Abstract
Objective
Diabetic complications and lipid peroxidation are known to have a close association. Lipid peroxidation commonly occurs at sites exposed to ischaemia, but distant organs and tissues also get damaged during ischaemia/reperfusion (I/R). Some of these targets are vital organs, such as the lung, liver, and kidney; the lung is the most frequently affected. The aim of our study was to investigate the effects of dexmedetomidine on I/R damage in lung tissue and on the oxidant/anti-oxidant system in diabetic rats.
Material and methods
Diabetes was induced with streptozotocin (55 mg/kg) in 18 Wistar Albino rats, which were then randomly divided into three groups (diabetes control (DC), diabetes plus ischaemia-reperfusion (DIR), and diabetes plus dexmedetomidine-ischaemia/reperfusion (DIRD)) after the effects of diabetes were clearly evident. The rats underwent a left thoracotomy and then ischaemia was produced in the myocardium muscle by a left anterior descending artery ligation for 30 min in the DIR and DIRD groups. I/R was performed for 120 min. The DIRD group received a single intraperitoneal dose of dexmedetomidine (100 µg/kg); the DIR group received no dexmedetomidine. Group DC was evaluated as the diabetic control group and also included six rats (C group) in which diabetes was not induced. These mice underwent only left thoracotomy and were closed without undergoing myocardial ischaemia. Histopathological changes, activities of catalase (CAT) and glutathione-S-transferase anti-oxidant enzymes, and malondialdehyde (MDA) levels were evaluated in the lung tissues of all rats.
Results
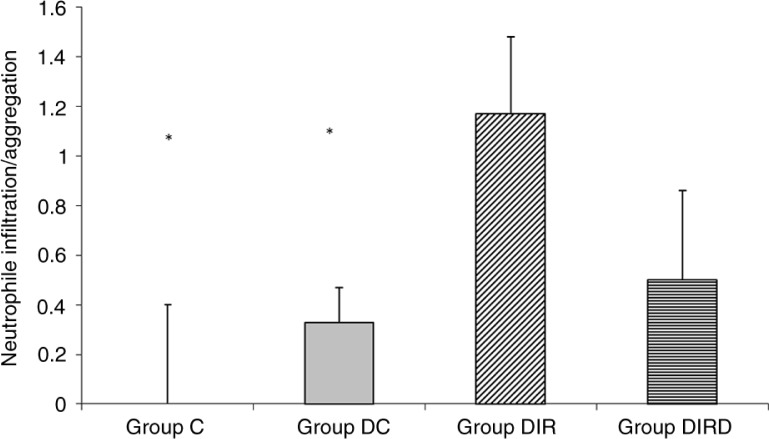
Neutrophil infiltration/aggregation was higher in the DIR group than in the C, DC, and DIRD groups (p=0.001, p=0.013, and p=0.042, respectively). The lung injury score was significantly higher in the DIR group than in the C and DC groups (p<0.0001 and p=0.024, respectively). The levels of MDA were significantly higher in the DIR group than in the C and DIRD groups. CAT activity was significantly higher in the DIR group than in the DIRD and C groups.
Conclusion
Our results confirm that dexmedetomidine has protective effects against the lung damage resulting from I/R in diabetic rats. Future studies conducted to evaluate the effects of the use of dexmedetomidine on damage to various organs following different I/R durations may help understanding possible protective effects of dexmedetomidine and underlying mechanisms in tissue damage related to I/R injury.
Keywords: Diabetes Mellitus, dexmedetomidine, ischaemia reperfusion, lung
Myocardial tissue ischaemia occurs due to perfusion loss, but it also arises in the microcirculation and distant organs because of reactive oxygen radicals generated during reperfusion and via the systemic inflammatory response (1, 2). Free oxygen radicals released from the ischaemic myocardium may cause damage to many distant organs such as lung, kidney, intestines, and pancreas. Oxidative stress and lipid peroxidation play important roles in distant organ damage after ischaemia/reperfusion (I/R) (3). Lipid peroxidation seen in diabetic patients further increases the damage to distant organs following reperfusion of the myocardium exposed to ischaemia (4). Among the distant organs, indirect damage seen after I/R is defined best in the lung (5) and is associated with these mechanisms. Therefore, acute lung injury may indirectly develop during and after pulmonary oedema.
