This paper describes a new protocol for producing insulin-producing cells in vitro that represents another potential cell source for a diabetes cell therapy. These cells can be loaded into a protective device that is implanted under the skin. The device is designed to protect the cells from immune rejection by the implant recipient. The implant can engraft and respond to glucose by secreting insulin, thus potentially replacing the β cells lost in patients with type 1 diabetes.
Keywords: Embryonic stem cells, Insulin-secreting cells, Diabetes, Drug delivery systems, Cell therapy, Pancreas development
Abstract
The PEC-01 cell population, differentiated from human embryonic stem cells (hESCs), contains pancreatic progenitors (PPs) that, when loaded into macroencapsulation devices (to produce the VC-01 candidate product) and transplanted into mice, can mature into glucose-responsive insulin-secreting cells and other pancreatic endocrine cells involved in glucose metabolism. We modified the protocol for making PEC-01 cells such that 73%–80% of the cell population consisted of PDX1-positive (PDX1+) and NKX6.1+ PPs. The PPs were further differentiated to islet-like cells (ICs) that reproducibly contained 73%–89% endocrine cells, of which approximately 40%–50% expressed insulin. A large fraction of these insulin-positive cells were single hormone-positive and expressed the transcription factors PDX1 and NKX6.1. To preclude a significant contribution of progenitors to the in vivo function of ICs, we used a simple enrichment process to remove remaining PPs, yielding aggregates that contained 93%–98% endocrine cells and 1%–3% progenitors. Enriched ICs, when encapsulated and implanted into mice, functioned similarly to the VC-01 candidate product, demonstrating conclusively that in vitro-produced hESC-derived insulin-producing cells can mature and function in vivo in devices. A scaled version of our suspension culture was used, and the endocrine aggregates could be cryopreserved and retain functionality. Although ICs expressed multiple important β cell genes, the cells contained relatively low levels of several maturity-associated markers. Correlating with this, the time to function of ICs was similar to PEC-01 cells, indicating that ICs required cell-autonomous maturation after delivery in vivo, which would occur concurrently with graft integration into the host.
Significance
Type 1 diabetes (T1D) affects approximately 1.25 million people in the U.S. alone and is deadly if not managed with insulin injections. This paper describes the production of insulin-producing cells in vitro and a new protocol for producing the cells, representing another potential cell source for a diabetes cell therapy. These cells can be loaded into a protective device that is implanted under the skin. The device is designed to protect the cells from immune rejection by the implant recipient. The implant can engraft and respond to glucose by secreting insulin, thus potentially replacing the β cells lost in patients with T1D.
Introduction
In type I diabetes, autoimmunity results in the loss of insulin-producing β cells in the pancreas, leading to a loss of blood glucose control. The standard treatment of insulin injection is life-saving; however, daily glucose fluctuations remain higher than in healthy persons. This leaves patients vulnerable to complications including vascular and kidney disease, blindness, and lower limb amputation. Transplantation of cadaveric islets has made great progress in recent years and can result in insulin independence or lower insulin requirements [1]. This approach, however, is greatly limited by donor availability and the need for chronic immunosuppression.
Human embryonic stem cells (hESCs) possess the key features of essentially unlimited expansion and the theoretical ability to differentiate to any cell type, thus there has been considerable effort toward developing protocols to produce β-like cells from hESCs for transplantation and drug development. Significant advances have been made over the past 10 years in recapitulating pancreatic development in vitro with hESCs, using the extensive knowledge gained from studies of pancreatic organogenesis in model organisms [2]. We have previously shown that pancreatic endoderm cells (PECs) differentiated from hESCs via a four-stage protocol can, when transplanted into immunodeficient mice, form functional β cells and reverse streptozotocin-induced diabetes [3, 4]; this result has been reproduced by others [5]. We also demonstrated a scaled manufacturing system capable of producing the required quantity and quality of PEC-01 cells (PECs derived from CyT49 hESCs) to enable clinical application [6].
To potentially circumvent the need for immune suppression when implanting allogeneic hESC-derived pancreatic cells into patients with type I diabetes, a particularly attractive approach is to encapsulate the cell product. We optimized a planar macroencapsulation device containing a semipermeable barrier (the Encaptra drug delivery system) that can be loaded with PEC-01 cells and implanted subcutaneously. The implanted cell/device combination product (the VC-01 candidate product; ViaCyte, Inc., San Diego, CA, http://viacyte.com) can mature and function in mice (unpublished data), as described for nonencapsulated grafts, and has the benefit of being easily retrievable. Others have recently published results using a similar device, the TheraCyte device (TheraCyte Inc., Laguna Hills, CA, http://www.theracyte.com/TheTechnology.htm), with similar outcomes [7, 8]. We are currently testing VC-01 in a clinical trial (ClinicalTrials.gov identifier NCT02239354).
