Abstract
Several factors can affect the perioperative immune function. We evaluated the effect of propofol and desflurane anesthesia on the surgery-induced immune perturbation in patients undergoing breast cancer surgery. The patients were randomly assigned to receive propofol (n = 20) or desflurane (n = 20) anesthesia. The total and differential white blood cell counts were determined with lymphocyte subpopulations before and 1 hr after anesthesia induction and at 24 hr postoperatively. Plasma concentrations of interleukin (IL)-2 and IL-4 were also measured. Both propofol and desflurane anesthesia preserved the IL-2/IL-4 and CD4+/CD8+ T cell ratio. Leukocytes were lower in the propofol group than in the desflurane group at 1 hr after induction (median [quartiles], 4.98 [3.87-6.31] vs. 5.84 [5.18-7.94] 103/µL) and 24 hr postoperatively (6.92 [5.54-6.86] vs. 7.62 [6.22-9.21] 103/µL). NK cells significantly decreased 1 hr after induction in the propofol group (0.41 [0.34-0.53] to 0.25 [0.21-0.33] 103/µL), but not in the desflurane group (0.33 [0.29-0.48] to 0.38 [0.30-0.56] 103/µL). Our findings indicate that both propofol and desflurane anesthesia for breast cancer surgery induce a favorable immune response in terms of preservation of IL-2/IL-4 and CD4+/CD8+ T cell ratio in the perioperative period. With respect to leukocytes and NK cells, desflurane anesthesia is associated with less adverse immune responses than propofol anesthesia during surgery for breast cancer. (Clinical trial registration at https://cris.nih.go.kr/cris number: KCT0000939)
Graphical Abstract
Keywords: Propofol, Desflurane, Immune System, Breast Neoplasms
INTRODUCTION
Breast cancer is the most commonly diagnosed cancer in the developed and developing countries and the leading cause of cancer mortality among women, most of which is attributable to recurrence and metastasis (1). Curative surgical resection remains the main treatment of breast cancer, but it is recognized that surgical resection may paradoxically create a critical window for cancer recurrence (2). Cancer surgery can induce an acute inflammatory response, related to local tissue damage and the shedding of malignant cells into the blood and lymphatic circulation despite meticulous surgical manipulation (3). Pre existing micrometastasis may be undetectable even in localized breast cancer (4). Accumulating evidence suggests that several factors during the perioperative period can affect the immune system (2,5,6,7). These include the surgery per se, pain, anxiety, hypothermia, blood transfusion, anesthetic technique, and anesthetic drugs. Impairment in the immune system may allow malignant cells to escape immunosurveillance and metastasize in the perioperative period (8) as well as increase the risk of postoperative complications, such as systemic inflammatory response syndrome, sepsis, and multi-organ failure (9).
Propofol and desflurane are widely used anesthetics for general anesthesia. Desflurane is the most recently introduced inhalation agent and facilitates a fast-track recovery when compared with other inhalation agents. It allows the patient to resume earlier normal activities after surgery (10,11). It has been reported that intravenous and volatile anesthetics produce different immune system effect. Several studies have demonstrated that propofol has a more favorable immunomodulating profile than other anesthetic agents (12,13). On the other hand, isoflurane and sevoflurane, volatile anesthetics, have been reported to suppress natural killer (NK) function and increase proinflammatory cytokines more than propofol (13,14,15,16). Moreover, there have been few investigations of the effect of desflurane on the immune system (17,18).
Leukocytes, including granulocytes and lymphocytes, are the cellular mediators of immune function. CD4+ T cells include type-1 helper T (Th1) and type-2 helper T (Th2) cells. Th1 cells secrete cytokines such as interferon γ, interleukin (IL-2), and tumor necrosis factor, which cooperate with cytotoxic CD8+ T cells. In contrast, Th2 cells are associated with IL-4, IL-6, IL-10, and IL-13 and promote a humoral, protumorigenic response (19). A decrease in the balance of the Th1/Th2 ratio is associated with suppression of cell-mediated immunity (CMI) and has been the focus of many previous studies on perioperative immune function (17,20,21).
