Abstract
Specific homologs of the plant Mildew Locus O (MLO) gene family act as susceptibility factors towards the powdery mildew (PM) fungal disease, causing significant economic losses in agricultural settings. Thus, in order to obtain PM resistant phenotypes, a general breeding strategy has been proposed, based on the selective inactivation of MLO susceptibility genes across cultivated species. In this study, PCR-based methodologies were used in order to isolate MLO genes from cultivated solanaceous crops that are hosts for PM fungi, namely eggplant, potato and tobacco, which were named SmMLO1, StMLO1 and NtMLO1, respectively. Based on phylogenetic analysis and sequence alignment, these genes were predicted to be orthologs of tomato SlMLO1 and pepper CaMLO2, previously shown to be required for PM pathogenesis. Full-length sequence of the tobacco homolog NtMLO1 was used for a heterologous transgenic complementation assay, resulting in its characterization as a PM susceptibility gene. The same assay showed that a single nucleotide change in a mutated NtMLO1 allele leads to complete gene loss-of-function. Results here presented, also including a complete overview of the tobacco and potato MLO gene families, are valuable to study MLO gene evolution in Solanaceae and for molecular breeding approaches aimed at introducing PM resistance using strategies of reverse genetics.
Electronic supplementary material
The online version of this article (doi:10.1007/s11248-015-9878-4) contains supplementary material, which is available to authorized users.
Keywords: MLO, Solanaceae, Powdery mildew, Resistance, Plant breeding
Introduction
Powdery mildew (PM) is a major fungal disease affecting thousands of plant species, caused by ascomycete fungi belonging to the order of Erysiphales (Glawe 2008). Chemical control of PM accounts for a large proportion of fungicides used in agricultural settings (Hewitt 1998). Therefore, the use of cultivars harbouring genetic sources of PM resistance is generally envisaged as a valuable strategy to reduce farming costs and to cope with public concerns related to environmental pollution and human health.
The Mildew Locus O (MLO) gene family encodes for plant-specific proteins harbouring several transmembrane domains, topologically reminiscent of metazoan G-protein coupled receptors (Devoto et al. 2003). Specific homologs of the MLO family act as susceptibility genes towards PM fungi. Indeed, their inactivation, through loss-of-function mutations or silencing, has been associated with a peculiar form of PM resistance, referred to as mlo resistance (Pavan et al. 2010). This is associated with the enhancement of exocytosis defence pathways at plant–pathogen interaction sites, which are thought to contribute to the prevention of fungal penetration into host cells (Assaad et al. 2004). Initially discovered in barley, mlo resistance has been later shown to occur in other plant species as well, specifically Arabidopsis, tomato, pea, pepper and bread wheat (Bai et al. 2008; Büschges et al. 1997; Consonni et al. 2006; Humphry et al. 2011; Pavan et al. 2011; Wang et al. 2014; Zheng et al. 2013). This eventually led to the formalization of a breeding approach based on the systematic inactivation of MLO susceptibility genes across cultivated species affected by the PM disease (Dangl et al. 2013; Pavan et al. 2010, 2011). Proof of concept for this strategy has been recently provided by the work of Wang et al. (2014), reporting the introduction of PM resistance in bread wheat following targeted mutagenesis of three MLO homoeoalleles. In contrast with most genetic sources of PM resistance, experimental data clearly indicate that mlo immunity is not specific towards particular fungal isolates and is extremely durable. For example, loss-of-function mutations of barley HvMLO confer resistance to all known isolates of the PM fungus Blumeria graminis f. sp. hordei, and is successfully employed in barley breeding since 1979 (Lyngkjaer et al. 2000). Similarly, pea er1 PM resistance, originating from the loss of function of PsMLO1, was first reported more than 60 years ago and is the only resistance source worldwide used for breeding purposes (Harland 1948; Humphry et al. 2011; Pavan et al. 2013).
Following the completion of the respective genome sequencing projects, a number of MLO homologs variable between 12 and 19 has been identified in the diploid species Arabidopsis, rice, grapevine, peach, woodland strawberry and cucumber (Devoto et al. 2003; Feechan et al. 2008; Liu and Zhu 2008; Pessina et al. 2014; Schouten et al. 2014). Remarkably, when placed in MLO protein family phylogenetic trees, all dicot MLO isoforms experimentally shown to be required for PM susceptibility group in the same clade, referred to as clade V in scientific literature (e.g. Feechan et al. 2008; Pavan et al. 2011; Acevedo-Garcia et al. 2014). This shows that evolutionary studies on MLO proteins may predict candidates for being PM susceptibility factors.
Concerning solanaceous crops, we have functionally characterized the two MLO orthologs SlMLO1 in tomato and CaMLO2 in pepper, whose inactivation is causally associated with PM resistance (Bai et al. 2008; Zheng et al. 2013). In this work, we report the isolation, through a PCR-based approach, of three MLO genes from other cultivated Solanaceae, namely eggplant, potato and tobacco, which are likely to share a relation of orthology with SlMLO1 and CaMLO2. The tobacco MLO homolog NtMLO1 was chosen for a transgenic complementation assay, resulting in its functional characterization and identification of a loss-of-function mutant allele. Finally, newly available tobacco and potato genome sequences (Sierro et al. 2014; The Potato Genome Sequencing Consortium 2011) were exploited to provide a comprehensive overview of the MLO gene families in these species.
Materials and methods
PCR-based isolation and phylogenetic characterization of MLO putative orthologs
Young leaves of eggplant (Solanum melongena cv. Half Lange Violette), potato (Solanum tuberosum cv. Desiree) and tobacco (Nicotiana tabacum cv. Petit Havana SR1) were collected for RNA extraction, which was performed using the Trizol reagent (Invitrogen). After RNA purification with the NucleoSpin RNA II kit (Macherey–Nagel), cDNA was synthesized using the SuperScript III RT first-strand cDNA synthesis kit (Invitrogen) with oligo(dT) primers.
