Abstract
Background Plants are sessile organisms that have the ability to integrate external cues into metabolic and developmental signals. The cues initiate specific signal cascades that can enhance the tolerance of plants to stress, and these mechanisms are crucial to the survival and fitness of plants. The adaption of plants to stresses is a complex process that involves decoding stress inputs as energy-deficiency signals. The process functions through vast metabolic and/or transcriptional reprogramming to re-establish the cellular energy balance. Members of the mitochondrial energy dissipation pathway (MEDP), alternative oxidases (AOXs) and uncoupling proteins (UCPs), act as energy mediators and might play crucial roles in the adaption of plants to stresses. However, their roles in plant growth and development have been relatively less explored.
Scope This review summarizes current knowledge about the role of members of the MEDP in plant development as well as recent advances in identifying molecular components that regulate the expression of AOXs and UCPs. Highlighted in particular is a comparative analysis of the expression, regulation and stress responses between AOXs and UCPs when plants are exposed to stresses, and a possible signal cross-talk that orchestrates the MEDP, reactive oxygen species (ROS), calcium signalling and hormone signalling.
Conclusions The MEDP might act as a cellular energy/metabolic mediator that integrates ROS signalling, energy signalling and hormone signalling with plant development and stress accumulation. However, the regulation of MEDP members is complex and occurs at transcriptional, translational, post-translational and metabolic levels. How this regulation is linked to actual fluxes through the AOX/UCP in vivo remains elusive.
Keywords: Alternative oxidase, AOX, calcium signals, energy signalling, mitochondrial energy dissipation pathway, MEDP, mitochondrial retrograde regulation, plant stress signalling, reactive oxygen species, ROS wave, uncoupling protein, UCP
INTRODUCTION
All chemical reactions in living cells are energy dependent. In biological processes, energy drives the cellular metabolism and transport processes that are necessary for plant growth and developmental switching (Wallimann et al., 1992; Borisjuk et al., 2003). Depending on the metabolic demands, production, transportation, utilization and conversion of energy within cells are typically dynamic and require the co-ordination of different organelles through diverse signalling pathways to achieve specific developmental switches (Suzuki et al., 2012). For instance, changes in energy demands were found to be closely associated with the greening process in embryos of Vicia faba during their development (Borisjuk et al., 2003), and endosperm differentiation of barley grains (Rolletschek et al., 2004) was reported to be developmentally regulated in a specific temporal and spatial manner (Borisjuk et al., 2003; Rolletschek et al., 2004). Stresses that affect plant growth and development can result in enhanced accumulation of reactive oxygen species (ROS) (Mittler, 2002), and alterations in the cellular energy state (Suzuki et al., 2012), which can cause oxidation of cellular components, developmental defects and even arrest of plant growth. The relationship between energy balance and plant stress adaption has been well documented (Huner et al., 1998; Wilson et al., 2006; Baena-González and Sheen, 2008). When plants respond to stresses, there is generally a decrease in the photosynthetic performance and fluctuations in respiration, which trigger an energy-deficit signal (Baena-González and Sheen, 2008). Specifically, some abiotic stresses such as extreme temperature (Stupnikova et al., 2006), hypoxia/anoxia (Huang et al., 2005; Liu et al., 2006; Branco-Price et al., 2008), low pH (Messerli et al., 2005), starvation (Vanlerberghe, 2013) and stresses that downregulate TOR (target of rapamycin) signalling (Caldana et al., 2013) are more likely to result in ATP overconsumption, causing a cellular energy crisis and thereby activating energy-conserving responses. In contrast, stresses that induce the autophage pathway generally do not trigger energy deficiency (Lv et al., 2014). A popular proposal has been that different stresses were partly decoded as signals of energy deficiency and that the adaption of plants to stresses is a process by which cellular metabolism and the transcriptome are reprogrammed to re-establish the balance of cellular energy (Wilson et al., 2006; Baena-González and Sheen, 2008; Rodrigues et al., 2013; Lastdrager et al., 2014). The metabolic re-programming has been suggested to be involved in inhibition of biosynthetic pathways, such as sugar synthesis (Stitt and Hurry, 2002), as well as induction of catabolic processes, photosynthesis, sugar remobilization (Tome et al., 2014), starch and storage lipid accumulation (Caldana et al., 2013), and alteration of primary and secondary metabolism (Caldana et al., 2013). Similarly, transcript profiling studies have provided a snapshot of the complex energy-associated gene network that operates during stress conditions (Baena-González et al., 2007; Avin-Wittenberg et al., 2012; Caldana et al., 2013). The changes in global gene expression in response to energy stress are followed by slowed photosynthesis, and activated energy-promoting gene networks, some of which are known to be correlated with tetrapyrrole biosynthesis, the aspartate family pathway, the tricarboxylic acid (TCA) cycle and mitochondrial electron transport chain (Avin-Wittenberg et al., 2012). Thus, the ability of plants to maintain the delicate balance of energy production and utilization may be of fundamental importance for their survival and fitness, particularly when exposed to stress conditions.
Plants have developed several mechanisms to minimize any damage that might result from energy imbalances, and two of the most important mechanisms have been well characterized. One of the mechanisms takes place in the chloroplast and is known as non-photochemical quenching-mediated energy dissipation (Huner et al., 1998; Szabo et al., 2005; Ruban et al., 2007), whereby excessively absorbed light energy is dissipated as heat (non-photochemical quenching) (Huner et al., 1998; Szabo et al., 2005). The other mechanism is a mitochondria–energy dissipation system, whereby ATP production and mitochondrial respiration are uncoupled by alternative oxidases (AOXs) and uncoupling proteins (UCPs) (Meeuse, 1975; Sluse and Jarmuszkiewicz, 2002). The AOXs and UCPs regulate coupling between the TCA cycle and ATP synthesis, allowing a stable carbon flow through glycolysis, which is essential for autotrophic organisms such as plants whose biosynthetic demands depend greatly on TCA intermediates (Vercesi et al., 2006). AOXs and UCPs were identified originally as being functionally related to thermogenic processes in plants and animals, respectively (Meeuse, 1975; Gimeno et al., 1997). It is now well documented that genes in the mitochondrial energy dissipation pathway (MEPD) are involved in a wide range of physiological and developmental processes (Chai et al., 2012; Diano and Horvath, 2012; Xu et al., 2012; Vanlerberghe, 2013). However, the precise mechanisms by which AOXs and UCPs are regulated are largely unknown.
In this review, we briefly summarize the advances in knowledge of the MEDP in plants with a timeline (Fig. 1) and describe the role of the MEDP in plant growth and development as well as in integrating metabolic processes in different compartments of the cell (Fig. 2). We also discuss recent advances in the understanding of components that regulate the expression of AOXs and UCPs with the focus on a comparative analysis of AOXs and UCPs in stress adaption, expression and regulation. We highlight stress-triggered ROS signals, calcium signals, redox signals and metabolic intermediates that might be important in integrating mitochondrial retrograde signalling with energy and stress signals under stress conditions.
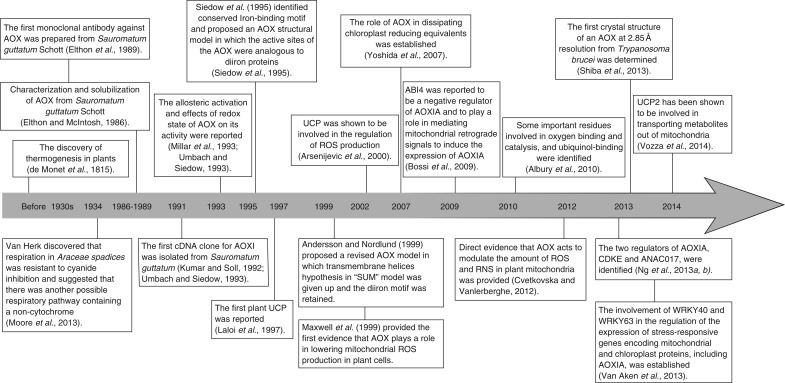
Fig. 1.
Timeline: a brief history of the understanding of the MEDP.
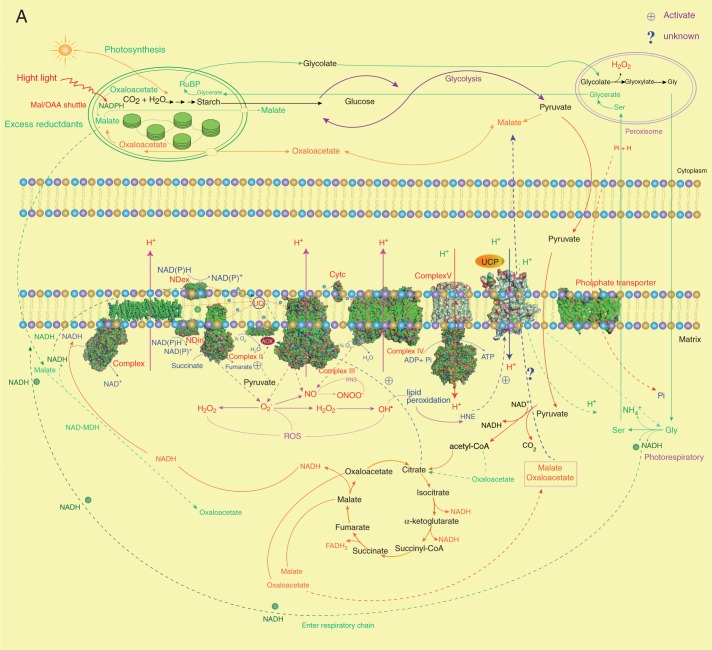
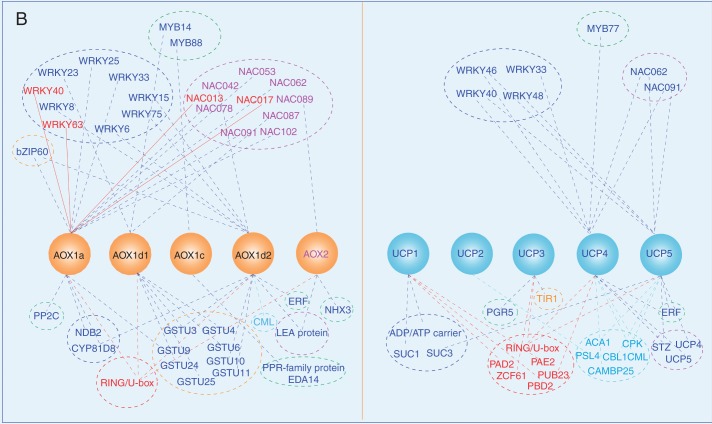
Fig. 2.
The intraorganelle metabolic association linked by the MEDP (A) and co-regulation gene network (B). (A) The major mitochondrial respiratory complexes (MCRs) and AOX are shown as crystal structures. For each MCR protein, structures are retrieved from the Protein Data Bank (PDB): complex I, PDB 3M9S (Efremov et al., 2010); complex II, PDB 1ZOY (Sun et al., 2005); complex III, cytochrome bc1, PDB 3H1J (Iwata et al., 1998); complex IV, cytochrome c oxidase, PDB 1OCO (Yoshikawa et al., 1998); complex V, F0, PDB 1C17 (Rastogi and Girvin, 1999), F1, PDB 1E79 (Gibbons et al., 2000); yeast NADH dehydrogenase, PDB 4G9K (Iwata et al., 2012); phosphate transporter, PDB 4J05 (Pedersen et al., 2013); AOX, PDB 3VV9 (Shiba et al., 2013); UCP, PDB 2LCK (Berardi et al., 2011). (B) To obtain gene sets of co-expressed MEDP members (Supplementary Data Tables S1–S5), each MEDP member is selected, respectively, to run the Perturbations tool, Co-Expression tool and Biclustering tool of the Genevestigator database (Zimmermann et al., 2004; Hruz et al., 2008). First, conditions that are relevant for each MEDP target gene are identified by using the Perturbations tool from the condition search tools to create samples based on a fold change value of 2 and P-value cut-off of 0·05, and then to correlate across these perturbations in the Co-Expression tool. The resulting co-expressed gene sets are used to produce the co-regulation gene network.
