Abstract
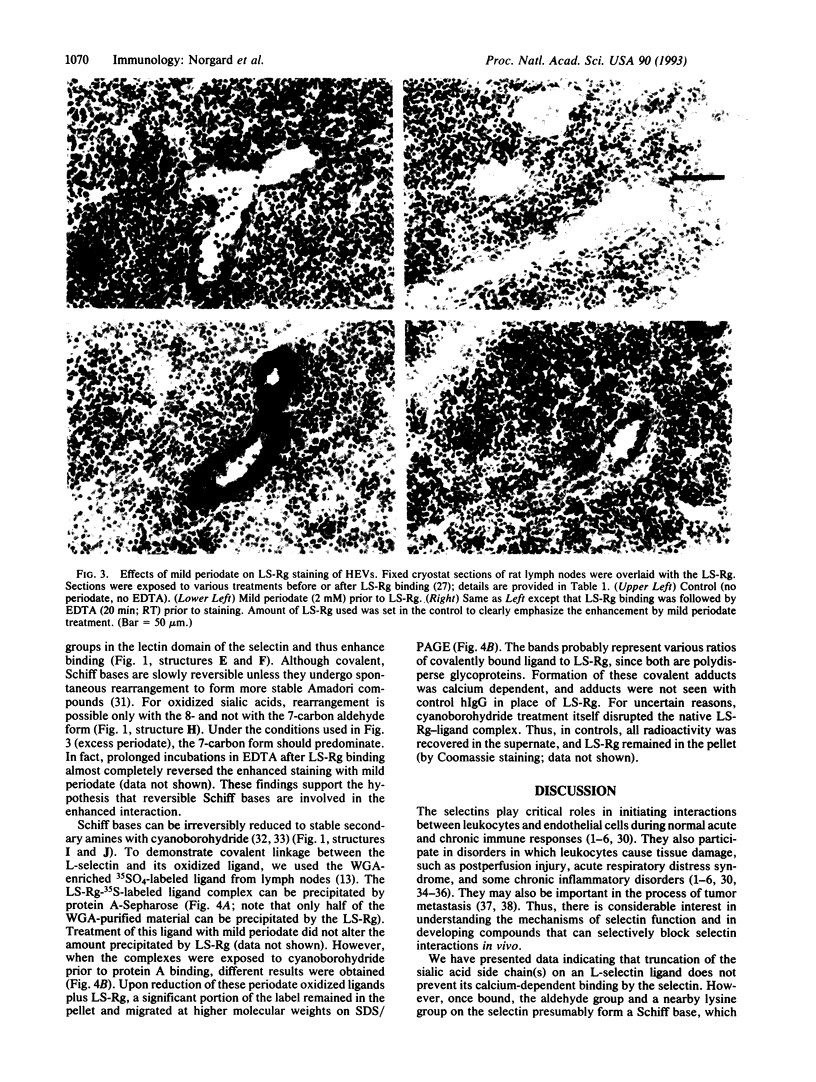
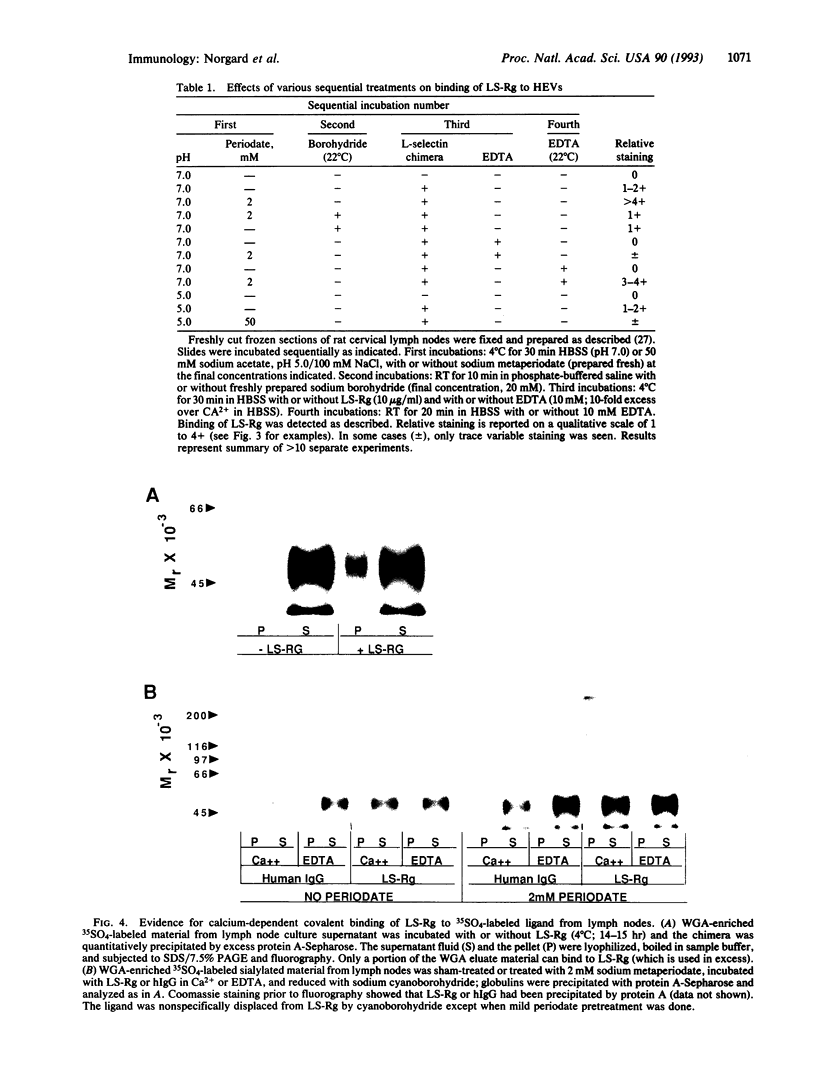
The selectins are adhesion receptors that play key roles in leukocyte trafficking. Each has an N-terminal C-type lectin domain that binds to specific carbohydrates in a calcium-dependent manner. L-selectin recognizes sulfated, sialylated ligands on lymph node high endothelial venules. This recognition is abolished by strong periodate oxidation under conditions that destroy oligosaccharides. In contrast, mild periodate oxidation, which selectively oxidizes the side chain of sialic acid residues without affecting the underlying oligosaccharide, markedly enhances this interaction. The enhancement is calcium dependent, indicating that lectin recognition is maintained. Reduction of the sialic acid aldehydes generated by mild periodate to alcohol groups abolishes this effect. Covalent cross-linking of the oxidized ligand to L-selectin can be demonstrated, suggesting Schiff base formation between lysine residues of the selectin and the newly formed aldehydes. Such selectively oxidized sialylated ligands could be used to probe the lectin domains of the selectins and to identify lysine residues near the binding site. Also, this approach could be used to design drugs for disrupting leukocyte-endothelial interactions leading to pathological inflammation.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brandley B. K., Swiedler S. J., Robbins P. W. Carbohydrate ligands of the LEC cell adhesion molecules. Cell. 1990 Nov 30;63(5):861–863. doi: 10.1016/0092-8674(90)90487-y. [DOI] [PubMed] [Google Scholar]
- Böyum A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand J Clin Lab Invest Suppl. 1968;97:77–89. [PubMed] [Google Scholar]
- Cheresh D. A., Reisfeld R. A., Varki A. P. O-acetylation of disialoganglioside GD3 by human melanoma cells creates a unique antigenic determinant. Science. 1984 Aug 24;225(4664):844–846. doi: 10.1126/science.6206564. [DOI] [PubMed] [Google Scholar]
- Foxall C., Watson S. R., Dowbenko D., Fennie C., Lasky L. A., Kiso M., Hasegawa A., Asa D., Brandley B. K. The three members of the selectin receptor family recognize a common carbohydrate epitope, the sialyl Lewis(x) oligosaccharide. J Cell Biol. 1992 May;117(4):895–902. doi: 10.1083/jcb.117.4.895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gundel R. H., Wegner C. D., Torcellini C. A., Clarke C. C., Haynes N., Rothlein R., Smith C. W., Letts L. G. Endothelial leukocyte adhesion molecule-1 mediates antigen-induced acute airway inflammation and late-phase airway obstruction in monkeys. J Clin Invest. 