Abstract
Background
Hypomagnesemia has been associated with an increased risk of postoperative atrial fibrillation (POAF). While earlier studies have suggested a beneficial effect of magnesium (Mg) therapy, almost all of these are limited by small sample size and relatively low Mg dose. We hypothesized that high-dose Mg decreases the occurrence of new-onset POAF, and we tested this hypothesis using data from a prospective trial assessing the effect of Mg on cognitive outcomes in cardiac surgical patients.
Methods
A total of 389 patients undergoing cardiac surgery were enrolled in this double-blind, placebo-controlled trial. Subjects were randomized to receive Mg as a 50 mg/kg bolus immediately after induction of anesthesia followed by another 50 mg/kg as an infusion given over 3 h (total dose 100 mg/kg) or placebo. The effect of Mg therapy on POAF was tested with logistic regression, adjusting for the risk of AF using the Risk Index for Atrial Fibrillation after Cardiac Surgery.
Results
Among the 363 patients analyzed, after excluding patients with chronic or acute preoperative AF (Placebo: n=177, Mg: n=186), the incidence of new-onset POAF was 42.5% (95% CI: 35 – 50%) in the Mg group compared to 37.9% (95% CI: 31 – 45%) in the placebo group (p=0.40). The 95% confidence interval for this absolute risk difference of 4.6% is −5.5% to 14.7%. The time to onset of POAF was also identical between the groups, and no significant effect of Mg was found in logistic regression analysis adjusting for AF risk (odds ratio 1.09 with 95% CI 0.69 – 1.72, p=0.73).
Conclusions
High-dose intraoperative Mg therapy did not decrease the incidence of new-onset POAF after cardiac surgery.
Introduction
New-onset postoperative atrial fibrillation (POAF) is a common complication of cardiac surgery. The incidence ranges between 27–63%, with the highest risk seen in combined coronary artery bypass graft (CABG) and valve surgery.1–3 POAF has a peak incidence on postoperative day 24 and is associated with several adverse outcomes, including increased cardiovascular, renal, and cerebral complications; increased cost of care and resource utilization; and decreased long-term survival.1,5–8
Hypomagnesemia has been associated with an increased risk of AF, both after cardiac surgery9,10 and in ambulatory settings.11,12 Hypomagnesemia is believed to promote AF through a variety of mechanisms, including effects on atrioventricular conduction, action potential duration, and the atrial effective refractory period.13 Specifically, in isolated rabbit hearts, magnesium (Mg) produces a significant and consistent prolongation of the atrial effective refractory period,13 thereby reducing myocardial excitability. Furthermore, Mg decreases the rate of diastolic depolarization of sinoatrial pacemaker cells.14 The interaction between surgical stress-induced sympathetic hyperactivity and Mg also appears to be important. The high concentration of norepinephrine released perioperatively promotes the efflux of Mg from cells,15 altering cardiomyocyte excitability and automaticity. Thus, the administration of Mg may attenuate sympathoadrenal-mediated arrhythmias.16,17
While several trials and meta-analyses have suggested a beneficial effect of Mg administration in preventing POAF,18–21 many of these studies were limited by small sample size, relatively low Mg dosage, and an inability to account for concomitant medications known to influence POAF. We hypothesized that high-dose Mg decreases the occurrence of new-onset POAF, and we tested this hypothesis using data from a prospective trial assessing the effect of Mg on cognitive outcomes in patients undergoing cardiac surgery.
Methods
Study Design
This study is a secondary analysis of a prospective, double-blind, randomized trial of IV Mg versus saline that was designed to evaluate neurocognitive outcomes, as previously published.22 The primary outcome for the current analyses was the incidence of new-onset POAF of any duration. Data relevant to this outcome were prospectively collected during the original clinical trial.
With approval from the Duke University Health System Institutional Review Board and written informed consent, 389 patients scheduled to undergo CABG, valve, or CABG plus valve surgery with cardiopulmonary bypass (CPB) were enrolled in this prospective, randomized, double-blind, placebo-controlled clinical trial. Exclusion criteria included planned circulatory arrest, history of symptomatic cerebrovascular disease (e.g., stroke with residual deficit), psychiatric illness (e.g., any clinical diagnosis requiring therapy), renal failure (defined as serum creatinine >2 mg/dL), hepatic insufficiency (defined as aspartate aminotransferase and/or alanine aminotransferase >1.5x the upper limit of normal), higher alcohol consumption (defined as >2 drinks/day), inability to read, less than a seventh-grade education, or score of <24 on baseline Mini Mental State examination. Enrolled subjects were randomized to receive either Mg, as a 50 mg/kg bolus over 20 min after the induction of anesthesia followed by another 50 mg/kg infused over 3 h (for a total dose of 100 mg/kg), or placebo (normal saline administered as a bolus and identical volume and duration infusion to the Mg group to preserve blinding). A group assignment schedule was prepared using a randomization function in SAS™ (Statistical Analysis System; SAS Institute Inc., Cary, NC, USA) and stored in consecutively numbered sealed envelopes until allocation. Randomization occurred prior to surgery after confirming the procedure type and planned use of CPB.
The patient’s ECG was continuously monitored after surgery with telemetry monitoring until hospital discharge. POAF was diagnosed by telemetry or 12-lead ECG and was defined as a supraventricular tachycardia with the absence of distinct p-waves and irregular R-R intervals.23 POAF was considered new-onset if it occurred postoperatively at any time prior to hospital discharge.
