Abstract
Considerable attention has been paid to the “gateway” pattern of drug use initiation in which individuals progress from tobacco and alcohol use to cannabis and other illicit drugs. The extent to which this sequence reflects a causal impact of licit substance use on illicit substance involvement remains unclear. Clarifying the mechanisms underlying substance use initiation may help inform our understanding of risk for psychopathology, as increasing research is demonstrating associations between initiation patterns and heavier involvement. This study examined patterns of substance use initiation using a discordant twin design. Participants were 3,476 monozygotic twins (37% male) from the Australian Twin Registry who reported on their ages of tobacco, alcohol, and cannabis initiation. Multilevel proportional hazard regression models were employed to (a) estimate within-twin-pair and between-twin-pair contributions to associations between the ages of onset of different drugs; and (b) examine whether the magnitude of effects differed as a function of the order of substance use initiation. Finding significant effects within twin pairs would support the hypothesis that the age of initiation of one substance causally influences the age of initiation of a subsequent substance. Finding significant effects between twin pairs would support the operation of familial influences that explain variation in the ages of initiation of multiple drugs. Within-twin-pair effects for typical patterns were modest. When initiation was atypical, however, larger within-twin-pair effects were observed and causal influences were more strongly implicated. Results support the utility of examining the timing and ordering of substance use initiation within sophisticated, genetically informative designs.
Keywords: Substance use initiation, discordant twins, multilevel modeling
Considerable attention has been paid to the “gateway effect.” This describes a pattern of drug use initiation in which individuals progress from use of tobacco and alcohol to cannabis and other illicit drugs (Degenhardt et al., 2009; Kandel, Yamaguchi, & Chen, 1992). Some studies support a causal impact of licit drug use on illicit drug involvement (Huizink et al., 2010; Kandel et al., 1992), while others indicate that the sequence results from liability to deviant behavior (Tarter, Vanyukov, Kirisci, Reynolds, & Clark, 2006), easier access to legal than illegal drugs, and differing drug prevalence (Degenhardt et al., 2010). Studies attempting to examine causal effects of earlier substance use on later initiation are limited in their ability to control for correlated risk factors that account for variation in the uptake of multiple substances. Genetic factors represent one mechanism; research strongly supports a genetic contribution to externalizing behavior and drug use (e.g., Kendler, Prescott, Myers, & Neale, 2003; Krueger et al., 2002). Aspects of the common environment that contribute to sibling similarity in substance use (for instance, low parental monitoring, parental attitudes toward use, and shared peer influences) may also explain variance in initiation patterns (Baker, Maes, & Kendler, 2012; Walden, McGue, Iacono, Burt, & Elkins, 2004). Genetically informative samples can allow for a higher degree of control over such factors.
Discordant twin modeling represents a powerful method with which to test questions regarding causality. Monozygotic twins reared together share 100% of their genes and 100% of their family environmental influences; however, some experiences are unique to each twin. Focusing on within-twin-pair differences with regard to these experiences – and associating them with within-twin-pair differences in an outcome of interest – can help establish a probable causal pathway between exposure and outcome by controlling for the correlated genetic and family environmental effects that might otherwise explain the association (Vitaro, Brendgen, & Arseneault, 2009). With regard to previous tests of “gateway effects,” this method has been used to demonstrate that early-onset cannabis use may increase risk for other drug use and dependence (Lessem et al., 2006; Lynskey et al., 2003) and nicotine dependence (Agrawal et al., 2008). However, demonstrating a significant association within twin pairs does not “prove” causality; the association is confounded by variables that differ within twins and are related to exposure and outcome. The current analysis concerns whether the age at which an individual starts to use one substance influences the age at which they start to use a subsequent substance; a within-twin-pair difference in age at first use that leads to a within-twin-pair difference in progression indicates the possibility (though not certainty) that the first use influences the outcome.
Discordant twin modeling can be performed using a multilevel approach, in which effects are modeled at the within-twin-pair and between-twin-pair levels (Snijders & Bosker, 1999). The relation between the ages of onset of two substances may be due to the age of onset of the first substance causally influencing that of the subsequent substance (this would be supported by finding a significant association within twin pairs). There also exist family-wide genetic and/or shared environmental effects that explain variation in the uptake of multiple substances (these are indicated by associations detected between twin pairs). Because (a) both processes may be operating and (b) stringent tests of within-twin-pair associations must account for these between-family influences, within- and between-twin-pair effects can be tested simultaneously within multilevel regression models to obtain accurate estimates (Arseneault et al., 2008).
We are aware of only one genetically informed study of the ages of initiation of multiple drugs. In a sample of African-American women, Sartor and colleagues (2009) found that genetic correlations across the ages of cigarette, alcohol, and cannabis initiation ranged from 0.25 to 0.70. These results support the proposition that shared genetic influences contribute to the similarity in the ages of initiation of various substances. These models were not, however, extended to examine the directionality of unique environmental effects across drugs. Evaluating within- and between-twin-pair effects within a survival analytic framework can help determine the extent to which the ages of tobacco, alcohol, and cannabis initiation directly influence each other.
Early first use of substances including tobacco (e.g., Everett et al., 1999), alcohol (e.g., Grant & Dawson, 1997), and cannabis (e.g., Lynskey et al., 2003) is robustly related to risk for later substance use disorder, and recent research supports elevated risk for other measures of psychopathology. For instance, Silins and colleagues (2014) detected robust dose-response relations between adolescent cannabis use and later outcomes including suicide attempt and educational attainment.
Early use of multiple substances is of increasing relevance, as adolescent polysubstance involvement is growing. A recent analysis of the National Longitudinal Study of Adolescent to Adult Health found that 34.1% of adolescents reported using alcohol and marijuana or alcohol, marijuana, and cigarettes prior to age 16, and early uptake of multiple substances exerted an additive effect on risk for later substance use disorder (Moss, Chen, & Yi, 2014). Additionally, use of multiple substances is associated with liability for and speed of transition to problematic use (Behrendt, Wittchen, Höfler, Lieb, & Beesdo, 2009).
With regard to the ordering of onset of multiple substances, research is increasingly demonstrating associations between initiation patterns and heavier involvement. Of particular interest are atypical patterns, in which individuals use illicit before licit substances. (This contrasts with the typical pattern of tobacco and/or alcohol use prior to cannabis and other illicit drug use). For instance, mounting research concerns “reverse gateway” sequences, in which cannabis initiation precedes tobacco initiation. Studies of reverse gateways produce mixed findings. For instance, Patton and colleagues (2005) detected increased risk for tobacco initiation and nicotine dependence among individuals who had used cannabis weekly during adolescence and young adulthood. Agrawal and colleagues (2008) found that risk for transitioning from cigarette initiation to regular smoking and from regular smoking to nicotine dependence was significantly elevated in women who had previously used cannabis, even after controlling for numerous measures of psychopathology. A later analysis, however, found that initiating cannabis use after tobacco use was associated with a greater likelihood of smoking 40 or more cigarettes per day than the reverse sequence, and the reverse gateway sequence was associated with fewer nicotine dependence symptoms (Agrawal et al., 2011). Further investigation of the ordering of substance use onset is needed to clarify the correlates of different onset patterns. Investigating the mechanisms underlying the timing and the sequence of substance use uptake may help inform our understanding of risk for later heavier substance use and problems.
