Abstract
Strategies to increase expanded criteria donor (ECD) transplantation are needed. We quantified the extent to which ECD kidneys provide recipients with a lifetime of allograft function by determining the difference between patient survival and death-censored allograft survival (graft survival). Initial analyses compared 5-year outcomes in the Eurotransplant Senior Program (European) and the United States Renal Data System. Among European recipients ≥65 years, patient survival exceeded graft survival, and ECD recipients returned to dialysis for an average of 5.2 months after transplant failure. Among United States recipients ≥60 years, graft survival exceeded patient survival. Although patient survival in elderly recipients in the United States was low (49% at 5 years), the average difference in patient survival at 10 years in elderly recipients in the United States with an ECD versus non-ECD transplant was only 7 months. The probability of patient survival with a functioning allograft at 5 years was higher with ECD transplantation within 1 year after activation to the waiting list than with delayed non-ECD transplantation ≥3 years after activation to the waiting list. Subsequent analyses demonstrated that ECD transplants do not provide a lifetime of allograft function in recipients <50 years in the United States. These findings should encourage ECD transplantation in patients ≥60 years, demonstrate that rapid ECD transplantation is superior to delayed non-ECD transplantation, and challenge the policy in the United States of allowing patients <50 years to receive an ECD transplant.
Keywords: transplantation, survival, expanded criteria donors, health policy, cadaver organ transplantation
The number of patients awaiting kidney transplantation in the United States recently eclipsed 100,000.1 Despite these staggering numbers, a high number of deceased donor kidneys from older aged donors are discarded, and in 2012, 60% of the kidneys recovered from deceased donors aged ≥65 years in the United States were not transplanted.2
In the new American kidney allocation scheme, to expedite placement and encourage utilization of expanded criteria donor (ECD) kidneys, priority for local allocation will be eliminated and ECD kidneys will be offered at the regional level.3 However, despite evidence that only certain patient groups will benefit from transplantation of ECD kidneys4 and that patients who will not benefit continue to be listed and transplanted with ECD kidneys,5 the new allocation scheme does not restrict which wait-list candidates can receive ECD kidneys.3 In contrast, the Eurotransplant Senior Program (ESP) restricts the transplantation of kidneys from deceased donors aged ≥65 years to recipients ≥65 years, but still allows patients aged ≥65 years the option of waiting for a kidney from a younger deceased donor.6 Previous work has shown that the discard of ECD kidneys in the ESP is significantly lower than that in the United States.7
In this study we report a series of analyses to inform the optimal use of ECD kidneys. The first group of analyses compares the use and outcomes of ECD kidneys in the ESP and the United States. The purpose of these analyses was to demonstrate the effect of different allocation policies on the utilization and outcome of ECD transplantation. The second set of analyses examined the consequences of continuing to allow any consenting wait-list candidates to undergo transplantation with an ECD kidney in the United States.
Under the new American kidney allocation system, ECDs will be defined by a Kidney Donor Profile Index (KDPI)>85%.8 The KDPI is a linear scale from 0% to 100% that transforms the relative risk of graft loss for any deceased donor kidney compared with that of a kidney from a donor aged 40 years, with 0% representing the longest projected survival and 100% representing the shortest survival.8 The KDPI is calculated on the basis of donor age, height, ethnicity, history of hypertension, diabetes, cause of death, serum creatinine, hepatitis C status, and donation after circulatory death status.8 Given that KDPI is not used in the ESP, and because most deceased donors aged ≥65 years would be classified as ECD in the United States, we defined ECD on the basis of deceased donor age of ≥65 years to maintain consistency in all analyses.
Results
Use and Outcomes of ECD Kidneys in the ESP and the United States
During the period 1999–2003, the number of transplants from deceased donors aged ≥65 years in the ESP was 1870 compared with only 1312 in the entire United States. ESP recipients received kidneys from donors that were older and more frequently had a history of hypertension compared with United States recipients (Table 1). Over 80% of ESP recipients were ≥65 years old compared with only 34% of United States recipients. ESP recipients were less likely to have diabetic ESRD or obesity than United States recipients, but they had a longer exposure to dialysis before transplantation (Table 1). Because the ESP excludes sensitized patients,6 97% of ESP patients had plasma renin activity <5%. Compared with ESP recipients, United States recipients were treated nearly twice as frequently with depleting antibody induction therapy (Table 1). The incidence of delayed graft function (ESP: 32%; United States: 34%) and primary nonfunction (ESP: 7%; United States: 6%) were clinically similar in ESP and United States recipients. Similar differences were observed among the subset of recipients aged ≥65 years (Table 2).