Aiming to identify sequences of SlMLO1 putative orthologs, the primer pairs Sol-F1 (5′-CATTTGACATTTCCCCTTCTTC-3′)/Sol-R1 (5′-GCACCATGCATGAGTACCTCT-3′) and Sol-F2 (5′-TTGGCAGTTGCTCATGTATTG-3′)/Sol-R2 (5′-ATGGTGCCAGCTTCTAAGAG-3′) were designed on the untranslated and coding sequences of the SlMLO1 gene (GeneBank accession number NM_001247885), respectively, (Primer3, Rozen and Skaletsky 2000) and used for PCR amplification of cDNAs. Amplicons obtained with the Sol-F2/Sol-R2 primer pair were purified using the NucleoSpin Extract II kit (Macherey–Nagel) and ligated (molar ratio 1:1) into the pGEM-T easy vector (Promega). Recombinant plasmids were cloned in E. coli DH10β chemically competent cells and recovered by using the Qiaprep spin miniprep kit (Qiagen). Sequencing reactions were performed using universal T7 and SP6 primers (Eurofins MWG Operon).
In order to obtain full-length coding sequences of potato and tobacco MLO genes, sequences overlapping with those of the amplicons above mentioned were retrieved by BLAST search, using the tomato SlMLO1 coding sequence as query against expressed sequence tags (ESTs) and predicted coding sequence repositories, both available at the Sol Genomic Network (SGN) database (http://solgenomics.net), and then used for local alignment. The expression and sequence of candidate genes was verified by PCR amplification of cDNAs, using the primer pairs StMLO1-F (5′-ATGGCTAAAGAACGGTCG-3′)/StMLO1-R (5′-TTATTTGTTTCCAAAAGT-3′) and NtMLO1-F (5′-ATGGAGGCAACTCCGACTTG-3′)/NtMLO1-R (5′-TCAACTCATTTTGTTGCCAAATG-3′), cloning and sequencing, which were performed as above described.
In order to amplify a full-length MLO sequence in eggplant, the following primer pair was used: SmMLO1-F2 (5′-ATGGCTAAAGAACGGTCG-3′)/SmMLO1-R1 (5′-TTATTTGTTTCCAAAAGTAAAATCTGA-3′). The corresponding PCR product was cloned and sequenced as indicated above.
Full-length eggplant, potato and tobacco MLO genes (named SmMLO1, StMLO1 and NtMLO1, respectively) were translated in silico. Corresponding protein sequences were used, together with those of dicot MLO proteins experimentally associated with PM susceptibility [Arabidopsis thaliana AtMLO2 (GenBank accession code NP172598), AtMLO6 (NP176350) and AtMLO12 (NP565902), Solanum lycopersicum SlMLO1 (NP001234814), Capsicum annuum CaMLO2 (AFH68055), Pisum sativum PsMLO1 (ACO07297), Lotus japonicus LjMLO1 (AAX77015) and Medicago truncatula MtMLO1 (ADV40949)] and those of the remaining twelve homologs of the Arabidopsis thaliana AtMLO protein family, for ClustalW alignment and the construction of a Unweighted Pair Group Method with Arithmetic Mean (UPGMA) phylogenetic tree. Bootstrap values were calculated from 100 replicates. All of these bioinformatic analyses were performed using the CLC sequence viewer software (http://www.clcbio.com/).
Generation of transgenic plants overexpressing NtMLO1
Two different NtMLO1 PCR products, differing for a single nucleotide polymorphism, were inserted into the Gateway-compatible vector pENTR D-TOPO (Invitrogen) and cloned in E. coli competent cells. Presence of the inserts was assessed by colony PCR, restriction enzyme digestion and sequencing using the universal M13 primer pair. Inserts were then transferred by LR recombination into the binary plasmid vector pK7WG2, harboring the 35S Cauliflower Mosaic Virus (CaMV) promoter for constitutive expression and the marker gene nptII for kanamycin resistance selection. Plasmids were inserted into E. coli competent cells and positive colonies were again screened by colony PCR and sequencing, as above. Recombinant vectors were finally extracted and transferred to the AGL1-virG strain of A. tumefaciens by electroporation. A selected PM resistant tomato line, named Slmlo1, described by Bai et al. (2008) and carrying a loss-of-function deletion in the SlMLO1 coding sequence, was used for transformation. This was performed according to the method described by McCormick et al. (1986). Briefly, seeds were surface-sterilized and sown on half-strength Murashige and Skoog (MS) agar supplemented with sucrose (10 g/l). Cotyledons were excised from 10-day-old seedlings, cut in two parts and submerged in an A. tumefaciens suspension with an OD600 value of about 0.125. Infected cotyledonary explants were placed abaxially on the GCF10 medium (4.3 g/l MS basal salt mixture, 8 g/l agar, 30 g/l sucrose, 108.73 mg/l Nitsch vitamins, 1.5 mg/l zeatin riboside, 0.2 mg/l indole-3-acetic acid, pH 5.8) supplemented with 1 ml/l acetosyringone at 25 °C for 48 h. Then, they were transferred to the GCF10 medium to which 100 mg/ml timentin and 50 mg/ml kanamycin were added and sub-cultured onto fresh medium every 3 weeks until shoot buds were observed. These were excised from the callus and transferred to the GCF11 medium (4.3 g/l MS basal salt mixture, 8 g/l agar, 30 g/l sucrose, 108.73 mg/l Nitsch vitamins, 1.9 mg/l zeatin riboside, pH 5.8) with 100 mg/ml timentin and 50 mg/ml kanamycin. After meristem development, the explants were transferred to the root-inducing medium MS30B5 (4.3 g/l MS basal salt mixture, 8 g/l agar, 30 g/l sucrose, 112 mg/l vitamin B5, 50 mg/ml kanamycin, pH 5.8). Once roots were developed, plantlets were finally located on woolen rock and grown in a greenhouse compartment.
