Abstract
Introduction
Conditional survival can provide valuable predictive information for both patients and caregivers for patients surviving over time. The purpose of this study was to estimate conditional survival for esophageal cancer patients through analysis of a national population-based cancer registry.
Methods
This retrospective cohort study analyzed 64,433 patients within the Surveillance, Epidemiology, and End Results (SEER) dataset who were diagnosed with esophageal cancer from 1988-2011. Covariates included cancer characteristics and demographics. Overall survival (defined as time from diagnosis until death), cause specific survival (defined as time from diagnosis until death from cancer), and five-year conditional survivals (the probability of surviving an additional five years) were calculated. Significant prognostic variables of univariate and multivariable models of survival were identified.
Results
The multivariable models of overall and cause specific survival included gender, age group, race, relationship status, year of diagnosis, site, grade, histology, and stage group. While all patients showed an improvement in conditional survival over time, more dramatic improvements were seen in more advanced stage groups. At the five-year mark, conditional cause specific survival of distant stage (defined as having spread by direct extension or metastasis to distant organs, tissues, or lymph nodes) increased from 4% to 79%, while regional stage increased from 18% to 77% and localized stage increased from 38% to 85.
Conclusions
Conditional survival showed improving prognosis over time. Patients with advanced stage had the most dramatic improvement. Clinicians, caregivers, and patients with esophageal cancer can feel encouraged by the improving prognosis with each year survived. This information has practical implications regarding longitudinal follow up guidelines and survivorship planning.
Keywords: esophageal carcinoma, conditional survival, SEER
Introduction
Esophageal cancer is the eighth most common cancer and sixth leading cause of cancer death worldwide, with more than 400,000 diagnoses and 350,000 deaths annually1. In the United States, an estimated 18,170 new cases were diagnosed in 2014 and median age of diagnosis was 672. Incidence rates have been increasing, especially in adenocarcinoma histology and white race1. Though survival of esophageal cancer has improved over the decades1, five-year relative survival was still only 17% in 2014 in the United States. Prognosis depends heavily on stage and the individual's response to treatment, but prognosis also changes for each individual over time. Conditional survival demonstrates quantitatively and visually how an individual's prognosis changes over time.
The purpose of this study was to calculate the conditional survival of esophageal cancer patients in the United States. The hypothesis was that conditional survival would improve over time, especially in more advanced stages, as has been shown for other gastrointestinal tract tumors like the stomach3, colon4, and rectum5. The national population-based SEER registry is a publicly available database that provides both large cohort size and long-term follow-up, two necessary components for studying conditional survival. These attributes are especially useful for esophageal cancer whose epidemiologic study can be difficult due to the short survival6.
Materials and Methods
The data were obtained from the Surveillance, Epidemiology, and End Results (SEER) registry of the National Cancer Institute (Bethesda, MD). SEER is a national population-based cancer registry that is globally recognized for its accuracy and completeness. It currently collects cancer incidence and survival data for over one fourth of the United States population7. More information on the geographic regions included as SEER regions can be found at its website7. Since the data from the SEER registry are deidentified and publicly available, no IRB approval was necessary.
The newest available SEER registry (1973-2011) found 81,686 patients that were diagnosed with esophageal cancer as their first and only cancer diagnosis using Collaborative Stage Schema. The anatomic sites included both the esophagus and esophagus/gastroesophageal junction (GEJ). Inclusion required microscopic diagnostic confirmation, malignant tumor behavior, active follow-up, and diagnosis after the year 1988 in order to have sufficient information for analysis. Cases were excluded if reported by death certificate, autopsy, or hospice. Pediatric cases were excluded (age at diagnosis <18 years). These criteria resulted in 64,433 cases in the final retrospective cohort analysis.
Covariates included demographic variables (gender, age group, race, and marital status) and diagnostic information (year of diagnosis, site, grade, histology, and SEER historic stage group). SEER historic stage grouped esophageal cancers into localized, regional, distant, or unknown stages as follows: (1) localized – an invasive neoplasm confined entirely to the organ of origin; (2) regional – a neoplasm that has extended directly into surrounding organs or tissues, into regional lymph nodes, or both; (3) distant – a neoplasm that has spread to parts of the body remote from the primary tumor either by direct extension or by discontinuous metastasis (e.g. implantation or seeding) to distant organs, tissues, or via the lymphatic system to distant lymph nodes; or (4) unknown – information is not sufficient to assign a stage. In order to maximize sample size, all sites (esophagus and esophagus/GEJ), grades (I-IV, unknown), and histology (adenocarcinoma, squamous cell carcinoma, all others) were included. Esophagus site can be loosely correlated with multiple regions (cervical, upper thoracic, and midthoracic). The lower (distal) esophagus and GEJ site can be considered as the lower thoracic region. Patients were divided into two age groups around 65 years, a commonly used definition of elderly patients according to the World Health Organization8. Race categories were white, black, other (American Indian/AK Native, Asian/Pacific Islander, or other unspecified), and unknown. Relationship status categories were single, married/partner, separated/divorced, widowed, and unknown. The chi-square test was used to compare the distribution of these groups across the stage groups.
