Abstract
Introduction
This mini-review discusses some of the parallels between rodent neurophysiological and human psychophysical data concerning temperature effects on sweet taste.
Methods and Purpose
“Sweet” is an innately rewarding taste sensation that is associated in part with foods that contain calories in the form of sugars. Humans and other mammals can show unconditioned preference for select sweet stimuli. Such preference is poised to influence diet selection and, in turn, nutritional status, which underscores the importance of delineating the physiological mechanisms for sweet taste with respect to their influence on human health. Advances in our knowledge of the biology of sweet taste in humans have arisen in part through studies on mechanisms of gustatory processing in rodent models. Along this line, recent work has revealed there are operational parallels in neural systems for sweet taste between mice and humans, as indexed by similarities in the effects of temperature on central neurophysiological and psychophysical responses to sucrose in these species. Such association strengthens the postulate that rodents can serve as effective models of particular mechanisms of appetitive taste processing. Data supporting this link are discussed here, as are rodent and human data that shed light on relationships between mechanisms for sweet taste and ingestive disorders, such as alcohol abuse.
Results and Conclusions
Rodent models have utility for understanding mechanisms of taste processing that may pertain to human flavor perception. Importantly, there are limitations to generalizing data from rodents, albeit parallels across species do exist.
Keywords: sweet, temperature, taste, ethanol, neural coding, psychophysics
Introduction
Taste is a chemical sense that likely evolved primarily as a mechanism for detection of chemicals in potential ingesta that signal nutritional value. One can intuit that an ability to discriminate the nutritional content of food on a sensory basis would allow organisms to effectively balance foraging and intake behavior against energy expenditure and nutritional status. As humans, we are intimately familiar with the taste qualities elicited by certain types of food stimuli, and the hedonia associated with select qualities. For example, the “saltiness” of table salt signals the availability of sodium, a critical cation necessary for survival and an appetitive taste stimulus that adds to the flavor of many foods, including meats, vegetables, and snacks such as potato chips. The “sweetness” of sugar is an inherently reinforcing taste sensation that can mark the presence of calories in potential foods. Such foods would serve as effective sources of energy and concomitantly provide for a pleasurable flavor experience during consumption.
On first glance, taste may appear to function as an accessory sensory modality from the standpoint of guiding the nutritional status of humans in the modern world. Nowadays, we rely on cognitive capacity and technology to seek and acquire food as opposed to sampling potential food sources in the wild to evaluate nutritional content. Grocery stores stocked with wide varieties of foods are conveniently located along common routes of passage, and it is now quite easy to locate additional food sources and to order foods using portable communication devices. Nevertheless, taste preferences in humans are influenced by unconditioned factors and also develop with experience with the tastes of consumed foods (Bertino et al. 1986; Mennella et al. 2011), leaving gustation as an important factor in diet selection. Unraveling mechanisms for taste has implications for understanding how sensory factors are involved with intake behavior and guide nutritional status in humans.
The rodent has proven to be a useful model for the study of the biology of taste in mammals, including mechanisms of appetitive sweet taste. For instance, prior studies in mice on genetic control of intake of human-defined “sweet” stimuli, which, in many cases, mice avidly consume (e.g., Bachmanov et al. 2001b), revealed sensitivity to the artificial sweetener saccharin was influenced by a single locus on mouse chromosome 4 (Lush et al. 1995), coined Sac (Fuller 1974). Sac was subsequently found to also affect neural and behavioral responses in mice to multiple types of sweet stimuli including sucrose (Bachmanov et al. 2001a; Bachmanov et al. 1997; Blizard et al. 1999; Lush 1989), a sugar that elicits an inborn hedonically positive taste in humans (e.g., Crook 1978; Nowlis and Kessen 1976; Steiner 1979). The identification of Sac in mice aided discovery within the human and murine genome of the syntenic gene Tas1r3 encoding the receptor protein T1R3, which is found in oral taste cells and shows amino acid sequences that segregate along with Sac phenotype (Bachmanov et al. 2001a; Kitagawa et al. 2001; Max et al. 2001; Montmayeur et al. 2001; Reed et al. 2004; Sainz et al. 2001). Genetic knockout studies in mice further revealed T1R3 as a functional component of the molecular receptor involved with the gustatory detection of diverse sweet stimuli, including saccharin and sucrose (Damak et al. 2003; Zhao et al. 2003). Several other studies have established a role for T1R3 in “sweet” taste in diverse mammalian species including humans (Fushan et al. 2009; Mennella et al. 2012), squirrel monkeys (Liu et al. 2012), and pandas (Jiang et al. 2014), findings that are tied to the initial discovery of Sac in the mouse system.
In addition to supporting discoveries on taste receptors, the use of rodent models in taste research has importantly contributed to our understanding of central neurophysiological mechanisms for gustation in mammals, including mechanisms of gustatory coding of information about appetitive sweet stimuli. Here we will focus on data that suggest there are operational parallels in neural systems for sweet taste between mice and humans. Such association strengthens the postulate that rodents can serve as effective models of particular aspects of taste perception and behavior in humans.
Temperature effects on sweet taste: psychophysical data
Psychophysics is a discipline that attempts to, in part, establish mathematical relationships that explain how features of external stimuli are transferred to perceptual experience. Because perception is a product of neural processing, quantitative effects in psychophysical data tap into and reflect principles of operation of sensory neurons. Thus, psychophysics provides a “window” to the workings of neural mechanisms for perception. Associations between psychophysical and neurophysiological data have provided insight into potential mechanistic relationships between neural circuitry and perceptual phenomena in diverse systems (e.g., LaMotte and Campbell 1978; Newsome et al. 1989). As follows, recent neurophysiological studies in mice have established mechanistic associations between neural processing in the hindbrain and human perceptual responses to sucrose, a prototype sweet-tasting stimulus, through evaluation of the effects of temperature on gustatory activity.
