion (Garcia-Bernal et al., 2005). Inefficient phosphorylation of Vav1 in response to CXCL12 in SLP-76–depleted T-cells correlated with their impaired Rac activation, whereas ADAP-knockdown cells displayed fully competent chemokine-promoted phosphorylation of this GEF, in spite of their defective Rac activation. These results might reflect the fact that ADAP does not directly interact with Vav1 but associates with the Vav1-SLP-76 module, possibly in a SLP-76–dependent manner. Interestingly, the increased Rac activation following CXCL12 stimulation in Pyk2-silenced cells was not coupled with enhanced Vav1 phosphorylation, but rather phosphorylation was not as efficient as in control transfectants. These data reveal that SLP-76 and Pyk2 but not ADAP contribute to optimal Vav1 tyrosine phosphorylation in T-cells in response to CXCL12, and indicate that net Rac activation is the result of Vav1-dependent and Vav1-independent mechanisms. It remains puzzling that inefficient Vav1 phosphorylation in SLP-76– and Pyk2-depleted T-cells led to opposite effects on Rac activation. The molecular basis underlying these distinct responses is not known at present. SLP-76 and Pyk2 knockdown might similarly affect recruitment and/or activity of tyrosine kinases involved in Vav1 tyrosine phosphorylation, such as ZAP-70 and Lck (Gulbins et al., 1993; Ottoson et al., 2001). But it is also clear that the subsequent Rac activation is differentially regulated and likely involves distinct GEFs and GTPase-activating proteins (GAPs). GEFs and GAPs that are known to regulate CXCL12-stimulated Rac activation and α4β1-mediated T-cell adhesion include the GEF DOCK2 and the GAP β-chimerin. Of note, lymphocytes from DOCK2 knockout mice display minimal chemokine-induced Rac activation compared with control cells (Fukui et al., 2001). Furthermore, we showed that DOCK2 cooperates with Vav1 for CXCL12-promoted Rac activation, and its silencing inhibits T-cell attachment mediated by α4β1 under flow conditions (Garcia-Bernal et al., 2006). Instead, β-chimerin overexpression in T-cells results in impaired Rac activation in response to CXCL12 associated with inhibition of α4β1-dependent T-cell adhesion (Siliceo et al., 2006). Although it has not yet been tested, the possibility exists that DOCK2 and β-chimerin might be recruited to the Vav1-Pyk2-SLP-76-ADAP module, thus contributing to the regulation of Rac activity and subsequent cell attachment mediated by α4β1. Other GEFs and GAPs expressed on T-lymphocytes that are known to regulate Rac activation include PlekhG2, Pix, and ArhGAP15 (Seoh et al., 2003; Phee et al., 2005; Runne and Chen, 2013), though their role in the control of α4β1-dependent T-cell adhesion has not been addressed.
SKAP-55 is an additional molecule that could modulate ADAP-controlled T-cell attachment to α4β1 ligands following chemokine stimulation. Thus ADAP binds SKAP-55, an adaptor protein that mediates up-regulation of T-cell adhesion to ICAM-1 and fibronectin upon TCR activation (Marie-Cardine et al., 1998; Wang et al., 2003, 2007). The ADAP-SKAP-55 module interacts with RIAM through SKAP-55, facilitating TCR-mediated integrin activation (Kliche et al., 2006; Menasche et al., 2007; Horn et al., 2009). Furthermore, independent ADAP-SKAP-55 modules regulate CCR7-mediated αLβ2 functions in T-cells (Kliche et al., 2012), and recent data indicate that ADAP can also bind talin and kindlin to promote integrin-αIIbβ3 activation and fibrinogen binding (Kasirer-Friede et al., 2014). Whether the ADAP-SKAP-55 association contributes to chemokine-stimulated Rac activation and T-cell adhesion involving α4β1 is currently unknown.
A model can be proposed from the present results (Figure 8). The net Rac1 activation levels needed to develop sufficient adhesion strength and cell spreading mediated by α4β1 are possibly the result of opposing activities inside the Vav1-SLP-76-ADAP-Pyk2 module. Thus Pyk2 kinase activity would oppose the Rac1 activation stimulated by Vav1-SLP-76-ADAP and possibly additional costimulatory molecules, potentially by activating a GAP or by inhibiting a GEF for Rac1. Talin can also be found in this protein module, but its role in the stability and function of the complex and whether it represents a different pool from β1-associated talin has not been addressed here. Furthermore, the present data, together with our previous results (Garcia-Bernal et al., 2009), raise the possibility that separate pools of SLP-76 with either Vav1 or talin might exist. Our data should add molecular mechanistic insights into the process of chemokine-stimulated T-cell adhesion mediated by α4β1, a key step during T-lymphocyte trafficking to sites of inflammation.
