Abstract
Background:
The variable numbers of tandem-repeat (VNTR) alleles at the phenylalanine hydroxylase (PAH) gene have been used in carrier detection and prenatal diagnosis in phenylketonuria families. This study was carried out to analyze VNTR alleles at the PAH gene in Iranian Azeri Turkish population.
Methods:
In this study, 200 alleles from general population were studied by PCR.
Results:
The frequencies of VNTR alleles were 45%, 46%, 2%, 3%, 1%, and 3% in studied group regarding 3, 8, 9, 11, 12, and 13 repeat copies, respectively. Statistically significant differences were not found between expected and observed frequencies of VNTR genotypes (P > 0.05).
Conclusions:
VNTR alleles with three and eight repeats were frequent, and the VNTR alleles with 13 repeats showed 3% frequency in the tested group. This study is the first report on tested population genetic structure using VNTR alleles at the PAH gene.
Key Words: Phenylalanine hydroxylase, Population genetics, Variable numbers of tandem-repeat
INTRODUCTION
The phenylalanine hydroxylase (PAH) is a hepatic enzyme involved in L-phenylalanine catabolism [1]. A large amount of variations and mutations within PAH gene result in defective form of enzyme [1]. Complete or nearly complete deficiency of PAH enzyme is responsible for different phenotypes of hyper-phenylalaninemia and phenyl-ketonuria (PKU) [2].
PAH gene is located on chromosome 12 (band region q22-24.1) and consists of 13 exons and 12 introns [3]. The human PAH gene is 100 kb long and covers 5’ and 3’ un-translated regions [3]. A wide range of (more than 500) disease-causing mutations has been found in human PAH gene [4]. In this regard, it is not possible to detect disease-causing mutations by a simple method.
For prenatal diagnosis and carrier screening of PKU, several linkage analyses may be informative for study such as short tandem repeats (TCTAT)n within intron 3 and multi-allelic variable numbers of tandem-repeat (VNTR), which is a 30-bp AT-rich tandem-repeat system located in 3’ un-translated region of the PAH gene [5, 6]. Multi-allelic VNTR at the PAH gene could be understood as a simple, rapid and highly informative molecular genetics procedure for diagnosis and carrier screening of PKU [6]. These polymorphic DNA markers have been studied in different populations and showed significant diversity between inter- and intra-geographical regions [7]. A DNA marker may be informative if any randomly selected individual is expected to be heterozygous for that marker, and the informativeness of the markers is one of the most important points in genetic linkage mapping of human chromosomes [8-10]. Importance of marker loci for genetic linkage analysis depends on the alleles and allele frequencies in the population [11]. A marker is known polymorphic if it has at least two alleles [11]. The rate of most common allele in the population is about 99% [12]. Informativeness of polymorphic DNA markers is determined by the calculation of heterozygosity and polymorphism information content (PIC) value, which was used for a codominant marker in a linkage study [12-14]. A proband is defined as informative if we conclude from proband genotype, which marker allele is linked to the disease-causing allele [13, 14]. Marker allele and disease-causing allele are co-inherited in subsequent generations [14]. Iran is defined as one of the most heterogeneous population in the world [9, 15]. Therefore in this study, VNTR alleles at PAH gene in an Iranian Azeri Turkish population were analyzed.
MATERIALS AND METHODS
This study was approved by the ethics committee of Urmia University of Medical Sciences (Urmia, Iran). Between 2012 and 2014, a total number of 100 healthy subjects with age ranging from 20 to 60 years voluntarily entered the study. We studied genetically unrelated healthy subjects who were resident in the West Azerbaijan Province of Iran. All participants were matched for ethnicity and geographical region. Studied individuals were selected in genetic counseling sessions taken place in Genetic Center of Motahari Hospital (Urmia). All healthy subjects were chosen randomly regarding their past medical history and exclusion of specific findings. A written informed consent was obtained from the studied subjects. DNA was isolated from blood samples using a ‘salting out’ method [16]. Analysis of the genomic DNA was carried out by PCR using the 5’-ttt taa tgt tct cac ccg cc-3’ and 5’-aag aat ccc atc tct cag ag-3’ primers [5]. The PCR conditions on the thermal cycler (Eppendorf) was as follows: an initial denaturation step at 95°C for 5 min, followed by 35 cycles of 1 min at 95°C, 1 min at 55°C, 1 min at 72°C, and a final extension step of 5 min at 72°C. Amplified PCR fragments were separated on 2% agarose gel and stained with ethidium bromide. Presence or absence of different amplified DNA fragments, which are responsible for different alleles, were visualized under UV light. Electrophoretic analysis of the amplified PCR products verified six distinct sizes 325, 475, 505, 565, 595, and 625 bp. These size differences reflected the presence of 3, 8, 9, 11, 12, and 13 copies of the repeated unit, respectively.
