Abstract
Nanotechnology has brought revolution in cancer detection and treatment. It has capability to detect even a single cancerous cell in vivo and deliver the highly toxic drugs to the cancerous cells. Nanoshells, carbon nanotubes, quantum dots, supermagnetic nanoparticles, nano wires, nanodiamonds, dandrimers, and recently synthesized nanosponges are some of the materials used for cancer detection. Using specific cross linkers, such as specific antibodies against cancer cells individual cancer cells can be located. With the aid of a novel set of lipid-coated, targeted quantum dots a method for quantifying multiple specific biomarkers on the surfaces of individual cancer cells was also developed. This approach to quantitative biomarker detection stands to improve the histopathology methods used to diagnosis pancreatic and other cancers and enable the development of methods to spot cancer cells circulating in the blood stream. Certain nano materials can also deliver cancer drugs at the site so the drug toxicity can also be reduced.
Keywords: Nanoparticles, Nanocensers, Nanosponges, Quantum dots, Nano-tubes and wires
Introduction
Nanotechnology gifted many applications for scientific knowledge from multiple disciplines in science and engineering to design, modify and monitor the properties of matter at nanoscale dimensions [1]. Nanotechnology holds enormous potential for overcoming many of the problems associated with conventional methods, faces difficulties in the detection, treatment, and diagnosis of cancer [2]. By using nanotechnology, nanomaterials have been developed and evaluated for cancer diagnostics. Thus, nanodiagnostics, defined as the use of nanotechnology for clinical diagnostic purposes were developed to meet the demands of clinical diagnostics for increased sensitivity and earlier detection of disease. There is an increasing desire for the developing of materials used in the diagnosis of cancer by nanotechnology such as, nanoshells, carbon nanotubes, quantum dots, polymeric nanoparticles, dendrimers and polynucleotide nanoparticles. The trend is also changed for designing the nanocarriers which are fused with target molecules to reach specific site. These include carbohydrates, antibodies, peptides, aptamers and other small molecules [3].
According to the National Nanotechnology Initiative nanotechnologic materials should be 1–100 nm in at least one dimension. This size requirement can be engineered through various rational designs, including top-down and bottom-up methods [4]. Many of the nanoparticles can be functionalized with several different types of molecules simultaneously—DNA, RNA, targeting molecules and peptides, carbohydrates, and imaging agents. Nanoparticles can selectively target cancer biomarkers and cancer cells, allowing more sensitive diagnosis; early detection requiring minimal amount of tissue, monitoring of the progress of therapy and tumor burden over time, and destruction of solely the cancer cells. However, there is nothing intrinsic about nanoparticles that allow them to specifically target tumor cells and distinguish them from the normal cells, or to distinguish among multiple cell types, resulting in minimal damage to healthy tissues. It is by design that they acquire the ability to recognize unique surface signatures of tumor cells. Moreover, developing clever strategies and knowing which molecules to attach to the nanoparticles require knowledge of tumor-specific receptors that would allow endocytosis of nanoparticles, tumor-specific biomarkers that facilitate identification of cancers, tissue-specific and tumor-specific homing proteins, and tumor-specific enzymes that can permit selective uptake into cells or accumulation in tumor microenvironments. In summary, basic knowledge of cell biology, tumor biology and immunology is essential to the rational design of nanoparticles for cancer therapeutics, and advancement in nanotechnology will be critically dependent on the advancements made in cancer biology [3, 5, 6].
Nanomaterials for Cancer Diagnosis
Nanoshells
Nanoshells are round shape nanoparticles synthesized using amphiphilic tercopolymorpholy and they are pH sensitive doxorubicin encapsulated shell, which are coated with thin metallic shells to improve biocompatibility and optical absorption. Hirsch et al. [7] have shown that surface of the nanoshells can be easily functionalized for targeting application. Nanoshells enhance chemical sensing by as much as 10 billion times. This makes them about 10 thousand times more effective at Raman scattering than that of traditional methods. Each individual nanoshells act as an independent Raman enhancer.
Antibodies can be attached to nanoshells to get them to specifically recognize and target cancer cells (e.g., breast adenocarcinoma cells over expressing human epidermal growth factor receptor-2) in vitro [8]. The antibodies were first attached to polyethylene glycol (PEG) and this antibody-PEG complex was then attached to the nanoshell surface through a sulfur-containing group located at the distal end of the PEG linker.