The objective of this prospective, randomized clinical study was to compare the effect of propofol and desflurane on perioperative immune cell populations in patients undergoing breast cancer surgery. We examined perioperative total and differential white blood cell counts, lymphocyte subsets, including CD4+ T cells, CD8+ T cells, and NK cells, and plasma concentrations of IL-2 (Th1 cytokine) and IL-4 (Th2 cytokine) in patients undergoing primary breast cancer surgery. We hypothesized that propofol would induce less adverse immune effects, as indicated by preservation of the Th1/Th2 ratio and attenuation of the NK cell count decrease.
MATERIALS AND METHODS
We studied female patients aged 18 to 85 yr, American Society of Anesthesiologists (ASA) physical status I-III, undergoing mastectomy, axillary node dissection or wide local tumor excision at Ewha Womans University Hospital. Exclusion criteria were previous breast cancer surgery (except diagnostic biopsy), inflammatory breast cancer, evidence of systemic infections (clinically defined or elevated C-reactive protein levels, leukocytosis, body temperature ≥37.8℃) and any contraindication to drugs used in this study.
On arrival at the operating room, standard ASA monitors were used throughout the surgery. The patients were assigned randomly to receive propofol (n=20) or desflurane (n=20) anesthesia using a computer-generated randomization table. In the propofol group, anesthesia was induced with propofol 1.5-2 mg/kg and fentanyl 1-2 µg/kg and maintained with a continuous infusion of propofol 4-8 mg/kg/h. In the desflurane group, anesthesia was induced with thiopental sodium 4-5 mg/kg and fentanyl 1-2 µg/kg and maintained with inhalation of desflurane at an end-tidal concentration of 3%-7%. Propofol and desflurane were titrated to maintain the bispectral index (BIS) value between 40 and 60.
In all patients, tracheal intubation was facilitated by administration of rocuronium 0.6 mg/kg and the lungs were ventilated with a mixture of 1:1 O2/N2O. A continuous infusion of vecuronium 0.02 mg/kg/hr was performed for muscle relaxation and a bolus of fentanyl 0.5-1 µg/kg was given as needed. Postoperative analgesia was provided with non-steroidal anti-inflammatory drugs and tramadol on request. Pain scores at 12 and 24 hr postoperatively were evaluated. The standardized protocol for the surgical procedure and postoperative patient care was used to minimize any difference in surgical impact between the randomized groups.
Sampling
Venous blood samples were collected before and 1 hr after anesthesia induction and at 24 hr postoperatively. The blood samples were collected in ethylenediamine tetraacetic acid (EDTA) tubes for blood cell counting and lymphocyte analysis and in a serum separator tube (SST) for cytokine concentration testing. Samples in SSTs were centrifuged (3,000 g, 10 min) and the resulting serum was stored at -20℃ until analysis.
Blood cell counts
A complete blood count was performed on all blood samples using an automated hemoanalyzer. Total and differential leukocyte counts were determined.
Flow cytometry
Lymphocyte subsets were analyzed by flow cytometry (Cytomics FC500, Beckman Coulter, Inc., FL, USA). The following antibodies (Beckman-Coulter, Inc., Marseille, France) to lymphocyte antigens were used to determine cell types: CD3-FITC/CD4-PE (helper T cells, CD4+ T cells), CD3-FITC/CD8-PE (suppressor/cytotoxic T cells, CD8+ T cells), and CD3-FITC/CD16+ CD56-PE (NK cells).
Cytokine measurements
Plasma concentrations of IL-2 and IL-4 were measured with an enzyme-linked immunosorbent assay (ELISA) using a commercially available kit (Human IL-2 and IL-4 Quantikine Elisa Kit, R&D Systems, Inc., USA) according to protocols provided. The assays were read using an ELISA microplate reader (Spectra Max 190, Molecular Devices, Sunnyvale, CA, USA). We determined the Th1/Th2 balance by calculating the IL-2/IL-4 ratio.
