Abstract
Pancreatic B-cell lymphoma is rare; it accounts for 0.2% to 2.0% of extranodal non-Hodgkin lymphoma, and constitutes less than 0.5% of all pancreatic malignancies. Most histologic types of the pancreatic lymphoma are diffuse large B-cell lymphoma, and follicular lymphoma is quite rare. We report here a case of pancreatic follicular lymphoma that was initially detected by acute pancreatitis. This is the first reported case of pancreatic follicular lymphoma presenting with acute pancreatitis. A 71-year-old woman had epigastric and left upper quadrant abdominal pain. Computed tomography (CT) revealed features of acute pancreatitis. After standard therapy for pancreatitis, enhanced CT showed a pancreatic tumor (50 × 35 mm) in the body of the pancreas with gradual enhancement. Endoscopic retrograde cholangiopancreatography and magnetic resonance cholangiopancreatography showed a complete interruption of the pancreatic duct in the body, with mild dilation of the duct in the tail of the pancreas. Endoscopic ultrasonography revealed hypervascularity of the pancreatic tumor. The patient underwent distal pancreatectomy to remove the cause of pancreatitis and to disclose the diagnosis. Histologic examination revealed follicular lymphoma of pancreas. Despite recent improvement in clinical strategies, differential diagnosis between pancreatic lymphoma and pancreatic cancer is still difficult without histologic information. Pancreatic lymphoma should be considered as a differential diagnosis in a patient who initially presents with acute pancreatitis.
Key words: Pancreatic lymphoma, Acute pancreatitis, Follicular lymphoma
Pancreatic B-cell lymphoma is rare; it accounts for 0.2% to 2.0% of extranodal non-Hodgkin lymphoma1 and constitutes less than 0.5% of all pancreatic malignancies.2,3 Most histologic types of pancreatic lymphoma are diffuse large B-cell lymphoma, and follicular lymphoma is quite rare. In addition, acute pancreatitis caused by pancreatic lymphoma is also rare.
Despite recent improvements in clinical strategies, differential diagnosis of pancreatic lymphoma with pancreatic cancer or autoimmune pancreatitis (AIP) is still difficult without histologic examination.
We report here a rare case of a 71-year-old female with pancreatic follicular lymphoma that initially presented as acute pancreatitis.
Case Report
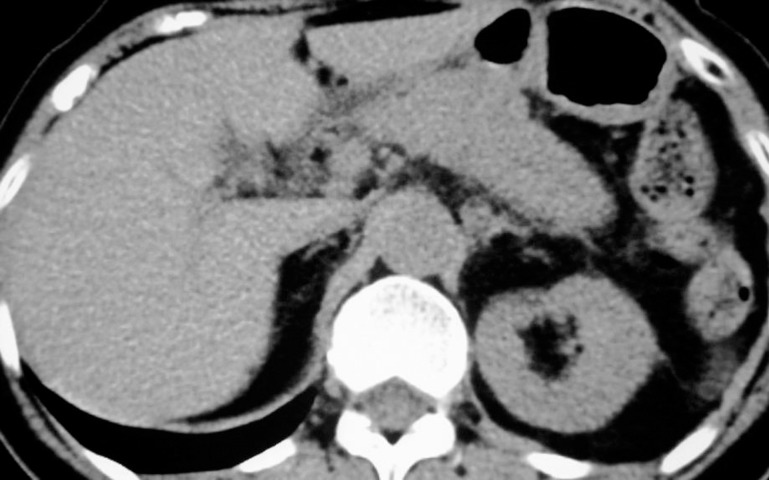
A 71-year-old woman visited our hospital for epigastric and left upper quadrant abdominal pain. On physical examination, she demonstrated tenderness in the left upper quadrant of the abdomen, when no mass was palpable. Laboratory investigations showed elevated serum total amylase (512 U/L; normal range, 40–129 U/L) and pancreatic amylase (403 U/L; normal range, 19–53 IU/L) concentrations. Further laboratory tests disclosed elevated serum lipase (1203 U/L; normal range, 5–35 U/L), elastase 1 (3800 ng/dL; normal value, <300 ng/dL), and lactase dehydrogenase (376 IU/L; normal range, 120–240 U/L) concentrations. Abdominal nonenhanced computed tomography (CT) revealed an edema around the tail of the pancreas, and a cloudy fat density in the anterior pararenal space (Fig. 1). There was no evidence of stones in the gallbladder or the common bile duct. Acute pancreatitis was initially suspected and the patient was admitted to the hospital.