The Kaplan-Meier method was used for the initial, unadjusted univariate survival analysis. Overall survival was defined as time from diagnosis until death. Cause specific survival was defined as time from diagnosis until death due to esophageal cancer. The log-rank test was used to identify and compare the outcome of significant covariates in univariate survival analysis. These clinically meaningful and significant prognostic variables were considered for inclusion in the final Cox proportional hazards multivariable regression models. The final model was the same result of forward, backward, and stepwise selections.
Five-year conditional [overall and cause specific] survivals were calculated for each of the covariates in the final multivariable regression models. Conditional survival was calculated as the proportion surviving five additional years, as shown in the equation below:
Analysis was conducted using SAS v9.3 and Microsoft Excel 2010. Conditional overall survival was also calculated for the subgroup of patients diagnosed during 2004-2011 with an American Joint Committee on Cancer (AJCC) 6th edition stage, to compare trends using SEER historic stage vs AJCC stage. Effort was made to present only conditional survival graphs with sufficient follow-up by displaying conditional survivals of only those values calculated from Kaplan-Meier survival estimates with at least 100 patients at risk; exceptions were required for unknown marital status (<100 cases at risk at 9 years after diagnosis, which was necessary for conditional survivals at ≥4 years) and for sub-analysis of cases diagnosed after 2004 (due to fewer cases, shorter follow-up, and poor survival).
Results
Stage distribution was statistically different across all demographic and diagnostic covariates, as shown in Table 1; all P-values from chi-square test were <0.0001. For all stages, there were more patients with male gender, white race, married, diagnosed in 2000-2011, non-GE junction site, grade II-III, and adenocarcinoma histology. Relatively lower stage was associated with female gender, older age group, white or unknown race, widowed or unknown relationship status, non-GE junction site, lower grade, and squamous cell histology. Diagnosis during 2000-2011 was associated with fewer unknown stage and more distant stage group diagnoses.
Table 1.
Demographic and cancer diagnosis distribution of studied cohort by stage group; all P-values <0.0001 by χ2 test. GEJ: gastroesophageal junction.
| Localized | Regional | Distant | Unknown | Total | P-value | ||
|---|---|---|---|---|---|---|---|
| Gender | Female | 3,529 | 4,050 | 4,490 | 2,516 | 14,585 | <0.0001 |
| % | 24 | 28 | 31 | 17 | |||
| Male | 10,592 | 15,345 | 18,524 | 5,387 | 49,848 | ||
| % | 21 | 31 | 37 | 11 | |||
| Age group | Age <65 | 5,170 | 9,241 | 11,948 | 2,271 | 28,630 | <0.0001 |
| % | 18 | 32 | 42 | 8 | |||
| Age 65+ | 8,951 | 10,154 | 11,066 | 5,632 | 35,803 | ||
| % | 25 | 28 | 31 | 16 | |||
| Race | White | 11,832 | 16,158 | 19,066 | 6,318 | 53,374 | <0.0001 |
| % | 22 | 30 | 36 | 12 | |||
| Black | 1,467 | 2,000 | 2,526 | 1,053 | 7,046 | ||
