Abstract
Purpose
To investigate the effect and safety of a selective Rho kinase inhibitor, ripasudil 0.4% eye drops, on corneal endothelial cells of healthy subjects.
Design
Prospective, interventional case series.
Methods
In this study, 6 healthy subjects were administered ripasudil 0.4% in the right eye twice daily for 1 week. Morphological changes and corneal endothelial cell density were examined by noncontact and contact specular microscopy. Central corneal thickness and corneal volume of 5 mm-diameter area of center cornea were analyzed by Pentacam Scheimpflug topography. All the above measurements were conducted in both eyes before administration, 1.5 and 6 hours after the initial administration on day 0; and in the same manner after the final administration on day 7.
Results
By noncontact specular microscopy, indistinct cell borders with pseudo guttae were observed, but by contact specular microscopy, morphological changes of corneal endothelial cells were mild and pseudo guttae was not observed after single and repeated administration of ripasudil in all subjects. These changes resolved prior to the next administration, and corneal endothelial cell density, central corneal thickness and corneal volume were not changed throughout the study period.
Conclusion
Transient morphological changes of corneal endothelial cells such as indistinct cell borders with pseudo guttae were observed by noncontact specular microscopy in healthy subjects after ripasudil administration. Corneal edema was not observed and corneal endothelial cell density did not decrease after 1 week repetitive administration. These morphological changes were reversible and corneal endothelial cell morphology returned to normal prior to the next administration.
Trial Registration
JAPIC Clinical Trials Information 142705
Introduction
Rho-associated, coiled-coil containing protein kinase (ROCK), serine/threonine kinase is involved in diverse physiological functions, such as cytoskeletal rearrangement related to cell shape, smooth muscle contraction, and gene expression [1,2]. Recently, by focusing on these physiological functions, many researchers are investigating clinical applications for ROCK inhibitors [3–6]. Some selective ROCK inhibitors are actually used in clinical practice, for example, fasudil and ripasudil are therapeutic agents for cerebral vasospasm and glaucoma, respectively. The mechanism of intraocular pressure-lowering by ROCK inhibitors, including ripasudil, is hypothesized to be due to ROCK inhibitors increase in conventional outflow of aqueous humor by directly altering cell shape in the trabecular meshwork and increasing the permeability of the Schlemm’s canal endothelial cells [4,7–9].
Currently, treatment of corneal endothelial dysfunction is mainly corneal transplantation, in addition, various alternative therapies are propounded [10,11]. Recently, attention has been focused on the effect of ROCK inhibitor on corneal endothelial cells (CECs). Okumura et al. reported that the selective ROCK inhibitor Y-27632 promotes cell adhesion and proliferation, and inhibits the apoptosis of primate CECs in culture [12]. In rabbit and monkey models of partial endothelial dysfunction, they showed that corneal endothelial wound healing was accelerated via the topical application of Y-27632 and Y-39983 to the ocular surface, resulting in regeneration of a corneal endothelial monolayer with a high endothelial cell density (ECD) [13–15]. In addition, the positive effect of Y-27632 eye drops in treating patients with central corneal edema due to Fuchs corneal endothelial dystrophy was reported [5,13,16].
Ripasudil 0.4% (K-115, Glanatec; Kowa Company, Ltd, Nagoya, Japan) is an ophthalmic solution which was approved in Japan for the treatment of glaucoma and ocular hypertension in September 2014 [17]. In a preclinical study of ripasudil in cynomolgus monkeys, some morphological changes in CECs and limited decrease in corneal thickness were observed after both single and repeated instillation of ripasudil [18]. These findings lead us to consider the possibility of the effect of ripasudil on CECs. In this prospective study, to investigate the effect and safety of ripasudil 0.4% ophthalmic solution on human CECs, we examined morphological changes and ECD in healthy subjects after single and 1 week of ripasudil administration.
Methods
A prospective, open-label clinical study was conducted at University Hospital, Kyoto Prefectural University of Medicine (KPUM) in accordance with the ethical principles of the Declaration of Helsinki. Heishinkai Medical Group Incorporated, OPHAC Hospital, which is not part of KPUM, was in charge of subject screening. The study protocol was approved prior to beginning the study by each institutional review board as follows, KPUM Institutional Review Board; OPHAC Hospital Institutional Review Board. Subjects for the clinical study received complete information regarding the protocol, and written informed consent was obtained from each participant before entry to the study. This study is registered at www.clinicaltrials.jp as study no. 142705.
Inclusion criteria were healthy Japanese subjects, aged from 20 to 64 years. Subjects with ocular disease (including corneal guttae) or who had undergone ocular surgery (including laser treatment) in either eye were excluded from the study. Subjects with central ECD of lower than 2000 cells/mm2, with CCT of 600 μm or more, or with their best-corrected visual acuity of worse than logMAR visual acuity 0.0 (decimal visual acuity 1.0) in either eye were also excluded. Throughout the study period, subjects were prohibited from receiving medical therapy (excluding medical therapy for adverse events), ocular surgery, laser treatment, ocular treatment, and wearing contact lenses.
Subjects fulfilling the above criteria were screened in OPHAC Hospital, and then transferred to KPUM within the 4 week observation period (during which they did not receive medical therapy or ripasudil). During the administration period, subjects were administered 1 drop of ripasudil 0.4% into the right eye twice daily for 1 week.
To investigate the effect of ripasudil administration on CECs, corneal endothelial cell morphology and ECD were examined by noncontact and specially-designed contact specular microscopy. Automated image capture and corneal endothelial cell count by the software associated with the device was performed by EM3000 noncontact specular microscopy (Tomey corporation, Nagoya, Japan). Subsequently, endothelial cell examination was performed with KSSP laser scanning wide-field contact specular microscopy (Konan Medical, Inc, Nishinomiya, Japan). Oxybuprocaine 0.4% eye drops were used to anaesthetize the eyes and subjects were asked to concentrate on the fixation light in the microscope and the contact cone lens (×40 magnification) was used to record pictures. The images from the center cornea with good contrast were selected and ECD was analyzed by using the manufacturer's software by the center method. To evaluate the corneal endothelial function, CCT and corneal volume of 5 mm-diameter area of the center cornea were analyzed by Pentacam Scheimpflug topography (Oculus Optikgeräte GmbH, Wetzlar, Germany).