Personal experience informs us that the perceived sweetness of sugary foods is strongly influenced by food temperature. For some humans, a piece of apple pie may taste sweeter if taken from a freshly baked, warmed pie as opposed to sampling a piece of the same pie allowed to cool for some time in a refrigerator. Sugar-rich sodas can taste noticeably sweeter when drank at room temperature rather than cooled on ice. As an aside, most sodas are consumed chilled, which can reduce their sweetness intensity and, conceivably, perceived sugar content to the consumer. Finally, chocolate ice cream can have a more pronounced sweetness if allowed to melt from its normally frozen state (i.e., to warm) prior to consumption. Thus, warming tends to enhance the sweetness of sugar, whereas cooling suppresses its sweetness.
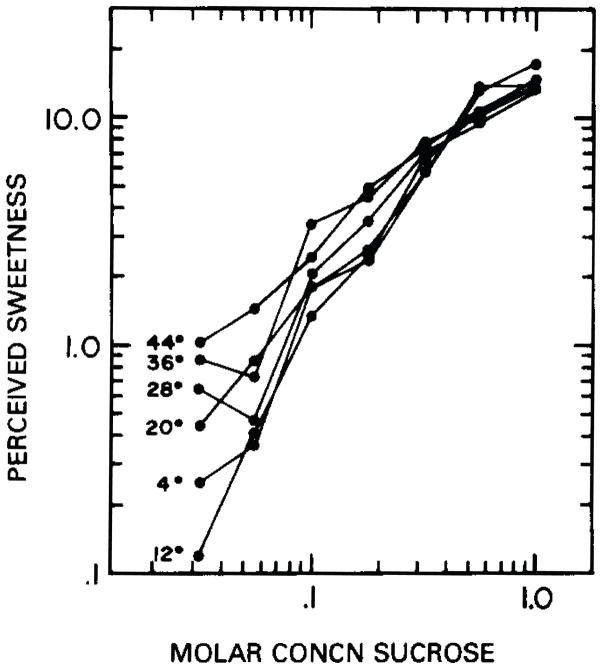
The effect of temperature on sweet taste perception in humans has been studied in the laboratory using psychophysical methods. Such experiments have revealed change in temperature can progressively change the perceived sweetness of sugars, such as sucrose, and, further, that the effect of stimulus concentration on sweetness intensity is conditioned on temperature. The perceived magnitude of sucrose generally decreases with cooling and increases with innocuous warming of stimulus solutions and also the tongue (Bartoshuk et al. 1982; Calvino 1986; Green and Frankmann 1987; Green and Nachtigal 2012). Increments in perceived intensity with warming can, in general, elevate the psychophysical concentration-response function for sucrose, which shows positive association between perceptual intensity (ordinate) and concentration (abscissa), yet shallow the slope of this function relative to cooling (Bartoshuk et al. 1982; Calvino 1986; Green and Frankmann 1987). In fact, stepwise warming and cooling progressively decrease and increase, respectively, the slope of the psychophysical function for sucrose (Figure 1; Bartoshuk et al. 1982). The slope of the sucrose concentration-response function indexes rate of growth in the intensity of the “sweetness” response that follows change in sucrose concentration. The inverse relationship of this slope with temperature suggests the perceived intensity of sucrose grows at a faster rate with concentration when sucrose is cooled rather than warmed (Calvino 1986). Moreover, the changing albeit positive slope of the psychophysical function across temperatures increases “spread” among thermally-varied functions at low concentrations of sucrose but causes convergence of functions at heightened intensities of this stimulus, near 0.4 to 0.5 M (Bartoshuk et al. 1982; Calvino 1986; Green and Frankmann 1987). This characteristic indicates temperature induces greater influence on sensations to reduced rather than elevated concentrations of sucrose (Green and Frankmann 1988), where thermal effects on perceived intensity are diminished.
Figure 1.
Effect of stimulus temperature on the perceived sweetness of sucrose in humans. Data represent magnitude estimates (ordinate) for a sucrose concentration series (abscissa) tested at 4°, 12°, 20°, 28°, 36°, and 44°C. Solid lines connect points representing responses to sequential concentrations measured at one temperature. Slopes for straight-line fits to data points acquired from 4° to 44°C are as follows: 1.31, 1.40, 1.02, 1.01, 0.89, and 0.80. Reprinted from Bartoshuk et al. (1982) with permission from the publisher.
