Abstract
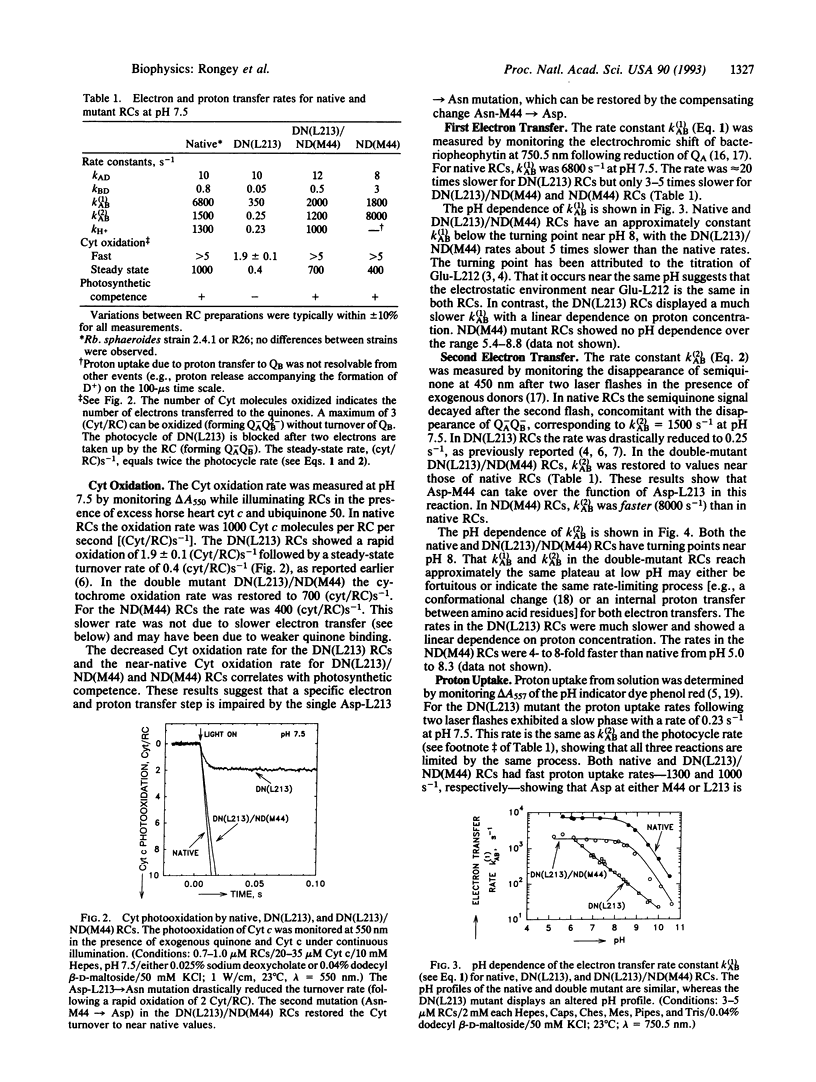
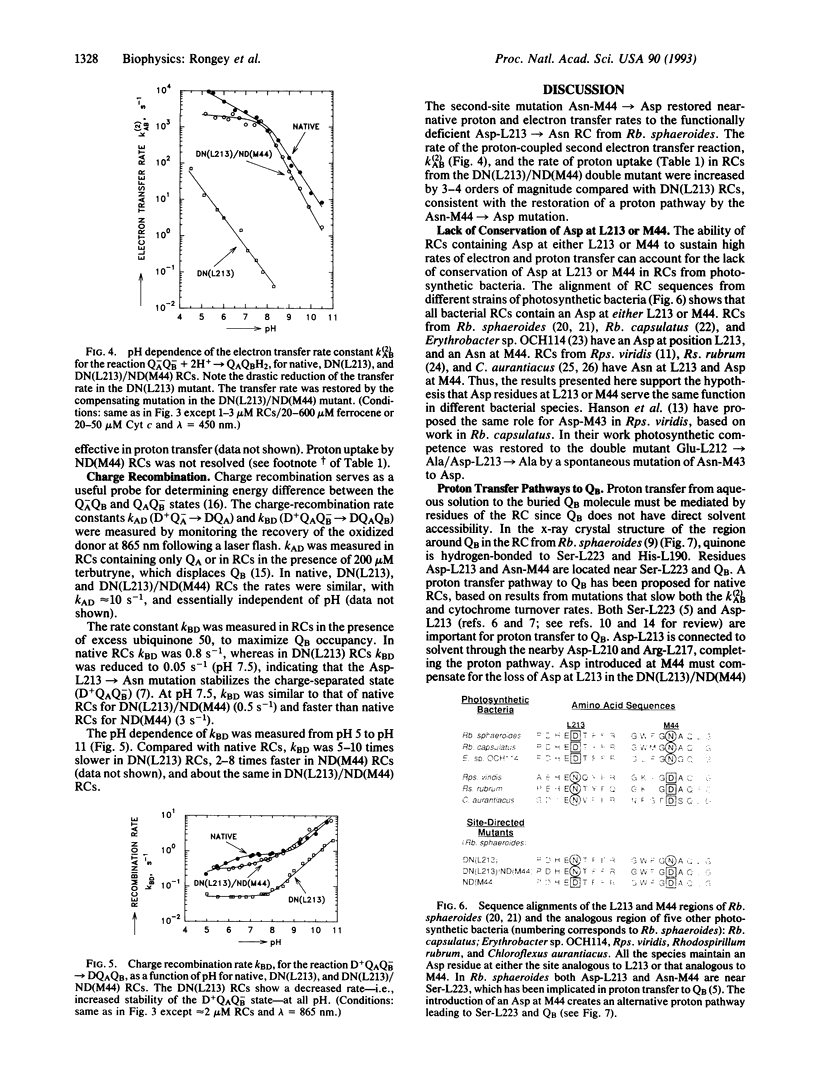
Site-directed mutagenesis of the photosynthetic reaction center (RC) from Rhodobacter sphaeroides has shown Asp-213 of the L subunit (Asp-L213) to be important for photosynthetic viability. Replacement of Asp-L213 with Asn resulted in a photosynthetically deficient mutant, due to the 10(4)-fold slower rate for the proton-coupled electron transfer reaction QA-QB- + 2H+-->QAQBH2 (k(2)AB). The detrimental effect of Asn-L213 is surprising since RCs from Rhodopseudomonas viridis, Rhodospirillum rubrum, and Chloroflexus aurantiacus have Asn at the homologous position. However, RCs from these bacteria have an Asp located near QB (the secondary quinone acceptor) at the position homologous to Asn-M44 in Rb. sphaeroides which might function in place of Asp-L213. To test this conjecture a "viridis-like" structure was introduced into Rb. sphaeroides by replacing Asp-L213 with Asn and Asn-M44 with Asp. The RCs from this double mutant displayed near-native rates for the electron transfer reaction k(2)AB and restored photosynthetic competence. The rates for the first electron transfer reaction QA-QB-->QAQB- (k(1)AB) and charge recombination D+QAQB--->DQAQB (kBD) were also restored to near-native values. These results indicate that Asp at either the L213 or the M44 site near QB can provide a pathway for rapid proton transfer and explain why Asp-L213 need not be conserved in different photosynthetic bacteria. To test further the effect of Asp at M44 on electron and proton transfer to QB a mutant containing Asp at both L213 and M44 was constructed. The RCs from this mutant (Asn-M44-->Asp) exhibited faster proton-coupled electron transfer to QB-. The increased rate of proton-coupled electron transfer (k(2)AB) in the presence of negatively charged Asp residues near QB suggests the role of an Asp near QB as (i) a proton donor group in the proton transfer chain and/or (ii) a negatively charged residue stabilizing proton transfer to reduced QB.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allen J. P., Feher G., Yeates T. O., Komiya H., Rees D. C. Structure of the reaction center from Rhodobacter sphaeroides R-26: protein-cofactor (quinones and Fe2+) interactions. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8487–8491. doi: 10.1073/pnas.85.22.8487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bélanger G., Bérard J., Corriveau P., Gingras G. The structural genes coding for the L and M subunits of Rhodospirillum rubrum photoreaction center. J Biol Chem. 1988 Jun 5;263(16):7632–7638. [PubMed] [Google Scholar]