These ocular examinations were conducted in both eyes before administration, 1.5 and 6 hours after the initial administration on day 0; and in the same manner after the final administration on day 7. Between 4 to 8 weeks after the final administration on day 7, the ocular examinations were repeated for follow-up observation. Other ocular examinations including slit-lamp microscopy, noncontact tonometry, and best-corrected visual acuity measurements were conducted, and adverse events were collected throughout the study period.
The changes of ECD, CCT, slit-lamp examination, best-corrected visual acuity, and intraocular pressure from before the initial administration to each subsequent time point was evaluated in both eyes for each subject at day 0, day 7 and at the follow-up observation. The palpebral and bulbar conjunctiva, cornea, anterior chamber, iris, and lens were examined with slit-lamp microscopy throughout the study.
Results
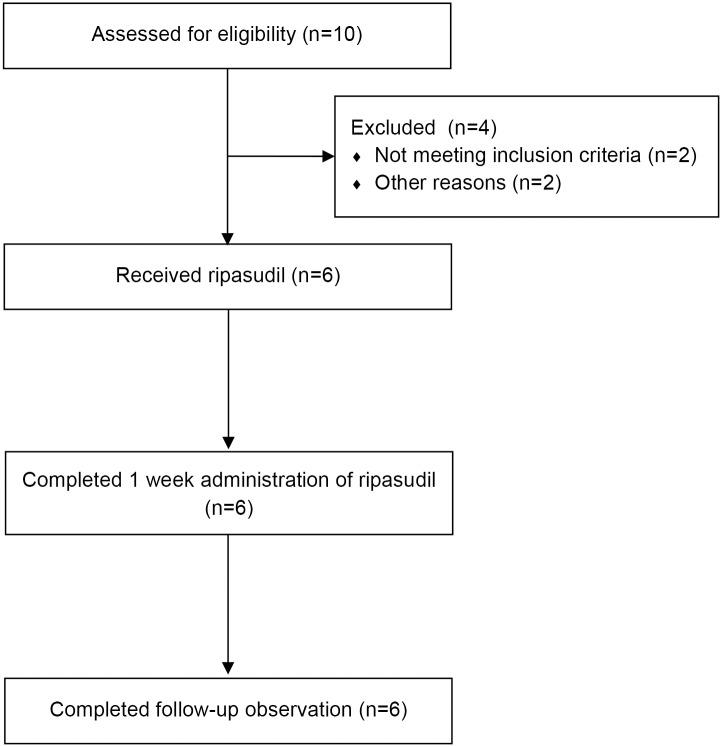
The enrollment of subjects began in November 2014 and follow-up observation was completed in February 2015. In this study, 10 subjects were enrolled for screening. Four subjects were excluded; 2 subjects did not satisfy inclusion criteria owing to abnormal laboratory value; 1 subject could not participate for study schedule; ocular examinations were difficult for another 1 subject due to having long eyelashes. The remaining 6 healthy male without any ocular diseases were enrolled in this study and had ripasudil 0.4% administered twice daily for 1 week (Fig 1). The mean age of the subjects was 27.7 ± 5.1 (mean ± SD) (ranged from 20 to 32).
Fig 1. Flow diagram of the study.
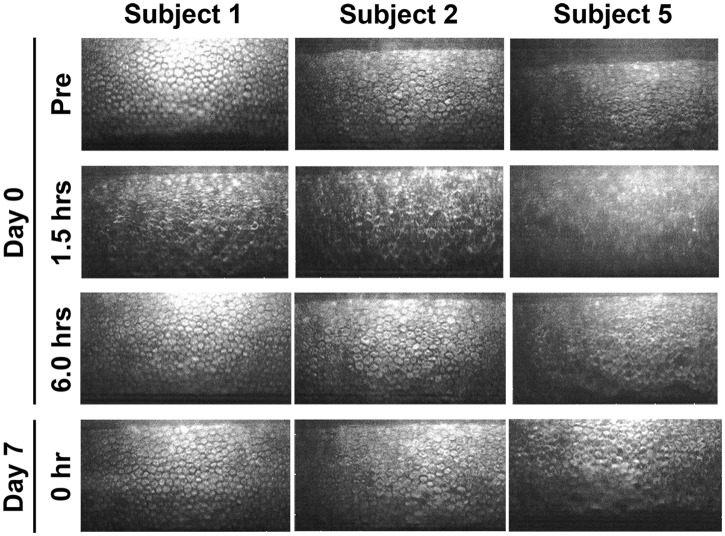
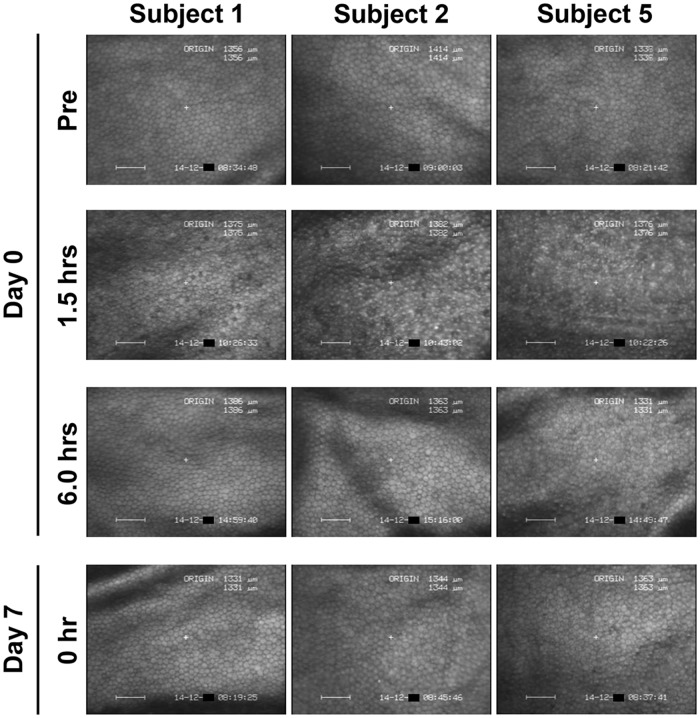
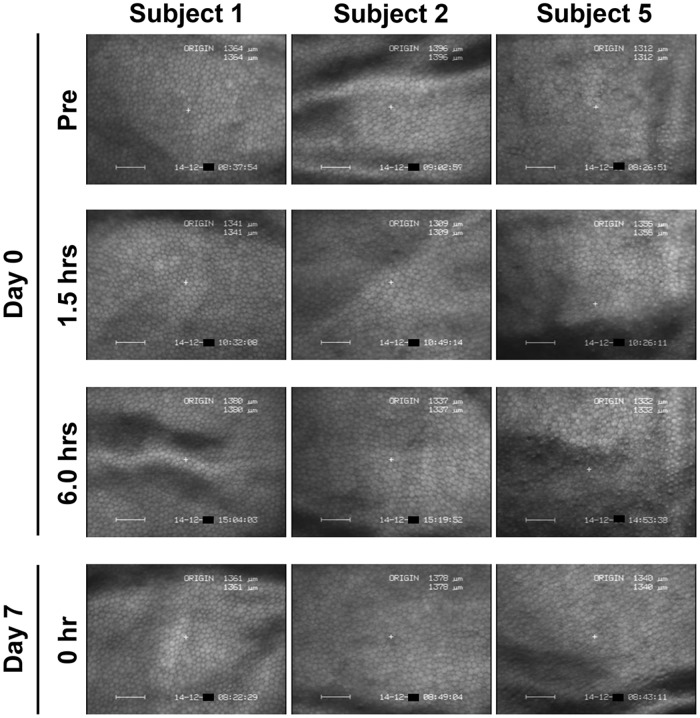
By noncontact specular microscopic examination, distinctive morphological changes of CECs were evident in all 6 subjects 1.5 hours after the initial administration of ripasudil (Fig 2). Highly irregular and blurred images by noncontact specular microscopy were obtained at 1.5 hours after ripasudil administration in all the subjects. Cell borders of CECs which are usually seen as black lines by specular microscopy were observed as white lines due to reflection of the light and as irregular forms. Guttae like dark cells with irregular and indistinct cell borders were detected in all the 6 subjects. In subject 5, the endothelial image was considerably affected and was hardly distinguished by noncontact specular microscopy (Fig 2, right, second). In contrast, using contact specular microscopy, 1.5 hours after ripasudil administration CECs showed less definitive morphological change though there were some irregular cell borders. A very few dark or swollen cells were observed only sporadically and they did not show the characteristic morphology of guttae (Fig 3, right, second). These morphological changes were detected in common in all of 6 subjects. These changes resolved and CEC morphology returned to normal by 6 hours after administration in 5 of 