Dexmedetomidine hydrochloride is an α2 agonist that has been used for sedation in anaesthesia in intensive care units (6). Besides its sedative effect, dexmedetomidine hydrochloride also has confirmed analgaesic and anxiolytic properties. Recent studies have shown that dexmedetomidine decreased the systemic inflammatory response, inhibited the release of inflammatory cytokines, and suppressed pulmonary and hepatic inflammation. The suppressive effects of inflammatory cytokines on tumour necrosis factor-α (TNF-α), interleukin-1β (IL-1β), IL-6, and macrophage inflammatory protein-2 (MIP-2) have been demonstrated; thus, inflammatory cytokine release is understood to alleviate acute organ damage and reduce inflammation during sepsis. Animal studies have demonstrated that dexmedetomidine decreased ischaemic reperfusion damage in various organs (intestines, myocardium, lung, liver, cerebrum, and kidney) (7–11). Dexmedetomidine alleviates lung damage effectively for the prevention of I/R damage, as well as for haemorrhagic shock, ventilator-related lung damage, rats in which pneumoperitoneum was induced, and pulmonary contusions occurring after blunt trauma (11). Protective effects of dexmedetomidine against inflammation secondary to cardiac, renal, hepatic, testicular, and cerebral IR injury have been described, but its effects on indirect lung damage developing after myocardial I/R have yet to be fully clarified. The objective of this study was to investigate the changes in the levels of malondialdehyde (MDA) and the activities of glutathione-S-transferase (GST) and catalase (CAT) anti-oxidant enzymes, as well as the histopathological features of lungs as indirectly damaged distant organs.
Material and methods
Animals and experimental protocol
This study was conducted in the GUDAM Laboratory of Gazi University with the consent of the Experimental Animals Ethics Committee at Gazi University. All procedures were performed according to the accepted standards of the Guide for the Care and Use of Laboratory Animals.
The study used 18 male Wistar albino rats, weighing between 225 and 275 g, raised under the same environmental conditions. For at least 1 week prior to surgery, the animals were housed in standard cages in a pathogen-free environment, with free access to food (until 2 h prior to the anaesthetic procedure) and water and with a 12-h light/dark cycle.
Diabetes was induced by a single intraperitoneal injection of streptozotocin (Sigma Chemical, St. Louis, MO, USA), at a dose of 55 mg/kg body weight. The blood glucose levels were measured 72 h and 4 weeks following this injection. Rats were classified as diabetic if their fasting blood glucose levels exceeded 250 mg/dl, and only animals with fasting blood glucose of >250 mg/dl were included in the diabetic groups (diabetes control (DC), diabetes plus ischaemia-reperfusion (DIR), and diabetes plus dexmedetomidine-ischaemia-reperfusion (DIRD)).
Rats were anaesthetised with an intraperitoneal injection of 100 mg/kg of ketamine. The trachea was cannulated for artificial respiration. The chest was shaved, and each animal was fixed in a supine position on the operating table. The chest was opened by a left thoracotomy followed by sectioning the fourth and fifth ribs about 2 mm to the left of the sternum. Positive-pressure artificial respiration was started immediately with room air, using a volume of 1.5 ml/100 g body weight at a rate of 60 strokes/min. Sodium heparin (500 IU/kg) was administered through the peripheral vein in the tail.
Following left thoracotomy, ischaemia was produced in the myocardium muscle by a left anterior descending (LAD) artery ligation for 30 min in the DIR and DIRD groups (12). Following ischaemia, the LAD ligation was removed and reperfusion was performed for 120 min. The DIRD group received a single intraperitoneal dose of dexmedetomidine (100 µg/kg); the DIR group received no dexmedetomidine (13). The DC group was evaluated as the diabetic control group and included six rats (C group) which were not induced with diabetes. These mice underwent only left thoracotomy and were closed without undergoing myocardial ischaemia. Activities of CAT and GST anti-oxidant enzymes and MDA quantities were measured. Histopathological changes were evaluated in 10 random samples by microscopic examination of lung sections stained with haematoxylin and eosin.
Histopathological evaluation of the lung
Lung samples were examined histopathologically by light microscopy by the same pathologist, who was blinded to the study. Lung injury in each area was evaluated by determining the alveolar thickness and neutrophil infiltration or aggregation. Each parameter was evaluated as none (0 point), slight (1 point), moderate (2 points), or severe (3 points); the two scores were added, and the total score was interpreted as the lung injury score (14).