| % | 21 | 28 | 36 | 15 | |||
| Other/unknown | 822 | 1,237 | 1,422 | 532 | 4,013 | ||
| % | 20 | 32 | 36 | 12 | |||
| Marital status | Single | 1,768 | 2,629 | 3,507 | 1,173 | 9,077 | <0.0001 |
| 19 | 29 | 39 | 13 | ||||
| Married/partner | 8,152 | 11,782 | 13,570 | 3,476 | 36,980 | ||
| 22 | 32 | 37 | 9 | ||||
| Separated/divorced | 1,311 | 2,117 | 2,673 | 729 | 6,830 | ||
| 19 | 31 | 39 | 11 | ||||
| Widowed | 2,266 | 2,269 | 2,501 | 1,847 | 8,883 | ||
| 26 | 26 | 28 | 21 | ||||
| Unknown | 624 | 598 | 763 | 678 | 2,663 | ||
| 23 | 22 | 29 | 25 | ||||
| Year of diagnosis | Dx 1988-1999 | 3,926 | 5,262 | 5,358 | 3,086 | 17,632 | <0.0001 |
| 22 | 30 | 30 | 18 | ||||
| Dx 2000-2011 | 10,195 | 14,133 | 17,656 | 4,817 | 46,801 | ||
| 22 | 30 | 38 | 10 | ||||
| Site | Esophagus | 9,597 | 13,050 | 14,896 | 5,876 | 43,419 | <0.0001 |
| 22 | 30 | 34 | 14 | ||||
| Esophagus/GEJ | 4,524 | 6,345 | 8,118 | 2,027 | 21,014 | ||
| 22 | 30 | 39 | 10 | ||||
| Grade | Grade I | 1,145 | 715 | 618 | 396 | 2,874 | <0.0001 |
| 40 | 25 | 22 | 14 | ||||
| Grade II | 5,146 | 6,339 | 6,268 | 2,401 | 20,154 | ||
| 26 | 31 | 31 | 12 | ||||
| Grade III/IV | 5,262 | 10,052 | 12,224 | 3,188 | 30,726 | ||
| 17 | 33 | 40 | 10 | ||||
| Unknown | 2,568 | 2,289 | 3,904 | 1,918 | 10,679 | ||
| 18 | 32 | 40 | 10 | ||||
| Histology | Adenocarcinoma | 8,622 | 11,339 | 14,517 | 3,928 | 38,406 | <0.0001 |
| 22 | 30 | 38 | 10 | ||||
| Squamous cell | 4,030 | 5,491 | 5,193 | 2,769 | 17,483 | ||
| 23 | 31 | 30 | 16 | ||||
| All others | 1,469 | 2,565 | 3,304 | 1,206 | 8,544 | ||
| 17 | 30 | 39 | 14 |
Analysis of univariate overall and cause specific survivals showed that better survival was associated with more distal site, lower stage, lower grade, adenocarcinoma histology, more recent diagnosis, younger age at diagnosis, and being married or non-widowed; all P-values from log-rank test for significance <0.0001. Five-year cause specific vs overall survival of localized stage was 38% vs 29%; regional stage was 18% vs 15%; and distant stage was 4% vs 3%. Ten-year cause specific vs overall survival of localized stage was 32% vs 19%; regional stage was 14% vs 9%; and distant stage was 3% vs 2%.
All of the covariates were also found to be significant in multivariable analysis, as shown in Table 2. Better prognosis was associated with younger age at diagnosis, non-black race, married or non-widowed relationship status, diagnosed in 2000-2011, more distal site, lower grade, adenocarcinoma histology, and lower stage after adjusting for other covariates. The single greatest hazard in the multivariable model was distant stage, with triple the risk of death compared to localized stage, and 3.5 times the risk of death from cancer compared to localized stage.
Table 2. Final Cox proportional hazards model multivariable regression with effect estimates for overall survival (defined as time from diagnosis until death from all causes) and cause specific survival (defined as time from diagnosis until death due to cancer).
| Overall survival | Cause specific survival | |||||
|---|---|---|---|---|---|---|