PECs generated from hESCs in vitro are mixed populations that contain multipotent pancreatic progenitors (PPs), pancreatic endocrine progenitors and precursors, and pancreatic endocrine cells. The endocrine cells in PECs often express multiple hormones and, in the case of cells expressing insulin, are deficient in expression of essential β cell markers (e.g., NKX6.1 and PDX1) and do not respond appropriately to glucose stimulation. These polyhormonal cells have been characterized in detail in recent studies [9–12]. Our group has demonstrated previously that purified and reaggregated cultures of PECs that were highly enriched for PPs and depleted of endocrine cells can differentiate to endocrine lineages in vivo including functional β cells, but purified and reaggregated polyhormonal endocrine cells do not [3]. This proves that PPs generated in vitro are sufficient to generate β cells in vivo. Whether the polyhormonal insulin-expressing cells are immature cells or whether they are inappropriately differentiated and not analogous to cell types produced during normal pancreas development is not known definitively.
In this study, we describe the development of a seven-stage differentiation protocol that produced highly enriched cultures of pancreatic endocrine cells in vitro (designated “ICs” [islet-like cells]). The first four stages of our IC protocol built on our PEC-01 protocol and increased the PP content into the range of 73%–80% while greatly reducing (polyhormonal) endocrine cell content. Stage 5 was designed to efficiently direct endocrine lineage commitment. Conditions supporting endocrine differentiation were optimized in stages 6 and 7, resulting in cultures consisting of 73%–89% endocrine cells and less than 10% PPs. Importantly, insulin-expressing cells produced by this method coexpressed other pancreatic hormones at a reduced frequency and coexpressed NKX6.1 and PDX1 at a higher frequency compared with the standard four-stage protocol. Methods producing endocrine cells from hESCs with these expression attributes have been described recently [13, 14].
The stage 7 cells, when encapsulated and implanted into mice, were capable of producing β-like cells that functioned at a level similar to that of implanted PEC-01 cells; however, because a low but significant percentage of PPs was present in these cultures, we could not rule out that it was PPs, and not endocrine cells produced in vitro, that resulted in functional cells in vivo. We used a dissociation/reaggregation process through which cell aggregates were enriched for endocrine cells and depleted of PPs. When implanted within devices, enriched ICs released human C-peptide in response to glucose, proving that highly purified endocrine cells differentiated in vitro from hESCs can produce functional β-like cells. Importantly, the kinetics of functional maturation in vivo are similar to those of PEC-01 cells, indicating that IC insulin-expressing cells are immature prior to implant.
Materials and Methods
All methods are described in the supplemental online data.
Results
Data from 10 independent experiments (numbered 1–10) are described.
Protocol Optimization for Pancreatic Progenitor Content
In our four-stage suspension culture-based PEC-01 protocol, hESC differentiation progresses through the intermediate stages of definitive endoderm (stage 1), primitive gut tube (stage 2), and posterior foregut (PF; stage 3) and then to a stage 4 population consisting of, on average (49 experiments), 32% PPs and 52% chromogranin A-expressing endocrine cells, the majority of which are polyhormonal [6]. In developing the IC protocol, we initially aimed to increase the PPs and reduce the endocrine content of the cell population at the end of stage 4. In PEC-01 and many similar differentiation protocols, neurogenin 3 (NEUROG3, also known as NGN3)—a master regulator of endocrine development [15]—is induced during stage 3. This occurs at the same time as PDX1 induction but well before other critical PP markers, such as NKX6.1 and PTF1A (i.e., in PF cells, not PPs). It seemed plausible that induction of the endocrine program in PF cells by NGN3 expression could be an important contributor to the generation of polyhormonal endocrine cells. We hypothesized that suppressing NGN3 through stages 3 and 4 might lead to decreased endocrine and increased PP content at the end of stage 4 and that, with further differentiation steps, endocrine cells that were more properly specified could be produced from PPs.
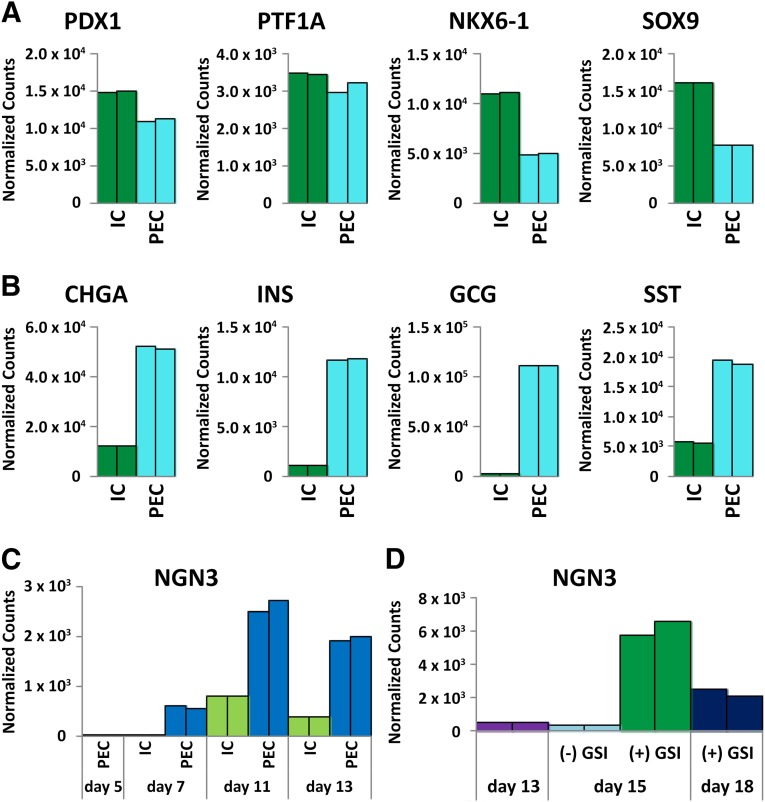