Patient Management
Anesthesia was induced with midazolam and fentanyl and maintained with isoflurane. All patients underwent nonpulsatile, hypothermic (30–32°C) CPB with a membrane oxygenator and arterial line filter via a pump primed with crystalloid. Serial hematocrit levels were maintained at ≥0.21. Patients were heparinized (300–400 U/kg) to a target activated coagulation time of >480 s prior to the initiation of CPB. Pump flow rates of 2–2.4 L•min−1•m2 were maintained throughout CPB to maintain a mean arterial pressure of 50–80 mmHg. Arterial blood gases were measured every 15–30 min to maintain PaCO2 at 35–40 mmHg unadjusted for temperature (α-stat) and PaO2 at 150–250 mmHg.
Preoperative and daily postoperative medication therapy was also recorded. The standard of care postoperatively was to administer 4 g of MgSO4 IV for serum levels ≤ 1.8 mg/dl or 2g of MgSO4 for serum levels 1.8 – 2.0 mg/dl.
Laboratory Assessments
In the first 121 patients, blood was sampled at baseline, on admission to the intensive care unit, and at 24 h and 48 h after the initial bolus for the measurement of serum Mg levels. Subsequently, to enhance patient safety, an additional blood sample was obtained immediately after the Mg bolus dose.
Statistical Analysis
Categorical and continuous demographic data were compared between the groups with χ2 test, Wilcoxon rank-sum, or t test, as appropriate. The primary analyses were conducted according to the intention-to-treat principle. Descriptive summaries are presented as counts and proportions for categorical variables and mean ± standard deviation for continuous variables. For the primary endpoint of the incidence of POAF, the effect of magnesium administration was evaluated using the χ2 test. A logistic regression model was pre-specified to test the effect of magnesium treatment and to account for the risk of developing AF using the Risk Index for Atrial Fibrillation after Cardiac Surgery score, as previously described,1 and subsequently modified to include preoperative statin use (Table 1). Briefly, from a derivation cohort of 3093 patients, associations between predictor variables and postoperative atrial fibrillation were identified to develop a risk model, which was subsequently assessed in a validation cohort of 1564 patients. Risk factors associated with atrial fibrillation were advanced age, history of atrial fibrillation, valve surgery and postoperative withdrawal of a beta-blocker or an angiotensin-converting enzyme inhibitor. Conversely, reduced risk was associated with postoperative administration of beta-blockers, ACE inhibitors, statins, potassium supplementation and nonsteroidal anti-inflammatory drugs. To develop relative weights for the predictors in the Multicenter Study of Perioperative Ischemia risk index, the parameter estimates of the final logistic model were multiplied by 10 and rounded to the nearest integer. The relative weight was then assigned to each binary predictor as well as to each age category (10-year increments). The risk score for each patient was calculated by summing the relative weights across the predictors present for a particular patient. The low risk group contained scores below 14 (25th percentile); the medium risk group contained scores between 14 and 31 (25th to 75th percentile); and the high risk group contained scores higher than 31 (75th percentile).
Table 1.
Modified Risk Index for Atrial Fibrillation after Cardiac Surgery
| Predictor | Incidence of Postoperative AF No./Total (%) | Risk Score | OR (95% CI) | P Value |
|---|---|---|---|---|
| Age, y | ||||
| < 30 | 0 | 0 | 1.74 (1.58 – 1.92) | <0.001 |
| 30–39 | 2/36 (5.6) | 6 | ||
| 40–49 | 19/229 (8.3) | 12 | ||
| 50–59 | 160/795 (20.1) | 18 | ||
| 60–69 | 378/1145 (33.0) | 24 | ||
| 70–79 | 377/817 (46.1) | 30 | ||
| ≥ 80 | 40/68 (58.8) | 36 | ||
| Medical history | ||||
| Atrial fibrillation | 142/268 (53.0) | 7 | 2.04 (1.52 – 2.75) | <0.001 |
| Chronic obstructive pulmonary disease | 137/320 (42.8) | 4 | 1.42 (1.09 – 1.86) | 0.010 |
| Concurrent valve surgery | 154/286 (53.9) | 5 | 1.67 (1.25 – 2.23) | <0.001 |
| Withdrawal of treatment | ||||
| Beta-blockers | 396/784 (50.5) | 7 | 1.95 (1.55 – 2.46) | <0.001 |
| ACE-I | 320/692 (46.2) | 5 | 1.65 (1.34 – 2.02) | <0.001 |
| Preoperative and postoperative treatment | ||||
| Beta-blockers | 228/1289 (17.7) | −7 | 0.50 (0.39 – 0.62) | <0.001 |
| ACE-I | 126/626 (20.1) | −5 | 0.61 (0.48 – 0.78) | <0.001 |
| Statins | 128/665 (19.3) | −5 | 0.58 (0.46 – 0.74) | <0.001 |
| Postoperative Beta-blockers | 51/228 (17.7) | −11 | 0.32 (0.22 – 0.46) | <0.001 |
| Other treatment | ||||
| Potassium supplementation | 774/2688 (28.8) | −5 | 0.62 (0.49 – 0.79) | <0.001 |
| NSAIDs | 173/934 (18.5) | −7 | 0.50 (0.41 – 0.62) | <0.001 |
OR, odds ratio; ACE-I, angiotensin-converting enzyme inhibitor; NSAID, nonsteroidal anti-inflammatory drug.
The linear regression model used to evaluate the primary endpoint included an interaction test and a quadratic test of the assumption of linearity between the AF risk index and the logit linearity of AF risk; linearity was also verified by inspection of AF proportions in deciles of risk. Model fit was assessed with the Hosmer-Lemeshow test. All analyses were performed with SAS™ version 9.3 (SAS Institute Inc., Cary, NC, USA); p <0.05 was considered significant.
