Abstract
Importance
Targeted magnetic resonance (MR)/ultrasound fusion prostate biopsy has been shown to detect prostate cancer. The implications of targeted biopsy alone vs standard extended-sextant biopsy or the 2 modalities combined are not well understood.
Objective
To assess targeted vs standard biopsy and the 2 approaches combined for the diagnosis of intermediate- to high-risk prostate cancer.
Design, Setting, And Participants
Prospective cohort study of 1003 men undergoing both targeted and standard biopsy concurrently from 2007 through 2014 at the National Cancer Institute in the United States. Patients were referred for elevated level of prostate-specific antigen (PSA) or abnormal digital rectal examination results, often with prior negative biopsy results. Risk categorization was compared among targeted and standard biopsy and, when available, whole-gland pathology after prostatectomy as the “gold standard.”
Interventions
Patients underwent multiparametric prostate magnetic resonance imaging to identify regions of prostate cancer suspicion followed by targeted MR/ultrasound fusion biopsy and concurrent standard biopsy.
Main Outcomes And Measures
The primary objective was to compare targeted and standard biopsy approaches for detection of high-risk prostate cancer (Gleason score ≥4 + 3); secondary end points focused on detection of low-risk prostate cancer (Gleason score 3 + 3 or low-volume 3 + 4) and the biopsy ability to predict whole-gland pathology at prostatectomy.
Results
Targeted MR/ultrasound fusion biopsy diagnosed 461 prostate cancer cases, and standard biopsy diagnosed 469 cases. There was exact agreement between targeted and standard biopsy in 690 men (69%) undergoing biopsy. Targeted biopsy diagnosed 30% more high-risk cancers vs standard biopsy (173 vs 122 cases, P < .001) and 17% fewer low-risk cancers (213 vs 258 cases, P < .001). When standard biopsy cores were combined with the targeted approach, an additional 103 cases (22%) of mostly low-risk prostate cancer were diagnosed (83% low risk, 12% intermediate risk, and 5% high risk). The predictive ability of targeted biopsy for differentiating low-risk from intermediate- and high-risk disease in 170 men with whole-gland pathology after prostatectomy was greater than that of standard biopsy or the 2 approaches combined (area under the curve, 0.73, 0.59, and 0.67, respectively; P < .05 for all comparisons).
Conclusions and Relevance
Among men undergoing biopsy for suspected prostate cancer, targeted MR/ultrasound fusion biopsy, compared with standard extended-sextant ultrasound-guided biopsy, was associated with increased detection of high-risk prostate cancer and decreased detection of low-risk prostate cancer. Future studies will be needed to assess the ultimate clinical implications of targeted biopsy.
Trial Registration
clinicaltrials.gov Identifier: NCT00102544
The current diagnostic procedure for men suspected of prostate cancer is a standard extended-sextant biopsy (ie, standard biopsy). Unlike many other solid tumors for which image-guided biopsy is common, prostate cancer has traditionally been detected by randomly sampling the entire organ. However, the recent introduction of multiparametric magnetic resonance imaging (MP-MRI) now allows for imaging-based identification of prostate cancer, which may improve diagnostic accuracy for higher-risk tumors.1-5
Advances in imaging have led to the development of targeted magnetic resonance (MR)/ultrasound fusion biopsy (ie, targeted biopsy) platforms in which MP-MRI images are electronically superimposed in real time on transrectal ultra-sound (TRUS) images6-9 (Figure 1). Numerous targeted biopsy platforms exist and are capable of performing biopsies of suspicious regions on the prostate MP-MRI.10-12
Figure 1. Steps for Magnetic Resonance/Ultrasound Fusion–Guided Biopsy.
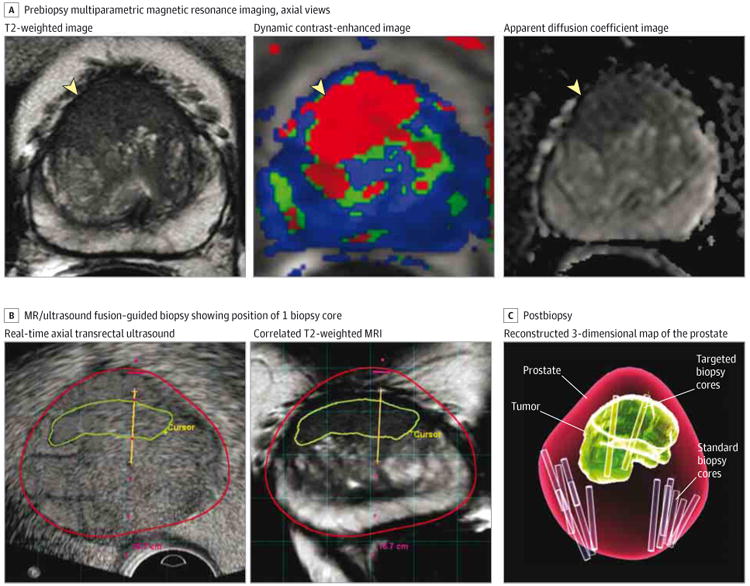
A, Prebiopsy multiparametric magnetic resonance imaging (MP-MRI) that includes anatomical (T2-weighted) and functional (dynamic contrast-enhanced and apparent diffusion coefficient) imaging is obtained and reviewed by a radiologist. Axial images all demonstrate a lesion suspicious for prostate cancer (yellow arrowheads). This lesion would be marked by a radiologist in preparation for fusion biopsy. B, At the time of MR/ultrasound fusion biopsy, a real-time axial transrectal ultrasound is performed to assist with needle guidance. The MR/ultrasound fusion platform overlays the outline of the lesion suspicious for prostate cancer (green line) and contour of the prostate (red line). The platform also synchronizes the transrectal ultrasound image with a location in the prostate and recreates an axial MRI based on the T2-weighted image to correlate with the location of the ultrasound image. A dotted red line demonstrates the path of the needle, and when a biopsy is performed, the location can be recorded as shown here with the yellow line. C, At the conclusion of the biopsy, a 3-dimensional map from the data above is generated, demonstrating the contour of the prostate (red), the location of the tumor lesion (green), the location of the standard extended-sextant biopsies (purple cores), and the location of the targeted MR/ultrasound fusion biopsies (yellow cores).