Statistical analysis
SPSS (ver. 18.0, Chicago, IL, USA) was used for the statistical analysis. The primary endpoint was the Th1/Th2 ratio (IL-2/IL-4) on the first postoperative day. Using a two-sided design at a significance level of 5% with a probability of 85%, an estimated 19 patients per group were needed to detect an effect size based on previous data with SD approximately 0.8 (22). Continuous variables were analysed by student's t-tests or Mann Whitney U-test after assessment for normality. Discrete variables were analysed using the chi-squared test or Fisher's exact test. Data of white blood cells, lymphocyte subsets counts and cytokines were analyzed by repeated measures analysis of variance (ANOVA) followed by Bonferroni correction. Mann Whitney U-test was used to analyse a difference between groups. Results are presented as the means±standard deviations (SD) or as medians with interquartile ranges (IQR) as appropriate. It was considered to be statistically significant if P<0.05.
Ethics statement
This study was approved by the institutional review board (IRB) of Ewha Womans Hospital (IRB No. ECT 13-29A-25). Written informed consent was obtained from all participating subjects. This trial was registered at https://cris.nih.go.kr/cris (Identifier number: KCT0000939).
RESULTS
The numbers of participants screened, enrolled, and who dropped out are shown in Fig. 1. This study was performed between September and December 2013. In total, 40 patients completed the study, 20 in each group. There was no significant difference between the groups in terms of age, height, weight, ASA class, anesthesia, and operation time, type of surgery, and TNM stage according to the American Joint Committee on Cancer criteria. Total consumption of fentanyl and pain score by numeric rating scale at 12 and 24 hr postoperatively were similar between the groups (Table 1). There was no difference in analgesic use during 24 hr postoperative between groups (data not shown).
Fig. 1. Study flow chart.
Table 1. Demographic and perioperative characteristics.
| Parameters | Propofol (n = 20) | Desflurane (n = 20) |
|---|---|---|
| Age (yr) | 50.00 ± 11.83 | 50.45 ± 9.37 |
| Weight (kg) | 55.80 ± 5.98 | 59.90 ± 7.91 |
| Height (cm) | 157.90 ± 5.37 | 158.40 ± 6.10 |
| ASA physical status (I/II/III) | 12/8/0 | 14/6/0 |
| Crystalloid infusion (mL) | 765.00 ± 188.56 | 785.00 ± 219.51 |
| Blood loss (mL) | 35.00 ± 26.46 | 44.50 ± 25.23 |
| Duration of anesthesia (min) | 178.00 ± 43.66 | 201.25 ± 54.14 |
| Duration of surgery (min) | 137.50 ± 42.04 | 164.00 ± 50.90 |
| Diagnosis | ||
| Intraductal carcinoma | 16 | 19 |
| Intraductal papilloma | 2 | 0 |
| DCIS | 2 | 1 |
| Stage (0/I/II/III)* | 0/13/6/1 | 1/10/6/2 |
| Operation | ||
| Mastectomy with axillary node clearance | 1 | 4 |
| Breast conserving surgery with sentinel node biopsy | 19 | 16 |
| Intraoperative fentanyl consumption (µg) | 175 ± 61 | 145 ± 42 |
| Pain score, 12 hr postoperatively | 1.90 ± 0.72 | 1.55 ± 0.10 |
| Pain score, 24 hr postoperatively | 0.25 ± 0.55 | 0.05 ± 0.22 |
*Stage by American Joint Committee on cancer staging system. ASA, American Society of Anesthesiologists; DCIS, ductal carcinoma in situ.
Plasma concentrations of IL-2 and IL-4 were not changed significantly after anesthesia induction. The IL-2/IL-4 ratio also showed no significant change between the groups (Table 2).
Table 2. Perioperative IL-2/IL-4 ratio.
| Cytokines | Propofol | Desflurane | ||||
|---|---|---|---|---|---|---|
| Baseline | 1 hr | 24 hr | Baseline | 1 hr | 24 hr | |
| IL-2 (pg/mL) | 20.45 (19.22-22.98) | 20.12 (19.23-20.08) | 20.37 (19.68-23.20) | 24.09 (22.52-26.98) | 23.23 (21.46-27.97) | 22.52 (20.59-25.03) |
| IL-4 (pg/mL) | 11.54 (11.09-12.19) | 11.68 (11.64-13.06) | 11.45 (11.06-12.19) | 12.66 (12.19-13.14) | 12.34 (11.64-13.06) | 12.50 (11.95-13.39) |
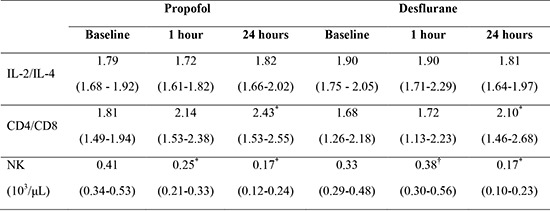
| IL-2/IL-4 | 1.79 (1.68-1.92) | 1.72 (1.61-1.82) | 1.82 (1.66-2.02) | 1.90 (1.75 - 2.05) | 1.90 (1.71-2.29) | 1.81 (1.64-1.97) |
Values are expressed as median (interquartile range).