FIGURE 8:
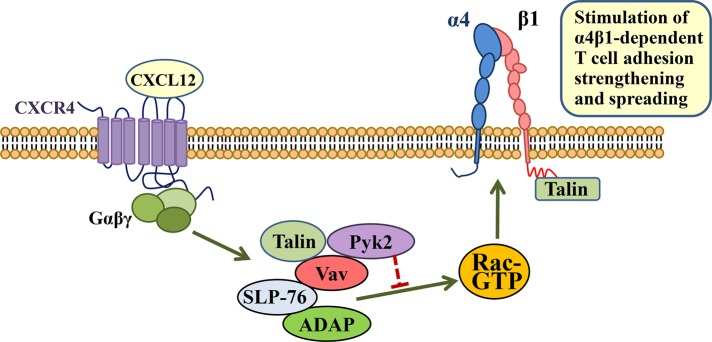
Model for regulation by Vav1-associated proteins of chemokine-stimulated, α4β1-dependent T-cell adhesion. Rac1 activation by CXCL12 is a well-known event for stimulation of the adhesion strength and spreading mediated by α4β1 in T-cells. A transient complex formed by Vav1, SLP-76, ADAP, and Pyk2 regulates this adhesion. While Vav1-SLP-76-ADAP stimulates chemokine-promoted Rac1 activation (green arrow), the presence of Pyk2 in the complex opposes this activation (red blocking symbol). The result of these opposing actions is a net Rac activation and up-regulation of T-cell adhesion strengthening and spreading dependent on α4β1. Talin can also be found in this complex, perhaps as an independent pool from β1-associated talin. Its role in the assembly and function of this complex has not been addressed in the present study.
MATERIALS AND METHODS
Cells, antibodies, and reagents
The human Molt-4 and Jurkat T-cell lines were cultured in RPMI 1640 medium (Lonza, Verviers, Belgium) and 10% fetal bovine serum (Life Technologies, Paisley, UK) (complete medium). The Jurkat-derived cell lines JCaM1.6 (American Type Culture Collection) and J14 (a gift from Arthur Weiss, University of California, San Francisco, CA) were grown in complete medium. Peripheral blood T-lymphocytes (PBL-T) were prepared as previously described (Garcia-Bernal et al., 2005). The Consejo Superior de Investigaciones Científicas Ethics Committee (Madrid, Spain) approved the protocols used to obtain and process the human blood samples. Control P3×63 and anti–β1 TS2/16 mAb were gifts from Francisco Sánchez-Madrid (Hospital de la Princesa, Madrid, Spain), and polyclonal anti–β1A antibodies were from Guido Tarone (Turin University, Italy). Antibodies to SLP-76 and Vav1 (C14) were from Santa Cruz Biotechnology (Santa Cruz, CA), anti–phospho-Vav (Y174; ab47282) was from Abcam (Cambridge, MA), anti-Rac1 and anti-ADAP were from BD Biosciences (San Diego, CA), and anti-GFP was from Molecular Probes (Eugene, OR). Antibodies to β-actin, vinculin, and talin clone 8D4 were from Sigma-Aldrich (St. Louis, MO), and antibody to Pyk2 was from Millipore (Billerica, MA). The HUTS-21 anti–β1 mAb has been previously characterized (Luque et al., 1996). CXCL12 was purchased from R&D Systems (Minneapolis, MN) and CCL21 was purchased from Peprotech (Rocky Hill, NJ).
Transfections and RNA interference
Vectors coding for Rac wild-type (wt), Rac V12, Rac N17, Pyk2-Y402F, Pyk2-K457A, and Pyk2-wt fused with GFP were provided by Francisco Sánchez-Madrid. The vector pRK5-myc-PRNK (Ivankovic-Dikic et al., 2000) was a gift from Ivan Dikic (Goethe University School of Medicine, Frankfurt, Germany). The siRNA against human SLP-76 (ON-TARGETplus, SMARTpool) was purchased from Dharmacon (Lafayette, CO). The siRNAs for ADAP-1, sense: CAAAUGUUGACCUGACGAAtt; ADAP-2, sense: GAGAUUUCAAGUUUGCCCUtt; Pyk2, sense: GAUGUGGUCCUGAAUCGUAtt; and control siRNA (Garcia-Bernal et al., 2005) were purchased from Sigma-Aldrich. Vectors and siRNA were nucleofected (Amaxa, Cologne, Germany) following a previously described procedure (Garcia-Bernal et al., 2005). Molt-4 or PBL-T siRNA transfectants were assayed 22–24 h posttransfection, and transfection did not affect cell viability, as assessed in cell cycle analyses by flow cytometry.
Cell adhesion and spreading assays
For static adhesions, we used the reported method (Garcia-Bernal et al., 2005). Briefly, cells were plated in triplicate on wells coated with the fibronectin fragment FN-H89, which contains the CS-1 region, or with VCAM-1 together with CXCL12 or CCL21 (650 ng/ml), and plates were incubated and adhesion measured. Adhesion data are presented relative to control untreated cells, which have been given an arbitrary value of 100. For flow-chamber adhesion assays, we followed the described protocol (Garcia-Bernal et al., 2006). In brief, cells were infused at a flow rate of 1 dyne/cm2 into flow chambers containing coimmobilized VCAM-1 and CXCL12. Rolling cells that subsequently firmly attached were expressed as stable arrest, whereas tethering cells that did not arrest at any moment were expressed as rolling cells. For evaluation of shear resistance, cells were allowed to attach and then were subjected to sequential increases of the flow rate. The number of cells remaining bound was determined as the percentage of total adhered cells after the adhesion step. For cellular spreading, CXCL12-stimulated cells attached on VCAM-1 were fixed and analyzed as previously described (Garcia-Bernal et al., 2006).