| Hazard ratio | 95% confidence interval of hazard ratio | P-value | Hazard ratio | 95% confidence interval of hazard ratio | P-value | |
| Gender | ||||||
| Female | 1 (baseline) | 1 (baseline) | ||||
| Male | 1.1 | 1.09-1.14 | <0.0001 | 1.1 | 1.07-1.12 | <0.0001 |
| Age group | ||||||
| Age <65 | 1 (baseline) | 1 (baseline) | ||||
| Age 65+ | 1.4 | 1.40-1.45 | <0.0001 | 1.3 | 1.31-1.36 | <0.0001 |
| Race | ||||||
| White | 1 (baseline) | 1 (baseline) | ||||
| Black | 1.2 | 1.12-1.19 | <0.0001 | 1.1 | 1.10-1.17 | <0.0001 |
| Other | 0.9 | 0.84-0.91 | <0.001 | 0.9 | 0.84-0.91 | <0.0001 |
| Unknown | 0.6 | 0.53-0.73 | <0.0001 | 0.6 | 0.50-0.72 | <0.0001 |
| Relationship status | ||||||
| Single | 1 (baseline) | 1 (baseline) | ||||
| Married/Partner | 0.8 | 0.79-0.83 | <0.0001 | 0.8 | 0.80-0.84 | <0.0001 |
| Separated/Divorced | 1.0 | 0.94-1.01 | 0.17 | 1.0 | 0.94-1.01 | 0.1536 |
| Widowed | 1.1 | 1.06-1.13 | <0.0001 | 1.1 | 1.06-1.14 | <0.0001 |
| Unknown | 0.9 | 0.87-0.96 | 0.0001 | 0.9 | 0.87-0.97 | 0.001 |
| Year of diagnosis | ||||||
| 1988-1999 | 1 (baseline) | 1 (baseline) | ||||
| 2000-2011 | 0.8 | 0.80-0.83 | <0.0001 | 0.8 | 0.79-0.83 | <0.0001 |
| Site | ||||||
| Esophagus | 1 (baseline) | 1 (baseline) | ||||
| Esophagus/GEJ | 1.0 | 0.94-0.98 | 0.0003 | 1.0 | 0.94-0.98 | <0.0001 |
| Grade | ||||||
| I | 1 (baseline) | 1 (baseline) | ||||
| II | 1.2 | 1.14-1.25 | <0.0001 | 1.2 | 1.18-1.30 | <0.0001 |
| III/IV | 1.4 | 1.33-1.46 | <0.0001 | 1.5 | 1.41-1.56 | <0.0001 |
| Unknown | 1.2 | 1.17-1.28 | <0.0001 | 1.3 | 1.21-1.34 | <0.0001 |
| Histology | ||||||
| Adenocarcinoma | 1 (baseline) | 1 (baseline) | ||||
| Squamous cell | 1.1 | 1.12-1.17 | <0.0001 | 1.1 | 1.12-1.17 | <0.0001 |
| All others | 1.1 | 1.11-1.17 | <0.0001 | 1.2 | 1.12-1.19 | <0.0001 |
| Stage | ||||||
| Localized | 1 (baseline) | 1 (baseline) | ||||
| Regional | 1.4 | 1.38-1.45 | <0.0001 | 1.6 | 1.54-1.63 | <0.0001 |
| Distant | 3.0 | 2.91-3.06 | <0.0001 | 3.5 | 3.44-3.63 | <0.0001 |
| Unknown | 2.0 | 1.90-2.02 | <0.0001 | 2.1 | 2.05-2.20 | <0.0001 |
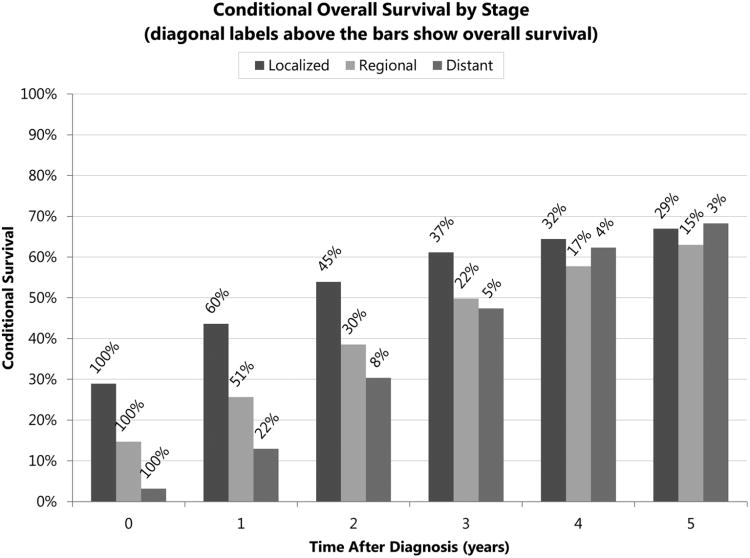
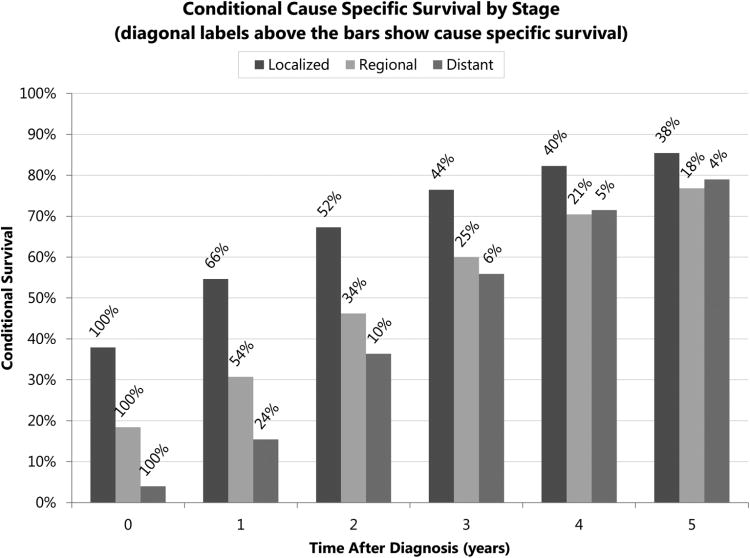
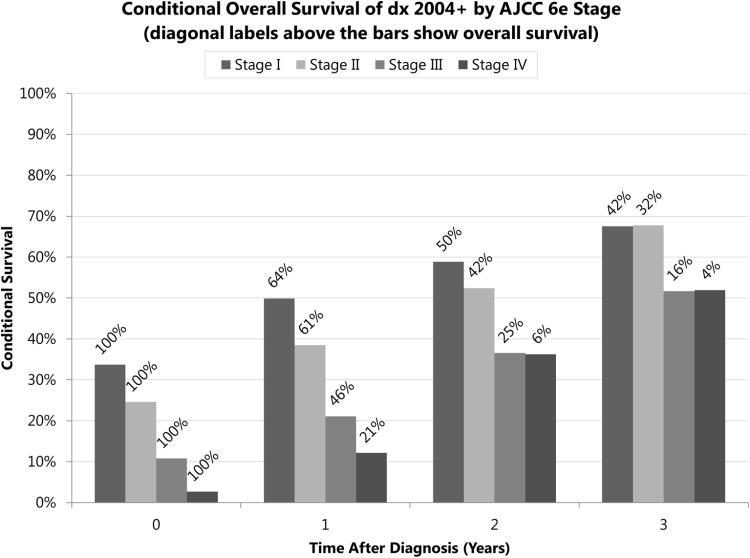
Conditional overall and cause specific survivals of all patients improved dramatically over time, as shown in the following Figures. Over the first five years after diagnosis, conditional overall survival improved from 29% to 67% in the localized stage group, 15% to 63% in the regional stage group, and 3% to 68% in the distant stage group (Figure 1a). Over the first five years after diagnosis, conditional cause specific survival improved from 38% to 85% in the localized stage group, 18% to 77% in the regional stage group, and 4% to 79% in the distant stage group (Figure 1b). Subanalysis of the 3,338 cases with AJCC 6th edition staging information showed the same trend, with conditional overall survival of stage IV increasing from 3% to 52% after 3 years, while stages III/II/I increased from 11%/25%/34% to 52%/68%/68%, respectively (Figure 3), although there were fewer cases in this analysis. More dramatic increases were seen in more advanced stages.