The early trials of targeted biopsy included a concurrent standard biopsy and thus actually were studies of combined biopsy in the diagnosis of prostate cancer.6,9,11,12 These studies suggested that targeted biopsy combined with standard biopsy is superior to standard biopsy alone in capturing the clinically significant tumors.10-15 This has led to questions about the necessity of performing standard biopsy if targeted biopsy was also performed.16 Therefore, the aim of this study was to assess targeted vs standard biopsy and the 2 approaches combined for the diagnosis of intermediate- to high-risk prostate cancer.
Methods
Patients were enrolled at the National Cancer Institute in Bethesda, Maryland, in a prospective clinical trial with institutional review board approval as part of an ongoing National Institutes of Health study on the use of electromagnetic tracking devices to locate disease during multimodality-navigated procedures. The tracking device is a sensor coil attached to the TRUS probe paired with a magnetic field generator to detect the location of the sensor coil in 3-dimensional space. Enrollment occurred between August 2007 and February 2014 with written informed consent. Inclusion criteria for this study were an elevated level of prostate-specific antigen (PSA) or anabnormal digital rectal examination finding and an MP-MRI that demonstrated at least 1 lesion in the prostate. Exclusion criteria included prior prostate cancer therapy and contraindication to MP-MRI. Patients ineligible for the trial were referred back to their providers.
Imaging
All patients underwent MP-MRI on a 3.0-T MRI (Achieva, Philips Healthcare) with 4 sequences–triplanar T2-weighted, dynamic contrast-enhanced, diffusion-weighted imaging, and MR spectroscopy–according to previously published protocols.17 Images were acquired with a 16-channel surface coil (SENSE, Philips Healthcare) and an endorectal coil (BPX-30, Medrad) in most cases or just a surface coil in a few patients. These MP-MRI studies underwent blinded, centralized radiologic evaluation, and lesions were assigned suspicion scores of low, moderate, or high. These scores are based on findings on each MP-MRI sequence using previously described criteria13 and have been associated with both the presence of prostate cancer and tumor grade6,18 (eMethods and eTable 1 in the Supplement). The now standardized PI-RADS criteria19 were not in use at our center during the time frame of this study. Two highly experienced genitourinary radiologists (B.T. and P.L.C.) with 8 and 14 years of experience interpreting prostate MP-MRI performed independent review and formed consensus reads of all studies in this series.
Biopsy Protocol
Prior to biopsy, an MP-MRI was interpreted by the radiologists; the images were segmented, and lesion locations were recorded (DynaCAD, Invivo). Patients with lesions identified on MP-MRI underwent a targeted biopsy performed by one physician followed in the same session by a standard biopsy performed by another physician who was not aware of the MR lesion locations. Using the UroNav MR/ultrasound fusion device (Invivo), or research iterations of the same device predating the commercially available device, the targeted biopsy was performed with the previously identified MP-MRI lesions superimposed using the T2-weighted sequence on the realtime TRUS images. Each lesion was sampled both in axial and sagittal planes by an end-fire TRUS probe (Philips). The standard biopsy was typically 12 cores collected in an extended-sextant template of biopsies from the lateral and medial aspects of the base, mid, and apical prostate on the left and right side. Only the TRUS images, with no MP-MRI target data available, were used for the standard biopsy portion of the case. More biopsy cores were obtained as part of the standard biopsy if any abnormality was noted on ultrasound. The median time from MP-MRI to biopsy was 43 days (interquartile range, 13-89 days). One genitourinary pathologist (M.J.M.) reviewed all pathologic specimens. The steps in performing an MR/ultrasound fusion biopsy are outlined in Figure 1.
Data Analysis
All data were collected prospectively by a dedicated data manager in a pretrial designed database. Criteria described by the START Consortium were followed in reporting this study.20 Patients were pathologically risk-stratified as low, intermediate, and high risk. Patients were assigned separate risk stratifications according to the standard biopsy, targeted biopsy, and whole-mount pathology, and then these risk strata were compared. Risk categories were chosen to reflect common clinical decision-making groups, eg, low risk (appropriate for active surveillance), intermediate risk (may benefit from intervention), and high risk (may benefit from more aggressive intervention). Alternative risk-stratification methods were also assessed and demonstrated similar outcomes to the study (eMethods in the Supplement). In cases where standard or targeted biopsy detected more than 1 tumor focus, the highest Gleason score reported was used to define the risk category established by that approach.