Table 1.
Characteristics of ECD transplant recipients
| Characteristic | ESP Recipients (n=1870) | United States Recipients (n=1312) | P Value |
|---|---|---|---|
| Donor characteristics | |||
| Age (yr), median (q1, q3) | 69 (67, 73) | 69 (66, 71) | <0.001 |
| Age ≥70 yr | 882 (47)a | 462 (35) | <0.001 |
| Male sex | 879 (47) | 586 (45) | 0.19 |
| History of diabetes | 130 (7) | 109 (8) | 0.15 |
| History of hypertension | 989 (53) | 626 (48) | <0.01 |
| Cerebrovascular accident as cause of death | 1309 (70) | 1022 (78) | <0.001 |
| Recipient characteristics | |||
| Age (yr), median (q1, q3) | 66 (65, 69) | 60 (52, 67) | <0.001 |
| Age (yr) | <0.001 | ||
| ≥65 | 1520 (81) | 446 (34) | |
| 60–64 | 118 (6) | 271 (21) | |
| 55–59 | 64 (3) | 194 (15) | |
| 50–54 | 54 (3) | 152 (12) | |
| <50 | 114 (6) | 249 (19) | |
| Male sex | 1211 (65) | 785 (60) | <0.01 |
| Race | N/A | N/A | |
| White | 820 (63) | ||
| Black | 411 (31) | ||
| Other | 81 (6) | ||
| Cause of ESRD | <0.001 | ||
| Diabetes | 171 (9) | 412 (33) | |
| Other | 1699 (91) | 821 (67) | |
| Missing | N/A | 71 | |
| Body mass index (kg/m2) | <0.001 | ||
| <30 | 1464 (90) | 743 (75) | |
| ≥30 | 171 (10) | 247 (25) | |
| Missing | 235 | 322 | |
| Time on dialysis (yr) | <0.001 | ||
| Median (q1, q3) | 3.36 (1.97, 5.25) | 2.85 (1.69, 4.61) | |
| Peak panel reactive antibody (%) | <0.001 | ||
| <5 | 1802 (97) | 850 (70) | |
| 5–30 | 44 (2) | 237 (20) | |
| >30 | 20 (1) | 122 (10) | |
| Missing | 4 | 103 | |
| Cold ischemic time (hr) | <0.001 | ||
| 0–12 | 824 (46) | 135 (12) | |
| 12.1–24 | 875 (49) | 585 (53) | |
| >24 | 99 (5) | 392 (35) | |
| Missing | 72 | 199 | |
| Induction therapy | <0.001 | ||
| Depleting antibody | 264 (16) | 375 (31) | |
| Nondepleting antibody | 489 (31) | 388 (32) | |
| Neither | 839 (53) | 444 (37) | |
| Missing | 278 | 105 | |
| Calcineurin inhibitor | <0.001 | ||
| Tacrolimus | 426 (28) | 660 (58) | |
| Cyclosporine | 1056 (71) | 390 (34) | |
| Sirolimus | 16 (1) | 93 (8) | |
| Missing | 372 | 169 | |
| Antimetabolite | <0.001 | ||
| Mycophenolate mofetil | 1339 (90) | 963 (96) | |
| Azathioprine | 144 (10) | 40 (4) | |
| Missing | 387 | 309 |
N/A, not applicable.
Values represent n (%) unless otherwise specified.
Table 2.