Statistical analysis. The frequencies of VNTR alleles and genotypes were determined via direct counting and dividing by the number of chromosomes and the number of subjects. The Hardy-Weinberg equilibrium (HWE) test for multiple VNTR alleles were carried out regarding equation (p + q + r + s + t + u)2 = 1 at a locus. In this study we used runs of homozygosity (ROH), heterozygosity (H), and PIC value to detect the informativeness of polymorphic VNTR alleles. ROH [17] and H [18] values at the PAH locus in the studied population were calculated by the following equations:
=
PIC value was determined via formula (12):
+
Where Pi and Pj are the population frequency of the ith and jth alleles, n= number of different alleles at single locus. Degree of freedom, the chi-square (χ2) distribution, χ2 critical value, and P value were analyzed for testing the agreement between the observed and expected frequencies of VNTR genotypes. Microsoft Office Excel 2010 was used for statistical analysis. χ2 critical value was found from χ2 table based on degree of freedom and α = 0.05. Significance level was accepted at P value less than 0.05.
RESULTS
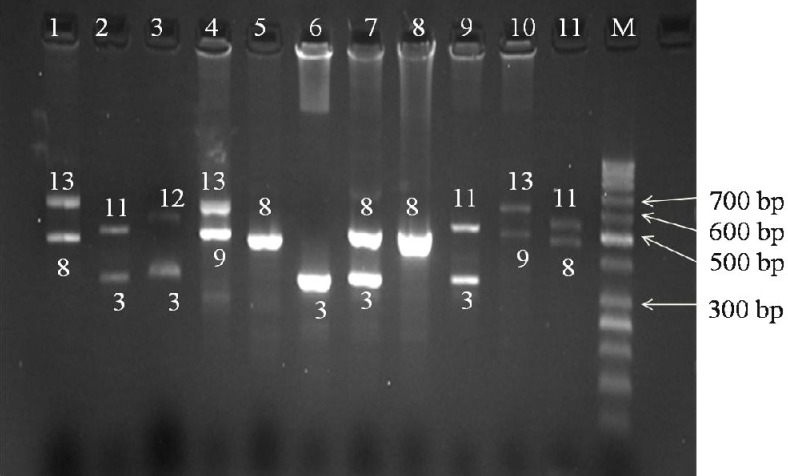
In this investigation, 200 alleles were studied in a general population. Number and size range of VNTR alleles, HWE, ROH, H, and PIC values were also analyzed. The results of this study are summarized in Table 1. Of 200 chromosomes, 6 VNTR alleles were identified at the PAH gene. The PCR products were six alleles of 325, 475, 505, 565, 595, and 625 bp containing 3, 8, 9, 11, 12, and 13 repeat copies, respectively. The frequencies of VNTR alleles at the PAH gene were 90 (45%), 92 (46%), 4 (2%), 6 (3%), 2 (1%), and 6 (3%) in studied group regarding 3, 8, 9, 11, 12, and 13 repeat copies, respectively. VNTR 5, 6, 7, and 10 alleles were not found in our study. The frequency of VNTR genotypes meets HWE, and the analysis of the χ2 distribution showed that there is an agreement between the observed and those of expected frequencies (χ2 = 2.33E-20 < 31.41 (χ2 CV), α = 0.05, degree of freedom = 20, P = 1). The findings of this study showed that the observed ROH and expected ROH were 0.146 and 0.416, demonstrating that the observed H and expected H values were 0.854 and 0.583, respectively. No statistically significant differences were found between observed H and expected H (χ2 = 0.18 < 3.84, P = 0.67). By calculation of PIC value using the formula for PIC, we found that PIC value for VNTR alleles at the PAH gene equals 0.496. Electrophoretic analysis of different VNTR alleles at the PAH gene, including 3, 8, 9, 11, 12, and 13 copies of the repeated units in 11 samples are shown in Figure 1.