Fig. 1.
Abdominal plain CT revealed edema around the tail of the pancreas and a cloudy fat density in the anterior pararenal space.
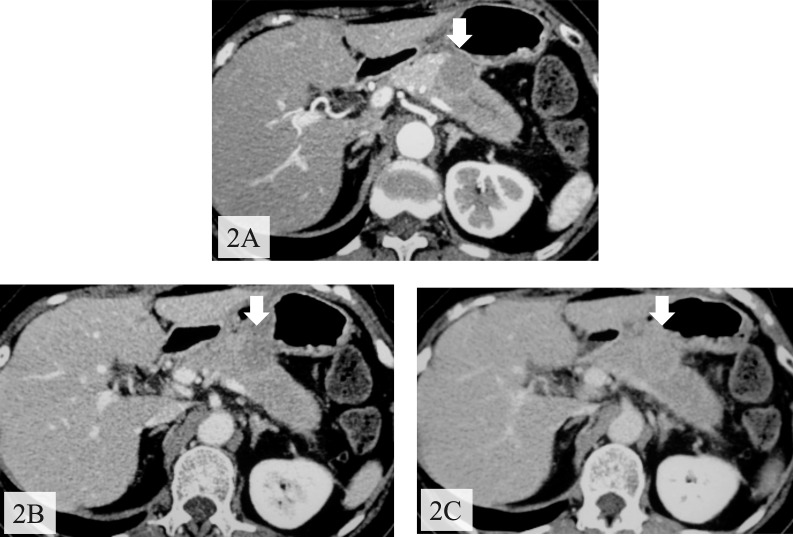
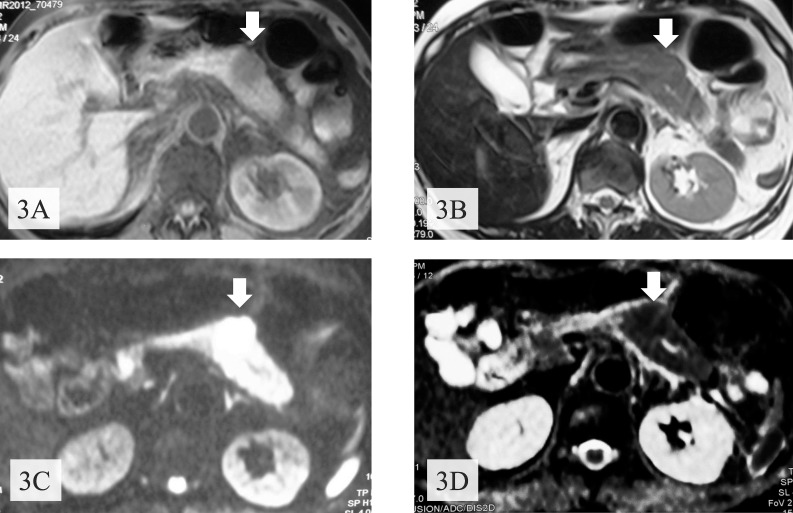
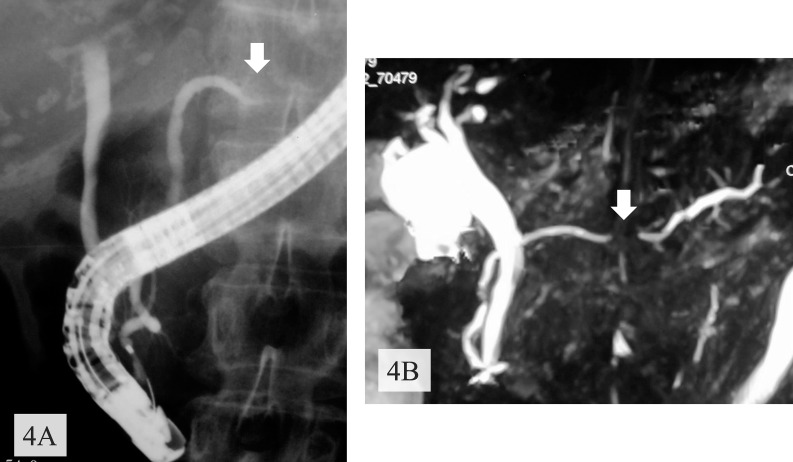
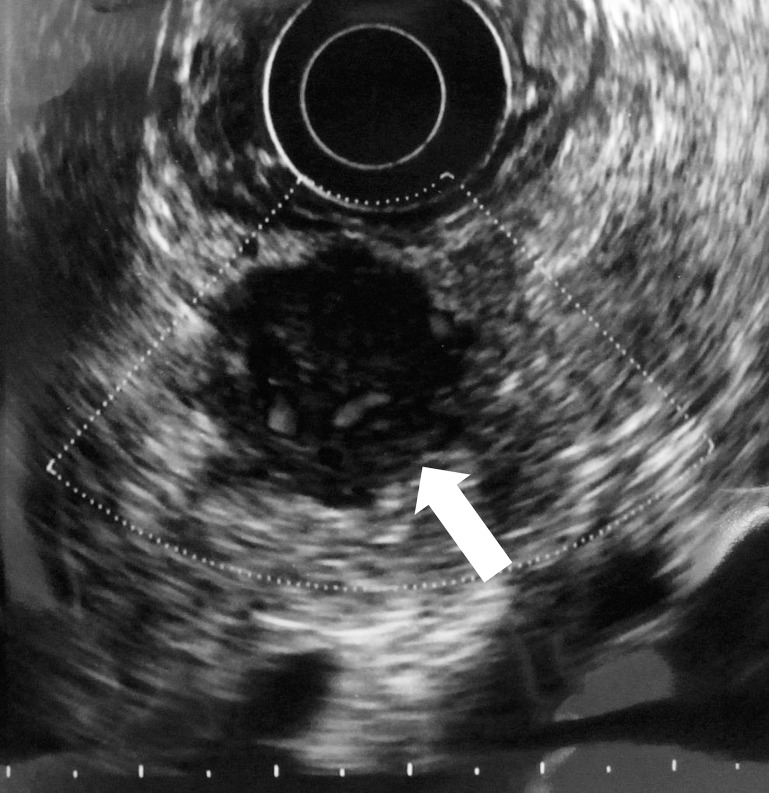
After the standard therapy for acute pancreatitis, the abdominal pain disappeared and abnormal laboratory data were normalized. Enhanced CT showed a nonenhanced round tumor (50 × 35 mm) of pancreatic body in the early phase (Fig. 2A), but the tumor was gradually more enhanced in the portal (Fig. 2B) and venous (Fig. 2C) phases. Furthermore, the right ovary and a mesenteric lymph node were found to be swollen. Magnetic resonance imaging revealed a tumor presenting with low intensity on T1-weighted images with fat suppression (Fig. 3A), isointensity on T2-weighted images (Fig. 3B), high intensity on diffusion magnified-weight imaging (Fig. 3C), and isointensity on apparent diffusion coefficient mapping (Fig. 3D). Endoscopic retrograde cholangiopancreatography (ERCP; Fig. 4A) and magnetic resonance cholangiopancreatography (Fig. 4B) showed a complete interruption of the main pancreatic duct (MPD) in the body with a mild dilation in the tail of the pancreas. Endoscopic ultrasound sonography (EUS) disclosed a hypoechoic mass in the body of the pancreas. We abandoned echoendoscope-guided fine-needle aspiration because the tumor was found to be hypervascular by Doppler scanning (Fig. 5). The levels of serum carcinoembryonic antigen, serum carbohydrate antigen 19-9, and immunoglobulin G were normal.
Fig. 2.
Enhanced CT showed a tumor in the body of the pancreas with delayed contrast enhancement. (A) Arterial phase. (B) Portal phase. (C) Venous phase.
Fig. 3.
Magnetic resonance imaging revealed a tumor (arrows) with low intensity on T1-weighted images with fat suppression (A), with isointensity on T2-weighted images (B), with high intensity on diffusion magnified-weight imaging (C), and with isointensity on apparent diffusion coefficient mapping (D).
Fig. 4.