6 subjects by specular microscopy (Figs 2 and 3, left and middle columns). In subject 5, minor guttae like dark cells was still detectable at 6 hours after administration using noncontact specular microscopy (Fig 2, right, third) but were not evident by contact specular microscopy (Fig 3, right, third). After repeated administration of ripasudil, contact specular microscopic images of CECs in all subjects showed healthy morphology prior to the final administration on day 7 (Fig 3, bottom). At the follow-up observation, no abnormal findings of CECs were detected by contact or noncontact specular microscopy in any subjects (data not shown). Serial observations of CECs by noncontact and contact specular microscopies on day 0 and day 7 revealed the morphological changes to be reversible and no morphological abnormalities were observed in the CECs.
Fig 2. Noncontact specular microscopy images of corneal endothelium before and after ripasudil administration in instilled eye.
In 3 of 6 typical healthy subjects, before administration (Top row), 1.5 hours after the initial administration on day 0 (Second row), 6 hours after the initial administration on day 0 (Third row), and before the final administration on day 7 (Bottom row), in subject 1 (Left column), subject 2 (Middle column), and subject 5 (Right column). Before ripasudil administration, corneal endothelial cells showed regular hexagonal monolayer with clear cell borders in all subjects (Top row). One and half hours after ripasudil administration, endothelial cell showed irregular and indistinct cell borders with guttae like dark cells in subjects 1 (Left, second) and 2 (Middle, second). In subject 5, no analyzable image was obtained by noncontact specular microscopy (Right, second). At 6 hours post-dose or later, corneal endothelial morphology returned to normal in subjects 1 and 2. Some guttae like dark cells were still observed in subject 5 at 6 hours on day 0 (Right, third) and prior to the final administration on day 7 (Right, bottom).
Fig 3. Contact specular microscopy images of corneal endothelium before and after ripasudil administration in instilled eye.
In 3 of 6 typical healthy subjects, before administration (Top row), 1.5 hours after the initial administration on day 0 (Second row), 6 hours after the initial administration on day 0 (Third row), and before the final administration on day 7 (Bottom row), in subject 1 (Left column), subject 2 (Middle column), and subject 5 (Right column). Before ripasudil administration, wide-field contact specular microscopy revealed regular hexagonal monolayer of corneal endothelial cells in all subjects (Top row). One and half hours after ripasudil administration, corneal endothelial cells showed indistinct cell borders with a very few dark cells (Second row), but characteristic morphology of guttae was not observed. These morphological changes were detected in common in all subjects. At 6 hours post-dose or later, corneal endothelial morphology came back to normal in all subjects (Third and bottom rows).