Measurements of MDA levels and CAT and GST activities in lung tissue
The lung tissues were first washed with cold-deionised water to remove blood contamination and then homogenised in a 10-fold volume of physiological saline solution (0.154 M) using a homogeniser (Heidolph DIAX900) at 3,000 rpm for 3 min. After centrifugation at 10,000×g for 10 min, the clear upper layer was removed and stored at −80°C until assessment. A supernatant of 10% (w/v) was used for MDA measurements as described by Van Ye et al. (15). All procedures were performed at +4°C throughout the experiments.
Part of the homogenate was extracted in an ethanol/chloroform mixture (5/3: v/v) to remove the lipid fraction, which interferes with the activity measurements of CAT and GST enzymes.
After centrifugation at 10,000×g for 60 min, the clear upper layer was removed and used for CAT and GST analyses as described by Aebi and by Habig (16, 17). The CAT activity method is based on the measurement of the absorbance decrease due to H2O2 consumption at 240 nm. The GST activity method is based on the measurement of absorbance changes at 340 nm due to formation of a GSH-CDNB complex. MDA level in lung supernatant was measured by monitoring the formation of thiobarbituric acid-reactive substances (TBARS) using the method of Van Ye et al. In brief, 0.1 ml of tissue homogenate (PBS; 100 mM, pH 6 Na2HPO4/KH2PO4) was treated with 2 ml of TBA–TCA–HCl reagent (0.2% thiobarbituric acid, 0.6 N HCl, and 20% TCA in absolute ethanol), placed in boiling water for 30 min, cooled, and centrifuged at room temperature for 10 min. The absorbance of the clear supernatant was measured spectrophotometrically against reference blank at 535 nm, using 1,1,3,3-tetraethoxy propane (TEP) as a standard.
Statistical analysis
The Statistical Package for the Social Sciences program (SPSS, version 20.0, Chicago, IL, USA) was used for statistical analysis. The Kolmogorov–Smirnov test was used for the comparisons to determine the distribution of all variable groups. Variations in CAT and GST activities, MDA levels, and histopathological parameters were assessed using the Kruskal–Wallis test. A Bonferroni-adjusted Mann–Whitney U test was used after a significant Kruskal–Wallis finding to determine which group differs from the others. Histopathological percentage was analysed with Fisher’ s test and χ2 test. Results are expressed as mean±standard deviation (SD). Statistical significance was set at a p<0.05.
Results
Comparison of the light microscopy findings for lung tissue neutrophil infiltration/aggregation revealed a significant difference among the groups (p=0.006). Neutrophil infiltration/aggregation was significantly higher in the DIR group than in the C and DC groups (p=0.001 and p=0.013, respectively) (Table 1; Figs. 1 and 2).
Table 1.
Histopathological findings of the lung tissue
| Group C (n=6) | Group DC (n=6) | Group DIR (n=6) | Group DIRD (n=6) | P | |
|---|---|---|---|---|---|
| Neutrophil infiltration/aggregation | 0.0±0.0* | 0.33±0.52* | 1.17±0.41 | 0.5±0.55 | 0.006 |
| Alveolar wall thickening | 0.33±0.52* | 0.67±0.52* | 2.67±0.52§ | 1.67±0.52*,§ | <0.0001 |
| Score (neutrophil infiltration/aggregation+alveolar wall thickening) | 0.33±0.52* | 1.00±0.63* | 4.00±0.89§ | 2.17±0.65*,§ | <0.0001 |
Values are mean±SD. C: control; DC: diabetes control; DIR: diabetes plus ischaemia-reperfusion; DIRD: diabetes plus dexmedetomidine-ischaemia-reperfusion.
Statistical significance was set at p<0.05 for Kruskal–Wallis test.
p<0.05 when compared with Group DIR.
p<0.05 when compared with Group C.
