Abstract
Background
Cancer cachexia is a complex syndrome characterized by an ongoing loss of skeletal muscle mass and progressive functional impairment. A proactive management approach is recommended, including physical exercise to maintain function via modulation of muscle metabolism, insulin sensitivity and levels of inflammation. The review aimed to determine the safety, acceptability and effectiveness of exercise in adults with cancer cachexia. Secondary aims, subject to the data availability, were to compare effectiveness according to the characteristics of the study intervention or population.
Methods
We sought randomised controlled trials (RCTs) in adults meeting international criteria for cancer cachexia, comparing a programme of exercise as a sole or adjunct intervention to usual care or an active control. CENTRAL, MEDLINE, EMBASE, DARE and HTA, ISI Web of Science, LILACS, PEDro, SciVerse SCOPUS, Biosis Previews PreMEDLINE and Open Grey databases were searched up to June 2014. Two authors independently assessed studies for eligibility.
Results
We screened 3154 separate titles and abstracts, and reviewed 16 full-texts. Corresponding authors were contacted to determine if samples met cachexia staging criteria. Most authors did not explore this concept. No trial met review eligibility criteria. We were unable to perform a meta-analysis to determine any effects from exercise intervention.
Conclusion
Despite a strong rationale for the use of exercise, there is insufficient evidence to determine safety and effectiveness in patients with cancer cachexia. Findings from ongoing studies are awaited. Assessment of cachexia domains, ideally against international criteria, is required for future trials of exercise and supportive care interventions.
Keywords: Exercise, Cachexia, Cancer, Human, Physical activity
Introduction
Cancer cachexia is characterized by an ongoing loss of skeletal muscle mass, with or without a loss of fat mass, leading to progressive functional impairment.1 Prevalence varies according to cancer type, with the highest rates found for cancers of the lung and upper gastrointestinal tract, where over half of all patients are affected at diagnosis.1,2 Variation in the development of cancer cachexia also occurs on an individual level, with genetic polymorphisms influencing hallmarks of the syndrome, for example inflammation, loss of lean or fat mass..3 The pathophysiology of cancer cachexia is complex but involves a combination of abnormal metabolism and a negative energy balance.4 Functional consequences of cachexia include limiting symptoms,5 reduced muscle function,6,7 reduced exercise capacity,8 and physical inactivity,9 all of which impact adversely on a patient's level of independence (Figure 1).
Figure 1.
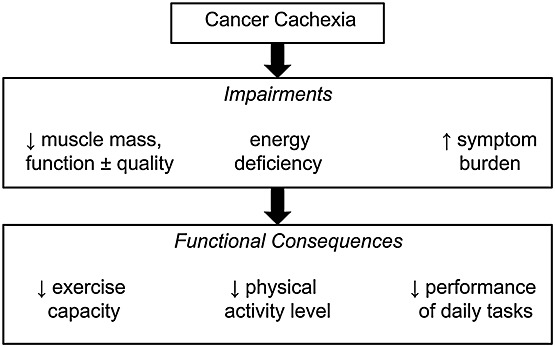
Functional impairments and consequences of cancer cachexia.
A proactive management approach is recommended, instigated before cachexia becomes refractory, with the aim to maintain or slow down the decline in physical function.1,4 Multimodal intervention is also considered necessary, as it is unlikely any single intervention will address the metabolic, nutritional, and functional deficits caused by cachexia concurrently.4 Three main component interventions are being developed: nutritional therapies, pharmacological agents, and exercise therapies.10
Exercise is defined as ‘a planned, structured and repetitive bodily movement done to maintain or improve one or more components of physical fitness’.11 Exercise programmes vary according to the frequency, intensity, volume, and type of training used, as well as contextual factors such as the setting and level of supervision. For example, both a 6 week hospital-based programme of twice weekly, high-intensity, treadmill walking, and a home-based programme of low-intensity, balance training throughout chemotherapy would be defined as exercise.11 Exercise may attenuate the effects of cancer cachexia via modulation of muscle metabolism, insulin sensitivity, and levels of inflammation.12,13 Exercise may help patients with or at risk of cancer cachexia to maintain their independence for longer, but empirical evidence is lacking. Despite a growing evidence base for nutritional and drug interventions for cancer cachexia,10 studies of exercise in advanced cancer are few. This paper provides an executive summary of a recent Cochrane Collaboration systematic review,14 which synthesizes evidence for the use of exercise for cancer cachexia.
The primary aim was to determine the effectiveness of exercise, compared with usual care or no treatment on lean muscle mass in adults with cancer. Secondary aim were to examine the acceptability and safety of exercise in this setting, and if possible, to compare effectiveness according to the characteristics of the exercise intervention or patient population.