Nanotubes
Carbon nanotubes are elongated, tube-structured fullerenes. These are made up by distinct atomic arrangement of carbons in the molecular form. The unique physical, chemical, electrical, and optical properties of fullerenes and their derivatives have led to their incorporation into new or improved devices and materials [9–12]. Carbon nanotubes have become popular tools due to their unique physicochemical properties in cancer diagnosis. These are considered as the most promising nanomaterials with the capability of both detecting the cancerous cells and delivering drugs or small therapeutic molecules to cancer cells. In this review, we will show how they have been introduced into the diagnosis and treatment of cancer. Novel SWNT-based tumor-targeted drug delivery systems (DDS) will be highlighted. Furthermore, the in vitro and in vivo toxicity of CNTs reported in recent years will be summarized. Ji et al. [13] reported that a new carbon nanotube device that is capable of detecting single cancer cells. If this is implemented in hospitals, this microfluidic device could help doctors more efficiently to detect the spread of cancer. The researchers at the Israel Institute of Technology have developed diagnostic system that may be able to diagnose lung cancer simply by sampling a patient’s breath with the nanotube microarray devices coated with a different organic material [14].
Quantum Dots
Quantum dots are semiconductors, the most commonly used cadmium selenide capped by zinc sulphide (CdSe/ZnS). The size of quantum dots range from 2 to 10 nm in diameter and these are composed of 10–50 atoms, containing electron–hole, pairs to a discrete quantized energy.
How Quantum Dots Work?
When energy is applied to an atom, electrons are energised and move to a higher level. When the electron returns to its lower and stable state, this additional energy is emitted as light corresponding to a particular frequency. QDs work in much the same way but a QD crystal acts as one very large atom. The energy source used to stimulate a QD is commonly ultraviolet light. The frequency or colour of light given off is not related to the material used in the quantum dot, but by the size of the QD.
Quantum Dot Size and Colour Relationship
Large QDs produce light with a long wavelength and small QDs produce light with small wavelengths. In terms of colour in the visible spectrum, this means large QDs produce red light and small QDs produce blue light—sizes in between account for all the other colours in the spectrum. By combining a range of sizes of QDs in the same sample, the entire light spectrum can be produced simultaneously and appears as white light.
Surface Modifications for In Vitro or In Vivo Applications
QDs are coated with hydrophobic surfactant molecules and can be only solved in organic solution. Before QDs can be applied to biological analysis, they have to meet several criteria. Most of the biomolecules, e.g. protein, DNA, peptides exist in aqueous environment. Modifying the surface of QDs to be hydrophilic and compatible to varieties of biomolecules is important. Designing techniques for specific labeling of cells and biomolecules with QDs is necessary. Nontoxic performance of QDs is required for in vivo applications.
Several strategies have been designed for hydrophilic bioconjugates of QDs. One of the strategies employs functional group reactions. Primary amines, carboxylic acids, alcohols, and thiols are major reacting groups. Another method involves thiol-exchange reactions. Mercapto-coated QDs are mixed with thiolated biomolecules, and chemical equilibrium is reached between absorbed thiols and the free thiols through overnight incubation. On the other hand, some research groups tagged QDs negative charged surfaces to an engineered recombinant protein with a polylysine chain via electrostatic interaction. Another strategy employs phospholipids micelle to encapsulate QDs. The hydrophobic core of the micelle adsorbed to QDs through hydrophobic interactions. The hydrophilic terminals on the outer side of the micelle will interact with biomolecules. In addition, QD-tagged microbeads can be adapted to various biomolecules, providing optical codes for target molecules. All the above strategies have been proved effective by in vitro or in vivo experiments.
Applications of Quantum Dots in Diagnostics
Bruchez et al. [15] reported the first biological applications of QDs in 1998. They used CdSe QDs coated with silica and mercaptoacetic acid layers and showed specific labeling by covalent coupling of ligands to these surfaces. Later, several authors have reported labeling of whole cells and tissue sections using several different surface modifications of QDs. When biomolecules are attached to nanometer-sized bits of semiconductors, a sensitive and widely applicable method for detecting biomolecules and for scrutinizing biomolecular processes was developed. Molecules that are labeled with the QD remain active for biochemical reactions and brightly colored products are produced by the tagged species. The methodology represents a new class of biological dyes that takes advantage of the efficient fluorescence and high photostability of the semiconductor QDs. Hydrophilic QDs in water medium and at physiological pH conditions, have the potential to expand conventional protocols used for cancer diagnostic, which needs previous tissue/cell fixation, and extend it to investigate living cellular and tissular neoplastic mechanisms in real time. QD applications in the investigation of pathological processes, such as neoplastic ones, which may give rise to a wide variety of cancer, constitute a topic of current interest, in which many questions still remain unanswered. In the pursuit of sensitive and quantitative methods to detect and diagnose cancer, nanotechnology has been identified as a field of great promise. For an optimal performance in biological imaging, semiconductor QDs are being developed in order to optimize their luminescent, surface and chemical stability properties. These conditions result in a very complex multilayered chemical assembly where the nanocrystal core determines its emission color, the passivation shell determines its brightness and photostability and the organic capping layer determines its stability and functionality.