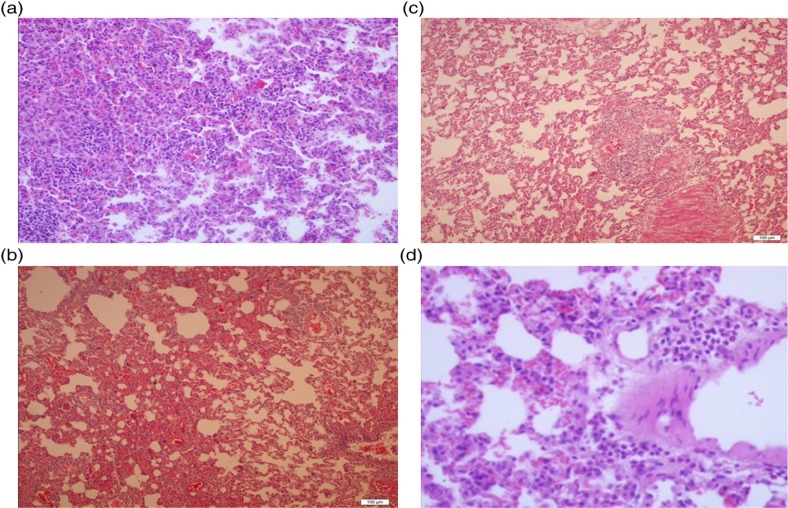
Fig. 1.
(a) Severe neutrophilic infiltration and increased alveolar wall thickness in the DIR group, HE×200. (b) Moderate neutrophilic infiltration and increased alveolar wall thickness in the DIRD group, HE×100. (c) Mild neutrophilic infiltration and increased alveolar wall thickness in the DC and D groups, HE×100. (d) Normal structure lung tissue parenchyma in the control group, HE×200.
Fig. 2.
Neutrophil infiltration/aggregation. *p<0.05 when compared with Group DIR.
Alveolar wall thickening in lung tissue was significantly higher in the DIR group than in the C, DC, and DIRD groups (p=0.003, p=0.003, and p=0.016, respectively) (Table 1; Fig. 1). Alveolar wall thickening was also significantly higher in the DIR and DIRD groups than in the C group (p=0.003 and p=0.007, respectively).
Comparison of lung tissue injury scores determined from light microscopy findings revealed a significant difference among the groups (p<0.0001). The lung tissue injury score was significantly higher in the DIR group than in the C and DC groups (p<0.0001 and p=0.024, respectively) (Table 1; Fig. 1). In addition, the lung tissue injury score was significantly higher in the DIR and DIRD groups than in the C group (p=0.003 and p=0.05, respectively).
In addition, percentage of comparison of the light microscopy findings for lung tissue neutrophil infiltration/aggregation, alveolar wall thickening, and lung tissue injury scores revealed a significant difference among the groups (p=0.005, p<0.0001, and p=0.001, respectively) (Table 2).
Table 2.
Histopathological findings of the lung tissue [n (%)]
| Points | Group C (n=6) | Group DC (n=6) | Group DIR (n=6) | Group DIRD (n=6) | P | |
|---|---|---|---|---|---|---|
| Neutrophil infiltration/aggregation | 0 (none) | 6 (100) | 4 (66.7) | 0 (0) | 3 (50) | 0.005 |
| 1 (slight) | 0 (0) | 2 (33.3) | 5 (83.3) | 3 (50) | ||
| 2 (moderate) | 0 (0) | 0 (0) | 1 (16.7) | 0 (0) | ||
| 3 (severe) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | ||
| Alveolar wall thickening | 0 (none) | 4 (66.7) | 2 (33.3) | 0 (0) | 0 (0) | <0.0001 |
| 1 (slight) | 2 (33.3) | 4 (66.7) | 0 (0) | 2 (33.3) | ||
| 2 (moderate) | 0 (0) | 0 (0) | 2 (33.3) | 4 (66.7) | ||
| 3 (severe) | 0 (0) | 0 (0) | 4 (66.7) | 0 (0) | ||
| Score (neutrophil infiltration/ | 0 | 4 (66.7) | 1 (16.7) | 0 (0) | 0 (0) | 0.001 |
| aggregation+Alveolar wall thickening) | 1 | 2 (33.3) | 4 (66.7) | 0 (0) | 1 (16.7) | |
| 2 | 0 (0) | 1 (16.7) | 0 (0) | 3 (50) | ||
| 3 | 0 (0) | 0 (0) | 2 (33.3) | 2 (33.3) | ||
| 4 | 0 (0) | 0 (0) | 2 (33.3) | 0 (0) | ||
| 5 | 0 (0) | 0 (0) | 2 (33.3) | 0 (0) |
Values are n (%). C: control; DC: diabetes control; DIR: diabetes plus ischaemia-reperfusion; DIRD: diabetes plus dexmedetomidine-ischaemia-reperfusion.