Temperature effects on sweet taste: neural correlates of perception
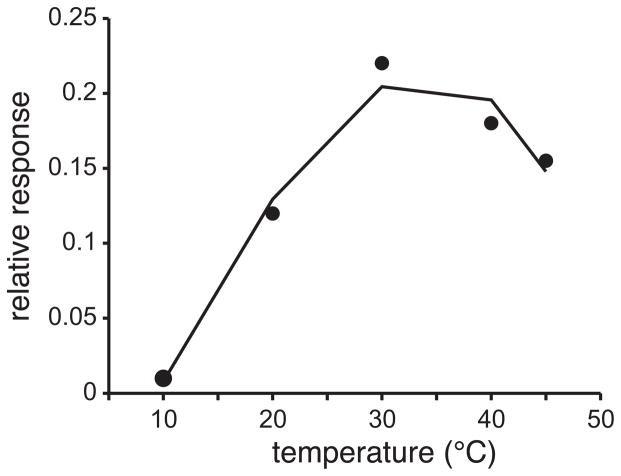
The psychophysical data above reflect the operation of an underlying neural construct in humans that transfers the physicochemical features of sucrose to gustatory experience. The effect of temperature on the operation of this construct is systematic and dependent on sucrose concentration: taste responses to sucrose show greater susceptibility to change with temperature with reductions in stimulus concentration. Where along the neuraxis does the neural machinery for this construct “reside”? There are certainly multiple levels of the nervous system poised to contribute to sucrose-temperature orosensory interaction, beginning with the molecular receptor mechanisms for sucrose expressed on gustatory epithelia in the mouth. For instance, the conversion of the chemical energy of sucrose to an electro-chemical message readable by the nervous system is contributed by current flow into taste receptor cells mediated by the transient receptor potential melastatin 5 (TRPM5) ion channel (Perez et al. 2002; Zhang et al. 2003), a nonselective cation channel that displays increased current passage in the presence of warmth (Talavera et al. 2005). Electrophysiological studies in rats and mice have revealed warming can, under certain conditions, augment gustatory responses to sucrose in fibers of the chorda tympani (CT) branch of the facial nerve (Figure 2; Breza et al. 2006; Lu et al. 2012; Talavera et al. 2005; Yamashita and Sato 1965), which supplies taste and, in part, cutaneous sensation to the rostral tongue. Further, the ability of warmth to enhance taste responses to sucrose in peripheral nerves is lost in mice genetically deficient for TRPM5 (Talavera et al. 2005). Thus, there is an important peripheral component to sucrose-temperature interaction, with temperature inducing direct modulatory effects on molecular components of taste receptors involved with sucrose transduction.
Figure 2.
The response of the chorda tympani nerve in rats to lingual delivery of 0.5 M sucrose tested at 10°, 20°, 30°, 40°, and 45°C. Points represent integrated, whole-nerve activity expressed relative to the response to a 0.1 M NaCl standard. Graph is reconstructed from data published in Yamashita and Sato (1965) with permission from the publisher.
To delineate a neural process for perception it is important to tie events occurring at the periphery to the actions of neurons in the brain, as perception and behavior are ultimately a product of interaction between neural elements residing at peripheral and central levels of the nervous system. Along this line, recent in vivo gustatory electrophysiological studies in mice have explored the impact of temperature on responses to sucrose in central taste-sensitive neurons in the nucleus of the solitary tract (NTS) in the hindbrain (Wilson and Lemon 2013; Wilson and Lemon 2014), with an emphasis on understanding whether quantitative parallels exist between central neurophysiological responses in this model animal system and the psychophysical effects of temperature on sucrose taste perception in humans, as above. These experiments targeted the NTS as this nucleus receives the terminals of gusto-somatic fibers of the facial nerve along with afferents of the glossopharyngeal and trigeminal nerves carrying taste and oral somatosensory information. Electrophysiological taste responses were recorded from NTS units during delivery to multiple regions of the tongue and mouth of several concentrations of sucrose adjusted to multiple temperatures, including temperatures identified as cool (e.g., 18°C) and warm (e.g., 30°C) relative to ambient “room” temperature. Many prior neurophysiological studies on gustatory processing in the brain, including studies I have co-authored, tested only uncontrolled-room temperature taste stimuli, which cannot capture effects imposed on sensory coding by change in stimulus temperature.
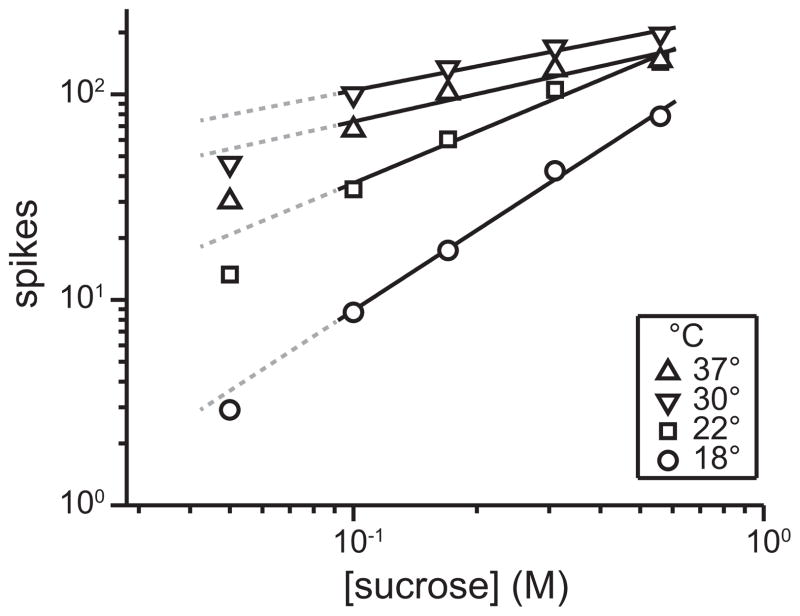
Recordings of temperature-varied activity revealed warming taste solutions markedly augmented gustatory responses to sucrose in mouse NTS neurons, including responses in neurons that were “tuned” to sucrose (Wilson and Lemon 2013; Wilson and Lemon 2014). This effect was similar to effects reported in studies of thermo-sucrose taste responses in nerves, as above. But it was further discovered that progressive warming of sucrose solutions systematically decreased the slope of the concentration-response function for this stimulus in sucrose-oriented cells, whereas stimulus cooling steepened this slope (Figure 3; Wilson and Lemon 2014). Thus, the slope of the concentration-response function for sucrose in hindbrain gustatory neurons in mice shows an inverse relation with temperature. Further inspection of this effect revealed change in temperature induced greater influence on neuronal responses to sucrose at reduced stimulus concentrations (Figure 3). For example, changing solution temperature from 18° to 30°C caused a 1,049% increase in the average neural response to 0.1 M sucrose, but only a 152% increase in mean activity to 0.56 M sucrose. This trend reflects that neural concentration-response functions for sucrose show separation at low concentrations but begin to converge across temperatures at high stimulus intensities (Figure 3). It is noteworthy that studies on temperature effects on peripheral gustatory activity to sucrose concentration series have not uniformly shown convergence of responses to elevated concentrations of sucrose at different temperatures (Lu et al. 2012; Talavera et al. 2005), leaving the potential for a central contribution to the hindbrain effect (Wilson and Lemon 2014).