Methods
Eligibility criteria
Criteria for review entry were randomized controlled trials or quasi-randomized studies in any setting involving adults (≥18 years of age) with a histological or clinical diagnosis of cancer, offered any type of exercising programme as a sole intervention or in combination with another intervention. We adopted an international criteria for cancer cachexia,1 and patients could be at any stage in the proposed classification:
Pre-cachexia; weight loss ≤5% with anorexia and metabolic changes
Cachexia; weight loss >5% in the past six months or body mass index (BMI) <20 kg/m2 and ongoing weight loss >2% or sarcopenia, anorexia or systemic inflammation
Refractory cachexia; active catabolism, ongoing weight loss, not responsive to treatment, and life expectancy of <3 months.
To include studies in populations relevant to the review, for example advanced cancer, but not explicitly performed to address cancer cachexia, we permitted studies where ≥50% of participants fell within the cachexia definitions. Where baseline demographic data were insufficient to assess participants against these criteria, we contacted study authors to seek additional data for this purpose.
Outcome measures
The primary endpoint was lean body mass assessed at the first study timepoint following a programme of exercise. Secondary outcomes included adherence to prescribed programmes, occurrence of adverse events and, subject to availability of data, muscle strength and endurance, maximal and submaximal exercise capacity, fatigue, and health-related quality of life.
Search strategy
We developed a comprehensive search strategy using a combination of terms based on the target population and intervention. The following electronic databases were searched from their inception until June 2014, CENTRAL, MEDLINE (Ovid), EMBASE (Ovid), DARE and HTA - Health Technology Assessments (on The Cochrane Library), ISI Web of Science (SCI-Expanded and CPCI), LILACS (Latin American and Caribbean Health Sciences), PEDro (the Physiotherapy Evidence Database), SciVerse SCOPUS; Biosis Previews PreMEDLINE, Open Grey (System for Information on Grey Literature).We identified ongoing studies using: Clinical Trials.gov; metaRegister of Controlled Trials (mRCT); International Clinical Trials Registry Platform (ICTRP); Pan African Clinical Trials; and the EU Clinical Trials Register. In addition, we hand-searched the following sources: proceedings from the Society on Sarcopenia, Cachexia and Wasting Disorders (SCWD); the American Cancer Society; the British Association for Cancer Research; and the European Clinical Guidelines. We checked reference lists and citation reports of studies review in full text. In addition, we contacted corresponding authors, experts, and organizations in the field to seek potentially relevant research, including unpublished and ongoing studies.
Data collection and analysis
Two review authors (A.J.G., V.S.) independently assess titles and abstracts of articles for relevance.14 We extracted relevant data relating to the study source, methods, participants, and intervention, Disagreements were discussed and resolved by consensus. We planned for two authors to independently assess each eligible study for risk of bias using the Cochrane Collaboration tool.15
Results
We identified 4786 references from the search. From a total of 3154 references after duplicates were removed, 3138 were deemed ineligible after title and abstract screening (Figure 2). Full texts of the remaining 16 potentially relevant titles were retrieved.14 We attempted to contact corresponding authors via electronic mail to determine the proportion of the sample meeting pre-cachexia or cachexia criteria. Most authors did not explore this concept, and others did not respond.14 We did not identify any suitable studies for inclusion. In the absence of any suitable trials, we were unable to perform a meta-analysis to determine any effects from exercise intervention.
Figure 2.

Review flow diagram.
Discussion
Using a comprehensive and systematic search strategy, we identified no randomized controlled trials of exercise for patients with cancer cachexia. In studies where exercise interventions were delivered to patients with cancer and at risk of cachexia, domains for the syndrome including weight loss, nutritional stores and intake, and muscle mass or function were seldom reported. To this end, it was not possible to determine the proportion of patients meeting pre-cachexia or cachexia criteria.
The lack of studies in this field is at odds with the strong rationale for examining exercise as a therapeutic intervention. Reviews have outlined the potential for exercise to impact positively on muscle mass and strength,16 inflammatory markers,13 and physical function,12 all of which are features of cancer cachexia which impairment the patient. It is plausible that the previous lack of a consensus definition has hindered study in the field. Without agreed diagnostic or staging criteria, the prevalence and impact of cancer cachexia have been difficult to assess. Moreover, interventions may have been inadvertently trialled in cachectic groups without formal recognition, as may be the case for exercise studies in advanced cancer. In this regard, the classification system proposed by Fearon et al.1 is an important step forward, and recent international validation studies will add further momentum.17–20
Two candidate studies may have included patients with cancer cachexia, although this cannot be confirmed. The first21 tested a multidisciplinary intervention with cognitive, emotional, social, and spiritual supportive care delivered over eight 90 min sessions. In 115 patients with advanced cancer undergoing radiation therapy, self-assessed physical well-being was improved in the intervention group, although no benefit in objectively measured physical function was observed. The second22 randomized 231 patients with incurable, metastatic cancer to physical exercise or usual care over 8 weeks. The exercise was delivered in two 60 min session each week, and led to improvements in handgrip strength and sit-to-stand performance. No adverse effects related to exercise were found in either study.