Statistical significance was set at p<0.05 for χ2 test.
Comparison of the groups to each other in terms of lung tissue MDA levels revealed a significant difference (p=0.001). MDA levels were significantly higher in the DIR group than in the C and DIRD groups (p=0.005 and p=0.002, respectively) (Table 3). Comparison of the groups to each other in terms of CAT enzyme activity also revealed a significant difference (p=0.015). CAT enzyme activity was significantly higher in the DIR group than in the C and DIRD groups (p=0.002 and p=0.012, respectively) (Table 3).
Table 3.
Oxidant status parameters of rat lung tissue (Mean±SD)
| Group C (n=6) | Group DC (n=6) | Group DIR (n = 6) | Group DIRD (n = 6) | P | |
|---|---|---|---|---|---|
| MDA (nmol/mg prot) | 0.81±0.32* | 1.25±0.39 | 1.61±0.27 | 0.73±0.23* | 0.001 |
| GST (IU/mg prot) | 1.15±0.21* | 1.87±0.98* | 6.65±1.63 | 1.16±0.07* | <0.0001 |
| CAT (IU/mg prot) | 676.70±124.82* | 936.47±267.01 | 1296.31±417.02 | 810.53±159.19* | 0.015 |
Values are mean±SD. C: control; DC: diabetes control; DIR: diabetes plus ischaemia-reperfusion; DIRD: diabetes plus dexmedetomidine-ischaemia-reperfusion. MDA: malondialdehyde; GST: glutathione-S-transferase; CAT: catalase.
Statistical significance was set at p<0.05 for Kruskal–Wallis test.
p<0.05 when compared with Group DIR.
When groups were compared with each other in terms of GST enzyme activity, a significant difference was observed (p<0.0001). GST enzyme activity was significantly higher in the DIR group compared with the C, DC, and DIRD groups (p<0.0001, p<0.0001, and p<0.0001, respectively) (Table 3).
Discussion
Damage related to I/R may develop in the lung under various conditions. It may occur directly due to ischaemia and subsequent reperfusion, or may arise due to mediators released into the systemic circulation due to reperfusion following the occurrence of ischaemia in other distant organs; numerous human and animal studies have been conducted on this subject (4, 13, 18). Changes in the microcirculation are among the leading causes of the damage that occurs to lung tissues, but diabetes affects and damages the microcirculation independently from all known factors (19). Changes occurring in the microcirculation may negatively affect the duration of ischaemia and may cause expansion of the ischaemic area (19, 20). Therefore, the present study was conducted on diabetic rats.
A number of studies have demonstrated that dexmedetomidine has a protective effect against I/R injury in several organs, including lung, heart, brain, and kidney, which is thought to be due to its anti-oxidant and anti-inflammatory properties (21–26). Some enzymes function as intracellular antioxidants against the damage caused by oxidative stress occurring during I/R. High activities of these enzymes can be considered as indicators that cells require protection against inflammation and subsequent pulmonary damage. CAT catalyses the destruction of H2O2 (27), and elevated blood levels of CAT indicate anti-oxidant activity. MDA is one of the end products of lipid peroxidation and is considered a marker of cell wall peroxidation. Plasma and tissue levels of MDA are accepted as good markers of the oxidative stress and the systemic response that follows I/R (28). Much more GST enzyme activity is observed in the liver tissue compared with lung and heart tissues; nevertheless, high GST activity can be accepted as a marker of elimination of metabolites related to peroxidation and mostly indicates damage caused by reactive oxygen products (29).
We measured CAT and GST enzyme activities and MDA levels to determine the effects of dexmedetomidine on the activities of these cellular protective factors. As a result, levels of tissue MDA, CAT enzyme activity, and GST enzyme activity were significantly lower in the DIRD group when compared with the DIR only group. These results indicate protective role of dexmedetomidine against inflammatory processes in alveolar tissue.