Due to the need to optimize the numerous parameters associated with imaging animals, tissue, or live cells, in vivo applications are especially problematic for any reporter species. These applications require many of the most advantageous optical properties exhibited by QDs, but also necessitate biocompatibility, low toxicity, proper attachment of biomolecules, and navigation of the cascade of events involved in the immune response. The following paragraphs address each of these issues to demonstrate the full capabilities of QDs.
Targeting
The work by Schroder et al. [16] demonstrated that QDs can target specific receptors in vivo. QDs were conjugated to folate, a critical nutrient necessary for rapid growth and cell division, to perform assays targeting the folate-specific receptor. TOPO-coated QDs (CdSe) were prepared in phospholipid micelles and assessed in animal studies. The folate-QD conjugates were specifically detected at the folate receptors in mouse lymphoma cells after incubation for two hours. An increase in fluorescence intensity over non-specific QDs in the same cell line was observed, demonstrating that folate was the main factor in bio-recognition and was highly specific in its targeting. As folate is critical for cell growth, the folate receptor will have higher expression levels in cancer cells than in normal cells. The determination of the extent of folate receptor expression could then be a possible diagnostic tool, as any significant intensity increase as compared to normal expression levels is an indication of over-expression, and may be important to cancer diagnosis. Antibody-QD conjugates were also used to optimize circulation times and provide specificity for in vivo applications. Noteworthy research by Jayagopal et al. [17] used these conjugates for standard microscopy determinations, flow cytometry assays, and in vivo imaging. The QD conjugates targeted cell adhesion molecules related to retinal vasculature in rats in a multiplexed fashion using a single excitation source. The researchers employed a PEG crosslinking scheme to link the antibodies and were able to discriminate between different cell adhesion molecules by conjugating specific monoclonal antibodies to QDs. The fluorescence intensities increased within 30 min, whereas non-specifically labeled QDs and the control showed no fluorescence localization in the vasculature.
Imaging
A specific advantage of QDs for in vivo applications is their photostability; QDs allow images to be recorded over a longer period of time than available with the use of fluorescent dyes or proteins due to their resistance to photobleaching. Maysinger visualized CdSe and CdTe QDs detectable at one hour-, one day-, three days-, and seven days post intracortical injection using in vivo imaging techniques. Mice were injected subcutaneously and scanned for fluorescence, in particular in the brain, where peak fluorescence was observable at three days post injection and persisted for seven days. Sub-cellular resolution was achieved and allowed the identification of the location of the conjugates (Fig. 1). This work had numerous important features [18]. The researchers demonstrated internalization of their QD conjugates, with varied rate and efficacy of internalization for different cell-types. Neuronal internalization is especially challenging, and in vivo imaging in neural cells has important relevance to QD toxicity, as this approach would allow the investigation of the neuronal immune response in real time. This approach was novel not only in the in vivo imaging aspects but the animal model was transgenic, and was monitored for an astrocyte-responsive luciferase reporter in addition to the injection of QD conjugates. The activation of the astrocyte response was observable as an increase in bioluminescence in response to foreign nanoparticles in mice. The work of Jiang et al. [19] demonstrated the ability of QDs with near-IR region emission wavelengths for in vivo analysis of deep tissue or non-invasive applications. The application of near-IR reporters minimized the absorption and scattering of light by native tissues, and allowed the researchers to employ longer wavelengths for a diagnostic emission window, generally between 650–900 nm. By conjugating transfer into a QD with an emission at 750 nm in vivo observations were performed on mouse heart and femur up to 0.8 mm deep beneath the skin. QD conjugates emitting in the near-IR allowed for greater visualization depth, yielding an increase of up to 800 microns from what was performed previously. Zimmer et al. [20] have also employed near-IR emitting QDs due to their ability to act as a reporter at a wavelength minimally absorbed by biological species. Building off of their previous work, the authors synthesized a series of InAs/ZnSe core/shell QDs with a smaller hydrodynamic diameter (less than 10 nm) than previously reported. The small core size, along with variation to the shell thickness and composition offered a range of size tunable emission wavelengths, between 750 and 920 nm. The conjugation of DHLA to the QDs allowed for the observations within the interstitial fluid in rats, where the QD conjugates exited the blood vessels. This visualization of the extravasation sites is important, and has potential to interrogate the delivery mechanism of QDs to tumor cells. No extravasation was observed in quantum dots without the DHLA coating.
