Abstract
Purpose
Previous studies show that inhibition of ABCB1 expression overcomes acquired docetaxel resistance in C4-2B-TaxR cells. In this study, we examined if anti-androgens, such as bicalutamide and enzalutamide, could inhibit ABCB1 activity and overcome resistance to docetaxel.
Experimental Design
ABCB1 efflux activity was determined using a rhodamine efflux assay. ABCB1 ATPase activity was determined by Pgp-Glo™ assay systems. The effects of the anti-androgens bicalutamide and enzalutamide on docetaxel sensitivity were determined by cell growth assays and tumor growth in vivo.
Results
We found that bicalutamide and enzalutamide inhibit ABCB1 ATP-binding cassette transporter activity through blocking ABCB1 efflux activity. Bicalutamide inhibited ABCB1 efflux activity by 40%, while enzalutamide inhibited ABCB1 efflux activity by ~60%. Both bicalutamide and enzalutamide inhibit ABCB1 ATPase activity. In addition, bicalutamide and enzalutamide inhibit ABCB1 efflux activity and desensitize docetaxel resistant and androgen receptor (AR)-negative DU145 cells. Combination of bicalutamide with docetaxel had a significant anti-tumor effect in both AR-positive and AR-negative docetaxel resistant xenograft models, suggesting that bicalutamide desensitizes docetaxel resistant cells to docetaxel treatment independent of AR status.
Conclusions
We identified a novel mechanism of action for anti-androgens such as bicalutamide and enzalutamide as inhibitors of ABCB1 efflux and ATPase activity. Bicalutamide and enzalutamide desensitize docetaxel resistant prostate cancer cells to docetaxel treatment independent of AR status. These studies may lead to the development of combinational therapies with bicalutamide/enzalutamide and docetaxel as an effective regiment to treat advanced castration resistant prostate cancer (CRPC) independent of AR status.
Keywords: prostate cancer, docetaxel, ABCB1, bicalutamide, enzalutamide, antiandrogens
Introduction
Prostate cancer is the most frequently diagnosed cancer and second most common cause of cancer-related death among men in the United States. Prostate cancer cells are dependent on androgen receptor (AR) signaling for growth and survival(1). Therefore, while prostate cancer patients will initially respond to androgen deprivation therapy (ADT), most will eventually develop castration-resistant prostate cancer (CRPC)(2, 3). Docetaxel, the standard first line treatment for CRPC, inhibits CRPC growth by binding β-tubulin and inhibiting mitotic cell division, leading to apoptotic cell death(4, 5). However, only approximately 50% of patients will respond to docetaxel and even those who initially benefit from the treatment will eventually develop resistance to the drug(6, 7). For patients with progressive CRPC previously treated with docetaxel, both abiraterone acetate (Zytiga) and enzalutamide (Xtandi) confer a statistically significant improvement in overall survival. This led to the recent FDA approval of both agents. Abiraterone inhibits intra-tumoral androgen biosynthesis by blocking CYP17A1(8). Enzalutamide impairs AR signaling by inhibiting AR nuclear translocation and DNA binding(9). These clinical results indicated that targeting AR signaling remains an important therapeutic strategy in docetaxel-resistant CRPC. Although both abiraterone and enzalutamide showed significant benefit to CRPC patients who failed docetaxel-based chemotherapy (both were also recently approved to treat CRPC patients prior docetaxel treatment), these treatment are quite expensive. According to recent cost-effectiveness analysis, abiraterone treatment cost about $123.4K/quality-adjusted life year compared to placebo, and the cost of enzalutamide was $437.6K/quality-adjusted life year compared to abiraterone (10). The cost of these two drugs makes treating patients with metastatic castration-resistant prostate cancer an extremely expensive process, and cost-effective therapeutic strategies are in urgent need.
Numerous studies have uncovered potential mechanisms involved in the development of docetaxel resistance in prostate cancer. Accumulation of certain β-tubulin isotypes such as βIII-tubulin and βIV-tubulin in tumor cells has been shown to be associated with reduced response rate in patients receiving docetaxel-based chemotherapy(11, 12). β-tubulin mutations, such as T26A, A595G and F270I, have been demonstrated to impair tubulin polymerization in the presence of docetaxel in prostate cancer and breast cancer cell lines (13, 14). Alterations of cell survival factors and apoptosis regulators such as Bcl-2, clusterin, HSPs and IAPs are also observed in docetaxel resistant prostate cancer cells (15–18). Aberrant activation of multiple cellular signaling pathways such as p53, NF-κB, PI3K-Akt and IL-6/STAT3 are associated with docetaxel resistance as well(13, 14, 19, 20). Additionally, recent studies also revealed that overexpressing Notch and Hedgehog signaling has high tumor-initiating capacity and the activation of GATA2-IGF axis confers taxane resistance in lethal prostate cancer (21, 22).
Our recent studies have also identified upregulation of ABCB1 as a common mechanism involved in acquired docetaxel resistance in CRPC(23). ABCB1 (P-glycoprotein, or MDR1) belongs to the ATP-binding cassette (ABC) transporters that use the energy produced during ATP hydrolysis to transport substrates, including taxanes such as docetaxel, across cell membranes and out of the cell. This diminishes the efficacy of the drug (24). Studies demonstrate that increased expression of ABCB1 confers resistance to chemotherapeutic agents (25–27). In addition, ABCB1 is overexpressed in many types of cancers including prostate, and ABCB1 expression is directly correlated with prostate tumor grade and stage (28).
In this study, we found that antiandrogens such as enzalutamide and bicalutamide inhibit ABCB1 efflux activity and resensitized docetaxel-resistant prostate cancer cells to docetaxel treatment. In addition, the previous-generation nonsteroidal antiandrogen, bicalutamide was able to overcome docetaxel resistance when combined with docetaxel in docetaxel-resistant prostate cancer cells both in vitro and in vivo. Our results suggest that co-treatment with bicalutamide and docetaxel could be developed as a combination therapy as an affordable regiment to treat docetaxel resistance CRPC.
Materials and Methods
Cell lines and cell culture
DU145 cells were obtained from the American Type Culture Collection (ATCC). All experiments with cell lines were conducted within 6 months of receipt from ATCC or resuscitation after cryopreservation. ATCC uses short tandem repeat (STR) profiling for testing and authentication of cell lines. C4-2B prostate cancer cells were kindly provided and authenticated by Dr. Leland Chung (Cedars-Sinai Medical Center, Los Angeles, CA). The cells were cultured in RPMI-1640 medium containing 10% complete fetal bovine serum (FBS) 100 units/mL penicillin and 0.1 mg/mL streptomycin and maintained at 37°C in a humidified incubator with 5% CO2. Docetaxel resistant C4-2B-TaxR (TaxR) and DU145-DTXR cells were generated from parental C4-2B and DU145 cells respectively by gradually increasing concentrations of docetaxel in the culture medium as previously described(23). TaxR and DU145-DTXR cells were maintained in 5 nM docetaxel-containing medium.