Jiang et al. found in their histopathological study that severe pulmonary damage occurred in the I/R group, whereas I/R damage was less in rats administered 2.5 µg/kg/h dexmedetomidine 1 h before I/R (30). Gu et al. also reported that more pulmonary damage developed in distant organs than in normal lung tissues following renal I/R and that dexmedetomidine decreased that damage (31). The histological results from that study revealed marked haemorrhage in the lamina propria, with loss of integrity, increased interstitial cellularity, and deterioration of alveolar and pulmonary structures in the group not receiving dexmedetomidine. A study by Shen et al. evaluated pulmonary histopathology by semi-quantitative scoring, using pulmonary oedema, existence of inflammatory cells, existence of alveolar haemorrhage, and presence of hyaline membranes and alveolar atelectasis as criteria (32). Pulmonary damage occurring following intestinal ischaemic reperfusion was less in the dexmedetomidine group. A study by Yang et al. investigated the effects of dexmedetomidine on ventilator-induced pulmonary damage and reported lung tissue morphology changes: lung tissue injury, swelling in the alveolar epithelium, alveolar wall changes, and infiltration of polymorphonuclear leukocytes (33). In our study, we found histopathological results resembling those mentioned above. First, alveolar wall thickening in lung tissue was significantly lower in the DIRD group than in the DIR group. Alveolar wall thickening is a well-known feature of inflammation triggered by leucocyte and macrophage accumulation plus perivascular oedema together with intra-alveolar oedema. Second, although the extent of neutrophil infiltration/aggregation was indifferent between the DIR and DIRD groups, the sum of alveolar wall thickening and neutrophil infiltration/aggregation (total injury score) was lower in the DIRD group than in the DIR group. Taken together (histopathological results and tissue anti-oxidant levels), we can postulate an anti-oxidant and anti-inflammatory effect of dexmedetomidine on alveolar tissue during alveolar I/R injury in diabetic rats.
Conclusion
Our study shows that dexmedetomidine could provide lung protection after myocardial I/R; for this reason, dexmedetomidine has potential value in decreasing lung injury following myocardial I/R, in addition to its sedative and analgesic properties. However, further studies are required on this subject.
Authors' contributions
Dr. Kip and Dr. Celik were the main authors of the article, and Dr. Şıvgın and Dr. Bilge collected the data, Dr. Ozer, Dr. Alkan, and Dr. Kiraz helped us in the experimental study. Dr. Erdem helped us in pathological interpretation, whereas Dr. Kavutcu and Dr. Arslan approved the final manuscript.
Conflict of interest and funding
The authors received no financial support for the research and/or authorship of this article.
References
- 1.Francischetti I, Moreno JB, Scholz M, Yoshida WB. Leukocytes and the inflammatory response in ischemia-reperfusion injury. Rev Bras Cir Cardiovasc. 2010;25:575–84. doi: 10.1590/s0102-76382010000400023. [DOI] [PubMed] [Google Scholar]
- 2.Werns SW, Shea MJ, Lucchesi BR. Free radicals and myocardial injury: pharmacologic implications. Circulation. 1986;74:1–5. doi: 10.1161/01.cir.74.1.1. [DOI] [PubMed] [Google Scholar]
- 3.Tsompos C, Panoulis C, Tomicronutouzas K, Zografos G, Papalois A. The effect of erythropoietin on sodium during ischemia reperfusion injury in rats. J Clin Anal Med. 2015;6:569–72. doi: http://dx.doi.org/10.4328/JCAM.2282. [Google Scholar]
- 4.Katz MA. The expanding role of oxygen free radicals in clinical medicine. West J Med. 1986;144:441–6. [PMC free article] [PubMed] [Google Scholar]