We modified the PEC-01 protocol by additionally treating with activin A, Wnt3A, and heregulin β1 at stage 3 (days 5–7) and with activin A and heregulin β1 at stage 4 (days 7–13). The PEC-01 and IC (stages 1–4) protocols are shown in schematic form in supplemental online Table 1. Relative to PEC-01, the IC population at stage 4 had more PPs (CHGA-negative [CHGA−], NKX6.1-positive [NKX6.1+], and PDX1+) and fewer endocrine (CHGA+) cells by flow cytometry (supplemental online Table 2). Specifically, IC differentiations yielded, on average, approximately 2.5-fold more PPs (75% vs. 32%) and more than 3-fold lower endocrine (14% vs. 52%) cell content (supplemental online Table 2). An activin concentration of either 50 or 75 ng/ml at stage 3 was used in the experiments shown in supplemental online Table 2. The higher concentration may have had a slightly increased effect (supplemental online Table 2) but also resulted in a lower cell yield (data not shown); therefore, activin at 50 ng/ml was used in all other experiments. The IC protocol was also compared with the PEC-01 protocol by digital RNA analysis. Stage 4 IC cultures had higher expression levels of PP genes (e.g., PDX1, NKX6.1, and SOX9) (Fig. 1A) and lower levels of endocrine genes (e.g., CHGA, INS, GCG, SST) (Fig. 1B) than did PEC-01 cultures. Looking at NGN3 expression over time (Fig. 1C), a marked reduction was observed during stage 3 in the IC protocol relative to PEC-01. Significant NGN3 expression occurred during stage 4 in ICs; however, it remained well below that in PEC-01 populations. Consequently, we achieved a cell population that was enriched in PP cells and that had much reduced (premature) endocrine commitment from which to optimize endocrine differentiation. Protocol stages 1–4 were essentially the same for all experiments except for a modestly higher stage 3 activin concentration in experiments 1 and 9, as noted.
Figure 1.
Digital RNA analysis of cell aggregates for pancreatic gene expression during the PEC-01 and IC protocols. Representative results from three or more experiments are shown. The two bars depicted for each sample represent biological replicates. RNA levels are shown for PP markers (PDX1, PTF1A, NKX6.1, and SOX9) (A) and endocrine markers (CHGA, INS, GCG, and SST) (B) on day 13 (experiment 8). (C): RNA levels of NGN3 over days 5–13 (stages 3 and 4; experiment 8). (D): NGN3 expression during stage 5 (days 13–15) and into stage 6 (day 18) of the IC protocol (experiment 6). Treatments were (+) GSI, GSI added during stage 5; (−) GSI, no GSI added. Abbreviations: GSI, γ-secretase inhibitor; IC, islet-like cells; NGN3, neurogenin 3; PEC, pancreatic endoderm cells derived from the PEC-01 protocol; PP, pancreatic progenitor.
Endocrine Lineage Commitment From PPs
The next step, stage 5 (days 13–15), was designed to maximize induction of NGN3. It is well documented that the Notch pathway is a negative regulator of NGN3 [16]. On ligand binding to Notch receptor, γ-secretase cleaves the receptor, releasing the Notch intracellular domain, which is part of a complex, including HES (hairy and enhancer of split) proteins, that represses NGN3 transcription [17]; therefore, γ-secretase inhibitors (GSIs) can inhibit Notch signaling activity and upregulate NGN3 expression. We tested the GSI designated RO4929097 [18] for its ability to promote endocrine differentiation from PPs. As seen in Figure 1D, NGN3 was strongly induced by this GSI, with a peak of expression at day 15 (of the days analyzed), followed by a reduction in expression by day 18 (i.e., 3 days after GSI withdrawal). Comparing NGN3 levels at day 15 in nontreated versus GSI-treated cells, a 17-fold enhancement was observed with treatment (Fig. 1D). Stage 5 treatments were the same in all experiments shown.
Optimization of Insulin Expression and Endocrine Maturation
During stages 6 and 7, we optimized for expression of insulin and mature β cell genes. A schematic of stages 5–7 is shown in supplemental online Table 3A. The resulting populations were analyzed by flow cytometry, digital gene expression, and immunofluorescence and for proinsulin and C-peptide content. Because testing was performed at intervals during the optimization process the data presented in this study had varying treatments at stages 6 and 7. This is detailed in the supplemental online data (Methods section) and in supplemental online Table 4. During the iterative process, some factors that had been beneficial in early experiments had no positive effect in later versions of the protocol. Examples of such factors are BMP4 and TTNPB, which were not used in the current IC protocol (supplemental online Table 4). The thyroid hormone triiodothyronine was added based on studies suggesting involvement in β cell maturation [19]. Nicotinamide has been shown to potentiate differentiation of fetal islets in vitro [20]. The Rho-associated kinases (ROCKs) have roles in multiple cellular functions such as adhesion, motility, proliferation, and apoptosis [21], and the ROCK inhibitor (ROCKi) Y-27632 can increase survival of hESCs [22].