Soluble VCAM-1 binding and flow cytometry
For cell binding to VCAM-1-Fc (R&D Systems), cells were stimulated for 20 s with CXCL12 before VCAM-1-Fc was added, which was detected by flow cytometry using phycoerythrin-conjugated AffiniPure F(ab′)2 fragment goat anti–human immunoglobulin G (IgG), Fcγ-fragment specific (Jackson ImmunoResearch, West Grove, PA). For the detection of high-affinity β1, cells were stimulated for 20 s with CXCL12 before HUTS-21 anti–β1 mAb (10 μg/ml) was added for 30 min at 4ºC. After washing, cells were incubated with FITC-conjugated rabbit anti–mouse IgG (Jackson Immunoresearch). Fluorescence intensity data indicate fold-induction values relative to those from control untreated cells, which were given an arbitrary value of one.
Immunoprecipitation, immunoblotting, and GTPase assays
For immunoprecipitation, we followed the described method (Garcia-Bernal et al., 2009). In brief, cell lysate supernatants were incubated with antibodies followed by coupling to protein G-Sepharose. Proteins were separated by SDS–PAGE and transferred to membranes that were sequentially incubated with primary antibodies and with horseradish peroxidase–conjugated secondary antibodies. Protein visualization was achieved using Immobilon Western chemiluminiscent substrate (Millipore). For GTPase assays, we used the reported method (Garcia-Bernal et al., 2005). Briefly, cells exposed to CXCL12 were lysed, and aliquots from extracts were separated for total lysate controls and for incubation with GST-PAK-CD fusion protein, followed by incubation with glutathione-Sepharose beads (GE Healthcare, Piscataway, NJ). Bound proteins were eluted and subjected to immunoblotting using anti-Rac1 antibodies.
Statistical analyses
Data were analyzed by one-way analysis of variance, followed by Tukey-Kramer multiple comparisons. In both analyses, the minimum acceptable level of significance was p < 0.05.
Supplementary Material
Acknowledgments
We thank Balbino Alarcón, Aldo Borroto, José Luis Rodríguez-Fernández, Danay Cibrian, and Angeles García-Pardo for reagents, technical help, and helpful discussions; and Pedro Lastres for help in flow cytometry experiments. This work was supported by grants SAF2011-24022 from Ministerio de Economía y Competitividad, RD12/0036/0061, and S2010/BMD-2314 from Comunidad de Madrid to J.T.
Abbreviations used:
- GAP
GTPase-activating protein
- GEF
guanine nucleotide exchange factor
- GFP
green fluorescent protein
- IgG
immunoglobulin G
- mAb
monoclonal antibody
- PRNK
Pyk2-related nonkinase
- PTx
pertussis toxin
- siRNA
small interfering RNA
- TCR
T-cell receptor.
Footnotes
This article was published online ahead of print in MBoC in Press (http://www.molbiolcell.org/cgi/doi/10.1091/mbc.3215) on July 22, 2015.
REFERENCES
- Avraham H, Park SY, Schinkmann K, Avraham S. RAFTK/Pyk2-mediated cellular signalling. Cell Signal. 2000;12:123–133. doi: 10.1016/s0898-6568(99)00076-5. [DOI] [PubMed] [Google Scholar]
- Avraham S, London R, Fu Y, Ota S, Hiregowdara D, Li J, Jiang S, Pasztor LM, White RA, Groopman JE, et al. Identification and characterization of a novel related adhesion focal tyrosine kinase (RAFTK) from megakaryocytes and brain. J Biol Chem. 1995;270:27742–27751. doi: 10.1074/jbc.270.46.27742. [DOI] [PubMed] [Google Scholar]
- Baker RG, Hsu CJ, Lee D, Jordan MS, Maltzman JS, Hammer DA, Baumgart T, Koretzky GA. The adapter protein SLP-76 mediates “outside-in” integrin signaling and function in T cells. Mol Cell Biol. 2009;29:5578–5589. doi: 10.1128/MCB.00283-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barda-Saad M, Shirasu N, Pauker MH, Hassan N, Perl O, Balbo A, Yamaguchi H, Houtman JC, Appella E, Schuck P, Samelson LE. Cooperative interactions at the SLP-76 complex are critical for actin polymerization. EMBO J. 2010;29:2315–2328. doi: 10.1038/emboj.2010.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beinke S, Phee H, Clingan JM, Schlessinger J, Matloubian M, Weiss A. Proline-rich tyrosine kinase-2 is critical for CD8 T-cell short-lived effector fate. Proc Natl Acad Sci USA. 2010;107:16234–16239. doi: 10.1073/pnas.1011556107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berlin-Rufenach C, Otto F, Mathies M, Westermann J, Owen MJ, Hamann A, Hogg N. Lymphocyte migration in lymphocyte function-associated antigen (LFA)-1-deficient mice. J Exp Med. 1999;189:1467–1478. doi: 10.1084/jem.189.9.1467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boerth NJ, Judd BA, Koretzky GA. Functional association between SLAP-130 and SLP-76 in Jurkat T cells. J Biol Chem. 2000;275:5143–5152. doi: 10.1074/jbc.275.7.5143. [DOI] [PubMed] [Google Scholar]