Fig. 1.
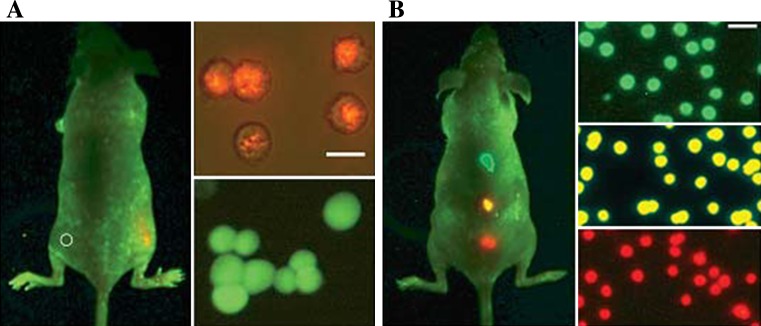
Sensitivity and multicolor capability of QD imaging in live animals. Sensitivity and spectral comparison between QD-tagged and GFP transfected cancer cells (a), and simultaneous in vivo imaging of multicolor QD-encoded microbeads (b)
Vasculature Imaging
Tumor growth requires a supply of nutrients from the blood stream. Angiogenesis is the process by which new vasculature establishes a blood supply to a growing tumor. Receptors, such as integrin, are highly expressed in tumor cells during angiogenesis, and diagnostics targeting such receptors can lend insight into the type and extent of diseases, including cancer. The work by Cai et al. [21] detailed the use of a tri-peptide-QD conjugate which specifically binds to integrin. The conjugation of arginine-glycine-aspartic acid to a QD with emission at 705 nm yielded a diagnostic tool specific for the integrin receptor that emits in the near-IR region. In vitro analysis was performed on human glioblastoma and human breast cancer cells, showing specificity for integrin-positive cells only. In addition, binding was inhibited by the presence of an integrin antagonist. These results illustrated that assays were possible with the potential to help differentiate cancer based on integrin expression levels. Injections of both the conjugates and non specific controls through the tail vein of mice also demonstrated the specificity, with a maximum fluorescence at six hours post-injection. This work showed successful in vivo tumor imaging by QDs, including the extensive vasculature network, and provides an important window into the importance of QDs for imaging applications. The work of Smith et al. [22] demonstrated how QDs allow for the non-invasive visualization of blood vessel development over time. The conjugation of biotinylated fibrinogen to quantum dots showed specificity towards the membrane of blood vessels during angiogenesis. The conjugates had robust biocompatibility, staying in circulation for days without noticeable toxicity. In addition, residence times could be influenced through alterations of the surface chemistry functionalities. Like work cited previously, the authors employed QDs with longer emission wavelengths to avoid native autofluorescence and increase their depth of field. Overall, as directly compared to the standard fluorophore FITC, these quantum dot conjugates imaged vasculature at a comparable intensity level at substantially lower concentrations, almost three orders of magnitude less concentrated.
Tracking
The work of Tada et al. [23] elucidated the delivery mechanism of QDs into human breast cancer cells. The authors employ a cell line that over-expressed the Human Epidermal growth factor Receptor 2 (HER2) on its cell membrane, which is a protein associated with higher aggressiveness in breast cancers. The conjugation of anti-HER2 monoclonal antibodies to QDs allowed for the visualization of the nanoparticles in blood vessels serving tumor cells in mice. The QDs helped determine the velocity and directionality, among other criteria, for the circulation in the blood vessel, extravasation, binding to HER2 on the cell membrane, and movement into the perinuclear region, etc. This important work allowed the tracking of movements that were random in orientation and speed, exhibited stop and go behavior, and diffused by Brownian motion. This work represents some of the most sophisticated efforts to track biomolecules and understand sub-cellular movements, and will serve as a landmark study in drug delivery.