In the parent trial, we expected that the incidence of cognitive deficits in patients undergoing cardiac surgery would be ≈ 40%. We hypothesized that magnesium treatment would decrease this incidence to 25%, and a sample size of 170 per group would yield power of 80% at a significance level of 0.05 to detect this difference. To allow for a 15% loss to follow-up, we intended to recruit a total of 400 patients. Since the sample size was determined by the parent trial, additional power analyses related to new-onset POAF were not conducted.
Results
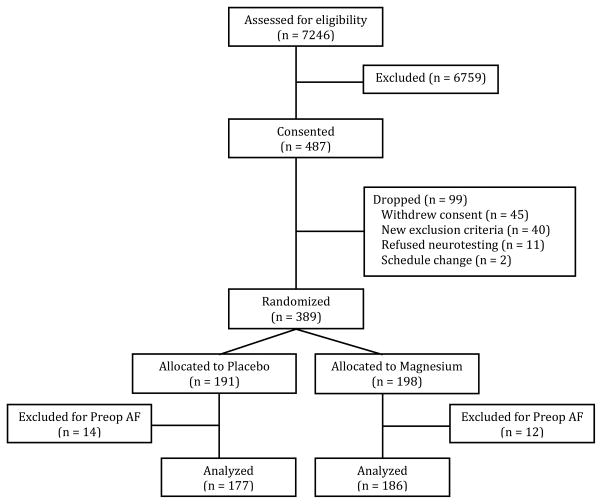
A CONSORT diagram detailing the screening, recruitment, and analysis of patients is presented in Figure 1. Prior to the analysis, 14 patients who received placebo and 11 patients who received Mg were excluded because of documented preoperative cardiac rhythm other than normal sinus rhythm (i.e., both acute AF and chronic persistent AF), leaving 177 patients in the placebo group and 186 patients in the Mg group for final analysis. Patient characteristics and pertinent intraoperative details are summarized in Table 2. As expected from randomization, there were no statistically significant differences between the Mg and placebo groups with respect to demographic characteristics, although there was a trend towards more females in the placebo group. Preoperative Mg levels were not different between the two groups. After the bolus dosing, serum Mg levels were significantly higher in the Mg treatment group at all time-points (Figure 2).
Figure 1.
CONSORT diagram showing the flow of study participants.
Table 2.
Characteristics of the subjects allocated to treatment with magnesium versus placebo.
| Variable | Magnesium (n = 186) | Placebo (n = 177) | P Value |
|---|---|---|---|
| Age (SD), y | 68.2 (8.3) | 67.8 (7.9) | 0.68 |
| Sex, % female | 26.3 | 35.6 | 0.06 |
| Race, % white | 81.2 | 75.1 | 0.16 |
| Weight (SD), kg | 83.6 (14.5) | 81.2 (15) | 0.12 |
| History of hypertension, % | 80.7 | 80.1 | 0.90 |
| Diabetes mellitus, % | 36.8 | 34.7 | 0.68 |
| Previous MI, % | 32.2 | 31.6 | 0.89 |
| Ejection fraction (SD) | 52.4 (11.8) | 52.9 (12.8) | 0.46 |
| Magnesium level at baseline (SD) | 1.92 (0.24) | 1.89 (0.27) | 0.16 |
| Surgical procedure, % | 0.94 | ||
| CABG | 80.7 | 79.7 | |
| CABG+valve | 14.5 | 14.7 | |
| Valve | 4.8 | 5.7 | |
| Redo surgery, % | 0.5 | 0.0 | 0.99 |
| Number of grafts | 3.2 (1.2) | 3.1 (1.3) | 0.56 |
| Cross-clamp time (SD), min | 82.8 (33.5) | 81.2 (33.9) | 0.45 |
| CPB time (SD), min | 134.8 (49.4) | 132.8 (52.7) | 0.46 |
CABG, coronary artery bypass grafting; CPB, cardiopulmonary bypass
Figure 2.
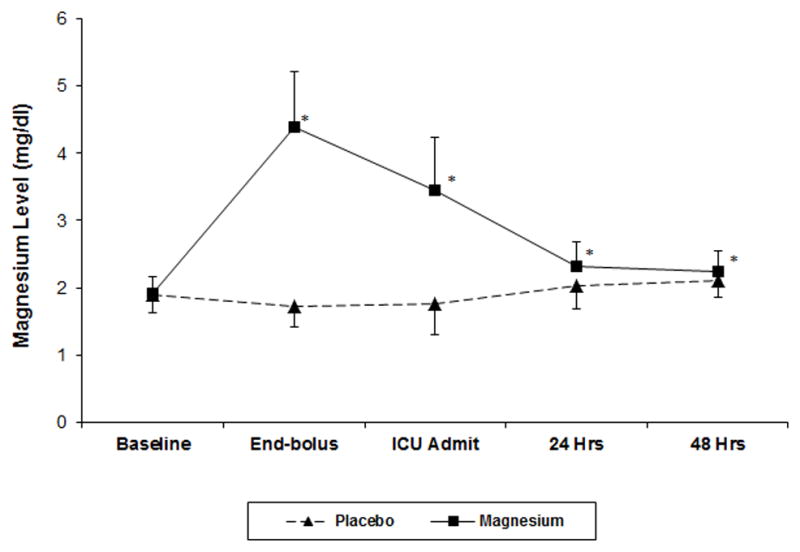
Serum concentration of magnesium at the five measurement time-points. Data are mean±SD. *P<0.001.