- Bolomini-Vittori M, Montresor A, Giagulli C, Staunton D, Rossi B, Martinello M, Constantin G, Laudanna C. Regulation of conformer-specific activation of the integrin LFA-1 by a chemokine-triggered Rho signaling module. Nat Immunol. 2009;10:185–194. doi: 10.1038/ni.1691. [DOI] [PubMed] [Google Scholar]
- Bubeck Wardenburg J, Pappu R, Bu JY, Mayer B, Chernoff J, Straus D, Chan AC. Regulation of PAK activation and the T cell cytoskeleton by the linker protein SLP-76. Immunity. 1998;9:607–616. doi: 10.1016/s1074-7613(00)80658-5. [DOI] [PubMed] [Google Scholar]
- Bustelo XR. Regulatory and signaling properties of the Vav family. Mol Cell Biol. 2000;20:1461–1477. doi: 10.1128/mcb.20.5.1461-1477.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calderwood DA, Campbell ID, Critchley DR. Talins and kindlins: partners in integrin-mediated adhesion. Nat Rev Mol Cell Biol. 2013;14:503–517. doi: 10.1038/nrm3624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman NM, Houtman JC. Functions of the FAK family kinases in T cells: beyond actin cytoskeletal rearrangement. Immunol Res. 2014;59:23–34. doi: 10.1007/s12026-014-8527-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coppolino MG, Krause M, Hagendorff P, Monner DA, Trimble W, Grinstein S, Wehland J, Sechi AS. Evidence for a molecular complex consisting of Fyb/SLAP, SLP-76, Nck, VASP and WASP that links the actin cytoskeleton to Fcγ receptor signalling during phagocytosis. J Cell Sci. 2001;114:4307–4318. doi: 10.1242/jcs.114.23.4307. [DOI] [PubMed] [Google Scholar]
- da Silva AJ, Li Z, de Vera C, Canto E, Findell P, Rudd CE. Cloning of a novel T-cell protein FYB that binds FYN and SH2-domain-containing leukocyte protein 76 and modulates interleukin 2 production. Proc Natl Acad Sci USA. 1997;94:7493–7498. doi: 10.1073/pnas.94.14.7493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis CB, Dikic I, Unutmaz D, Hill CM, Arthos J, Siani MA, Thompson DA, Schlessinger J, Littman DR. Signal transduction due to HIV-1 envelope interactions with chemokine receptors CXCR4 or CCR5. J Exp Med. 1997;186:1793–1798. doi: 10.1084/jem.186.10.1793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dikic I, Tokiwa G, Lev S, Courtneidge SA, Schlessinger J. A role for Pyk2 and Src in linking G-protein-coupled receptors with MAP kinase activation. Nature. 1996;383:547–550. doi: 10.1038/383547a0. [DOI] [PubMed] [Google Scholar]
- Feigelson SW, Grabovsky V, Winter E, Chen LL, Pepinsky RB, Yednock T, Yablonski D, Lobb R, Alon R. The Src kinase p56(lck) up-regulates VLA-4 integrin affinity. Implications for rapid spontaneous and chemokine-triggered T cell adhesion to VCAM-1 and fibronectin. J Biol Chem. 2001;276:13891–13901. doi: 10.1074/jbc.M004939200. [DOI] [PubMed] [Google Scholar]
- Fischer KD, Kong YY, Nishina H, Tedford K, Marengere LE, Kozieradzki I, Sasaki T, Starr M, Chan G, Gardener S, et al. Vav is a regulator of cytoskeletal reorganization mediated by the T-cell receptor. Curr Biol. 1998;8:554–562. doi: 10.1016/s0960-9822(98)70224-6. [DOI] [PubMed] [Google Scholar]
- Fukui Y, Hashimoto O, Sanui T, Oono T, Koga H, Abe M, Inayoshi A, Noda M, Oike M, Shirai T, Sasazuki T. Haematopoietic cell-specific CDM family protein DOCK2 is essential for lymphocyte migration. Nature. 2001;412:826–831. doi: 10.1038/35090591. [DOI] [PubMed] [Google Scholar]
- Ganju RK, Hatch WC, Avraham H, Ona MA, Druker B, Avraham S, Groopman JE. RAFTK, a novel member of the focal adhesion kinase family, is phosphorylated and associates with signaling molecules upon activation of mature T lymphocytes. J Exp Med. 1997;185:1055–1063. doi: 10.1084/jem.185.6.1055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao C, Blystone SD. A Pyk2-Vav1 complex is recruited to β3-adhesion sites to initiate Rho activation. Biochem J. 2009;420:49–56. doi: 10.1042/BJ20090037. [DOI] [PubMed] [Google Scholar]