The current study employed a multilevel discordant twin design to examine progression in substance use initiation in a large volunteer twin registry. We had two primary aims. First, we investigated within-twin-pair and between-twin-pair contributions to associations between the ages of onset of different substances of abuse. Second, we explored whether the magnitude of within- and between-twin-pair effects differed as a function of the order in which individuals initiated substances. Specifically, we compared effects across models examining typical and atypical patterns of substance use uptake. Finding significant effects within twin pairs would provide evidence consistent with the hypothesis that the age of initiation of one substance causally influences the age of initiation of a subsequent substance. Finding significant effects between twin pairs would support the operation of familial (genetic and/or shared environmental) influences that explain variation in the ages of initiation of multiple drugs.
Method
Participants
Participants were 3,476 members of the Australian Twin Registry Cohort II (n = 6,265 twins, mean age = 30.0 years (range = 24–36)) and Cohort III (n = 3,824 twins, mean age = 32.1 years (range = 27–40); for more information about participants, see Knopik et al., 2004 and Lynskey et al., 2012). Only monozygotic twins were included in order to provide the strongest control for familial influences and isolate potential within-twin-pair differences. The current sample consisted of 1,738 twin pairs in which both twins reported having used tobacco, alcohol, and/or cannabis – 37% were male and 63% were female.
Procedure
Both cohorts completed interviews based on the Semi-Structured Assessment for the Genetics of Alcoholism (SSAGA-OZ; Bucholz et al., 1994). Cohort II was surveyed by telephone in 1996–2000 (participation rate = 84%; Knopik et al., 2004). Cohort III was surveyed by computer-assisted telephone interview in 2005–2009 (participation rate = 76%; Lynskey et al., 2012).
Retest data were collected 3.7 years (SD = 0.4, range = 1.1–4.3) after the interview for a subsample of twins (n = 216) from Cohort II. Initial and retest interviews were conducted by trained lay-interviewers who were blind to the status of the co-twin. Informed consent was obtained from participants and the study was approved by the Institutional Review Boards at Washington University School of Medicine, the University of Missouri, and the Queensland Institute of Medical Research.
Measures
Lifetime substance use
Lifetime substance use was coded as a binary response to a question regarding ever use of the drug. Lifetime cigarette, alcohol, and cannabis use were examined. Regarding cigarettes, Cohort II was asked, “Have you ever tried cigarettes?” and Cohort III was asked, “Have you ever tried cigarette smoking, even a puff?” Concerning alcohol, Cohort II was queried, “Have you ever had a drink of alcohol?” and Cohort III was queried, “Have you ever had a full drink of alcohol?” Regarding cannabis, Cohort II was asked if they had ever used cannabis when not prescribed or used more than the prescribed dose, or if they had experimented even once with cannabis. Cohort III was asked if they had ever used cannabis. Cannabis was defined as marijuana or hashish. Lifetime abstainers from cigarette, alcohol, and cannabis use were 13.6%, 0.4%, and 39.6% of the combined sample, respectively.
Age of tobacco initiation
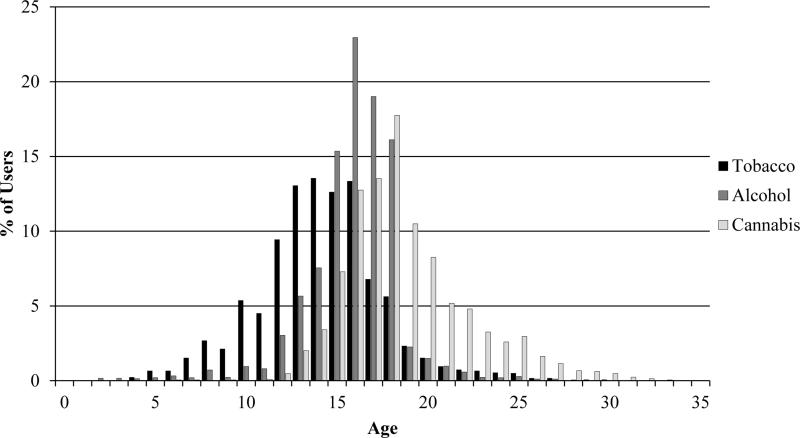
In Cohort II, non-abstainers were asked, “How old were you when you tried your first cigarette?” Cohort III was queried, “How old were you the first time you smoked even a puff of a cigarette?” Figure 1 displays the frequency distributions for reported ages of tobacco, alcohol, and cannabis initiation. Responses for tobacco initiation ranged from 2 to 33 years. To reduce the influence of low-lying values, individuals who reported ages below 5 years were equated to 5 years. Reported mean ages of smoking onset were 14.1 years in Cohort II and 14.5 years in Cohort III.1 In the current combined sample, mean ages of smoking onset for men and women were 13.7 years (SD = 3.5) and 14.5 years (SD = 3.4), respectively. Retest data demonstrated good reliability in respondent recall (r(74) = .70, p < .0001).2 Mean ages reported by the retest sample at times 1 and 2 did not differ significantly (t(73) = −.49, p = .62). Mean age at first smoking for the retest sample was 13.4 years (SD = 3.3) at time 1 and 13.8 years (SD = 3.6) at time 2. Potential age-related bias was examined by correlating individuals' ages with their reported ages of cigarette initiation. The correlation of r = −.01 suggested minimal bias.
Figure 1.
Frequency distributions for the ages of tobacco, alcohol, and cannabis initiation.
Age of alcohol initiation
In Cohort II, non-abstainers were asked, “How old were you the first time you had more than just a sip of beer, wine or spirits?” Cohort III was asked, “How old were you the first time you had a full drink of beer, wine or spirits?” Responses ranged from 1 to 28 years (see Figure 1). To reduce the influence of low-lying values, individuals reporting ages below 5 years were equated to 5 years. Reported mean ages of drinking onset were 15.9 years in Cohort II and 16.0 years in Cohort III. In the current combined sample, mean ages of drinking initiation for men and women were 15.4 years (SD = 2.6) and 16.2 years (SD = 2.5), respectively. These data demonstrated excellent retest reliability in respondent recall (r(80) = .86, p < .0001), and the mean ages reported at each time point did not significantly differ (t(79) = −1.39, p = .17). Mean age at first drink for the retest sample was 15.1 years (SD = 3.3) at time 1 and 15.4 years (SD = 3.2) at time 2. Age-related bias was minimal (r = .02).
Age of cannabis initiation
Non-abstainers in both cohorts were queried, “How old were you the first time you used cannabis?” Responses ranged from 6 to 33 years (Figure 1). Reported mean ages of cannabis initiation were 19.1 years in Cohort II and 18.0 years in Cohort III. In the current sample, mean ages of first cannabis use were 18.6 years (SD = 3.4) for men and 18.8 years (SD = 3.5) for women. Retest data indicated good reliability in respondent recall (r(60) = .78, p < .0001). Mean ages of cannabis onset reported at times 1 and 2 did not significantly differ (t(59) = −1.85, p = .07). Mean age of cannabis initiation was 18.2 years (SD = 2.5) at time 1 and 19.0 years (SD = 4.0) at time 2. The correlation between age and reported age at first cannabis use (r = .08) was slightly higher than for the ages of tobacco and alcohol initiation, but still small, suggesting limited age-related bias.
Initiation Versus Experimentation
We investigated the degree of experimentation in the sample (i.e., the percentage of individuals who progressed beyond first substance use). Because of slight differences in the assessment of substance use across cohorts, we only evaluated experimentation in Cohort II. In Cohort II, 68.2% of lifetime tobacco users reported having smoked cigarettes more than “one or two times ‘just to try.’” 92.3% of lifetime alcohol users reported having become regular drinkers (drinking at least once a month for six months or more) and/or having gotten drunk after their first experience with alcohol. 83.3% of lifetime cannabis users reported having used cannabis three or more times. Therefore, it appears that for the majority of participants, age at first use was not simply experimentation. Results should, however, be interpreted in the context of higher rates of experimentation for tobacco compared with alcohol and cannabis.