Table 1.
Variable numbers of tandem-repeat genotypes in Iranian Azeri Turkish population
| No. | Genotype | Observed | % Observed | Expected | % Expected |
|---|---|---|---|---|---|
| 1 | 3,3 | 26 | 0.26 | 0.2025 | 20.25 |
| 2 | 3,8 | 32 | 0.32 | 0.4140 | 41.40 |
| 3 | 3,11 | 4 | 0.04 | 0.0270 | 2.70 |
| 4 | 3,12 | 2 | 0.02 | 0.009 | 0.90 |
| 5 | 8,8 | 28 | 0.28 | 0.2116 | 21.16 |
| 6 | 8,11 | 2 | 0.02 | 0.0276 | 2.76 |
| 7 | 8,13 | 2 | 0.02 | 0.0276 | 2.76 |
| 8 | 9,13 | 4 | 0.04 | 0.0012 | 0.12 |
| 9 | 9,3 | 0 | 0 | 0.0180 | 1.80 |
| 10 | 13,3 | 0 | 0 | 0.0270 | 2.70 |
| 11 | 8,9 | 0 | 0 | 0.0184 | 1.84 |
| 12 | 8,12 | 0 | 0 | 0.0092 | 0.92 |
| 13 | 9,9 | 0 | 0 | 0.0004 | 0.04 |
| 14 | 9,11 | 0 | 0 | 0.0012 | 0.12 |
| 15 | 9,12 | 0 | 0 | 0.0004 | 0.04 |
| 16 | 11,11 | 0 | 0 | 0.0009 | 0.09 |
| 17 | 11,12 | 0 | 0 | 0.0006 | 0.06 |
| 18 | 11,13 | 0 | 0 | 0.0018 | 0.18 |
| 19 | 12,12 | 0 | 0 | 0.0001 | 0.01 |
| 20 | 12,13 | 0 | 0 | 0.0006 | 0.06 |
| 21 | 13,13 | 0 | 0 | 0.0009 | 0.09 |
Fig. 1.
Identification of VNTR alleles in 11 samples by PCR test in Iranian Azeri Turkish general population. PCR yields fragments of 325, 475, 505, 565, 595, and 625 bp containing 3, 8, 9, 11, 12, and 13 repeat copies, respectively. Lane 1, 8/13; Lane 2, 3/11; Lane 3, 3/12; Lane 4, 9/13; Lane 5, 8/8; Lane 6, 3/3; Lane 7, 3/8; Lane 8, 8/8; Lane 9, 3/11; Lane 10, 9/13; Lane 11, 8/11; M: 50 bp DNA ladder.
DISCUSSION
The frequencies of multi-allelic VNTR at the PAH gene is significantly different among ethnic groups [19]. A total of six VNTR alleles at the PAH gene were recognized. The frequencies of VNTR were 45%, 46%, 2%, 3%, 1%, and 3% at the PAH gene in Iranian Azeri Turkish population regarding 3, 8, 9, 11, 12, and 13 repeat copies, respectively. In genetic loci, including multiple alleles (more than 10), the number of genotype(s) is/are large and some of the other genotypes are absent. VNTR 5, 6, 7, and 10 were also absent in this study. The VNTR allele with three and eight repeat copies had the highest frequency in the studied group. In agreement with other studies [15, 20, 21], we here reported the VNTR alleles with 13 copies in Iranian Azeri Turkish population (West Azerbaijan Province of Iran). Interestingly, the VNTR alleles with 13 copies at the PAH locus are specific to Iranian population [15, 20, 21]. Our findings implied that VNTR alleles at the PAH gene are known to be moderately informative [22]. The high rate of heterozygosity at the PAH earns a great number of genetic variability. In this investigation, the observed heterozygosity was 0.854. This high rate of heterozygosity was consistent to Fazeli and Vallian (70%) [21] and Kamkar et al. (66%) [15]. These findings demonstrate a high degree of heterozygosity for the VNTR alleles at the PAH gene in Iranian population. Considering random mating, PIC value for VNTR alleles at the PAH gene was 49.6% in this study. PIC value was also 70%, 32%, and 66% in European Caucasians, Chinese, and Iranian [23, 24], respectively. Highly informative level of PIC is set at PIC value ≥ 0.7 [22]. At a PIC of 1, the marker would have multiple alleles at a locus [13, 24]. It is clear that markers with higher number of alleles have higher PIC value, and they are considered to be more informative. VNTR alleles in the PAH gene was found to be moderately informative in tested group (0.49 vs. 0.70) [22]. The findings of the present study indicate that the degree of heterozygosity for the VNTR markers at the PAH locus was high.