For each of the two transformations with a different NtMLO1 gene sequence, 20 T1 plants and two T2 families (each composed by fifteen individuals derived from self-pollination of individual T1 plants) were assayed for the presence of the construct, using the primer pair ntpIIF (5′-TCGGCTATGACTGGGCACAAC-3′)/ntpIIR (5′-AAGAAGGCGATAGAAGGCGA-3′), designed on the ntpII gene sequence, and the primer pair 35S-F (5′-GCTCCTACAAATGCCATCA-3′)/35S-R (5′-GATAGTGGGATTGTGCGTCA-3′), designed on the 35S promoter sequence. Expression of the transgene was assessed by qPCR using the primer pair NtMLO1_qFw (5′-GTGGAAATAAGTCCAGCATTATG-3′)/NtMLO1_qRev (5′-CACCCAAAGGTACGAGTACAATC-3′).
Disease tests and Oidium neolycopersici quantification on transgenic plants
Three cuttings per T1 individuals and plants of the T2 families mentioned above were challenged with an isolate of the tomato PM fungus Oidium neolycopersici maintained at the Plant Breeding Department of the University of Wageningen, The Netherlands. The Slmlo1 mutant line and the susceptible cultivar Moneymaker (MM) were used as controls. Inoculation was performed as described by Pavan et al. (2008), by spraying plants with a suspension of conidiospores obtained from freshly sporulating leaves of heavily infected plants and adjusted to a final concentration of 4 × 104 spores/ml. Inoculated plants were grown in a greenhouse compartment at 20 ± 2 °C with 70 ± 15 % relative humidity and day-length of 16 h. Disease evaluation was carried out 15 days after inoculation, based on a visual scoring as described by Bai et al. (2008) and/or analytically, by the relative quantification of the ratio between fungal and plant gDNAs. The latter was performed by the qPCR assay reported by Huibers et al. (2013). Specifically, plant and fungal genomic DNAs were extracted from O. neolycopersici infected tomato leaves (Qiagen DNeasy Plant Mini Kit) and used for amplification with the primer pairs On-F (5′-CGCCAAAGACCTAACCAAAA-3′)/On-R (5′-AGCCAAGAGATCCGTTGTTG-3′), designed on O. neolycopersici internal transcribed spacer (ITS) sequences (GenBank accession number EU047564), and Ef-F (5′-GGAACTTGAGAAGGAGCCTAAG-3′)/Ef-R (5′-CAACACCAACAGCAACAGTCT-3′), designed on the tomato Elongation Factor 1α (Ef1α) gene (Løvdal and Lillo 2009). Relative quantification was performed by the 2–ΔΔCt method (Livak and Schmittgen 2001; Pfaffl 2001).
In silico characterization of the tobacco and potato MLO gene families
In order to retrieve tobacco and potato MLO homologs, nucleotide sequences of NtMLO1 and StMLO1 and corresponding translated sequences were used as query for BLAST (BLASTn and tBLASTn) search against the Sol Genomics Network (SGN) and the Potato Genomics Resource (Spud DB) databases, using default parameters.
The number of transmembrane domains was predicted using the online software TMHMM (http://www.cbs.dtu.dk/services/TMHMM/). The putative number of introns was obtained using the online service FGENESH of Softberry (http://www.softberry.com/). Chromosomal localization and gene position of potato MLO genes were inferred by the annotations of the Potato Genome Consortium. Finally, the MEME (http://meme.nbcr.net/) (Bailey et al. 2009) package was used to predict functional motifs in the NtMLO and StMLO protein families. Predicted tobacco NtMLO and potato StMLO proteins were used to integrate the phylogenetic tree described in the previous section, according to the same methodologies above mentioned.
Results
Identification of MLO gene sequences from cultivated Solanaceae
Two primer pairs, one designed on the untranslated sequence and the other on the coding sequence of tomato SlMLO1, were used to amplify homologous sequences from eggplant, potato and tobacco cDNAs. PCRs performed with the Sol-F1/R1 primer pair failed, thus suggesting the occurrence of polymorphic sequences in untranslated regions. In contrast, PCR performed with the Sol-F2/R2 primer pair, designed within the SlMLO1 coding sequence, resulted in single amplification products of 876 bp. Full-length sequences of a 1560 bp tobacco gene, named NtMLO1, and a 1557 bp potato gene, named StMLO1, were obtained by assembling partial gene sequences of PCR products with overlapping sequences retrieved by the interrogation of the SGN database. Amplification and sequencing of StMLO1 and NtMLO1 from potato and tobacco cDNAs provided evidence for their actual expression in leaves and validated their sequences. These were deposited in the GenBank database with the accession codes KM244715 (StMLO1) and KM244716 (NtMLO1).
In order to clone an eggplant MLO gene putatively involved in PM susceptibility, several primers were designed, based on the identification of conserved regions from the alignment of SlMLO1, StMLO1 and NtMLO1. These primers were then tested on eggplant cDNA. The SmMLO1-F2/SmMLO1-R1 primer pair produced a single PCR amplification product. The corresponding sequence of 1572 bp was named SmMLO1 and deposited in the GenBank database with the accession code KM244717.