Figure 3.
Effect of stimulus temperature on neurophysiological activity to sucrose in taste-sensitive neurons in the mouse hindbrain. Data represent mean responses (spikes per unit time; ordinate) across 22 sucrose-oriented neurons to a sucrose concentration series (abscissa) tested at relatively cool (e.g., 18°C) to physiologically warm (37°C) temperatures. Data are plotted in doubly-logarithmic coordinates. Slopes for least-squares fits (solid lines) to activity to 0.1, 0.17, 0.31, and 0.56 M sucrose measured at 18°, 22°, 30°, and 37°C are as follows: 1.44, 0.94, 0.39, and 0.50. Dashed lines extend fits for comparison against activity to 0.05 M, a near-threshold concentration of sucrose (Treesukosol and Spector 2012). Reprinted from Wilson and Lemon (2014) with permission from the publisher.
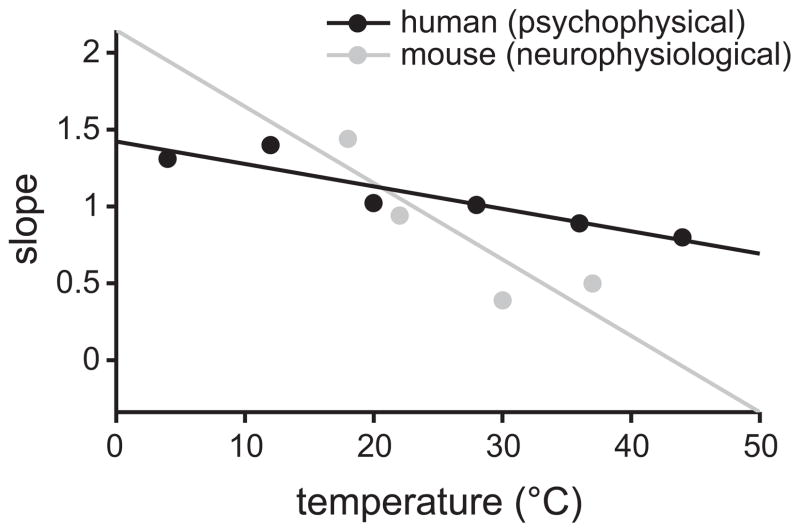
It follows, then, that there exist certain mathematical parallels between neurophysiological responses to sucrose in the mouse hindbrain and perceptual responses to sucrose in humans, as revealed by parallel quantitative shifts in sensory activity to this input between species under similar temperature conditions. In both mice and humans, the slope of the sucrose concentration-response function showed an inverse relation with temperature (Figure 4), with temperature exerting greater relative influence on perceptual and neural responses to weak/low rather than high concentrations of sucrose. Although one must proceed cautiously when comparing data across species and levels of analysis, this association raises the possibility that there exists a common neurophysiological construct mediating the taste of appetitive sucrose across diverse mammals, and that the mouse system may be a reasonable model of particular aspects of sucrose taste processing in humans. Moreover, it is remarkable that neurophysiological correlates of thermal effects on sucrose taste perception emerged in the hindbrain, which suggests modulatory effects of temperature can originate at an “early” stage of the neuraxis and would complement further integration of taste with somatosensation and smell, as arises in higher brain centers (cf. Small and Green 2012) during flavor processing. It would be oversimplification, however, to infer that the actions of hindbrain circuits largely or sufficiently account for central mechanisms of sweet taste perception, as these circuits project to, and receive descending modification from, other brain areas that imbue gustatory neural codes with, for example, cognitive features such as expectancy (cf. Samuelsen et al. 2012; Smith et al. 2005). Nonetheless, processing in hindbrain circuitry critically shapes “bottom-up” drive in central pathways for sweet taste.
Figure 4.
Effect of temperature on the slope of the sucrose concentration-response function in mice and man. Lines represent least-squares fits of data points representing slopes (ordinate) for human psychophysical (Bartoshuk et al. 1982) and mouse neurophysiological (Wilson and Lemon 2014) concentration-response functions to sucrose tested at different temperatures (abscissa). Values for data point are given in Figures 1 and 3.
It is important to point out that the above relationship between neurophysiological and psychophysical response data in mice and humans represents only one example of a functional parallel in the processing of sweet taste between these species. Other parallels between rodent and man include modification of the gustatory response to sweet stimuli by hormones common across species, such as leptin (e.g., Kawai et al. 2000; Nakamura et al. 2008), and common neurotransmitter systems associated with the rewarding orosensory response to sucrose and pleasantness rating of food, including dopamine (DA) pathways (e.g., Hajnal et al. 2004; Small et al. 2003). These and the above data suggest studies on neural circuitry for sweet taste in rodent models will be important for elucidating mechanisms of perceptual binding involving sweet stimuli and, relatedly, for understanding how activation of brain mechanisms mediating appetitive taste associate with patterns of intake behavior.