Materials and reagents
Docetaxel (CAS#114977-28-5) was purchased from TSZ CHEM (Framingham, MA). Bicalutamide (Cat: #B3209) were purchased from LKT Laboratories, Int. Elacridar (Cat: #143664-11-3) and Rhodamine 123 (Cat: #62667-70-9) were purchased from Sigma-Aldrich. Anti-ser15 phosphorylated p53 antibody was obtained from Cell Signaling Technology, Int. Cleaved PARP, wt-p53 and tubulin antibodies were obtained from Santa Cruz Biotechnologies (Santa Cruz, CA).
Preparation of Whole Cell Extracts
Cells were harvested, rinsed twice with PBS, and lysed in high-salt buffer (10 mM HEPES[pH 7.9], 0.25 M NaCl, 0.1% NP-40) supplemented with protease inhibitors (Roche, Basel, Switzerland). Whole cell extracts were prepared as previously described(29). Protein concentration was determined with Coomassie Plus protein assay kit (Piece, Rockford, IL).
Western blot Analysis
Equal amounts of total cell lysates (40 μg of protein) were loaded on 10% or 12% SDS-PAGE, and electrophoretically transferred onto nitrocellulose membranes. After blocking in 5% non-fat milk in 1×PBS/0.1% Tween-20 at room temperature for 1 hour, membranes were washed three times with 1×PBS/0.1% Tween-20. The membranes were incubated overnight with primary antibodies at 4°C. Proteins were visualized by enhanced chemiluminescence kit (Millipore) after incubation with the appropriate horseradish peroxidase-conjugated secondary antibodies as previously described(30).
Rhodamine 123 efflux assay
Cells were seeded in 6-well plates at a density of 2×105 cells per well. Cells were treated with elacridar, enzalutamide, and bicalutamide overnight, and then incubated with 1 μM Rhodamine 123 for 4 hours. The cells were then washed three times with PBS. Fluorescent pictures were taken. Each experiment was repeated at least 3 times.
Cell Growth Assay
Cells were seeded in 12-well plates at a density of 1×105 cells per well. Cells were treated and total cell numbers were counted using a Coulter cell counter.
Cell Death ELISA
TaxR cells were seeded in 12-well plates at a density of 1×105 cells per well and were treated. DNA fragmentation in the cytoplasmic fraction of cell lysates was determined using the Cell Death Detection ELISA kit (Roche, Indianapolis, IN) according to the manufacturer’s instructions. Apoptotic cell death was measured at 405 nm absorbance.
Clonogenic Assay
TaxR cells were treated for 6 hours. Two thousands cells were then plated in 100 mm dishes for 14 days. The cells were fixed with 4% formaldehyde for 10 min, stained with 0.5% crystal violet for 30 minutes, and the number of colonies was counted.
ABCB1 ATPase activity assay
ATPase activity was determined by Pgp-Glo™ assay systems (Promega, Madison, WI) following the user protocol provided by the manufacturer. Na3VO4 was used as an ABCB1 ATPase inhibitor. The activity of ABCB1 ATPase was measured in the presence of test compounds incubated with 25 μM recombinant human ABCB1 membranes at 37°C for 40 min. Luminescence was initiated by ATP detection buffer and luminescent activity was immediately read on Glomax 96-microplate luminometer (Promega, Madison, WI). To rule out the possibility of false positives (unexpected stimulation of ABCB1 ATPase activity by bicalutamide or enzalutamide), the activity of ABCB1 ATPase in cells receiving different concentrations of bicalutamide or enzalutamide plus Na3VO4 was compared with treatment with Na3VO4 alone. The differences between the average luminescent signals from Na3VO4 samples and bicalutamide or enzalutamide plus Na3VO4 samples reflect the luciferase inhibitory values (ΔRLU) by different concentrations of bicalutamide or enzalutamide. The ABCB1 ATPase activity was measured in the presence of docetaxel (50 μM) plus different concentrations of bicalutamide or enzalutamide. The average luminescent signals from docetaxel plus bicalutamide or enzalutamide were normalized to the luciferase inhibitory values (ΔRLU) of bicalutamide or enzalutamide. The difference in luminescent signal between Na3VO4-treated samples and untreated samples represents the basal ABCB1 ATPase activity. The ABCB1 ATPase activity affected by bicalutamide or enzalutamide was calculated by the difference in luminescent signal between Na3VO4-treated samples and the adjusted bicalutamide or enzalutamide-treated samples.
In vivo tumorigenesis assay
C4-2B, TaxR and DU145-DTXR cells (4×106) were mixed with matrigel (1:1) and injected subcutaneously into the flanks of 6 to 8 week-old male SCID mice. C4-2B derived tumor-bearing mice (tumor volume around 50–100 mm3) were randomized into two groups (with six tumors each group) and treated as follows: (i) vehicle control (5% Tween 80 and 5% ethanol in PBS, i.p.), (ii) docetaxel (10 mg/kg, p.o.). TaxR derived tumor-bearing mice and DU145-DTXR derived tumor-bearing mice (tumor volume around 50–100 mm3) were randomized into four groups (with six tumors each group) and treated as follows: (i) vehicle control (5% Tween 80 and 5% ethanol in PBS, i.p.), (ii) docetaxel (10 mg/kg, i.p., once a week), (iii) bicalutamide (25 mg/kg, p.o., 5 days a week), and (iv) docetaxel (10 mg/kg, i.p., once a week) + bicalutamide (25 mg/kg, p.o., 5 days a week). Tumors were measured using calipers twice a week and tumor volumes were calculated using length × width2/2. Tumor tissues were harvested after 3 weeks of treatment.
Immunohistochemistry
Tumors were fixed by formalin and paraffin-embedded tissue blocks were dewaxed, rehydrated, and blocked for endogenous peroxidase activity as previously described(31). Antigen retrieving was performed in sodium citrate buffer (0.01 mol/L, pH 6.0) in a microwave oven at 1,000 W for 3 minutes and then at 100W for 2-minutes. Nonspecific antibody binding was blocked by incubating with 10% FBS in PBS for 30 minutes at room temperature. Slides were then incubated with anti-Ki67 (1:500, NeoMarker) at room temperature for 30 minutes. Slides were then washed and incubated with biotin-conjugated secondary antibodies for 30 minutes, followed by incubation with avidin DH-biotinylated horseradish peroxidase complex for 30 minutes (Vectastain ABC Elite Kit; Vector Laboratories). The sections were developed with the Diaminobenzidine Substrate Kit (Vector Laboratories) and counterstained with hematoxylin. Nuclear stained cells were scored and counted in 3 different areas of the tumor. Images were taken with an Olympus BX51 microscope equipped with DP72 camera.
Statistical Analysis
All data presented are depicted as mean ± SD. Statistical significance between groups was determined by one-way ANOVA followed by the Scheffer procedure for comparison of means. P<0.05 was considered significant.