Bioinformatic analyses support the identification of solanaceous MLO functional orthologs required for PM susceptibility
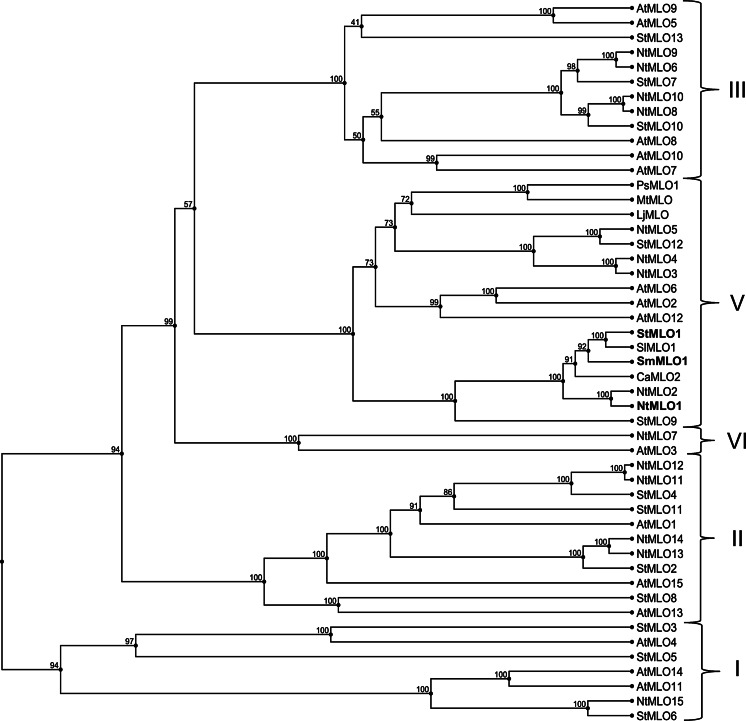
StMLO1, NtMLO1 and SmMLO1 protein sequences were used to perform a phylogenetic analysis. With strong bootstrap support, they were found to group in the phylogenetic clade V, containing all the dicot MLO homologs so far experimentally shown to be required for PM susceptibility (AtMLO2, AtMLO6, AtMLO12, SlMLO1, CaMLO2, PsMLO1, LjMLO1 and MtMLO1) (Fig. 1), thus indicating they could possibly be functionally related.
Fig. 1.
UPGMA-based tree of full-length MLO proteins. The dataset includes the tobacco NtMLO, potato StMLO and Arabidopsis AtMLO protein families, tomato SlMLO1, pepper CaMLO2, eggplant SmMLO1, pea PsMLO1, lotus LjMLO1 and barrel clover MtMLO1. Phylogenetic clades are designated with Roman numbers based on the position of AtMLO homologs, according to the nomenclature indicated by Feechan et al. (2008). Homologs identified by means of a PCR-based approach in this study (SmMLO1, StMLO1 and NtMLO1) are indicated in bold red. Numbers at each node represent bootstrap support values (out of 100 replicates)
Previous studies highlighted the presence of amino acid residues highly conserved either in the whole MLO protein family or in MLO orthologs involved in the interaction with PM fungi, which are predicted to play a key functional role (Elliott et al. 2005; Panstruga 2005). All of these residues were found to be present in the StMLO1, NtMLO1 and SmMLO1 protein sequences (Supplementary Fig. 1), providing further evidence for the identification of MLO genes required for PM susceptibility.
Finally, another strong bioinformatic indication for the identification of solanaceous MLO susceptibility genes was provided by aligning the coding sequences of StMLO1, NtMLO1 and SmMLO1 with those of the PM susceptibility genes SlMLO1 and CaMLO2, functionally characterized in tomato and pepper, respectively (Bai et al. 2008; Zheng et al. 2013) (Supplementary Fig. 2). Indeed, this revealed a very high percentage of nucleotide identity (81.4 % between tomato and tobacco, 87.5 % between tomato and eggplant and 94.8 % between tomato and potato), suggesting that all of these solanaceous MLO genes are orthologs.
Tobacco NtMLO1 complements tomato SlMLO1 in a functional complementation assay
In order to characterize NtMLO1 at the functional level, we set up an assay based on its transgenic overexpression in the previously described tomato line Slmlo1, which carries a loss-of-function mutation in the tomato SlMlo1 homolog and is thus resistant to the PM fungus O. neolycopersici (Bai et al. 2008). We hypothesised that overexpression of NtMLO1 would have restored PM susceptibility in the tomato Slmlo1 mutant line, thereby demonstrating functional conservation between NtMLO1 and SlMLO1.
After transformation, cuttings of 20 T1 transgenic individuals were challenged with O. neolycopersici. Fifteen of the tested T1 individuals showed restoration of PM symptoms (data not shown). In order to confirm this result, two T2 families of the fifteen individuals (T2_a and T2_b) derived from self-pollination of two different T1 plants were also inoculated, together with MM (the susceptible control) and the Slmlo1 mutant line (the resistant control). The presence of the overexpression construct in segregating T2 families was assessed by PCR amplification with primer pairs designed on the nptII gene and the 35S promoter (Supplementary Fig. 3). T2 individuals not carrying the overexpression construct [T2(−)], as well as individuals of the Slmlo1 mutant line, showed no NtMLO1 expression and an average of disease score of about 0.5. In contrast, T2 individuals of the two families positive for the presence of the construct [T2(+)_a and T2(+)_b] showed NtMLO1 expression and an average disease score of 1.8 and 1.7, respectively (Fig. 2 and Supplementary Fig. 4).
Fig. 2.