Statistical Analysis
A series of multi-level frailty models were constructed using PROC PHREG in SAS 9.3. A frailty model is a random effects model for survival data that estimates a Cox proportional hazard regression that accounts for a correlated data structure (Hougaard, 2014). Frailty models are constructed similarly to standard proportional hazard regression models, with the inclusion of a random intercept term specifying the clustering variable. In the current data, twins were nested within families; therefore, a random intercept (the family number for each twin pair) was included to obtain unbiased parameter estimates. The “time to event” variable was an individual's age of substance use initiation. Abstainers were included in analyses (see below); they were treated as censored at interview, with their age at interview modeled as time.
The first set of proportional hazard models analyzed typical progression patterns (tobacco initiation before alcohol initiation, tobacco initiation before cannabis initiation, and alcohol initiation before cannabis initiation; see Table 2). The second set evaluated atypical progression patterns (alcohol initiation before tobacco initiation, cannabis initiation before tobacco initiation, and cannabis initiation before alcohol initiation; Table 2). For a twin pair to be included in a model, both twins had to report having used the first substance, and at least one twin had to report having used the second substance in the requisite pattern (typical or atypical). Co-twins who had not used the second substance or had used it in the opposite pattern were considered censored (see Table S3 in supplemental materials for more information about these censored cases). Baseline models included within-twin-pair (WTP) and between-twin-pair (BTP) indicators of substance use onset, sex, and age predicting the age of onset of the subsequent substance. Within- and between-twin-pair predictors were group mean centered (WTP: individual onset minus the twin pair average onset; BTP: twin pair average onset minus the average onset of all other twins). Data were structured in “long format,” such that each member of a pair contributed data at the within- and between-twin-pair levels.
Table 2.
Prevalence (%) of Different Substance Use Initiation Patterns
| Onset Pair | Typical | Atypical | Same Age |
|---|---|---|---|
| Tobacco and Alcohol (n = 2,984) | 61.6 | 22.7 | 15.7 |
| Tobacco and Cannabis (n = 2,023) | 88.4 | 3.9 | 7.7 |
| Alcohol and Cannabis (n = 2,083) | 81.7 | 6.1 | 12.2 |
Notes. Prevalences calculated among individuals who reported having used both substances. Typical sequencing patterns are: tobacco before alcohol, tobacco before cannabis, and alcohol before cannabis.
Subsequently, we aimed to consider progression between each pair of substances in the context of use of the third substance. That is, we tested whether within- and between-twin-pair associations between the ages of initiation of each pair of substances differed depending on whether an individual had used a substance that was not modeled in each baseline analysis. Therefore, the second series of models controlled for lifetime use of the third substance (e.g., within- and between-twin-pair effects for lifetime cannabis use were included in models predicting age of alcohol initiation from age of tobacco initiation). Within the current models, a hazard ratio of 1.0 for WTP and BTP effects indicates no effect of the predictor on the outcome. Hazard ratios closer to zero designate a stronger effect of the predictor.
7.7%–15.7% of the sample reported having first tried each pair of substances at the same age. Because age of initiation was recorded on a yearly scale, it is possible that individuals coded as “same aged” users first used one substance before the other (e.g., in a different month or day), but the assessment did not capture this difference. To avoid misclassifying such “ambiguous cases,” individuals reporting initiating two substances at the same age were included in models evaluating both typical and atypical progression patterns. Results were comparable to those from models excluding “same aged” users (not shown).
Proportional hazards models assume that risk remains constant over time (that a constant relative hazard is observed across individuals). This assumption was tested by examining interactions between predictors and the log of the time-to-event variable. To remain conservative in our estimates of violations, an alpha level of .10 was used to determine significance. To adjust for violations, all significant interactions were entered into the models. Results of tests for proportional hazard violations are presented in Tables S1-S2 in supplemental materials.
Results
Descriptive Analyses
Table 1 displays the discordance between twins for their ages of tobacco, alcohol, and cannabis onset. Ages of onset for all substances were moderately correlated (rs = 0.23–0.29). Discordance was treated as a continuous variable representing the difference in years of the age of onset of the first twin minus the age of onset of the second twin; twins whose ages of onset differed by one or more years were considered discordant. Most pairs were discordant: 1,177 pairs (83% of users), 1,243 pairs (72% of users), and 662 pairs (79% of users) were discordant for their ages of tobacco, alcohol, and cannabis initiation, respectively. Table 2 displays the prevalences of individuals adopting typical and atypical patterns and using substances at the same age. Atypical sequencing was more common with regard to licit substances (e.g., first use of alcohol before tobacco [22.7%]) than illicit drugs (e.g., first trying cannabis before tobacco [3.9%] or alcohol [6.1%]).3 In the current sample, use of tobacco before alcohol was more prevalent than the opposite pattern and therefore classified as a typical initiation sequence. However, this is not universal; other studies find that alcohol use is more or equally likely to precede tobacco use as the reverse sequence (e.g., Kandel, 1975; White, Jarrett, Valencia, Loeber, & Wei, 2007).
Table 1.
Twin Discordance for Age of Substance Use Initiation
| Mean (standard deviation) |
|||
|---|---|---|---|
| Full Sample | Men | Women | |
| Age of tobacco initiation | 2.5 (2.4) | 2.8 (2.6) | 2.3 (2.3) |
| Age of alcohol initiation | 1.7 (2.0) | 1.8 (1.9) | 1.6 (2.0) |
| Age of cannabis initiation | 2.2 (2.3) | 2.1 (2.1) | 2.3 (2.4) |
Notes. Twin discordance for tobacco, alcohol, and cannabis initiation indicates the average absolute value of the difference in co-twins' reported ages of initiation for each substance.
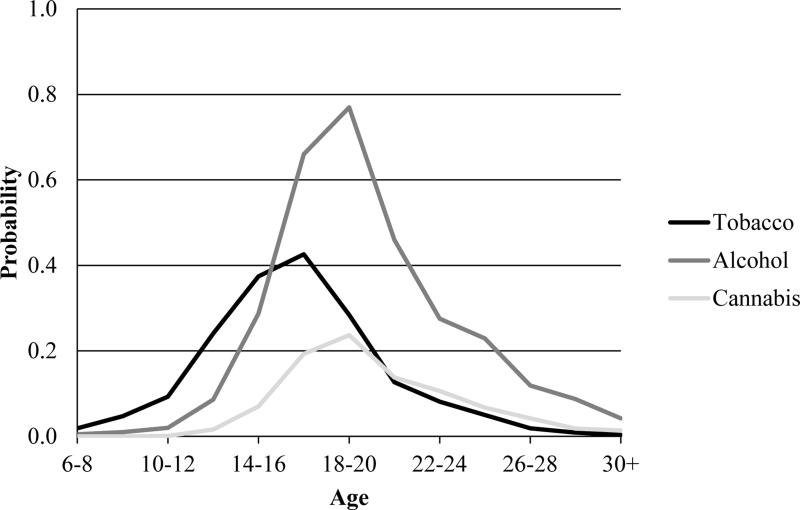
Figure 2 depicts the hazards for initiating tobacco, alcohol, and cannabis use at different ages for the sample. Peak periods of risk for initiation were between ages 16 and 18 for tobacco and between ages 18 and 20 for alcohol and cannabis. This is not unexpected given that when participants were adolescents and young adults, the legal ages to use tobacco and alcohol in Australia were 16 and 18, respectively.