ECD was evaluated at 6 time points and at the follow-up observation by noncontact and contact specular microscopies (Tables 1 and 2). ECD did not change throughout observation period other than 1.5 hours after ripasudil administration. At 1.5 hours, ECD was difficult to evaluate for all 6 subjects by noncontact specular microscopy and in 3 subjects by contact specular microscopy due to the blurred images with considerable morphological changes. Reliable data was obtained from subjects 1, 3 and 4 at 1.5 hours by contact specular microscopy and decreased ECD was not observed in these subjects. Moreover none of the 6 subjects showed decreased ECD at the follow-up observation.
Table 1. Endothelial Cell Density Examined by Noncontact Specular Microscopy Before and After the Administrations of Ripasudil in Instilled Eye.
N/A = not able to be calculated or unreliable data, SD = standard deviation. Values are the number of corneal endothelial cells (/mm2).
| Subject | Day 0 | Day 7 | Follow-up | ||||
|---|---|---|---|---|---|---|---|
| Pre a | 1.5 hrs. | 6 hrs. | 0 hr b | 1.5 hrs. | 6 hrs. | ||
| 1 | 3252 | N/A | 3280 | 3201 | N/A | 3210 | 3175 |
| 2 | 2692 | N/A | 2583 | 2664 | N/A | 2478 | 2590 |
| 3 | 3001 | N/A | 2874 | 2974 | N/A | 2929 | 2881 |
| 4 | 2567 | N/A | 2426 | 2628 | N/A | 2613 | 2597 |
| 5 | 2384 | N/A | 2296 | 2283 | N/A | 2427 | 2614 |
| 6 | 2824 | N/A | 2702 | 3010 | N/A | 2825 | 2982 |
| Mean | 2786.7 | - | 2693.5 | 2793.3 | - | 2747.0 | 2806.5 |
| SD | 310.9 | - | 351.7 | 331.7 | - | 298.8 | 244.9 |
a Before the administration of ripasudil.
b Before the final administrations of ripasudil (Twelve hours after administration).
Table 2. Endothelial Cell Density Examined by Contact Specular Microscopy Before and After the Administrations of Ripasudil in Instilled Eye.
N/A = not able to be calculated or unreliable data, SD = standard deviation. Values are the number of corneal endothelial cells (/mm2).
| Subject | Day 0 | Day 7 | Follow-up | ||||
|---|---|---|---|---|---|---|---|
| Pre a | 1.5 hrs. | 6 hrs. | 0 hr b | 1.5 hrs. | 6 hrs. | ||
| 1 | 3175 | 3135 | 3155 | 3390 | 3378 | 3378 | 3300 |
| 2 | 2646 | N/A | 2688 | 2604 | N/A | 2611 | 2545 |
| 3 | 3125 | 3115 | 3226 | 3215 | 3145 | 3165 | 3145 |
| 4 | 2283 | 2247 | 2353 | 2525 | 2506 | 2564 | 2421 |
| 5 | 2755 | N/A | 2725 | 2849 | N/A | 2933 | 2833 |
| 6 | 3058 | N/A | 2941 | 2967 | N/A | 2994 | 3096 |
| Mean | 2840.3 | - | 2848.0 | 2925.0 | - | 2940.8 | 2890.0 |
| SD | 345.3 | - | 326.0 | 337.9 | - | 314.7 | 351.5 |
a Before the administration of ripasudil.
b Before the final administrations of ripasudil (Twelve hours after administration).
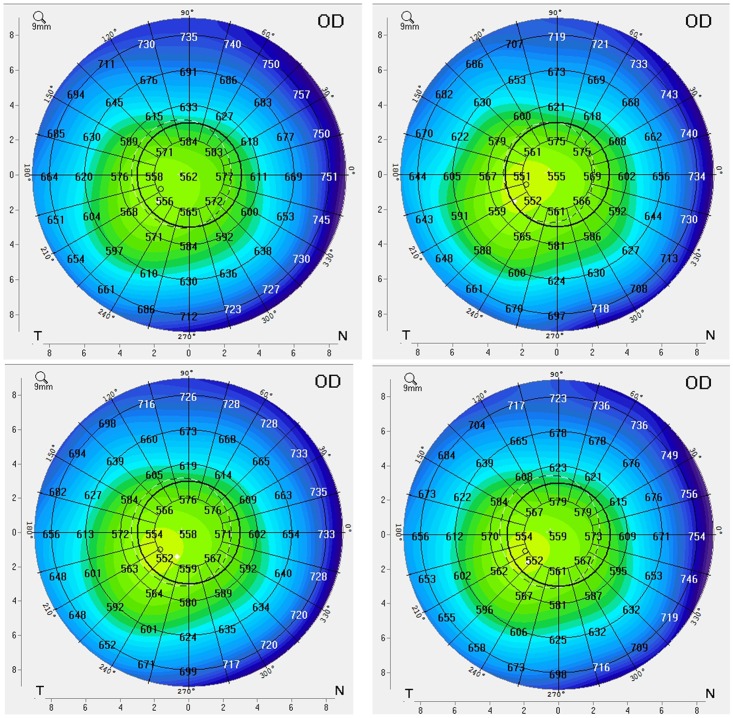
To examine the effect of ripasudil on corneal endothelial function of healthy subjects, we evaluated CCT (Table 3) and corneal volume of the 5 mm-diameter area at the central cornea (Table 4) at 6 time points and at the follow-up observation using Pentacam Scheimpflug topography. We did not find any changes for either CCT or corneal volume for any of the 6 subjects. Corneal pachymetry mapping data obtained by Pentacam Scheimpflug topography supported the findings obtained by CCT and corneal volume (Fig 4). Best-corrected visual acuity did not change in any of the subjects eyes. In the non-instilled eye, there were no morphological changes in CECs (Figs 5 and 6), changes of ECD (Tables 5 and 6), or change in CCT (Table 7) or corneal volume (Table 8) observed in any of the subjects. The only drug related adverse events observed were mild conjunctival hyperemia, which was detected in 5 of 6 subjects (except subject 1), and resolved prior to the next administration in all cases. By slit-lamp microscopy, the cornea remained clear throughout the observation period and corneal stromal and epithelial edema was not detected in any subject. Mild guttae like endothelial reflection, corresponding to the specular microscopic image, was observed at 1.5 hours after the initial administration in 1 subject (subject 2). This finding was resolved by 6 hours.