- Deisenhofer J., Michel H. Nobel lecture. The photosynthetic reaction centre from the purple bacterium Rhodopseudomonas viridis. EMBO J. 1989 Aug;8(8):2149–2170. doi: 10.1002/j.1460-2075.1989.tb08338.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinfeld D., Okamura M. Y., Feher G. Electron transfer in reaction centers of Rhodopseudomonas sphaeroides. I. Determination of the charge recombination pathway of D+QAQ(-)B and free energy and kinetic relations between Q(-)AQB and QAQ(-)B. Biochim Biophys Acta. 1984 Jul 27;766(1):126–140. doi: 10.1016/0005-2728(84)90224-x. [DOI] [PubMed] [Google Scholar]
- Liebetanz R., Hornberger U., Drews G. Organization of the genes coding for the reaction-centre L and M subunits and B870 antenna polypeptides alpha and beta from the aerobic photosynthetic bacterium Erythrobacter species OCH114. Mol Microbiol. 1991 Jun;5(6):1459–1468. doi: 10.1111/j.1365-2958.1991.tb00792.x. [DOI] [PubMed] [Google Scholar]
- Michel H., Weyer K. A., Gruenberg H., Dunger I., Oesterhelt D., Lottspeich F. The 'light' and 'medium' subunits of the photosynthetic reaction centre from Rhodopseudomonas viridis: isolation of the genes, nucleotide and amino acid sequence. EMBO J. 1986 Jun;5(6):1149–1158. doi: 10.1002/j.1460-2075.1986.tb04340.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ovchinnikov YuA, Abdulaev N. G., Shmuckler B. E., Zargarov A. A., Kutuzov M. A., Telezhinskaya I. N., Levina N. B., Zolotarev A. S. Photosynthetic reaction centre of Chloroflexus aurantiacus. Primary structure of M-subunit. FEBS Lett. 1988 May 23;232(2):364–368. doi: 10.1016/0014-5793(88)80770-1. [DOI] [PubMed] [Google Scholar]
- Ovchinnikov YuA, Abdulaev N. G., Zolotarev A. S., Shmukler B. E., Zargarov A. A., Kutuzov M. A., Telezhinskaya I. N., Levina N. B. Photosynthetic reaction centre of Chloroflexus aurantiacus. I. Primary structure of L-subunit. FEBS Lett. 1988 Apr 11;231(1):237–242. doi: 10.1016/0014-5793(88)80739-7. [DOI] [PubMed] [Google Scholar]