As shown in supplemental online Table 5, stage 7 IC populations averaged 78% endocrine cells, of which 45% were INS+ and 22% were GCG+ cells. Approximately 66% of the INS+ cells were GCG− (i.e., single-hormone positive). SST has been observed to coexpress with INS in hESC-derived endocrine cells [23], but there were very few SST+ cells present in IC cultures (described in “Histological analysis of native and reaggregated ICs”). RNA analyses at stages 6 and 7 (Fig. 2) showed the strong induction of early endocrine genes (e.g., NGN3, PAX4), followed by hormone genes (e.g., INS, GCG and SST) and mature β cell markers (e.g., GCK, PCSK1 and SLC30A8). We note that these maturity genes are not necessarily specific to β cells. Other mature β cell genes were expressed at very low levels relative to islets (e.g., IAPP and MAFA; data not shown). The expression patterns of PAX4 and ARX are interesting regarding possible dominance of GCG over INS expression in various paradigms [3, 9, 24].
Figure 2.
RNA analysis of IC aggregates during stages 5–7 of differentiation (experiment 7) showing early endocrine (NGN3, PAX4, and ARX), hormone (INS, GCG, and SST), and mature β cell (GCK, IAPP, and G6PC2) markers. Representative results from three or more experiments are shown. The two bars depicted for each sample represent biological replicates. Abbreviations: GCG, glucagon; INS, insulin; NGN3, neurogenin 3; SST, somatostatin.
Enrichment of Endocrine Cells via Dissociation and Reaggregation
A major goal of this work was to prove that endocrine cells produced in vitro were able to mature and function in vivo. To accomplish this, a highly enriched endocrine preparation would be required, to preclude any contribution from PPs present in heterogeneous populations. We built on a relatively simple enrichment technique described previously [3]. That study showed that PEC-01 aggregates could be enzymatically dissociated and then reaggregated in rotating suspension culture. The resulting aggregates were enriched in endocrine cells and depleted of PPs. In contrast, the inclusion of ROCKi in the aggregation medium allowed survival and incorporation of PP into the aggregates. We removed ROCKi from the IC differentiation medium on the last day of stage 6, then dissociated and reaggregated ICs on the first day of stage 7, when the hormone genes were nearing peak expression. The cells within the resulting endocrine-enriched clusters were very loosely adhered, as observed previously [3]. The following day, we added ROCK inhibitor and a source of extracellular matrix in the form of very dilute, and thus soluble, Matrigel (Corning Inc., Corning, NY, https://www.corning.com). The combination of these two components greatly increased the degree of cell-cell interaction within the clusters and, consequently, the stability of the aggregates (supplemental online Fig. 1).
Reaggregated ICs were then differentiated through stage 7. Stages 5–7 for native (i.e., not reaggregated) ICs and reaggregated ICs are shown in schematic form in supplemental online Table 3. In Experiments 2 and 3, as measured by flow cytometry, reaggregated ICs contained 98% and 93% endocrine cells and 1% and 3% PPs, respectively (supplemental online Table 6). Compared with native ICs, reaggregated ICs were modestly enriched in endocrine markers (e.g., CHGA, INS, and GCG) and highly depleted for SOX9, a PP marker that is not expressed in endocrine cells (Fig. 3). The mature β cell genes PCSK1 and SLC30A8 were expressed in both native and reaggregated ICs at levels approaching (particularly in experiment 3) those found in isolated human islets (Fig. 3). As noted, these maturity genes are not necessarily specific to β cells.
Figure 3.
RNA analysis of IC aggregates at stage 7 of differentiation (experiments 2 and 3) comparing pancreatic gene expression in native ICs (IC), native ICs that had been cryopreserved and thawed (Cryo), and reaggregated ICs (RA) to purified human islets. Representative results from three or more experiments are shown. The two bars depicted for each sample represent biological replicates. Abbreviations: Expt., experiment; GCG, glucagon; IC, islet-like cells; INS, insulin; SST, somatostatin.