THE MITOCHONDRIAL ENERGY DISSIPATION PATHWAY IN PLANTS
There are at least two sets of functionally related pathways in plant mitochondria. One pathway consists of members of the small nuclear gene family, which couple the oxidation of ubiquinol with the reduction of molecular oxygen to water (Vanlerberghe and McIntosh, 1997). This pathway mediates cyanide-insensitive plant respiration with a terminal AOX and is known as the alternative pathway or the cyanide-resistant respiratory pathway (Vanlerberghe and McIntosh, 1997). The alternative pathway branches at a ubiquinol pool and bypasses ATP-producing complexes III and IV, which results in uncoupling between electron transport and ATP synthesis. Thus, a direct consequence of alternative pathway engagement is a decrease of the tight coupling of oxidative phosphorylation and an increase in heat release. The other pathway is involved in energy dissipation in plant mitochondria and consists of members of the mitochondrial anion carrier protein superfamily, the UCPs, which mediate free fatty acid-activated, purine nucleotide-inhibited proton conductance (Echtay et al., 2002; Jarmuszkiewicz et al., 2010). The presence of UCPs creates an alternative way for the re-entry of protons into the mitochondrial matrix, and leads to uncoupled substrate oxidation from the phosphorylation of ADP to ATP to the release of energy as heat (Pecqueur et al., 2009). In animals, uncoupled mitochondrial respiration mediated by UCPs was considered to be involved with the control of the NAD+/NADH ratio and the regulation of metabolic pathways such as ketogenesis and amino acid synthesis (Ricquier and Bouillaud, 2000). Recently, Gandin et al. (2014) addressed the possible role of UCP1 and AOX1A in balancing the energy partitioning between nitrogen and carbon metabolism by feeding aox1a and ucp1 plants with and NH4+ (Gandin et al., 2014). The rate of foliar assimilation was enhanced in both aox1a and ucp1 mutants compared with the wild-type plants, suggesting that the decreased capacity of the mitochondrial electron transport chain and increased reductant within the cytosol probably contribute to driving foliar assimilation (Gandin et al., 2014). These results suggested the important role of AOX and UCP in maintaining cellular metabolism.
TEMPORAL AND SPATIAL EXPRESSION PATTERNS OF MEMBERS OF THE MEDP
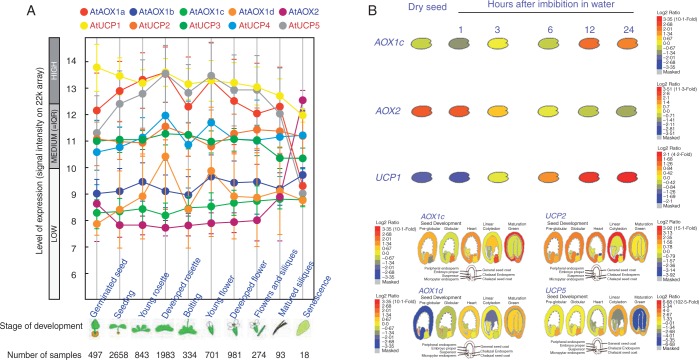
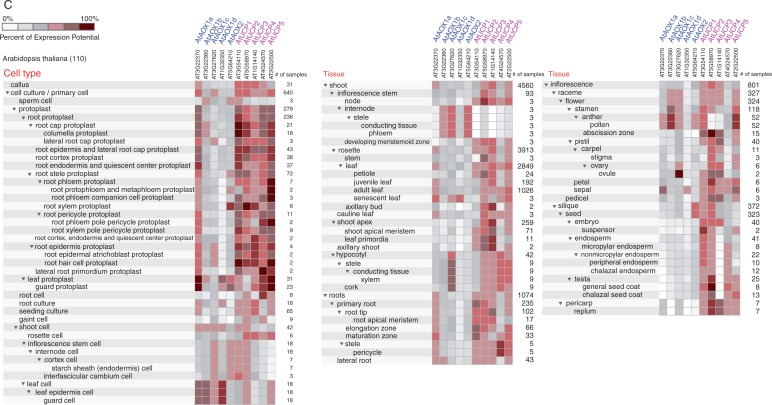
The availability of public databases, such as Genevestigator database (Zimmermann et al., 2004; Hruz et al., 2008) and BAR (Bio-Array Resource for plant biology) (Winter et al., 2007), provides opportunities to explore the expression and regulation of members of the MEDP in plants. The Development tool and Anatomy tool in the condition search tools of the Genevestigator database were used to examine the expression patterns of MEDP gene members at different developmental stages of the life cycle of Arabidopsis thaliana (arabidopsis) as well as in different tissues and organs (Fig. 3A, B). The expression of MEDP members during seed development is retrieved from the BAR database using the Arabidopsis eFP brower (Fig. 3C). The Co-Expression tool, Perturbations tool and Biclustering tool of the Genevestigator database (Hruz et al., 2008) were used to analyse the expression and regulation of members of the MEDP in arabidopsis. The expression levels of UCP genes in arabidopsis were higher than those of AOX genes in different developmental stages (Fig. 3A). Similarly, high transcript levels of five UCP genes were detected in most cell types and tissues; whereas, of five AOX genes, only AOX1A displayed high levels of expression similar to those of the UCP genes (Fig. 3B). Notably, the MEDP genes were differentially expressed in most tissues, and each gene displaying a distinct expression pattern. For instance, in the ovule, AOX1C was the predominant transcript, while AOX1D and AOX2 were predominant in pollen and seeds. A co-expression analysis based on the Co-Expression tool in Genevestigator (Hruz et al., 2008), further suggested that genes involved in embryo sac development arrest (AT3G60360), embryo defects (AT2G31340) and embryogenesis, such as the pentatricopeptide repeat-containing protein (AT3G49240), were co-expressed with AOX1C. Therefore, AOX1C may play a specific role in ovule development, which is consistent with a study that showed that AOX1C was regulated by growth and developmental signals (Ho et al., 2007). Unlike the other MEDP genes, the expression of UCP2 was ubiquitous in many tissues and organs, with higher expression levels occurring in the root protoplast, abscission zone, petal and seed coat; UCP4 was present at much lower levels than the other UCP genes, particularly in root endodermis and the quiescent centre (Fig. 3B). In a recent study, a transcription factor, UPBEAT1 (UPB1), was identified as a key regulator that modulates transition from proliferation to differentiation in the root cells by regulating ROS balance and distribution (Tsukagoshi et al., 2010). The upb1 mutants exhibit an altered accumulation of ROS, with reduced accumulation of hydrogen peroxide and increased accumulation of superoxide in the elongation zone (Tsukagoshi et al., 2010). The plants lacking upb1 have longer roots due to an increase in both meristem size and root cell length (Tsukagoshi et al., 2010), suggesting that alteration of the gradient and compartmentation of ROS in the root result in changes in the size of the meristem and in root length. Moreover, the size of the wild-type meristem was significantly affected by salicylhydroxamic acid (SHAM), an inhibitor of peroxidase activity as well as AOX, and KCN treatment (Tsukagoshi et al., 2010), indicating that AOX might be involved in the regulation of the size of the root meristem, and that upregulation of AOX is not sufficient to compensate effects of KCN on other antioxidant systems. Despite the fact that UCP4 (At4g24570) is not a direct target of UPB1, the UCP4 expression was significantly upregulated in UPB1-overexpressing plants (Tsukagoshi et al. (2010). Whether AOX genes and UCP4 are directly involved in the regulation of the root meristem needs further investigation. The highest expression levels of UCP3 were observed in the shoot apical meristem, auxiliary bud, leaf primordia and guard cell (Fig. 3B), and in stomatal closure under perturbations, suggesting that UCP3 may play a specific role in shoot meristem development. Interestingly, regulators responsible for stomatal development, such as the epidermal patterning factors EPF1 (Hara et al., 2007) and STOMAGEN (Sugano et al., 2009), as well as proteins involved in the regulation of metabolic fluxes during abscisic acid (ABA)-controlled seed development and germination (Pudelski et al., 2012), were co-expressed with UCP3. However, it remains unknown whether UCP3 is involved in the regulation of the shoot meristem. Nitric oxide (NO) and ROS are known to regulate stomatal closure in response to ABA in various species (Desikan et al., 2004). The inter-relationship between NO, ROS and AOX was confirmed in a study that found that the knock-down of AOX in tobacco plants led to increased content of NO in guard cells and decreased hydrogen peroxide levels compared with wild-type plants (Cvetkovska et al., 2014). While no noticeable impact on stomatal function was found in AOX knock-down plants, the repression of AOX reduced the response of guard cells to NO and compromised the ability of stomata to change the irradiance response (Cvetkovska et al., 2014), highlighting the role of AOX in stomata and NO homeostasis regulation. Therefore, the differential expression suggested that the MEDP genes might have specific functions in particular developmental stages.
Fig. 3.
Analysis of MEDP gene expression during development and organ distribution. (A) Genevestigator expression map describing the expression patterns of MEDP members during arabidopsis development. Growth stages from seed germination to senescence are arbitrarily grouped into sub-categories based on Boyes standard (Boyes et al., 2001). Each category contains averaged raw signal data and the standard error derived from all ATH1 chips hydridized with RNA from the corresponding growth stages in the Genevestigator database, including all organs available at that stage. Each of the arabidopsis growth stages is shown, and plant age in each category is indicated underneath the graph, as is the number of chips contributing to each category. Error bars represent the standard error. Data were retrieved from the Genevestigator database (https://genevestigator.com/gv/plant.jsp) using the Development tool. (B) Transcript expression of the MEDP members in wild-type arabidopsis. Relative expression level of MEDP members in plant tissues and organs and in particular cell types. Data were obtained from the Genevestigator database using the condition search tool set, the Anatomy tool. (C) The expression level of MEDP members during seed development and germination. The log2 ratio graphs on the right indicate the highest level of expression in red and the lowest level of expression in blue for each gene. The data were retrieved from the BAR database (http://bar.utoronto.ca/).
INVOLVEMENT OF THE MEDP IN POLLEN GERMINATION AND POLLEN TUBE GROWTH
The growth and development stages of plants have distinct cellular demands (Clifton et al., 2006). Co-ordination between the cellular energy status and gene expression is required to achieve specific development programmes. Thus, the increased expression of a specific sub-set of genes involved in cellular energy processes at particular growth stages or in specific tissues/organs is probably indicative of specific cellular energy demands. Pollen germination (PG) and pollen tube growth (PTG) are complex processes that requires high rates of energy flow (Rounds et al., 2011), the co-ordination of ion fluxes (Holdaway-Clarke and Hepler, 2003) and the regulation of ROS production (Martin et al., 2013; Lassig et al., 2014). As an energy mediator as well as a regulator of ROS production, it is tempting to speculate that the MEDP might play a role in these developmental processes. It has been suggested that the expression of AOX1B from arabidopsis was specific to floral tissues (Saisho et al., 1997; Clifton et al., 2006), while transcripts of AOX1D were highly expressed in stamens and pollen (Fig. 3B). Similarly, high expression levels of UCP5 were found in stamens and pollen, suggesting that UCP5 may play a role similar to AOX1D in floral development (Fig. 3B). Generation of an AOX1b/UCP5 double mutant would resolve this question and provide more insight into the role of MEDP genes in floral development. The links between the MEDP genes and PG and PTG were supported by a study in arabidopsis that found that the MEDP genes were upregulated during the transition from mature pollen grains to germinating pollen and to growing pollen tubes (Wang et al., 2008). In their study, Wang et al. (2008) showed that the expression of AOX1A, AOX1B, UCP1, UCP4 and UCP5 was detected during PG and PTG; the expression of UCP4 and UCP5 increased during PG, while increases in AOX1A and UCP4 expression were detected only during PTG, indicating that each stage of PG and PTG had different energy demands and growth rate oscillations. In a separate study of gene expression profiles associated with PG and PTG, 137 and 186 genes (both included AOX1A), respectively, were identified. The expression levels of all these genes increased during pollen hydration and PTG in vitro. The expression of AOX1A in 4 h PTG was upregulated >2-fold compared with its expression in dry pollen (Qin et al., 2009). It has been demonstrated that ion fluxes and transporters play critical roles in controlling PTG by controlling pollen tube polarity and also by providing sufficient materials for PTG (Hepler et al., 2001; Holdaway-Clarke and Hepler, 2003). In the pre-germination phase, the respiration rate is high with the conversion of sugar to starch; as PG continues, respiration slows (Rounds et al., 2011). The upregulation of transporters such as UCP4 and UCP5 during PG probably reflects an increased demand for nutrients for PG and PTG. Conversely, increased expression of AOX1A and UCP4 during PTG might be associated with the control of ROS accumulation. It was reported that ROS preferentially accumulated in central cells of the embryo sac before fertilization, and that stigma pollination triggered a localized oxidative burst that was restricted to the synergid cells of mature embryo sacs and was maintained until pollen tube arrival (Martin et al., 2013). Although an oxidative environment was postulated to be involved in pollen tube reception (Martin et al., 2013, 2014), the incorrect localization of ROS could result in developmental defects. In a mitochondrial manganese superoxide dismutase (MSD) mutant plant, the disruption of temporal and spatial ROS homeostasis along the embryo sac during development was found to lead to aberrant female gametophytes, because impaired MSD expression resulted in high levels of ROS in central cells, which further disturbed the determination of central cell fates (Martin et al., 2013). The production of ROS by pollen tube NAD(P)H oxidases has also been shown to be associated with stabilization of PTG rate oscillations (Lassig et al., 2014). These results suggested that the compartmentation and gradient of ROS is critical to initiate specific developmental processes, such as PG and PTG. The differential expression of MEDP genes at the PG and PTG stages may reflect specific respiration demands and a possible mechanism by which the balance of both ROS and NAD+ could be regulated precisely.