ERCP (A) and magnetic resonance cholangiopancreatography (B) showed complete interruption (arrows) of the pancreatic duct in the body of the pancreas.
Fig. 5.
EUS disclosed a hypoechoic mass in the body of the pancreas with hypervascularity of the tumor (arrow).
We judged that pancreatitis had been caused by the pancreatic tumor. In addition, the tumor was suspected to be the pancreatic cancer. Thus, we performed distal pancreatectomy with lymph node dissection so as to remove the cause of pancreatitis and to disclose the diagnosis.
Operative findings indicated that the tumor, located in the body of the pancreas, was elastic and hard on palpation. Intraoperative histologic examination indicated that the tumor was diagnosed as a lymphoid proliferative mass. We also excised both swollen ovaries and a mesenteric lymph node.
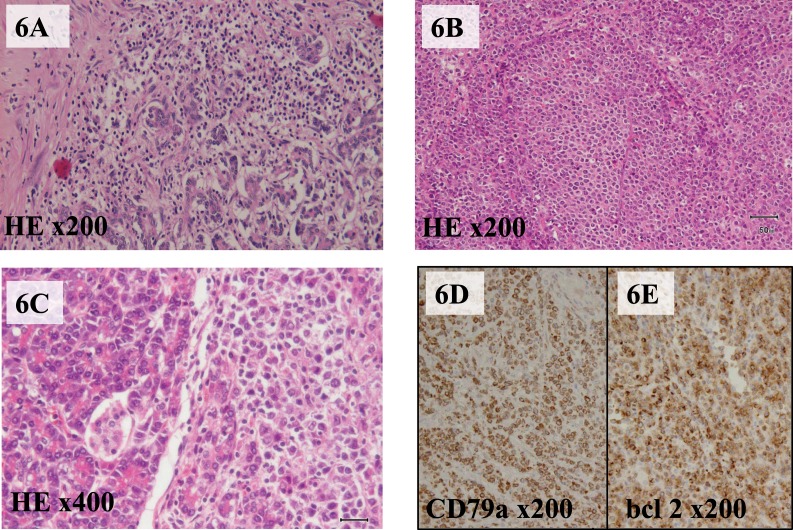
The final histologic diagnosis was acute pancreatitis and a B-cell type follicular lymphoma of the pancreas (Fig. 6). Immunohistochemical analysis demonstrated that the tumor stained positive for CD79a, CD20, CD10, and bcl-2 protein. The ovaries and the mesenteric lymph node indicated the same appearance. The lymphoma was classified as grade 2 (6–15 centroblasts per high-power field) using the Berard classification. According to the Ann Arbor Staging System, this case was categorized as stage II. After the operation, the patient was discharged uneventfully. The patient was readmitted and received chemotherapy.
Fig. 6.
(A) Microscopic examination following hematoxylin-eosin staining, which showed acute pancreatitis with lymphocyte-infiltrating parenchyma of the pancreas tail. (B) Hematoxylin-eosin staining, which demonstrated follicular-type lymphoma cells. (C) Lymphoma cells involving the parenchyma of the exocrine pancreas and Langerhans islets. Both CD79a immunostaining (D) and bcl-2 immunostaining (E) are positive.
Discussion
Pancreatic B-cell lymphoma is rare, in which follicular type is even rarer. Baylor and Berg4 reported the estimated frequency of pancreatic lymphoma in a surgical series of pancreatic tumor to be 0.2%. Most cases are intermediate or high-grade non-Hodgkin lymphoma, with diffuse large B-cell lymphoma being the predominant (about 60%) histotype.5 Shia et al6 reported 26 cases of primary follicular lymphoma of the gastrointestinal tract, and none of them involved the pancreas. Salvatore et al7 summarized only 3 of 60 cases of the pancreatic tumor as being follicular lymphoma.
The clinical presentation of pancreatic lymphoma is nonspecific, including abdominal pain, weight loss, jaundice, nausea, and abdominal mass in 83%, 50%, 37%, 34%, and 58% of cases, respectively.8 Systemic symptoms, such as fever, night sweat, and weight loss, which are often seen in malignant lymphoma, are uncommon in pancreatic lymphoma.9 Pancreatic B-cell lymphoma presenting with acute pancreatitis as an initial pathogenesis is quite rare. To our knowledge, only 13 such cases have been described in the literature.9–14 This is the first case of pancreatic follicular lymphoma presenting with acute pancreatitis.