There was no difference in the proportion of patients with new-onset POAF between the Mg group (42.5%, 95% confidence interval [CI], 35 – 50%)] and the placebo group (37.9%, 95% CI, 31 – 45%; p=0.40). In a secondary “as-treated” analysis, 5 placebo patients were reassigned to the Mg group, but once again there was no difference in the incidence of POAF (Mg 41.4%, 95% CI, 34–48% vs. Placebo, 39.0%, 95% CI, 32–46%; p=0.64). Of these 5 placebo patients reassigned to the Mg group for the “as-treated” analysis, 1 had been incorrectly randomized and the other 4 had received magnesium (2 g) intraoperatively for the management of ventricular arrhythmias. The proportion of patients receiving other medications known to have an association with AF (either with AF risk or those medications used as treatment for AF) preoperatively and postoperatively, but prior to the onset of AF, was not different between patients receiving Mg compared to placebo (Table 3). Finally, there was no difference between treatment groups with regard to duration of POAF (Mg: 2 [1–4] days vs. Placebo: 2 [1–4] days; p=0.91), time to onset of POAF (Mg: 2 [2–3] days vs. Placebo: 2 [1–3] days; p=0.71), recurrence of atrial fibrillation (mg: 44.3% vs. Placebo: 40.3%; p=0.63) or in the rate of atrial fibrillation at discharge (Mg: 7.2% vs. Placebo: 10.7%; p=0.54).
Table 3.
Perioperative Medication Administration
| Variable | Magnesium (n = 186) | Placebo (n = 177) | P value |
|---|---|---|---|
| Medications given before AF onset | |||
| Beta-blocker preop but not postop, % | 16.1 | 10.7 | 0.13 |
| Beta-blocker preop and postop, % | 51.1 | 50.9 | 0.97 |
| Beta-blocker postop only, % | 22.6 | 26.6 | 0.38 |
| ACE-I preop but not postop, % | 43.6 | 35.0 | 0.10 |
| ACE-I preop and postop, % | 12.4 | 18.1 | 0.13 |
| Statin preop and postop, % | 38.7 | 37.3 | 0.78 |
| Potassium postop, % | 81.2 | 83.6 | 0.54 |
| NSAID or COX-2 inhibitor postop, % | 19.4 | 17.0 | 0.55 |
All results were analyzed with the chi-square test. AF, atrial fibrillation; ACE-I, angiotensin-converting enzyme inhibitor; NSAID, nonsteroidal anti-inflammatory drug; COX, cyclooxygenase.
The AF risk index score, reported as (median, mean±SD), was similar between the treatment groups (Mg: 14, 16.5±11 vs. Placebo: 13, 14.8±11; p=0.142). As expected, the AF risk index score was different between patients who did (20, 21±13) and those who did not (12, 12.0±9) develop postoperative AF (p<0.0001). The interaction between treatment groups and risk level for AF was also not significant (Low Risk – Mg: 30.7% vs. Placebo: 23.0%, p=0.23; Medium Risk – Mg: 46.2% vs. Placebo: 51.6%, p=0.52; High Risk – Mg 80.0% vs. Placebo 84.6%, p=0.40). Finally, no significant effect of Mg on POAF was found in logistic regression analysis adjusting for the AF risk score (Mg treatment odds ratio 1.09 with 95% CI 0.69 – 1.72, p=0.73, area under the ROC curve, 0.72). The Hosmer-Lemeshow test for goodness of fit was non-significant for lack of fit (p=0.51).
Discussion
In this prospective, randomized, double-blinded, placebo-controlled study of the administration of IV Mg versus saline during adult cardiac surgery with CPB, we found that systemic, high-dose intraoperative Mg therapy did not reduce the incidence of new-onset POAF.
Mg has several antiarrhythmic properties that make it a good pharmacologic candidate for the prevention and treatment of arrhythmias.24,25 Consistent with these electrophysiologic actions, Mg administration has been shown in several clinical studies to significantly improve the rate of chemical26–28 and electrical cardioversion29 of atrial arrhythmias. Investigations of Mg prophylaxis for the prevention of POAF in the context of cardiac surgery appeared to confirm these beneficial results.18–21 However, several of these studies were limited by small sample size, improper blinding, and the use of a lower dose of Mg than in the current study. In fact, by combining studies on Mg prophylaxis in CABG surgery that were properly blinded and with sufficient patient sample sizes, Hazelrigg et al. 34 failed to find an overall reduction in postoperative atrial or ventricular arrhythmias. Cook et al.35 were also unable to demonstrate a difference in the rate of clinically important atrial arrhythmia in patients receiving Mg versus placebo in addition to routine beta-blockade. Most of these studies, however, had not adequately adjusted for the confounding effects of beta-blockers, ACE inhibitors, statins, potassium, and non-steroidal anti-inflammatory drugs. In our randomized, placebo controlled, double-blind study, despite adjusting for all relevant medications given prior to the onset of POAF, we also could not find a benefit for intraoperative Mg administration in preventing POAF after cardiac surgery.