It can be concluded that the VNTR alleles with three and eight repeats are frequent and the frequency of the VNTR allele with 13 repeats is 3% in Iranian Azeri Turkish population. This study is the first report on tested population genetic structure using VNTR alleles at the PAH gene.
The present report is the first in its own kind in tested group and describes population genetic structure using VNTR alleles at the PAH gene.
ACKNOWLEDGMENTS
The authors would like to thank all of the participants for their involvement in this study.
References
- 1.Flydal MI, Martinez A. Phenylalanine hydroxylase: function, structure, and regulation. IUBMB Life. 2013 Apr;65(4):341–9. doi: 10.1002/iub.1150. [DOI] [PubMed] [Google Scholar]
- 2.Mitchell JJ, Trakadis YJ, Scriver CR. Phenylalanine hydroxylase deficiency. Genet Med. 2011 Aug;13:697–707. doi: 10.1097/GIM.0b013e3182141b48. [DOI] [PubMed] [Google Scholar]
- 3.Hoang L, Byck S, Prevost L, Scriver CR. PAH Mutation Analysis Consortium Database: a database for disease-producing and other allelic variation at the human PAH locus. Nucleic Acids Res. 1996 Jan;24(1):127–31. doi: 10.1093/nar/24.1.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Williams RA, Mamotte CD, Burnett JR. Phenyl-ketonuria: an inborn error of phenylalanine metabolism. Clin Biochem Rev. 2008 Feb;29(1):31–41. [PMC free article] [PubMed] [Google Scholar]
- 5.Zschocke J, Graham CA, Carson DJ, Nevin NC. Phenylketonuria mutation analysis in Northern Ireland: a rapid stepwise approach. Am J Hum Genet. 1995 Dec;57(6):1311–7. [PMC free article] [PubMed] [Google Scholar]
- 6.Haghighatnia A, Vallian S, Mowla J, Fazeli Z. Genetic Diversity and Balancing Selection within the Human Phenylalanine Hydroxylase (PAH) Gene Region in Iranian Population. Iran J Public Health. 2012;41(5):97–104. [PMC free article] [PubMed] [Google Scholar]
- 7.Goltsov AA, Eisensmith RC, Konecki DS, Lichter-Konecki U, Woo SL. Associations between mutations and a VNTR in the human phenylalanine hydroxylase gene. Am J Hum Genet. 1992 Sep;51(3):627–36. [PMC free article] [PubMed] [Google Scholar]
- 8.Ellegren H. Microsatellites: simple sequences with complex evolution. Nat Rev Genet. 2004 Jun;5(6):435–45. doi: 10.1038/nrg1348. [DOI] [PubMed] [Google Scholar]
- 9.Fazeli Z, Vallian S. An artifact band frequently associated with variable number of tandem repeat marker at phenylalanine hydroxylase gene: application in carrier detection and prenatal diagnosis of phenylketonuria. Mol Biol Rep. 2011 Jun;38(5):3395–9. doi: 10.1007/s11033-010-0448-9. [DOI] [PubMed] [Google Scholar]
- 10.Todd S, Sherman SL, Naylor SL. Dinucleotide repeat loci contribute highly informative genetic markers to the human chromosome 2 linkage map. Genomics. 1993 Jun;16(3):612–8. doi: 10.1006/geno.1993.1238. [DOI] [PubMed] [Google Scholar]
- 11.Goddard KA, Hopkins PJ, Hall JM, Witte JS. Linkage disequilibrium and allele-frequency distributions for 114 single-nucleotide polymorphisms in five populations. Am J Hum Genet. 2000 Jan;66(1):216–34. doi: 10.1086/302727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shete S, Tiwari H, Elston RC. On estimating the heterozygosity and polymorphism information content value. Theor Popul Biol. 2000 May;57(3):265–71. doi: 10.1006/tpbi.2000.1452. [DOI] [PubMed] [Google Scholar]