Table 3. Central Corneal Thickness Before and After the Administrations of Ripasudil in Instilled Eye.
SD = standard deviation. Values are the central corneal thickness (μm) examined by Pentacam Scheimpflug topography.
| Subject | Day 0 | Day 7 | Follow-up | ||||
|---|---|---|---|---|---|---|---|
| Pre a | 1.5 hrs. | 6 hrs. | 0 hr b | 1.5 hrs. | 6 hrs. | ||
| 1 | 559 | 550 | 556 | 558 | 555 | 555 | 555 |
| 2 | 559 | 575 | 569 | 564 | 561 | 561 | 564 |
| 3 | 587 | 575 | 575 | 568 | 566 | 575 | 580 |
| 4 | 560 | 554 | 556 | 558 | 559 | 554 | 567 |
| 5 | 558 | 553 | 559 | 554 | 556 | 555 | 562 |
| 6 | 561 | 559 | 555 | 561 | 562 | 561 | 559 |
| Mean | 564.0 | 561.0 | 561.7 | 560.5 | 559.8 | 560.2 | 564.5 |
| SD | 11.3 | 11.2 | 8.3 | 5.0 | 4.1 | 7.9 | 8.6 |
a Before the administration of ripasudil.
b Before the final administrations of ripasudil (Twelve hours after administration).
Table 4. Corneal Volume of 5mm Diameter Area of Central Cornea Before and After the Administrations of Ripasudil in Instilled Eye.
SD = standard deviation. Values are the corneal volume of 5mm diameter area (mm3).
| Subject | Day 0 | Day 7 | Follow-up | ||||
|---|---|---|---|---|---|---|---|
| Pre a | 1.5 hrs. | 6 hrs. | 0 hr b | 1.5 hrs. | 6 hrs. | ||
| 1 | 12.1 | 11.9 | 12.0 | 12.1 | 12.1 | 12.0 | 12.0 |
| 2 | 11.9 | 12.2 | 12.1 | 12.1 | 11.9 | 11.9 | 11.9 |
| 3 | 12.0 | 12.1 | 12.1 | 12.1 | 11.9 | 12.1 | 12.3 |
| 4 | 11.9 | 11.7 | 11.8 | 11.8 | 11.8 | 11.7 | 12.0 |
| 5 | 11.9 | 11.8 | 12.0 | 11.9 | 11.9 | 11.8 | 12.0 |
| 6 | 11.9 | 11.9 | 11.8 | 11.9 | 12.0 | 11.9 | 11.9 |
| Mean | 11.95 | 11.93 | 11.97 | 11.98 | 11.93 | 11.90 | 12.02 |
| SD | 0.08 | 0.19 | 0.14 | 0.13 | 0.10 | 0.14 | 0.15 |
a Before the administration of ripasudil.
b Before the final administrations of ripasudil (Twelve hours after administration).
Fig 4. Corneal pachymetry mapping in instilled eye.
Corneal pachymetry mapping of subject 4 on day 0 before administration (Top left), 1.5 hours (Top right) and 6 hours (Bottom left) after the initial administration and on day 7 prior to the final administration (Bottom right) of ripasudil in instilled eye. Throughout the observation period, there was no change of corneal thickness of the center and peripheral cornea.
Fig 5. Noncontact specular microscopy images of corneal endothelium before and after ripasudil administration in non-instilled eye.
In 3 of 6 typical healthy subjects, before administration (Top row), 1.5 hours after the initial administration on day 0 (Second row), 6 hours after the initial administration on day 0 (Third row), and before the final administration on day 7 (Bottom row) of ripasudil in non-instilled eye, in subject 1 (Left column), subject 2 (Middle column), and subject 5 (Right column). Throughout the observation period, there were no morphological changes of corneal endothelial cells in non-instilled eye.
Fig 6. Contact specular microscopy images of corneal endothelium before and after ripasudil administration in non-instilled eye.
In 3 of 6 typical healthy subjects, before administration (Top row), 1.5 hours after the initial administration on day 0 (Second row), 6 hours after the initial administration on day 0 (Third row), and before the final administration on day 7 (Bottom row) of ripasudil in non-instilled eye, in subject 1 (Left column), subject 2 (Middle column), and subject 5 (Right column). Throughout the observation period, there were no morphological changes of corneal endothelial cells in non-instilled eye.
Table 5. Endothelial Cell Density Examined by Noncontact Specular Microscopy Before and After the Administrations of Ripasudil in Non-instilled Eye.
SD = standard deviation. Time points indicate administrations of ripasudil in instilled eye. Values are the number of corneal endothelial cells (/mm2).
| Subject | Day 0 | Day 7 | Follow-up | ||||
|---|---|---|---|---|---|---|---|
| Pre a | 1.5 hrs. | 6 hrs. | 0 hr b | 1.5 hrs. | 6 hrs. | ||
| 1 | 3187 | 3175 | 3201 | 3300 | 3274 | 3135 | 3221 |
| 2 | 2648 | 2578 | 2517 | 2543 | 2566 | 2544 | 2626 |
| 3 | 3006 | 3074 | 2886 | 3130 | 3042 | 3036 | 3074 |
| 4 | 2459 | 2404 | 2472 | 2465 | 2437 | 2380 | 2476 |
| 5 | 2690 | 2548 | 2657 | 2507 | 2611 | 2697 | 2618 |
| 6 | 2864 | 2744 | 2873 | 2978 | 2805 | 2819 | 2955 |
| Mean | 2809.0 | 2753.8 | 2767.7 | 2820.5 | 2789.2 | 2768.5 | 2828.3 |
| SD | 263.6 | 308.4 | 274.0 | 361.2 | 317.7 | 288.1 | 296.6 |
a Before the administrations of ripasudil in instilled eye.
b Twelve hours after the administrations of ripasudil in instilled eye.