Because of the marked difference in treatment between pancreatic cancer and lymphoma, differential diagnosis of them is very important. However, diagnosis of pancreatic lymphoma is difficult without the aid of histologic examination because patients' symptoms, laboratory findings, or imaging findings are nonspecific.1,15
ERCP seems useful in differentiating pancreatic lymphoma from pancreatic cancer. A previous study reported that MPD was normal in almost 30% of patients with pancreatic lymphoma on ERCP. Furthermore, there is no distal dilatation of MPD.5 In contrast, the MPD of pancreatic adenocarcinoma is usually associated with moderate to severe dilatation.16 In the present case, MPD was completely interrupted on ERCP and magnetic resonance cholangiopancreatography, suggesting adenocarcinoma.
EUS has also been used to evaluate pancreatic tumors. Flamenbaum et al17 described the typical findings of pancreatic lymphoma as a strongly hypoechoic mass with a hyperechoic wall in MPD with multiple isoechogenic peripancreatic lymph nodes. Furthermore, EUS enables an accurate fine-needle aspiration or core biopsy. These histological examinations have become a standard tool for the diagnosis of pancreatic lymphoma because they are highly accurate and easy.18 Volmer et al19 examined a total of 1050 pancreatic fine-needle biopsies and could diagnose 14 cases of pancreatic lymphoma, including 4 cases (0.4%) of follicular type. In the current case, EUS showed a hypoechoic mass in the body of the pancreas and a nontypical hypoechoic wall of MPD. It also revealed hypervascularity of the tumor by Doppler scanning. AIP is an autoimmune-mediated chronic pancreatitis characterized by the formation of tumors and irregular stenosis of the pancreatic duct, presenting as abdominal pain, epigastric discomfort, and jaundice in the middle-aged male patient. AIP may mimic pancreatic lymphoma. Therefore, differential diagnosis between pancreatic lymphoma and AIP is important. AIP is usually associated with hyperglobulinemia and autoantibodies. Wu et al20 reported 19 cases of AIP, and almost all of them showed elevated serum γ-globulin levels, particularly immunoglobulin G. In addition, serum CA19-9 levels were slightly elevated compared with patients with nonautoimmune pancreatitis. In the current patient, male sex and the normal levels of serum CA19-9 and autoimmune antibodies were not confusing.
To the best of our knowledge, this is the first reported case of pancreatic follicular lymphoma presenting with acute pancreatitis. Pancreatic lymphoma needs to be included in the differential diagnosis of acute pancreatitis.
References
- 1.Mortenson MM, Katz MH, Tamm EP, Bhutani MS, Wang H, Evans DB. Current diagnosis and management of unusual pancreatic tumors. Am J Surg. 2008;196(1):100–113. doi: 10.1016/j.amjsurg.2008.02.005. [DOI] [PubMed] [Google Scholar]
- 2.Weingrad DN, Decosse JJ, Sherlock P, Straus D, Lieberman PH, Flippa DA. Primary gastrointestinal lymphoma: a 30-year review. Cancer. 1982;49(6):1258–1265. doi: 10.1002/1097-0142(19820315)49:6<1258::aid-cncr2820490630>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 3.Tanaka T, Matsugu Y, Koide K, Miura Y, Ichiba Y, Dohi K. Malignant lymphoma of the pancreas. Dig Dis Sci. 1996;41(2):402–404. doi: 10.1007/BF02093835. [DOI] [PubMed] [Google Scholar]
- 4.Baylor SM, Berg JW. Cross-classification and survival characteristics of 5,000 cases of cancer of the pancreas. J Surg Oncol. 1973;5(4):335–358. doi: 10.1002/jso.2930050410. [DOI] [PubMed] [Google Scholar]