Prostate cancer treatment options, including active surveillance, focal therapy, radiation, and radical prostatectomy, were discussed with each patient as appropriate. Whole-mount pathology slides of the full prostate gland were available for inclusion in this study in patients who opted for radical prostatectomy. Low risk on biopsy was defined as Gleason score 6 or low-volume Gleason score 3 + 4 (ie, <50% of any core containing cancer and <33% of standard biopsy cores positive for cancer).21-23 Intermediate risk was defined as Gleason score 3 + 4 with 50% or more of any core positive for cancer or 33% or more of standard biopsy cores positive for cancer. High-risk tumors were Gleason score 4 + 3 or greater cancers.24,25 Static patient-specific risk factors such as age, PSA level, and digital rectal examination results were not included in the risk stratification as they do not change between targeted and standard biopsy yet may mask differences observed between these modalities. Prostatectomy whole-mount pathology was risk stratified as follows: low risk (Gleason score 6 or Gleason score 3 + 4 pathology in <20% of the total prostate26), intermediate risk (Gleason score 3 + 4 pathology in ≥20% of the prostate), and high risk (Gleason score ≥4+325).
The main objective of this study was to test the hypothesis that targeted biopsy has a higher rate of high-risk prostate cancer detection than standard biopsy. Secondary objectives included comparing the rates of low-risk prostate cancer detection and evaluating the accuracy of the targeted biopsy, standard biopsy, and combined biopsy approaches to predict whole-gland pathology. Further analyses were performed to quantify the clinical utility of the different biopsy paradigms as a decision aid for radical prostatectomy.
Statistical Methods
Reported statistical significance levels were all 2-sided, and the threshold of statistical significance was P < .05. The Wilcoxon rank sum test was used for comparing the distribution of quantitative variables between the cohorts of patients with and without prior biopsy and the cohorts of patients who did and did not undergo prostatectomy. Fisher exact test was used to compare proportions such as proportion with high-risk disease between these cohorts. Fisher exact test was also used to compare the proportion of intermediate- and high-risk patients among those upgraded from no-cancer or low-risk standard biopsy to the proportion of intermediate- or high-risk patients among those upgraded from a no-cancer or low-risk targeted biopsy. The McNemar test was used for comparing the proportion of patients with high-risk (or low-risk) disease based on targeted biopsy vs standard biopsy.
For generating metrics of accuracy, the risk strata from the biopsy and whole-gland pathology were dichotomized to a no-cancer/low-risk group and an intermediate-/high-risk group. The ability of targeted biopsy, standard biopsy, and the modalities combined was examined to predict the whole-gland pathology risk which was used as the “gold standard.” We compared the sensitivity, specificity, negative predictive value, positive predictive value, accuracy, and area under the receiver operator characteristic curves (AUC) for each of the 3 biopsy approaches. The AUC was calculated based on the receiver operator characteristic curves from the logistic regression of the biopsy type against the whole-gland pathology. The DeLong test was used to determine if the difference between the AUCs was statistically significant. Number needed to biopsy was computed by dividing the total number of men undergoing biopsy by the number of events. All statistical computations were performed using the R statistics package (version 3.1.1).
Decision curve analysis is an analytic instrument to assess net benefit of a diagnostic tool for which there are competing benefits and harms.27 Decision curve analysis was performed on the prostatectomy cohort to compute the net benefit of decisions for prostatectomy based on biopsy results from targeted biopsy alone, standard biopsy alone, and combined biopsy. The desirable outcome, or “benefit,” was defined as operative intervention limited to intermediate-and high-risk tumors, while the undesirable outcome, or “harm,” was operative intervention for low-risk tumors. The decision curve analysis generates a graph of net benefit as a function of a threshold probability (pt) of intermediate- to high-risk disease at which an individual considers the potential benefit and harm of surgery to be equivalent. The net benefit was measured as the rate that incorporating the decision guide of interest (such as targeted biopsy) would lead to additional beneficial decision to treat intermediate-/high-risk cancer without causing any additional harmful decision to overtreat low-risk disease. Decision curve analysis was performed using publicly available code28 and the R statistics package. See the eMethods in the Supplement for more details regarding the decision curve analysis.
Results
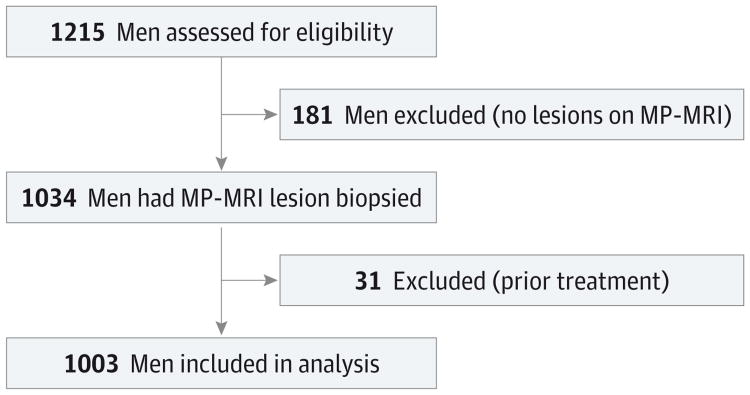
During the study period, 1215 men had an MP-MRI, of whom 181 had no lesions, leading to 1034 patients who underwent biopsy. Thirty-one patients were excluded because of prior treatment for prostate cancer. After exclusions, 1003 unique patients were included in the study (Figure 2). In patients with 2 or more MR/ultrasound fusion biopsy sessions, only the first biopsy session was evaluated in this analysis. Patient demographics are listed in Table 1. In addition to the main sample of all patients who underwent prostate biopsy, 2 subgroups were analyzed in this study: patients with no prior prostate biopsy (n = 196) and patients who ultimately underwent prostatectomy (n = 170). The patients with no history of prior biopsy had a lower prebiopsy PSA level (median, 5.3 vs 7.1 ng/mL, P = .002), smaller prostate volume (median, 42 vs 52 cm3, P < .001), fewer anterior lesions (34% vs 47%, P = .001), and more MRI lesions to biopsy (mean, 2.9 vs 2.6, P = .001) compared with those patients with prior biopsy (n = 807). Anterior lesions (ie, prostate cancer lesions located in the anterior aspect of the prostate) are preferentially detected on targeted biopsy and thus described here. Compared with all patients who underwent biopsy, patients who ultimately underwent prostatectomy were younger (mean age, 60.2 vs 62.1 years, P < .001), had smaller prostate volumes (median, 39 vs 49 cm3, P < .001), and had more MRI lesions (mean, 3.1 vs 2.7, P < .001).