Table 6. Endothelial Cell Density Examined by Contact Specular Microscopy Before and After the Administrations of Ripasudil in Non-instilled Eye.
SD = standard deviation. Time points indicate administrations of ripasudil in instilled eye. Values are the number of corneal endothelial cells (/mm2).
| Subject | Day 0 | Day 7 | Follow-up | ||||
|---|---|---|---|---|---|---|---|
| Pre a | 1.5 hrs. | 6 hrs. | 0 hr b | 1.5 hrs. | 6 hrs. | ||
| 1 | 3185 | 3344 | 3236 | 3247 | 3236 | 3226 | 3195 |
| 2 | 2681 | 2695 | 2710 | 2618 | 2717 | 2625 | 2558 |
| 3 | 3040 | 3289 | 3030 | 3106 | 3155 | 3125 | 3086 |
| 4 | 2381 | 2463 | 2364 | 2353 | 2488 | 2433 | 2358 |
| 5 | 2681 | 2786 | 2710 | 2865 | 2770 | 2747 | 2762 |
| 6 | 2976 | 2915 | 2778 | 2809 | 2801 | 2809 | 3067 |
| Mean | 2824.0 | 2915.3 | 2804.7 | 2833.0 | 2861.2 | 2827.5 | 2837.7 |
| SD | 295.7 | 344.5 | 299.9 | 323.8 | 282.4 | 300.3 | 333.5 |
a Before the administrations of ripasudil in instilled eye.
b Twelve hours after the administrations of ripasudil in instilled eye.
Table 7. Central Corneal Thickness in Non-instilled Eye.
SD = standard deviation. Time points indicate administrations of ripasudil in instilled eye. Values are the central corneal thickness measured by Pentacam Scheimpflug topography (μm).
| Subject | Day 0 | Day 7 | Follow-up | ||||
|---|---|---|---|---|---|---|---|
| Pre a | 1.5 hrs. | 6 hrs. | 0 hr b | 1.5 hrs. | 6 hrs. | ||
| 1 | 552 | 551 | 549 | 554 | 557 | 549 | 551 |
| 2 | 556 | 562 | 563 | 565 | 562 | 567 | 555 |
| 3 | 565 | 573 | 575 | 567 | 571 | 575 | 572 |
| 4 | 559 | 564 | 557 | 564 | 559 | 560 | 562 |
| 5 | 551 | 550 | 553 | 554 | 551 | 549 | 553 |
| 6 | 552 | 550 | 554 | 558 | 551 | 552 | 556 |
| Mean | 555.8 | 558.3 | 558.5 | 560.3 | 558.5 | 558.7 | 558.2 |
| SD | 5.4 | 9.5 | 9.3 | 5.8 | 7.5 | 10.7 | 7.7 |
a Before the administrations of ripasudil in instilled eye.
b Twelve hours after the administrations of ripasudil in instilled eye.
Table 8. Corneal Volume of 5mm Diameter Area of Central Cornea in Non-instilled Eye.
SD = standard deviation. Time points indicate administrations of ripasudil in instilled eye. Values are the corneal volume (mm3).
| Subject | Day 0 | Day 7 | Follow-up | ||||
|---|---|---|---|---|---|---|---|
| Pre a | 1.5 hrs. | 6 hrs. | 0 hr b | 1.5 hrs. | 6 hrs. | ||
| 1 | 11.8 | 11.9 | 11.8 | 11.9 | 12.0 | 11.8 | 11.8 |
| 2 | 11.8 | 11.9 | 11.9 | 12.0 | 11.9 | 12.0 | 11.7 |
| 3 | 12.0 | 12.2 | 12.2 | 12.1 | 12.1 | 12.2 | 12.2 |
| 4 | 11.9 | 12.0 | 11.8 | 12.0 | 11.8 | 11.9 | 12.0 |
| 5 | 11.8 | 11.8 | 11.8 | 11.9 | 11.8 | 11.8 | 11.8 |
| 6 | 11.6 | 11.6 | 11.7 | 11.8 | 11.6 | 11.6 | 11.7 |
| Mean | 11.82 | 11.90 | 11.87 | 11.95 | 11.87 | 11.88 | 11.87 |
| SD | 0.13 | 0.20 | 0.18 | 0.10 | 0.18 | 0.20 | 0.20 |
a Before the administrations of ripasudil in instilled eye.
b Twelve hours after the administrations of ripasudil in instilled eye.
Discussion
This study was designed and conducted to examine the effect and safety of a selective ROCK inhibitor, ripasudil 0.4% ophthalmic solution, on CECs in healthy subjects. Six healthy subjects with healthy cornea were instilled with ripasudil 0.4% twice daily for 1 week. Considerable morphological changes in CECs were observed by noncontact specular microscopy in all subjects at 1.5 hours after ripasudil administration. Since these changes were detected only in ripasudil instilled eyes but not in non-instilled eyes, it was considered to be a phenomenon associated with ripasudil administration.
One and half hours after ripasudil administration, irregularity of cell borders with guttae like dark cells was observed in CEC images taken by noncontact specular microscope. These findings were similar to the results from previous studies in cynomolgus monkeys [18], so these corneal endothelial responses to ripasudil might be common in primates. The reason for these findings detected in the animal study was considered to be due to the reflected light of specular microscopy being scattered by CECs with morphological changes [18]. However, the actual changes in CECs were not adequately examined due to the limitation of quality of CECs observed by noncontact specular microscopy.
In this clinical study, we used specially designed, slit-scanning, wide-field contact specular microscope to analyze the effect of ripasudil on corneal endothelial morphology, and we successfully obtained clearer images than those of noncontact specular microscopy. The contact specular microscope we used in this study is a machine KSSP made by Konan Medical, Inc. This machine has a slit-scanning system and effectively reduces the light scattering by CECs. In this study, we were able to observe the morphological changes of CECs caused by ripasudil administration in detail using contact specular microscopy. We found that CECs showed indistinct cell borders with a very few dark or swollen cells but there was no findings of loss of CECs. Considerable morphological changes with multiple guttae like cells which was observed by noncontact specular microscopy was not actually detected using contact specular microscopy.