Histological Analysis of Native and Reaggregated ICs
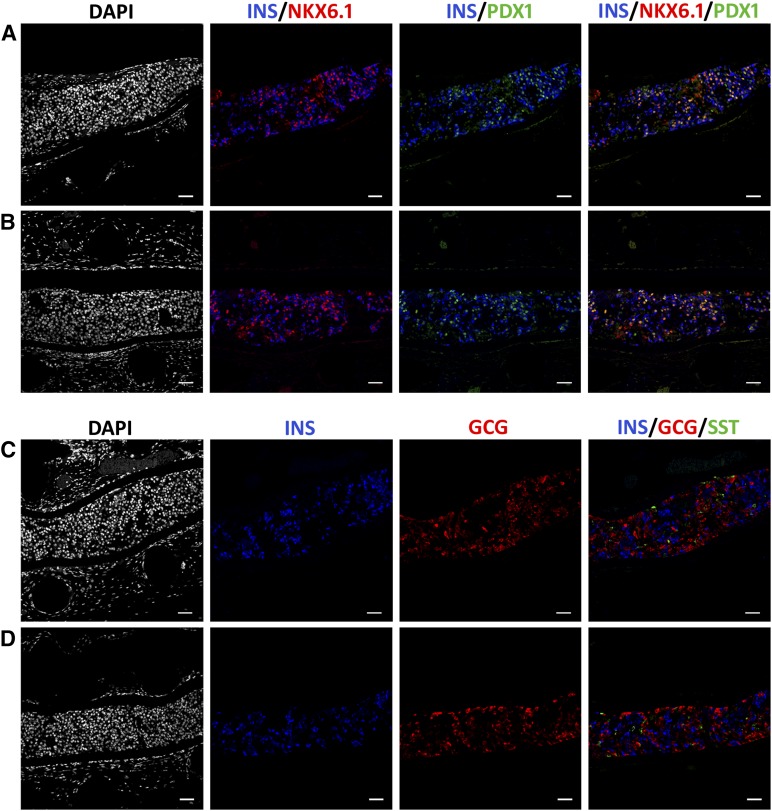
As noted, previously studied polyhormonal insulin-expressing cells lacked expression of NKX6.1 and PDX1. In contrast, many, if not most, stage 7 INS+ cells coexpressed these transcription factors, as seen by immunofluorescence, in native IC, reaggregated IC, and cryopreserved IC samples, shown in Figure 4 (experiment 3) and supplemental online Figures 2–4 (experiments 2, 3, and 4, respectively). Depletion of PPs in reaggregated samples can be observed (supplemental online Fig. 2). In correlation with noted flow cytometry data, when costained for INS, GCG, and SST, most of the stage 7 cells expressed only one hormone, although polyhormonal cells were observed (Fig. 4; supplemental online Figs. 2–4). There were very few SST+ cells in the cultures. These data indicate that stage 7 endocrine cells are more appropriately specified compared with polyhormonal cells and that cryopreservation and thaw or enrichment by reaggregation maintains this phenotype.
Figure 4.
Immunostaining of stage 7 islet-like cell (IC) aggregates (experiment 3, day 27). (A, B): Immunofluorescence for colocalization of INS, NKX6.1, and PDX1 in native IC aggregates (A) and reaggregated ICs (B). (C, D): Immunofluorescence for INS, GCG, and SST in native IC aggregates (C) and reaggregated ICs (D). Scale bars = 50 μm. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; GCG, glucagon; INS, insulin; SST, somatostatin.
Proinsulin Processing by ICs Compared With Human Islets
To further assess the maturity of stage 7 insulin-producing cells, we quantified the amount of C-peptide (which is generated by processing of proinsulin to insulin) present in the cells and the percentage of proinsulin that was fully processed compared with purified human islets (Fig. 5). The mean values for C-peptide levels, in micrograms of C-peptide per microgram of DNA (i.e., normalized to cell content), were 0.31 for islets and 0.15 for ICs (Fig. 5A). The mean percentages of proinsulin processed in islets and ICs were 96% and 90%, respectively (Fig. 5B). We concluded that ICs produced very significant levels of insulin and C-peptide (albeit approximately twofold lower than islets) and processed proinsulin essentially as efficiently as human islets do.
Figure 5.
Proinsulin processing by stage 7 IC aggregates and human islets. Samples tested were three lots of purified human islets, designated a, b, and c, as IC-a (experiment 9, day 34), IC-b (experiment 10, day 28), and IC-c (experiment 10, day 33). (A): Enzyme-linked immunosorbent assay (ELISA) for C-peptide content (in micrograms) normalized to DNA content (in micrograms). (B): Percentage of proinsulin processed, calculated using the ratio of C-peptide to C-peptide plus proinsulin (both determined by ELISA). Error bars represent the SEM of C-peptide. Abbreviations: C-PEP, C-peptide; IC, islet-like cells.
In Vivo Assessment of IC Populations
We tested whether ICs could mature and exhibit glucose-stimulated insulin secretion (GSIS) in the in vivo setting. Five cohorts of SCID/beige mice were implanted with ICs loaded into Encaptra macroencapsulation devices in 3 experiments (experiments 2–4). In experiment 2, native ICs were compared with reaggregated ICs; experiment 3 compared native ICs that had been cryopreserved with reaggregated ICs; and experiment 4 consisted of native ICs only. The results of GSIS assays in grafted mice from experiments 2 and 3, tested at 8, 12, and 16 weeks after transplant, are shown in Figure 6. The serum C-peptide values plotted in Figure 6, and additional values from experiment 4 are shown in supplemental online Table 7. The IC-implanted mouse cohort data were compared with our database of mice similarly implanted with VC-01 in unrelated experiments. At 8 weeks, the median glucose-stimulated C-peptide values were comparable for all cohorts and ranged from 326 to 539 pmol/l with the VC-01 controls at 375 pmol/l. As described (supplemental online Table 6), the reaggregated IC populations contained 98% (experiment 2) and 93% (experiment 3) CHGA+ endocrine cells. Importantly, these data prove that in vitro differentiated insulin-producing cells can mature and function in vivo. These cohorts were not tested at time points earlier than 8 weeks because in pilot experiments with ICs, we did not see a robust GSIS response prior to this time (data not shown); however, these earlier experiments, which were not yet well optimized, also had relatively low GSIS values at later time points (data not shown). At 12 weeks after implant, the GSIS values increased considerably in all cohorts, with median values for the IC-engrafted groups between 468 and 848 pm serum C-peptide at 60 minutes following glucose injection. There was no clear correlation between median C-peptide values and native, reaggregated, or cryopreserved cell implants. The median value for the PEC-01 grafts was considerably higher, at 1830 pmol/l C-peptide, compared with the IC grafts; however, the significance of this is unclear because of the relatively small number of animals implanted with ICs. Stimulated C-peptide values increased again at 16 weeks after implant, and the native IC and VC-01 groups had similar median glucose-stimulated C-peptide values. At this time point there was a trend toward the reaggregated IC cohorts having median values below those of the native IC groups and the PEC-01 controls. This would not be particularly surprising considering the harsh nature of the reaggregation process; however, as noted, the number of animals implanted was too few to be definitive. In summary, ICs, including populations that were essentially pure endocrine cells or that had been cryopreserved, were capable of maturing in a device in vivo and subsequently responding appropriately to glucose stimulation by releasing insulin.