Figure 2. Flowchart for Study Inclusion Among Men Undergoing Both Targeted and Standard Biopsy.
MP-MRI indicates multiparametric magnetic resonance imaging.
Table 1. Patient Demographics.
| All Patients Who Had Biopsy | No-Prior-Biopsy Cohort | Prostatectomy Cohort | |
|---|---|---|---|
| No. of men (% of total) | 1003 | 196 (20) | 170 (17) |
| Age, mean (SD), y | 62.1 (7.5) | 61.2 (8.1) | 60.2 (7.3) |
| PSA, median (IQR), ng/mL | 6.7 (4.4-10.7) | 5.3 (3.3-8.1) | 6.8 (4.4-10.7) |
| Prostate volume, median (IQR), cm3 | 49 (36-71) | 42 (34-54) | 39 (30-48) |
| Prior negative biopsy, No. (%) | 432 (43) | 0 | 45 (26) |
| Cancer suspicion score on MP-MRI, No. (%)a | |||
| Low | 176 (18) | 37 (19) | 28 (16) |
| Moderate | 718 (72) | 129 (66) | 114 (67) |
| High | 109 (11) | 30 (15) | 28 (16) |
| Tumor staging, No. (%)b | |||
| T1c | 902 (89) | 161 (82) | |
| T2a | 93 (9) | 30 (15) | 22 (13) |
| T2b | 3 (0.3) | 2 (1) | 3 (2) |
| T2c | 5 (0.5) | 3 (1.5) | 110 (65) |
| T3a | 25 (15) | ||
| T3b | 8 (5) | ||
| T4 | 2 (1) | ||
| No. of lesions on MP-MRI, mean (SD) | 2.7 (1.3) | 2.9 (1.4) | 3.1 (1.3) |
| Patients with anterior lesions, No. (%) | 446 (44) | 67 (34) | 87 (51) |
| Targeted MR/ultrasound fusion biopsy cores per patient, mean (SD) | 5.3 (2.6) | 5.8 (2.7) | 6.2 (2.5) |
| Systematic extended-sextant biopsy cores per patient, mean (SD) | 12.3 (0.7) | 12.4 (1.0) | 12.3 (0.7) |
Abbreviations: IQR, interquartile range; MP-MRI, multiparametric magnetic resonance imaging; PSA, prostate-specific antigen.
Based on appearance of lesion on the 4 different MRI parameters as noted in the Methods.
Clinical staging for biopsies and pathologic staging for prostatectomy cohort.
Comparisonof Targeted MR/Ultrasound Fusion Biopsy With Standard Extended-Sextant Biopsy
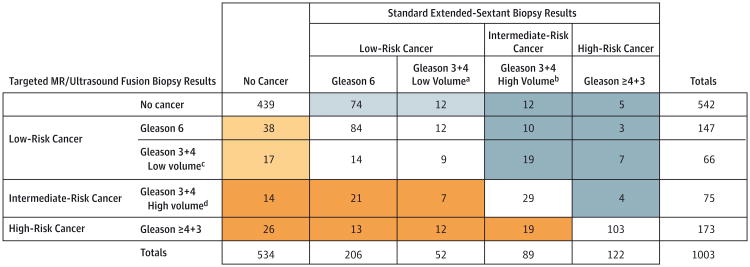
Figure 3 demonstrates a comparison of pathologic outcomes by targeted vs standard biopsy in the entire cohort. Six hundred ninety patients of the total cohort (69%) demonstrated exact agreement between targeted and standard biopsy pathologic risk categories. Targeted biopsy diagnosed a similar number of cancer cases (461 patients) to standard biopsy (469 patients). However, the 2 approaches differed in that targeted biopsy diagnosed 30% more high-risk cancers vs standard biopsy (173 vs 122 cases, P < .001) and 17% fewer low-risk cancers (213 vs 258 cases, P = .002). Targeted biopsy demonstrated a higher risk category in 167 cases (17%) (orange shading) while standard biopsy demonstrated a higher risk category in 146 cases (15%) (blue shading). Among cases in which targeted biopsy revealed a higher risk category, 112 (67%) (dark orange) were upgraded to intermediate- or high-risk pathology by targeted biopsy, whereas in cases where standard biopsy demonstrated a higher risk category, only 60 (41%) (dark blue) were upgraded to intermediate- or high-risk pathology by standard biopsy (P < .001).
Figure 3. Comparison of Pathology From Standard Extended-Sextant Biopsy and Targeted MR/Ultrasound Fusion Biopsy for Prostate Cancer.