Characteristics of patients aged ≥65 years who received an ECD transplant
| Characteristic | ESP Recipients (n=1520) | United States Recipients (n=446) | P Value |
|---|---|---|---|
| Donor characteristics | |||
| Age (yr), median (q1, q3) | 69 (67, 73) | 69 (67, 72) | 0.07 |
| Age ≥70 yr | 741 (49)a | 196 (44) | 0.07 |
| Male sex | 704 (46) | 192 (43) | 0.22 |
| History of diabetes | 113 (7) | 40 (9) | 0.29 |
| History of hypertension | 842 (55) | 228 (51) | 0.11 |
| Cerebrovascular accident as cause of death | 1063 (70) | 358 (80) | <0.01 |
| Recipient characteristics | |||
| Male sex | 988 (65) | 286 (64) | 0.73 |
| Race | N/A | N/A | |
| White | 315 (71) | ||
| Black | 102 (23) | ||
| Other | 49 (6) | ||
| Cause of ESRD | <0.001 | ||
| Diabetes | 141 (9) | 132 (30) | |
| Other | 1379 (91) | 307 (70) | |
| Missing | 0 | 7 | |
| Body mass index (kg/m2) | <0.001 | ||
| <30 | 1208 (89) | 293 (82) | |
| ≥30 | 150 (11) | 65 (18) | |
| Missing | 162 | 88 | |
| Time on dialysis (yr), median (q1, q3) | 3.36 (2.08, 5.16) | 2.58 (1.58, 3.87) | <0.001 |
| Panel reactive antibody | <0.001 | ||
| <5% | 1489 (98) | 315 (73) | |
| 5%–30% | 25 (2) | 86 (20) | |
| >30% | 3 (0) | 30 (7) | |
| Missing | 3 | 15 | |
| Cold ischemic time (hr) | <0.001 | ||
| 0–12 | 774 (53) | 50 (13) | |
| 12.1–24 | 652 (44) | 210 (56) | |
| >24 | 42 (3) | 118 (31) | |
| Missing | 52 | 68 | |
| Induction therapy | <0.01 | ||
| Depleting antibody | 240 (19) | 98 (24) | |
| Nondepleting antibody | 421 (32) | 151 (36) | |
| Neither | 639 (49) | 165 (40) | |
| Missing | 220 | 32 | |
| Calcineurin inhibitor | <0.001 | ||
| Tacrolimus | 344 (29) | 213 (54) | |
| Cyclosporine | 840 (70) | 142 (36) | |
| Sirolimus | 13 (1) | 41 (10) | |
| Missing | 323 | 50 | |
| Antimetabolite | <0.01 | ||
| Mycophenolate mofetil | 1107 (90) | 328 (95) | |
| Azathioprine | 119 (10) | 16 (5) | |
| Neither | 294 | 102 |
Values represent n (%) unless otherwise specified.
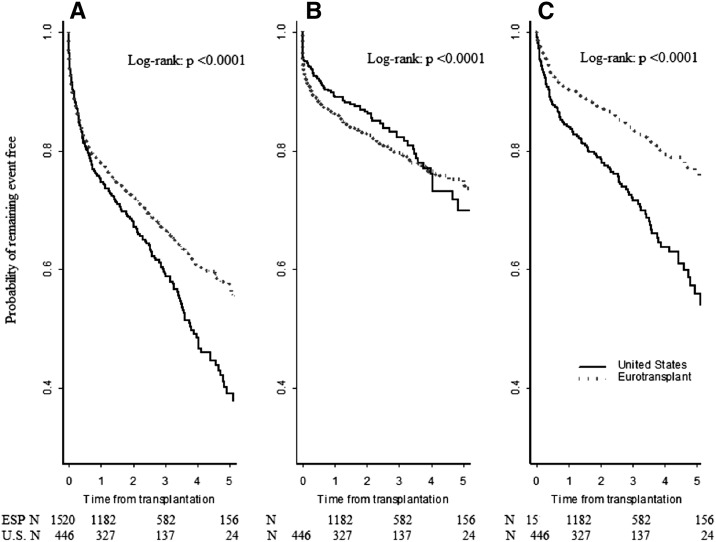
Figure 1 shows Kaplan–Meier plots comparing transplant outcomes among the 1520 ESP and 446 United States recipients who were ≥65 years of age at the time of kidney transplantation from a deceased donor ≥65 years old. The higher allograft survival in ESP recipients was the result of a higher incidence of death with a functioning graft in United States recipients.
Figure 1.
Kaplan–Meier analyses comparing transplant outcomes among the 1520 ESP and 446 United States recipients who were ≥65 years of age at the time of kidney transplantation from a deceased donor ≥65 years. (A) All cause graft loss. (B) Death censored graft loss. (C) Death with a functioning graft.
Table 3 shows the results of separate Cox multivariate regression analyses for the outcomes of graft loss from any cause, death-censored allograft loss, and death with a functioning graft. Among transplant recipients ≥65 years of age, there was a lower risk of graft loss from any cause and death with a functioning graft in ESP compared with United States recipients, but the risk of death-censored allograft loss was similar in both groups.
Table 3.