Neural circuits for sweet taste and ingestive disorders
Parallels in sucrose taste processing between humans and rodents suggest rodents provide a reasonable model system to study how activation of circuits for sweet taste may also associate with patterns of intake in disease states, including disorders involving motivated ingestive behaviors such as alcohol abuse. Alcohol is an ingested drug whose initial direct interaction with the body during consumption is of orosensory nature. Electrophysiological studies in mice and rats have consistently shown elevated, clinically-relevant concentrations of ethanol are potent and largely selective stimuli for sucrose-responsive neurons in the NTS, resulting in reliable positive correlations between central gustatory codes for ethanol and appetitive sucrose (Brasser et al. 2010; Lemon et al. 2004; Lemon et al. 2011). This association is, in certain ways, not surprising considering ethanol is derived naturally during the fermentation of sugars in ripe fruits. An appetitive sensory component to ethanol may have evolutionary advantages for frugivores and the fruits they consume in terms of, respectively, detection of a stimulus associated with calories contained by fruits and, following egestion, dispersion of matured seeds of ripened fruits by the fruit consumer (Dudley 2000). Furthermore, the ability of ethanol to selectively stimulate central neural pathways for sweet taste could lead to a rapid, pharmacological change in brain circuitry associated with motivated behavior that is elicited by sensory cues (cf. Lemon et al. 2004). The activation of neural circuitry encoding the taste of sucrose induces a near immediate increase in DA efflux in the nucleus accumbens (NAc; Avena et al. 2006; Hajnal et al. 2004), a region of the striatum where DA transmission has been implicated in select approach and effort related behaviors (e.g., Nicola 2010; Salamone et al. 2007). Microdialysis work on the role of the striatum in alcohol-motivated behavior in rats has shown that upon consumption of ethanol there is an initial increase in DA efflux in the NAc that is not directly related to the concentration of ethanol that reaches this structure, implicating the stimulus properties of ethanol as contributing to an “early” release of NAc DA during alcohol drinking (Doyon et al. 2003). The ability of oral ethanol to selectively stimulate central pathways mediating the taste of appetitive sucrose, as revealed in studies of rats and mice, seems a plausible route underlying this effect.
Additional data from rodents indicate neurobiological mechanisms for oral sensation and sweet taste are involved with ethanol-guided behaviors in these species. Recent data have revealed selectively-bred alcohol-preferring “P” rats, a proposed rodent model of excessive alcohol intake and alcoholism, display heightened chemosensory-mediated attraction to ethanol and sucrose compared to selectively-bred alcohol non-preferring “NP” and outbred Wistar rats (Brasser et al. 2012). Genetic mapping studies in mice have shown co-localization of genes mediating sweet taste preference and preference for alcohol (Bachmanov et al. 2002). Further, normally ethanol-preferring C57BL/6J mice (Belknap et al. 1993) engineered with genetic deficiencies in proteins involved with sweet taste transduction display significant and marked reductions in ethanol preference (Blednov et al. 2008; Brasser et al. 2010), with an associated reduction in gustatory activity to ethanol in central taste-sensitive neurons (Brasser et al. 2010).
Although neural mechanisms for sweet taste do contribute to ethanol preference in rodents (see Brasser et al. 2014 for additonal review), how gustation is precisely involved with ethanol drinking behavior in humans remains elusive. Yet there exist ties between ethanol intake and sweet processing in humans. Alcoholics show greater preference for concentrated solutions of sucrose compared to non-alcoholic controls (Kampov-Polevoy et al. 1997; Kampov-Polevoy et al. 1998). Moreover, preference for concentrated sucrose is greater in humans with a family history of alcoholism compared to humans who lack such history, suggesting sweet preference associates with genetic vulnerability to excessive alcohol consumption (Kampov-Polevoy et al. 2003a; Kampov-Polevoy et al. 2003b). A recent study using functional magnetic resonance imaging revealed positive correlations between the rated “liking” of sucrose, the number of alcoholic drinks consumed by individuals per drinking day, and the neural response to oral delivery of sucrose in cortical regions involved with reward (Kareken et al. 2013). From these data Kareken et al. (2013) proposed the neuronal response to sweet taste in the brain could phenotypically associate with risk for alcohol abuse. Further studies may elucidate mechanisms of the association between ethanol stimulation of sweet taste pathways and alcohol preference. Similarities in the operation of neural circuits for sweet taste between humans, mice, and rats, as above, render the rodent an important model for this pursuit.
In closing
One goal of gustatory neurobiology is to delineate how physiological mechanisms for taste contribute to diet selection in our species. This issue has implications for understanding the function of the taste system in the homeostatic regulation of ingestive behavior, a process for which the sense of taste likely evolved in humans and diverse animals. Studies in non-human models, such as rodents, have importance for understanding the organization of the human taste system. Non-human models provide, for one, a platform to carry out experiments that would not be ethical or feasible in humans, but are needed to understand circuit organization. It is noteworthy, however, that there are limitations to using rodents as models for appetitive taste in humans, including species differences in the perceptual categorization of taste stimuli. For instance, select non-nutritive stimuli that taste pleasurably “sweet” to humans can elicit only variable preference responses or avoidance in some rodent lines (Bachmanov et al. 2001b; Sclafani and Abrams 1986; Sclafani and Clare 2004). Rodents also show attraction towards polysaccharides that humans can report as having an unpleasant flavor at high molar concentrations (Feigin et al. 1987). What is more, outbred stocks and inbred strains of laboratory rodents have obvious genomic differences with genetically heterogeneous humans that may impose challenges for developing model systems of human physiology. Nevertheless, parallels in the operation of neural substrates for the taste of sucrose in mice and man, as evidenced in part by positive associations between neurophysiological and psychophysical response data, indicate biological mechanisms mediating appetitive taste can share common functional features across species. This overlap promotes the use of the rodent system in studies on particular aspects of appetitive taste perception and behavior.
Acknowledgments
Finding
Supported in part by National Institutes of Health grant DC-011579 to C.H.L.