1991 Oct;88(4):1407–1411. doi: 10.1172/JCI115447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imai Y., Singer M. S., Fennie C., Lasky L. A., Rosen S. D. Identification of a carbohydrate-based endothelial ligand for a lymphocyte homing receptor. J Cell Biol. 1991 Jun;113(5):1213–1221. doi: 10.1083/jcb.113.5.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jentoft N., Dearborn D. G. Labeling of proteins by reductive methylation using sodium cyanoborohydride. J Biol Chem. 1979 Jun 10;254(11):4359–4365. [PubMed] [Google Scholar]
- Knibbs R. N., Goldstein I. J., Ratcliffe R. M., Shibuya N. Characterization of the carbohydrate binding specificity of the leukoagglutinating lectin from Maackia amurensis. Comparison with other sialic acid-specific lectins. J Biol Chem. 1991 Jan 5;266(1):83–88. [PubMed] [Google Scholar]
- Kojima N., Handa K., Newman W., Hakomori S. Inhibition of selectin-dependent tumor cell adhesion to endothelial cells and platelets by blocking O-glycosylation of these cells. Biochem Biophys Res Commun. 1992 Feb 14;182(3):1288–1295. doi: 10.1016/0006-291x(92)91872-n. [DOI] [PubMed] [Google Scholar]
- Lasky L. A. Lectin cell adhesion molecules (LEC-CAMs): a new family of cell adhesion proteins involved with inflammation. J Cell Biochem. 1991 Feb;45(2):139–146. doi: 10.1002/jcb.240450204. [DOI] [PubMed] [Google Scholar]
- Lasky L. A. Selectins: interpreters of cell-specific carbohydrate information during inflammation. Science. 1992 Nov 6;258(5084):964–969. doi: 10.1126/science.1439808. [DOI] [PubMed] [Google Scholar]
- Lasky L. A., Singer M. S., Dowbenko D., Imai Y., Henzel W. J., Grimley C., Fennie C., Gillett N., Watson S. R., Rosen S. D. An endothelial ligand for L-selectin is a novel mucin-like molecule. Cell. 1992 Jun 12;69(6):927–938. doi: 10.1016/0092-8674(92)90612-g. [DOI] [PubMed] [Google Scholar]
- Leung D. Y., Pober J. S., Cotran R. S. Expression of endothelial-leukocyte adhesion molecule-1 in elicited late phase allergic reactions. J Clin Invest. 1991 May;87(5):1805–1809. doi: 10.1172/JCI115201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matrosovich M. N., Gambaryan A. S., Reizin F. N., Chumakov M. P. Recognition by human A and B influenza viruses of 8- and 7-carbon analogues of sialic acid modified in the polyhydroxyl side chain. Virology. 1991 Jun;182(2):879–882. doi: 10.1016/0042-6822(91)90634-n. [DOI] [PubMed] [Google Scholar]