INVOLVEMENT OF THE MEDP IN SEED DEVELOPMENT AND FRUIT RIPENESS
Seed germination is a complex process that integrates environmental cues with internal signals to initiate a developmental programme that orchestrates the temporal and spatial expression of a set of genes. This process involves mitochondrial energy metabolism, hormone metabolism, RNA processing and degradation, protein synthesis, degradation and transportation, which re-establish mitochondrial respiratory metabolism and the subsequent emergence of radicals (Nakabayashi et al., 2005; Finch-Savage and Leubner-Metzger, 2006; Holdsworth et al., 2008; Nonogaki et al., 2010; Narsai et al., 2011). A prominent feature of seed germination is that genes related to energy metabolism and mitochondrial electron transport were activated specifically in the micropylar and chalazal endosperm (Dekkers et al., 2013), and it is accompanied by a rapid acceleration in oxygen consumption and mitochondrial respiration activity (Ehrenshaft and Brambl, 1990). The production of ROS has been observed during seed germination in various species (Oracz et al., 2009; Leymarie et al., 2012; Ishibashi et al., 2013) and was considered to act as a positive signal in seed dormancy release (El-Maarouf-Bouteau and Bailly, 2008). Based on these findings, it is reasonable to suppose that MEDP proteins such as AOXs and UCPs play roles in seed development. Indeed, from seed germination to maturation, the expression of MEDP genes occurs in a temporal- and spatial-specific manner. During germination stages, the AOX1A and AOX2 genes were found to be differentially expressed in dry seed and in stratified and germinating seedlings, whereas all the UCP genes were found to be differentially expressed during stratification, germination and post-germination (Narsai et al., 2011). The genome-wide network model that captured seed germination also showed that AOX1A was upregulated by dormancy in wheat, whereas UCP1 was upregulated by germination (Bassel et al., 2011). Furthermore, the transcripts of AOX1A and UCP5 decreased >3-fold from embryo emergence at around 24 h after imbibition in the light to 48 h after imbibition in the light. Conversely, the expression of UCP1 was upregulated >3-fold by stratification, reaching a maximum at 48 h stratification (Narsai et al., 2011). It has been suggested that the expression of AOX2 may be mature seed specific and that the expression decreased upon imbibition (Clifton et al., 2006), implying that AOX2 probably plays a role in mature seed development rather than seed germination. Direct evidence for the involvement of MEDP genes in seed development was demonstrated in a study that showed that the suppression of AOX2A expression in soybean resulted in reduced pod production, and thus seed yields (Chai et al., 2012). Interestingly, some MEDP genes were found in previously reported differentially expressed gene data sets, revealing transcriptomic changes associated with seed development, including arabidopsis seed germination, rice seed germination (Howell et al., 2009) and legume seed maturation (Verdier et al., 2013a, b). This result suggested that the upregulation or downregulation of mitochondrial energy components may be an evolutionarily conserved mechanism that is associated with the developmental processes that initiate seed germination or development.
Genes of the MEDP have also been demonstrated to be involved in the process of fruit ripening as regulators of cellular energy and ROS (Almeida et al., 1999; Considine et al., 2001; Xu et al., 2012). It was recently reported that explosive seed discharge in dwarf mistletoe is triggered by thermogenesis, and that the timing of explosive seed discharge in dwarf mistletoe corresponds to the presence of AOX in its mitochondria (deBruyn et al., 2015). It is therefore reasonable to suppose that AOX may play a critical role in triggering seed discharge in dwarf mistletoe. To the best of our knowledge, the MEDP may regulate ROS and the energy status of cells, and thus participate in these developmental processes.
LINKS BETWEEN MEDP MEMBERS, STRESS AND ENERGY SIGNALLING
Controlling energy and metabolic homeostasis is a great challenge for plants, and adaptations to stress conditions are typically linked to adjustment of cellular energy and/or metabolism (Baena-González and Sheen, 2008). In plants, two important protein kinases, plant Snf-1-related kinase 1 (SnRK1) homologue of yeast sucrose non-fermenting-1 (SNF1) kinase and the plant TOR kinase, are central regulators integrating plant growth, stress and energy signalling (Lastdrager et al., 2014; Tome et al., 2014). The arabidopsis SnRK superfamily consists of three distinct sub-families, i.e. the SnRK1, SnRK2 and SnRK3 sub-families. The SnRK2 and SnRK3 sub-family members have been suggested to be involved in ABA-mediated signalling and other signalling pathways that regulate plant response to abiotic stresses such as cold, drought and salt (Hey et al., 2010), whereas SnRK1 is a metabolic sensor that mediates an extensive metabolic reprogramming when plants are subjected to an energy crisis (Baena-González et al., 2007). In arabidopsis, the activation of KIN10, a catalytic subunit of SnRK1, initiates a global gene expression, promoting catabolism and suppressing anabolism (Baena-González et al., 2007). In addition to SnRK genes, MEDP members also play roles in stress and energy signalling, possibly by integrating ROS signalling with controlling the metabolic/redox state.
The expression of AOXs is known to be induced by a wide range of biotic and abiotic stresses (for reviews, see Vanlerberghe et al., 2009; Vanlerberghe, 2013), whereas the induction of UCPs by stress is less well characterized despite evidence from the Genevestigator microarray database suggesting that it might be involved in a range of biotic and abiotic stress responses (Fig. 4). The induced expression of MEDP genes under stress conditions is partly associated with the demand for decreasing ROS production and remodelling of the cellular metabolic status. Low temperature is known to reduce membrane fluidity by affecting the positional redistribution of saturated and unsaturated fatty acids within lipid molecules (Moon et al., 1995; Somerville, 1995), thereby reducing proton leakage, which can cause an increase in the potential for adenylate restriction of the electron transport chain (Atkin and Tjoelker, 2003). In this scenario, electron transport from ubiquinone to oxygen through AOX can occur without being restricted by adenylate control or the cellular ATP/ADP ratio, resulting in an increase in the respiration rate and heat release, which might, in turn, rescue membrane fluidity and growth. There is evidence that a chilling-sensitive maize cultivar showed higher electron partitioning through the alternative pathway compared with a chilling-tolerant cultivar when exposed to low temperatures (Ribas-Carbo et al., 2000), suggesting that upregulation of genes in the alternative pathway helped alleviate cold damage so as to sustain growth after recovery from chilling. Exposure of plants to low temperature results in accumulation of ROS (Suzuki and Mittler, 2006) and soluble sugars (Guy et al., 1992). Accumulation of ROS can cause oxidative damage, whereas accumulation of soluble sugars can protect plants against oxidative stress via their impact on ascorbate synthesis (Couée et al., 2006), as they can serve as a substrate for the synthesis of the antioxidant ascorbate (Smirnoff et al., 2001). Temperature acclimation is a complex process that involves extensive reprogramming of gene expression and metabolism (Stitt and Hurry, 2002; Zhu et al., 2007); for example, low temperature acclimation is closely associated with a selective stimulation of sucrose synthesis and re-establishment of a high rate of photosynthesis (Stitt and Hurry, 2002). Several studies have shown that MEDP genes play an important role under low temperature conditions. Suppressing AOX1A in arabidopsis plants leads to reduction of growth, with impaired leaf area development and smaller rosette size, whereas overexpressing it has the opposite effect at low temperatures (Fiorani et al., 2005). Interestingly, transgenic tobacco plants with enhanced AOX levels showed enhanced sugar accumulation upon exposure to low temperature (Wang et al., 2011). Furthermore, the compensatory upregulation of UCP1 expression was observed in an AOX1A arabidopsis mutant when it was exposed to low temperatures (Watanabe et al., 2008), suggesting that both AOX and UCP are involved in the response to low temperatures. It has been shown that MEDP members function at different time points following exposure of arabidopsis to cold, with increased alternative pathway activity being important only in the early stages of cold treatment (Armstrong et al., 2008), whereas an increase in UCP transcripts (Watanabe et al., 2008) and protein levels (Armstrong et al., 2008) was observed during long-term exposure to cold. It has also been shown that the transcripts of both AOX and UCP greatly increased in a chilling-tolerant tomato (Zhefen No. 208) but not in chilling-sensitive plants during chilling (Shi et al., 2013), which has been associated with a decrease in the mitochondrial production of ROS. It should be noted that the expression of some glycolysis-metabolic genes, such as PPi-dependent phosphofructokinase and sucrose synthase, was also upregulated in that study. The simultaneous engagement of the two energy dissipation systems would theoretically lead to greatly decreased ATP yields. Therefore, cytosol glycolysis-metabolic genes may be switched on to compensate for the loss of energy, reflecting the flexible energetic and metabolic cross-talk between the two organelles and the cytosol. Taken together, it is probable that AOX plays a significant role in short-term cold acclimation by providing plants with a means, independent of the energy state, to oxidize excess cellular reductant during early stages of cold stress and a way to maintain the respiration rate. Given that cold acclimation involves re-establishment of a high rate of photosynthesis (Stitt and Hurry, 2002), it is possible that induction of UCP transcripts and protein levels facilitate photosynthesis as the absence of UCP1 results in restriction of photorespiration, and thereby a decreased photosynthetic carbon assimilation rate (Sweetlove et al., 2006). It should be noted that the response of MEDP members to low temperature might be species/stress dependent; for example, in tomato, the alternative splicing event in AOX pre-mRNAs occurred preferentially at low temperature (Fung et al., 2006), whereas UCP but not AOX experienced such defective pre-mRNA processing when rice is exposured to cold environments (Watanabe and Hirai, 2002).
Fig. 4.
Clustering of responses of MEDP genes to a wide range of perturbations. Data are taken from Genevestigator using the Perturbations tool and Biclustering tool (Zimmermann et al., 2004, 2008). Red indicates that gene expression was induced by the treatment, and green indicates the opposite. A dendrogram is shown using the Euclidean distance.
In addition to low temperature, poor oxygen availability is also a factor that leads to a local energy crisis within the tissue (van Dongen and Licausi, 2015). Unlike animals, plants lack efficient systems to distribute oxygen to heterotrophic tissues, such as roots, tubers, meristems, germinating pollen and developing seeds, making them vulnerable to oxygen deprivation (Geigenberger, 2003; Mustroph et al., 2010). When plants suffer from oxygen deficiency, cytochrome oxidase activity becomes oxygen limited as the oxygen concentration drops, and ATP production through oxidative phosphorylation is inhibited (Geigenberger, 2003). Thus, hypoxia/anoxia restricts respiration and leads to a decrease in the ATP/ADP ratio and adenylate energy status (Geigenberger, 2003; Geigenberger et al., 2010). Low oxygen and/or re-oxygenation have also been shown to be associated with ROS signalling (van Dongen and Licausi, 2015), cellular metabolic remodelling (Branco-Price et al., 2008; Gupta et al., 2012; Shingaki-Wells et al., 2014) and mitochondrial responses, including the TCA cycle and respiratory chain (Shingaki-Wells et al., 2014). It is probable that MEDP members play roles in hypoxia/anoxia similar to that in low temperature. On the one hand, AOX was downregulated by oxygen deprivation at early stages due to its extreme affinity for oxygen (Igamberdiev et al., 2005), which may facilitate transient ROS accumulation. Accumulated ROS might act as a signal to trigger a signal cascade to activate genes involved in ROS scavenging (van Dongen and Licausi, 2015). On the other hand, hypoxia-induced production of NO inhibits aconitase and increases the level of citrate, which, in turn, induces the expression of AOX. The increased level of AOX expression might contribute to minimizing oxidative damage by regulating ROS production when ROS are extensively accumulated in the post-anoxic stages. Although there is little evidence that UCP is involved in the plant hypoxia/anoxia response, the induced expression of UCP4 and UCP5 is observed based on Genevestigator microarray analysis (Fig. 4C).