Pathologic outcomes per individual of targeted magnetic resonance (MR)/ultrasound fusion biopsies compared with standard extended-sextant biopsies for total cohort of 1003 men. Orange shading indicates patients in whom targeted MR/ultrasound fusion biopsy upgraded prostate cancer risk category in relation to standard extended-sextant biopsy. Dark orange indicates cases in which the upgrade was to an intermediate- or high-risk category, Blue shading indicates patients in whom standard extended-sextant biopsy upgraded prostate cancer risk category in relation to targeted MR/ultrasound fusion biopsy. Dark blue indicates cases in which the upgrade was to an intermediate- or high-risk category.
In addition, the utility of targeted biopsy alone vs targeted and standard biopsy combined was examined. Adding standard biopsy to targeted biopsy lead to 103 more cases of cancer (22%); however, of these, 86 (83%) were low risk while only 5 (5%) were high risk (Figure 3). This equated to a number needed to biopsy with standard biopsy in addition to targeted biopsy of 200 men to diagnose 1 additional high-risk cancer. Furthermore, for every additional case of high-risk cancer diagnosed, 17 additional cases of low-risk cancer would be diagnosed. Incorporation of standard biopsy in addition to targeted biopsy led to no change in Gleason score risk stratification in 857 cases (85%) (eTable 2 in the Supplement). Of those patients with a change in risk category, 86 (9%) increased from no cancer to low-risk cancer, while only 19 (2%) increased from no cancer or low- or intermediate-risk disease to high-risk prostate cancer.
Comparison of Biopsy With Whole-Gland Pathology
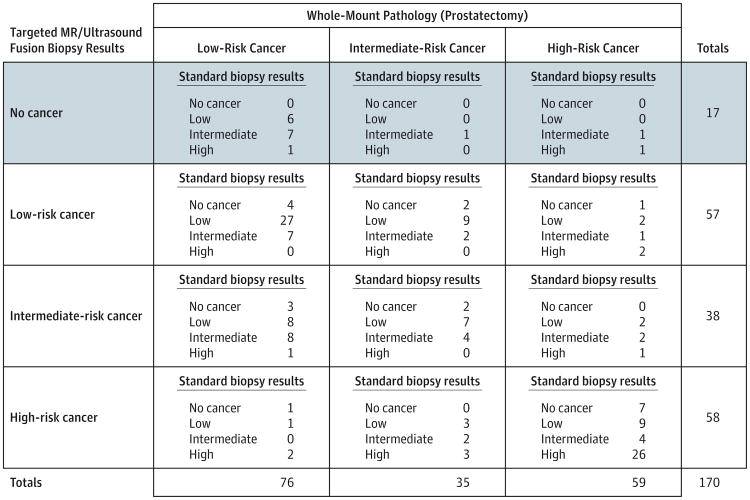
The subset of 170 patients who underwent a radical prostatectomy was also studied so that pathology results from the targeted biopsy and standard biopsy could be compared against the whole-gland prostatectomy pathology (Figure 4). Within this group, 17 patients were diagnosed preprostatectomy with prostate cancer only on standard biopsy, of whom 3 (18%) had intermediate- or high-risk cancer on whole-mount pathology. By contrast, 20 patients (the sum of all “no cancer” values for standard biopsy in all the cells; ie, 4 + 2 + 1+3 + 2 + 1+7) were diagnosed with prostate cancer only on targeted biopsy, of whom 12 (60%) had intermediate- or high-risk cancer on whole-mount pathology. When the ability of preoperative biopsy to predict whole-gland pathology was examined, the sensitivity of targeted biopsy was 77% vs 53% for standard biopsy while the specificities were similar (targeted, 68%, vs standard, 66%). The AUC for targeted biopsy (0.73) was significantly greater than that of either standard biopsy (0.59, P = .005) or combined biopsy (0.67, P = .04) (Table 2).
Figure 4. Comparison of Whole-Mount Prostatectomy Pathology Outcome With Targeted Magnetic Resonance (MR)/Ultrasound Fusion Biopsy and Standard Extended-Sextant Biopsy Pathology for Prostate Cancer.
Pathologic outcomes of the standard and targeted prostate biopsy as well as the subsequent pathology from the prostatectomy specimen for all 170 men in the study who underwent the surgery. The table can be read by examining the whole-mount pathology of interest on the top, the targeted biopsy pathology of interest on the side, and then the standard biopsy pathology of interest within each cell. For example, the shaded row is the distribution of patients not diagnosed with cancer on targeted biopsy but were ultimately found to have cancer on standard biopsy and prostatectomy.
Table 2. Performance of Different Biopsy Approaches in the Detection of Intermediate- toHigh-Risk Prostate Cancer on Whole-Gland Prostatectomy Specimen.
| Targeted MR/Ultrasound Fusion Biopsy | Standard Extended-Sextant Biopsy | Combined Biopsy | |
|---|---|---|---|
| Sensitivity, % (95% CI) | 77 (67-84) | 53 (43-63) | 85 (76-91) |
| Specificity, % (95% CI) | 68 (57-78) | 66 (54-76) | 49 (37-60) |
| Negative predictive value, % (95% CI) | 70 (58-80) | 53 (43-63) | 73 (58-84) |
| Positive predictive value, % (95% CI) | 75 (65-83) | 66 (54-76) | 67 (58-75) |
| Accuracy, % (95% CI) | 73 (70-76) | 59 (55-63) | 69 (65-72) |
| AUC (95% CI) | 0.73 (0.66-0.79) | 0.59 (0.52-0.67) | 0.67 (0.60-0.74) |
| P value of comparison with targeted MR/ultrasound biopsy | .005 | .04 |
Abbreviations: AUC, area under the curve; MR, magnetic resonance.