Superparamagnetic Nanoparticles
Iron oxide particles (Fe3O4) are reffered as magnetic nanoparticles and developed as superparamagnetic nanoparticles. The sizes of the nanoparticles are less than 10 nm in diameter. These nanoparticles are having potential application in magnetic resonance imaging. Many reports revealed that superparamagnetic nanoparticles can be functionalized with other type of nanoparticle, so as to permit specific tumor targeting. These are also having importance in the use of magnetic fields to localize magnetic nanoparticles to targeted sites, a system known as magnetic drug targeting [24]. Superparamagnetic nanoparticles can be modified by improving the solubility and specificity of iron oxide particles. Nanoparticles are attaching to polyethylene glycol this modification preventing sterical opsonization of nanoparticles in the serum and decreasing their uptake by the reticuloendothelial system. This design of nanoparticles effectively enhances biocompatibility and increases the circulation time of nanoparticles [25]. Superparamagnetic nanoparticles are developed as hydrophobic by coating with aliphatic surfactants or liposomes [26]. Iron oxide nanoparticles can also be used in imaging techniques that can selectively image proliferating cells in vivo can provide critically important insights of tumor growth rate, degree of tumor angiogenesis, effectiveness of treatment and vigor of normal cells. Superparamagnetic nanoparticles undergo Brownian relaxation by induction of an alternating field, in which heat is generated by the rotation of particles in the field. However, iron oxide concentrations are necessary to raise the tissue to critical temperatures for thermal ablation [27, 28].
Nanowires
Nanowires are nanoparticles with diameters of only a few nanometres and extended lengths. Predictably, the length and width ration is extremely large making them effectively one-dimensional structures. These are revolutionized innovative compounds these would be used to link together tiny components into extremely small circuits. These nanowires are purported to have functions in monitoring brain electrical activity without having to use a brain probe and violating the brain parenchyma. Using platinum nanowires, researchers have used blood vessels as the guiding pathway to determining the electrical activity of neurons that are adjacent to the blood vessels. These nanowires can deliver and receive electrical impulses. Because of the potential for targeting specific areas of the brain, they can enable new treatment modalities for various neurological diseases [29]. Another form of nanowires, polymer nanowires has the advantage of changing shape in response to electrical fields allowing precise steering through the circulatory system of brain towards the exact spot of interest. These also have the benefit of being 20–30 times smaller than the platinum ones and are biodegradable, allowing for short-term use of brain implants [30].
Nanodiamonds
Nanodiamonds are synthesized in 1962 by detonation and also can be prepared by covalent and noncovalent modification to absorb or graft a variety of functional groups and complex moieties, including proteins and DNA [31]. These are attractive agents for use in biological and medical applications largely due to their greater biocompatibility than other carbon nanomaterials, stable photoluminescence, commercial availability and minimal cytotoxicity [31–33]. Nanodiamonds can be modified and conjugated to a variety of molecules for the use of cell labeling and drug delivery. Nanodiamonds which are substituted with certain functional groups can improve their solubility and direct them to specific binding sites on target cells and tissues and reduce their effects on normal tissues.
The different types of functional groups substituted on nanodiamonds which broadened the scope of their potential diagnostic and therapeutic applications. Nanodiamonds can be used for cell labeling and tracing because they do not interrupt cell division or differentiation, have minimal cytotoxicity, and are easily functionalized with proteins and other markers for targeting purposes. Nanodiamonds have successfully been used as biomarkers or tracers to label or trace HeLa cells, lung cancer cells, and murine fibroblasts [34–37].
Nanosponge
Nanosponge is like a three-dimensional network or scaffold. Its backbone is a long length of polyester and the size is of about virus. Nanosponge is mixed in solution with small molecules called cross-linkers that act like tiny seizing hooks to tie up different parts of the polymer together. The net effect of this arrangement is to form spherically shaped particles filled with cavities where drug molecules can be stored and then injected into the body. This tiny sponge circulates around the tumor cell until they encounter the surface to sustain releasing their drug cargo. Nanosponge is three to five times more effective at reducing tumor growth than direct injection. The targeted delivery systems of nanosponge have several basic advantages like, the drug is released at the tumor instead of circulating widely through the body, it is more effective for a given dosage [38, 39]. The nanosponge should have basic features such as fewer harmful side effects because smaller amounts of the drug will come into contact with healthy tissue [40].
Dendrimers
Dendrimers are highly complex molecules with a core, branched and end groups. The size and shape depends on the core, branches and surface functional groups, these helps to identify the reactivity of dendrimers. Dendrimers’ biocompatibity has been used for delivering the potent drugs, such as cisplatin and doxorubicin in cancer treatment [41]. The surface chemistry of dendrimers can be modified to attach ligands, this modified nanostructure was used to target the dendrimer to tumor cells. Dendrimers were studied extensively for targeting and delivering the therapeutic agents for cancer and contrast agents for magnetic resonance imaging [42, 43]. The gold coating on surface of dendrimer was greatly reduced their toxicity without significant altering their size and to provide an anchor for attachment of targeting molecules with high affinity to tumor cells [44, 45].