In animal cells, an involvement of UCPs in hypoxia/anoxia has been verified by several studies (Lu and Sack, 2008; Deng et al., 2012; Dromparis et al., 2013) and this is partly achieved by controlling ROS production (Lu and Sack, 2008; Deng et al., 2012), calcium transfer from the endoplasmic reticulum into the mitochondria (Dromparis et al., 2013) and possibly a metabolic switch toward glycolysis (Greer et al., 2012). In rice, hypoxia-induced elevation of cytosolic calcium is involved in anaerobic repression of AOX1A expression (Tsuji et al., 2000). Comparative analysis based on 21 organisms from four kingdoms has further revealed that low oxygen stress is involved in metabolic reconfigurations which enhance ATP production and NAD+ regeneration, and that some genes encoding the ROS network, such as UCP, appear to be conserved in both plants and animals in response to low oxygen conditions (Mustroph et al., 2010).
A co-expression analysis suggested that some WRKY transcription factors such as WRKY33 and WRKY40 are co-expressed with some AOX and UCP members (Fig. 2B), suggesting that AOX and UCP might share similar regulation mechanisms.
THE MEDP AS AN INTEGRATOR OF INTRAORGANELLE CROSS-TALK
The AOXs and UCPs provide plants with considerable metabolic flexibility by regulating cellular energy expenditure, and thus exert control over the cellular energy balance. In good agreement with this, under micronutrient- (phosphorus or nitrogen) limited conditions, the amounts of the AOX proteins were greatly increased in tobacco cells, whereas the lack of AOXs resulted in enhanced carbohydrate accumulation, suggesting that the upregulation of AOXs may help the cell to consume excess carbohydrates, thus maintaining the cellular carbon balance (Sieger et al., 2005). Similarly, the carbon to nitrogen ratio in an arabidopsis AOX1A mutant increased under low temperatures compared with the ratio in the wild-type plants (Watanabe et al., 2008), which indicated the role of AOX in maintaining cellular metabolic balance. Additional studies have suggested that the accumulation of NO in wild-type plants under hypoxia resulted in the inhibition of aconitase and accumulation of citrate, which in turn activated AOX and caused a shift of metabolism toward amino acid synthesis (Gupta et al., 2012). AOX is known to be activated by pyruvate or succinate, depending on whether a cysteine or serine is present in the conserved position (Millar et al., 1993; Holtzapffel et al., 2003; Umbach et al., 2006; Grant et al., 2009). This flexible biochemical control of AOX activity by specific α-keto acids might confer an additional layer of regulation for reprogramming cellular metabolism through integrated cellular compartment or organelle signals. Indeed, it was found that a decrease in cytosolic pyruvate in transgenic potato plants that exhibited silencing of pyruvate kinase genes resulted in the repression of the AOXs and changes in carbon partitioning, with glycolysis being diverted towards starch synthesis (Oliver et al., 2008). This result suggested that signals originating from alterations of glycolytic metabolism in the cytosol could be transmitted through AOX to the mitochondria, resulting in the readjustment of cellular metabolism. In animals, UCP2 has been identified as a key regulator of cellular fuel utilization and whole-body glucose and lipid metabolism (Diano and Horvath, 2012). It was shown recently that UCP2 may be involved in transportation of C4 metabolites out of the mitochondria (Vozza et al., 2014), making it possible for UCP2 to regulate influxes of the TCA cycle. The overexpression of arabidopsis UCP1 in tobacco was found to alter mitochondrial–nuclear communication, which resulted in decreased cellular ATP levels, and this in turn triggered a signal that initiated the expression of mitochondrial and stress-related genes (Barreto et al., 2014).
In addition to being a mediator that regulates cellular energy expenditure and metabolic balance, the involvement of AOXs in maintaining redox balance in chloroplasts has also been established. Under high light conditions, excess reducing equivalents were accumulated in the chloroplasts, which is concomitant with the upregulation of AOX, resulting in excess reductants that were dissipated in the mitochondria through an AOX and malate/oxalacetate shuttle (Yoshida et al., 2007; Noguchi and Yoshida, 2008; Taniguchi and Miyake, 2012). Interestingly, after etiolated seedlings were exposed to high light, delayed accumulation of chlorophyll and the increased accumulation of reducing reductant were observed in AOX1A mutant plants compared with the wild-type plants (Zhang et al., 2010), suggesting that AOXs play a role in optimizing photosynthesis. When cyanide-resistant respiration is inhibited, excess NADH accumulates in mitochondria. In this situation, excess NADH can be exported from mitochondria to chloroplasts, probably via the malate/oxaloacetate shuttle, resulting in an increase in the plastid NADPH/NADP ratio, which subsequently blocks the importation of plastidial protein (Zhang et al., 2014). This result suggests that the cross-talk between mitochondria and chloroplasts at the metabolic level is partly mediated by MEDP members. Furthermore, it was found that increased AOX activity correlated well with the elevation of intracellular pyruvate and that increased cellular pyruvate and malate levels paralleled increases in AOX under high light (Dinakar et al., 2010). These results suggested that the post-translational regulation of AOX by metabolites under high light may be critical to optimal photosynthesis.
As sensors for environmental perturbations and metabolic status, mitochondria not only can dissipate reducing equivalents, but also can generate reductants; in particular, in the photorespiratory process, a large number of NADHs were produced through the oxidation of glycine. The increased availability of NADH and electron flow into the mitochondrial electron transport chain through the photorespiratory pathway might result in the accumulation of NADH when the malate/oxalacetate shuttle cannot consume all of the NADH produced. This could result in the build-up a high proton gradient across the inner mitochondrial membrane when cellular ATP requirements are low, which will ultimately impose great constraints on electron flux and photorespiratory flux (Sweetlove et al., 2006). Under such conditions, up-regulation of MEDP genes could help to consume excess NADH, thus maintaining the cellular redox balance. The loss of UCP1 in arabidopsis restricted the photorespiratory flux of glycine to serine, suggesting that UCP allows NADH to be oxidized in the mitochondrial matrix by regulating the dissipation of the proton gradient across the inner mitochondrial membrane, thus ensuring continued photorespiration (Sweetlove et al., 2006). Interestingly, calcium-mediated signalling cross-talk in different organelles, including the endoplasmic reticulum (ER), mitochondria and nucleus, was also found to be involved in the induction of AOX1A in arabidopsis during salt stress conditions (Vanderauwera et al., 2012; Ng et al., 2014), suggesting that the ER might also be involved in plant mitochondrial retrograde regulation. Indeed, an analysis of transcription profiling using the Diff-Expression tools in Genevestigator revealed the differential gene expression of AOX1A in wild-type arabidopsis after treatment with tunicamycin compared with mock-treated (control) plants (Mishiba et al., 2013; Supplementary Data Table S5D), suggesting that an unfolded protein response or the ER stress response could induce the expression of AOX genes. Remarkably, the transcription factor bZIP60 involved in the unfolded protein response was found to be co-expressed with AOX1A and AOX1D under perturbations (Fig. 2B). Thus it is possible that bZIP60 could regulate the expression of AOX1A and AOX1D during ER stress. Together, these findings support the idea that interactions among different organelles are mediated partly through the MEDP by regulating NADH, NADPH or metabolite signals.
CROSS-TALK OF MEDP, PHYTOHORMONE AND CALCIUM SIGNALLING
Plant hormones regulate many physiological and developmental processes. They are also involved in integrating diverse environmental cues with a plant’s genetic programme (Santner and Estelle, 2009), and in cross-talk between stress signalling pathways (Fujita et al., 2006). ROS signalling has been shown to interact with plant hormone signalling (Fujita et al., 2006; Kwak et al., 2006). Hormone stability may be affected by unbalanced ROS accumulation (Kokkinakis and Brooks, 1979; Sewelam et al., 2014). The involvement of members of the MEDP in limiting the generation of ROS and in the adaptation of plants to stresses might be the link that integrates mitochondrial energy systems to hormone signalling. Indeed, it has been shown that some plant hormones, including salicylic acid (Lennon et al., 1997), ABA (Giraud et al., 2009; Liu et al., 2010), methyl salicylate and methyl jasmonate (Fung et al., 2004, 2006) and ethylene (Ederli et al., 2006; Wang et al., 2010), may be involved in the induction of AOXs, whereas auxin was shown to repress the induction of AOX1A by antimycin A (Ivanova et al., 2014). An examination of the Genevestigator database suggested that of the five UCP genes, UCP4 and UCP5 were stress-responsive genes and that the expression of UCP4 and UCP5 could be induced by ABA and auxin in arabidopsis seedlings (Fig. 4A). As an integrator of light and hormone, the HY5 transcription factor and its close homologue HYH have been suggested to play roles in suppressing auxin signalling (Sibout et al., 2006). Interestingly, the differential expression of UCP5 in HY5 and HYH mutants was observed, and its expression was further induced in a HY5/HYH double mutant (Supplementary Data Table S5A, B), suggesting that the derepression of HY5 and/or HYH on auxin signalling contributed to the expression of UCP genes. Therefore, auxin signalling and MEDP signalling were reciprocally regulated, depending on the growth and stress conditions.
Reactive oxygen species are signalling molecules that mediate a diverse range of cellular responses in plant cells, including development (Swanson and Gilroy, 2010), hormone signalling (Gapper and Dolan, 2006; Kwak et al., 2006), programmed cell death (Lam et al., 2001) and plant immunity (Kadota et al., 2014). Furthermore, ROS have been shown to interact directly or indirectly with other signalling pathways, such as NO and calcium signalling (Overmyer et al., 2003; Mittler et al., 2004; Mazars et al., 2010). Therefore, cross-talk between members of the MEDP and plant hormones may be mediated partly by ROS and/or calcium signals. Consistent with this finding, it has been observed that chilling-induced expression of AOX genes in arabidopsis callus was blocked completely by a calcium chelator and NADPH oxidase inhibitor (Wang et al., 2010, 2012).
Mitochondria, chloroplasts and peroxisomes are considered as sources of ROS in plants (Overmyer et al., 2003). The production of ROS was enhanced when plants were exposed to stress conditions (Mittler et al., 2004). Two sources of ROS production have been well characterized: metabolism-triggered ROS and enzyme-generating ROS. Metabolism-triggered ROS arise from metabolic imbalances after the changes in environmental conditions and/or electron leakage from the electron transportation chain of mitochondria and chloroplasts. Enzyme-generating ROS are produced by enzymes, most notably plant NADPH oxidases, also known as respiratory burst oxidase homologues (RBOHs) (Mittler et al., 2004; Suzuki et al., 2011; Vaahtera et al., 2014). The production of ROS by RBOHs was found to be associated closely with a variety of growth and development processes and stress responses (Suzuki et al., 2011), and RBOHD and RBOHF have been shown to be involved in the response of plants to biotic stresses (Drerup et al., 2013). Although the role of MEDP genes in dampening ROS production in mitochondria has been well characterized (Maxwell et al., 1999; Smith et al., 2004; Cvetkovska and Vanlerberghe, 2012), members of the MEDP were also found to be involved in protecting plants from pathogen infection and in decreasing the generation of ROS triggered by pathogen infection (Fu et al., 2010). These results suggested that the MEDP might play an additional role in limiting other non-mitochondrial sources of ROS. The exogenous application of hydrogen peroxide has been shown to increase the amount of AOX in many species and tissues (Vanlerberghe and Mclntosh, 1996; Ho et al., 2008), suggesting that the induction of AOX by hydrogen peroxide is not source specific. It has been proposed that cells activate their own ROS-producing mechanism, resulting in the generation of ROS, which in turn triggers an autopropagating wave in the adjacent cell (Mittler et al., 2011). ROS waves of this type have been suggested to depend largely on RBOHD and are considered to be an engine of systematic signalling (Gilroy et al., 2014). It has been suggested that ROS could be used as rapid, long-distance autopropagating signals that spread from the initial site to the entire plant via a cell to cell communication mechanism and that the ROS waves integrate with other signalling pathways, such as calcium, electrical and hormone signals (Miller et al., 2009; Mittler et al., 2011; Dubiella et al., 2013; Suzuki et al., 2013). In accordance with this idea, it has been shown that RBOHB, RBOHD and RBOHF can be phosphorylated by calcium-dependent protein kinases (CDPK4/5, CPK5) (Kobayashi et al., 2007; Dubiella et al., 2013) and calcineurin B-like protein-interacting protein kinase (CIPK26), respectively (Drerup et al., 2013; Kimura et al., 2013). Therefore, the regulation of ROS production by calcium signalling partly accounts for the reported inhibition of the expression of MEDP genes by the calcium chelator. Interestingly, we found that components involved in calcium signalling, such as the calcium-dependent protein kinases, calmodulin-like protein and calmodulin-binding protein, were co-expressed with MEDP genes under perturbations (Fig. 2C). This observation further suggested that the induced expression of members of the MEDP under stress conditions might be involved in both ROS production and calcium signals.