- 5.Önem G, Saçar M, Aybek H, Kocamaz E, Adalı F, Saçkan KG, et al. Protective effects of cilostazol and levosimendan on lung injury induced by lower limb ischemia-reperfusion. Turk J Thorac Cardiovasc Surg. 2012;20:577–83. [Google Scholar]
- 6.Kamibayashi T, Maze M. Clinical uses of alpha2-adrenergic agonists. Anesthesiology. 2000;93:1345–9. doi: 10.1097/00000542-200011000-00030. [DOI] [PubMed] [Google Scholar]
- 7.Cai Y, Xu H, Yan J, Zhang L, Lu Y. Molecular targets and mechanism of action of dexmedetomidine in treatment of ischemia/reperfusion injury. Mol Med Rep. 2014;9:1542–50. doi: 10.3892/mmr.2014.2034. [DOI] [PubMed] [Google Scholar]
- 8.Kocoglu H, Karaaslan K, Gonca E, Bozdogan O, Gulcu N. Preconditioning effects of dexmedetomidine on myocardial ischemia/reperfusion injury in rats. Curr Ther Res Clin Exp. 2008;69:150–8. doi: 10.1016/j.curtheres.2008.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kocoglu H, Ozturk H, Ozturk H, Yilmaz F, Gulcu N. Effect of dexmedetomidine on ischemia-reperfusion injury in rat kidney: a histopathologic study. Ren Fail. 2009;31:70–4. doi: 10.1080/08860220802546487. [DOI] [PubMed] [Google Scholar]
- 10.Kucuk A, Yaylak F, Cavunt-Bayraktar A, Tosun M, Arslan M, Comu FM, et al. The protective effects of dexmedetomidine on hepatic ischemia reperfusion injury. Bratisl Med J. 2014;115:680–4. doi: 10.4149/bll_2014_132. [DOI] [PubMed] [Google Scholar]
- 11.Geze S, Cekic B, Imamoğlu M, Yörük MF, Yuluğ E, Alver A, et al. Use of dexmedetomidine to prevent pulmonary injury after pneumoperitoneum in ventilated rats. Surg Laparosc Endosc Percutan Tech. 2012;22:447–53. doi: 10.1097/SLE.0b013e31826183df. [DOI] [PubMed] [Google Scholar]
- 12.Arslan M, Comu FM, Isik B, Ozturk L, Kesimci E. Effect of dexmedetomidine on erythrocyte deformability during ischemia-reperfusion injury of liver in diabetic rats. Bratisl Lek Listy. 2012;113:687–91. doi: 10.4149/bll_2012_156. [DOI] [PubMed] [Google Scholar]
- 13.Tüfek A, Tokgöz O, Aliosmanoglu I, Alabalik U, Evliyaoglu O, Çiftçi T, et al. The protective effects of dexmedetomidine on the liver and remote organs against hepatic ischemia reperfusion injury in rats. Int J Surg. 2013;11:96–100. doi: 10.1016/j.ijsu.2012.12.003. [DOI] [PubMed] [Google Scholar]
- 14.Peng CK, Huang KL, Wu CP, Li MH, Hu YT, Hsu CW, et al. Glutamine protects ischemia-reperfusion induced acute lung injury in isolated rat lungs. Pulm Pharmacol Ther. 2011;24:153–61. doi: 10.1016/j.pupt.2010.07.002. [DOI] [PubMed] [Google Scholar]
- 15.Van Ye TM, Roza AM, Pieper GM, Henderson J, Jr, Johnson JP, Adams MB. Inhibition of intestinal lipid peroxidation does not minimize morphological damage. J Surg Res. 1993;55:553–8. doi: 10.1006/jsre.1993.1183. [DOI] [PubMed] [Google Scholar]
- 16.Aebi H. Catalase in vitro . Methods Enzymol. 1984;105:121–6. doi: 10.1016/s0076-6879(84)05016-3. [DOI] [PubMed] [Google Scholar]
- 17.Habig WH, Pabst MJ, Jakoby WB. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem. 1974;249:7130–9. [PubMed] [Google Scholar]
- 18.Wang Y, Ji M, Chen L, Wu X, Wang L. Breviscapine reduces acute lung injury induced by left heart ischemic reperfusion in rats by inhibiting the expression of ICAM-1 and IL-18. Exp Ther Med. 2013;6:1322–6. doi: 10.3892/etm.2013.1287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Steiner G. How can we improve the management of vascular risk in type 2 diabetes: insights from FIELD. Cardiovasc Drugs Ther. 2009;23:403–8. doi: 10.1007/s10557-009-6190-7. [DOI] [PubMed] [Google Scholar]