Conclusion
The application of nanotechnology in the field of cancer nanotechnology has experienced exponential growth in the past few years. Nanotechnology offers innovative exposure of the destiny of medicine and surgery. With the advent and popularity of minimally invasive surgeries and interventional techniques, nanotechnology provides a future platform for further development of drugs for cancer diagnosis. The multidisciplinary field of nanotechnology holds the promise of delivering a scientific breakthrough and may move very fast from concept to reality.
References
- 1.Mousa SA, Bharali DJ, Armstrong D. From nutraceuticals to pharmaceuticals to nanopharmaceuticals: a case study in angiogenesis modulation during oxidative stress. Mol Biotechnol. 2007;37:72–80. doi: 10.1007/s12033-007-0064-7. [DOI] [PubMed] [Google Scholar]
- 2.Davis ME, Chen ZG, Shin DM. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat Rev Drug Discov. 2008;7:771–782. doi: 10.1038/nrd2614. [DOI] [PubMed] [Google Scholar]
- 3.Frank A, June-Wha R, Jerome PR, Radovic-Moreno AF, Robert L, Omid CF. New frontiers in nanotechnology for cancer treatment. Urol Oncol Semin Orig Invest. 2008;26:74–85. doi: 10.1016/j.urolonc.2007.03.017. [DOI] [PubMed] [Google Scholar]
- 4.Koning GA, Krijger GC. Targeted multifunctional lipid-based nanocarriers for image-guided drug delivery. Anticancer Agents Med Chem. 2007;7:425–440. doi: 10.2174/187152007781058613. [DOI] [PubMed] [Google Scholar]
- 5.Ranjita M, Sarbari A, Sanjeeb KS. Cancer nanotechnology: application of nanotechnology in cancer therapy. Drug Discovery Today. 2010;15:19–20. doi: 10.1016/j.drudis.2010.08.006. [DOI] [PubMed] [Google Scholar]
- 6.Kewal K, Jain T. Nanotechnology in clinical laboratory diagnostics, review. Clin Chim Acta. 2005;358:37–54. doi: 10.1016/j.cccn.2005.03.014. [DOI] [PubMed] [Google Scholar]
- 7.Hirsch LR, Gobin AM, Lowery AR, et al. Metal nanoshells. Ann Biomed Eng. 2006;34:15–22. doi: 10.1007/s10439-005-9001-8. [DOI] [PubMed] [Google Scholar]
- 8.Loo C, Lowery A, Halas N, West J, Drezek R. Immunotargeted nanoshells for integrated cancer imaging and therapy. Nano Lett. 2005;5(4):709–711. doi: 10.1021/nl050127s. [DOI] [PubMed] [Google Scholar]
- 9.Avouris P, Chen Z, Perebeinos V. Carbon-based electronics. Nat Nanotechnol. 2007;2(10):605–615. doi: 10.1038/nnano.2007.300. [DOI] [PubMed] [Google Scholar]
- 10.Bakry R, Vallant RM, Najam-ul-Haq M, Rainer M, Szabo Z, Huck CW, Bonn GK. Medicinal applications of fullerenes. Int J Nanomed. 2007;2(4):639–649. [PMC free article] [PubMed] [Google Scholar]
- 11.Bosi S, Da Ros T, Spalluto G, Prato M. Fullerene derivatives: an attractive tool for biological applications. Eur J Med Chem. 2003;38:913–923. doi: 10.1016/j.ejmech.2003.09.005. [DOI] [PubMed] [Google Scholar]
- 12.Guldi DM, Prato M. Excited-state properties of C(60) fullerene derivatives. Acc Chem Res. 2000;33(10):695–703. doi: 10.1021/ar990144m. [DOI] [PubMed] [Google Scholar]
- 13.Ji SR, Liu C, Zhang B, Yang F, Xu J, Long J, Jin C, Fu DL, Ni QX, Yu XJ. Carbon nanotubes in cancer diagnosis and therapy. Biochim Biophys Acta. 2010;1806:29–35. doi: 10.1016/j.bbcan.2010.02.004. [DOI] [PubMed] [Google Scholar]
- 14.Kam NW, O’Connell M, Wisdom JA, Dai H. Carbon nanotubes as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction. Proc Natl Acad Sci USA. 2005;102(33):11600–11605. doi: 10.1073/pnas.0502680102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bruchez M, Moronne M, Gin P, Weiss S, Alivisatos AP. Semiconductor nanocrystals as fluorescent biological labels. Science. 1998;281:2013–2016. doi: 10.1126/science.281.5385.2013. [DOI] [PubMed] [Google Scholar]