- Garcia-Bernal D, Parmo-Cabanas M, Dios-Esponera A, Samaniego R, Hernan PdlOD, Teixido J. Chemokine-induced Zap70 kinase-mediated dissociation of the Vav1-talin complex activates α4β1 integrin for T cell adhesion. Immunity. 2009;31:953–964. doi: 10.1016/j.immuni.2009.09.021. [DOI] [PubMed] [Google Scholar]
- Garcia-Bernal D, Sotillo-Mallo E, Nombela-Arrieta C, Samaniego R, Fukui Y, Stein JV, Teixido J. DOCK2 is required for chemokine-promoted human T lymphocyte adhesion under shear stress mediated by the integrin α4β1. J Immunol. 2006;177:5215–5225. doi: 10.4049/jimmunol.177.8.5215. [DOI] [PubMed] [Google Scholar]
- Garcia-Bernal D, Wright N, Sotillo-Mallo E, Nombela-Arrieta C, Stein JV, Bustelo XR, Teixido J. Vav1 and Rac control chemokine-promoted T lymphocyte adhesion mediated by the integrin α4β1. Mol Biol Cell. 2005;16:3223–3235. doi: 10.1091/mbc.E04-12-1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geng L, Raab M, Rudd CE. Cutting edge: SLP-76 cooperativity with FYB/FYN-T in the up-regulation of TCR-driven IL-2 transcription requires SLP-76 binding to FYB at Tyr595 and Tyr651. J Immunol. 1999;163:5753–5757. [PubMed] [Google Scholar]
- Ghandour H, Cullere X, Alvarez A, Luscinskas FW, Mayadas TN. Essential role for Rap1 GTPase and its guanine exchange factor CalDAG-GEFI in LFA-1 but not VLA-4 integrin mediated human T-cell adhesion. Blood. 2007;110:3682–3690. doi: 10.1182/blood-2007-03-077628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gismondi A, Jacobelli J, Strippoli R, Mainiero F, Soriani A, Cifaldi L, Piccoli M, Frati L, Santoni A. Proline-rich tyrosine kinase 2 and Rac activation by chemokine and integrin receptors controls NK cell transendothelial migration. J Immunol. 2003;170:3065–3073. doi: 10.4049/jimmunol.170.6.3065. [DOI] [PubMed] [Google Scholar]
- Grabovsky V, Feigelson S, Chen C, Bleijs DA, Peled A, Cinamon G, Baleux F, Arenzana-Seisdedos F, Lapidot T, van Kooyk Y, et al. Subsecond induction of α4 integrin clustering by immobilized chemokines stimulates leukocyte tethering and rolling on endothelial vascular cell adhesion molecule 1 under flow conditions. J Exp Med. 2000;192:495–506. doi: 10.1084/jem.192.4.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths EK, Krawczyk C, Kong YY, Raab M, Hyduk SJ, Bouchard D, Chan VS, Kozieradzki I, Oliveira-Dos-Santos AJ, Wakeham A, et al. Positive regulation of T cell activation and integrin adhesion by the adapter Fyb/Slap. Science. 2001;293:2260–2263. doi: 10.1126/science.1063397. [DOI] [PubMed] [Google Scholar]
- Griffiths EK, Penninger JM. Communication between the TCR and integrins: role of the molecular adapter ADAP/Fyb/Slap. Curr Opin Immunol. 2002;14:317–322. doi: 10.1016/s0952-7915(02)00334-5. [DOI] [PubMed] [Google Scholar]
- Gulbins E, Coggeshall KM, Baier G, Katzav S, Burn P, Altman A. Tyrosine kinase-stimulated guanine nucleotide exchange activity of Vav in T cell activation. Science. 1993;260:822–825. doi: 10.1126/science.8484124. [DOI] [PubMed] [Google Scholar]
- Hogg N, Patzak I, Willenbrock F. The insider’s guide to leukocyte integrin signalling and function. Nat Rev Immunol. 2011;11:416–426. doi: 10.1038/nri2986. [DOI] [PubMed] [Google Scholar]
- Horn J, Wang X, Reichardt P, Stradal TE, Warnecke N, Simeoni L, Gunzer M, Yablonski D, Schraven B, Kliche S. Src homology 2-domain containing leukocyte-specific phosphoprotein of 76 kDa is mandatory for TCR-mediated inside-out signaling, but dispensable for CXCR4-mediated LFA-1 activation, adhesion, and migration of T cells. J Immunol. 2009;183:5756–5767. doi: 10.4049/jimmunol.0900649. [DOI] [PubMed] [Google Scholar]
- Ivankovic-Dikic I, Gronroos E, Blaukat A, Barth BU, Dikic I. Pyk2 and FAK regulate neurite outgrowth induced by growth factors and integrins. Nat Cell Biol. 2000;2:574–581. doi: 10.1038/35023515. [DOI] [PubMed] [Google Scholar]
- Kasirer-Friede A, Kang J, Kahner B, Ye F, Ginsberg MH, Shattil SJ. ADAP interactions with talin and kindlin promote platelet integrin αIIbβ3 activation and stable fibrinogen binding. Blood. 2014;123:3156–3165. doi: 10.1182/blood-2013-08-520627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katagiri K, Maeda A, Shimonaka M, Kinashi T. RAPL, a Rap1-binding molecule that mediates Rap1-induced adhesion through spatial regulation of LFA-1. Nat Immunol. 2003;4:741–748. doi: 10.1038/ni950. [DOI] [PubMed] [Google Scholar]