Figure 1a.
Five-year conditional overall survival of esophageal cancer patients by SEER historic stage group, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Figure 1b.
Five-year conditional cause specific survival of esophageal cancer patients by SEER historic stage group, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Figure 3.
Five-year conditional cause specific survival of esophageal cancer patients by American Joint Committee on Cancer (AJCC) 6th edition stage, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
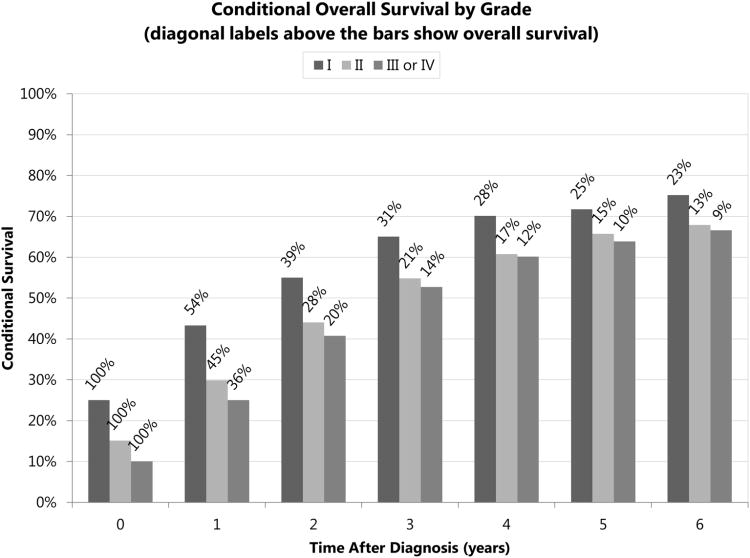
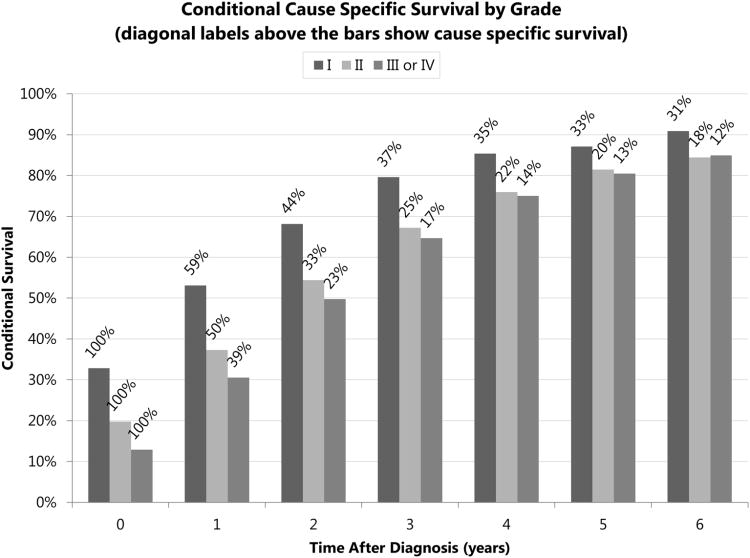
A similar trend was seen in conditional overall and cause specific survival of the higher grade group as well (Figure 2a, Figure 2b). When grouped by site (Supplemental Digital Content 4a, Supplemental Digital Content 4b), histology (Supplemental Digital Content 5a, Supplemental Digital Content 5b), year of diagnosis (Supplemental Digital Content 6a, Supplemental Digital Content 6b), gender (Supplemental Digital Content 7a, Supplemental Digital Content 7b), age at diagnosis (Supplemental Digital Content 8a, Supplemental Digital Content 8b), race (Supplemental Digital Content 9a, Supplemental Digital Content 9b), or relationship status (Supplemental Digital Content 10a, Supplemental Digital Content 10b), the conditional survivals increased more uniformly so that the relative relationship between subgroups was maintained over time.