Footnotes
Conflict of interest statement
The author declares no conflict of interest
References
- Avena NM, Rada P, Moise N, Hoebel BG. Sucrose sham feeding on a binge schedule releases accumbens dopamine repeatedly and eliminates the acetylcholine satiety response. Neuroscience. 2006;139:813–820. doi: 10.1016/j.neuroscience.2005.12.037. [DOI] [PubMed] [Google Scholar]
- Bachmanov AA, et al. Positional cloning of the mouse saccharin preference (Sac) locus. Chem Senses. 2001a;26:925–933. doi: 10.1093/chemse/26.7.925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachmanov AA, Reed DR, Li X, Li S, Beauchamp GK, Tordoff MG. Voluntary ethanol consumption by mice: genome-wide analysis of quantitative trait loci and their interactions in a C57BL/6ByJ x 129P3/J F2 intercross. Genome Res. 2002;12:1257–1268. doi: 10.1101/gr.129702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachmanov AA, Reed DR, Ninomiya Y, Inoue M, Tordoff MG, Price RA, Beauchamp GK. Sucrose consumption in mice: major influence of two genetic loci affecting peripheral sensory responses. Mamm Genome. 1997;8:545–548. doi: 10.1007/s003359900500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachmanov AA, Tordoff MG, Beauchamp GK. Sweetener preference of C57BL/6ByJ and 129P3/J mice. Chem Senses. 2001b;26:905–913. doi: 10.1093/chemse/26.7.905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartoshuk LM, Rennert K, Rodin J, Stevens JC. Effects of temperature on the perceived sweetness of sucrose. Physiol Behav. 1982;28:905–910. doi: 10.1016/0031-9384(82)90212-8. [DOI] [PubMed] [Google Scholar]
- Belknap JK, Crabbe JC, Young ER. Voluntary consumption of ethanol in 15 inbred mouse strains. Psychopharmacology (Berl) 1993;112:503–510. doi: 10.1007/BF02244901. [DOI] [PubMed] [Google Scholar]
- Bertino M, Beauchamp GK, Engelman K. Increasing dietary salt alters salt taste preference. Physiol Behav. 1986;38:203–213. doi: 10.1016/0031-9384(86)90155-1. [DOI] [PubMed] [Google Scholar]
- Blednov YA, Walker D, Martinez M, Levine M, Damak S, Margolskee RF. Perception of sweet taste is important for voluntary alcohol consumption in mice. Genes Brain Behav. 2008;7:1–13. doi: 10.1111/j.1601-183X.2007.00309.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blizard DA, Kotlus B, Frank ME. Quantitative trait loci associated with short-term intake of sucrose, saccharin and quinine solutions in laboratory mice. Chem Senses. 1999;24:373–385. doi: 10.1093/chemse/24.4.373. [DOI] [PubMed] [Google Scholar]
- Brasser SM, Castro N, Feretic B. Alcohol Sensory Processing and its Relevance for Ingestion. Physiol Behav. 2014 doi: 10.1016/j.physbeh.2014.09.004. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brasser SM, Norman MB, Lemon CH. T1r3 taste receptor involvement in gustatory neural responses to ethanol and oral ethanol preference. Physiol Genomics. 2010;41:232–243. doi: 10.1152/physiolgenomics.00113.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brasser SM, Silbaugh BC, Ketchum MJ, Olney JJ, Lemon CH. Chemosensory responsiveness to ethanol and its individual sensory components in alcohol-preferring, alcohol-nonpreferring and genetically heterogeneous rats. Addict Biol. 2012;17:423–436. doi: 10.1111/j.1369-1600.2011.00415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breza JM, Curtis KS, Contreras RJ. Temperature modulates taste responsiveness and stimulates gustatory neurons in the rat geniculate ganglion. J Neurophysiol. 2006;95:674–685. doi: 10.1152/jn.00793.2005. [DOI] [PubMed] [Google Scholar]
- Calvino AM. Perception of sweetness: the effects of concentration and temperature. Physiol Behav. 1986;36:1021–1028. doi: 10.1016/0031-9384(86)90474-9. [DOI] [PubMed] [Google Scholar]
- Crook C. Taste perception in the newborn infant. Infant Behav Dev. 1978;1:52–69. [Google Scholar]
- Damak S, et al. Detection of sweet and umami taste in the absence of taste receptor T1r3. Science. 2003;301:850–853. doi: 10.1126/science.1087155. [DOI] [PubMed] [Google Scholar]
- Doyon WM, York JL, Diaz LM, Samson HH, Czachowski CL, Gonzales RA. Dopamine activity in the nucleus accumbens during consummatory phases of oral ethanol self-administration. Alcohol Clin Exp Res. 2003;27:1573–1582. doi: 10.1097/01.ALC.0000089959.66222.B8. [DOI] [PubMed] [Google Scholar]
- Dudley R. Evolutionary origins of human alcoholism in primate frugivory Q. Rev Biol. 2000;75:3–15. doi: 10.1086/393255. [DOI] [PubMed] [Google Scholar]
- Feigin MB, Sclafani A, Sunday SR. Species differences in polysaccharide and sugar taste preferences. Neurosci Biobehav Rev. 1987;11:231–240. doi: 10.1016/s0149-7634(87)80031-3. [DOI] [PubMed] [Google Scholar]
- Fuller JL. Single-locus control of saccharin preference in mice. J Hered. 1974;65:33–36. doi: 10.1093/oxfordjournals.jhered.a108452. [DOI] [PubMed] [Google Scholar]
- Fushan AA, Simons CT, Slack JP, Manichaikul A, Drayna D. Allelic polymorphism within the TAS1R3 promoter is associated with human taste sensitivity to sucrose. Curr Biol. 2009;19:1288–1293. doi: 10.1016/j.cub.2009.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green BG, Frankmann SP. The effect of cooling the tongue on the perceived intensity of taste. Chem Senses. 1987;12:609–619. [Google Scholar]