Hypomagnesemia is common after cardiac surgery, and it is standard practice to correct the deficit with IV Mg. However, serum ionized Mg levels do not necessarily reflect intracellular levels, and the equilibration between tissue compartments and intracellular concentrations is often a slow process.24 Thus, serum Mg is a poor marker of intracellular magnesium homeostasis, and its administration has unproven effects on tissue and intracellular levels of Mg.30 Moreover, although patients with arrhythmias have been shown to have a relative deficiency in intracellular Mg,24,27 the dose and duration of treatment required to adequately restore intracellular Mg concentrations is not known. Importantly, the timing of prophylactic Mg therapy has been variable among studies and may represent a critical factor in the success or failure of this intervention. A previous meta-analysis indicated that lower doses of Mg (defined as mean cumulative dose of 8.2 g) are more effective than moderate-to-higher doses (mean cumulative dose of 15 g) in reducing postoperative AF.32 In our study, we administered a combination of a bolus dose plus infusion to achieve a total cumulative Mg dose of 100 mg/kg (mean dose of 8.4 g). This dosing produced statistically significantly higher serum Mg concentrations at all measured time points when compared to patients who received placebo. The immediate and postoperative serum levels achieved in our study were similar to those achieved by Cook et al.35 Although statistically higher than placebo, the difference in the mean serum concentration of Mg between our treatment and placebo groups fell to a mean of 0.27 mg/dL by 24 h [Mg, 2.31±0.38 mg/dL vs. Placebo, 2.04±0.33 mg/dL] and 0.15 mg/dL by 48 h [Mg, 2.25±0.30 mg/dL vs. Placebo, 2.10±0.25 mg/dL] differences that are not likely to be clinically significant. In fact, with the peak incidence of POAF occurring around day 2, the minimal elevation in serum Mg in the treatment group at 48 h may partially explain the lack of benefit in preventing POAF. However, Cook et al.35 extended their treatment to postoperative day 4 and were still unable to reduce the incidence of POAF.
Sympathetic activation is postulated to be a mechanism underlying POAF.33 It has been hypothesized that Mg exerts a prophylactic effect by inhibiting adrenergic-mediated automaticity. However, there is a discrepancy in the time course of sympathetic activation and POAF, with the peak of sympathetic activation occurring around 24 h postoperatively whereas the peak in the onset of POAF occurs between 2 and 4 d postoperatively.34 The inflammatory response induced by cardiac surgery, however, has a time course that more closely mirrors that of POAF.35,36 The apparent efficacy of specific anti-inflammatory drugs, particularly statins and colchicine,37,38 in reducing the incidence of POAF, combined with the failure of Mg and other modulators of sympathetic hyperactivity to fully prevent POAF39 suggest that the pursuit of inflammatory mechanisms of POAF may be more fruitful.
The primary limitation of this study is that it is a secondary analysis of a prospective trial designed to evaluate neurocognitive outcomes in the setting of intraoperative high-dose Mg administration. As such, the administration of postoperative medications that are potentially relevant to POAF was not standardized, although we did not see a difference between groups in the preoperative or postoperative administration of relevant medication. Furthermore, we recorded all medications administered prior to the onset of POAF, and thus, unlike prior studies, we are able to more accurately account for the temporal relationships between all (i.e., not just beta-blockers) medication therapy and POAF. A second limitation is that our intervention was limited to the intraoperative period. It is possible that high-dose Mg therapy extended into the postoperative period could alter the occurrence of POAF. However, a standardized postoperative magnesium treatment protocol was used, and we note that the Mg levels were statistically higher in the treatment group. Finally, our definition of new-onset POAF included POAF of any duration and may have been too liberal. We therefore repeated the analysis in the cohort of patients (n=340) who received treatment for POAF (i.e. “significant” AF). In this subgroup, once again, the incidence of POAF did not differ between the treatment groups (Mg: 21% vs Placebo: 15%, adjusted p=0.33).
Although many pharmacological approaches to preventing atrial fibrillation after cardiac surgery have been investigated,40,41 the studies evaluating Mg have yielded mixed results. We found that, despite significantly higher Mg levels in the intraoperative and immediate postoperative period, high-dose intraoperative Mg therapy did not decrease the incidence of new-onset POAF. Considering the high incidence of AF after cardiac surgery and its potential for significant short- and long-term morbidity, especially embolic stroke, further investigation into preventative strategies is warranted. Given the presumptive multifactorial etiology of POAF, its effective prevention will likely require a multimodal pharmacologic approach.
Acknowledgments
Funding: This work was supported in part by grants HL069081 (Dr. Newman), HL096978, HL108280, and HL109971 (Dr. Mathew) and grant M01-RR-30 from the National Institutes of Health, Washington, DC, and by the Division of Cardiothoracic Anesthesiology and Critical Care Medicine, Department of Anesthesiology, Duke University Medical Center, Durham, NC.
Footnotes
The authors declare no conflicts of interest.
Reprints will not be available from the authors.