Figure 2a.
Five-year conditional overall survival of esophageal cancer patients by grade, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Figure 2b.
Five-year conditional cause specific survival of esophageal cancer patients by grade, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Discussion
This is the first analysis to describe overall conditional and cause specific survival for esophageal cancer. Two studies of perioperative mortality published what they called conditional survival: one following esophagectomy or gastrectomy for esophagogastric cancer9, and another following surgery for non-metastatic colon, esophageal, gastric, liver, lung, pancreatic, or rectal cancers10. What they actually calculated was survival excluding perioperative mortality (survival among patients who did not die within 30 or 60 days of surgery). These are different from the more standard five-year conditional survival calculated here.
A meeting abstract by German investigators calculated conditional survival using 25,306 SEER cases diagnosed between 1988 and 200411. It is unclear from the abstract exactly what selection criteria or analytical methods they used. Their “multivariate [sic]” analysis does not seem to have included stage, site, grade, histology, year of diagnosis, or age group. Dubecz et al showed the same dramatic increase in conditional overall survival in more advanced stages, with slightly lower conditional survival values (58%, 56%, and 61% for localized, regional, and distant SEER historic stages after five years, compared to the current, larger analysis, 67%, 63%, and 68%, respectively). Other gastrointestinal tract3,4,5 and solid tumors12,13,14,15 show a similar pattern, with the five-year conditional survival of higher stage disease increasing more rapidly and starting to approach the conditional survivals of lower stages over time. As shown in Figure 2b, though conditional cause specific survival of distant stage increases from 4% to 15% after just 1 year, this is only for the 24% who survive for one year. Likewise, though conditional cause specific survival of distant stage increases to almost 20-fold over 5 years (from 4% to 79%), this is only for the 4% who are still alive at the five year mark following the original diagnosis.
Our higher survivals are reasonable given that Dubecz et al used an older release of the same SEER registry. Patients diagnosed in 2000-2011 had better outcomes than those from 1988-1999 in both univariate and multivariable survival analysis. This is consistent with established national trends1, and is likely a result of a combination of factors including advances in treatment methods, better use of combined modalities, and changes in dominant histology type. Incidence of adenocarcinoma has been increasing while that of squamous cell carcinoma has been decreasing1; adenocarcinoma had better overall and cause specific survivals than squamous cell carcinoma in both univariate and multivariable survival analysis.
There are many possible reasons for the greater increase in conditional survival for more advanced stages. Stage is thought to be the single most important prognostic factor1. In our multivariable analysis as well, the largest predictors of hazard were advanced stages (Table 2). Unlike localized or regional stages, distant stage includes a much wider spectrum of disease; distant stage could mean one distant lymph node, or it could mean widely metastatic cancer. In a sub-analysis of cases diagnosed after 2004, when SEER started including AJCC 6th edition stage, localized stage group were mostly stage I (ranging stages I-II), regional stage group were mostly stages II and III (ranging stages I-IV), and distant stage group were mostly stage IV (ranging stage I-IV). This would exacerbate the range in therapies received by patients with distant stage, due to different treatment options, aggressiveness of and patients' tolerance of these treatments, and varying goals of treatment. Initial mortality was high from more advanced stages, with one-year cause specific survival of only 24%, but the patients who did survive one or two years may have been those with relatively less severe disease and thus better survival.
SEER can provide data to quantify the potential impact of different modalities and their sequence of administration including preoperative therapy16. The generation of outcome prediction tools using this data may serve as decision aids for patients and practitioners16,17. One limitation of using SEER data is the lack of detailed treatment information. Treatment information is limited to surgery or radiation therapy received within 4 months of diagnosis; it does not have treatments started later, or details on the specific agents that comprised systemic therapies. Dubecz et al included surgery as a covariate, but we did not feel that there were adequate details regarding surgery or other treatment information to include it in the analysis without potentially compromising the quality of the results due to incomplete treatment information.
The grade may not be as reliable as some of the other tumor characteristics since central pathology review was not and cannot be performed for all of the cases in the SEER database. Another limitation is the inconsistency of staging information and potential understaging, particularly in older cases. SEER's historic stage is certainly useful, but AJCC staging information provides much finer classifications and is the basis of modern clinical decision-making and prognostic assessment. For esophageal cancer in the SEER registry, AJCC stage information is only available as 6th edition for cases diagnosed during or after 2004. The quality of diagnostic procedures may have led to understaging of patients, which could artificially inflate the survival of more advanced stages.