Figure 2.
Hazard functions for initiation of tobacco, alcohol, and cannabis use.
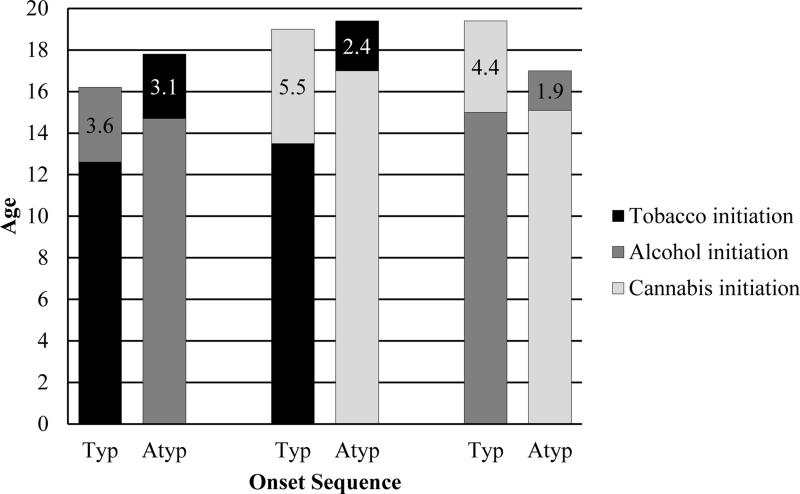
Figure 3 displays the differences between the mean ages of onset of tobacco, alcohol, and cannabis use for typical and atypical patterns. Across all pairs of substances, individuals who adopted an atypical sequence started using their first substance later than respondents who followed a typical sequence. For instance, among individuals who tried tobacco and cannabis, mean age of onset of the first drug was 13.5 years for typical progression and 17.0 years for atypical progression. Although respondents adopting uncharacteristic patterns waited longer to try their first substance, they progressed more rapidly to use of their subsequent drug. Among individuals who tried tobacco and cannabis, the mean difference in ages of onset of both substances was 5.5 years and 2.4 years for typical and atypical sequencing, respectively. This “telescoped” pattern of initiation was evident across all pairs of substances; however, it was stronger for cannabis than licit substances. Among respondents who tried tobacco and alcohol, the mean difference in the ages of onset of both substances was 3.6 years for typical progression and 3.1 years for atypical progression.
Figure 3.
Differences between mean ages of onset of tobacco, alcohol, and cannabis use for typical and atypical sequencing patterns. Typ = typical, Atyp = atypical. Typical sequencing patterns are: tobacco before alcohol, tobacco before cannabis, and alcohol before cannabis. Data labels indicate the difference between the mean ages of onset for the first and second substance in the sequence.
Frailty Models: Typical Progression
Age of tobacco initiation predicting age of alcohol initiation
Results from baseline models evaluating typical progression patterns are presented in Table 3. In the model for age of tobacco initiation predicting age of alcohol initiation, the within-twin-pair and between-twin-pair effects were significant (WTP: HR = .82, 95% confidence interval (CI) [.80, .84]; BTP: HR = .34, 95% CI [.25, .46]). The within-twin-pair effect of .82 indicated a potentially causal relation between age of tobacco onset and age of alcohol onset, such that for every year that an individual started to smoke cigarettes earlier than their co-twin, their risk of subsequently trying alcohol increased by 18%. The between-twin-pair effect of .34 indicated that there was also a familial contribution to the association between the ages of tobacco and alcohol initiation, such that for every year that a twin pair's average age of smoking onset was earlier than the age of onset among all other twins, their risk of subsequently starting to drink increased by 66%. Both the within- and between-twin-pair effects remained significant after including within- and between-twin-pair indicators of lifetime cannabis use in the model (WTP: HR = .82, 95% CI [.80, .84]; BTP: HR = .28, 95% CI [.20, .38], not tabled (results available upon request)).
Table 3.
Typical and Atypical Sequencing Pattern Results from Baseline Multilevel Proportional Hazard Regression Models
| Typical Sequencing |
Atypical Sequencing |
||
|---|---|---|---|
| HR (95% CI) |
HR (95% CI) |
||
| Tobacco Initiation → Alcohol Initiation (n = 2,558) |
Alcohol Initiation → Tobacco Initiation (n = 1,722) |
||
| Age | 1.00 (.98, 1.02) | Age | 1.00 (.97, 1.02) |
| Sex | .97 (.89, 1.06) | Sex | 1.05 (.93, 1.19) |
| WTP age of tobacco initiation | .82 (.80, .84) | WTP age of alcohol initiation | .74 (.71, .77) |
| BTP age of tobacco initiation | .34 (.25, .46) | BTP age of alcohol initiation | .87 (.85, .90) |
|
Tobacco Initiation → Cannabis Initiation (n = 2,224) |
Cannabis Initiation → Tobacco Initiation (n = 338) |
||
| Age | .99 (.97, 1.01) | Age | .98 (.92, 1.04) |
| Sex | 1.00 (.91, 1.10) | Sex | .88 (.66, 1.17) |
| WTP age of tobacco initiation | .93 (.90, .95) | WTP age of cannabis initiation | .07 (.02, .28) |
| BTP age of tobacco initiation | .52 (.40, .68) | BTP age of cannabis initiation | .01 (.004, .03) |
|
Alcohol Initiation → Cannabis Initiation (n = 2,410) |
Cannabis Initiation → Alcohol Initiation (n = 552) |
||
| Age | .99 (.98, 1.01) | Age | 1.04 (.99, 1.08) |
| Sex | .96 (.88, 1.06) | Sex | .004 (.00, .96) |
| WTP age of alcohol initiation | .89 (.86, .91) | WTP age of cannabis initiation | .58 (.53, .63) |
| BTP age of alcohol initiation | .55 (.39, .77) | BTP age of cannabis initiation | .002 (.001, .006) |
Notes. HR = hazard ratio, CI = confidence interval, WTP = within-twin-pair, BTP = between-twin-pair. Each model included individuals from twin pairs in which at least one twin followed the designated initiation sequence and the co-twin had used the first substance. Individuals who reported using both substances at the same age were included in models for both typical and atypical sequencing. When a more conservative alpha level of .01 was used for calculation of confidence limits, the only effect that did not remain significant was the between-twin-pair effect for the age of cannabis initiation predicting the age of alcohol initiation.
Age of tobacco initiation predicting age of cannabis initiation
Significant within- and between-twin-pair effects of the age of tobacco initiation on the age of cannabis initiation were detected (WTP: HR = .93, 95% CI [.90, .95]; BTP: HR = .52, 95% CI [.40, .68]). Because nearly everyone in this group had used alcohol, minimal changes in estimates were observed after controlling for lifetime alcohol use (WTP: HR = .93, 95% CI [.90, .95]; BTP: HR = .55, 95% CI [.42, .72]).
Age of alcohol initiation predicting age of cannabis initiation
Age of alcohol initiation exerted significant within- and between-twin-pair effects on the age of cannabis initiation (WTP: HR = .89, 95% CI [.86, .91]; BTP: HR = .55, 95% CI [.39, .77]), and these effects remained significant after controlling for lifetime tobacco use (WTP: HR = .89, 95% CI [.86, .91]; BTP: HR = .91, 95% CI [.89, .93]).