Leukocytes revealed a decrease at 1 hr after induction only in the propofol group, with borderline significance (P=0.05) and were lower in the propofol group than in the desflurane group at 1 hr after induction and 24 hr postoperatively. They increased in all patients at 24 hr postoperatively, largely as a result of increased neutrophils, which was statistically significant only in the desflurane group. Lymphocytes in the propofol group were lower than those in the desflurane group 1 hr after induction (P=0.019). Monocytes at 24 hr increased versus baseline values in the desflurane group, with a significant difference between the groups. Neutrophils at 24 hr increased, compared with baseline values, in both groups with a difference between groups (Table 3).
Table 3. The counts of total leukocytes, lymphocytes, monocytes, and neutrophils in patients undergoing surgery under propofol and desflurane anesthesia.
| Cell counts | Propofol | Desflurane | ||||
|---|---|---|---|---|---|---|
| Baseline | 1 hr | 24 hr | Baseline | 1 hr | 24 hr | |
| Leukocytes (103/µL) | 5.50 (4.61-6.60) | 4.98 (3.87-6.31) | 6.92 (5.54-6.86) | 6.11 (4.55-7.15) | 5.84† (5.18-7.94) | 7.62*,† (6.22-9.21) |
| Lymphocytes (103/µL) | 2.03 (1.59-2.21) | 1.65 (1.32-2.04) | 1.58 (1.33-1.91) | 1.74 (1.64-2.26) | 2.14† (1.96-2.44) | 1.68 (1.39-1.90) |
| Monocytes (103/µL) | 0.30 (0.27-0.46) | 0.34 (0.28-0.40) | 0.47 (0.38-0.59) | 0.42 (0.36-0.48) | 0.49† (0.34-0.61) | 0.59*,† (0.47-0.77) |
| Neutrophils (103/µL) | 2.87 (2.34-3.88) | 2.78 (2.07-3.58) | 4.54* (3.52-5.49) | 2.90 (2.52-4.78) | 2.95 (2.57-4.09) | 5.43*,† (4.42-6.04) |
Values are expressed as median (interquartile range). *P<0.05 vs. preoperative value; †P<0.05 vs. propofol group.
As shown in Table 4, CD4+ and CD8+ T cells showed no statistically significant change after anesthesia induction in either group. The CD4+/CD8+ T cell ratio remained unchanged at 1 hr after induction and then increased at 24 hr in both groups (P<0.001). NK cells decreased significantly 1 hr after induction in the propofol group (P=0.040), but not in the desflurane group. At 24 hr postoperative, NK cell counts decreased further in all patients (P<0.001) with no difference between groups. There was no adverse event related to anesthesia or surgery during the study period.
Table 4. The counts of CD4+ T cell, CD8+ T cell and NK cell in patients undergoing surgery under propofol and desflurane anesthesia.
| Cells | Propofol | Desflurane | ||||
|---|---|---|---|---|---|---|
| Baseline | 1 hr | 24 hr | Baseline | 1 hr | 24 hr | |
| CD4 (103/µL) | 0.78 (0.53-1.00) | 0.79 (0.52-0.91) | 0.76 (0.53-0.91) | 0.73 (0.62-0.80) | 0.87 (0.72-1.06) | 0.79 (0.64-1.06) |
| CD8 (103/µL) | 0.45 (0.30-0.57) | 0.38 (0.28-0.52) | 0.38 (0.24-0.49) | 0.43 (0.33-0.48) | 0.46 (0.40-0.64) | 0.36 (0.28-0.44) |
| CD4/CD8 | 1.81 (1.49-1.94) | 2.14 (1.53-2.38) | 2.43* (1.53-2.55) | 1.68 (1.26-2.18) | 1.72 (1.13-2.23) | 2.10* (1.46-2.68) |
| NK (103/µL) | 0.41 (0.34-0.53) | 0.25* (0.21-0.33) | 0.17* (0.12-0.24) | 0.33 (0.29-0.48) | 0.38† (0.30-0.56) | 0.17* (0.10-0.23) |
Values are expressed as median (interquartile range). *P<0.05 vs. preoperative value; †P<0.05 vs. propofol group.