Results
ABCB1 efflux activity is increased in docetaxel resistant cells
Overexpression of the ABC transporters is known to be involved in multidrug resistance (MDR) in cancer(32). One of the major members of the ABC transporters related to MDR in cancer cells is ABCB1 (P-glycoprotein, ABCB1/MDR1). ABCB1 transports a large number of hydrophobic compounds out of cells including cancer chemotherapeutics agents such as taxanes (like docetaxel,) anthracyclines, and alkaloids (33). We previously demonstrated that ABCB1 is overexpressed in docetaxel resistant TaxR cells compared to docetaxel sensitive C4-2B parental cells (23). Similarly, others have shown that ABCB1 is also overexpressed in docetaxel resistant DU145R and CWR22rv1R cells derived from parental DU145 and CWR22rv1 cells, respectively (34). In the present study, we first examined ABCB1 activity in both parental C4-2B cells and ABCB1 overexpressing TaxR cells by using rhodamine efflux assay. Rhodamine 123 is a member of the rhodamine family of fluorescent dyes which is exported by ABCB1 and is routinely used to examine membrane transport by ABCB1(35). Due to the fact that the efflux of rhodamine 123 can be inhibited by other ABCB1 modulators, quantification of rhodamine 123 uptake into cells is an excellent indicator of ABCB1 transport activity, and is very useful in screening for novel ABCB1 inhibitors (36, 37). After 4 hours of incubation in 1μg/mL rhodamine 123, both parental C4-2B and TaxR cells were washed with 1×PBS three times and the intracellular rhodamine 123 intensities were observed under fluorescent microscope. As shown in Figure 1A, the intracellular rhodamine accumulation decreased significantly in ABCB1 overexpressing TaxR cells compared to C4-2B cells. TaxR cells had 6.9% rhodamine intake, whereas the rhodamine intake in C4-2B cells was 98.9%. These results suggest that almost all of the docetaxel was taken up into C4-2B parental cells and remained inside of the cells. Conversely, the majority of docetaxel was transported out of the TaxR cells.
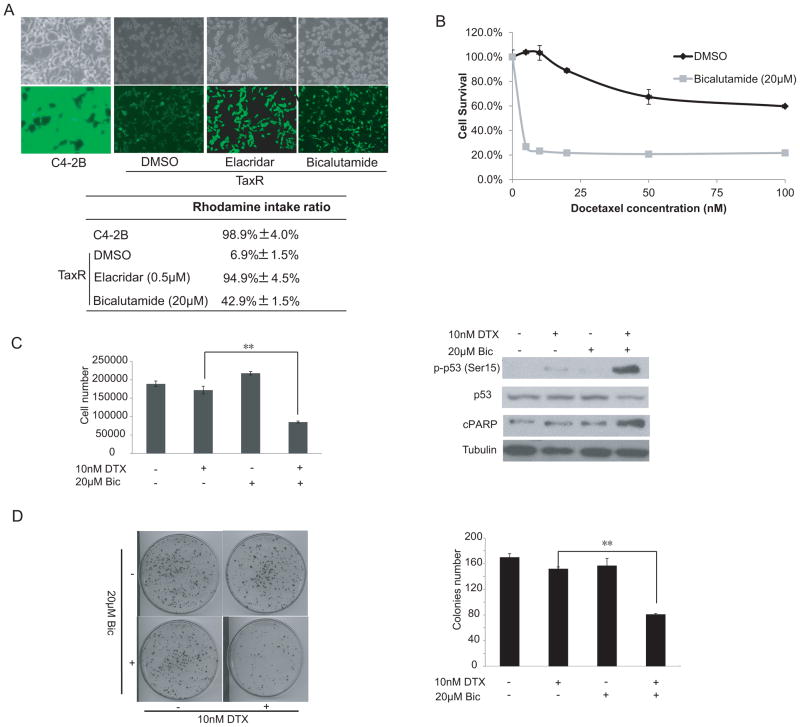
Figure 1.
Bicalutamide reverses docetaxel resistance of TaxR cells. (A) C4-2B and TaxR cells were seeded in 6-well plates at a density of 2×105 cells per well. TaxR cells were treated with 0.5 μM elacridar or 20 μM bicalutamide for 24 hrs. Cells were incubated with 1 μg/mL Rhodamine 123 for another 4 hrs. The cells were then washed 3 times with 1×PBS. Fluorescence was detected at an excitation wavelength of 480 nm and an emission wavelength of 534 nm. Top panel, fluorescent and phase contrast pictures were shown. Lower panel, rhodamine intake ratio. (B) TaxR cells were plated in 12-well plates and treated with increasing concentrations of docetaxel in the presence or absence of 20 μM bicalutamide. Cell number was counted after 24 hrs of treatment. (C) TaxR cells were treated with either 20 μmol/L bicalutamide or 10 nM docetaxel. After 24 hrs of treatment, cell number was counted (left panel). Whole-cell extracts were analyzed by Western blot using specific antibodies as indicated (right panel). (D) TaxR cells were treated with 10 nM docetaxel in the presence or absence of 20 μM bicalutamide. After 6 hrs of treatment, 1,000 cells were plated in 100 mm dishes in media containing complete FBS. The number of colonies was counted after 3 weeks and the results are presented as means ± SD of 2 experiments performed in duplicate. **, P < 0.01..
Bicalutamide inhibits the efflux activity of ABCB1 and reverses docetaxel resistance in TaxR cells in vitro
It has been reported that combination of androgen deprivation therapy with taxanes could improve therapeutic response of taxanes in CRPC (38). Therefore, we determined if antiandrogens such as bicalutamide could affect the response of TaxR cells to docetaxel treatment. We first determined if bicalutamide affects the efflux activity of ABCB1 in TaxR cells. A rhodamine efflux assay was performed in TaxR cells treated with bicalutamide or vehicle control. After the cells were incubated with bicalutamide or control for 24 hrs, the rhodamine intake ratio significantly increased from 6.9% to 42.9% (Fig 1A). This suggests that bicalutamide has the ability to affect ABCB1 efflux activity.
Reversal of ABCB1-mediated multidrug resistance can be achieved by either altering ABCB1 expression or inhibiting ABCB1 efflux activity (39, 40). Since ABCB1 was identified as a critical mechanism leading to docetaxel resistance in TaxR cells (23), we determined whether bicalutamide could restore docetaxel sensitivity by inhibiting ABCB1 efflux activity. In order to analyze the effect of bicalutamide on docetaxel resistance in prostate cancer cells, a cell growth assay was performed in TaxR cells. TaxR cells were treated with increasing concentrations of docetaxel for 24 hrs alone and in combination with 20 μM bicalutamide. As depicted in Fig 1B, bicalutamide decreased the IC50 value of docetaxel in TaxR cells from 140 nM to ~5 nM.
To further examine whether bicalutamide restores docetaxel sensitivity of TaxR cells, we treated TaxR cells with 10 nM docetaxel alone or in combination with 20 μM bicalutamide. Treatment with 20 μM bicalutamide had minimal effects on the growth of TaxR cells (Fig 1C, left panel). However, the combination of 10 nM docetaxel and 20 μM bicalutamide reduced the growth of TaxR cells to ~40%–50%. The combination treatment led to induction of p53 phosphorylation and cPARP expression (Fig 1C, right panel). The effect of combination bicalutamide/docetaxel treatment on TaxR cell clonogenic ability was also determined. The combination treatment reduced clone number by ~40%–50% (Fig 1D). Taken together, these data suggest that bicalutamide restores docetaxel sensitivity in TaxR cells in vitro.