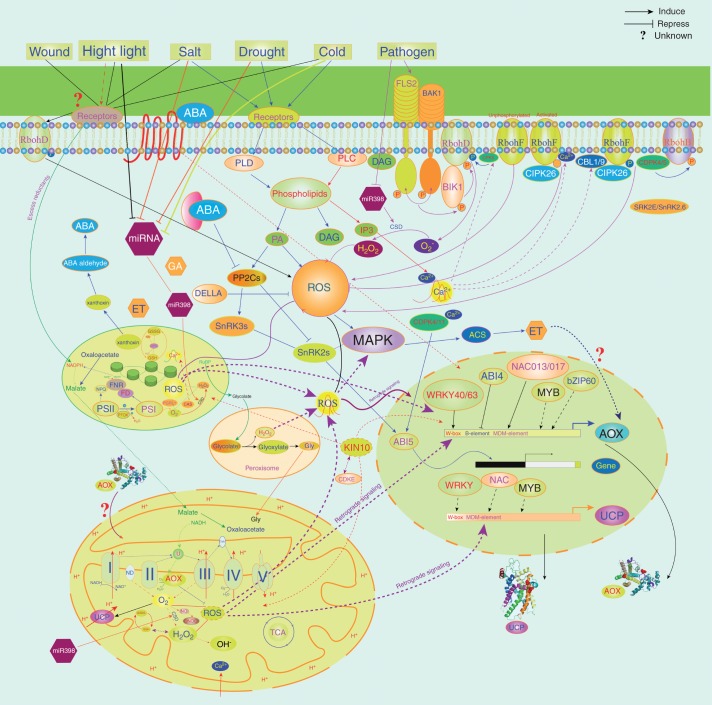
In addition to RBOH-derived ROS that induce the expression of MEDP genes, metabolic-derived ROS can also affect the transcription of MEDP genes. A recent study showed that different organelle-derived ROS differentially affected the expression of MEDP genes (Sewelam et al., 2014). It was found that the expression of AOX1D (At1g32350) was specifically upregulated after 8 h of induction of hydrogen peroxide in chloroplasts, whereas AOX1A was induced specifically by chloroplast-produced and peroxisomal-produced hydrogen peroxide (Sewelam et al., 2014), indicating that ROS might not induce the expression of MEDP genes directly. Several regulators involved in the regulation of AOX expression have been identified, including cyclin-dependent kinase E1 (CDKE1) (Ng et al., 2013a), WRKY40 and WRKY60 (Van Aken et al., 2013), and ANAC017 (Ng et al., 2013b). CDKE1 was shown to play a role in mitochondrial retrograde signalling and was proposed to integrate environmental signals with plant development (Ng et al., 2013a). CDKE1 was also predicted to interact with arabidopsis kinase 10 (KIN10), a central regulator involved in integrating stress, darkness and energy signalling with growth (Baena-González and Sheen, 2007, 2008). Therefore, it is possible that ROS produced in specific cellular compartments or organelles use a general signal to initiate and integrate different cellular signalling pathways of the cells and that ROS from different sources converge with mitochondrial retrograde regulation (MRR), thus deciding plant growth, development and the stress response. Moreover, as a player in MRR, members of the MEDP may, to some extent, act as sensors that transmit the signals from different organelles (e.g. chloroplass, mitochondria and peroxisomes) to the nucleus by sensing the balance between energy and NADPH or/and NADH levels. Furthermore, the small RNAs, including microRNAs and small interfering RNAs, have been shown to be involved in stress responses of plants, controlling ROS production and SnRK1-dependent energy signalling in arabidopsis (Khraiwesh et al., 2012; Confraria et al., 2013). Therefore, the regulation of the MEDP genes involves complex cross-talk between ROS signalling, calcium signalling, hormone signalling and possibly small RNAs (Fig. 5).
Fig. 5.
Signal cross-talk between the MEDP, ROS, calcium signalling and hormone signalling. Different stress signals coming from outside the cell are transmitted by a variety of messenger molecules, leading to alteration of the calcium signature and/or the production of ROS, which could trigger cell to cell communication events and ROS wave propagation between organelles (Mittler et al., 2011). This is concomitant with (de)activation of transcription factors, which, in turn, regulate the cellular antioxidant response. As mediators of ROS and energy, MEDP genes may play a role in controlling organellar ROS signalling, cellular redox and energy homeostasis.
CONCLUSIONS AND PERSPECTIVES
There is a strong evidence to support the role of ROS in influencing the status of cellular energy in plant development. Thus, the co-ordination of ROS production and their activities as well as cellular energy and/or redox balance are emerging as important themes in understanding how plant growth and development and stress responses are integrated. The involvement of members of the MEDP in lowering ROS production as well as in the regulation of cellular energy may, to some extent, provide a mechanism by which the developmental programme of plants is integrated with their cellular energy needs. However, the regulation of MEDP members is complex and occurs at the transcriptional level, translational level, post-translational level and metabolic level. How this regulation is linked to actual fluxes through the AOXs/UCPs in vivo remains elusive; for instance, the correlation between allosteric activation of AOX by organic acids and the in vivo electron flux to AOX has not been fully understood. A detailed analysis on the relationship between AOX activation state and electron flow to AOX is required.
SUPPLEMENTARY DATA
Supplementary data are available online at www.aob.oxfordjournals.org and consist of Tables S1–S4: gene sets co-expressed with AOX and UCP under perturbation (with positive and negative correlations). Table S5: differentially expressed gene sets between Col and hy5, Col and hyh, Col and the hy5-hyh double mutant, and control and tunicamycin-treated wild-type plants.
ACKNOWLEDGEMENTS
We apologize to the authors whose work was not cited, because of space limitations. This study was supported by the National Natural Science Foundation of China (31470342, 91417305, 31400211), the National Basic Research Program of China (973 Program) (2015CB150100) and Sichuan Natural Science Foundation (2015JY0101, 2015JY0223). We thank two anonymous reviewers for helpful comments on the paper.
LITERATURE CITED
- Albury MS, Elliott C, Moore AL. 2010. Ubiquinol-binding site in the alternative oxidase: mutagenesis reveals features important for substrate binding and inhibition. Biochimica et Biophysia Acta 1797: 1933–1939. [DOI] [PubMed] [Google Scholar]
- Almeida AM, Jarmuszkiewicz W, Khomsi H, Arruda P, Vercesi AE, Sluse FE. 1999. Cyanide-resistant, ATP-synthesis-sustained, and uncoupling-protein-sustained respiration during postharvest ripening of tomato fruit. Plant Physiology 119: 1323–1330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersson ME, Nordlund P. 1999. A revised model of the active site of alternative oxidase. FEBS Letters 449: 17–22. [DOI] [PubMed] [Google Scholar]
- Arsenijevic D, Onuma H, Pecqueur C, et al. 2000. Disruption of the uncoupling protein-2 gene in mice reveals a role in immunity and reactive oxygen species production. Nature Genetics 26: 435–439. [DOI] [PubMed] [Google Scholar]
- Armstrong AF, Badger MR, Day DA, et al. 2008. Dynamic changes in the mitochondrial electron transport chain underpinning cold acclimation of leaf respiration. Plant, Cell and Environment 31: 1156–1169. [DOI] [PubMed] [Google Scholar]
- Atkin OK, Tjoelker MG. 2003. Thermal acclimation and the dynamic response of plant respiration to temperature. Trends in Plant Science 8: 343–351. [DOI] [PubMed] [Google Scholar]
- Avin-Wittenberg T, Tzin V, Angelovici R, Galili G. 2012. Deciphering energy-associated gene networks operating in the response of Arabidopsis plants to stress and nutritional cues. The Plant Journal 70: 954–966. [DOI] [PubMed] [Google Scholar]
- Baena-González E, Rolland F, Thevelein JM, Sheen J. 2007. A central integrator of transcription networks in plant stress and energy signalling. Nature 448: 938–942. [DOI] [PubMed] [Google Scholar]
- Baena-González E, Sheen J. 2008. Convergent energy and stress signaling. Trends in Plant Science 13: 474–482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barreto P, Okura VK, Neshich IA, Maia Ide G, Arruda P. 2014. Overexpression of UCP1 in tobacco induces mitochondrial biogenesis and amplifies a broad stress response. BMC Plant Biology 14: 144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassel GW, Lan H, Glaab E, et al. 2011. Genome-wide network model capturing seed germination reveals coordinated regulation of plant cellular phase transitions. Proceedings of the National Academy of Sciences, USA 108: 9709–9714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berardi MJ, Shih WM, Harrison SC, Chou JJ. 2011. Mitochondrial uncoupling protein 2 structure determined by NMR molecular fragment searching. Nature 476: 109–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borisjuk L, Rolletschek H, Walenta S, Panitz R, Wobus U, Weber H. 2003. Energy status and its control on embryogenesis of legumes: ATP distribution within Vicia faba embryos is developmentally regulated and correlated with photosynthetic capacity. The Plant Journal 36: 318–329. [DOI] [PubMed] [Google Scholar]
- Bossi F, Cordoba E, Dupré P, Mendoza MS, Román CS, León P. 2009. The Arabidopsis ABA-INSENSITIVE (ABI) 4 factor acts as a central transcription activator of the expression of its own gene, and for the induction of ABI5 and SBE2.2 genes during sugar signaling. The Plant Journal 59: 359–374. [DOI] [PubMed] [Google Scholar]
- Boyes DC, Zayed AM, Ascenzi R, et al. 2001. Growth stage-based phenotypic analysis of Arabidopsis a model for high throughput functional genomics in plants. The Plant Cell 13: 1499–1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Branco-Price C, Kaiser KA, Jang CJH, Larive CK, Bailey-Serres J. 2008. Selective mRNA translation coordinates energetic and metabolic adjustments to cellular oxygen deprivation and reoxygenation in Arabidopsis thaliana. The Plant Journal 56: 743–755. [DOI] [PubMed] [Google Scholar]
- Caldana C, Li Y, Leisse A, et al. 2013. Systemic analysis of inducible target of rapamycin mutants reveal a general metabolic switch controlling growth in Arabidopsis thaliana. The Plant Journal 73: 897–909. [DOI] [PubMed] [Google Scholar]
- Chai TT, Simmonds D, Day DA, Colmer TD, Finnegan PM. 2012. A GmAOX2b antisense gene compromises vegetative growth and seed production in soybean. Planta 236: 199–207. [DOI] [PubMed] [Google Scholar]
- Clifton R, Millar AH, Whelan J. 2006. Alternative oxidases in Arabidopsis: a comparative analysis of differential expression in the gene family provides new insights into function of non-phosphorylating bypasses. Biochimica et Biophysica Acta 1757: 730–741. [DOI] [PubMed] [Google Scholar]
- Confraria A, Martinho C, Elias A, Rubio-Somoza I, Baena-Gonzalez E. 2013. miRNAs mediate SnRK1-dependent energy signaling in Arabidopsis. Frontiers in Plant Science 4: 197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Considine MJ, Daley DO, Whelan J. 2001. The expression of alternative oxidase and uncoupling protein during fruit ripening in mango. Plant Physiology 126: 1619–1629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Couée I, Sulmon C, Gouesbet G, El Amrani A. 2006. Involvement of soluble sugars in reactive oxygen species balance and responses to oxidative stress in plants. Journal of Experimental Botany 57: 449–459. [DOI] [PubMed] [Google Scholar]
- Cvetkovska M, Vanlerberghe GC. 2012. Alternative oxidase modulates leaf mitochondrial concentrations of superoxide and nitric oxide. New Phytologist 195: 32–39. [DOI] [PubMed] [Google Scholar]
- Cvetkovska M, Dahal K, Alber NA, Jin C, Cheung M, Vanlerberghe GC. 2014. Knockdown of mitochondrial alternative oxidase induces the ‘stress state’ of signaling molecule pools in Nicotiana tabacum, with implications for stomatal function. New Phytologist 203: 449–461. [DOI] [PubMed] [Google Scholar]
- deBruyn RA, Paetkau M, Ross KA, Godfrey DV, Friedman CR. 2015. Thermogenesis-triggered seed dispersal in dwarf mistletoe. Nature Communications 6: 6262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dekkers BJ, Pearce S, van Bolderen-Veldkamp RP, et al. 2013. Transcriptional dynamics of two seed compartments with opposing roles in Arabidopsis seed germination. Plant Physiology 163: 205–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng S, Yang Y, Han Y, et al. 2012. UCP2 inhibits ROS-mediated apoptosis in A549 under hypoxic conditions. PLoS One 7: e30714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desikan R, Cheung MK, Bright J, Henson D, Hancock JT, Neill SJ. 2004. ABA, hydrogen peroxide and nitric oxide signalling in stomatal guard cells. Journal of Experimental Botany 55: 205–212. [DOI] [PubMed] [Google Scholar]
- Diano S, Horvath TL. 2012. Mitochondrial uncoupling protein 2 (UCP2) in glucose and lipid metabolism. Trends in Molecular Medicine 18: 52–58. [DOI] [PubMed] [Google Scholar]
- Dinakar C, Raghavendra AS, Padmasree K. 2010. Importance of AOX pathway in optimizing photosynthesis under high light stress: role of pyruvate and malate in activating AOX. Physiologia Plantarum 139: 13–26. [DOI] [PubMed] [Google Scholar]