DISCUSSION
Whether anesthesia and surgery affect immune function is an issue for breast cancer patients undergoing cancer surgery, and the least possible immunosuppressive regimen would be beneficial for them. Our results show that surgery with both anesthetic agents preserve the IL-2/IL-4 ratio in the perioperative period, although desflurane anesthesia is even less associated with a decrease in leukocyte and NK cell counts at 1 hr after induction, when compared to propofol anesthesia.
IL-2 and IL-4 are associated with Th1 and Th2 cells, respectively (21,23). As a shift from a Th1 to Th2 response appears to be related with infectious complications and cancer progression, the balance between Th1 and Th2 cell has the significance in those patients with cancer (24,25,26). An increased Th1/Th2 cell ratio indicates enhanced CMI and would be potentially beneficial for cancer elimination, while a decreased ratio would raise the risk of cancer progression. In an animal study using mice, Hiroki et al. (27) demonstrated that combined spinal and sevoflurane general anesthesia preserved the Th1/Th2 balance with a resulting reduction in liver metastasis, whereas general anesthesia alone reduced the Th1/Th2 ratio. In another study in craniotomy patients, propofol anesthesia preserved the Th1/Th2 ratio, while isoflurane anesthesia reduced the Th1/Th2 ratio significantly (22). The authors concluded that propofol anesthesia attenuated the adverse immune effects related with surgical stress when compared to isoflurane anesthesia. Our results showed that the IL-2/IL-4 ratio was not significantly changed after propofol or desflurane anesthesia induction. The CD4+/CD8+ T cell ratio has been regarded as an important index of CMI (28). In our patients, it remained unchanged at 1 hr after induction and then even increased, compared with baseline values. A significant decrease in lymphocytes and monocytes, which are usually found in changes of the immune system after surgery, also did not occur in our patients. From this, desflurane is considered to provide a favorable effect on the immune system, at least as much as propofol anesthesia, which is known not to have major adverse effect on immune system.
With respect to NK cells and leukocytes, desflurane anesthesia showed more favorable effect on the immune system than propofol anesthesia. CMI consists of two immune system components, the innate and adaptive systems (19). The innate immune system is a non-specific system, including macrophages, NK cells, and dendritic cells. NK cells play a key role in destroying tumors as a first-line of defense against cancer cells. NK cell activity in the perioperative period has been reported to be inversely related to the development of metastasis (26). In the present study, NK cells decreased significantly 1 hr after induction only in the propofol group, suggesting the possibility of somewhat impaired innate immunity. Leukocytes decreased only in the propofol group at 1 hr after induction and were lower in propofol group than in the desflurane group at 1 hr after induction and 24 hr postoperatively.
NK cells decreased significantly in all patients at 24 hr postoperatively. This finding is consistent with Liu et al. (23) regarding immune cell populations in craniotomy patients. They reported that a decrease in immune cell populations was most obvious early after anesthesia induction. Most immune cells, such as leukocytes, total lymphocytes, T cells, and B cells decreased to their minimum levels after anesthesia induction and recovered by the end of surgery, while NK cells remained at a decreased level.
Several studies have reported markedly different impacts of propofol and desflurane after surgery. Schilling et al. (17) investigated proinflammatory reactions using bronchoalveolar lavage during one-lung ventilation (OLV) and concluded that desflurane anesthesia alleviated alveolar inflammatory responses to OLV, compared with propofol anesthesia. Another study showed that desflurane anesthesia produced less inflammatory response than propofol anesthesia, in terms of lower plasma concentrations of IL-6 and IL-8, after cardiopulmonary bypass (18).