We also identified ongoing studies targeting cancer cachexia, most notably a randomized feasibility trial of a 6 week multimodal intervention comprising nutritional supplementation, home-based exercise, and anti-inflammatory treatment for patients with advanced non-small cell lung or pancreatic cancer (NCT01419145). Further ongoing studies are examining combined physical activity/exercise and nutrition interventions, over 12 weeks for patients with head and neck cancer during or following treatment (NCT01681654) and over 20 weeks for cachectic patients with lung cancer (ACTRN12611000870954). Findings from these trials are eagerly awaited and will pave the way for future exercise interventions for cachexia.
Conclusions
There is insufficient evidence to determine the safety and effectiveness of exercise for patients with cancer cachexia. Despite a strong rationale for the use of exercise, there are no present data from RCTs to elucidate the specific effects in this population. International criteria to diagnose and classify cancer cachexia provide a strong basis to define study populations within trials of interventions for this condition. An assessment of these domains must be included in cachexia trials and is strongly encouraged in populations where cachexia may be present. This would enhance the interpretation and generalization of findings from relevant study populations to the cachexia setting.
Acknowledgments
A.J.G. is funded by the Science Without Borders-Cnpq, Brazil. M.M. is supported by a National Institute for Health Research (NIHR) Clinical Trials Fellowship and the NIHR Collaboration for Leadership in Applied Health Research and Care (CLAHRC) for South London. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the NIHR nor the Department of Health.
Conflict of interest
Antonio Jose Grande, Valter Silva, and Matthew Maddocks declare that they have no conflict of interest.
Ethical guidelines
The authors certify that they comply with the ethical guidelines for authorship and publishing of the Journal of Cachexia, Sarcopenia and Muscle (von Haehling S, Morley JE, Coats AJS, Anker SD. Ethical guidelines for authorship and publishing in the Journal of Cachexia, Sarcopenia and Muscle. J Cachexia Sarcopenia Muscle. 2010;1:7–8.).
References
- 1.Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011;12:489–495. doi: 10.1016/S1470-2045(10)70218-7. [DOI] [PubMed] [Google Scholar]
- 2.Laviano A, Meguid MM, Inui A, Muscaritoli M, Rossi-Fanelli F. Therapy insight: cancer anorexia-cachexia syndrome—when all you can eat is yourself. Nat Clin Pract Oncol. 2005;2:158–165. doi: 10.1038/ncponc0112. [DOI] [PubMed] [Google Scholar]
- 3.Johns N, Tan BH, MacMillan M, Solheim TS, Ross JA, Baracos VE, et al. Genetic basis of interindividual susceptibility to cancer cachexia: selection of potential candidate gene polymorphisms for association studies. J Genet. 2014;93:893–916. doi: 10.1007/s12041-014-0405-9. [DOI] [PubMed] [Google Scholar]
- 4.Fearon K, Arends J, Baracos V. Understanding the mechanisms and treatment options in cancer cachexia. Nat Rev Clin Oncol. 2013;10:90–99. doi: 10.1038/nrclinonc.2012.209. [DOI] [PubMed] [Google Scholar]
- 5.Wilcock A, Maddocks M, Lewis M, England R, Manderson C. Symptoms limiting activity in cancer patients with breathlessness on exertion: ask about muscle fatigue. Thorax. 2008;63:91–92. doi: 10.1136/thx.2007.084665. [DOI] [PubMed] [Google Scholar]
- 6.Stephens NA, Gray C, MacDonald AJ, Tan BH, Gallagher IJ, Skipworth RJ, et al. Sexual dimorphism modulates the impact of cancer cachexia on lower limb muscle mass and function. Clin Nutr. 2012;31:499–505. doi: 10.1016/j.clnu.2011.12.008. [DOI] [PubMed] [Google Scholar]
- 7.Weber MA, Krakowski-Roosen H, Schröder L, Kinscherf R, Krix M, Kopp-Schneider A, et al. Morphology, metabolism, microcirculation, and strength of skeletal muscles in cancer-related cachexia. Acta Oncol. 2009;48:116–124. doi: 10.1080/02841860802130001. [DOI] [PubMed] [Google Scholar]