- van Dongen JT, Licausi F. 2015. Oxygen sensing and signaling. Annual Review of Plant Biology (in press). [DOI] [PubMed] [Google Scholar]
- Drerup MM, Schlucking K, Hashimoto K, et al. 2013. The calcineurin B-like calcium sensors CBL1 and CBL9 together with their interacting protein kinase CIPK26 regulate the Arabidopsis NADPH oxidase RBOHF. Molecular Plant 6: 559–569. [DOI] [PubMed] [Google Scholar]
- Dromparis P, Paulin R, Sutendra G, Qi AC, Bonnet S, Michelakis ED. 2013. Uncoupling protein 2 deficiency mimics the effects of hypoxia and endoplasmic reticulum stress on mitochondria and triggers pseudohypoxic pulmonary vascular remodeling and pulmonary hypertension. Circulation Research 113: 126–136. [DOI] [PubMed] [Google Scholar]
- Dubiella U, Seybold H, Durian G, et al. 2013. Calcium-dependent protein kinase/NADPH oxidase activation circuit is required for rapid defense signal propagation. Proceedings of the National Academy of Sciences, USA 110: 8744–8749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elthon TE, McIntosh L. 1986. Characterization and solubilization of the alternative oxidase of Sauromatum guttatum mitochondria. Plant Physiology 82: 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elthon TE, Nickels RL, McIntosh L. 1989. Monoclonal antibodies to the alternative oxidase of higher plant mitochondria. Plant Physiology 89: 1311–1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Echtay KS, Roussel D, St-Pierre J, et al. 2002. Superoxide activates mitochondrial uncoupling proteins. Nature 415: 96–99. [DOI] [PubMed] [Google Scholar]
- Ederli L, Morettini R, Borgogni A, et al. 2006. Interaction between nitric oxide and ethylene in the induction of alternative oxidase in ozone-treated tobacco plants. Plant Physiology 142: 595–608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Efremov RG, Baradaran R, Sazanov LA. 2010. The architecture of respiratory complex I. Nature 465: 441–445. [DOI] [PubMed] [Google Scholar]
- Ehrenshaft M, Brambl R. 1990. Respiration and mitochondrial biogenesis in germinating embryos of maize. Plant Physiology 93: 295–304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Maarouf-Bouteau H, Bailly C. 2008. Oxidative signaling in seed germination and dormancy. Plant Signaling and Behavoior 3: 175–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finch-Savage WE, Leubner-Metzger G. 2006. Seed dormancy and the control of germination. New Phytologist 171: 501–523. [DOI] [PubMed] [Google Scholar]
- Fiorani F, Umbach AL, Siedow JN. 2005. The alternative oxidase of plant mitochondria is involved in the acclimation of shoot growth at low temperature. A study of Arabidopsis AOX1a transgenic plants. Plant Physiology 139: 1795–1805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu LJ, Shi K, Gu M, et al. 2010. Systemic induction and role of mitochondrial alternative oxidase and nitric oxide in a compatible tomato–Tobacco mosaic virus interaction. Molecular Plant-Microbe Interactions 23: 39–48. [DOI] [PubMed] [Google Scholar]
- Fujita M, Fujita Y, Noutoshi Y, et al. 2006. Crosstalk between abiotic and biotic stress responses: a current view from the points of convergence in the stress signaling networks. Current Opinion in Plant Biology 9: 436–442. [DOI] [PubMed] [Google Scholar]
- Fung RW, Wang CY, Smith DL, Gross KC, Tao Y, Tian M. 2006. Characterization of alternative oxidase (AOX) gene expression in response to methyl salicylate and methyl jasmonate pre-treatment and low temperature in tomatoes. Journal of Plant Physiology 163: 1049–1060. [DOI] [PubMed] [Google Scholar]
- Fung RW, Wang CY, Smith DL, Gross KC, Tian M. 2004. MeSA and MeJA increase steady-state transcript levels of alternative oxidase and resistance against chilling injury in sweet peppers (Capsicum annuum L.). Plant Science 166: 711–719. [Google Scholar]
- Gandin A, Denysyuk M, Cousins AB. 2014. Disruption of the mitochondrial alternative oxidase (AOX) and uncoupling protein (UCP) alters rates of foliar nitrate and carbon assimilation in Arabidopsis thaliana. Journal of Experimental Botany 65: 3133–3142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gapper C, Dolan L. 2006. Control of plant development by reactive oxygen species. Plant Physiology 141: 341–345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geigenberger P. 2003. Response of plant metabolism to too little oxygen. Current Opinion in Plant Biology 6: 247–256. [DOI] [PubMed] [Google Scholar]
- Geigenberger P, Riewe D, Fernie AR. 2010. The central regulation of plant physiology by adenylates. Trends in Plant Science 15: 98–105. [DOI] [PubMed] [Google Scholar]
- Gibbons C, Montgomery MG, Leslie AG, Walker JE. 2000. The structure of the central stalk in bovine F1-ATPase at 2·4 Å resolution. Nature Structural and Molecular Biology 7: 1055–1061. [DOI] [PubMed] [Google Scholar]
- Gilroy S, Suzuki N, Miller G, et al. 2014. A tidal wave of signals: calcium and ROS at the forefront of rapid systemic signaling. Trends in Plant Science 19: 623–630. [DOI] [PubMed] [Google Scholar]
- Gimeno RE, Dembski M, Weng X, et al. 1997. Cloning and characterization of an uncoupling protein homolog: a potential molecular mediator of human thermogenesis. Diabetes 46: 900–906. [DOI] [PubMed] [Google Scholar]
- Giraud E, Van Aken O, Ho LHM, Whelan J. 2009. The transcription factor ABI4 is a regulator of mitochondrial retrograde expression of ALTERNATIVE OXIDASE1a. Plant Physiology 150: 1286–1296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grant N, Onda Y, Kakizaki Y, Ito K, Watling J, Robinson S. 2009. Two cys or not two cys? That is the question; alternative oxidase in the thermogenic plant sacred lotus. Plant Physiology 150: 987–995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greer SN, Metcalf JL, Wang Y, Ohh M. 2012. The updated biology of hypoxia-inducible factor. EMBO Journal 31: 2448–2460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta KJ, Shah JK, Brotman Y, et al. 2012. Inhibition of aconitase by nitric oxide leads to induction of the alternative oxidase and to a shift of metabolism towards biosynthesis of amino acids. Journal of Experimental Botany 63: 1773–1784. [DOI] [PubMed] [Google Scholar]
- Guy CL, Huber JL, Huber SC. 1992. Sucrose phosphate synthase and sucrose accumulation at low temperature. Plant Physiology 100: 502–508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara K, Kajita R, Torii KU, Bergmann DC, Kakimoto T. 2007. The secretory peptide gene EPF1 enforces the stomatal one-cell-spacing rule. Genes and Development, 21: 1720–1725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hepler PK, Vidali L, Cheung AY. 2001. Polarized cell growth in higher plants. Annual Review of Cell and Developmental Biology 17: 159–187. [DOI] [PubMed] [Google Scholar]
- Hey SJ, Byrne E, Halford NG. 2010. The interface between metabolic and stress signalling. Annals of Botany 105: 197–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho LH, Giraud E, Uggalla V, et al. 2008. Identification of regulatory pathways controlling gene expression of stress-responsive mitochondrial proteins in Arabidopsis. Plant Physiology 147: 1858–1873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho LHM, Giraud E, Lister R, et al. 2007. Characterization of the regulatory and expression context of an alternative oxidase gene provides insights into cyanide-insensitive respiration during growth and development. Plant Physiology 143: 1519–1533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holdaway-Clarke TL, Hepler PK. 2003. Control of pollen tube growth: role of ion gradients and fluxes. New Phytologist 159: 539–563. [DOI] [PubMed] [Google Scholar]
- Holdsworth MJ, Finch-Savage WE, Grappin P, Job D. 2008. Post-genomics dissection of seed dormancy and germination. Trends in Plant Science 13: 7–13. [DOI] [PubMed] [Google Scholar]
- Holtzapffel RC, Castelli J, Finnegan PM, Millar AH, Whelan J, Day DA. 2003. A tomato alternative oxidase protein with altered regulatory properties. Biochimica et Biophysica Acta 1606: 153–162. [DOI] [PubMed] [Google Scholar]
- Howell KA, Narsai R, Carroll A, et al. 2009. Mapping metabolic and transcript temporal switches during germination in rice highlights specific transcription factors and the role of RNA instability in the germination process. Plant Physiology 149: 961–980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hruz T, Laule O, Szabo G, et al. 2008. Genevestigator v3: a reference expression database for the meta-analysis of transcriptomes. Advances in Bioinformatics 2008: 420747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang S, Greenway H, Colmer TD, Millar AH. 2005. Protein synthesis by rice coleoptiles during prolonged anoxia: implications for glycolysis, growth and energy utilization. Annals of Botany 96: 703–715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huner N, Öquist G, Sarhan F. 1998. Energy balance and acclimation to light and cold. Trends in Plant Science 3: 224–230. [Google Scholar]
- Igamberdiev AU, Baron K, Manac’h-Little N, Stoimenova M, Hill RD. 2005. The haemoglobin/nitric oxide cycle: involvement in flooding stress and effects on hormone signalling. Annals of Botany 96: 557–564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishibashi Y, Koda Y, Zheng S-H, Yuasa T, Iwaya-Inoue M. 2013. Regulation of soybean seed germination through ethylene production in response to reactive oxygen species. Annals of Botany 111: 95–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivanova A, Law SR, Narsai R, et al. 2014. A functional antagonistic relationship between auxin and mitochondrial retrograde signaling regulates alternative oxidase1a expression in Arabidopsis. Plant Physiology 165: 1233–1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwata M, Lee Y, Yamashita T, et al. 2012. The structure of the yeast NADH dehydrogenase (Ndi1) reveals overlapping binding sites for water- and lipid-soluble substrates. Proceedings of the National Academy of Sciences, USA 109: 15247–15252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwata S, Lee JW, Okada K, et al. 1998. Complete structure of the 11-subunit bovine mitochondrial cytochrome bc1 complex. Science 281: 64–71. [DOI] [PubMed] [Google Scholar]
- Jarmuszkiewicz W, Woyda-Ploszczyca A, Antos-Krzeminska N, Sluse FE. 2010. Mitochondrial uncoupling proteins in unicellular eukaryotes. Biochimica et Biophysica Acta 1797: 792–799. [DOI] [PubMed] [Google Scholar]
- Kadota Y, Sklenar J, Derbyshire P, et al. 2014. Direct regulation of the NADPH oxidase RBOHD by the PRR-associated kinase BIK1 during plant immunity. Molecular Cell 54: 43–55. [DOI] [PubMed] [Google Scholar]
- Khraiwesh B, Zhu JK, Zhu J. 2012. Role of miRNAs and siRNAs in biotic and abiotic stress responses of plants. Biochimica et Biophysuca Acta 1819: 137–148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura S, Kawarazaki T, Nibori H, et al. 2013. The CBL-interacting protein kinase CIPK26 is a novel interactor of Arabidopsis NADPH oxidase AtRbohF that negatively modulates its ROS-producing activity in a heterologous expression system. Journal of Biochemistry 153: 191–195. [DOI] [PubMed] [Google Scholar]
- Kobayashi M, Ohura I, Kawakita K, et al. 2007. Calcium-dependent protein kinases regulate the production of reactive oxygen species by potato NADPH oxidase. The Plant Cell 19: 1065–1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kokkinakis DM, Brooks JL. 1979. Hydrogen peroxide-mediated oxidation of indole-3-acetic acid by tomato peroxidase and molecular oxygen. Plant Physiology 64: 220–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar AM, Soll D. 1992. Arabidopsis alternative oxidase sustains Escherichia coli respiration. Proceedings of the National Academy of Sciences, USA 89: 10842–10846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwak JM, Nguyen V, Schroeder JI. 2006. The role of reactive oxygen species in hormonal responses. Plant Physiology 141: 323–329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laloi M, Klein M, Riesmeier JW, et al. 1997. A plant cold-induced uncoupling protein. Nature 389: 135–136. [DOI] [PubMed] [Google Scholar]