Effects of the transgenic expression of NtMLO1 in a tomato mlo loss-of-function genetic background. a From left to right as follows: one individual of a T2 family positive for the presence the NtMLO1 overexpression construct [T2_a(+)]; one individual of another independent T2 family positive for the presence of the NtMLO1 overexpression construct [T2_b(+)]; one T2 individual negative for the presence of the overexpression construct [T2(−)]; one individual of the tomato Slmlo1 mutant line, carrying a loss of function deletion in the SlMLO1 gene; one individual of the susceptible cultivar Moneymaker (MM). b Reports the average visual scoring of disease incidence observed on the individuals of the same two T2 families [T2_a(+) and T2_b(+)]; individuals of both T2_a and T2_b families negative for the presence of the 35S::NtMLO1 construct [T2(−)]; individuals of the Slmlo1 mutant line; individuals of the cultivar MM. The scale from 0 (completely resistant) to 3 (fully susceptible) reported by Bai et al. (2008), was used for scoring. Bars and standard errors refer to 11 T2(+)_a plants, 10 T2(+)_b plants, 9 T2(−) plants, 10 Slmlo1 plants and 10 MM plants
A NtMLO1 point mutation causing the substitution of a conserved glutamine residue results in gene loss of function
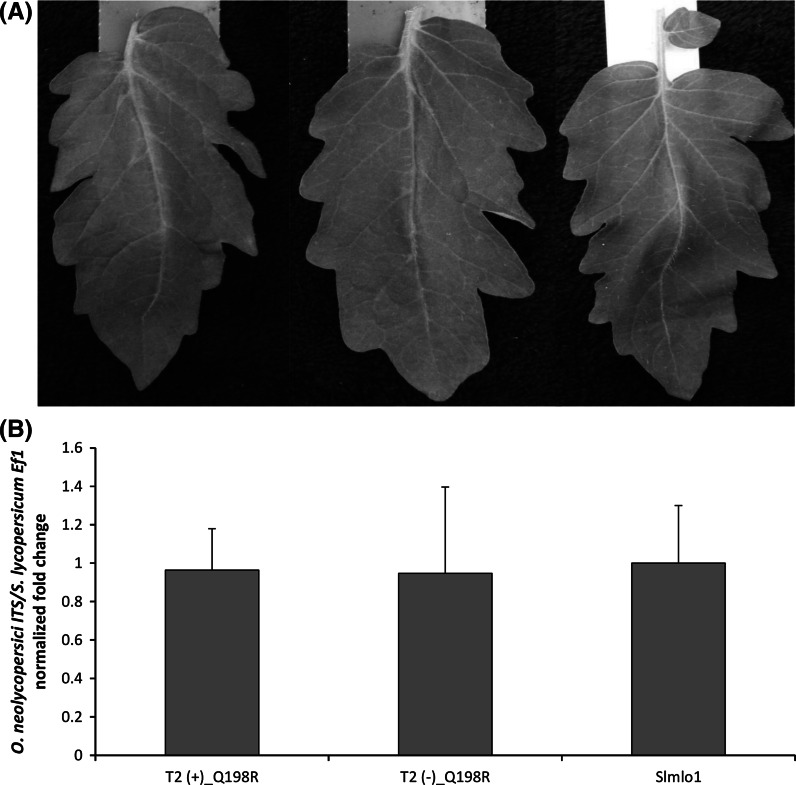
During the preparation of the 35S::NtMLO1 overexpression vector, we accidentally cloned another insert, carrying a single nucleotide polymorphism in the tobacco NtMLO1 gene. This resulted in the substitution of a glutamine residue, located in the protein second intracellular loop and previously reported to be invariable throughout the whole MLO protein family, with arginine (Q198R, Fig. 3). We could not get the same arginine-coding insert by repeating the cloning procedure several times from tobacco cDNA, so we assumed that this resulted from a mutation due to an error by the Taq polymerase used for amplification. Nonetheless, in order to study the effect of this substitution on protein function, we developed transgenic lines carrying an overexpression construct for this insert. Following O. neolycopersici inoculation, none of 20 individual T1 plants developed disease symptoms. Individuals of two independent T2 families positive for the presence of the construct [T2(+)_Q198R-a and b] were found to express the transgene, as assessed by qPCR (Supplementary Fig. 4). Nevertheless, following O. neolycopersici challenge, no PM symptoms were visible on [T2(+)_Q198R] individuals, which were phenotypically undistinguishable from those of the Slmlo1 line (Fig. 4a). In order to test whether the mutated NtMLO1 sequence maintained some residual functional activity, even so still resulting in a macroscopically resistant phenotype, we quantified, in transgenic individuals of the two T2 families, the relative fold-change of the ratio between O. neolycopersici and tomato gDNAs. Compared to the Slmlo1 line, no significant difference was found (Fig. 4b), indicating that the point nucleotide mutation causing the substitution of glutamine with arginine in the NtMLO1 protein sequence leads to complete gene loss of function.
Fig. 3.
Alignment of part of the second MLO intracellular loop from several MLO proteins experimentally shown to be required for powdery mildew susceptibility (Arabidopsis AtMLO2, AtMLO6 and AtMLO12, tomato SlMLO1, pepper CaMLO2, pea PsMLO1, lotus LjMLO1, barrel clover MtMLO1 and barley HvMLO), and NtMLO1 proteins derived from the conceptual translation of the two inserts obtained during the cloning procedure (NtMLO1 and NtMLO1-Q198R). The latter is characterized by the substitution of an invariable glutamine with arginine, whose position is indicated by an arrow
Fig. 4.
Effects of the transgenic expression of a NtMLO1 mutant sequence, resulting in the substitution of a glutamine residue with arginine in the protein second intracellular loop (Q198R). a The phenotype of a plant of the tomato loss-of-function Slmlo1 line (right) and transgenic individuals from two different T2 families (left and centre) assessed for transgene overexpression. b The relative quantification of the ratio between Oidium neolycopersici and plant gDNAs in transgenic individuals of the same T2 families assessed for the presence or absence of the overexpression construct [T2(+)_Q198R and T2(−)_Q198R, respectively] and in the tomato Slmlo1 mutant line. Bars and standard errors refer to 11 and 7 transgenic individuals for NtMLO1_Q198R-a and b, respectively, and 10 Slmlo1 plants
In silico characterization of tobacco and potato MLO families
Recently released sequences from potato (group Phureja DM1) and tobacco (cv. Basma Xanthi) prompted us to perform a genome-wide search aiming to characterize the MLO gene families in these species. This search revealed a total of 15 and 13 predicted tobacco NtMLO and potato StMLO loci, respectively, which were named according to the nomenclature specified in Supplementary Tables 1 and 2. A predicted tobacco coding sequence, referred to as mRNA_127718_cds in the Sol Genomics Database, was found to be identical to NtMLO1. No sequence fully matching with StMLO1 could be identified by the interrogation of the Potato Genomics Resource database, but in its place a partial gene sequence showing 100 % of identity with the same gene.