- 16.Schroeder JE, Shweky I, Shmeeda H, Banin U, Gabizon A. Folate-mediated tumor cell uptake of quantum dots entrapped in lipid nanoparticles. J Control Release. 2007;124:2834. doi: 10.1016/j.jconrel.2007.08.028. [DOI] [PubMed] [Google Scholar]
- 17.Jayagopal A, Yan RS, Blakemore JL, Linton MF, Fazio S, Haselton FR. Quantum dot mediated imaging of atherosclerosis. Nanotechnology. 2009;20:165102. doi: 10.1088/0957-4484/20/16/165102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Maysinger D. Nanoparticles and cells: good companions and doomed relationships. Org Biomol Chem. 2007;5:2335–2342. doi: 10.1039/b704275b. [DOI] [PubMed] [Google Scholar]
- 19.Jiang W, Singhal A, Kim BYS, Zheng J, Rutka JT, Wang C, Chan WCW. Assessing near-infrared quantum dots for deep tissue, organ, and animal imaging applications. J Assoc Lab Autom. 2008;13:6–12. doi: 10.1016/j.jala.2007.09.002. [DOI] [Google Scholar]
- 20.Zimmer JP, Kim SW, Ohnishi S, Tanaka E, Frangioni JV, Bawendi MG. Size series of small indium arsenide-zinc selenide core-shell nanocrystals and their application to in vivo imaging. J Am Chem Soc. 2006;128:2526–2527. doi: 10.1021/ja0579816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cai W, Chen X. Preparation of peptide-conjugated quantum dots for tumor vasculature-targeted imaging. Nat Protoc. 2008;3(1):89–96. doi: 10.1038/nprot.2007.478. [DOI] [PubMed] [Google Scholar]
- 22.Smith AM, Dave S, Nie S, True L, Gao X. Multicolor quantum dots for molecular diagnostics of cancer. Expert Rev Mol Diagn. 2006;6:231–244. doi: 10.1586/14737159.6.2.231. [DOI] [PubMed] [Google Scholar]
- 23.Tada H, Higuchi H, Wanatabe TM, Ohuchi N. In vivo real-time tracking of single quantum dots conjugated with monoclonal anti-HER2 antibody in tumors of mice. Cancer Res. 2007;67(3):1138–1144. doi: 10.1158/0008-5472.CAN-06-1185. [DOI] [PubMed] [Google Scholar]
- 24.Wust P, Hildebrandt B, Sreenivasa G, Rau B, Gellermann J, Riess H, et al. Hyperthermia in combined treatment of cancer. Lancet Oncol. 2002;3(8):487–497. doi: 10.1016/S1470-2045(02)00818-5. [DOI] [PubMed] [Google Scholar]
- 25.Harris JM, Martin NE, Modi M. Pegylation: a novel process for modifying pharmacokinetics. Clin Pharmcokinetics. 2001;40:539–551. doi: 10.2165/00003088-200140070-00005. [DOI] [PubMed] [Google Scholar]
- 26.Kubo T, Sugita T, Shimose S, Nitta Y, Ikuta Y, Murakami T. Targeted delivery of anticancer drugs with intravenously administered magnetic liposomes in osteosarcoma-bearing hamsters. Int J Oncol. 2000;17(2):309–315. doi: 10.3892/ijo.17.2.309. [DOI] [PubMed] [Google Scholar]
- 27.Johannsen M, Gneveckow U, Thiesen B, Taymoorian K, Cho CH, Waldofner N, Scholz R, Jordan A, Loening SA, Wust P. Thermotherapy of prostate cancer using magnetic nanoparticles: feasibility, imaging, and three-dimensional temperature distribution. Eur Urol. 2007;52:1653–1661. doi: 10.1016/j.eururo.2006.11.023. [DOI] [PubMed] [Google Scholar]
- 28.Neuwelt E, Varallyay P, Bago A, Muldoon L, Nesbit G, Nixon R. Imaging of iron oxide nanoparticles by MR and light microscopy in patients with malignant brain tumours. Neuropathol Appl Neurobiol. 2004;30(5):456–471. doi: 10.1111/j.1365-2990.2004.00557.x. [DOI] [PubMed] [Google Scholar]
- 29.Kupsch A, Earl C. Neurosurgical interventions in the treatment of idiopathic Parkinson disease: neurostimulation and neural implantation. J Mol Med. 1999;77(1):178–184. doi: 10.1007/s001090050332. [DOI] [PubMed] [Google Scholar]