Subanalysis of conditional overall survival using only this subgroup diagnosed during 2004 to 2011 found the same conclusion as using SEER historic stage. Due to recent data with limited follow-up, conditional survival could only be calculated up to three years after diagnosis for this subanalysis, and stages III and IV in particular had very small sample sizes (111 and 53 at risk after 3 years) due to the poor survival of advanced stages, resulting in potentially less representative results. Follow-up time is particularly important for conditional survival since the conditional survival calculated at the two year mark, for instance, requires follow-up information until at least seven years after diagnosis. The SEER registry is a useful resource because it has long-term follow-up information for many tumor types, including esophageal cancer.
Conclusions
This is the first known manuscript of conditional survival for esophageal cancer. Conditional overall and cause specific survivals here provide visual and quantitative evidence of the changing prognosis of a patient over time. Patients with more advanced stages in particular can feel encouraged by their improving prognosis with every year survived. Conditional survival is a valuable resource in cancer survivorship and should continue to be investigated with other databases with more treatment and staging information and longer follow-up. In summary, conditional survival can be a useful tool to predict survival for esophageal patients, their family and friends, and healthcare professionals.
Supplementary Material
Supplemental Digital Content 4a. Five-year conditional overall survival of esophageal cancer patients by site, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis; GEJ: gastroesophageal junction
Supplemental Digital Content 4b. Five-year conditional cause specific survival of esophageal cancer patients by site, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis; GEJ: gastroesophageal junction
Supplemental Digital Content 5a. Five-year conditional overall survival of esophageal cancer patients by histology, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 5b. Five-year conditional cause specific survival of esophageal cancer patients by histology, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 6a. Five-year conditional overall survival of esophageal cancer patients by year of diagnosis, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 6b. Five-year conditional cause specific survival of esophageal cancer patients by year of diagnosis, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 7a. Five-year conditional overall survival of esophageal cancer patients by gender, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 7b. Five-year conditional cause specific survival of esophageal cancer patients by gender, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 8a. Five-year conditional overall survival of esophageal cancer patients by age at diagnosis, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 8b. Five-year conditional cause specific survival of esophageal cancer patients by age at diagnosis, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 9a. Five-year conditional overall survival of esophageal cancer patients by race, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 9b. Five-year conditional cause specific survival of esophageal cancer patients by race, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 10a. Five-year conditional overall survival of esophageal cancer patients by relationship status, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 10b. Five-year conditional cause specific survival of esophageal cancer patients by relationship status, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Acknowledgments
This paper uses data obtained from the Research Data File of the SEER Program, provided to persons interested in research by the SEER Program. Dr. Koroukian was supported in part by the Clinical and Translational Science Collaborative of Cleveland, UL1TR000439 from the National Center for Advancing Translational Sciences (NCATS) component of the National Institutes of Health and National Institutes of Health roadmap for Medical Research.The contents are solely the responsibility of the authors and do not necessarily represent the official views of the National Institutes of Health or National Cancer Institute.
Guarantor Statement and Contributions: CRT, SK, and EK had full access to all of the data in the study; contributed substantially to the study design, data analysis and interpretation, and writing of the manuscript; and take responsibility for the integrity of the work as a whole, from inception to published article.
Contributor Information
Charles R Thomas, Jr., Email: thomasch@ohsu.edu, Oregon Health & Science University, 3181 SW Sam Jackson Park Road, Mail Code L-337, Portland, Oregon 97239-3098, United States, 503-494-8758, work: 503-494-8756, Fax: 503-494-6967.
Ellen Kim, Case Western Reserve University School of Medicine.
Siran Koroukian, Case Western Reserve University School of Medicine, Department of Epidemiology and Biostatistics.
References
- 1.Brown LM, Devesa SS. Epidemiology and risk of esophageal cancer: Clinical. In: Jobe BA, Thomas CR Jr, Hunter JG, editors. Esophageal cancer: Principles and practice. New York: Demos Medical Publishing; 2009. pp. 103–14. [Google Scholar]
- 2.DeSantis CE, Lin CC, Mariotto AB, et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin. 2014;64(4):252–71. doi: 10.3322/caac.21235. [DOI] [PubMed] [Google Scholar]
- 3.Wang SJ, Emery R, Fuller CD, Kim JS, Sittig DF, Thomas CR., Jr Conditional survival in gastric cancer: a SEER database analysis. Gastric Cancer. 2007;10:153–8. doi: 10.1007/s10120-007-0424-9. [DOI] [PubMed] [Google Scholar]
- 4.Zamboni BA, Yothers G, Choi M, et al. Conditional survival and the choice of conditioning set for patients with colon cancer: an analysis of NSABP trials C-03 through C-07. J Clin Oncol. 2010;28:2544–8. doi: 10.1200/JCO.2009.23.0573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang SJ, Fuller CD, Emery R, Thomas CR., Jr Conditional survival in rectal cancer: a SEER database analysis. Gastrointest Cancer Res. 2007;1:84–9. [PMC free article] [PubMed] [Google Scholar]
- 6.Pickens A. Ethnic disparities in cancer of the esophagus. In: Jobe BA, Thomas CR Jr, Hunter JG, editors. Esophageal cancer: Principles and practice. New York: Demos Medical Publishing; 2009. pp. 137–43. [Google Scholar]
- 7.National Cancer Institute. Surveillance, Epidemiology, and End Results. Overview of the SEER Program. [Accessed online January 5, 2015]; http://seer.cancer.gov/about.