- McEver R. P. Leukocyte-endothelial cell interactions. Curr Opin Cell Biol. 1992 Oct;4(5):840–849. doi: 10.1016/0955-0674(92)90109-p. [DOI] [PubMed] [Google Scholar]
- McLean R. L., Suttajit M., Beidler J., Winzler R. J. N-acetylneuraminic acid analogues. I. Preparation of the 8-carbon and 7-carbon compounds. J Biol Chem. 1971 Feb 10;246(3):803–809. [PubMed] [Google Scholar]
- Moore K. L., Stults N. L., Diaz S., Smith D. F., Cummings R. D., Varki A., McEver R. P. Identification of a specific glycoprotein ligand for P-selectin (CD62) on myeloid cells. J Cell Biol. 1992 Jul;118(2):445–456. doi: 10.1083/jcb.118.2.445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulligan M. S., Varani J., Dame M. K., Lane C. L., Smith C. W., Anderson D. C., Ward P. A. Role of endothelial-leukocyte adhesion molecule 1 (ELAM-1) in neutrophil-mediated lung injury in rats. J Clin Invest. 1991 Oct;88(4):1396–1406. doi: 10.1172/JCI115446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray M. C., Bhavanandan V. P., Davidson E. A., Reinhold V. Modification of sialyl residues of glycoconjugates by reductive amination. Characterization of the modified sialic acids. Carbohydr Res. 1989 Mar 15;186(2):255–265. doi: 10.1016/0008-6215(89)84039-x. [DOI] [PubMed] [Google Scholar]
- Picker L. J., Butcher E. C. Physiological and molecular mechanisms of lymphocyte homing. Annu Rev Immunol. 1992;10:561–591. doi: 10.1146/annurev.iy.10.040192.003021. [DOI] [PubMed] [Google Scholar]
- Polley M. J., Phillips M. L., Wayner E., Nudelman E., Singhal A. K., Hakomori S., Paulson J. C. CD62 and endothelial cell-leukocyte adhesion molecule 1 (ELAM-1) recognize the same carbohydrate ligand, sialyl-Lewis x. Proc Natl Acad Sci U S A. 1991 Jul 15;88(14):6224–6228. doi: 10.1073/pnas.88.14.6224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reuter G., Schauer R., Szeiki C., Kamerling J. P., Vliegenthart J. F. A detailed study of the periodate oxidation of sialic acids in glycoproteins. Glycoconj J. 1989;6(1):35–44. doi: 10.1007/BF01047888. [DOI] [PubMed] [Google Scholar]
- Rosen S. D., Singer M. S., Yednock T. A., Stoolman L. M. Involvement of sialic acid on endothelial cells in organ-specific lymphocyte recirculation. Science. 1985 May 24;228(4702):1005–1007. doi: 10.1126/science.4001928. [DOI] [PubMed] [Google Scholar]
- Roy R., Katzenellenbogen E., Jennings H. J. Improved procedures for the conjugation of oligosaccharides to protein by reductive amination. Can J Biochem Cell Biol. 1984 May;62(5):270–275. doi: 10.1139/o84-037. [DOI] [PubMed] [Google Scholar]