Frailty Models: Atypical Progression
Age of alcohol initiation predicting age of tobacco initiation
Results from baseline models evaluating atypical progression patterns are displayed in Table 3. There were significant within-twin-pair and between-twin-pair effects of the age of alcohol initiation on the age of tobacco initiation (WTP: HR = .74, 95% CI [.71, .77]; BTP: HR = .87, 95% CI [.85, .90]). The within-twin-pair effect was larger than that obtained when examining the influence of the age of tobacco initiation on the age of alcohol initiation, and the confidence limits did not overlap across models.4 The within-twin-pair effect demonstrated an 8% increase in the hazard of initiation; this indicates a stronger potentially causal effect of the age of onset of the first drug on the age of onset of the subsequent drug when progression follows an atypical pattern. The within- and between-twin-pair parameters remained significant after controlling for lifetime cannabis use (WTP: HR = .74, 95% CI [.72, .77]; BTP: HR = .88; 95% CI [.86, .91]).
Age of cannabis initiation predicting age of tobacco initiation
Significant within- and between-twin-pair effects of the age of cannabis initiation on the age of tobacco initiation were observed (WTP: HR = .07, 95% CI [.02, .28]; BTP: HR = .01, 95% CI [.004, .03]). Both the within- and between-twin-pair effects were notably larger than those observed when examining the influence of tobacco onset on cannabis onset, with non-overlapping confidence intervals across models. Because everyone in this group had tried alcohol in their lifetime, controlling for lifetime alcohol use did not change the within- or between-twin-pair estimates or associated confidence limits.
Age of cannabis initiation predicting age of alcohol initiation
Significant within- and between-twin-pair effects of the age of cannabis initiation on the age of alcohol initiation were observed (WTP: HR = .58, 95% CI [.53, .63]; BTP: HR = .002, 95% CI [.001, .006]).5 Both effects were larger than those obtained when evaluating the influence of alcohol onset on cannabis onset, with non-overlapping confidence limits across models. The within- and between-twin-pair effects remained largely unchanged after controlling for lifetime tobacco use (WTP: HR = .57; 95% CI [.53, .62]; BTP: HR = .002, 95% CI [.001, .007]).
Discussion
This study employed a sample of monozygotic twins who reported on their ages of initiation of tobacco, alcohol, and cannabis use. Using a multilevel discordant twin approach, we investigated within-twin-pair and between-twin-pair contributions to associations between the ages of onset of these three substances of abuse. As a secondary aim, we evaluated whether the magnitude of within- and between-twin-pair effects differed as a function of whether individuals initiated substance use in a typical or atypical pattern.
Typical Versus Atypical Progression
When examining typical progression, significant but modest within-twin-pair effects were observed for all models. When evaluating atypical progression, however, much larger within-twin-pair effects were obtained. Notably, this pattern persisted across all models after controlling for lifetime use of the third substance. It was most evident for models evaluating cannabis onset as a predictor of tobacco and alcohol onset. Baseline models for atypical progression of tobacco and cannabis initiation demonstrated an 86% increase and for cannabis and alcohol initiation a 31% increase in the within-twin-pair hazard risk compared to typical progression models of these substances. This suggests that the age at which an individual tries a substance may exert a greater causal effect on the age at which they try their subsequent substance when initiation follows an uncharacteristic pattern. This phenomenon may be strongest with regard to use of cannabis before licit substances.
Several factors might contribute to stronger potentially causal associations for atypical progression. First, experimenting with one's initial drug in an uncharacteristic fashion may lead to reduced perceived barriers to trying additional drugs. This is particularly relevant to use of cannabis prior to other substances; very few individuals who try cannabis are subjected to legal sanctions (Hall, 2001), and seemingly successful first experiences with cannabis may reduce the perceived risk of trying legal substances. A related mechanism is that of access: if individuals are able to experiment with illicit drugs, licit drugs are likely easily available. Third, pharmacological processes might mediate causal associations. Common receptor sensitization following drug exposure may increase sensitivity to the effects of subsequent drugs (Tanda, Pontieri, & DiChiara, 1997). It is unclear, however, whether the degree of exposure inherent in simply trying a drug is sufficient to induce neuro-pharmacological sensitization.
With the exception of models examining the ages of tobacco and alcohol initiation, the between-twin-pair effects were larger for atypical compared with typical progression. This, in combination with the larger within-twin-pair effects obtained for atypical progression, suggests a stronger overall association between the ages of onset of the first and second substance when initiation follows an uncharacteristic sequence. The pattern of progressively stronger within- and between-twin-pair effects for atypical models is reflected in the faster rates of transition between substances for atypical compared with typical progression. The greatest change in hazard ratios for within- and between-twin-pair parameters was observed for initiation of tobacco and cannabis use, followed by alcohol and cannabis use and then tobacco and alcohol use. Similarly, among atypical progression models, the difference between mean ages of initiation was 3.1 years shorter for tobacco and cannabis, 2.5 years shorter for alcohol and cannabis, and 0.5 years shorter for tobacco and alcohol.
Few behavioral genetic studies have examined the age of substance use initiation; however, it is interesting to compare our results for typical progression to those regarding the genetic and environmental overlap between the ages of tobacco, alcohol, and cannabis initiation. Sartor and colleagues (2009) detected the highest degree of genetic covariation between the ages of cigarette and alcohol onset (rG = .70), with lower correlations for alcohol and cannabis (rG = .49) and cigarettes and cannabis (rG = .25). Similarly, we observed the largest between-twin-pair effect for the ages of tobacco and alcohol onset (HR = .34). The effect sizes for alcohol and cannabis (HR = .55) and tobacco and cannabis (HR = .52) were smaller, but comparable.6 The importance of familial factors in influencing typical and atypical progression is also consistent with previous findings that the family environment explains a notable proportion of the variance in substance use initiation (Dick, Prescott, & McGue, 2009). It will be informative for future studies to examine initiation using approaches that are sensitive to progression. For instance, recent methods enable simultaneous modeling of the genetic and environmental liabilities to initiation and progression of substance use (causal-common contingent models; Fowler et al., 2007; Neale, Harvey, Maes, Sullivan, & Kendler, 2006) and determination of genetic and environmental contributions to inter- and intra-individual changes in substance involvement (biometric latent growth curve models; Neale & McArdle, 2000).
Relationship to Disorder
Current results support the assertion that the frequently observed “gateway sequence” is largely a function of correlated risk factors that account for variation in the uptake of multiple drugs. “Violations” of this sequence, by contrast, are associated with more rapid progression between substances that may be more likely to reflect causal processes. We emphasize that although this analysis can inform studies of substance use progression, it cannot speak directly to how initiation patterns may relate to risk for disorder. Existing studies regarding this question produce inconsistent results. Some cite a causal impact of gateway violations on later polydrug involvement and dependence (Ginzler et al., 2003; Mackesy-Amiti et al., 1997), while others note that such associations are attributable to confounding variables (Degenhardt et al., 2009; Wells & McGee, 2008). The prognostic significance of atypical patterns is of increasing relevance, as they are likely to become more common in the US. Cannabis use is growing due to increased social approval and legalization, while tobacco involvement has been controlled by social sanctions and legislation (Johnston et al., 2013; Palamar, Ompad, & Petkova, 2014). Indeed, such changes may lead currently atypical patterns to become normative. Future research incorporating measures of disorder into models of substance use progression will help clarify the nature of associations between gateway violations and later problems.