The present study has several limitations. First, although the anesthetic drugs may have caused complex effects in cancer cell biology, in terms of cancer proliferation, migration, invasion, and interference with the CMI system (6), we did not evaluate actual long-term clinical outcomes of the cancers, the interactions between immune cells and surrounding factors in these individuals. Second, we used fentanyl to provide analgesia, which is known to have immunomodulating effects, which might also have influenced our findings. Experimental studies concerning the effects of opioids on immune function have shown conflicting results (29,30,31) and clinical evidence is lacking. Third, we may be criticized for the use of nitrous oxide with respect to hematologic, myocardial, neurological, and immunological concerns (32). A randomized controlled trial in colorectal cancer patients demonstrated that cancer recurrence risks were similar in those who were assigned to 65% nitrous oxide or nitrogen during cancer surgery (33). We used nitrous oxide because of familiarity and ease of use with regard to low blood/gas partition coefficients, its own anesthetic properties, and second gas effects. We sought to compare the effects of propofol and desflurane in the same condition so we used nitrous oxide in both groups. Further work without the use of nitrous oxide might show different results.
In conclusion, surgery with both propofol and desflurane reveals favorable effect on immune system in terms of IL-2/IL-4 and CD4+/CD8+ T cell ratio in the perioperative period. With respect to leukocytes and NK cells, desflurane anesthesia is even less associated with adverse effect on immune system than propofol anesthesia during surgery for breast cancer. Further studies are needed to determine the clinical implications of anesthetic drug choices.
Footnotes
DISCLOSURE: The authors declare that no potential conflicts of interest exist with any companies. Our research including data analysis was not influenced by a pharmaceutical company offering research grants.
AUTHOR CONTRIBUTION: Conception and coordination of the study: Baik HJ, Kim CH. Design of ethical issues: Woo JH, Baik HJ. Acquisition of data: Chung RK, Kim DY, Lee GY, Chun EH. Statistical analysis: Woo JH, Baik HJ. Manuscript preparation: Woo JH, Baik HJ, Chun EH. Manuscript approval: all authors.
References
- 1.Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. doi: 10.3322/caac.20107. [DOI] [PubMed] [Google Scholar]
- 2.Tavare AN, Perry NJ, Benzonana LL, Takata M, Ma D. Cancer recurrence after surgery: direct and indirect effects of anesthetic agents. Int J Cancer. 2012;130:1237–1250. doi: 10.1002/ijc.26448. [DOI] [PubMed] [Google Scholar]
- 3.Tracey KJ. Reflex control of immunity. Nat Rev Immunol. 2009;9:418–428. doi: 10.1038/nri2566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mansi JL, Gogas H, Bliss JM, Gazet JC, Berger U, Coombes RC. Outcome of primary-breast-cancer patients with micrometastases: a long-term follow-up study. Lancet. 1999;354:197–202. doi: 10.1016/s0140-6736(98)10175-7. [DOI] [PubMed] [Google Scholar]
- 5.Heaney A, Buggy DJ. Can anaesthetic and analgesic techniques affect cancer recurrence or metastasis? Br J Anaesth. 2012;109:i17–i28. doi: 10.1093/bja/aes421. [DOI] [PubMed] [Google Scholar]
- 6.Niwa H, Rowbotham DJ, Lambert DG, Buggy DJ. Can anesthetic techniques or drugs affect cancer recurrence in patients undergoing cancer surgery? J Anesth. 2013;27:731–741. doi: 10.1007/s00540-013-1615-7. [DOI] [PubMed] [Google Scholar]
- 7.Green JS, Tsui BC. Impact of anesthesia for cancer surgery: continuing professional development. Can J Anaesth. 2013;60:1248–1269. doi: 10.1007/s12630-013-0037-1. [DOI] [PubMed] [Google Scholar]