As pharmacological effects of ROCK inhibitor, cell morphological changes induced by disruption of actin microfilament bundles and impairment of focal adhesion formation were reported [4,19], and similar morphological changes of CECs might also be induced by ripasudil. Moreover, similar findings were reported from previous studies in cynomolgus monkeys by instillation of both ripasudil and Y-39983 [18], and therefore, morphological changes of CECs could be a common characteristic of ROCK inhibitors. The reason that images using contact specular microscopy were clearer than those by noncontact specular microscopy, was thought to be due to a decrease in the influence of scattering of reflected light owing to the device being in contact with the corneal surface.
Similar morphological change of CECs detected by specular microscopy was previously reported in cynomolgus monkey after the administration of H-7, serine-threonine kinase inhibitor [20]. In that paper, it was reported that CEC borders became indistinct by 1 hour after H-7 administration but cell morphology was mostly recovered by 3–5 hours and had completely recovered to normal by 24 hours without changes in ECD. H-7 is a serine-threonine kinase which decreases intraocular pressure by relaxing and expanding the trabecular meshwork and Schlemm’s canal, which is a similar mechanism as ripasudil on intraocular pressure. It is interesting that CECs of human subjects with ripasudil administration showed a similar change with that of cynomolgus monkey administered H-7. Though the mechanism of this pseudo guttae formation is yet to be elucidated, it might have occurred due to morphological changes of CECs induced by the pharmacological effect of ripasudil on actin microfilament which might lead to strongly-scattered reflected light from the specular microscopy.
In previous studies, pseudo guttae with circumscribed black zones were observed after insertion of soft contact lenses in unadapted patients and they were referred to as “endothelial bleb” [21]. The causes of endothelial blebs from wearing soft contact lenses were ascribed to several factors, including the mechanical effects of the lens, reduced corneal oxygenation, interference with carbon dioxide (CO2) efflux and changes in corneal pH [22]. These factors are not likely to be involved in subjects with ripasudil administration, so the mechanism of pseudo guttae formation in ripasudil instilled eyes is likely different from patients wearing soft contact lenses.
The morphological changes in CECs after ripasudil administration were confirmed to be reversible effects and CECs returned to normal prior to the next administration for all subjects. In addition, ECD, CCT, corneal volume, and best-corrected visual acuity remained stable for all subjects. We conducted post hoc Wilcoxon signed rank test for changes from before administration on Day 0 in ECD, CCT and corneal volume. No significant differences were found in the instilled eye, and significant differences were found for only 1 time point for 2 ocular examinations in the non-instilled eye; CCT before administration on Day 7 (P = 0.03, two-sided); and corneal volume before administration on Day 7 (P = 0.03, two-sided). These differences were thought to be due to random fluctuations in the non-instilled eye. Thus, no findings of cell loss in CECs might have occurred, and we assumed that the findings similar to guttae were pseudo guttae which might have occurred due to transient morphological change of CECs. Pseudo guttae formation was a transient effect of ripasudil and these changes were different from “real” guttae where accumulation of abnormal extracellular matrix is often observed in corneal endothelial diseases such as Fuchs endothelial corneal dystrophy. Additionally, 1-year clinical evaluation of ripasudil 0.4% in patients with open-angle glaucoma and ocular hypertension revealed acceptable safety profile and there were no corneal endothelial disorders reported as adverse events in the long-term study [23].
In conclusion, this study revealed that transient morphological changes identified by indistinct cell borders and appearance of pseudo guttae were observed in CECs after ripasudil administration using noncontact specular microscopy. This may induce a transient functional alteration of corneal endothelium. However, the morphological changes were mild when observed by contact specular microscopy and there were no findings of CEC loss. These morphological changes did not effect corneal thickness or ECD. The recovery of CECs was confirmed by contact specular microscopy prior to the next administration of ripasudil. These results support the safety of short-term use of ripasudil on CECs in subjects with healthy cornea. Further studies are needed in order to explore the long-term effects of ripasudil and the underlying cellular mechanism of the effect on CECs.
Supporting Information
(PDF)
(PDF)
Acknowledgments
We thank Hiroshi Mikami, Heishinkai Medical Group Incorporated, OPHAC Hospital, Yodogawa, Osaka, contribution to data collection.
Data Availability
All relevant data are within the paper and Supporting Information files.
Funding Statement
This study was sponsored by Kowa Company, Ltd, Nagoya, Japan. The sponsor participated in design, study conduct, data collection, data management, data analysis, interpretation of the data of the study, and preparation, review, and approval of the manuscript.