- Lam E, Kato N, Lawton M. 2001. Programmed cell death, mitochondria and the plant hypersensitive response. Nature 411: 848–853. [DOI] [PubMed] [Google Scholar]
- Lassig R, Gutermuth T, Bey TD, Konrad KR, Romeis T. 2014. Pollen tube NAD (P) H oxidases act as a speed control to dampen growth rate oscillations during polarized cell growth. The Plant Journal 78: 94–106. [DOI] [PubMed] [Google Scholar]
- Lastdrager J, Hanson J, Smeekens S. 2014. Sugar signals and the control of plant growth and development. Journal of Experimental Botany 65: 799–807. [DOI] [PubMed] [Google Scholar]
- Lennon AM, Neuenschwander UH, Ribas-Carbo M, Giles L, Ryals JA, Siedow JN. 1997. The effects of salicylic acid and tobacco mosaic virus infection on the alternative oxidase of tobacco. Plant Physiology 115: 783–791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leymarie J, Vitkauskaite G, Hoang HH, et al. 2012. Role of reactive oxygen species in the regulation of Arabidopsis seed dormancy. Plant and Cell Physiology 53: 96–106. [DOI] [PubMed] [Google Scholar]
- Liu L, Cash TP, Jones RG, Keith B, Thompson CB, Simon MC. 2006. Hypoxia-induced energy stress regulates mRNA translation and cell growth. Molecular Cell 21: 521–531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, He J, Chen Z, Ren X, Hong X, Gong Z. 2010. ABA overly-sensitive 5 (ABO5), encoding a pentatricopeptide repeat protein required for cis-splicing of mitochondrial nad2 intron 3, is involved in the abscisic acid response in Arabidopsis. The Plant Journal 63: 749–765. [DOI] [PubMed] [Google Scholar]
- Lu Z, Sack MN. 2008. ATF-1 is a hypoxia-responsive transcriptional activator of skeletal muscle mitochondrial-uncoupling protein 3. Journal of Biological Chemistry 283: 23410–23418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lv X, Pu X, Qin G, Zhu T, Lin H. 2014. The roles of autophagy in development and stress responses in Arabidopsis thaliana. Apoptosis 19: 905–921. [DOI] [PubMed] [Google Scholar]
- Martin MV, Distefano AM, Bellido A, et al. 2014. Role of mitochondria during female gametophyte development and fertilization in A. thaliana. Mitochondrion 19B: 350–356. [DOI] [PubMed] [Google Scholar]
- Martin MV, Fiol DF, Sundaresan V, Zabaleta EJ, Pagnussat GC. 2013. oiwa, a female gametophytic mutant impaired in a mitochondrial manganese-superoxide dismutase, reveals crucial roles for reactive oxygen species during embryo sac development and fertilization in Arabidopsis. The Plant Cell 25: 1573–1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxwell DP, Wang Y, McIntosh L. 1999. The alternative oxidase lowers mitochondrial reactive oxygen production in plant cells. Proceedings of the National Academy of Sciences, USA 96: 8271–8276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazars C, Thuleau P, Lamotte O, Bourque S. 2010. Cross-talk between ROS and calcium in regulation of nuclear activities. Molecular Plant 3: 706–718. [DOI] [PubMed] [Google Scholar]
- Meeuse BJD. 1975. Thermogenic respiration in aroids. Annual Review of Plant Physiology 26: 117–126. [Google Scholar]
- Messerli MA, Amaral-Zettler LA, Zettler E, Jung S-K, Smith PJ, Sogin ML. 2005. Life at acidic pH imposes an increased energetic cost for a eukaryotic acidophile. Journal of Experimental Biology 208: 2569–2579. [DOI] [PubMed] [Google Scholar]
- Millar AH, Wiskich JT, Whelan J, Day DA. 1993. Organic acid activation of the alternatlve oxidase of plant mitochondria. FEBS Letters 329: 259–262. [DOI] [PubMed] [Google Scholar]
- Miller G, Schlauch K, Tam R, et al. 2009. The plant NADPH oxidase RBOHD mediates rapid systemic signaling in response to diverse stimuli. Science Signaling 2: ra45. [DOI] [PubMed] [Google Scholar]
- Mishiba K-i, Nagashima Y, Suzuki E, et al. 2013. Defects in IRE1 enhance cell death and fail to degrade mRNAs encoding secretory pathway proteins in the Arabidopsis unfolded protein response. Proceedings of the National Academy of Sciences, USA 110: 5713–5718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mittler R. 2002. Oxidative stress, antioxidants and stress tolerance. Trends in Plant Science 7: 405–410. [DOI] [PubMed] [Google Scholar]
- Mittler R, Vanderauwera S, Gollery M, Van Breusegem F. 2004. Reactive oxygen gene network of plants. Trends in Plant Science 9: 490–498. [DOI] [PubMed] [Google Scholar]
- Mittler R, Vanderauwera S, Suzuki N, et al. 2011. ROS signaling: the new wave? Trends in Plant Science 16: 300–309. [DOI] [PubMed] [Google Scholar]
- de Monet JBPA, Lamarck D, De Candolle AP. 1815. Flore française, ou descriptions succinctes de toutes les plantes qui croissent naturellement en France. Paris: Agasse. [Google Scholar]
- Moon BY, Higashi S, Gombos Z, Murata N. 1995. Unsaturation of the membrane lipids of chloroplasts stabilizes the photosynthetic machinery against low-temperature photoinhibition in transgenic tobacco plants. Proceedings of the National Academy of Sciences, USA 92: 6219–6223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore AL, Shiba T, Young L, Harada S, Kita K, Ito K. 2013. Unraveling the heater: new insights into the structure of the alternative oxidase. Annual Review of Plant Biology 64: 637–663. [DOI] [PubMed] [Google Scholar]
- Mustroph A, Lee SC, Oosumi T, et al. 2010. Cross-kingdom comparison of transcriptomic adjustments to low-oxygen stress highlights conserved and plant-specific responses. Plant Physiology 152: 1484–1500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakabayashi K, Okamoto M, Koshiba T, Kamiya Y, Nambara E. 2005. Genome-wide profiling of stored mRNA in Arabidopsis thaliana seed germination: epigenetic and genetic regulation of transcription in seed. The Plant Journal 41: 697–709. [DOI] [PubMed] [Google Scholar]
- Narsai R, Law SR, Carrie C, Xu L, Whelan J. 2011. In-depth temporal transcriptome profiling reveals a crucial developmental switch with roles for RNA processing and organelle metabolism that are essential for germination in Arabidopsis. Plant Physiology 157: 1342–1362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S, Giraud E, Duncan O, et al. 2013a. Cyclin-dependent kinase E1 (CDKE1) provides a cellular switch in plants between growth and stress responses. Journal of Biological Chemistry 288: 3449–3459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S, Ivanova A, Duncan O, et al. 2013b. A membrane-bound NAC transcription factor, ANAC017, mediates mitochondrial retrograde signaling in Arabidopsis. The Plant Cell 25: 3450–3471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S, De Clercq I, Van Aken O, et al. 2014. Anterograde and retrograde regulation of nuclear genes encoding mitochondrial proteins during growth, development, and stress. Molecular Plant 7: 1075–1093. [DOI] [PubMed] [Google Scholar]
- Noguchi K, Yoshida K. 2008. Interaction between photosynthesis and respiration in illuminated leaves. Mitochondrion 8: 87–99. [DOI] [PubMed] [Google Scholar]
- Nonogaki H, Bassel GW, Bewley JD. 2010. Germination – still a mystery. Plant Science 179: 574–581. [Google Scholar]
- Oliver SN, Lunn JE, Urbanczyk-Wochniak E, et al. 2008. Decreased expression of cytosolic pyruvate kinase in potato tubers leads to a decline in pyruvate resulting in an in vivo repression of the alternative oxidase. Plant Physiology 148: 1640–1654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oracz K, El-Maarouf-Bouteau H, Kranner I, Bogatek R, Corbineau F, Bailly C. 2009. The mechanisms involved in seed dormancy alleviation by hydrogen cyanide unravel the role of reactive oxygen species as key factors of cellular signaling during germination. Plant Physiology 150: 494–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Overmyer K, Brosché M, Kangasjärvi J. 2003. Reactive oxygen species and hormonal control of cell death. Trends in Plant Science 8: 335–342. [DOI] [PubMed] [Google Scholar]
- Pecqueur C, Alves-Guerra C, Ricquier D, Bouillaud F. 2009. UCP2, a metabolic sensor coupling glucose oxidation to mitochondrial metabolism? IUBMB Life 61: 762–767. [DOI] [PubMed] [Google Scholar]
- Pedersen BP, Kumar H, Waight AB, et al. 2013. Crystal structure of a eukaryotic phosphate transporter. Nature 496: 533–536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pudelski B, Schock A, Hoth S, et al. 2012. The plastid outer envelope protein OEP16 affects metabolic fluxes during ABA-controlled seed development and germination. Journal of Experimental Botany 63: 1919–1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin Y, Leydon AR, Manziello A, et al. 2009. Penetration of the stigma and style elicits a novel transcriptome in pollen tubes, pointing to genes critical for growth in a pistil. PLoS Genetics 5: e1000621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rastogi VK, Girvin ME. 1999. Structural changes linked to proton translocation by subunit c of the ATP synthase. Nature 402: 263–268. [DOI] [PubMed] [Google Scholar]
- Ribas-Carbo M, Aroca R, Gonzàlez-Meler MA, Irigoyen JJ, Sánchez-Díaz M. 2000. The electron partitioning between the cytochrome and alternative respiratory pathways during chilling recovery in two cultivars of maize differing in chilling sensitivity. Plant Physiology 122: 199–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ricquier D, Bouillaud F. 2000. The uncoupling protein homologues: UCP1, UCP2, UCP3, StUCP and AtUCP. Biochemical Journal 345: 161–179. [PMC free article] [PubMed] [Google Scholar]
- Rodrigues A, Adamo M, Crozet P, et al. 2013. ABI1 and PP2CA phosphatases are negative regulators of Snf1-related protein kinase1 signaling in Arabidopsis. The Plant Cell 25: 3871–3884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolletschek H, Weschke W, Weber H, Wobus U, Borisjuk L. 2004. Energy state and its control on seed development: starch accumulation is associated with high ATP and steep oxygen gradients within barley grains. Journal of Experimental Botany 55: 1351–1359. [DOI] [PubMed] [Google Scholar]
- Rounds CM, Winship LJ, Hepler PK. 2011. Pollen tube energetics: respiration, fermentation and the race to the ovule. AoB Plants 2011: plr019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruban AV, Berera R, Ilioaia C, et al. 2007. Identification of a mechanism of photoprotective energy dissipation in higher plants. Nature 450: 575–578. [DOI] [PubMed] [Google Scholar]
- Saisho D, Nambara E, Naito S, Tsutsumi N, Hirai A, Nakazono M. 1997. Characterization of the gene family for alternative oxidase from Arabidopsis thaliana. Plant Molecular Biology 35: 585–596. [DOI] [PubMed] [Google Scholar]
- Santner A, Estelle M. 2009. Recent advances and emerging trends in plant hormone signalling. Nature 459: 1071–1078. [DOI] [PubMed] [Google Scholar]
- Sewelam N, Jaspert N, Van Der Kelen K, et al. 2014. Spatial H2O2 signaling specificity: H2O2 from chloroplasts and peroxisomes modulates the plant transcriptome differentially. Molecular Plant 7: 1191–1210. [DOI] [PubMed] [Google Scholar]
- Shi K, Fu LJ, Zhang S, et al. 2013. Flexible change and cooperation between mitochondrial electron transport and cytosolic glycolysis as the basis for chilling tolerance in tomato plants. Planta 237: 589–601. [DOI] [PubMed] [Google Scholar]
- Shiba T, Kido Y, Sakamoto K, et al. 2013. Structure of the trypanosome cyanide-insensitive alternative oxidase. Proceedings of the National Academy of Sciences, USA 110: 4580–4585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shingaki-Wells R, Millar AH, Whelan J, Narsai R. 2014. What happens to plant mitochondria under low oxygen? An omics review of the responses to low oxygen and reoxygenation. Plant, Cell and Environment 37: 2260–2277. [DOI] [PubMed] [Google Scholar]