- Katagiri T, Takahashi T, Sasaki T, Nakamura S, Hattori S. Protein-tyrosine kinase Pyk2 is involved in interleukin-2 production by Jurkat T cells via its tyrosine 402. J Biol Chem. 2000;275:19645–19652. doi: 10.1074/jbc.M909828199. [DOI] [PubMed] [Google Scholar]
- Kim C, Ye F, Ginsberg MH. Regulation of integrin activation. Annu Rev Cell Dev Biol. 2011;27:321–345. doi: 10.1146/annurev-cellbio-100109-104104. [DOI] [PubMed] [Google Scholar]
- Kliche S, Breitling D, Togni M, Pusch R, Heuer K, Wang X, Freund C, Kasirer-Friede A, Menasche G, Koretzky GA, Schraven B. The ADAP/SKAP55 signaling module regulates T-cell receptor-mediated integrin activation through plasma membrane targeting of Rap1. Mol Cell Biol. 2006;26:7130–7144. doi: 10.1128/MCB.00331-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kliche S, Worbs T, Wang X, Degen J, Patzak I, Meineke B, Togni M, Moser M, Reinhold A, Kiefer F, et al. CCR7-mediated LFA-1 functions in T cells are regulated by 2 independent ADAP/SKAP55 modules. Blood. 2012;119:777–785. doi: 10.1182/blood-2011-06-362269. [DOI] [PubMed] [Google Scholar]
- Koretzky GA, Abtahian F, Silverman MA. SLP76 and SLP65: complex regulation of signalling in lymphocytes and beyond. Nat Rev Immunol. 2006;6:67–78. doi: 10.1038/nri1750. [DOI] [PubMed] [Google Scholar]
- Krause M, Sechi AS, Konradt M, Monner D, Gertler FB, Wehland J. Fyn-binding protein (Fyb)/SLP-76-associated protein (SLAP), Ena/vasodilator-stimulated phosphoprotein (VASP) proteins and the Arp2/3 complex link T cell receptor (TCR) signaling to the actin cytoskeleton. J Cell Biol. 2000;149:181–194. doi: 10.1083/jcb.149.1.181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee D, Kim J, Baker RG, Koretzky GA, Hammer DA. SLP-76 is required for optimal CXCR4-stimulated T lymphocyte firm arrest to ICAM-1 under shear flow. Eur J Immunol. 2012;42:2736–2743. doi: 10.1002/eji.201142303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lev S, Moreno H, Martinez R, Canoll P, Peles E, Musacchio JM, Plowman GD, Rudy B, Schlessinger J. Protein tyrosine kinase PYK2 involved in Ca2+-induced regulation of ion channel and MAP kinase functions. Nature. 1995;376:737–745. doi: 10.1038/376737a0. [DOI] [PubMed] [Google Scholar]
- Ley K, Laudanna C, Cybulsky MI, Nourshargh S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol. 2007;7:678–689. doi: 10.1038/nri2156. [DOI] [PubMed] [Google Scholar]
- Li J, Avraham H, Rogers RA, Raja S, Avraham S. Characterization of RAFTK, a novel focal adhesion kinase, and its integrin-dependent phosphorylation and activation in megakaryocytes. Blood. 1996;88:417–428. [PubMed] [Google Scholar]
- Li X, Dy RC, Cance WG, Graves LM, Earp HS. Interactions between two cytoskeleton-associated tyrosine kinases: calcium-dependent tyrosine kinase and focal adhesion tyrosine kinase. J Biol Chem. 1999;274:8917–8924. doi: 10.1074/jbc.274.13.8917. [DOI] [PubMed] [Google Scholar]
- Luque A, Gomez M, Puzon W, Takada Y, Sanchez-Madrid F, Cabanas C. Activated conformations of very late activation integrins detected by a group of antibodies (HUTS) specific for a novel regulatory region (355–425) of the common β1 chain. J Biol Chem. 1996;271:11067–11075. doi: 10.1074/jbc.271.19.11067. [DOI] [PubMed] [Google Scholar]
- Luster AD, Alon R, von Andrian UH. Immune cell migration in inflammation: present and future therapeutic targets. Nat Immunol. 2005;6:1182–1190. doi: 10.1038/ni1275. [DOI] [PubMed] [Google Scholar]
- Malinin NL, Zhang L, Choi J, Ciocea A, Razorenova O, Ma YQ, Podrez EA, Tosi M, Lennon DP, Caplan AI, et al. A point mutation in KINDLIN3 ablates activation of three integrin subfamilies in humans. Nat Med. 2009;15:313–318. doi: 10.1038/nm.1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manevich E, Grabovsky V, Feigelson SW, Alon R. Talin 1 and paxillin facilitate distinct steps in rapid VLA-4-mediated adhesion strengthening to vascular cell adhesion molecule 1. J Biol Chem. 2007;282:25338–25348. doi: 10.1074/jbc.M700089200. [DOI] [PubMed] [Google Scholar]
- Manevich-Mendelson E, Feigelson SW, Pasvolsky R, Aker M, Grabovsky V, Shulman Z, Kilic SS, Rosenthal-Allieri MA, Ben-Dor S, Mory A, et al. Loss of kindlin-3 in LAD-III eliminates LFA-1 but not VLA-4 adhesiveness developed under shear flow conditions. Blood. 2009;114:2344–2353. doi: 10.1182/blood-2009-04-218636. [DOI] [PubMed] [Google Scholar]