- 5.Saif MW. Primary pancreatic lymphomas. JOP. 2006;7(3):262–273. [PubMed] [Google Scholar]
- 6.Shia J, Teruya-Feldstein J, Pan D, Hegde A, Klimstra DS, Chaganti RS. Primary follicular lymphoma of the gastrointestinal tract: a clinical and pathologic study of 26 cases. Am J Surg Pathol. 2002;26(2):216–224. doi: 10.1097/00000478-200202000-00008. [DOI] [PubMed] [Google Scholar]
- 7.Salvatore JR, Cooper B, Shah I, Kummet T. Primary pancreatic lymphoma: a case report, literature review, and proposal for nomenclature. Med Oncol. 2000;17(3):237–247. doi: 10.1007/BF02780536. [DOI] [PubMed] [Google Scholar]
- 8.Mofredj A, Cadranel JF, Cazier A, Traore I, Coutarel P, Levy P. Malignant pancreatic non-Hodgkin's lymphoma manifesting as severe acute pancreatitis. Gastroenterol Clin Biol. 1999;23(4):528–531. [PubMed] [Google Scholar]
- 9.Federico E, Falconi M, Zuodar G, Falconieri G, Puglisi F. B-cell lymphoma presenting as acute pancreatitis. Pancreatology. 2011;11(6):553–556. doi: 10.1159/000332038. [DOI] [PubMed] [Google Scholar]
- 10.Saif MW, Khubchandani S, Walczak M. Secondary pancreatic involvement by a diffuse large B-cell lymphoma presenting as acute pancreatitis. World J Gastroenterol. 2007;13(36):4909–4911. doi: 10.3748/wjg.v13.i36.4909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee MK, Jeon SW, Lee YD, Seo HE, Cho CM, Kim SG. A case of primary pancreatic non-Hodgkin's lymphoma. Korean J Intern Med. 2006;21(2):123–126. doi: 10.3904/kjim.2006.21.2.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bernardeau M, Auroux J, Cavicchi M, Haioun C, Tsakiris L, Delchier JC. Secondary pancreatic involvement by diffuse large B-cell lymphoma presenting as acute pancreatitis: treatment and outcome. Pancreatology. 2002;2(4):427–430. doi: 10.1159/000065093. [DOI] [PubMed] [Google Scholar]
- 13.Sugishita H, Watanabe Y, Yamamoto Y, Yoshida M, Sato K, Horiuchi A. Primary pancreatic lymphoma: the role of surgical treatment. Case Rep Gastroenterol. 2010;4(1):104–110. doi: 10.1159/000283405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.To CA, Quigley MM, Saven A, Nicholson L. Masquerade without a mass: an unusual cause of severe acute pancreatitis. J Gastrointest Oncol. 2013;4(1):114–117. doi: 10.3978/j.issn.2078-6891.2012.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ravi K, Sanchez W, Sweetser S. Primary pancreatic follicular lymphoma mimicking adenocarcinoma. Clin Gastroenterol Hepatol. 2010;8(10):e101–e102. doi: 10.1016/j.cgh.2010.05.011. [DOI] [PubMed] [Google Scholar]
- 16.Tuchek JM, De Jong SA, Pickman J. Diagnosis, surgical intervention, and prognosis of primary pancreatic lymphoma. Am Surg. 1993;59(8):513–518. [PubMed] [Google Scholar]
- 17.Flamenbaum M, Pujol B, Souquet JC, Cassan P. Endoscopic ultrasonography of a pancreatic lymphoma. Endoscopy. 1998;30(3):S43. doi: 10.1055/s-2007-1001273. [DOI] [PubMed] [Google Scholar]
- 18.Markle EM, Bender GN, Brambs HJ. Imaging findings in pancreatic lymphoma: differential aspects. AJR Am J Roentgenol. 2000;174(3):671–675. doi: 10.2214/ajr.174.3.1740671. [DOI] [PubMed] [Google Scholar]
- 19.Volmar KE, Routbort MJ, Jones CK, Xie HB. Primary pancreatic lymphoma evaluated by fine-needle aspiration: findings in 14 cases. Am J Clin Pathol. 2004;121(6):898–903. doi: 10.1309/UAD9-PYFU-A82X-9R9U. [DOI] [PubMed] [Google Scholar]
- 20.Wu WC, Yao XZ, Jin DY, Wang DS, Lou WH, Qin XY. Clinical strategies for identifying autoimmune pancreatitis from pancreaticobiliary malignancy to avoid unnecessary surgical resection. J Dig Dis. 2013;14(9):500–508. doi: 10.1111/1751-2980.12075. [DOI] [PubMed] [Google Scholar]