A decision curve analysis was performed to assess the clinical utility of using each of these 3 biopsy approaches to guide the decision in whom to recommend surgery. Two additional decision guidance approaches (surgery for no one and surgery for everyone with cancer, regardless of risk) were also incorporated for comparison. The eFigure in the Supplement demonstrates that within the clinical range where treat no one and treat everyone with surgery are not the optimal options, the optimal approach to deciding on surgery was by using targeted biopsy to guide decision making as reflected by the higher net benefit seen on the targeted biopsy curve compared with the standard biopsy and combined biopsy curves.
No-Prior-Biopsy Subcohort Analysis
Patients with no prior prostate biopsies were examined separately to assess for potential bias in patients with a history of prior prostate biopsy sessions (such as enrichment for anterior or other standard biopsy occult lesions). Within the 196-patient no-prior-biopsy cohort, 46 patients (42%) had low-risk, 18 patients (16%) had intermediate-risk, and 46 patients (42%) had high-risk prostate cancer. There was no significant difference between the targeted biopsy risk distribution in the no-prior-biopsy patient cohorts and the cohort with prior biopsies (P = .52). The standard biopsy risk distribution was higher among patients without prior biopsy and not significantly different from the targeted biopsy distribution of that cohort. The effect of adding standard biopsy to targeted biopsy was similar as well in the no-prior-biopsy cohort with no change in risk status in 85% of both the no-prior-biopsy cohort and the total cohort. Upgrading to high-risk disease by use of combined biopsy occurred in 7 patients (4%) in the no-prior-biopsy patient cohort, which was similar to the rate in the total cohort (2%) (eTable 2 in the Supplement).
Discussion
In this study, targeted biopsy significantly increased the detection of high-risk prostate cancer while decreasing the detection of low-risk prostate cancer compared with standard biopsy. Targeted biopsy had a greater accuracy than standard biopsy or the 2 combined for intermediate- to high-risk disease on prostatectomy and a higher sensitivity of 77% vs 53%. The utility of standard biopsy in addition to targeted biopsy was also found to be limited. The number needed to biopsy by standard biopsy in addition to targeted biopsy to diagnose 1 additional high-risk tumor was 200 men. Furthermore, for every 1 additional high-risk tumor diagnosed, 17 additional low-risk tumors would also be diagnosed.
This study demonstrated that targeted biopsy could significantly change the distribution of risk in men newly diagnosed with prostate cancer toward diagnosis of more high-risk disease. Although these improvements in risk stratification could translate into substantial clinical benefits, it is important to recognize that this study is preliminary with regard to clinical end points such as recurrence of disease and prostate cancer–specific mortality. These findings provide a strong rationale for the conduct of randomized clinical trials to determine the effect of targeted biopsy on clinical outcomes.
Cost is another important issue that will have to be addressed with the dissemination of this technology. While the technology itself has some cost associated with it, the greatest increase in cost is due to the MRI performed on each patient. A related topic has been studied for MRI in-gantry biopsy in an extensive analysis demonstrating that when the benefits of improved risk stratification were considered, the expected costs per patient were virtually the same. Similar studies in relation to MR/ultrasound fusion biopsy will need to be performed.29
This study has a number of limitations. The majority of the 1003 patients had 1 or more previous biopsies. The study population consisted of patients referred to a single institution, which could have introduced selection bias. Future validation of these findings in a large biopsy-naive screening population may clarify the implications of this paradigm for screening. Second, patients with no lesions visible on MP-MRI were excluded from the study. Data from institutions where standard biopsy is routine for patients with negative MP-MRI results have demonstrated that a negative MRI has a negative predictive value of 83% for any prostate cancer and 98% for Gleason 7 or greater prostate cancer.30 Thus, the cohort of negative MP-MRI results is unlikely to harbor many patients with undiagnosed intermediate- to high-risk disease, and it is therefore unlikely that inclusion of these patients with negative MP-MRI results in the study would have changed the outcome significantly. Third, all of the MP-MRIs in our study cohort were read by 2 highly experienced genitourinary radiologists. Reproducing these findings may be challenging until sufficient experience in the interpretation of these studies has been attained at centers newly adapting this technology.
Conclusions
Among men undergoing biopsy for suspected prostate cancer, targeted MR/ultrasound fusion biopsy, compared with standard extended-sextant ultrasound-guided biopsy, was associated with increased detection of high-risk prostate cancer and decreased detection of low-risk prostate cancer. Future studies will be needed to assess the ultimate clinical implications of targeted biopsy.
Supplementary Material
Acknowledgments
Funding/Support: This research was supported by the Intramural Research Program of the National Institutes of Health (NIH), National Cancer Institute, Center for Cancer Research, and Center for Interventional Oncology. The NIH, Philips Healthcare, and Invivo have a cooperative research and development agreement. The NIH, Philips Healthcare, and Invivo share intellectual property in the field. This research was also made possible through the NIH Medical Research Scholars Program, a public-private partnership supported jointly by the NIH and generous contributions to the Foundation for the NIH from Pfizer, the Leona M. and Harry B. Helmsley Charitable Trust, and the Howard Hughes Medical Institute, as well as other private donors. For a complete list, visit the foundation website at http://www.fnih.org.
Role of the Funder/Sponsor: The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Footnotes
Author Contributions: Drs Siddiqui and Pinto had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Siddiqui, Rais-Bahrami, Turkbey, George, Rothwax, Linehan, Merino, Wood, Pinto.