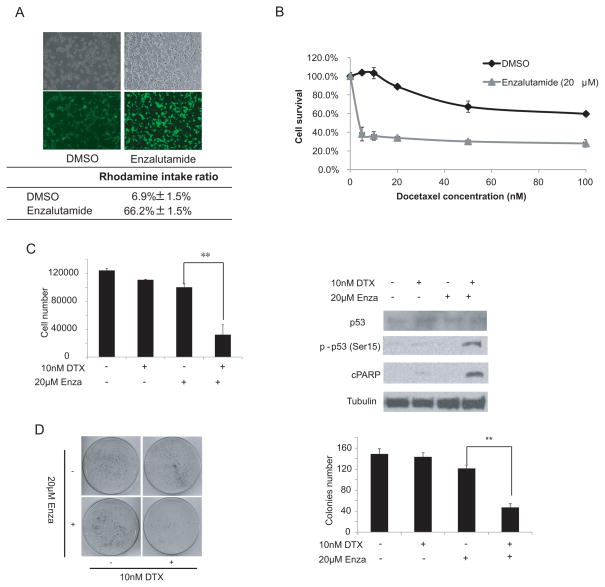
Enzalutamide is a recently FDA-approved drug for patients with CRPC who have failed docetaxel-based chemotherapy as well as for patients with CRPC prior to docetaxel treatment. Since enzalutamide is considered the second generation drug of bicalutamide, we examined if enzalutamide could affect ABCB1 activity similar to bicalutamide. TaxR cells were treated with enzalutamide for 48 hrs and rhodamine efflux activity was determined. TaxR cells had an increased rhodamine intake from 6.9% to 66.2% in the presence of enzalutamide treatment (Fig 2A), suggesting that enzalutamide could inhibit ABCB1 efflux activity. Next, we examined if enzalutamide could reverse resistance to docetaxel in TaxR cells. TaxR cells were treated with increasing concentrations of docetaxel for 24 hr alone or in combination with 20 μM enzalutamide. As shown in Fig 2B, 20 μM enzalutamide decreased the IC50 value of docetaxel in TaxR cells from 140nM to ~3nM. Similar to bicalutamide, the combination of 20 μM of enzalutamide with 10 nM docetaxel significantly reduced the growth of TaxR cells (Fig 2C, left panel) and induced phosphorylation of p53 at Ser15 and the cleavage of PARP (Fig 2C, right panel). Additionally, combination of docetaxel with enzalutamide reduced clone number by ~50% in clonogenic assays (Fig 2D), suggesting that the combination of docetaxel and enzalutamide induces apoptotic cell death.
Figure 2.
Enzalutamide reverses docetaxel resistance of TaxR cells. (A) TaxR cells were treated with either 20 μmol/L enzalutamide or with 10 nM docetaxel for 24 hrs. Cells were incubated with 1 μg/mL Rhodamine 123 for another 4 hrs. The cells were then washed three times with 1×PBS. Fluorescence was detected at an excitation wavelength of 480 nm and an emission wavelength of 534 nm. Fluorescent and phase contrast pictures are shown. Lower panel is the rhodamine intake ratio. (B) TaxR cells were plated in 12-well plates and treated with increasing concentrations of docetaxel in the presence or absence of 20 μM enzalutamide. Cell number was counted after 24 hrs of treatment. (C) TaxR cells were treated with either 20 μmol/L enzalutamide or with 10 nM docetaxel. After 24 hrs of treatment, the cell number was counted (left panel). Whole-cell extracts were analyzed by Western blot using specific antibodies as indicated (right panel). (D) TaxR cells were treated with 10 nM docetaxel in the presence or absence of 20 μM enzalutamide. After 6 hrs of treatment, 1,000 cells were plated in 100 mm dishes in media containing complete FBS. The number of colonies was counted after 3 weeks. Results are presented as means ± SD of 2 experiments performed in duplicate. **, P < 0.01.
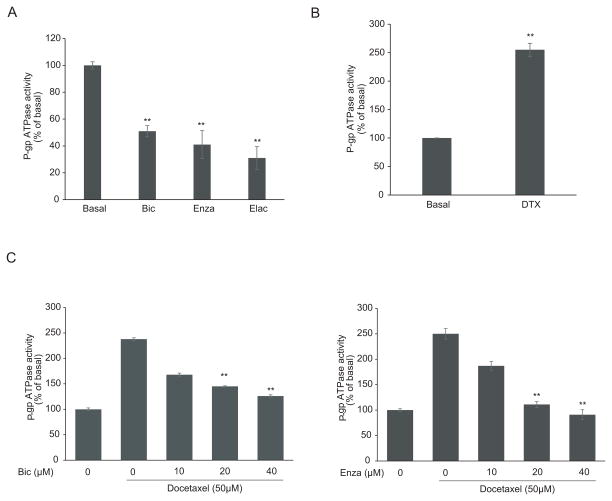
Antiandrogens inhibit the ATPase activity of ABCB1
ABCB1, also known as P-glycoprotein, functions as an ATP-dependent drug efflux transporter. Compounds that interact with ABCB1 can either be identified as substrates or inhibitors which stimulate or inhibit the ATPase activity of ABCB1 respectively. In this study, we tested the effects of bicalutamide and enzalutamide on ABCB1 ATPase activity using the Pgp-Glo™ Assay Systems. As shown in Fig 3A, the ABCB1 ATPase inhibitor elacridar at 2.5μM inhibited ABCB1 ATPase activity by 69%. Anti-androgens bicalutamide and enzalutamide decreased ABCB1 ATPase activity in a dose-dependent manner, with 45% and 60% inhibition at concentration of 40μM respectively, indicating both anti-androgens functioned as ABCB1 ATPase inhibitors. It has been reported that paclitaxel and docetaxel are both substrates for ABCB1-mediated efflux and were able to stimulate ABCB1 ATPase activity (41, 42). In addition, we tested the effect of docetaxel on ABCB1 ATPase activity. As shown in Fig 3B, 50 μM of docetaxel significantly stimulated ABCB1 ATPase activity by ~150%. Bicalutamide (Fig 3C, left panel) and enzalutamide (Fig 3C, right panel) inhibit the docetaxel-stimulated ABCB1 ATPase activity in a dose-dependent manner. Taken together, these data along with the rhodamine efflux assay suggested bicalutamide and enzalutamide reversed the docetaxel resistance in ABCB1-overexpressing cells through inhibiting the ATPase activity of ABCB1.
Figure 3.
Effects of bicalutamide and enzalutamide on ABCB1 ATPase activity. ABCB1 ATPase activity in response to 40 μM bicalutamide and 40 μM enzalutamide and 2.5 μM elacridar (A) or 50 μM docetaxel (B) (C) Effects of bicalutamide (10, 20 and 40 μM) and enzalutamide (10, 20 and 40 μM) on 50 μM docetaxel-induced ABCB1 ATPase activity. The value was normalized to the basal P-gp ATPase activity as described in Materials and Methods. **, P < 0.01.