For tobacco and potato MLO proteins, amino acid length and number of transmembrane domains were inferred (Supplementary Table 1 and Supplementary Table 2). In addition, information on chromosomal localization and intron number was available for predicted StMLO genes (Supplementary Table 2).
The tobacco NtMLO and potato StMLO protein families were used as input to search for conserved motifs, using an approach similar to the one previously reported by Deshmukh et al. (2014). We looked for motifs with length ranging from 40 to 70 residues and shared by at least three homologs. For each of the two families, seven motifs were identified. Of these, five were found to be at least partially matching with those identified in the soybean protein family (Deshmukh et al. (2014) (Supplementary Table 3).
A comparative analysis was carried out in order to establish phylogenetic relationships between the NtMLO and the StMLO protein families and MLO proteins from other dicot plant species. The analysis resulted in the distinction of five clades, designated with Roman numbers based on the position of Arabidopsis AtMLO homologs, according to the nomenclature indicated by Feechan et al. (2008) (Fig. 1). Besides NtMLO1 and StMLO1, additional NtMLO (NtMLO2, NtMLO3, NtMLO4 and NtMLO5) and StMLO (StMLO9 and StMLO12) homologs were found to group in clade V together with all dicot MLO proteins previously associated with PM susceptibility.
Discussion
In previous studies, we functionally characterized tomato SlMLO1 and pepper CaMLO2 as two solanaceous MLO susceptibility genes, as their inactivation was causally associated with PM resistance (Bai et al. 2008; Zheng et al. 2013). Starting from this information, we followed here a combined approach based on database search and PCR amplification, which resulted in the isolation of three MLO genes from other widely distributed solanaceous species affected by the PM disease, namely eggplant (SmMLO1), potato (StMLO1) and tobacco (NtMLO1). PM disease represents one of the most important fungal diseases of tobacco and eggplant (Bubici and Cirulli 2008; Darvishzadeh et al. 2010) and in conducive environments may lead to important economic losses in potato cultivation (Glawe et al. 2004).
A chain of evidence, based on phylogenetic relatedness (Fig. 1) and sequence conservation with other known PM susceptibility genes and proteins (Supplementary Fig. 1 and Supplementary Fig. 2) was provided, suggesting the identification of solanaceous orthologs of SlMLO1 and CaMLO2. Aiming at the functional characterization of NtMLO1, we set up an assay based on its heterologous overexpression in a tomato mlo-mutant genetic background, taking advantage from the availability of a tomato resistant line and routine protocols for tomato genetic transformation (Bai et al. 2008). Success of such an assay, as demonstrated by the restoration of symptoms in transgenic plants (Fig. 2), provides a final evidence for the role of NtMLO1 as a PM susceptibility gene. Although it was not proven at the functional level, we speculate that both StMLO1 and SmMLO1 are involved in PM susceptibility in potato and eggplant, as they are, at the nucleotide level, even closer than NtMLO1 to SlMLO1 and CaMLO2.
While completing this work, newly released sequences of potato and tobacco became available. Thus, a genome-wide search was performed, which allowed to retrieve additional MLO homologs and, presumably, to characterize the complete tobacco and potato MLO gene families. Phylogenetic analysis using these sequences highlighted the presence of additional NtMLO and StMLO proteins in clade V, previously shown to group dicot MLO homologs acting as PM susceptibility factors (Fig. 1). Functional redundancy of MLO homologs belonging to this clade has been shown to occur in Arabidopsis thaliana, as the simultaneous inactivation of the three homolog genes AtMLO2, AtMLO6 and AtMLO12 is required to result in complete PM resistance. Thus, functional analyses, such as the transgenic complementation test above mentioned, might lead to the identification of additional solanaceous MLO homologs playing a role in the interaction with PM fungi.
Interestingly, due to a polymerase error during the cloning procedure, we also had the opportunity to verify the crucial role of a glutamine residue localized in the second intracellular MLO domain. This amino acid has been shown to be invariable throughout the whole MLO protein family and therefore predicted to be fundamental for the role of MLO proteins as PM susceptibility factors (Elliott et al. 2005). Indeed, its replacement with arginine in tobacco NtMLO1 (Fig. 3) resulted in complete failure of transgenic complementation, as inferred by visual scoring and relative quantification of fungal gDNA with respect to plant gDNA (Fig. 4). This result represents a complement to earlier investigations addressed to the functional characterization of MLO proteins (Reinstädler et al. 2010; Pavan et al. 2013).
A growing body of experimental evidence supports the view that mlo-based resistance can be conveniently pursued as a strategy to cope with the PM disease in practical breeding (Pavan et al. 2010). Therefore, we predict that results here provided might be of great interest for future activities aimed at the introduction of PM resistance in Solanaceae. Targeted identification of mutations of MLO susceptibility genes can be achieved through conventional approaches of TILLING (targeted induced local lesions in genomes) or RNA interference (McCallum et al. 2000; Matthew 2004). In addition, cutting-edge technologies of genome editing are also available to the breeder, based on zinc finger nucleases (ZFNs), clustered regularly interspaced short palindromic repeat (CRISPR) and transcription activator-like effector nucleases (TALEN) (Gaj and Gersbach 2013; Terns and Terns 2014). Noteworthy, a TALEN-based approach has been recently successfully applied to introduce PM resistance in bread wheat through simultaneous targeting of three homoelog MLO alleles, as mentioned in Wang et al. (2014).