- 8.Neeman E, Ben-Eliyahu S. Surgery and stress promote cancer metastasis: new outlooks on perioperative mediating mechanisms and immune involvement. Brain Behav Immun. 2013;30:S32–S40. doi: 10.1016/j.bbi.2012.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wakefield CH, Carey PD, Foulds S, Monson JR, Guillou PJ. Changes in major histocompatibility complex class II expression in monocytes and T cells of patients developing infection after surgery. Br J Surg. 1993;80:205–209. doi: 10.1002/bjs.1800800224. [DOI] [PubMed] [Google Scholar]
- 10.White PF, Tang J, Wender RH, Yumul R, Stokes OJ, Sloninsky A, Naruse R, Kariger R, Norel E, Mandel S, et al. Desflurane versus sevoflurane for maintenance of outpatient anesthesia: the effect on early versus late recovery and perioperative coughing. Anesth Analg. 2009;109:387–393. doi: 10.1213/ane.0b013e3181adc21a. [DOI] [PubMed] [Google Scholar]
- 11.Mahmoud NA, Rose DJ, Laurence AS. Desflurane or sevoflurane for gynaecological day-case anaesthesia with spontaneous respiration? Anaesthesia. 2001;56:171–174. doi: 10.1046/j.1365-2044.2001.01528.x. [DOI] [PubMed] [Google Scholar]
- 12.Kushida A, Inada T, Shingu K. Enhancement of antitumor immunity after propofol treatment in mice. Immunopharmacol Immunotoxicol. 2007;29:477–486. doi: 10.1080/08923970701675085. [DOI] [PubMed] [Google Scholar]
- 13.Melamed R, Bar-Yosef S, Shakhar G, Shakhar K, Ben-Eliyahu S. Suppression of natural killer cell activity and promotion of tumor metastasis by ketamine, thiopental, and halothane, but not by propofol: mediating mechanisms and prophylactic measures. Anesth Analg. 2003;97:1331–1339. doi: 10.1213/01.ANE.0000082995.44040.07. [DOI] [PubMed] [Google Scholar]
- 14.Müller-Edenborn B, Roth-Z'graggen B, Bartnicka K, Borgeat A, Hoos A, Borsig L, Beck-Schimmer B. Volatile anesthetics reduce invasion of colorectal cancer cells through down-regulation of matrix metalloproteinase-9. Anesthesiology. 2012;117:293–301. doi: 10.1097/ALN.0b013e3182605df1. [DOI] [PubMed] [Google Scholar]
- 15.Ke JJ, Zhan J, Feng XB, Wu Y, Rao Y, Wang YL. A comparison of the effect of total intravenous anaesthesia with propofol and remifentanil and inhalational anaesthesia with isoflurane on the release of pro- and anti-inflammatory cytokines in patients undergoing open cholecystectomy. Anaesth Intensive Care. 2008;36:74–78. doi: 10.1177/0310057X0803600113. [DOI] [PubMed] [Google Scholar]
- 16.Tylman M, Sarbinowski R, Bengtson JP, Kvarnström A, Bengtsson A. Inflammatory response in patients undergoing colorectal cancer surgery: the effect of two different anesthetic techniques. Minerva Anestesiol. 2011;77:275–282. [PubMed] [Google Scholar]
- 17.Schilling T, Kozian A, Kretzschmar M, Huth C, Welte T, Bühling F, Hedenstierna G, Hachenberg T. Effects of propofol and desflurane anaesthesia on the alveolar inflammatory response to one-lung ventilation. Br J Anaesth. 2007;99:368–375. doi: 10.1093/bja/aem184. [DOI] [PubMed] [Google Scholar]
- 18.Baki ED, Aldemir M, Kokulu S, Koca HB, Ela Y, Sivaci RG, öztürk NK, Emmiler M, Adali F, Uzel H. Comparison of the effects of desflurane and propofol anesthesia on the inflammatory response and s100beta protein during coronary artery bypass grafting. Inflammation. 2013;36:1327–1333. doi: 10.1007/s10753-013-9671-6. [DOI] [PubMed] [Google Scholar]
- 19.Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity's roles in cancer suppression and promotion. Science. 2011;331:1565–1570. doi: 10.1126/science.1203486. [DOI] [PubMed] [Google Scholar]