Figure 6.
Enzyme-linked immunosorbent assay of human C-peptide levels in sera of implanted mice. Mice implanted with Encaptra-loaded aggregates were analyzed at the indicated postengraftment times (8, 12, or 16 weeks) for human C-peptide at fasting and 60 minutes after intraperitoneal glucose administration. Human C-peptide released by IC implants was compared with our historical data from mice with VC-01 implants. The x-axis labels for IC implants are as follows: IC (native ICs, experiment 2), Cryo (native ICs that had been cryopreserved and thawed, experiment 3), RA-1 (reaggregated ICs, experiment 2), RA-2 (reaggregated ICs, experiment 3). C-peptide values are shown both in picomoles per liter and nanograms per milliliter. Horizontal bars show median serum C-peptide content. Abbreviation: IC, islet-like cells.
Histological Analysis of Explanted IC Grafts
IC grafts were removed between 18 and 24 weeks after implant. GSIS assay results in the week of or the week prior to explant are shown in supplemental online Tables 8–10 (experiments 2–4, respectively). Robust function was maintained through the latest time point examined (24 weeks) (supplemental online Table 9). High-functioning grafts were chosen for immunofluorescent staining (Fig. 7; supplemental online Figs. 5, 6) (experiments 2, 3, and 4, respectively). Almost all INS+ cells expressed both PDX1 and NKX6.1. Hormones were very rarely, if ever, coexpressed. As with IC aggregates in vitro, there were few SST+ cells in the IC grafts. The immunofluorescent analyses of grafts from native, reaggregated, and cryopreserved native IC appeared substantially similar.
Figure 7.
Immunostaining of IC grafts in Encaptra devices at 22 weeks after implantation (experiment 2). (A, B): Immunofluorescence for colocalization of INS, NKX6.1, and PDX1 in grafts of native ICs (A) and reaggregated ICs (B). (C, D): Immunofluorescence for INS, GCG, and SST in grafts of native ICs (C) and reaggregated ICs (D). Scale bars = 50 μm. Abbreviations: DAPI, 4′,6-diamidino-2-phenylindole; GCG, glucagon; IC, islet-like cells; INS, insulin; SST, somatostatin.
Discussion
We developed an in vitro differentiation protocol that produces pancreatic endocrine cells from hESCs. This protocol built on our previous 4-stage protocol [6] by modifying stages 3 and 4 such that the NKX6.1+/PDX1+ PP pool was increased to 73%–80% (supplemental online Table 2). This was accomplished with the addition of activin A, Wnt3A, and heregulin β1. We previously found that activin A is capable of suppressing NGN3 and increasing PP content; however, at effective concentrations, activin A treatment resulted in a reduction of cell yield (data not shown). Wnt3A was capable of counteracting this cell loss, allowing the use of relatively high concentrations of activin A and, in fact, resulted in a net increase in cells at the end of stage 3 (data not shown). We have not addressed whether this effect of Wnt3A was due to increased proliferation, decreased cell death, decreased migration of cells from aggregates (and into the medium and subsequently aspirated on daily feeding), or a combination of these. Wnt signaling has been shown to promote proliferation of PPs in the developing mouse pancreas [25], and activin family signaling has been implicated in pancreas specification [26], although because of the complexity of these pathways, the particular ligands used in vivo are not well established. Others have used relatively low concentrations of activin A similarly in their hESC-to-pancreatic cell differentiation protocols [10]. Heregulin β1 also decreased the loss of cells, although not as potently as did Wnt3A. In addition, low concentrations of heregulin β1 were used because higher concentrations increased expression of CDX2, a posterior gut marker (data not shown).