Electronic supplementary material
Acknowledgments
This work was supported by the Italian Ministry of University and Research through the project GenHort PON R&C.
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Michela Appiano and Stefano Pavan have contributed equally to this work.
References
- Acevedo-Garcia J, Kusch S, Panstruga R. Magical mystery tour: MLO proteins in plant immunity and beyond. N Phytol. 2014 doi: 10.1111/nph.12889. [DOI] [PubMed] [Google Scholar]
- Assaad FF, Qiu J-L, Youngs H, Ehrhardt D, Zimmerli L, Kalde M, Wanner G, Peck SC, Edwards H, Ramonell K, Somerville CR, Thordal-Christensen H. The PEN1 syntaxin defines a novel cellular compartment upon fungal attack and is required for the timely assembly of papillae. Mol Biol Cell. 2004;15:5118–5129. doi: 10.1091/mbc.E04-02-0140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai Y, Pavan S, Zheng Z, Zappel NF, Reinstädler A, Lotti C, De Giovanni C, Ricciardi L, Lindhout P, Visser R, Theres K, Panstruga R. Naturally occurring broad-spectrum powdery mildew resistance in a Central American tomato accession is caused by loss of Mlo function. Mol Plant Microbe Interact. 2008;21:30–39. doi: 10.1094/MPMI-21-1-0030. [DOI] [PubMed] [Google Scholar]
- Bailey TL, Boden M, Buske FA, Frith M, Grant CE, Clementi L, Ren J, Li WW, Noble WS. MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res. 2009;37:W202–W208. doi: 10.1093/nar/gkp335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bubici G, Cirulli M. Screening and selection of eggplant and wild related species for resistance to Leveillula taurica. Euphytica. 2008;164:339–345. doi: 10.1007/s10681-008-9663-z. [DOI] [Google Scholar]
- Büschges R, Hollricher K, Panstruga R, Simons G, Wolter M, Frijters A, Van Daelen R, Van der Lee T, Diergaarde P, Groenendijk J, Töpsch S, Vos P, Salamini F, Schulze-Lefert P. The barley Mlo gene: a novel control element of plant pathogen resistance. Cell. 1997;88:695–705. doi: 10.1016/S0092-8674(00)81912-1. [DOI] [PubMed] [Google Scholar]
- Consonni C, Humphry ME, Hartmann HA, Livaja M, Durner J, Westphal L, Vogel J, Lipka V, Kemmerling B, Schulze-Lefert P, Somerville SC, Panstruga R. Conserved requirement for a plant host cell protein in powdery mildew pathogenesis. Nat Genet. 2006;38:716–720. doi: 10.1038/ng1806. [DOI] [PubMed] [Google Scholar]
- Dangl JL, Horvath DM, Staskawicz BJ. Pivoting the plant immune system from dissection to deployment. Science. 2013;341:746–751. doi: 10.1126/science.1236011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darvishzadeh R, Alavi R, Sarrafi A. Resistance to Powdery Mildew (Erysiphe cichoracearum DC.) in oriental and semi-oriental tobacco germplasm under field conditions. J Crop Improv. 2010;24:122–130. doi: 10.1080/15427520903559425. [DOI] [Google Scholar]
- Deshmukh R, Singh VK, Singh BD. Comparative phylogenetic analysis of genome-wide Mlo gene family members from Glycine max and Arabidopsis thaliana. Mol Genet Genomic. 2014 doi: 10.1007/s00438-014-0811-y. [DOI] [PubMed] [Google Scholar]
- Devoto A, Hartmann HA, Piffanelli P, Elliott C, Simmons C, Taramino G, Goh CS, Cohen FE, Emerson BC, Schulze-Lefert P, Panstruga R. Molecular phylogeny and evolution of the plant-specific seven-transmembrane MLO family. J Mol Evol. 2003;56:77–88. doi: 10.1007/s00239-002-2382-5. [DOI] [PubMed] [Google Scholar]
- Elliott C, Müller J, Miklis M, Bhat RA, Schulze-Lefert P, Panstruga R. Conserved extracellular cysteine residues and cytoplasmic loop–loop interplay are required for functionality of the heptahelical MLO protein. Biochem J. 2005;385:243–254. doi: 10.1042/BJ20040993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feechan A, Jermakow AM, Torregrosa L, Panstruga R, Dry IB. Identification of grapevine MLO gene candidates involved in susceptibility to powdery mildew. Funct Plant Biol. 2008;35:1255–1266. doi: 10.1071/FP08173. [DOI] [PubMed] [Google Scholar]
- Gaj T, Gersbach CA, Barbas CF., III ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol. 2013;31:397–405. doi: 10.1016/j.tibtech.2013.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glawe DA. The powdery mildews: a review of the world’s most familiar (yet poorly known) plant pathogens. Annu Rev Phytopathol. 2008;46:27–51. doi: 10.1146/annurev.phyto.46.081407.104740. [DOI] [PubMed] [Google Scholar]
- Glawe DA, Toit LJd, Pelter GQ. First report of powdery mildew on potato caused by Leveillula taurica in North America. Plant Health Prog. 2004 [Google Scholar]
- Harland SC. Inheritance of immunity to mildew in Peruvian forms of Pisum sativum. Heredity. 1948;2:263–269. doi: 10.1038/hdy.1948.15. [DOI] [PubMed] [Google Scholar]
- Hewitt HG. Fungicides in crop protection. Wallingford: CAB International; 1998. [Google Scholar]