Acquisition, analysis, or interpretation of data: Siddiqui, Rais-Bahrami, Turkbey, George, Rothwax, Shakir, Okoro, Raskolnikov, Parnes, Linehan, Merino, Simon, Choyke, Wood, Pinto.
Drafting of the manuscript: Siddiqui, Rais-Bahrami, Turkbey, Wood.
Critical revision of the manuscript for important intellectual content: Siddiqui, Rais-Bahrami, Turkbey, George, Rothwax, Shakir, Okoro, Raskolnikov, Parnes, Linehan, Merino, Simon, Choyke, Wood, Pinto.
Statistical analysis: Siddiqui, Turkbey, Simon, Wood.
Obtained funding: Linehan, Wood, Pinto.
Administrative, technical, or material support: Siddiqui, Rais-Bahrami, Turkbey, Rothwax, Shakir, Okoro, Raskolnikov, Linehan, Merino, Choyke, Wood, Pinto.
Study supervision: Rais-Bahrami, George, Parnes, Linehan, Choyke, Wood, Pinto.
Conflict of Interest Disclosures: All authors have completed and submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Dr Choyke and Dr Pinto reported holding a patent related to the MR/ultrasound fusion biopsy platform. Dr Wood reported holding multiple related patents in the field, including a method and system for performing biopsies, a system and method for fusing real-time ultrasound images with preacquired medical images, and others, and contributing to 3 invention reports related to prostate disease diagnosis and treatment.
References
- 1.Haider MA, van der Kwast TH, Tanguay J, et al. Combined T2-weighted and diffusion-weighted MRI for localization of prostate cancer. AJR Am J Roentgenol. 2007;189(2):323–328. doi: 10.2214/AJR.07.2211. [DOI] [PubMed] [Google Scholar]
- 2.Kitajima K, Kaji Y, Fukabori Y, Yoshida K, Suganuma N, Sugimura K. Prostate cancer detection with 3 T MRI: comparison of diffusion-weighted imaging and dynamic contrast-enhanced MRI in combination with T2-weighted imaging. J Magn Reson Imaging. 2010;31(3):625–631. doi: 10.1002/jmri.22075. [DOI] [PubMed] [Google Scholar]
- 3.Fütterer JJ, Heijmink SW, Scheenen TW, et al. Prostate cancer localization with dynamic contrast-enhanced MR imaging and proton MR spectroscopic imaging. Radiology. 2006;241(2):449–458. doi: 10.1148/radiol.2412051866. [DOI] [PubMed] [Google Scholar]
- 4.Villeirs GM, Oosterlinck W, Vanherreweghe E, De Meerleer GO. A qualitative approach to combined magnetic resonance imaging and spectroscopy in the diagnosis of prostate cancer. Eur J Radiol. 2010;73(2):352–356. doi: 10.1016/j.ejrad.2008.10.034. [DOI] [PubMed] [Google Scholar]
- 5.Turkbey B, Mani H, Shah V, et al. Multiparametric 3T prostate magnetic resonance imaging to detect cancer: histopathological correlation using prostatectomy specimens processed in customized magnetic resonance imaging based molds. J Urol. 2011;186(5):1818–1824. doi: 10.1016/j.juro.2011.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pinto PA, Chung PH, Rastinehad AR, et al. Magnetic resonance imaging/ultrasound fusion guided prostate biopsy improves cancer detection following transrectal ultrasound biopsy and correlates with multiparametric magnetic resonance imaging. J Urol. 2011;186(4):1281–1285. doi: 10.1016/j.juro.2011.05.078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kaplan I, Oldenburg NE, Meskell P, Blake M, Church P, Holupka EJ. Real time MRI-ultrasound image guided stereotactic prostate biopsy. Magn Reson Imaging. 2002;20(3):295–299. doi: 10.1016/s0730-725x(02)00490-3. [DOI] [PubMed] [Google Scholar]
- 8.Xu S, Kruecker J, Guion P, et al. Closed-loop control in fused MR-TRUS image-guided prostate biopsy. Med Image Comput Comput Assist Interv. 2007;10(pt 1):128–135. doi: 10.1007/978-3-540-75757-3_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sonn GA, Natarajan S, Margolis DJ, et al. Targeted biopsy in the detection of prostate cancer using an office based magnetic resonance ultrasound fusion device. J Urol. 2013;189(1):86–91. doi: 10.1016/j.juro.2012.08.095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Siddiqui MM, Rais-Bahrami S, Truong H, et al. Magnetic resonance imaging/ultrasound-fusion biopsy significantly upgrades prostate cancer versus systematic 12-core transrectal ultrasound biopsy. Eur Urol. 2013;64(5):713–719. doi: 10.1016/j.eururo.2013.05.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wysock JS, Rosenkrantz AB, Huang WC, et al. A prospective, blinded comparison of magnetic resonance (MR) imaging-ultrasound fusion and visual estimation in the performance of MR-targeted prostate biopsy: the PROFUS trial. Eur Urol. 2014;66(2):343–351. doi: 10.1016/j.eururo.2013.10.048. [DOI] [PubMed] [Google Scholar]
- 12.Kasivisvanathan V, Dufour R, Moore CM, et al. Transperineal magnetic resonance image targeted prostate biopsy versus transperineal template prostate biopsy in the detection of clinically significant prostate cancer. J Urol. 2013;189(3):860–866. doi: 10.1016/j.juro.2012.10.009. [DOI] [PubMed] [Google Scholar]
- 13.Yerram NK, Volkin D, Turkbey B, et al. Low suspicion lesions on multiparametric magnetic resonance imaging predict for the absence of high-risk prostate cancer. BJU Int. 2012;110(11 pt B):783–788. doi: 10.1111/j.1464-410X.2012.11646.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sonn GA, Chang E, Natarajan S, et al. Value of targeted prostate biopsy using magnetic resonance-ultrasound fusion in men with prior negative biopsy and elevated prostate-specific antigen. Eur Urol. 2014;65(4):809–815. doi: 10.1016/j.eururo.2013.03.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.van den Bergh RC, van der Poel HG, Siddiqui M Minhaj, Rais-Bahrami Soroush, Truong Hong, et al. Magnetic resonance imaging/ultrasound-fusion biopsy significantly upgrades prostate cancer versus systematic 12-core transrectal ultrasound biopsy. Eur Urol. 2013;64:713–9. doi: 10.1016/j.eururo.2013.05.059. and Re: Emberton Mark. Has magnetic resonance-guided biopsy of the prostate become the standard of care? Eur Urol. 2013;64:720–1. doi: 10.1016/j.eururo.2013.06.050.. Eur Urol. 2014; 65(6):e106-e107.