- Sibout R, Sukumar P, Hettiarachchi C, Holm M, Muday GK, Hardtke CS. 2006. Opposite root growth phenotypes of hy5 versus hy5 hyh mutants correlate with increased constitutive auxin signaling. PLoS Genetics 2: e202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siedow JN, Umbach AL, Moore AL. 1995. The active site of the cyanide-resistant oxidase from plant mitochondria contains a binuclear iron center. FEBS Letters 362: 10–14. [DOI] [PubMed] [Google Scholar]
- Sieger SM, Kristensen BK, Robson CA, et al. 2005. The role of alternative oxidase in modulating carbon use efficiency and growth during macronutrient stress in tobacco cells. Journal of Experimental Botany 56: 1499–1515. [DOI] [PubMed] [Google Scholar]
- Sluse FE, Jarmuszkiewicz W. 2002. Uncoupling proteins outside the animal and plant kingdoms: functional and evolutionary aspects. FEBS Letters 510: 117–120. [DOI] [PubMed] [Google Scholar]
- Smirnoff N, Conklin PL, Loewus FA. 2001. Biosynthesis of ascorbic acid in plants: a renaissance. Annual Review of Plant Biology 52: 437–467. [DOI] [PubMed] [Google Scholar]
- Smith AM, Ratcliffe RG, Sweetlove LJ. 2004. Activation and function of mitochondrial uncoupling protein in plants. Journal of Biological Chemistry 279: 51944–51952. [DOI] [PubMed] [Google Scholar]
- Somerville C. 1995. Direct tests of the role of membrane lipid composition in low-temperature-induced photoinhibition and chilling sensitivity in plants and cyanobacteria. Proceedings of the National Academy of Sciences, USA 92: 6215–6218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stitt M, Hurry V. 2002. A plant for all seasons: alterations in photosynthetic carbon metabolism during cold acclimation in Arabidopsis. Current Opinion in Plant Biology 5: 199–206. [DOI] [PubMed] [Google Scholar]
- Stupnikova I, Benamar A, Tolleter D, et al. 2006. Pea seed mitochondria are endowed with a remarkable tolerance to extreme physiological temperatures. Plant Physiology 140: 326–335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugano SS, Shimada T, Imai Y, et al. 2009. Stomagen positively regulates stomatal density in Arabidopsis. Nature 463: 241–244. [DOI] [PubMed] [Google Scholar]
- Sun F, Huo X, Zhai Y, et al. 2005. Crystal structure of mitochondrial respiratory membrane protein complex II. Cell 121: 1043–1057. [DOI] [PubMed] [Google Scholar]
- Suzuki N, Mittler R. 2006. Reactive oxygen species and temperature stresses: a delicate balance between signaling and destruction. Physiologia Plantarum, 126: 45–51. [Google Scholar]
- Suzuki N, Miller G, Morales J, Shulaev V, Torres MA, Mittler R. 2011. Respiratory burst oxidases: the engines of ROS signaling. Current Opinion in Plant Biology 14: 691–699. [DOI] [PubMed] [Google Scholar]
- Suzuki N, Koussevitzky S, Mittler R, Miller G. 2012. ROS and redox signalling in the response of plants to abiotic stress. Plant, Cell and Environment 35: 259–270. [DOI] [PubMed] [Google Scholar]
- Suzuki N, Miller G, Salazar C, et al. 2013. Temporal–spatial interaction between reactive oxygen species and abscisic acid regulates rapid systemic acclimation in plants. The Plant Cell 25: 3553–3569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swanson S, Gilroy S. 2010. ROS in plant development. Physiologia Plantarum 138: 384–392. [DOI] [PubMed] [Google Scholar]
- Sweetlove LJ, Lytovchenko A, Morgan M, et al. 2006. Mitochondrial uncoupling protein is required for efficient photosynthesis. Proceedings of the National Academy of Sciences, USA 103: 19587–19592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szabo I, Bergantino E, Giacometti GM. 2005. Light and oxygenic photosynthesis: energy dissipation as a protection mechanism against photo-oxidation. EMBO Reports 6: 629–634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniguchi M, Miyake H. 2012. Redox-shuttling between chloroplast and cytosol: integration of intra-chloroplast and extra-chloroplast metabolism. Current Opinion in Plant Biology 15: 252–260. [DOI] [PubMed] [Google Scholar]
- Tome F, Nagele T, Adamo M, et al. 2014. The low energy signaling network. Frontiers in Plant Science 5: 353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuji H, Nakazono M, Saisho D, Tsutsumi N, Hirai A. 2000. Transcript levels of the nuclear-encoded respiratory genes in rice decrease by oxygen deprivation: evidence for involvement of calcium in expression of the alternative oxidase 1a gene. FEBS Letters 471: 201–204. [DOI] [PubMed] [Google Scholar]
- Tsukagoshi H, Busch W, Benfey PN. 2010. Transcriptional regulation of ROS controls transition from proliferation to differentiation in the root. Cell 143: 606–616. [DOI] [PubMed] [Google Scholar]
- Umbach AL, Siedow JN. 1993. Covalent and noncovalent dimers of the cyanide-resistant alternative oxidase protein in higher-plant mitochondria and their relationship to enzyme-activity. Plant Physiology 103: 845–854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umbach AL, Ng VS, Siedow JN. 2006. Regulation of plant alternative oxidase activity: a tale of two cysteines. Biochimica et Biophysica Acta 1757: 135–142. [DOI] [PubMed] [Google Scholar]
- Vaahtera L, Brosche M, Wrzaczek M, Kangasjarvi J. 2014. Specificity in ROS signaling and transcript signatures. Antioxidants and Redox Signaling 21: 1422–1441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Aken O, Zhang B, Law S, Narsai R, Whelan J. 2013. AtWRKY40 and AtWRKY63 modulate the expression of stress-responsive nuclear genes encoding mitochondrial and chloroplast proteins. Plant Physiology 162: 254–271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanderauwera S, Vandenbroucke K, Inzé A, et al. 2012. AtWRKY15 perturbation abolishes the mitochondrial stress response that steers osmotic stress tolerance in Arabidopsis. Proceedings of the National Academy of Sciences, USA 109: 20113–20118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanlerberghe GC. 2013. Alternative oxidase: a mitochondrial respiratory pathway to maintain metabolic and signaling homeostasis during abiotic and biotic stress in plants. International Journal of Molecular Sciences 14: 6805–6847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanlerberghe GC, Mclntosh L. 1996. Signals regulating the expression of the nuclear gene encoding alternative oxidase of plant mitochondria. Plant Physiology 111: 589–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanlerberghe GC, McIntosh L. 1997. Alternative oxidase: from gene to function. Annual Review of Plant Biology 48: 703–734. [DOI] [PubMed] [Google Scholar]
- Vanlerberghe GC, Cvetkovska M, Wang J. 2009. Is the maintenance of homeostatic mitochondrial signaling during stress a physiological role for alternative oxidase? Physiologia Plantarum 137: 392–406. [DOI] [PubMed] [Google Scholar]
- Vercesi AE, Borecký J, Maia Ide G, Arruda P, Cuccovia IM, Chaimovich H. 2006. Plant uncoupling mitochondrial proteins. Annual Review of Plant Biology 57: 383–404. [DOI] [PubMed] [Google Scholar]
- Verdier J, Lalanne D, Pelletier S, et al. 2013a. A regulatory network-based approach dissects late maturation processes related to the acquisition of desiccation tolerance and longevity of Medicago truncatula seeds. Plant Physiology 163: 757–774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verdier J, Torres-Jerez I, Wang M, et al. 2013b. Establishment of the Lotus japonicus Gene Expression Atlas (LjGEA) and its use to explore legume seed maturation. The Plant Journal 74: 351–362. [DOI] [PubMed] [Google Scholar]
- Vozza A, Parisi G, De Leonardis F, et al. 2014. UCP2 transports C4 metabolites out of mitochondria, regulating glucose and glutamine oxidation. Proceedings of the National Academy of Sciences, USA 111: 960–965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallimann T, Wyss M, Brdiczka D, Nicolay K, Eppenberger H. 1992. Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: the ‘phosphocreatine circuit’for cellular energy homeostasis. Biochemical Journal 281: 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Liang X, Huang J, et al. 2010. Involvement of ethylene and hydrogen peroxide in induction of alternative respiratory pathway in salt-treated Arabidopsis calluses. Plant and Cell Physiology 51: 1754–1765. [DOI] [PubMed] [Google Scholar]
- Wang H, Huang J, Liang X, Bi Y. 2012. Involvement of hydrogen peroxide, calcium, and ethylene in the induction of the alternative pathway in chilling-stressed Arabidopsis callus. Planta 235: 53–67. [DOI] [PubMed] [Google Scholar]
- Wang J, Rajakulendran N, Amirsadeghi S, Vanlerberghe GC. 2011. Impact of mitochondrial alternative oxidase expression on the response of Nicotiana tabacum to cold temperature. Physiologia Plantarum 142: 339–351. [DOI] [PubMed] [Google Scholar]
- Wang Y, Zhang W-Z, Song L-F, Zou J-J, Su Z, Wu W-H. 2008. Transcriptome analyses show changes in gene expression to accompany pollen germination and tube growth in Arabidopsis. Plant Physiology 148: 1201–1211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe A, Hirai A. 2002. Two uncoupling protein genes of rice (Oryza sativa L.): molecular study reveals the defects in the pre-mRNA processing for the heat-generating proteins of the subtropical cereal. Planta 215: 90–100. [DOI] [PubMed] [Google Scholar]
- Watanabe CK, Hachiya T, Terashima I, Noguchi K. 2008. The lack of alternative oxidase at low temperature leads to a disruption of the balance in carbon and nitrogen metabolism, and to an up-regulation of antioxidant defence systems in Arabidopsis thaliana leaves. Plant, Cell and Environment 31: 1190–1202. [DOI] [PubMed] [Google Scholar]
- Wilson KE, Ivanov AG, Öquist G, Grodzinski B, Sarhan F, Huner NP. 2006. Energy balance, organellar redox status, and acclimation to environmental stress. Botany 84: 1355–1370. [Google Scholar]
- Winter D, Vinegar B, Nahal H, Ammar R, Wilson GV, Provart NJ. 2007. An ‘Electronic Fluorescent Pictograph’ browser for exploring and analyzing large-scale biological data sets. PLoS One 2: e718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu F, Yuan S, Zhang DW, Lv X, Lin HH. 2012. The role of alternative oxidase in tomato fruit ripening and its regulatory interaction with ethylene. Journal of Experimental Botany 63: 5705–5716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida K, Terashima I, Noguchi K. 2007. Up-regulation of mitochondrial alternative oxidase concomitant with chloroplast over-reduction by excess light. Plant and Cell Physiology 48: 606–614. [DOI] [PubMed] [Google Scholar]
- Yoshikawa S, Shinzawa-Itoh K, Nakashima R, et al. 1998. Redox-coupled crystal structural changes in bovine heart cytochrome c oxidase. Science 280: 1723–1729. [DOI] [PubMed] [Google Scholar]
- Zhang DW, Xu F, Zhang ZW, et al. 2010. Effects of light on cyanide-resistant respiration and alternative oxidase function in Arabidopsis seedlings. Plant, Cell and Environment 33: 2121–2131. [DOI] [PubMed] [Google Scholar]
- Zhang DW, Yuan S, Xu F, et al. 2014. Light intensity affects chlorophyll synthesis during greening process by metabolite signal from mitochondrial alternative oxidase in Arabidopsis. Plant, Cell and Environment (in press). [DOI] [PubMed] [Google Scholar]
- Zhu J, Dong C-H, Zhu J-K. 2007. Interplay between cold-responsive gene regulation, metabolism and RNA processing during plant cold acclimation. Current Opinion in Plant Biology 10: 290–295. [DOI] [PubMed] [Google Scholar]
- Zimmermann P, Hirsch-Hoffmann M, Hennig L, Gruissem W. 2004. GENEVESTIGATOR. Arabidopsis microarray database and analysis toolbox. Plant Physiology 136: 2621–2632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmermann P, Laule O, Schmitz J, Hruz T, Bleuler S, Gruissem W. 2008. Genevestigator transcriptome meta-analysis and biomarker search using rice and barley gene expression databases. Molecular Plant 1: 851–857. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.