DISCLOSURES:
Name: Rebecca Y. Klinger, MD, MS
Contribution: This author helped analyze the data, and write the manuscript
Attestation: Rebecca Y. Klinger has seen the original study data, reviewed the analysis of the data, and approved the final manuscript
Name: Christopher A. Thunberg, MD
Contribution: This author helped design the study, conduct the study, analyze the data, and write the manuscript
Attestation: Christopher A. Thunberg has seen the original study data, reviewed the analysis of the data, and approved the final manuscript
Name: William D. White, MPH
Contribution: This author helped design the study, analyze the data, and write the manuscript
Attestation: William D. White has seen the original study data, reviewed the analysis of the data, and approved the final manuscript
Name: Manuel Fontes, MD
Contribution: This author helped design the study, conduct the study, and write the manuscript
Attestation: Manuel Fontes has seen the original study data, reviewed the analysis of the data, and approved the final manuscript
Name: Nathan H. Waldron, MD
Contribution: This author helped write the manuscript
Attestation: Nathan H. Waldron reviewed the analysis of the data and approved the final manuscript
Name: Jonathan P. Piccini, MD, MHSc
Contribution: This author helped design the study and write the manuscript
Attestation: Jonathan P. Piccini reviewed the analysis of the data and approved the final manuscript
Name: G. Chad Hughes, MD
Contribution: This author helped design the study and write the manuscript
Attestation: G. Chad Hughes reviewed the analysis of the data and approved the final manuscript
Name: Mihai V. Podgoreanu, MD
Contribution: This author helped design the study and write the manuscript
Attestation: Mihai V. Podgoreanu reviewed the analysis of the data and approved the final manuscript
Name: Mark Stafford-Smith, MD
Contribution: This author helped design the study and write the manuscript
Attestation: Mark Stafford-Smith reviewed the analysis of the data and approved the final manuscript
Name: Mark F. Newman, MD
Contribution: This author helped design the study and write the manuscript
Attestation: Mark F. Newman reviewed the analysis of the data and approved the final manuscript
Name: Joseph P. Mathew, MD, MHSc
Contribution: This author helped design the study, conduct the study, analyze the data, and write the manuscript
Attestation: Joseph P. Mathew has seen the original study data, reviewed the analysis of the data, approved the final manuscript, and is the author responsible for archiving the study files
This manuscript was handled by: Charles W. Hogue, MD
Contributor Information
Rebecca Y. Klinger, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina.
Christopher A. Thunberg, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina (Current Affiliation: Department of Anesthesiology, Mayo Clinic, Scottsdale, Arizona).
William D. White, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina.
Manuel Fontes, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina.
Nathan H. Waldron, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina.
Jonathan P. Piccini, Department of Medicine-Cardiology, Duke University Medical Center, Durham, North Carolina.
G. Chad Hughes, Department of Surgery, Duke University Medical Center, Durham, North Carolina.
Mihai V. Podgoreanu, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina.
Mark Stafford-Smith, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina.
Mark F. Newman, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina.
Joseph P. Mathew, Department of Anesthesiology, Duke University Medical Center, Durham, North Carolina.
References
- 1.Mathew JP, Fontes ML, Tudor IC, Ramsay J, Duke P, Mazer CD, Barash PG, Hsu PH, Mangano DT. A multicenter risk index for atrial fibrillation after cardiac surgery. JAMA. 2004;291(14):1720–1729. doi: 10.1001/jama.291.14.1720. [DOI] [PubMed] [Google Scholar]
- 2.Ommen SR, Odell JA, Stanton MS. Atrial arrhythmias after cardiothoracic surgery. N Engl J Med. 1997;336(20):1429–1434. doi: 10.1056/NEJM199705153362006. [DOI] [PubMed] [Google Scholar]
- 3.Echahidi N, Pibarot P, O’Hara G, Mathieu P. Mechanisms, prevention, and treatment of atrial fibrillation after cardiac surgery. J Am Coll Cardiol. 2008;51(8):793–801. doi: 10.1016/j.jacc.2007.10.043. [DOI] [PubMed] [Google Scholar]
- 4.Aranki SF, Shaw DP, Adams DH, Rizzo RJ, Couper GS, VanderVliet M, Collins JJ, Cohn LH, Burstin HR. Predictors of atrial fibrillation after coronary artery surgery. Current trends and impact on hospital resources. Circulation. 1996;94(3):390–397. doi: 10.1161/01.cir.94.3.390. [DOI] [PubMed] [Google Scholar]
- 5.Kim MH, Deeb GM, Morady F, Bruckman D, Hallock LR, Smith KA, Karavite DJ, Bolling SF, Pagani FD, Wahr JA, Sonnad SS, Kazanjian PE, Watts C, Williams M, Eagle KA. Effect of postoperative atrial fibrillation on length of stay after cardiac surgery (The Postoperative Atrial Fibrillation in Cardiac Surgery study [PACS(2)] Am J Cardiol. 2001;87(7):881–885. doi: 10.1016/s0002-9149(00)01530-7. [DOI] [PubMed] [Google Scholar]