- 13.Hildebrand CE, David C, Torney WRP, Wagner P. Informativeness of polymorphic DNA markers. Los Alamos Science. 1992;20(20):100–102. [Google Scholar]
- 14.Botstein D, White RL, Skolnick M, Davis RW. Construction of a genetic linkage map in man using restriction fragment length polymorphisms. Am J Hum Genet. 1980 May;32(3):314–31. [PMC free article] [PubMed] [Google Scholar]
- 15.Kamkar M, Saadat M, Saadat I, Haghighi G. Report of VNTR with 13 repeats linked to phenylalanine hydroxylase locus in unaffected members of two PKU families. Iran Biomed J. 2003;7:89–90. [Google Scholar]
- 16.Miller SA, Dykes DD, Polesky HF. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988 Feb;16 doi: 10.1093/nar/16.3.1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Guldberg P, Levy HL, Hanley WB, Koch R, Matalon R, Rouse BM, Trefz F, de la Cruz F, Henriksen KF, Güttler F. Phenylalanine hydroxylase gene mutations in the United States: report from the Maternal PKU Collaborative Study. Am J Hum Genet. 1996 Jul;59(1):84–94. [PMC free article] [PubMed] [Google Scholar]
- 18.Kidd JR, Pakstis AJ, Zhao H, Lu RB, Okonofua FE, Odunsi A, et al. Haplotypes and linkage disequilibrium at the phenylalanine hydroxylase locus, PAH, in a global representation of populations. Am J Hum Genet. 2000 Jun;66(6):1882–99. doi: 10.1086/302952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Akhmetova VL, Viktorova TV, Khusnutdinova EK. Molecular-genetic analysis of VNTR polymorphism in alleles of the phenylalanine hydroxylase gene in inhabitants of the Volga-Urals region. Genetika. 2000 Aug;36(8):1161–5. [PubMed] [Google Scholar]
- 20.Vallian S, Barahimi E. Analysis of the importance of PAHVNTR marker in carrier detection of phenyl ketonuria disease in Isfahan population. Genetics in the 3rd Millennium. 2005;3:572–576. [Google Scholar]
- 21.Fazeli Z, Vallian S. Estimation haplotype frequency of BglII/EcoRI/VNTR markers at the PAH gene region in Iranian population. Int J Hum Genet. 2009;9(2):115–121. [Google Scholar]
- 22.Maddox JF, Davies KP, Crawford AM, Hulme DJ, Vaiman D, Cribiu EP, Freking BA, Beh KJ, Cockett NE, Kang N, Riffkin CD, Drinkwater R, Moore SS, Dodds KG, Lumsden JM, van Stijn TC, Phua SH, Adelson DL, Burkin HR, Broom JE, Buitkamp J, Cambridge L, Cushwa WT, Gerard E, Galloway SM, Harrison B, Hawken RJ, Hiendleder S, Henry HM, Medrano JF, Paterson KA, Schibler L, Stone RT, van Hest B. An enhanced linkage map of the sheep genome comprising more than 1000 loci. Genome Res. 2001 Jul;11(7):1275. doi: 10.1101/gr.135001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Eisensmith RC, Goltsov AA, Woo SLC. A simple, rapid, and highly informative PCR based procedure for prenatal diagnosis and carrier screening of phenylketonuria. Prenat Diagn. 1994 Dec;14:1113–1118. doi: 10.1002/pd.1970141204. [DOI] [PubMed] [Google Scholar]
- 24.Hosseini-Mazinani SM, Koochmeshgi J, Khazaee-Koohpar Z, Hosein-Pur-Nobari N, Seifati SM. Carrier detection of phenylketonuria in Iranian families by variable number tandem-repeat polymorphism analysis. East Mediterr Health J. 2008;14(6):1445–51. [PubMed] [Google Scholar]