When differentiated through stage 7, IC populations could be macroencapsulated, implanted, and matured in vivo to glucose-responsive β-like cells (Fig. 6). The ability to form functional grafts while protected from cell-cell interactions with the immune system is a critical component of any potential allogeneic cell therapy for diabetes. Furthermore, as proof of the efficacy of in vitro-derived endocrine cells, we showed that enriched ICs, largely depleted of PPs, can function similarly to native ICs (Fig. 6; supplemental online Table 6). Because ICs contain some polyhormonal cells, as seen in supplemental online Table 5, it is possible that they can mature and contribute to in vivo function. The small fraction of PPs remaining in enriched ICs (supplemental online Table 6) likely contribute, at most, a small fraction of the in vivo activity. Another critical component of a potential cell therapy is the ability to cryopreserve and recover the cell product, and that ability was demonstrated in this study (Fig. 6).
Two important reports that pancreatic endocrine cells produced from hESCs in vitro can also function in vivo were published recently [13, 14]. The in vivo studies in the reports were performed with nonencapsulated cells. Remarkably, the endocrine cells produced in vitro were shown to respond to glucose in vitro in both publications, with an approximately twofold stimulation index (which was similar to human islet controls). We do not see a reproducible response to glucose by insulin-producing cells generated with our IC protocol (data not shown). We have not directly compared the cells generated by the three protocols; nonetheless, some interesting observations can be made. All three protocols produce endocrine cells that are approximately 50% INS+, and a high proportion of these are GCG−. In addition, a high proportion of INS+ cells also are positive for the β cell markers PDX1 and NKX6.1. These important attributes are all in contrast to previously reported polyhormonal cells derived from hESCs. All three protocols produce relatively high proportions of PPs by the end of stage 4, (>55% in [13], 62% in [14] as shown in one flow cytometry plot, and an average of 75% shown in this study). We noted that the time at which endocrine induction was promoted by addition of a GSI was the same (day 13) among the 3 protocols. This may be related to the previously described [27] competence windows of pancreatic progenitors for the formation of the various endocrine cell types. Consequently, cells produced by all three regimens have very important similarities despite relatively divergent protocols. Concerning the time required to first observe in vivo function, we consistently saw a significant GSIS response at 8 weeks with both IC and PEC-01 protocols, not far from the 6 weeks reported by Rezania et al. [14]. This indicates that ICs require additional developmental maturation to acquire a β cell-like glucose response. Taken together, our data strongly suggest that the insulin-expressing cells within ICs most closely resemble immature β cells. These cells should prove to be a great starting point for gaining a better understanding of the final steps of β cell maturation.
Supplementary Material
Acknowledgments
We thank the California Institute for Regenerative Medicine for funding support in the form of Strategic Partnership Award SP1-06513. Confocal microscopy was performed at the University of California San Diego School of Medicine Microscopy Core, which uses funding from National Institute of Neurological Disorders and Stroke P30 Core Grant NS047101.
Author Contributions
A.D.A.: conception and design, collection and assembly of data, data analysis and interpretation, wrote the manuscript, final approval of manuscript; D.M.A.: design, collection and assembly of data, data analysis and interpretation, manuscript writing; M.A.M.: design, data analysis; A.B.: design, collection and assembly of data, data analysis; R.M.C.: assembly of data, data analysis J.K.P., J.R.K., C.H., R.S., A.Z.W., J.K., and M.A.F.: collection and assembly of data; E.J.K.: design, data analysis and interpretation; K.A.D.: design, data analysis and interpretation, manuscript writing, final approval of manuscript.
Disclosure of Potential Conflicts of Interest
All authors are employees of ViaCyte Inc. A.D.A. has uncompensated patents assigned to ViaCyte Inc., has compensated research funding, and is an uncompensated ViaCyte, Inc. shareholder. D.M.A. has uncompensated stock options. M.A.M., A.B., R.M.C., J.K.P., J.R.K., C.H., R.S., A.Z.W., J.K., and M.A.F. have compensated research funding and uncompensated stock options. E.J.K. and K.A.D. have compensated research funding and are uncompensated ViaCyte, Inc. shareholders.