- Marie-Cardine A, Hendricks-Taylor LR, Boerth NJ, Zhao H, Schraven B, Koretzky GA. Molecular interaction between the Fyn-associated protein SKAP55 and the SLP-76-associated phosphoprotein SLAP-130. J Biol Chem. 1998;273:25789–25795. doi: 10.1074/jbc.273.40.25789. [DOI] [PubMed] [Google Scholar]
- Menasche G, Kliche S, Chen EJ, Stradal TE, Schraven B, Koretzky G. RIAM links the ADAP/SKAP-55 signaling module to Rap1, facilitating T-cell-receptor-mediated integrin activation. Mol Cell Biol. 2007;27:4070–4081. doi: 10.1128/MCB.02011-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitra SK, Hanson DA, Schlaepfer DD. Focal adhesion kinase: in command and control of cell motility. Nat Rev Mol Cell Biol. 2005;6:56–68. doi: 10.1038/nrm1549. [DOI] [PubMed] [Google Scholar]
- Moser M, Bauer M, Schmid S, Ruppert R, Schmidt S, Sixt M, Wang HV, Sperandio M, Fassler R. Kindlin-3 is required for β2 integrin-mediated leukocyte adhesion to endothelial cells. Nat Med. 2009a;15:300–305. doi: 10.1038/nm.1921. [DOI] [PubMed] [Google Scholar]
- Moser M, Legate KR, Zent R, Fassler R. The tail of integrins, talin, and kindlins. Science. 2009b;324:895–899. doi: 10.1126/science.1163865. [DOI] [PubMed] [Google Scholar]
- Musci MA, Hendricks-Taylor LR, Motto DG, Paskind M, Kamens J, Turck CW, Koretzky GA. Molecular cloning of SLAP-130, an SLP-76-associated substrate of the T cell antigen receptor-stimulated protein tyrosine kinases. J Biol Chem. 1997;272:11674–11677. doi: 10.1074/jbc.272.18.11674. [DOI] [PubMed] [Google Scholar]
- Okabe S, Fukuda S, Kim YJ, Niki M, Pelus LM, Ohyashiki K, Pandolfi PP, Broxmeyer HE. Stromal cell-derived factor-1α/CXCL12-induced chemotaxis of T cells involves activation of the RasGAP-associated docking protein p62Dok-1. Blood. 2005;105:474–480. doi: 10.1182/blood-2004-03-0843. [DOI] [PubMed] [Google Scholar]
- Okigaki M, Davis C, Falasca M, Harroch S, Felsenfeld DP, Sheetz MP, Schlessinger J. Pyk2 regulates multiple signaling events crucial for macrophage morphology and migration. Proc Natl Acad Sci USA. 2003;100:10740–10745. doi: 10.1073/pnas.1834348100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ottoson NC, Pribila JT, Chan AS, Shimizu Y. Cutting edge: T cell migration regulated by CXCR4 chemokine receptor signaling to ZAP-70 tyrosine kinase. J Immunol. 2001;167:1857–1861. doi: 10.4049/jimmunol.167.4.1857. [DOI] [PubMed] [Google Scholar]
- Owen KA, Pixley FJ, Thomas KS, Vicente-Manzanares M, Ray BJ, Horwitz AF, Parsons JT, Beggs HE, Stanley ER, Bouton AH. Regulation of lamellipodial persistence, adhesion turnover, and motility in macrophages by focal adhesion kinase. J Cell Biol. 2007;179:1275–1287. doi: 10.1083/jcb.200708093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pauker MH, Reicher B, Fried S, Perl O, Barda-Saad M. Functional cooperation between the proteins Nck and ADAP is fundamental for actin reorganization. Mol Cell Biol. 2011;31:2653–2666. doi: 10.1128/MCB.01358-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson EJ, Woods ML, Dmowski SA, Derimanov G, Jordan MS, Wu JN, Myung PS, Liu QH, Pribila JT, Freedman BD, et al. Coupling of the TCR to integrin activation by Slap-130/Fyb. Science. 2001;293:2263–2265. doi: 10.1126/science.1063486. [DOI] [PubMed] [Google Scholar]
- Phee H, Abraham RT, Weiss A. Dynamic recruitment of PAK1 to the immunological synapse is mediated by PIX independently of SLP-76 and Vav1. Nat Immunol. 2005;6:608–617. doi: 10.1038/ni1199. [DOI] [PubMed] [Google Scholar]
- Qian D, Lev S, van Oers NS, Dikic I, Schlessinger J, Weiss A. Tyrosine phosphorylation of Pyk2 is selectively regulated by Fyn during TCR signaling. J Exp Med. 1997;185:1253–1259. doi: 10.1084/jem.185.7.1253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raab M, da Silva AJ, Findell PR, Rudd CE. Regulation of Vav-SLP-76 binding by ZAP-70 and its relevance to TCR zeta/CD3 induction of interleukin-2. Immunity. 1997;6:155–164. doi: 10.1016/s1074-7613(00)80422-7. [DOI] [PubMed] [Google Scholar]