References
- 1. Ishizaki T, Maekawa M, Fujisawa K, Okawa K, Iwamatsu A, Fujita A, et al. The small GTP-binding protein Rho binds to and activates a 160 kDa Ser/Thr protein kinase homologous to myotonic dystrophy kinase. EMBO J 1996;15: 1885–1893. [PMC free article] [PubMed] [Google Scholar]
- 2. Shimokawa H, Takeshita A. Rho-kinase is an important therapeutic target in cardiovascular medicine. Arterioscler Thromb Vasc Biol 2005;25: 1767–1775. [DOI] [PubMed] [Google Scholar]
- 3. Shibuya M, Suzuki Y, Sugita K, Saito I, Sasaki T, Takakura K, et al. Effect of AT877 on cerebral vasospasm after aneurysmal subarachnoid hemorrhage. Results of a prospective placebo-controlled double-blind trial. J Neurosurg 1992;76: 571–577. [DOI] [PubMed] [Google Scholar]
- 4. Honjo M, Tanihara H, Inatani M, Kido N, Sawamura T, Yue BY, et al. Effects of rho-associated protein kinase inhibitor Y-27632 on intraocular pressure and outflow facility. Invest Ophthalmol Vis Sci 2001;42: 137–144. [PubMed] [Google Scholar]
- 5. Koizumi N, Okumura N, Ueno M, Kinoshita S. New therapeutic modality for corneal endothelial disease using Rho-associated kinase inhibitor eye drops. Cornea 2014;33: S25–31. [DOI] [PubMed] [Google Scholar]
- 6. Arita R, Hata Y, Nakao S, Kita T, Miura M, Kawahara S, et al. Rho kinase inhibition by fasudil ameliorates diabetes-induced microvascular damage. Diabetes 2009;58: 215–226. 10.2337/db08-0762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Isobe T, Mizuno K, Kaneko Y, Ohta M, Koide T, Tanabe S. Effects of K-115, a rho-kinase inhibitor, on aqueous humor dynamics in rabbits. Curr Eye Res 2014;39: 813–822. 10.3109/02713683.2013.874444 [DOI] [PubMed] [Google Scholar]
- 8. Koga T, Koga T, Awai M, Tsutsui J, Yue BY, Tanihara H. Rho-associated protein kinase inhibitor, Y-27632, induces alterations in adhesion, contraction and motility in cultured human trabecular meshwork cells. Exp Eye Res 2006;82: 362–370. [DOI] [PubMed] [Google Scholar]
- 9. Kameda T, Inoue T, Inatani M, Fujimoto T, Honjo M, Kasaoka N, et al. The effect of rho-associated protein kinase inhibitor on monkey Schlemm's canal endothelial cells. Invest Ophthalmol Vis Sci 2012;53: 3092–3103. 10.1167/iovs.11-8018 [DOI] [PubMed] [Google Scholar]
- 10. Jhanji V, Mehta JS, Sharma N, Sharma B, Vajpayee RB. Targeted corneal transplantation. Curr Opin Ophthalmol. 2012;23:324–329. [DOI] [PubMed] [Google Scholar]
- 11. Jhanji V, Sharma N, Agarwal T, Vajpayee RB. Alternatives to allograft corneal transplantation. Curr Opin Ophthalmol. 2010;21:301–309. [PubMed] [Google Scholar]
- 12. Okumura N, Ueno M, Koizumi N, Sakamoto Y, Hirata K, Hamuro J, et al. Enhancement on primate corneal endothelial cell survival in vitro by a ROCK inhibitor. Invest Ophthalmol Vis Sci 2009;50: 3680–3687. 10.1167/iovs.08-2634 [DOI] [PubMed] [Google Scholar]
- 13. Okumura N, Koizumi N, Kay EP, Ueno M, Sakamoto Y, Nakamura S, et al. The ROCK inhibitor eye drop accelerates corneal endothelium wound healing. Invest Ophthalmol Vis Sci 2013;54: 2493–2502. 10.1167/iovs.12-11320 [DOI] [PubMed] [Google Scholar]
- 14. Okumura N, Nakano S, Kay EP, Numata R, Ota A, Sowa Y, et al. Involvement of cyclin D and p27 in cell proliferation mediated by ROCK inhibitors Y-27632 and Y-39983 during corneal endothelium wound healing. Invest Ophthalmol Vis Sci. 2014;55: 318–329. 10.1167/iovs.13-12225 [DOI] [PubMed] [Google Scholar]
- 15. Okumura N, Koizumi N, Ueno M, Sakamoto Y, Takahashi H, Hirata K, et al. Enhancement of corneal endothelium wound healing by Rho-associated kinase (ROCK) inhibitor eye drops. Br J Ophthalmol 2011;95: 1006–1009. 10.1136/bjo.2010.194571 [DOI] [PubMed] [Google Scholar]
- 16. Koizumi N, Okumura N, Ueno M, Nakagawa H, Hamuro J, Kinoshita S. Rho-associated kinase inhibitor eye drop treatment as a possible medical treatment for Fuchs corneal dystrophy. Cornea 2013;32: 1167–1170. [DOI] [PubMed] [Google Scholar]
- 17. Garnock-Jones KP. Ripasudil: first global approval. Drugs 2014;74: 2211–2215. 10.1007/s40265-014-0333-2 [DOI] [PubMed] [Google Scholar]
- 18. Wato E, Omichi K, Yoneyama S, Tanaka M, Kagawa M, Amano Y. Safety evaluation of morphological changes in corneal endothelial cells induced by K-115 in cynomolgus monkeys. Fund Toxicol Sci 2014;1: 39–47. [Google Scholar]
- 19. Nagayama K, Matsumoto T. Contribution of actin filaments and microtubules to quasi-in situ tensile properties and internal force balance of cultured smooth muscle cells on a substrate. Am J Physiol Cell Physiol 2008;295: C1569–1578. 10.1152/ajpcell.00098.2008 [DOI] [PubMed] [Google Scholar]
- 20. Tian B, Sabanay I, Peterson JA, Hubbard WC, Geiger B, Kaufman PL. Acute effects of H-7 on ciliary epithelium and corneal endothelium in monkey eyes. Curr Eye Res 2001;22: 109–120 [DOI] [PubMed] [Google Scholar]
- 21. Zantos SG, Holden BA. Transient endothelial changes soon after wearing soft contact lenses. Am J Optom Physiol Opt 1977;54: 856–858. [DOI] [PubMed] [Google Scholar]
- 22. Holden BA, Williams L, Zantos SG. The etiology of transient endothelial changes in the human cornea. Invest Ophthalmol Vis Sci 1985;26: 1354–1359. [PubMed] [Google Scholar]
- 23. Tanihara H, Inoue T, Yamamoto T, Kuwayama Y, Abe H, Suganami H, et al. One-Year Clinical Evaluation of 0.4% Ripasudil (K-115) in Patients with Open-Angle Glaucoma and Ocular Hypertension. Acta Ophthalmol 2015; in press. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PDF)
(PDF)
Data Availability Statement
All relevant data are within the paper and Supporting Information files.