- 6.Villareal RP, Hariharan R, Liu BC, Kar B, Lee V-V, Elayda M, Lopez JA, Rasekh A, Wilson JM, Massumi A. Postoperative atrial fibrillation and mortality after coronary artery bypass surgery. J Am Coll Cardiol. 2004;43(5):742–748. doi: 10.1016/j.jacc.2003.11.023. [DOI] [PubMed] [Google Scholar]
- 7.Helgadottir S, Sigurdsson MI, Ingvarsdottir IL, Arnar DO, Gudbjartsson T. Atrial fibrillation following cardiac surgery: risk analysis and long-term survival. J Cardiothorac Surg. 2012;7:87. doi: 10.1186/1749-8090-7-87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mathew JP, Parks R, Savino JS, Friedman AS, Koch C, Mangano DT, Browner WS. Atrial fibrillation following coronary artery bypass graft surgery: predictors, outcomes, and resource utilization. MultiCenter Study of Perioperative Ischemia Research Group. JAMA. 1996;276(4):300–306. [PubMed] [Google Scholar]
- 9.Singh RB, Manmohan MD, Dube KP, Singh VP. Serum magnesium concentrations in atrial fibrillation. Acta Cardiol. 1976;31(3):221–226. [PubMed] [Google Scholar]
- 10.DeCarli C, Sprouse G, LaRosa JC. Serum magnesium levels in symptomatic atrial fibrillation and their relation to rhythm control by intravenous digoxin. Am J Cardiol. 1986;57(11):956–959. doi: 10.1016/0002-9149(86)90738-1. [DOI] [PubMed] [Google Scholar]
- 11.Khan AM, Lubitz SA, Sullivan LM, Sun JX, Levy D, Vasan RS, Magnani JW, Ellinor PT, Benjamin EJ, Wang TJ. Low serum magnesium and the development of atrial fibrillation in the community: the Framingham Heart Study. Circulation. 127(1):33–38. doi: 10.1161/CIRCULATIONAHA.111.082511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Misialek JR, Lopez FL, Lutsey PL, Huxley RR, Peacock JM, Chen LY, Soliman EZ, Agarwal SK, Alonso A. Serum and dietary magnesium and incidence of atrial fibrillation in whites and in African Americans--Atherosclerosis Risk in Communities (ARIC) study. Circ J. 2013;77(2):323–329. doi: 10.1253/circj.cj-12-0886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Watanabe Y, Dreifus LS. Electrophysiological effects of magnesium and its interactions with potassium. Cardiovasc Res. 1972;6(1):79–88. doi: 10.1093/cvr/6.1.79. [DOI] [PubMed] [Google Scholar]
- 14.Op’t Hof T, Mackaay AJ, Bleeker WK, Jongsma HJ, Bouman LN. Differences between rabbit sinoatrial pacemakers in their response to Mg, Ca and temperature. Cardiovas Res. 1983;17(9):526–532. doi: 10.1093/cvr/17.9.526. [DOI] [PubMed] [Google Scholar]
- 15.Romani A, Scarpa A. Hormonal control of Mg2+ transport in the heart. Nature. 1990;346(6287):841–844. doi: 10.1038/346841a0. [DOI] [PubMed] [Google Scholar]
- 16.Wesley RC, Haines DE, Lerman BB, DiMarco JP, Crampton RS. Effect of intravenous magnesium sulfate on supraventricular tachycardia. Am J Cardiol. 1989;63(15):1129–1131. doi: 10.1016/0002-9149(89)90092-1. [DOI] [PubMed] [Google Scholar]
- 17.Hasegawa J, Matsumoto T, Takami T, Fujimoto Y, Kotake H, Mashiba H. Suppression of catecholamine-induced abnormal pacemaker activities by magnesium ion in guinea pig cardiac muscle cells. Magnesium. 1989;8(2):94–99. [PubMed] [Google Scholar]
- 18.Miller S, Crystal E, Garfinkle M, Lau C, Lashevsky I, Connolly SJ. Effects of magnesium on atrial fibrillation after cardiac surgery: a meta-analysis. Heart. 2005;91(5):618–623. doi: 10.1136/hrt.2004.033811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Alghamdi AA, Al-Radi OO, Latter DA. Intravenous magnesium for prevention of atrial fibrillation after coronary artery bypass surgery: a systematic review and meta-analysis. J Card Surg. 2005;20(3):293–299. doi: 10.1111/j.1540-8191.2005.200447.x. [DOI] [PubMed] [Google Scholar]
- 20.Shiga T, Wajima Z, Inoue T, Ogawa R. Magnesium prophylaxis for arrhythmias after cardiac surgery: a meta-analysis of randomized controlled trials. Am J Med. 2004;117(5):325–333. doi: 10.1016/j.amjmed.2004.03.030. [DOI] [PubMed] [Google Scholar]
- 21.Gu W-J, Wu Z-J, Wang P-F, Aung LHH, Yin R-X. Intravenous magnesium prevents atrial fibrillation after coronary artery bypass grafting: a meta-analysis of 7 double-blind, placebo-controlled, randomized clinical trials. Trials. 2012;13:41. doi: 10.1186/1745-6215-13-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mathew JP, White WD, Schinderle DB, Podgoreanu MV, Berger M, Milano CA, Laskowitz DT, Stafford-Smith M, Blumenthal JA, Newman MF. Intraoperative magnesium administration does not improve neurocognitive function after cardiac surgery. Stroke. 2013;44(12):3407–3413. doi: 10.1161/STROKEAHA.113.002703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC, Conti JB, Ellinor PT, Ezekowitz MD, Field ME, Murray KT, Sacco RL, Stevenson WG, Tchou PJ, Tracy CM, Yancy CW. 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014;64(21):e1–e76. doi: 10.1016/j.jacc.2014.03.022. [DOI] [PubMed] [Google Scholar]
- 24.Shah SA, Clyne CA, Henyan N, Migeed M, Yarlagadda R, Silver BB, Kluger J, White CM. Impact of magnesium sulfate on serum magnesium concentrations and intracellular electrolyte concentrations among patients undergoing radio frequency catheter ablation. Conn Med. 2008;72(5):261–265. [PubMed] [Google Scholar]