- 8.World Health Organization. Definition of an older or elderly person. [Accessed online January 5, 2015]; www.who.int/healthinfo/survey/ageingdefnolder/en/
- 9.Smith RC, Creighton N, Lord RV, Merrett ND, Keogh GW, et al. Survival, mortality and morbidity outcomes after oesophagogastric cancer surgery in New South Wales, 2001-2008. Med J Aust. 2014;200(7):408–13. doi: 10.5694/mja13.11182. [DOI] [PubMed] [Google Scholar]
- 10.Bilimoria KY, Bentrem DJ, Feinglass JM, Stewart AK, Winchester DP, et al. Directing surgical quality improvement initiatives: Comparison of perioperative mortality and long-term survival for cancer surgery. J Clin Oncol. 2008;26(28):4626–33. doi: 10.1200/JCO.2007.15.6356. [DOI] [PubMed] [Google Scholar]
- 11.Dubecz A, Stadlhuber RJ, Stein HJ. Long term conditional survival in esophageal cancer: a SEER database analysis [abstract] [Accessed online December 31, 2014];Society for Surgery of the Alimentary Tract 51st Annual Meeting. http://meetings.ssat.com/abstracts/10ddw/P26.cgi.
- 12.Choi M, Fuller CD, Thomas CR, Wang SJ. Conditional survival in ovarian cancer: results from the SEER dataset 1988-2001. Gyn Oncol. 2008;109:203–9. doi: 10.1016/j.ygyno.2008.01.033. [DOI] [PubMed] [Google Scholar]
- 13.Fuller CD, Wang SJ, Thomas CR, Jr, Hoffman HT, Weber RS, Rosenthal DI. Conditional survival in head and neck squamous cell carcinoma: results from the SEER dataset 1973-1998. Cancer. 2007;109:1331–43. doi: 10.1002/cncr.22563. [DOI] [PubMed] [Google Scholar]
- 14.Wang SJ, Fuller CD, Thomas CR. Ethnic disparities in conditional survival of patients with non-small cell lung cancer. J Thorac Oncol. 2007;2:180–90. doi: 10.1097/JTO.0b013e318031cd4e. [DOI] [PubMed] [Google Scholar]
- 15.Kurta ML, Edwards RP, Moysich KB, et al. Prognosis and conditional disease-free survival among patients with ovarian cancer. J Clin Oncol. 2014;32(36):4102–13. doi: 10.1200/JCO.2014.55.1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Eil R, Diggs BS, Wang SJ, et al. Nomogram for predicting the benefit of neoadjuvant chemoradiotherapy for patients with esophageal cancer: a SEER-Medicare analysis. Cancer. 2013;120(4):492–8. doi: 10.1002/cncr.28447. [DOI] [PubMed] [Google Scholar]
- 17.Eil R, Voncken F, Torres-Roca J, Thomas CR., Jr . Esophageal cancer. In: Gaspar LE, Nieder C, editors. Decision Tools for Radiation Oncology: Prognosis, Treatment response and Toxicity. Heidelberg, Germany: Springer; 2014. pp. 107–25. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Digital Content 4a. Five-year conditional overall survival of esophageal cancer patients by site, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis; GEJ: gastroesophageal junction
Supplemental Digital Content 4b. Five-year conditional cause specific survival of esophageal cancer patients by site, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis; GEJ: gastroesophageal junction
Supplemental Digital Content 5a. Five-year conditional overall survival of esophageal cancer patients by histology, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 5b. Five-year conditional cause specific survival of esophageal cancer patients by histology, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 6a. Five-year conditional overall survival of esophageal cancer patients by year of diagnosis, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 6b. Five-year conditional cause specific survival of esophageal cancer patients by year of diagnosis, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 7a. Five-year conditional overall survival of esophageal cancer patients by gender, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 7b. Five-year conditional cause specific survival of esophageal cancer patients by gender, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 8a. Five-year conditional overall survival of esophageal cancer patients by age at diagnosis, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 8b. Five-year conditional cause specific survival of esophageal cancer patients by age at diagnosis, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 9a. Five-year conditional overall survival of esophageal cancer patients by race, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 9b. Five-year conditional cause specific survival of esophageal cancer patients by race, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis
Supplemental Digital Content 10a. Five-year conditional overall survival of esophageal cancer patients by relationship status, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing overall survival from diagnosis
Supplemental Digital Content 10b. Five-year conditional cause specific survival of esophageal cancer patients by relationship status, calculated from Kaplan-Meier survival estimates, with diagonal labels above the bars showing cause specific survival from diagnosis