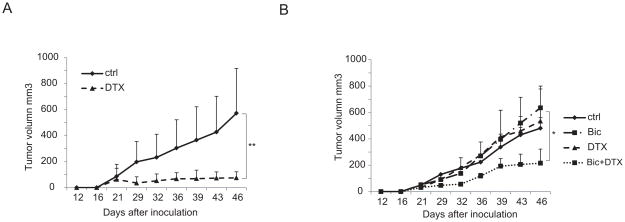
Bicalutamide reverses docetaxel resistance in TaxR cells in vivo
To test if bicalutamide can overcome docetaxel resistance in vivo, docetaxel resistant TaxR cells and parental C4-2B cells were injected into SCID mice s.c. on the flank. The mice developed tumors three weeks after injection. The mice injected with C4-2B cells were then divided into two groups to receive either vehicle or docetaxel treatments. The mice injected with TaxR cells were divided into four groups to receive either vehicle as controls, docetaxel or bicalutamide alone or with combination treatment. As hypothesized, docetaxel significantly repressed C4-2B tumor growth (Fig 4A). Neither docetaxel nor bicalutamide treatment alone inhibited tumor growth in TaxR cells, while combinatory treatment with docetaxel and bicalutamide significantly inhibited tumor growth of TaxR cells (Fig 4B). These results indicate that TaxR cells are resistant to docetaxel, and that combination of bicalutamide with docetaxel overcomes this resistance both in vitro and in vivo.
Figure 4.
Combination treatment with bicalutamide and docetaxel overcomes docetaxel resistance. Six to eight week-old SCID mice were inoculated s.c. with 4×106 C4-2B or TaxR cells on the flank. (A) Mice carrying C4-2B tumors were divided into two groups to receive either vehicle or docetaxel (10mg/kg body weight, i.p.) once a week. (B) Mice carrying TaxR tumors were divided into four groups and treated with vehicle, docetaxel (10 mg/kg body weight, i.p., one day a week) or bicalutamide (25 mg/kg body weight, esophageal gavaging, 5 days a week) alone or a combination of docetaxel (10 mg/kg body weight, i.p., one day a week )+ bicalutamide (25 mg/kg body weight, esophageal gavaging, 5 days a week). Tumor sizes were measured twice a week. *, P < 0.05. **, P < 0.01. DTX, docetaxel, Bic, bicalutamide.
Bicalutamide reverses docetaxel resistance in DU145-DTXR cells
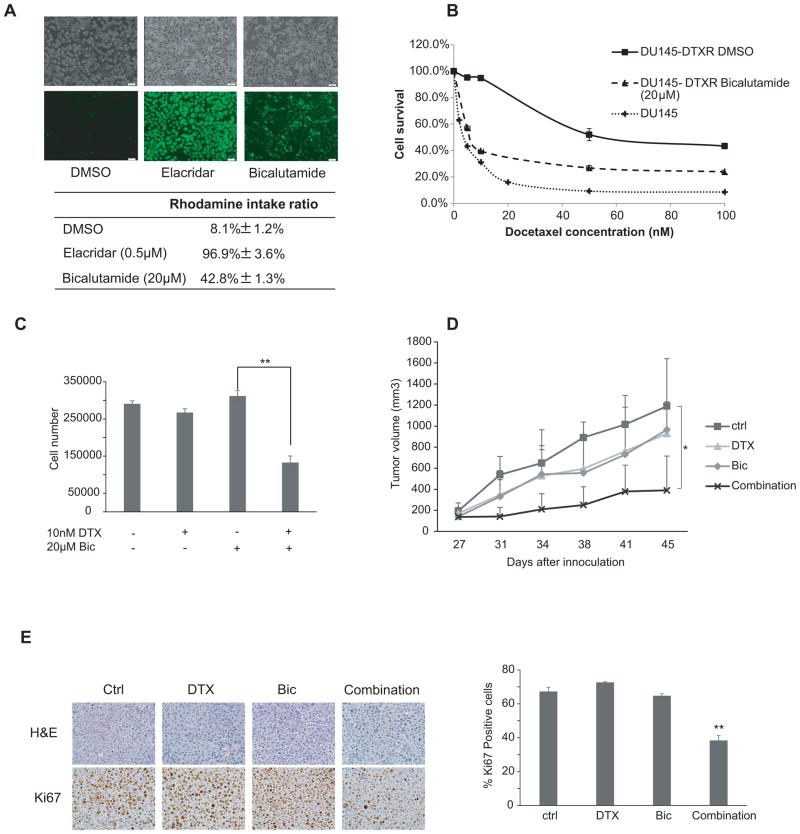
DU145-DTXR cells are another docetaxel resistant prostate cancer cell line generated from AR-negative parental DU145 cells in our laboratory (23). Our previous data showed that similar to TaxR cells, ABCB1 is overexpressed in DU145-DTXR cells compared to parental DU145 cells (23). These data suggested that overexpression of ABCB1 serves as a general mechanism of docetaxel resistance in prostate cancer. To determine whether the effect of bicalutamide on ABCB1 efflux activity in DU145-DTXR is similar to TaxR cells, rhodamine efflux assay was performed. Bicalutamide increased the rhodamine intake ratio from 8.1% to 42.8% (Fig 5A). The IC50 to docetaxel in DU145-DTXR cells is 52.0 nM compared with 4.3 nM in parental DU145 cells (Fig 5B). Further study has shown that 20μM bicalutamide decreased the IC50 value of docetaxel in DU145-DTXR cells from 52.0 nM to ~7.5 nM (Fig 5B). To test the effects of bicalutamide on docetaxel resistance of DU145-DTXR cells, we treated DU145-DTXR cells with 10 nM docetaxel alone or in combination with 20 μM bicalutamide. Although docetaxel or bicalutamide alone had only minimal effects on cell growth, combining docetaxel and bicalutamide significantly inhibited the growth of DU145-DTXR cells (Fig 5C). These effects were further verified in vivo. Four million DU145-DTXR cells were injected under the flank of 4–6 week old male SCID mice. The mice were randomly divided into four groups when the tumor size reached 50–100 mm3 to receive vehicle control, docetaxel or bicalutamide alone or in a combined treatment. The combination group had a significantly slower tumor growth rate and resulted in much smaller tumors compared with the other three treatment groups (Fig 5D). Representative tumor samples were analyzed by IHC for Ki67 expression. Combination treatment further decreased Ki67 expression (Fig 5E). Altogether, these data indicate that bicalutamide reversed ABCB1-mediated docetaxel resistance in DU145-DTXR cells in vitro and in vivo.
Figure 5.