- 20.Decker D, Schondorf M, Bidlingmaier F, Hirner A, von Ruecker AA. Surgical stress induces a shift in the type-1/type-2 T-helper cell balance, suggesting down-regulation of cell-mediated and up-regulation of antibody-mediated immunity commensurate to the trauma. Surgery. 1996;119:316–325. doi: 10.1016/s0039-6060(96)80118-8. [DOI] [PubMed] [Google Scholar]
- 21.Cata JP, Bauer M, Sokari T, Ramirez MF, Mason D, Plautz G, Kurz A. Effects of surgery, general anesthesia, and perioperative epidural analgesia on the immune function of patients with non-small cell lung cancer. J Clin Anesth. 2013;25:255–262. doi: 10.1016/j.jclinane.2012.12.007. [DOI] [PubMed] [Google Scholar]
- 22.Inada T, Yamanouchi Y, Jomura S, Sakamoto S, Takahashi M, Kambara T, Shingu K. Effect of propofol and isoflurane anaesthesia on the immune response to surgery. Anaesthesia. 2004;59:954–959. doi: 10.1111/j.1365-2044.2004.03837.x. [DOI] [PubMed] [Google Scholar]
- 23.Liu S, Wang B, Li S, Zhou Y, An L, Wang Y, Lv H, Zhang G, Fang F, Liu Z, et al. Immune cell populations decrease during craniotomy under general anesthesia. Anesth Analg. 2011;113:572–577. doi: 10.1213/ANE.0b013e3182278237. [DOI] [PubMed] [Google Scholar]
- 24.Raman D, Baugher PJ, Thu YM, Richmond A. Role of chemokines in tumor growth. Cancer Lett. 2007;256:137–165. doi: 10.1016/j.canlet.2007.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Han YF, Zhao J, Ma LY, Yin JH, Chang WJ, Zhang HW, Cao GW. Factors predicting occurrence and prognosis of hepatitis-B-virus-related hepatocellular carcinoma. World J Gastroenterol. 2011;17:4258–4270. doi: 10.3748/wjg.v17.i38.4258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Snyder GL, Greenberg S. Effect of anaesthetic technique and other perioperative factors on cancer recurrence. Br J Anaesth. 2010;105:106–115. doi: 10.1093/bja/aeq164. [DOI] [PubMed] [Google Scholar]
- 27.Wada H, Seki S, Takahashi T, Kawarabayashi N, Higuchi H, Habu Y, Sugahara S, Kazama T. Combined spinal and general anesthesia attenuates liver metastasis by preserving TH1/TH2 cytokine balance. Anesthesiology. 2007;106:499–506. doi: 10.1097/00000542-200703000-00014. [DOI] [PubMed] [Google Scholar]
- 28.Hori Y, Ibuki T, Hosokawa T, Tanaka Y. The effects of neurosurgical stress on peripheral lymphocyte subpopulations. J Clin Anesth. 2003;15:1–8. doi: 10.1016/s0952-8180(02)00455-5. [DOI] [PubMed] [Google Scholar]
- 29.Yeager MP, Procopio MA, DeLeo JA, Arruda JL, Hildebrandt L, Howell AL. Intravenous fentanyl increases natural killer cell cytotoxicity and circulating CD16(+) lymphocytes in humans. Anesth Analg. 2002;94:94–99. doi: 10.1097/00000539-200201000-00018. [DOI] [PubMed] [Google Scholar]
- 30.Beilin B, Shavit Y, Hart J, Mordashov B, Cohn S, Notti I, Bessler H. Effects of anesthesia based on large versus small doses of fentanyl on natural killer cell cytotoxicity in the perioperative period. Anesth Analg. 1996;82:492–497. doi: 10.1097/00000539-199603000-00011. [DOI] [PubMed] [Google Scholar]
- 31.Yardeni IZ, Beilin B, Mayburd E, Alcalay Y, Bessler H. Relationship between fentanyl dosage and immune function in the postoperative period. J Opioid Manag. 2008;4:27–33. doi: 10.5055/jom.2008.0005. [DOI] [PubMed] [Google Scholar]
- 32.Sanders RD, Weimann J, Maze M. Biologic effects of nitrous oxide: a mechanistic and toxicologic review. Anesthesiology. 2008;109:707–722. doi: 10.1097/ALN.0b013e3181870a17. [DOI] [PubMed] [Google Scholar]
- 33.Fleischmann E, Marschalek C, Schlemitz K, Dalton JE, Gruenberger T, Herbst F, Kurz A, Sessler DI. Nitrous oxide may not increase the risk of cancer recurrence after colorectal surgery: a follow-up of a randomized controlled trial. BMC Anesthesiol. 2009;9:1. doi: 10.1186/1471-2253-9-1. [DOI] [PMC free article] [PubMed] [Google Scholar]