- Springer T. A. The sensation and regulation of interactions with the extracellular environment: the cell biology of lymphocyte adhesion receptors. Annu Rev Cell Biol. 1990;6:359–402. doi: 10.1146/annurev.cb.06.110190.002043. [DOI] [PubMed] [Google Scholar]
- Stamper H. B., Jr, Woodruff J. J. An in vitro model of lymphocyte homing. I. Characterization of the interaction between thoracic duct lymphocytes and specialized high-endothelial venules of lymph nodes. J Immunol. 1977 Aug;119(2):772–780. [PubMed] [Google Scholar]
- Takada A., Ohmori K., Takahashi N., Tsuyuoka K., Yago A., Zenita K., Hasegawa A., Kannagi R. Adhesion of human cancer cells to vascular endothelium mediated by a carbohydrate antigen, sialyl Lewis A. Biochem Biophys Res Commun. 1991 Sep 16;179(2):713–719. doi: 10.1016/0006-291x(91)91875-d. [DOI] [PubMed] [Google Scholar]
- True D. D., Singer M. S., Lasky L. A., Rosen S. D. Requirement for sialic acid on the endothelial ligand of a lymphocyte homing receptor. J Cell Biol. 1990 Dec;111(6 Pt 1):2757–2764. doi: 10.1083/jcb.111.6.2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyrrell D., James P., Rao N., Foxall C., Abbas S., Dasgupta F., Nashed M., Hasegawa A., Kiso M., Asa D. Structural requirements for the carbohydrate ligand of E-selectin. Proc Natl Acad Sci U S A. 1991 Nov 15;88(22):10372–10376. doi: 10.1073/pnas.88.22.10372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Lenten L., Ashwell G. Studies on the chemical and enzymatic modification of glycoproteins. A general method for the tritiation of sialic acid-containing glycoproteins. J Biol Chem. 1971 Mar 25;246(6):1889–1894. [PubMed] [Google Scholar]
- Varki A. Selectins and other mammalian sialic acid-binding lectins. Curr Opin Cell Biol. 1992 Apr;4(2):257–266. doi: 10.1016/0955-0674(92)90041-a. [DOI] [PubMed] [Google Scholar]
- Watson S. R., Imai Y., Fennie C., Geoffroy J. S., Rosen S. D., Lasky L. A. A homing receptor-IgG chimera as a probe for adhesive ligands of lymph node high endothelial venules. J Cell Biol. 1990 Jun;110(6):2221–2229. doi: 10.1083/jcb.110.6.2221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yednock T. A., Rosen S. D. Lymphocyte homing. Adv Immunol. 1989;44:313–378. doi: 10.1016/s0065-2776(08)60645-8. [DOI] [PubMed] [Google Scholar]
- Zhou Q., Moore K. L., Smith D. F., Varki A., McEver R. P., Cummings R. D. The selectin GMP-140 binds to sialylated, fucosylated lactosaminoglycans on both myeloid and nonmyeloid cells. J Cell Biol. 1991 Oct;115(2):557–564. doi: 10.1083/jcb.115.2.557. [DOI] [PMC free article] [PubMed] [Google Scholar]