- Huibers RP, Loonen AEHM, Gao D, Van den Ackerveken G, Visser RGF, Bai Y. Powdery mildew resistance in tomato by impairment of SlPMR4 and SlDMR1. PLoS One. 2013;8(6):e67467. doi: 10.1371/journal.pone.0067467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humphry M, Reinstädler A, Ivanov S, Bisseling T, Panstruga R. Durable broad-spectrum powdery mildew resistance in pea er1 plants is conferred by natural loss-of-function mutations in PsMLO1. Mol Plant Pathol. 2011;12:866–878. doi: 10.1111/j.1364-3703.2011.00718.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Q, Zhu H. Molecular evolution of the MLO gene family in Oryza sativa and their functional divergence. Gene. 2008;409:1–10. doi: 10.1016/j.gene.2007.10.031. [DOI] [PubMed] [Google Scholar]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Løvdal T, Lillo C. Reference gene selection for quantitative real-time PCR normalization in tomato subjected to nitrogen, cold, and light stress. Anal Biochem. 2009;387:238–242. doi: 10.1016/j.ab.2009.01.024. [DOI] [PubMed] [Google Scholar]
- Lyngkjaer MF, Newton AC, Atzema JL, Baker SJ. The Barley mlo-gene: an important powdery mildew resistance source. Agronomie. 2000;20:745–756. doi: 10.1051/agro:2000173. [DOI] [Google Scholar]
- Matthew L. RNAi for plant functional genomics. Comp Funct Genomics. 2004;5:240–244. doi: 10.1002/cfg.396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCallum CM, Comai L, Greene EA, Henikoff S. Targeting induced local lesions in genomes (TILLING) for plant functional genomics. Plant Physiol. 2000;123:439–442. doi: 10.1104/pp.123.2.439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCormick S, Niedermeyer J, Fry J, Barnason A, Horsch R, Fraley R. Leaf disc transformation of cultivated tomato (L. esculentum) using Agrobacterium tumefaciens. Plant Cell Rep. 1986;5:81–84. doi: 10.1007/BF00269239. [DOI] [PubMed] [Google Scholar]
- Panstruga R. Discovery of novel conserved peptide domains by ortholog comparison within plant multi-protein families. Plant Mol Biol. 2005;59:485–500. doi: 10.1007/s11103-005-0353-0. [DOI] [PubMed] [Google Scholar]
- Pavan S, Zheng Z, Borisova M, Van Den Berg P, Lotti C, De Giovanni C, Lindhout P, De Jong H, Ricciardi L, Visser RGF, Bai Y. Map- vs. homology-based cloning for the recessive gene ol-2 conferring resistance to tomato powdery mildew. Euphytica. 2008;162:91–98. doi: 10.1007/s10681-007-9570-8. [DOI] [Google Scholar]
- Pavan S, Jacobsen E, Visser RGF, Bai Y. Loss of susceptibility as a novel breeding strategy for durable and broad-spectrum resistance. Mol Breed. 2010;25:1–12. doi: 10.1007/s11032-009-9323-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavan S, Schiavulli A, Appiano M, Marcotrigiano AR, Cillo F, Visser RGF, Bai Y, Lotti C, Ricciardi L. Pea powdery mildew er1 resistance is associated to loss-of-function mutations at a MLO homologous locus. Theor Appl Genet. 2011;123:1425–1431. doi: 10.1007/s00122-011-1677-6. [DOI] [PubMed] [Google Scholar]
- Pavan S, Schiavulli A, Appiano M, Miacola C, Visser RGF, Bai Y, Lotti C, Ricciardi L. Identification of a complete set of functional markers for the selection of er1 powdery mildew resistance in Pisum sativum L. Mol Breed. 2013;31:247–253. doi: 10.1007/s11032-012-9781-0. [DOI] [Google Scholar]
- Pessina S, Pavan S, Catalano D, Gallotta A, Visser R, Bai Y, Malnoy M, Schouten H. Characterization of the MLO gene family in Rosaceae and gene expression analysis in Malus domestica. BMC Genom. 2014;15:618. doi: 10.1186/1471-2164-15-618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfaffl MW. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001;29(9):e45. doi: 10.1093/nar/29.9.e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinstädler A, Müller J, Czembor JH, Piffanelli P, Panstruga R. Novel induced mlo mutant alleles in combination with site-directed mutagenesis reveal functionally important domains in the heptahelical barley Mlo protein. BMC Plant Biol. 2010;10:31. doi: 10.1186/1471-2229-10-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rozen S, Skaletsky H. Primer3 on the WWW for general users and for biologist programmers. Method Mol Biol. 2000;132:365–386. doi: 10.1385/1-59259-192-2:365. [DOI] [PubMed] [Google Scholar]
- Schouten HJ, Krauskopf J, Visser RGF, Bai Y. Identification of candidate genes required for susceptibility to powdery or downy mildew in cucumber. Euphytica. 2014 [Google Scholar]
- Sierro N, Battey JND, Ouadi S, Bakaher N, Bovet L, Willig A, Goepfert S, Peitsch MC, Ivanov NV. The tobacco genome sequence and its comparison with those of tomato and potato. Nat Commun. 2014;5:3833. doi: 10.1038/ncomms4833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terns RM, Terns MP. CRISPR-based technologies: prokaryotic defense weapons repurposed. Trends Genet. 2014;30(3):111–118. doi: 10.1016/j.tig.2014.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The Potato Genome Sequencing Consortium Genome sequence and analysis of the tuber crop potato. Nature. 2011;475:189–195. doi: 10.1038/nature10158. [DOI] [PubMed] [Google Scholar]
- Wang Y, Cheng X, Shan Q, Zhang Y, Liu J, Gao C, Qiu JL. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat Biotechnol. 2014 doi: 10.1038/nbt.2969. [DOI] [PubMed] [Google Scholar]
- Zheng Z, Nonomura T, Appiano M, Pavan S, Matsuda Y, Toyoda H, Wolters AMA, Visser RGF, Bai Y. Loss of function in Mlo orthologs reduces susceptibility of pepper and tomato to powdery mildew disease caused by Leveillula taurica. PLoS One. 2013;8(7):e70723. doi: 10.1371/journal.pone.0070723. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.