- Paddock M. L., McPherson P. H., Feher G., Okamura M. Y. Pathway of proton transfer in bacterial reaction centers: replacement of serine-L223 by alanine inhibits electron and proton transfers associated with reduction of quinone to dihydroquinone. Proc Natl Acad Sci U S A. 1990 Sep;87(17):6803–6807. doi: 10.1073/pnas.87.17.6803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paddock M. L., Rongey S. H., Feher G., Okamura M. Y. Pathway of proton transfer in bacterial reaction centers: replacement of glutamic acid 212 in the L subunit by glutamine inhibits quinone (secondary acceptor) turnover. Proc Natl Acad Sci U S A. 1989 Sep;86(17):6602–6606. doi: 10.1073/pnas.86.17.6602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi E., Wraight C. A. Proton and electron transfer in the acceptor quinone complex of Rhodobacter sphaeroides reaction centers: characterization of site-directed mutants of the two ionizable residues, GluL212 and AspL213, in the QB binding site. Biochemistry. 1992 Jan 28;31(3):855–866. doi: 10.1021/bi00118a031. [DOI] [PubMed] [Google Scholar]
- Takahashi E., Wraight C. A. Small weak acids stimulate proton transfer events in site-directed mutants of the two ionizable residues, GluL212 and AspL213, in the QB-binding site of Rhodobacter sphaeroides reaction center. FEBS Lett. 1991 May 20;283(1):140–144. doi: 10.1016/0014-5793(91)80572-k. [DOI] [PubMed] [Google Scholar]
- Vermeglio A., Clayton R. K. Kinetics of electron transfer between the primary and the secondary electron acceptor in reaction centers from Rhodopseudomonas sphaeroides. Biochim Biophys Acta. 1977 Jul 7;461(1):159–165. doi: 10.1016/0005-2728(77)90078-0. [DOI] [PubMed] [Google Scholar]
- Williams J. C., Steiner L. A., Feher G., Simon M. I. Primary structure of the L subunit of the reaction center from Rhodopseudomonas sphaeroides. Proc Natl Acad Sci U S A. 1984 Dec;81(23):7303–7307. doi: 10.1073/pnas.81.23.7303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams J. C., Steiner L. A., Ogden R. C., Simon M. I., Feher G. Primary structure of the M subunit of the reaction center from Rhodopseudomonas sphaeroides. Proc Natl Acad Sci U S A. 1983 Nov;80(21):6505–6509. doi: 10.1073/pnas.80.21.6505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wraight C. A. Electron acceptors of bacterial photosynthetic reaction centers. II. H+ binding coupled to secondary electron transfer in the quinone acceptor complex. Biochim Biophys Acta. 1979 Nov 8;548(2):309–327. doi: 10.1016/0005-2728(79)90138-5. [DOI] [PubMed] [Google Scholar]
- Youvan D. C., Bylina E. J., Alberti M., Begusch H., Hearst J. E. Nucleotide and deduced polypeptide sequences of the photosynthetic reaction-center, B870 antenna, and flanking polypeptides from R. capsulata. Cell. 1984 Jul;37(3):949–957. doi: 10.1016/0092-8674(84)90429-x. [DOI] [PubMed] [Google Scholar]