- 30.Lee Min-Ho, Lee Dong-Ho, Jung Suk-Won, Lee Kuk-Nyung, Park YoungSoo, Seong Woo-Kyeong. Measurements of serum C-reactive protein levels in patients with gastric cancer and quantification using silicon nanowire arrays. Nanomed Nanotechnol Biol Med. 2010;6:78–83. doi: 10.1016/j.nano.2009.04.004. [DOI] [PubMed] [Google Scholar]
- 31.Krueger A. New carbon materials: biological applications of functionalized nanodiamond materials. Chemistry. 2008;14:1382–1390. doi: 10.1002/chem.200700987. [DOI] [PubMed] [Google Scholar]
- 32.Enoki T, Takai K, Osipov V, Baidakova M, Vul A. Nanographene and nanodiamond; new members in the nanocarbon family. Chem Asian J. 2009;4:796–804. doi: 10.1002/asia.200800485. [DOI] [PubMed] [Google Scholar]
- 33.Holt KB. Diamond at the nanoscale: applications of diamond nanoparticles from cellular biomarkers to quantum computing. Philos Transact A Math Phys Eng Sci. 2007;365:2845–2861. doi: 10.1098/rsta.2007.0005. [DOI] [PubMed] [Google Scholar]
- 34.Vaijayanthimala V, Chang HC. Functionalized fluorescent nanodiamonds for biomedical applications. Nanomed. 2009;4(1):47–55. doi: 10.2217/17435889.4.1.47. [DOI] [PubMed] [Google Scholar]
- 35.Fu CC, Lee HY, Chen K, Lim TS, Wu HY, Lin PK, et al. Characterization and application of single flourescent ananodiamonds as cellular biomarkers. Proc Natl Acad Sci USA. 2007;104:727–732. doi: 10.1073/pnas.0605409104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chang YP, Pinaud F, Antelman J, Weiss S. Tracking bio-molecules in live cells using quantum dots. J Biophotonics. 2008;1:287–298. doi: 10.1002/jbio.200810029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chang YR, Lee HY, Chen K, Chang CC, Tsai DS, Fu CC, et al. Mass production and dynamic imaging of fluorescent nanodiamonds. Nat Nanotechnol. 2008;3(5):284–288. doi: 10.1038/nnano.2008.99. [DOI] [PubMed] [Google Scholar]
- 38.Cavalli R, Trotta F, Tumiatti V. Cyclodextrin-based nanosponges for drug delivery. J Incl Phenom Macrocycl Chem. 2006;56:209–213. doi: 10.1007/s10847-006-9085-2. [DOI] [Google Scholar]
- 39.Guo L, Gao G, Liu X, Liu F. Preparation and characterization of TiO2 nanosponge. Mater Chem Phys. 2008;111:322–325. doi: 10.1016/j.matchemphys.2008.04.016. [DOI] [Google Scholar]
- 40.Swaminathan S, Vavia PR, Trotta F, Torne S. Formulation of betacyclodextrin based nanosponges of itraconazole. J Incl Phenom Macrocycl Chem. 2007;57:337–341. doi: 10.1007/s10847-006-9216-9. [DOI] [Google Scholar]
- 41.Sampathkumar SG, Yarema KJ. Targeting cancer cells with dendrimers. Chem Biol. 2005;12:5–6. doi: 10.1016/j.chembiol.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 42.Lee CC, MacKay JA, Frechet JM, Szoka FC. Designing dendrimers for biological applications. Nat Biotechnol. 2005;23:1517–1526. doi: 10.1038/nbt1171. [DOI] [PubMed] [Google Scholar]
- 43.Gillies ER, Frechet JMJ. Dendrimers and dendritic polymers in drug delivery. Drug Discov Today. 2005;10:35–43. doi: 10.1016/S1359-6446(04)03276-3. [DOI] [PubMed] [Google Scholar]
- 44.Shi X, Wang S, Meshinchi S, Van Antwerp ME, Bi X, Lee I, et al. Dendrimer-entrapped gold nanoparticles as a platform for cancer-cell targeting and imaging. Small. 2007;3:1245–1252. doi: 10.1002/smll.200700054. [DOI] [PubMed] [Google Scholar]
- 45.Link S, El-Sayed MA. Shape and size dependence of radiative, non-radiative and photothermal properties of gold nanocrystals. Int Rev Phys Chem. 2000;19:409–453. doi: 10.1080/01442350050034180. [DOI] [Google Scholar]