Bicalutamide reverses docetaxel resistance in DU145-DTXR cells. (A) DU145-DTXR cells were treated with 0.5 μM elacridar or 20μM bicalutamide for 24 hrs. Cells were incubated with 1 μg/mL Rhodamine 123 for another 4 hrs. The cells were then washed 3 times with 1×PBS. Fluorescence was detected at an excitation wavelength of 480 nm and an emission wavelength of 534 nm. Top panel, fluorescent and phase contrast pictures were shown. Lower panel, rhodamine intake ratio. (B) DU145 and DU145-DTXR cells were plated in 12-well plates. DU145 were treated with different concentrations of docetaxel as indicated. DU145-DTXR cells were treated with different concentrations of docetaxel as indicated in the presence or absence of 20 μM bicalutamide. Cell number was counted after 24 hrs of treatment. (C) DU145-DTXR cells were plated in 12-well plates and treated with 10nM docetaxel in the presence or absence of 20 μM bicalutamide. Cell number was counted after 24 hrs of treatment. (D) Six to eight week-old SCID mice were inoculated s.c. with 4×106 DU145-DTXR cells subcutaneously. Mice carrying tumors were divided into four groups and treated with vehicle, docetaxel (10 mg/kg body weight, i.p., one day a week) or bicalutamide (25 mg/kg body weight, esophageal gavaging, 5 days a week) alone or a combination of docetaxel (10 mg/kg body weight, i.p., one day a week )+ bicalutamide (25 mg/kg body weight, esophageal gavaging, 5 days a week). Tumor sizes were measured twice a week. (E) Ki67 was analyzed in tumor tissues by IHC staining and quantified as described in Materials and Methods. *, P < 0.05. **, P < 0.01.
Discussion
Docetaxel has been the first-line treatment for metastatic CRPC since 2004. Acquired resistance to docetaxel treatment is one of the major concerns regarding prostate cancer therapy(43). Previous studies from our laboratory have observed that ABCB1 is up-regulated in docetaxel resistant TaxR prostate cancer cells and that knockdown of ABCB1 expression sensitized resistant TaxR cells to docetaxel(23). In the current study, we demonstrate that ABCB1 efflux activity in TaxR cells is significantly increased compared to parental C4-2B cells. Anti-androgens such as enzalutamide and bicalutamide inhibit ABCB1 efflux activity and reverse the ABCB1-mediated docetaxel resistance in prostate cancer. Furthermore, combination of bicalutamide or enzalutamide with docetaxel significantly enhances the cytotoxicity of docetaxel independent of AR status.
Overexpression of ABCB1 is one of the main mechanisms leading to docetaxel resistance in CRPC. ABCB1 activity in cultured cells has been assessed by monitoring the efflux of several small, fluorescent molecules such as rhodamine 123 from cells (35). Efflux of rhodamine 123 was reduced when TaxR cells were treated with either bicalutamide or enzalutamide. Bicalutamide inhibited ABCB1 efflux activity by 40% and decreased the IC50 of docetaxel in TaxR cells from 140 nM to around 5 nM, while enzalutamide inhibited ABCB1 efflux activity by ~60%. Reversal of ABCB1-mediated docetaxel resistance can be achieved by altering ABCB1 expression.. The ability of blockage of ABCB1 efflux activity by bicalutamide and enzalutamide is through inhibition of ABCB1 ATPase activity. Both bicalutamide and enzalutamide act as anti-androgens to treat AR -positive prostate cancer. Our finding that bicalutamide/enzalutamide block ABCB1 efflux activity via inhibition of ABCB1 ATPase activity provides a new mechanism of action of these drugs. This novel mechanism of action provides a scientific rational for a combination treatment strategy of bicalutamide/enzalutamide with docetaxel for advanced prostate cancer independent of the status of androgen receptor.
For patients with progressive CRPC previously treated with docetaxel, enzalutamide confers a statistically significant improvement in overall survival, leading to its recent US FDA approval. Enzalutamide impairs AR signaling by inhibiting AR nuclear translocation and DNA binding (31). Clinical studies indicate that targeting AR signaling remains an important therapeutic strategy in docetaxel-resistant CRPC. In this study, we showed a significant growth inhibition effect of enzalutamide on TaxR cells when combined with docetaxel through a reduction of ABCB1 efflux activity. Combination of enzalutamide and docetaxel inhibited cell growth and clonogenic ability accompanied with increased cellular apoptosis of TaxR cells. These findings suggest an additional mechanism of action by enzalutamide in CPRC through inhibition of ABCB1 efflux activity.
Similar to enzalutamide, the previous generation of anti-androgen, bicalutamide, was also found to inhibit ABCB1 efflux activity and resensitize docetaxel resistant cells to docetaxel treatment. Clinically, patients with advanced prostate cancer were initially treated with gonadotropin releasing hormone (GnRH) analogs (which suppress active androgens) alone or in combination with anti-androgens such as bicalutamide. Launched in 1995, bicalutamide is one of the first-generation pure anti-androgens binding to and inhibiting the androgen receptor and is widely used to treat prostate cancer in patients who fail androgen deprivation therapy. Despite the initial response to bicalutamide treatment, almost all of the patients develop CRPC and no longer respond to the drug. Docetaxel is the standard first-line treatment for CRPC. However, relapse eventually occurs due to the development of resistance to docetaxel. Enzalutamide and abiraterone are recently approved therapies for CRPC after the cancer fails to respond to docetaxel. Based on the fact that bicalutamide inhibited ABCB1 efflux activity and reversed the resistance to docetaxel similar to enzalutamide, bicalutamide could be developed as a combination therapy with docetaxel to effectively treat docetaxel resistant CRPC. Although both abiraterone and enzalutamide have significant benefit for CRPC patients who fail docetaxel-based chemotherapy, these treatments are expensive. Compared to enzalutamide or abiraterone treatment, combination treatment of bicalutamide with docetaxel is more cost effective, and could be developed to treat patients with CRPC who fail docetaxel therapy.
In summary, we identified a novel mechanism of action of the anti-androgens such as bicalutamide and enzalutamide as inhibitors for ABCB1 efflux and ATPase activity. Bicalutamide and enzalutamide desensitize docetaxel resistant prostate cancer cells to docetaxel treatment. Our studies suggest for the first time that bicalutamide and enzalutamide reverses docetaxel resistance in vitro and in vivo by inhibition of ABCB1 efflux activity, and may be developed as a combination therapy with docetaxel as an effective regiment to treat advanced CRPC independent of AR status.
Translational Relevance.
Acquired resistance to docetaxel is a big challenge in the treatment of advanced prostate cancer. Reduced intracellular concentration of the drug through alteration of ABCB1 activity contributes to docetaxel resistance. In this study, we identified a novel mechanism of action for anti-androgen drugs such as bicalutamide and enzalutamide as inhibitors for ABCB1 efflux and ATPase activity. The anti-androgens increase the cytotoxicity of docetaxel in docetaxel resistant cells independent of androgen receptor status. Furthermore, combination of bicalutamide with docetaxel had a significant anti-tumor effect in both AR-positive and AR-negative docetaxel resistant xenograft models, suggesting that bicalutamide resensitizes docetaxel resistant cells to docetaxel treatment independent of AR status. These studies may lead to the development of combinational therapies with bicalutamide/enzalutamide and docetaxel as effective regiments to treat advanced CRPC independent of AR status.