Limitations
This study has limitations. The first concerns generalizability, as this sample is largely Caucasian and the cultural context of substance use in Australia is different from that in other countries. The current sample has a very low abstinence rate from alcohol. Additionally, in Australia, cannabis-containing joints often include tobacco (Bélanger, Akre, Kuntsche, Gmel, & Suris, 2011), which may explain some effects of cannabis initiation on tobacco initiation. Cannabis and tobacco also have a common route of administration, and some data suggest the increased likelihood of cannabis use observed in tobacco users is limited to individuals who use smoked rather than smokeless forms of tobacco (Agrawal & Lynskey, 2009). We also reiterate that using tobacco prior to alcohol was more common in the current sample than the reverse sequence, and was therefore classified as a typical pattern. However, this finding may not generalize to other groups.
Mixing tobacco with cannabis may also limit analyses of progression in that individuals who try cannabis before cigarettes will be exposed to tobacco; therefore, subsequent cigarette use will not capture first tobacco use. Individuals who tried cannabis before cigarettes represented only 3.9% of the current sample; nevertheless, studies of progression employing samples that do not co-use cannabis with tobacco will be important to examine the generalizability of the current findings.
It is also unclear to what extent the effect sizes obtained in the current study may generalize to unrelated individuals. There is evidence that siblings influence each other's substance involvement, particularly during adolescence (Rende, Slomkowski, Lloyd-Richardson, & Niaura, 2005). To our knowledge, no studies have yet examined sibling effects on the age of substance use initiation; however, such imitation is plausible given that initiation often occurs in adolescence, when levels of sibling contact are higher than in later life. Imitative dyadic sibling interaction may increase heritability estimates in genetically-informed designs (Carey, 1992). The present analytic framework did not allow for adjustment for reciprocal twin influence for the age of substance use initiation. Therefore, it is possible that the current within-twin-pair and between-twin-pair effects were under- and over-estimates, respectively.
Cohort II and Cohort III were interviewed approximately 10 years apart; this might lead to differences in substance involvement across cohorts that reflect a changing social milieu. Participant age was included as a covariate in analyses; however, we also re-ran models controlling for cohort membership. Minimal differences in results were observed. Our assessment of age of onset asked about the year in which individuals first used a substance. An individual who reported having tried two substances within the same year could therefore have been recorded as having tried both at the same age. To determine the extent to which inclusion of “same aged” users influenced results, baseline models were re-run excluding these cases. Weaker between-twin-pair effects were observed for models of typical sequencing; however, the pattern of results across typical and atypical models was the same as that obtained from analyses excluding these cases, suggesting that findings were not notably affected by the operationalization of age of onset. Assessment of age of onset on a more fine-grained (e.g., monthly or daily) scale, however, would provide more detailed information. This approach is most difficult to accomplish via retrospective studies, but more easily employed within longitudinal designs. Prospective studies can also help to minimize the bias in retrospective reports on age of substance use initiation, for which people have been shown to report progressively later ages of onset over time (Parra, O'Neill, & Sher, 2003). Although this study's participants were interviewed in adulthood, correlations between age at interview and reported ages of onset were minimal (rs = .01-.08), suggesting limited age-related bias. Nevertheless, accuracy in reporting may be increased via prospective assessment.
We included twin pairs who were discordant for initiation sequence in analyses and modeled the twin discordant for the requisite sequence as censored. This was done to avoid losing valuable co-twin data and preserve statistical power, particularly in analyses of the less prevalent atypical patterns. We considered co-twins censored in order to limit bias; however, analyses of twin pairs concordant for sequence may produce more robust results. Larger sample sizes will be necessary to accomplish this aim.
Finally, strong causal conclusions cannot be drawn solely from this study. Future research employing longitudinal designs and incorporating measures of unique environmental influences that relate to early-onset substance use would help to (a) clarify the mechanisms underlying associations between the ages of onset of different drugs; and (b) determine whether putatively causal mechanisms are explained by third variables.
Conclusions
Notwithstanding limitations, the current study offers an advance in our understanding of progression in substance use initiation. Results highlight the importance of considering both the timing and the ordering of substance use onset, and demonstrate that when individuals adopt an atypical sequence of progression, their ages of onset of prior substances may exert a stronger causal effect on their ages of initiation of subsequent substances. Continued investigation of substance use phenotypes within sophisticated, genetically informed designs will allow for rigorous tests of causal hypotheses regarding progression in substance involvement.
Supplementary Material
Lay summary.
This study suggests that individuals’ ages of onset of prior drugs more strongly influence their ages of onset of subsequent drugs when substance use initiation follows an atypical rather than a typical pattern of progression.
Acknowledgments
This work was supported by National Institute on Alcohol Abuse and Alcoholism grants AA023419 (LR), AA023133 (AD), AA21235 (AA), and AA007728 (AH), and National Institute on Drug Abuse grants DA18267 (ML) and DA23668 (AA). We thank Bronwyn Morris, Megan Fergusson, David Smyth, Olivia Zheng, Harry Beeby, Anjali Henders, Dixie Statham, Richard Parker, Soad Hancock, Judith Moir, Sally Rodda, Pieta-Maree Shertock, Heather Park, Jill Wood, Pam Barton, Fran Husband, and Adele Somerville for data collection and data management. We thank the Australian Twin Registry twins for their continued participation. Gratitude is also expressed to Lesa Hoffman for providing consultation regarding the analytic approach.
Footnotes
There are no conflicts of interest to declare.
The small difference in age of smoking onset across cohorts is not likely to be due to the difference in wording of the question. Cohort III's operationalization of first smoking was more liberal (“even a puff of a cigarette”) than Cohort II's definition (“your first cigarette”). If wording had significantly influenced results, an earlier age of onset would be expected for Cohort III than Cohort II; however, the opposite pattern of results was observed.
For all onset variables, separate test-retest correlations were calculated for men and women and pooled via a Fisher's z-transformation to compute the overall correlation coefficient.
Twins exhibited much higher concordance rates for typical than atypical initiation patterns. For typical progression, concordance rates were 45.9% (ages of tobacco and alcohol initiation), 79.7% (ages of tobacco and cannabis initiation), and 66.7% (ages of alcohol and cannabis initiation). For atypical progression, concordance rates were 8.4% (ages of alcohol and tobacco initiation), 0.5% (ages of cannabis and tobacco initiation), and 1.4% (ages of cannabis and alcohol initiation). We also investigated twin concordance for use of all three substances in a typical (tobacco before alcohol before cannabis) versus an atypical pattern. Of the 804 twin pairs who reported having used all three substances, 25.6% were concordant for typical sequencing. By contrast, no twin pairs were concordant for the atypical sequence of cannabis before alcohol before tobacco.
Within the current analytic framework, a direct statistical comparison of within- and between-twin-pair effect sizes across typical and atypical models was not possible. However, we are reasonably confident that the effects were significantly different given that confidence intervals did not overlap across any models (and were well separated, with the exception of the within-twin-pair parameter for the ages of tobacco and alcohol initiation). We acknowledge, however, that this provides a less stringent gauge of significance than a statistical test.
We re-ran all baseline analyses adopting a more conservative alpha level of .01 for calculation of confidence limits. The only effect that did not remain significant was the between-twin-pair effect for the age of cannabis initiation predicting the age of alcohol initiation.
Typical progression models represent the most appropriate comparison to Sartor and colleagues’ (2009) findings as the majority of participants adopt a typical pattern, thus contributing more strongly to findings regarding genetic and environmental overlap.