- 16.Emberton M. Has magnetic resonance-guided biopsy of the prostate become the standard of care? Eur Urol. 2013;64(5):720–721. doi: 10.1016/j.eururo.2013.06.050. [DOI] [PubMed] [Google Scholar]
- 17.Turkbey B, Mani H, Aras O, et al. Prostate cancer: can multiparametric MR imaging help identify patients who are candidates for active surveillance? Radiology. 2013;268(1):144–152. doi: 10.1148/radiol.13121325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rais-Bahrami S, Siddiqui MM, Turkbey B, et al. Utility of multiparametric magnetic resonance imaging suspicion levels for detecting prostate cancer. J Urol. 2013;190(5):1721–1727. doi: 10.1016/j.juro.2013.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Barentsz JO, Richenberg J, Clements R, et al. European Society of Urogenital Radiology. ESUR prostate MR guidelines 2012. Eur Radiol. 2012;22(4):746–757. doi: 10.1007/s00330-011-2377-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Moore CM, Kasivisvanathan V, Eggener S, et al. START Consortium. Standards of reporting for MRI-targeted biopsy studies (START) of the prostate: recommendations from an International Working Group. Eur Urol. 2013;64(4):544–552. doi: 10.1016/j.eururo.2013.03.030. [DOI] [PubMed] [Google Scholar]
- 21.Klotz L, Zhang L, Lam A, Nam R, Mamedov A, Loblaw A. Clinical results of long-term follow-up of a large, active surveillance cohort with localized prostate cancer. J Clin Oncol. 2010;28(1):126–131. doi: 10.1200/JCO.2009.24.2180. [DOI] [PubMed] [Google Scholar]
- 22.Selvadurai ED, Singhera M, Thomas K, et al. Medium-term outcomes of active surveillance for localised prostate cancer. Eur Urol. 2013;64(6):981–987. doi: 10.1016/j.eururo.2013.02.020. [DOI] [PubMed] [Google Scholar]
- 23.Bul M, van den Bergh RC, Zhu X, et al. Outcomes of initially expectantly managed patients with low or intermediate risk screen-detected localized prostate cancer. BJU Int. 2012;110(11):1672–1677. doi: 10.1111/j.1464-410X.2012.11434.x. [DOI] [PubMed] [Google Scholar]
- 24.Cooperberg MR, Pasta DJ, Elkin EP, et al. The University of California, San Francisco Cancer of the Prostate Risk Assessment score: a straightforward and reliable preoperative predictor of disease recurrence after radical prostatectomy. J Urol. 2005;173(6):1938–1942. doi: 10.1097/01.ju.0000158155.33890.e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stark JR, Perner S, Stampfer MJ, et al. Gleason score and lethal prostate cancer: does 3 + 4 = 4 + 3? J Clin Oncol. 2009;27(21):3459–3464. doi: 10.1200/JCO.2008.20.4669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ramos CG, Roehl KA, Antenor JA, Humphrey PA, Catalona WJ. Percent carcinoma in prostatectomy specimen is associated with risk of recurrence after radical prostatectomy in patients with pathologically organ confined prostate cancer. J Urol. 2004;172(1):137–140. doi: 10.1097/01.ju.0000132139.40964.75. [DOI] [PubMed] [Google Scholar]
- 27.Vickers AJ, Elkin EB. Decision curve analysis: a novel method for evaluating prediction models. Med Decis Making. 2006;26(6):565–574. doi: 10.1177/0272989X06295361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Memorial Sloan Kettering Cancer Center; [Accessed January 2, 2015]. Epidemiology and biostatistics: decision curve analysis. http://www.mskcc.org/research/epidemiology-biostatistics/health-outcomes/decision-curve-analysis-0. [Google Scholar]
- 29.de Rooij M, Crienen S, Witjes JA, Barentsz JO, Rovers MM, Grutters JP. Cost-effectiveness of magnetic resonance (MR) imaging and MR-guided targeted biopsy versus systematic transrectal ultrasound-guided biopsy in diagnosing prostate cancer: a modelling study from a health care perspective. Eur Urol. 2014;66(3):430–436. doi: 10.1016/j.eururo.2013.12.012. [DOI] [PubMed] [Google Scholar]
- 30.Wysock JS, Rosenkrantz AB, Meng X, et al. Predictive value of negative 3T multiparametric prostate MRI on 12 core biopsy results; Presented at: American Urologic Association Annual Meeting; Orlando, FL. May 20, 2014. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.