Acknowledgments
Grant Support:
This work is supported in part by grants NIH/NCI CA140468, CA168601, CA179970 (A.C. Gao), US Department of Veterans Affairs, Office of Research and Development VA Merits I01 BX000526 (A.C. Gao), and by resources from the VA Northern California Health Care System, Sacramento, California.
Footnotes
Potential conflicts of interest: None
References
- 1.Trapman J, Cleutjens KB. Androgen-regulated gene expression in prostate cancer. Semin Cancer Biol. 1997;8:29–36. doi: 10.1006/scbi.1997.0050. [DOI] [PubMed] [Google Scholar]
- 2.Dutt SS, Gao AC. Molecular mechanisms of castration-resistant prostate cancer progression. Future Oncology. 2009;5:1403–13. doi: 10.2217/fon.09.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Harris WP, Mostaghel EA, Nelson PS, Montgomery B. Androgen deprivation therapy: progress in understanding mechanisms of resistance and optimizing androgen depletion. Nat Clin Pract Urol. 2009;6:76–85. doi: 10.1038/ncpuro1296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fitzpatrick JM, de Wit R. Taxane Mechanisms of Action: Potential Implications for Treatment Sequencing in Metastatic Castration-resistant Prostate Cancer. European Urology. doi: 10.1016/j.eururo.2013.07.022. [DOI] [PubMed] [Google Scholar]
- 5.Herbst RS, Khuri FR. Mode of action of docetaxel – a basis for combination with novel anticancer agents. Cancer Treatment Reviews. 2003;29:407–15. doi: 10.1016/s0305-7372(03)00097-5. [DOI] [PubMed] [Google Scholar]
- 6.Kim SJ, Kim SI. Current treatment strategies for castration-resistant prostate cancer. Korean J Urol. 2011;52:157–65. doi: 10.4111/kju.2011.52.3.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Galletti E, Magnani M, Renzulli ML, Botta M. Paclitaxel And Docetaxel Resistance: Molecular Mechanisms and Development of New Generation Taxanes. Chem Med Chem. 2007;2:920–42. doi: 10.1002/cmdc.200600308. [DOI] [PubMed] [Google Scholar]
- 8.de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L, et al. Abiraterone and Increased Survival in Metastatic Prostate Cancer. New England Journal of Medicine. 2011;364:1995–2005. doi: 10.1056/NEJMoa1014618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Scher HI, Fizazi K, Saad F, Taplin M-E, Sternberg CN, Miller K, et al. Increased Survival with Enzalutamide in Prostate Cancer after Chemotherapy. New England Journal of Medicine. 2012;367:1187–97. doi: 10.1056/NEJMoa1207506. [DOI] [PubMed] [Google Scholar]
- 10.Wilson L, Tang J, Zhong L, Balani G, Gipson G, Xiang P, et al. New therapeutic options in metastatic castration-resistant prostate cancer: Can cost-effectiveness analysis help in treatment decisions? Journal of Oncology Pharmacy Practice. 2013 doi: 10.1177/1078155213509505. [DOI] [PubMed] [Google Scholar]
- 11.Shalli K, Brown I, Heys SD, Schofield AC. Alterations of β-tubulin isotypes in breast cancer cells resistant to docetaxel. The FASEB Journal. 2005 doi: 10.1096/fj.04-3178fje. [DOI] [PubMed] [Google Scholar]
- 12.Berrieman HK, Lind MJ, Cawkwell L. Do β-tubulin mutations have a role in resistance to chemotherapy? The Lancet Oncology. 2004;5:158–64. doi: 10.1016/S1470-2045(04)01411-1. [DOI] [PubMed] [Google Scholar]
- 13.Hari M, Loganzo F, Annable T, Tan X, Musto S, Morilla DB, et al. Paclitaxel-resistant cells have a mutation in the paclitaxel-binding region of β-tubulin (Asp26Glu) and less stable microtubules. Molecular Cancer Therapeutics. 2006;5:270–8. doi: 10.1158/1535-7163.MCT-05-0190. [DOI] [PubMed] [Google Scholar]
- 14.Hara T, Ushio K, Nishiwaki M, Kouno J, Araki H, Hikichi Y, et al. A mutation in β-tubulin and a sustained dependence on androgen receptor signalling in a newly established docetaxel-resistant prostate cancer cell line. Cell Bio Int. 2010;34:177–84. doi: 10.1042/CBI20090030. [DOI] [PubMed] [Google Scholar]
- 15.Seruga B, Ocana A, Tannock IF. Drug resistance in metastatic castration-resistant prostate cancer. Nat Rev Clin Oncol. 2011;8:12–23. doi: 10.1038/nrclinonc.2010.136. [DOI] [PubMed] [Google Scholar]
- 16.Yoshino T, Shiina H, Urakami S, Kikuno N, Yoneda Y, Shigeno K, et al. Bcl-2 Expression as a Predictive Marker of Hormone-Refractory Prostate Cancer Treated with Taxane-Based Chemotherapy. Clin Cancer Res. 2006;12:6116–24. doi: 10.1158/1078-0432.CCR-06-0147. [DOI] [PubMed] [Google Scholar]
- 17.Sowery RD, Hadaschik BA, So AI, Zoubeidi A, Fazli L, Hurtado-Coll A, et al. Clusterin knockdown using the antisense oligonucleotide OGX-011 re-sensitizes docetaxel-refractory prostate cancer PC-3 cells to chemotherapy. BJU International. 2008;102:389–97. doi: 10.1111/j.1464-410X.2008.07618.x. [DOI] [PubMed] [Google Scholar]
- 18.Zhong B, Sallman DA, Gilvary DL, Pernazza D, Sahakian E, Fritz D, et al. Induction of clusterin by AKT-Role in cytoprotection against docetaxel in prostate tumor cells. Mol Cancer Ther. 2010;9:1831–41. doi: 10.1158/1535-7163.MCT-09-0880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kosaka T, Miyajima A, Shirotake S, Suzuki E, Kikuchi E, Oya M. Long-Term Androgen Ablation and Docetaxel Up-Regulate Phosphorylated Akt in Castration Resistant Prostate Cancer. The Journal of Urology. 2011;185:2376–81. doi: 10.1016/j.juro.2011.02.016. [DOI] [PubMed] [Google Scholar]
- 20.Liu C, Zhu Y, Lou W, Nadiminty N, Chen X, Zhou Q, et al. Functional p53 determines docetaxel sensitivity in prostate cancer cells. Prostate. 2013;73:418–27. doi: 10.1002/pros.22583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Domingo-Domenech J, Vidal SJ, Rodriguez-Bravo V, Castillo-Martin M, Quinn SA, Rodriguez-Barrueco R, et al. Suppression of acquired docetaxel resistance in prostate cancer through depletion of notch- and hedgehog-dependent tumor-initiating cells. Cancer Cell. 2012;22:373–88. doi: 10.1016/j.ccr.2012.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vidal Samuel J, Rodriguez-Bravo V, Quinn SA, Rodriguez-Barrueco R, Lujambio A, Williams E, et al. A Targetable GATA2-IGF2 Axis Confers Aggressiveness in Lethal Prostate Cancer. Cancer Cell. 2015;27:223–39. doi: 10.1016/j.ccell.2014.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhu Y, Liu C, Nadiminty N, Lou W, Tummala R, Evans CP, et al. Inhibition of ABCB1 Expression Overcomes Acquired Docetaxel Resistance in Prostate Cancer. Molecular Cancer Therapeutics. 2013;12:1829–36. doi: 10.1158/1535-7163.MCT-13-0208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sharom FJ. ABC multidrug transporters: structure, function and role in chemoresistance. Pharmacogenomics. 2008:9. doi: 10.2217/14622416.9.1.105. [DOI] [PubMed] [Google Scholar]