References
- Agrawal A, Lynskey MT. Tobacco and cannabis co-occurrence: Does route of administration matter? Drug and Alcohol Dependence. 2009;99:240–247. doi: 10.1016/j.drugalcdep.2008.08.007. doi:10.1016/j.drugalcdep.2008.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agrawal A, Lynskey MT, Pergadia ML, Bucholz KK, Heath AC, Martin NG, Madden PAF. Early cannabis use and DSM-IV nicotine dependence: A twin study. Addiction. 2008;103:1896–1904. doi: 10.1111/j.1360-0443.2008.02354.x. doi:10.1111/j.1360-0443.2008.02354.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agrawal A, Madden PAF, Bucholz KK, Heath AC, Lynskey MT. Transitions to regular smoking and to nicotine dependence in women using cannabis. Drug and Alcohol Dependence. 2008;95:107–114. doi: 10.1016/j.drugalcdep.2007.12.017. doi:10.1016/j.drugalcdep.2007.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agrawal A, Scherrer JF, Lynskey MT, Sartor CE, Grant JD, Haber JR, Xian H. Patterns of use, sequence of onsets and correlates of tobacco and cannabis. Addictive Behaviors. 2011;36:1141–1147. doi: 10.1016/j.addbeh.2011.07.005. doi:10.1016/j.addbeh.2011.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arseneault L, Milne BJ, Taylor A, Adams F, Delgado K, Caspi A, Moffitt TE. Being bullied as an environmentally mediated contributing factor to children's internalizing problems. Archives of Pediatric and Adolescent Medicine. 2008;162:145–150. doi: 10.1001/archpediatrics.2007.53. doi:10.1001/archpediatrics.2007.53. [DOI] [PubMed] [Google Scholar]
- Baker JH, Maes HH, Kendler KS. Shared environmental contributions to substance use. Behavior Genetics. 2012;42:345–353. doi: 10.1007/s10519-011-9516-8. doi:10.1007/S20519-011-9516-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behrendt S, Wittchen H-U, Höfler M, Lieb R, Beesdo K. Transitions from first substance use to substance use disorders in adolescence: Is early onset associated with a rapid escalation? Drug and Alcohol Dependence. 2009;99:68–78. doi: 10.1016/j.drugalcdep.2008.06.014. doi:10.1016/j.drugalcdep.2008.06.014. [DOI] [PubMed] [Google Scholar]
- Bélanger RE, Akre C, Kuntsche E, Gmel G, Surie JC. Adding tobacco to cannabis – its frequency and likely implications. Nicotine & Tobacco Research. 2011;13:746–750. doi: 10.1093/ntr/ntr043. doi:10.1093/ntr/ntr043. [DOI] [PubMed] [Google Scholar]
- Bucholz KK, Cadoret R, Cloninger CR, Dinwiddie SH, Hesselbrock VM, Nurnberger JL, Schuckit MA. A new, semi-structured psychiatric interview for use in genetic linkage studies: A report on the reliability of the SSAGA. Journal of Studies on Alcohol and Drugs. 1994;55:149–158. doi: 10.15288/jsa.1994.55.149. [DOI] [PubMed] [Google Scholar]
- Carey G. Twin imitation for antisocial behavior: Implications for genetic and family environment research. Journal of Abnormal Psychology. 1992;101:18–25. doi: 10.1037//0021-843x.101.1.18. doi:10.1037/0021-843X.101.1.18. [DOI] [PubMed] [Google Scholar]
- Degenhardt L, Chiu WT, Conway K, Dierker L, Glantz M, Kalaydjian A, Kessler RC. Does the ‘gateway’ matter? Associations between the order of drug use initiation and the development of drug dependence in the National Comorbidity Study Replication. Psychological Medicine. 2009;39:157–167. doi: 10.1017/S0033291708003425. doi:10.1017/S0033291708003425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Degenhardt L, Dierker L, Chiu WT, Medina-Mora ME, Neumark Y, Sampson N, Kessler RC. Evaluating the drug use “gateway” theory using cross-national data: Consistency and associations of the order of initiation of drug use among participants in the WHO World Mental Health Surveys. Drug and Alcohol Dependence. 2010;108:84–97. doi: 10.1016/j.drugalcdep.2009.12.001. doi:10.1016/j.drugalcdep.2009.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dick DM, Prescott C, McGue M. The genetics of substance use and substance use disorders. In: Kim Y-K, editor. Handbook of behavior genetics. 1st ed. Springer; New York, NY: 2009. pp. 443–453. doi:10.1007/978-0-387-76727-7_29. [Google Scholar]
- Everett SA, Warren CW, Sharp D, Kann L, Husten CG, Crossett LS. Initiation of cigarette smoking and subsequent smoking behavior among U.S. high school students. Preventive Medicine. 1999;29:327–333. doi: 10.1006/pmed.1999.0560. doi:10.1006/pmed.1999.0560. [DOI] [PubMed] [Google Scholar]
- Fowler T, Lifford K, Shelton K, Rice R, Thapar A, Neale MC, van den Bree MBM. Exploring the relationship between genetic and environmental influences on initiation and progression of substance use. Addiction. 2007;101:413–422. doi: 10.1111/j.1360-0443.2006.01694.x. doi:10.1111/j.1360-0443.2006.01694.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ginzler J, Cochran B, Domenech-Rodriguez M, Cauce A, Whitbeck L. Sequential progression of substance use among homeless youth: An empirical investigation of the gateway theory. Substance Use and Misuse. 2003;38:725–758. doi: 10.1081/ja-120017391. [DOI] [PubMed] [Google Scholar]
- Grant BF, Dawson DA. Age at onset of alcohol use and its association with DSM-IV alcohol abuse and dependence: Results from the national longitudinal alcohol epidemiologic survey. Journal of Substance Abuse. 1997;9:103–110. doi: 10.1016/s0899-3289(97)90009-2. doi:10.1016/S0899-3289(97)90009-2. [DOI] [PubMed] [Google Scholar]
- Hall W. Reducing the harms caused by cannabis use: The policy debate in Australia. Drug and Alcohol Dependence. 2001;62:163–174. doi: 10.1016/s0376-8716(00)00171-x. doi:10.1016/S0376-8716(00)00171-X. [DOI] [PubMed] [Google Scholar]
- Hougaard P. Frailty models. In: Klein JP, van Houwelingen HC, Ibrahim JG, Scheike TH, editors. Handbook of survival analysis. Taylor & Francis; Boca Raton, FL: 2014. pp. 458–472. [Google Scholar]
- Huizink AC, Levälahti E, Korhonen T, Dick DM, Pulkkinen L, Rose RJ, Kaprio J. Tobacco, cannabis, and other illicit drug use among Finnish adolescent twins: Causal relationship or correlated liabilities? Journal of Studies on Alcohol and Drugs. 2010;71:5–14. doi: 10.15288/jsad.2010.71.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston LD, O’Malley PM, Bachman JG, Schulenberg JE. Monitoring the future national results on drug use: 2012 overview, key findings on adolescent drug use. Institute for Social Research, The University of Michigan; Ann Arbor: 2013. [Google Scholar]
- Kandel D. Stages in adolescent involvement in drug use. Science. 1975;190:912–914. doi: 10.1126/science.1188374. doi:10.1126/science.1188374. [DOI] [PubMed] [Google Scholar]
- Kandel DB, Yamaguchi K, Chen K. Stages of progression in drug involvement from adolescence to adulthood: Further evidence for the gateway theory. Journal of Studies on Alcohol. 1992;53:447–457. doi: 10.15288/jsa.1992.53.447. [DOI] [PubMed] [Google Scholar]