- 20.Tapuria N, Junnarkar SP, Dutt N, Abu-Amara M, Fuller B, Seifalian AM, et al. Effect of remote ischemic preconditioning on hepatic microcirculation and function in a rat model of hepatic ischemia reperfusion injury. HPB (Oxford) 2009;11:108–17. doi: 10.1111/j.1477-2574.2009.00006.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang CL, Chen CH, Tsai PS, Wang TY, Huang CJ. Protective effects of dexmedetomidine-ketamine combination against ventilator-induced lung injury in endotoxemia rats. J Surg Res. 2011;167:e273–81. doi: 10.1016/j.jss.2010.02.020. [DOI] [PubMed] [Google Scholar]
- 22.Yoshitomi O, Hara T, Akiyama D, Oshibuchi M, Sumikawa K. Direct protective effects of dexmedetomidine against myocardial ischemia reperfusion injury in anesthetized pigs. Anesthesiology. 2004;101:A633. doi: 10.1097/SHK.0b013e318254d3fb. [DOI] [PubMed] [Google Scholar]
- 23.Engelhard K, Werner C, Eberspacher E, Bachl M, Blobner M, Hildt E, et al. The effect of the alpha 2- agonist dexmedetomidine and the N-methyl-D-aspartate antagonist S (þ)-ketamine on the expression of apoptosis regulating proteins after incomplete cerebral ischemia and reperfusion in rats. Anesth Analg. 2003;96:524–31. doi: 10.1097/00000539-200302000-00041. [DOI] [PubMed] [Google Scholar]
- 24.Gu J, Sun P, Zhao H, Watts HR, Sanders RD, Terrando N, et al. Dexmedetomidine provides renoprotection against ischemia-reperfusion injury in mice. Crit Care. 2011;15:R153. doi: 10.1186/cc10283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lai YC, Tsai PS, Huang CJ. Effects of dexmedetomidine on regulating endotoxin-induced up-regulation of inflammatory molecules in murine macrophages. J Surg Res. 2009;154:212–19. doi: 10.1016/j.jss.2008.07.010. [DOI] [PubMed] [Google Scholar]
- 26.Dong X, Xing Q, Li Y, Han X, Sun L. Dexmedetomidine protects against ischemia-reperfusion injury in rat skeletal muscle. J Surg Res. 2014;186:240–5. doi: 10.1016/j.jss.2013.07.052. [DOI] [PubMed] [Google Scholar]
- 27.Basel H, Kavak S, Demir H, Meral I, Ekim H, Bektas H. Effect of levosimendan injection on oxidative stress of rat myocardium. Toxicol Ind Health. 2013;29:435–40. doi: 10.1177/0748233712436643. [DOI] [PubMed] [Google Scholar]
- 28.Baltalarli A, Ozcan V, Bir F, Aybek H, Sacar M, Onem G, et al. Ascorbic acid (vitamin C) and iloprost attenuate the lung injury caused by ischemia/reperfusion of the lower extremities of rats. Ann Vasc Surg. 2006;20:49–55. doi: 10.1007/s10016-005-9284-0. [DOI] [PubMed] [Google Scholar]
- 29.Vázquez-Medina JP, Zenteno-Savín T, Elsner R. Antioxidant enzymes in ringed seal tissues: potential protection against dive-associated ischemia/reperfusion. Comp Biochem Physiol C Toxicol Pharmacol. 2006;142:198–204. doi: 10.1016/j.cbpc.2005.09.004. [DOI] [PubMed] [Google Scholar]
- 30.Jiang L, Li L, Shen J, Qi Z, Guo L. Effect of dexmedetomidine on lung ischemia reperfusion injury. Mol Med Rep. 2014;9:419–26. doi: 10.3892/mmr.2013.1867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gu J, Chen J, Xia P, Tao G, Zhao H, Ma D. Dexmedetomidine attenuates remote lung injury induced by renal ischemia reperfusion in mice. Acta Anaesthesiol Scand. 2011;55:1272–8. doi: 10.1111/j.1399-6576.2011.02526.x. [DOI] [PubMed] [Google Scholar]
- 32.Shen J, Fu G, Jiang L, Xu J, Li L, Fu G. Effect of dexmedetomidine pretreatment on lung injury following intestinal ischemia-reperfusion. Exp Ther Med. 2013;6:1359–64. doi: 10.3892/etm.2013.1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yang CL, Tsai PS, Huang CJ. Effects of dexmedetomidine on regulating pulmonary inflammation in a rat model of ventilator-induced lung injury. Acta Anaesthesiol Taiwan. 2008;46:151–9. doi: 10.1016/S1875-4597(09)60002-3. [DOI] [PubMed] [Google Scholar]