- Green BG, Frankmann SP. The effect of cooling on the perception of carbohydrate and intensive sweeteners. Physiol Behav. 1988;43:515–519. doi: 10.1016/0031-9384(88)90127-8. [DOI] [PubMed] [Google Scholar]
- Green BG, Nachtigal D. Somatosensory factors in taste perception: effects of active tasting and solution temperature. Physiol Behav. 2012;107:488–495. doi: 10.1016/j.physbeh.2012.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hajnal A, Smith GP, Norgren R. Oral sucrose stimulation increases accumbens dopamine in the rat. Am J Physiol Regul Integr Comp Physiol. 2004;286:R31–37. doi: 10.1152/ajpregu.00282.2003. [DOI] [PubMed] [Google Scholar]
- Jiang P, et al. The bamboo-eating giant panda (Ailuropoda melanoleuca) has a sweet tooth: behavioral and molecular responses to compounds that taste sweet to humans. PLoS One. 2014;9:e93043. doi: 10.1371/journal.pone.0093043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kampov-Polevoy A, Garbutt JC, Janowsky D. Evidence of preference for a high-concentration sucrose solution in alcoholic men. Am J Psychiatry. 1997;154:269–270. doi: 10.1176/ajp.154.2.269. [DOI] [PubMed] [Google Scholar]
- Kampov-Polevoy AB, Garbutt JC, Davis CE, Janowsky DS. Preference for higher sugar concentrations and Tridimensional Personality Questionnaire scores in alcoholic and nonalcoholic men. Alcohol Clin Exp Res. 1998;22:610–614. doi: 10.1111/j.1530-0277.1998.tb04300.x. [DOI] [PubMed] [Google Scholar]
- Kampov-Polevoy AB, Garbutt JC, Khalitov E. Family history of alcoholism and response to sweets. Alcohol Clin Exp Res. 2003a;27:1743–1749. doi: 10.1097/01.ALC.0000093739.05809.DD. [DOI] [PubMed] [Google Scholar]
- Kampov-Polevoy AB, et al. Association between sweet preference and paternal history of alcoholism in psychiatric and substance abuse patients. Alcohol Clin Exp Res. 2003b;27:1929–1936. doi: 10.1097/01.ALC.0000099265.60216.23. [DOI] [PubMed] [Google Scholar]
- Kareken DA, Dzemidzic M, Oberlin BG, Eiler WJ., 2nd A preliminary study of the human brain response to oral sucrose and its association with recent drinking. Alcohol Clin Exp Res. 2013;37:2058–2065. doi: 10.1111/acer.12194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawai K, Sugimoto K, Nakashima K, Miura H, Ninomiya Y. Leptin as a modulator of sweet taste sensitivities in mice. Proc Natl Acad Sci U S A. 2000;97:11044–11049. doi: 10.1073/pnas.190066697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitagawa M, Kusakabe Y, Miura H, Ninomiya Y, Hino A. Molecular genetic identification of a candidate receptor gene for sweet taste. Biochem Biophys Res Commun. 2001;283:236–242. doi: 10.1006/bbrc.2001.4760. [DOI] [PubMed] [Google Scholar]
- LaMotte RH, Campbell JN. Comparison of responses of warm and nociceptive C-fiber afferents in monkey with human judgments of thermal pain. J Neurophysiol. 1978;41:509–528. doi: 10.1152/jn.1978.41.2.509. [DOI] [PubMed] [Google Scholar]
- Lemon CH, Brasser SM, Smith DV. Alcohol activates a sucrose-responsive gustatory neural pathway. J Neurophysiol. 2004;92:536–544. doi: 10.1152/jn.00097.2004. [DOI] [PubMed] [Google Scholar]
- Lemon CH, Wilson DM, Brasser SM. Differential neural representation of oral ethanol by central taste-sensitive neurons in ethanol-preferring and genetically heterogeneous rats. J Neurophysiol. 2011;106:3145–3156. doi: 10.1152/jn.00580.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu B, Ha M, Meng XY, Khaleduzzaman M, Zhang Z, Li X, Cui M. Functional characterization of the heterodimeric sweet taste receptor T1R2 and T1R3 from a New World monkey species (squirrel monkey) and its response to sweet-tasting proteins. Biochem Biophys Res Commun. 2012;427:431–437. doi: 10.1016/j.bbrc.2012.09.083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu B, Breza JM, Nikonov AA, Paedae AB, Contreras RJ. Leptin increases temperature-dependent chorda tympani nerve responses to sucrose in mice. Physiol Behav. 2012;107:533–539. doi: 10.1016/j.physbeh.2012.04.018. [DOI] [PubMed] [Google Scholar]
- Lush IE. The genetics of tasting in mice. VI. Saccharin, acesulfame, dulcin and sucrose. Genet Res. 1989;53:95–99. doi: 10.1017/s0016672300027968. [DOI] [PubMed] [Google Scholar]
- Lush IE, Hornigold N, King P, Stoye JP. The genetics of tasting in mice. VII. Glycine revisited, and the chromosomal location of Sac and Soa. Genet Res. 1995;66:167–174. doi: 10.1017/s0016672300034510. [DOI] [PubMed] [Google Scholar]
- Max M, et al. Tas1r3, encoding a new candidate taste receptor, is allelic to the sweet responsiveness locus. Sac Nat Genet. 2001;28:58–63. doi: 10.1038/ng0501-58. [DOI] [PubMed] [Google Scholar]