- Kendler KS, Prescott CA, Myers J, Neale MC. The structure of genetic and environmental risk factors for common psychiatric and substance use disorders in men and women. Archives of General Psychiatry. 2003;60:929–937. doi: 10.1001/archpsyc.60.9.929. doi:10.1001/archpsyc.60.9.929. [DOI] [PubMed] [Google Scholar]
- Knopik VS, Heath AC, Madden PAF, Bucholz KK, Slutske WS, Nelson EC, Martin NG. Genetic effects on alcohol dependence risk: Re-evaluating the importance of psychiatric and other heritable risk factors. Psychological Medicine. 2004;34:1519–1530. doi: 10.1017/s0033291704002922. doi:10.1017/S0033291704002922. [DOI] [PubMed] [Google Scholar]
- Krueger RF, Hicks BM, Patrick CJ, Carlson SR, Iacono WG, McGue M. Etiologic connections among substance dependence, antisocial behavior and personality: Modeling the externalizing spectrum. Journal of Abnormal Psychology. 2002;111:411–424. doi:10.1037//0021-843X.111.3.411. [PubMed] [Google Scholar]
- Lessem JM, Hopfer CJ, Haberstick BC, Timberlake D, Ehringer MA, Smolen A, Hewitt JK. Relationship between adolescent marijuana use and young adult illicit drug use. Behavior Genetics. 2006;36:498–506. doi: 10.1007/s10519-006-9064-9. doi:10.1007/S20519-006-9064-9. [DOI] [PubMed] [Google Scholar]
- Lynskey MT, Agrawal A, Henders A, Nelson EC, Madden PAF, Martin NG. An Australian twin study of cannabis and other illicit drug use and misuse, and other psychopathology. Twin Research and Human Genetics. 2012;15:631–641. doi: 10.1017/thg.2012.41. doi:10.1017/thg.2012.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynskey MT, Heath AC, Bucholz KK, Slutske WS, Madden PAF, Nelson EC, Martin NG. Escalation of drug use in early-onset cannabis users vs. co-twin controls. Journal of the American Medical Association. 2003;289:427–433. doi: 10.1001/jama.289.4.427. doi:10.1001/jama.289.4.427. [DOI] [PubMed] [Google Scholar]
- Mackesy-Amiti ME, Fendrich M, Goldstein PJ. Sequence of drug use among serious drug users: Typical vs. atypical progression. Drug and Alcohol Dependence. 1997;45:185–196. doi: 10.1016/s0376-8716(97)00032-x. doi:10.1016/S0376-8716(97)00032-X. [DOI] [PubMed] [Google Scholar]
- Moss HB, Chen CM, Yi H. Early adolescent patterns of alcohol, cigarettes, and marijuana polysubstance use and young adult substance use outcomes in a nationally representative sample. Drug and Alcohol Dependence. 2014;136:51–62. doi: 10.1016/j.drugalcdep.2013.12.011. doi:10.1016/j.drugalcdep.2013.12.011. [DOI] [PubMed] [Google Scholar]
- Neale MC, Harvey E, Maes HM, Sullivan PF, Kendler KS. Extensions to the modeling of initiation and progression: Applications to substance use and abuse. Behavior Genetics. 2006;36:507–524. doi: 10.1007/s10519-006-9063-x. doi:10.1007/S20519-006-9063-x. [DOI] [PubMed] [Google Scholar]
- Neale MC, McArdle JJ. Structured latent growth curves for twin data. Twin Research. 2000;3:165–177. doi: 10.1375/136905200320565454. doi:10.1375/136905200320565454. [DOI] [PubMed] [Google Scholar]
- Palamar JJ, Ompad DC, Petkova E. Correlates of intentions to use cannabis among US high school seniors in the case of cannabis legalization. International Journal of Drug Policy. 2012;25:424–435. doi: 10.1016/j.drugpo.2014.01.017. doi:10.1016/j.drugpo.2014.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parra GR, O'Neill SE, Sher KJ. Reliability of self-reported age of substance involvement onset. Psychology of Addictive Behaviors. 2003;17:211–218. doi: 10.1037/0893-164X.17.3.211. doi:10.1037/0893-164X.17.3.211. [DOI] [PubMed] [Google Scholar]
- Patton GC, Coffey C, Carlin JB, Sawyer SM, Lynskey M. Reverse gateways? Frequent cannabis use as a predictor of tobacco initiation and nicotine dependence. Addiction. 2005;100:1518–1525. doi: 10.1111/j.1360-0443.2005.01220.x. doi:10.1111/j.1360-0443.2005.01220.x. [DOI] [PubMed] [Google Scholar]
- Rende R, Slomkowski C, Lloyd-Richardson E, Niaura R. Sibling effects on substance use in adolescence: Social contagion and genetic relatedness. Journal of Family Psychology. 2005;19:611–618. doi: 10.1037/0893-3200.19.4.611. doi:10.1037/0893-3200.19.4.611. [DOI] [PubMed] [Google Scholar]
- Sartor CE, Agrawal A, Lynskey MT, Bucholz KK, Madden PAF, Heath AC. Common genetic influences on the timing of first use for alcohol, cigarettes, and cannabis in young African-American women. Drug and Alcohol Dependence. 2009;102:49–55. doi: 10.1016/j.drugalcdep.2008.12.013. doi:10.1016/j.drugalcdep.2008.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silins E, Horwood LJ, Patton GC, Fergusson DM, Olsson CA, Hutchinson DM, Mattick RP. Young adult sequelae of adolescent cannabis use: An integrative analysis. Lancet Psychiatry. 2014;1:286–293. doi: 10.1016/S2215-0366(14)70307-4. [DOI] [PubMed] [Google Scholar]
- Snijders T, Bosker R. Multilevel analysis: An introduction to basic and advanced multilevel modeling. Sage; London, UK: 1999. [Google Scholar]
- Tanda G, Pontieri FE, DiChiara G. Cannabinoid and heroin activation of mesolimbic dopamine transmission by a common mu1 opioid receptor mechanism. Science. 1997;276:2048–2050. doi: 10.1126/science.276.5321.2048. [DOI] [PubMed] [Google Scholar]
- Tarter RE, Vanyukov M, Kirisci L, Reynolds M, Clark DB. Predictors of marijuana use in adolescents before and after licit drug use: Examination of the gateway hypothesis. American Journal of Psychiatry. 2006;163:2134–2140. doi: 10.1176/ajp.2006.163.12.2134. doi:10.1176/appi.ajp.163.12.2134. [DOI] [PubMed] [Google Scholar]
- Vitaro F, Brendgen M, Arseneault L. The discordant MZ-twin method: One step closer to the holy grail of causality. International Journal of Behavioral Development. 2009;33:376–382. doi:10.1177/0165025409340805. [Google Scholar]
- Walden B, Mcgue M, Iacono W, Burt SA, Elkins I. Identifying shared environmental contributions to early substance use: The respective roles of peers and parents. Journal of Abnormal Psychology. 2004;113:440–450. doi: 10.1037/0021-843X.113.3.440. doi:10.1037/0021-843X.113.3.440. [DOI] [PubMed] [Google Scholar]
- Wells JE, McGee MA. Violations of the usual sequence of drug initiation: Prevalence and associations with the development of dependence in the New Zealand Mental Health Survey. Journal of Studies on Alcohol and Drugs. 2008;69:789–795. doi: 10.15288/jsad.2008.69.789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White HR, Jarrett N, Valencia EY, Loeber R, Wei E. Stages and sequences of initiation and regular substance use in a longitudinal cohort of Black and White male adolescents. Journal of Studies on Alcohol and Drugs. 2007;68:173–181. doi: 10.15288/jsad.2007.68.173. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.