- 8.England R, Maddocks M, Manderson C, Wilcock A. Factors influencing exercise performance in thoracic cancer. Respir Med. 2012;106:294–299. doi: 10.1016/j.rmed.2011.11.002. [DOI] [PubMed] [Google Scholar]
- 9.Maddocks M, Wilcock A. Exploring physical activity level in patients with thoracic cancer: implications for use as an outcome measure. Support Care Cancer. 2012;20:1113–1116. doi: 10.1007/s00520-012-1393-z. [DOI] [PubMed] [Google Scholar]
- 10.Solheim TS, Laird BJ. Evidence base for multimodal therapy in cachexia. Curr Opin Support Palliat Care. 2012;6:424–431. doi: 10.1097/SPC.0b013e328359b668. [DOI] [PubMed] [Google Scholar]
- 11.Thompson WR, Gordon NF, Pescatello LS. ACSM's Guidelines for Exercise Testing and Prescription. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010. [Google Scholar]
- 12.Maddocks M, Murton AJ, Wilcock A. Therapeutic exercise in cancer cachexia. Crit Rev Oncog. 2012;17:285–292. doi: 10.1615/critrevoncog.v17.i3.60. [DOI] [PubMed] [Google Scholar]
- 13.Maddocks M, Jones LW, Wilcock A. Immunological and hormonal effects of exercise: implications for cancer cachexia. Curr Opin Support Palliat Care. 2013;7:376–382. doi: 10.1097/SPC.0000000000000010. [DOI] [PubMed] [Google Scholar]
- 14.Grande AJ, Silva V, Riera R, Medeiros A, Vitoriano SG, Peccin MS, et al. Exercise for cancer cachexia in adults. Cochrane Database Syst Rev. 2014;11 doi: 10.1002/14651858.CD010804.pub2. CD010804. [DOI] [PubMed] [Google Scholar]
- 15.Higgins JPT, Altman DG. Chapter 8: assessing risk of bias in included studies. In: Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. 2011. (updated March 2011]. The Cochrane Collaboration. Available from http://www.cochrane-handbook.org. [Google Scholar]
- 16.Stene GB, Helbostad JL, Balstad TR, Riphagen II, Kaasa S, Oldervoll LM. Effect of physical exercise on muscle mass and strength in cancer patients during treatment—a systematic review. Crit Rev Oncol Hematol. 2013;88:573–593. doi: 10.1016/j.critrevonc.2013.07.001. [DOI] [PubMed] [Google Scholar]
- 17.Cheville AL, Kollasch J, Vandenberg J, Shen T, Grothey A, Gamble G, et al. A home-based exercise program to improve function, fatigue, and sleep quality in patients with stage IV lung and colorectal cancer: a randomized controlled trial. J Pain Symptom Manage. 2013;45:811–821. doi: 10.1016/j.jpainsymman.2012.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Oldervoll LM, Loge JH, Paltiel H, Asp MB, Vidvei U, Wiken AN, et al. The effect of a physical exercise program in palliative care: a phase II study. J Pain Symptom Manage. 2006;31:421–430. doi: 10.1016/j.jpainsymman.2005.10.004. [DOI] [PubMed] [Google Scholar]
- 19.Martin L, Senesse P, Gioulbasanis I, Antoun S, Bozzetti F, Deans C, et al. Diagnostic criteria for the classification of cancer-associated weight loss. J Clin Oncol. 2015;33:90–99. doi: 10.1200/JCO.2014.56.1894. [DOI] [PubMed] [Google Scholar]
- 20.Prado CM, Lieffers JR, McCargar LJ, Reiman T, Sawyer MB, Martin L, et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol. 2008;9:629–635. doi: 10.1016/S1470-2045(08)70153-0. [DOI] [PubMed] [Google Scholar]
- 21.Cheville AL, Girardi J, Clark MM, Rummans TA, Pittelkow T, Brown P, et al. Therapeutic exercise during outpatient radiation therapy for advanced cancer: feasibility and impact on physical well-being. Am J Phys Med Rehabil. 2010;89:611–619. doi: 10.1097/PHM.0b013e3181d3e782. [DOI] [PubMed] [Google Scholar]
- 22.Oldervoll LM, Loge JH, Lydersen S, Paltiel H, Asp MB, Nygaard UV, et al. Physical exercise for cancer patients with advanced disease: a randomized controlled trial. Oncologist. 2011;16:1649–1657. doi: 10.1634/theoncologist.2011-0133. [DOI] [PMC free article] [PubMed] [Google Scholar]