References
- 1.McCall M, Shapiro AM. Update on islet transplantation. Cold Spring Harb Perspect Med. 2012;2:a007823. doi: 10.1101/cshperspect.a007823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pagliuca FW, Melton DA. How to make a functional β-cell. Development. 2013;140:2472–2483. doi: 10.1242/dev.093187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kelly OG, Chan MY, Martinson LA, et al. Cell-surface markers for the isolation of pancreatic cell types derived from human embryonic stem cells. Nat Biotechnol. 2011;29:750–756. doi: 10.1038/nbt.1931. [DOI] [PubMed] [Google Scholar]
- 4.Kroon E, Martinson LA, Kadoya K, et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat Biotechnol. 2008;26:443–452. doi: 10.1038/nbt1393. [DOI] [PubMed] [Google Scholar]
- 5.Rezania A, Bruin JE, Riedel MJ, et al. Maturation of human embryonic stem cell-derived pancreatic progenitors into functional islets capable of treating pre-existing diabetes in mice. Diabetes. 2012;61:2016–2029. doi: 10.2337/db11-1711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schulz TC, Young HY, Agulnick AD, et al. A scalable system for production of functional pancreatic progenitors from human embryonic stem cells. PLoS One. 2012;7:e37004. doi: 10.1371/journal.pone.0037004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bruin JE, Rezania A, Xu J, et al. Maturation and function of human embryonic stem cell-derived pancreatic progenitors in macroencapsulation devices following transplant into mice. Diabetologia. 2013;56:1987–1998. doi: 10.1007/s00125-013-2955-4. [DOI] [PubMed] [Google Scholar]
- 8.Kirk K, Hao E, Lahmy R, et al. Human embryonic stem cell derived islet progenitors mature inside an encapsulation device without evidence of increased biomass or cell escape. Stem Cell Res (Amst) 2014;12:807–814. doi: 10.1016/j.scr.2014.03.003. [DOI] [PubMed] [Google Scholar]
- 9.Basford CL, Prentice KJ, Hardy AB, et al. The functional and molecular characterisation of human embryonic stem cell-derived insulin-positive cells compared with adult pancreatic beta cells. Diabetologia. 2012;55:358–371. doi: 10.1007/s00125-011-2335-x. [DOI] [PubMed] [Google Scholar]
- 10.Bruin JE, Erener S, Vela J, et al. Characterization of polyhormonal insulin-producing cells derived in vitro from human embryonic stem cells. Stem Cell Res (Amst) 2014;12:194–208. doi: 10.1016/j.scr.2013.10.003. [DOI] [PubMed] [Google Scholar]
- 11.Hrvatin S, O’Donnell CW, Deng F, et al. Differentiated human stem cells resemble fetal, not adult, β cells. Proc Natl Acad Sci USA. 2014;111:3038–3043. doi: 10.1073/pnas.1400709111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Xie R, Everett LJ, Lim HW, et al. Dynamic chromatin remodeling mediated by polycomb proteins orchestrates pancreatic differentiation of human embryonic stem cells. Cell Stem Cell. 2013;12:224–237. doi: 10.1016/j.stem.2012.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pagliuca FW, Millman JR, Gürtler M, et al. Generation of functional human pancreatic β cells in vitro. Cell. 2014;159:428–439. doi: 10.1016/j.cell.2014.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rezania A, Bruin JE, Arora P, et al. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat Biotechnol. 2014;32:1121–1133. doi: 10.1038/nbt.3033. [DOI] [PubMed] [Google Scholar]
- 15.Rukstalis JM, Habener JF. Neurogenin3: A master regulator of pancreatic islet differentiation and regeneration. Islets. 2009;1:177–184. doi: 10.4161/isl.1.3.9877. [DOI] [PubMed] [Google Scholar]
- 16.Afelik S, Jensen J. Notch signaling in the pancreas: Patterning and cell fate specification. Wiley Interdiscip Rev Dev Biol. 2013;2:531–544. doi: 10.1002/wdev.99. [DOI] [PubMed] [Google Scholar]
- 17.Jorissen E, De Strooper B. Gamma-secretase and the intramembrane proteolysis of Notch. Curr Top Dev Biol. 2010;92:201–230. doi: 10.1016/S0070-2153(10)92006-1. [DOI] [PubMed] [Google Scholar]
- 18.Huynh C, Poliseno L, Segura MF, et al. The novel gamma secretase inhibitor RO4929097 reduces the tumor initiating potential of melanoma. PLoS One. 2011;6:e25264. doi: 10.1371/journal.pone.0025264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Aguayo-Mazzucato C, Zavacki AM, Marinelarena A, et al. Thyroid hormone promotes postnatal rat pancreatic β-cell development and glucose-responsive insulin secretion through MAFA. Diabetes. 2013;62:1569–1580. doi: 10.2337/db12-0849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Otonkoski T, Beattie GM, Mally MI, et al. Nicotinamide is a potent inducer of endocrine differentiation in cultured human fetal pancreatic cells. J Clin Invest. 1993;92:1459–1466. doi: 10.1172/JCI116723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Olson MF. Applications for ROCK kinase inhibition. Curr Opin Cell Biol. 2008;20:242–248. doi: 10.1016/j.ceb.2008.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Watanabe K, Ueno M, Kamiya D, et al. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat Biotechnol. 2007;25:681–686. doi: 10.1038/nbt1310. [DOI] [PubMed] [Google Scholar]
- 23.D’Amour KA, Bang AG, Eliazer S, et al. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat Biotechnol. 2006;24:1392–1401. doi: 10.1038/nbt1259. [DOI] [PubMed] [Google Scholar]
- 24.Spijker HS, Ravelli RB, Mommaas-Kienhuis AM, et al. Conversion of mature human β-cells into glucagon-producing α-cells. Diabetes. 2013;62:2471–2480. doi: 10.2337/db12-1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Murtaugh LC. The what, where, when and how of Wnt/β-catenin signaling in pancreas development. Organogenesis. 2008;4:81–86. doi: 10.4161/org.4.2.5853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wiater E, Vale W. Roles of activin family in pancreatic development and homeostasis. Mol Cell Endocrinol. 2012;359:23–29. doi: 10.1016/j.mce.2012.02.015. [DOI] [PubMed] [Google Scholar]
- 27.Johansson KA, Dursun U, Jordan N, et al. Temporal control of neurogenin3 activity in pancreas progenitors reveals competence windows for the generation of different endocrine cell types. Dev Cell. 2007;12:457–465. doi: 10.1016/j.devcel.2007.02.010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.