- Reynolds LF, Smyth LA, Norton T, Freshney N, Downward J, Kioussis D, Tybulewicz VL. Vav1 transduces T cell receptor signals to the activation of phospholipase C-γ1 via phosphoinositide 3-kinase-dependent and -independent pathways. J Exp Med. 2002;195:1103–1114. doi: 10.1084/jem.20011663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Runne C, Chen S. PLEKHG2 promotes heterotrimeric G protein βγ-stimulated lymphocyte migration via Rac and Cdc42 activation and actin polymerization. Mol Cell Biol. 2013;33:4294–4307. doi: 10.1128/MCB.00879-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasaki H, Nagura K, Ishino M, Tobioka H, Kotani K, Sasaki T. Cloning and characterization of cell adhesion kinase β, a novel protein-tyrosine kinase of the focal adhesion kinase subfamily. J Biol Chem. 1995;270:21206–21219. doi: 10.1074/jbc.270.36.21206. [DOI] [PubMed] [Google Scholar]
- Seoh ML, Ng CH, Yong J, Lim L, Leung T. ArhGAP15, a novel human RacGAP protein with GTPase binding property. FEBS Lett. 2003;539:131–137. doi: 10.1016/s0014-5793(03)00213-8. [DOI] [PubMed] [Google Scholar]
- Siliceo M, Garcia-Bernal D, Carrasco S, Diaz-Flores E, Coluccio Leskow F, Teixido J, Kazanietz MG, Merida I. β2-chimaerin provides a diacylglycerol-dependent mechanism for regulation of adhesion and chemotaxis of T cells. J Cell Sci. 2006;119:141–152. doi: 10.1242/jcs.02722. [DOI] [PubMed] [Google Scholar]
- Svensson L, Howarth K, McDowall A, Patzak I, Evans R, Ussar S, Moser M, Metin A, Fried M, Tomlinson I, Hogg N. Leukocyte adhesion deficiency-III is caused by mutations in KINDLIN3 affecting integrin activation. Nat Med. 2009;15:306–312. doi: 10.1038/nm.1931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thelen M, Stein JV. How chemokines invite leukocytes to dance. Nat Immunol. 2008;9:953–959. doi: 10.1038/ni.f.207. [DOI] [PubMed] [Google Scholar]
- Tokiwa G, Dikic I, Lev S, Schlessinger J. Activation of Pyk2 by stress signals and coupling with JNK signaling pathway. Science. 1996;273:792–794. doi: 10.1126/science.273.5276.792. [DOI] [PubMed] [Google Scholar]
- Tuosto L, Michel F, Acuto O. p95vav associates with tyrosine-phosphorylated SLP-76 in antigen-stimulated T cells. J Exp Med. 1996;184:1161–1166. doi: 10.1084/jem.184.3.1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner M, Billadeau DD. VAV proteins as signal integrators for multi-subunit immune-recognition receptors. Nat Rev Immunol. 2002;2:476–486. doi: 10.1038/nri840. [DOI] [PubMed] [Google Scholar]
- Tybulewicz VL. Vav-family proteins in T-cell signalling. Curr Opin Immunol. 2005;17:267–274. doi: 10.1016/j.coi.2005.04.003. [DOI] [PubMed] [Google Scholar]
- van Seventer GA, Mullen MM, van Seventer JM. Pyk2 is differentially regulated by β1 integrin- and CD28-mediated co-stimulation in human CD4+ T lymphocytes. Eur J Immunol. 1998;28:3867–3877. doi: 10.1002/(SICI)1521-4141(199811)28:11<3867::AID-IMMU3867>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- Wang H, Liu H, Lu Y, Lovatt M, Wei B, Rudd CE. Functional defects of SKAP-55-deficient T cells identify a regulatory role for the adaptor in LFA-1 adhesion. Mol Cell Biol. 2007;27:6863–6875. doi: 10.1128/MCB.00556-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Moon EY, Azouz A, Wu X, Smith A, Schneider H, Hogg N, Rudd CE. SKAP-55 regulates integrin adhesion and formation of T cell-APC conjugates. Nat Immunol. 2003;4:366–374. doi: 10.1038/ni913. [DOI] [PubMed] [Google Scholar]
- Wu J, Motto DG, Koretzky GA, Weiss A. Vav and SLP-76 interact and functionally cooperate in IL-2 gene activation. Immunity. 1996;4:593–602. doi: 10.1016/s1074-7613(00)80485-9. [DOI] [PubMed] [Google Scholar]
- Xiong W, Parsons JT. Induction of apoptosis after expression of PYK2, a tyrosine kinase structurally related to focal adhesion kinase. J Cell Biol. 1997;139:529–539. doi: 10.1083/jcb.139.2.529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong WC, Macklem M, Parsons JT. Expression and characterization of splice variants of PYK2, a focal adhesion kinase-related protein. J Cell Sci. 1998;111:1981–1991. doi: 10.1242/jcs.111.14.1981. [DOI] [PubMed] [Google Scholar]
- Zheng C, Xing Z, Bian ZC, Guo C, Akbay A, Warner L, Guan JL. Differential regulation of Pyk2 and focal adhesion kinase (FAK). The C-terminal domain of FAK confers response to cell adhesion. J Biol Chem. 1998;273:2384–2389. doi: 10.1074/jbc.273.4.2384. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.