- Mennella JA, Finkbeiner S, Reed DR. The proof is in the pudding: children prefer lower fat but higher sugar than do mothers. Int J Obes. 2012;36:1285–1291. doi: 10.1038/ijo.2012.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mennella JA, Lukasewycz LD, Griffith JW, Beauchamp GK. Evaluation of the Monell forced-choice, paired-comparison tracking procedure for determining sweet taste preferences across the lifespan. Chem Senses. 2011;36:345–355. doi: 10.1093/chemse/bjq134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montmayeur JP, Liberles SD, Matsunami H, Buck LB. A candidate taste receptor gene near a sweet taste locus. Nat Neurosci. 2001;4:492–498. doi: 10.1038/87440. [DOI] [PubMed] [Google Scholar]
- Nakamura Y, et al. Diurnal variation of human sweet taste recognition thresholds is correlated with plasma leptin levels. Diabetes. 2008;57:2661–2665. doi: 10.2337/db07-1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newsome WT, Britten KH, Movshon JA. Neuronal correlates of a perceptual decision. Nature. 1989;341:52–54. doi: 10.1038/341052a0. [DOI] [PubMed] [Google Scholar]
- Nicola SM. The flexible approach hypothesis: unification of effort and cue-responding hypotheses for the role of nucleus accumbens dopamine in the activation of reward-seeking behavior. J Neurosci. 2010;30:16585–16600. doi: 10.1523/JNEUROSCI.3958-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nowlis GH, Kessen W. Human newborns differentiate differing concentrations of sucrose and glucose. Science. 1976;191:865–866. doi: 10.1126/science.1251200. [DOI] [PubMed] [Google Scholar]
- Perez CA, et al. A transient receptor potential channel expressed in taste receptor cells. Nat Neurosci. 2002;5:1169–1176. doi: 10.1038/nn952. [DOI] [PubMed] [Google Scholar]
- Reed DR, et al. Polymorphisms in the taste receptor gene (Tas1r3) region are associated with saccharin preference in 30 mouse strains. J Neurosci. 2004;24:938–946. doi: 10.1523/JNEUROSCI.1374-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sainz E, Korley JN, Battey JF, Sullivan SL. Identification of a novel member of the T1R family of putative taste receptors. J Neurochem. 2001;77:896–903. doi: 10.1046/j.1471-4159.2001.00292.x. [DOI] [PubMed] [Google Scholar]
- Salamone JD, Correa M, Farrar A, Mingote SM. Effort-related functions of nucleus accumbens dopamine and associated forebrain circuits. Psychopharmacology (Berl) 2007;191:461–482. doi: 10.1007/s00213-006-0668-9. [DOI] [PubMed] [Google Scholar]
- Samuelsen CL, Gardner MP, Fontanini A. Effects of cue-triggered expectation on cortical processing of taste. Neuron. 2012;74:410–422. doi: 10.1016/j.neuron.2012.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sclafani A, Abrams M. Rats show only a weak preference for the artificial sweetener aspartame. Physiol Behav. 1986;37:253–256. doi: 10.1016/0031-9384(86)90228-3. [DOI] [PubMed] [Google Scholar]
- Sclafani A, Clare RA. Female rats show a bimodal preference response to the artificial sweetener sucralose. Chem Senses. 2004;29:523–528. doi: 10.1093/chemse/bjh055. [DOI] [PubMed] [Google Scholar]
- Small DM, Green BG. A Proposed Model of a Flavor Modality. In: Murray MM, Wallace MT, editors. The Neural Bases of Multisensory Processes. Frontiers in Neuroscience; Boca Raton (FL): 2012. [Google Scholar]
- Small DM, Jones-Gotman M, Dagher A. Feeding-induced dopamine release in dorsal striatum correlates with meal pleasantness ratings in healthy human volunteers. Neuroimage. 2003;19:1709–1715. doi: 10.1016/s1053-8119(03)00253-2. [DOI] [PubMed] [Google Scholar]
- Smith DV, Ye MK, Li CS. Medullary taste responses are modulated by the bed nucleus of the stria terminalis. Chem Senses. 2005;30:421–434. doi: 10.1093/chemse/bji037. [DOI] [PubMed] [Google Scholar]
- Steiner JE. Human facial expressions in response to taste and smell stimulation. Adv Child Dev Behav. 1979;13:257–295. doi: 10.1016/s0065-2407(08)60349-3. [DOI] [PubMed] [Google Scholar]
- Talavera K, et al. Heat activation of TRPM5 underlies thermal sensitivity of sweet taste. Nature. 2005;438:1022–1025. doi: 10.1038/nature04248. [DOI] [PubMed] [Google Scholar]
- Treesukosol Y, Spector AC. Orosensory detection of sucrose, maltose, and glucose is severely impaired in mice lacking T1R2 or T1R3, but Polycose sensitivity remains relatively normal. Am J Physiol Regul Integr Comp Physiol. 2012;303:R218–235. doi: 10.1152/ajpregu.00089.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson DM, Lemon CH. Modulation of central gustatory coding by temperature. J Neurophysiol. 2013;110:1117–1129. doi: 10.1152/jn.00974.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson DM, Lemon CH. Temperature systematically modifies neural activity for sweet taste. J Neurophysiol. 2014;112:1667–1677. doi: 10.1152/jn.00368.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamashita S, Sato M. The effects of temperature on gustatory response of rats. J Cell Compar Physl. 1965;66:1–18. doi: 10.1002/jcp.1030660102. [DOI] [PubMed] [Google Scholar]
- Zhang Y, et al. Coding of sweet, bitter, and umami tastes: different receptor cells sharing similar signaling pathways. Cell. 2003;112:293–301. doi: 10.1016/s0092-8674(03)00071-0. [DOI] [PubMed] [Google Scholar]
- Zhao GQ, Zhang Y, Hoon MA, Chandrashekar J, Erlenbach I, Ryba NJ, Zuker CS. The receptors for mammalian sweet and umami taste. Cell. 2003;115:255–266. doi: 10.1016/s0092-8674(03)00844-4. [DOI] [PubMed] [Google Scholar]