- 25.Rasmussen HS, Thomsen PE. The electrophysiological effects of intravenous magnesium on human sinus node, atrioventricular node, atrium, and ventricle. Clin Cardiol. 1989;12(2):85–90. doi: 10.1002/clc.4960120204. [DOI] [PubMed] [Google Scholar]
- 26.Klevay LM, Milne DB. Low dietary magnesium increases supraventricular ectopy. Am J Clin Nutr. 2002;75(3):550–554. doi: 10.1093/ajcn/75.3.550. [DOI] [PubMed] [Google Scholar]
- 27.Steinwender C, Hönig S, Kypta A, Kammler J, Schmitt B, Leisch F, Hofmann R. Pre-injection of magnesium sulfate enhances the efficacy of ibutilide for the conversion of typical but not of atypical persistent atrial flutter. Int J Cardiol. 2010;141(3):260–265. doi: 10.1016/j.ijcard.2008.12.012. [DOI] [PubMed] [Google Scholar]
- 28.Tercius AJ, Kluger J, Coleman CI, White CM. Intravenous magnesium sulfate enhances the ability of intravenous ibutilide to successfully convert atrial fibrillation or flutter. Pacing Clin Electrophysiol. 2007;30(11):1331–1335. doi: 10.1111/j.1540-8159.2007.00866.x. [DOI] [PubMed] [Google Scholar]
- 29.Coleman CI, Sood N, Chawla D, Talati R, Ghatak A, Kluger J. Intravenous magnesium sulfate enhances the ability of dofetilide to successfully cardiovert atrial fibrillation or flutter: results of the Dofetilide and Intravenous Magnesium Evaluation. Europace. 2009;11(7):892–895. doi: 10.1093/europace/eup084. [DOI] [PubMed] [Google Scholar]
- 30.Sultan A, Steven D, Rostock T, Hoffmann B, Müllerleile K, Servatius H, Drewitz I, Lueker J, Meyer P, Salukhe T, Willems S. Intravenous administration of magnesium and potassium solution lowers energy levels and increases success rates electrically cardioverting atrial fibrillation. J Cardiovasc Electrophysiol. 2012;23(1):54–59. doi: 10.1111/j.1540-8167.2011.02146.x. [DOI] [PubMed] [Google Scholar]
- 31.DiCarlo LA, Morady F, de Buitleir M, Krol RB, Schurig L, Annesley TM. Effects of magnesium sulfate on cardiac conduction and refractoriness in humans. J Am Coll Cardiol. 1986;7(6):1356–1362. doi: 10.1016/s0735-1097(86)80157-7. [DOI] [PubMed] [Google Scholar]
- 32.Christiansen EH, Frost L, Andreasen F, Mortensen P, Thomsen PE, Pedersen AK. Dose-related cardiac electrophysiological effects of intravenous magnesium. A double-blind placebo-controlled dose-response study in patients with paroxysmal supraventricular tachycardia. Europace. 2000;2(4):320–326. doi: 10.1053/eupc.2000.0123. [DOI] [PubMed] [Google Scholar]
- 33.Onalan O, Crystal E, Daoulah A, Lau C, Crystal A, Lashevsky I. Meta-analysis of magnesium therapy for the acute management of rapid atrial fibrillation. Am J Cardiol. 2007;99(12):1726–1732. doi: 10.1016/j.amjcard.2007.01.057. [DOI] [PubMed] [Google Scholar]
- 34.Hazelrigg SR, Boley TM, Cetindag IB, Moulton KP, Trammell GL, Polancic JE, Shawgo TS, Quin JA, Verhulst S. The efficacy of supplemental magnesium in reducing atrial fibrillation after coronary artery bypass grafting. Ann Thorac Surg. 2004;77(3):824–830. doi: 10.1016/j.athoracsur.2003.08.027. [DOI] [PubMed] [Google Scholar]
- 35.Cook RC, Humphries KH, Gin K, Janusz MT, Slavik RS, Bernstein V, Tholin M, Lee MK. Prophylactic intravenous magnesium sulphate in addition to oral {beta}-blockade does not prevent atrial arrhythmias after coronary artery or valvular heart surgery: a randomized, controlled trial. Circulation. 2009;120(11 Suppl):S163–9. doi: 10.1161/CIRCULATIONAHA.108.841221. [DOI] [PubMed] [Google Scholar]
- 36.Henyan NN, Gillespie EL, White CM, Kluger J, Coleman CI. Impact of intravenous magnesium on post-cardiothoracic surgery atrial fibrillation and length of hospital stay: a meta-analysis. Ann Thorac Surg. 2005;80(6):2402–2406. doi: 10.1016/j.athoracsur.2005.03.036. [DOI] [PubMed] [Google Scholar]
- 37.Kalman JM, Munawar M, Howes LG, Louis WJ, Buxton BF, Gutteridge G, Tonkin AM. Atrial fibrillation after coronary artery bypass grafting is associated with sympathetic activation. Ann Thorac Surg. 1995;60(6):1709–1715. doi: 10.1016/0003-4975(95)00718-0. [DOI] [PubMed] [Google Scholar]
- 38.Workman AJ. Cardiac adrenergic control and atrial fibrillation. Naunyn Schmiedebergs Arch Pharmacol. 2010;381(3):235–249. doi: 10.1007/s00210-009-0474-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hogue CW, Hyder ML. Atrial fibrillation after cardiac operation: risks, mechanisms, and treatment. Ann Thorac Surg. 2000;69(1):300–306. doi: 10.1016/s0003-4975(99)01267-9. [DOI] [PubMed] [Google Scholar]
- 40.Jacob KA, Nathoe HM, Dieleman JM, van Osch D, Kluin J, van Dijk D. Inflammation in new-onset atrial fibrillation after cardiac surgery: a systematic review. Eur J Clin Invest. 2014;44(4):402–428. doi: 10.1111/eci.12237. [DOI] [PubMed] [Google Scholar]
- 41.Maesen B, Nijs J, Maessen J, Allessie M, Schotten U. Post-operative atrial fibrillation: a maze of mechanisms. Europace. 2012;14(2):159–174. doi: 10.1093/europace/eur208. [DOI] [PMC free article] [PubMed] [Google Scholar]