- 25.O’Neill Amanda JMP, Dowling Catherine, Fan Yue, Mulrane Laoighse, Gallagher William m, O’Connor Darran, O’Connor Robert, Devery Aoife, Corcoran Claire, Rani Sweta, O’Driscoll Lorraine, Fitzpatrick John M, William R, Watson G. Characterisation and manipulation of docetaxel resistant prostate cancer cell lines. Molecular Cancer. 2011:10. doi: 10.1186/1476-4598-10-126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gergely Szakacs JKP, Ludwig Joseph A, Booth-Genthe Catherine, Gottesman Michael M. Targeting multidrug resistance in cancer. Nature reviews of drug discovery. 2006;5:219–34. doi: 10.1038/nrd1984. [DOI] [PubMed] [Google Scholar]
- 27.Catherine Sanchez AM, Contreras Hector R, Mendoza Patricia, Cabezas Juan, Acevedo Cristian, Huidobro Christian, Castellon Enrique A. Chemotherapy sensitivity recovery of prostate cancer cells by functional inhibition and knock down of multidrug resistance proteins. The Prostate. 2011;71:1810–7. doi: 10.1002/pros.21398. [DOI] [PubMed] [Google Scholar]
- 28.Bhangal G, BS, Halford S, MA, MRCP, Wang J, BSc, MRCP, Roylance R, BSc, MRCP, Shah R, MRCP, Waxman J., MD, FRCP, BSc Expression of the multidrug resistance gene in human prostate cancer. Urologic Oncology. 2000;5:118–21. doi: 10.1016/s1078-1439(99)00055-1. [DOI] [PubMed] [Google Scholar]
- 29.Zhu Y, Liu C, Cui Y, Nadiminty N, Lou W, Gao AC. Interleukin-6 induces neuroendocrine differentiation (NED) through suppression of RE-1 silencing transcription factor (REST) The Prostate. 2014;74:1086–94. doi: 10.1002/pros.22819. [DOI] [PubMed] [Google Scholar]
- 30.Zhu Y, Tummala R, Liu C, Nadiminty N, Lou W, Evans CP, et al. RhoGDIα suppresses growth and survival of prostate cancer cells. The Prostate. 2012;72:392–8. doi: 10.1002/pros.21441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu C, Lou W, Zhu Y, Nadiminty N, Schwartz CT, Evans CP, et al. Niclosamide Inhibits Androgen Receptor Variants Expression and Overcomes Enzalutamide Resistance in Castration-Resistant Prostate Cancer. Clinical Cancer Research. 2014;20:3198–210. doi: 10.1158/1078-0432.CCR-13-3296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sharom FJ. ABC multidrug transporters: structure, function and role in chemoresistance. Pharmacogenomics. 2007;9:105–27. doi: 10.2217/14622416.9.1.105. [DOI] [PubMed] [Google Scholar]
- 33.Shi Z, Tiwari AK, Shukla S, Robey RW, Singh S, Kim I-W, et al. Sildenafil Reverses ABCB1- and ABCG2-Mediated Chemotherapeutic Drug Resistance. Cancer Research. 2011;71:3029–41. doi: 10.1158/0008-5472.CAN-10-3820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Domingo-Domenech J, Vidal SJ, Rodriguez-Bravo V, Castillo-Martin M, Quinn SA, Rodriguez-Barrueco R, et al. Suppression of acquired docetaxel resistance in prostate cancer through depletion of notch- and hedgehog-dependent tumor-initiating cells. Cancer Cell. 2012;22:373–88. doi: 10.1016/j.ccr.2012.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kimchi-Sarfaty C, Oh JM, Kim I-W, Sauna ZE, Calcagno AM, Ambudkar SV, et al. A “Silent” Polymorphism in the MDR1 Gene Changes Substrate Specificity. Science. 2007;315:525–8. doi: 10.1126/science.1135308. [DOI] [PubMed] [Google Scholar]
- 36.Sheu M-J, Teng Y-N, Chen Y-Y, Hung C-C. The Functional Influences of Common ABCB1 Genetic Variants on the Inhibition of P-glycoprotein by Antrodia cinnamomea Extracts. PLoS ONE. 2014;9:e89622. doi: 10.1371/journal.pone.0089622. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 37.Caceres G, Robey RW, Sokol L, McGraw KL, Clark J, Lawrence NJ, et al. HG-829 Is a Potent Noncompetitive Inhibitor of the ATP-Binding Cassette Multidrug Resistance Transporter ABCB1. Cancer Research. 2012;72:4204–13. doi: 10.1158/0008-5472.CAN-12-0743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhu ML, Horbinski CM, Garzotto M, Qian DZ, Beer TM, Kyprianou N. Tubulin-targeting chemotherapy impairs androgen receptor activity in prostate cancer. Cancer Res. 2010;70:7992–8002. doi: 10.1158/0008-5472.CAN-10-0585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sanchez C, Mercado A, Contreras HR, Mendoza P, Cabezas J, Acevedo C, et al. Chemotherapy sensitivity recovery of prostate cancer cells by functional inhibition and knock down of multidrug resistance proteins. Prostate. 2011;71:1810–7. doi: 10.1002/pros.21398. [DOI] [PubMed] [Google Scholar]
- 40.Szakács G, Paterson JK, Ludwig JA, Booth-Genthe C, Gottesman M. Targeting multidrug resistance in cancer. Nat Rev Drug Discov. 2006;5:219–34. doi: 10.1038/nrd1984. [DOI] [PubMed] [Google Scholar]
- 41.Malofeeva EV, Domanitskaya N, Gudima M, Hopper-Borge EA. Modulation of the ATPase and transport activities of broad-acting multidrug resistance factor ABCC10 (MRP7) Cancer Res. 2012;72:6457–67. doi: 10.1158/0008-5472.CAN-12-1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Brooks TA, Minderman H, O’Loughlin KL, Pera P, Ojima I, Baer MR, et al. Taxane-based reversal agents modulate drug resistance mediated by P-glycoprotein, multidrug resistance protein, and breast cancer resistance protein. Mol Cancer Ther. 2003;2:1195–205. [PubMed] [Google Scholar]
- 43.Huang X, Chau C, Figg W. Challenges to improved therapeutics for metastatic castrate resistant prostate cancer: from recent successes and failures. Journal of Hematology & Oncology. 2012;5:35. doi: 10.1186/1756-8722-5-35. [DOI] [PMC free article] [PubMed] [Google Scholar]