Relative risk of transplant failure in ESP compared with United States transplant recipients
| Graft and Patient Outcomes | Univariate | Multivariate |
|---|---|---|
| All-cause graft loss | 0.73 (0.62, 0.86) | 0.74 (0.60, 0.93) |
| Death-censored graft loss | 1.07 (0.83, 1.36) | 1.10 (0.79, 1.54) |
| Death with function | 0.52 (0.42, 0.64) | 0.54 (0.40, 0.73) |
Separate Cox multivariate regression analyses adjusted for differences in donor age, sex, diabetes, and hypertension; recipient age, sex, cause of end-stage kidney disease, body mass index, peak panel reactive antibody, time on dialysis before transplantation; and immunosuppression and induction therapy at time of transplantation. Values in parentheses are expressed as 95% confidence intervals.
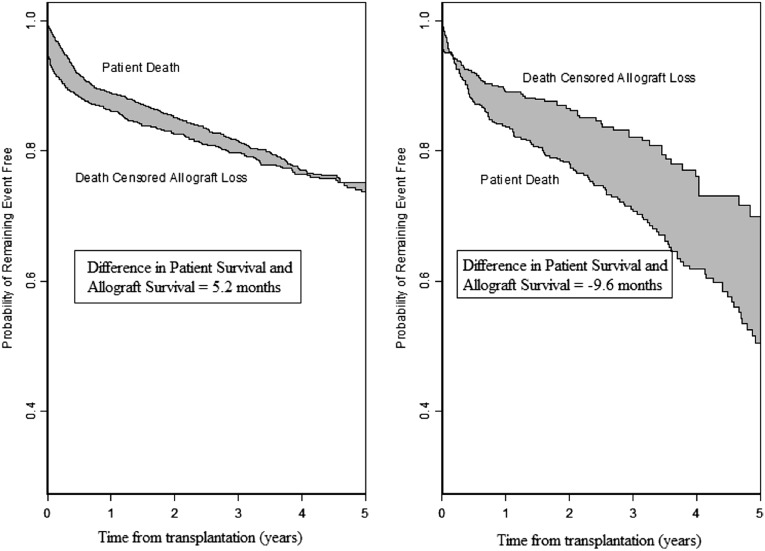
Figure 2 shows the difference in patient and death-censored allograft survival at 5 years after transplantation in ESP and United States recipients aged ≥65 years who underwent transplantation with a deceased donor kidney from a donor aged ≥65 years. Table 4 quantifies the difference in mean 5-year patient and death-censored allograft survival in ESP and United States recipients aged ≥65 years. ESP recipients were more likely to outlive their transplant, requiring them to return to dialysis for an average of 5.2 months over the 5-year follow-up period. Among United States recipients, death-censored allograft survival exceeded patient survival by 9.6 months, indicating that, on average, elderly United States recipients aged ≥65 years die with a functioning allograft after ECD transplantation (Table 5).
Figure 2.
Patient and death-censored allograft survival for recipients aged ≥65 years who received transplantation from a deceased donor aged ≥65 years in the ESP and the United States. The shaded area represents the difference between patient and death-censored allograft survival. ESP recipients were more likely to outlive their transplant requiring them to return to dialysis for an average of 5.2 months over the 5-year follow-up period. Among United States recipients, the death-censored allograft survival exceeded patient survival by 9.6 months, indicating that on average, elderly United States recipients aged ≥65 years die with a functioning allograft after ECD transplantation.
Table 4.
Mean difference in patient versus death-censored allograft survival in elderly ESP recipients versus United States recipients after 5 years of transplantation
| Patient and Graft Survival | ESP Recipients (n=1520) | United States Recipients (n=446) |
|---|---|---|
| Patient survival (yr) | 4.18±0.04 | 3.63±0.09 |
| Graft survival (yr) | 3.75±0.04 | 4.05±0.08 |
| Difference in patient and graft survival (mo) | 0.43±0.06 | −0.42±0.12 |
| 5.2±0.72 | −5.0±1.44a |
Values are mean±SEM.
Negative value indicates mean death-censored allograft survival exceeds mean patient survival and on average patients will die with a functioning allograft.
Table 5.
Adjusted differences in 10-year patient survival among transplant recipients aged ≥60 years after transplantation with a deceased donor aged <65 and ≥65 years
| Recipient Age Group | Donor Characteristics | Area under Survival Curve (mo)a | Difference in Patient Survival (mo) Compared with Transplantation from a Donor Aged ≥65 yr |
|---|---|---|---|
| Recipient 60–64 yr | Donor <65 yr | ||
| 60%–69% | 84.4±1.1 | 8.2±1.8 | |
| 70%–79% | 85.6±1.1 | 9.4±1.8 | |
| 80%–85% | 85.1±1.3 | 8.9±2.0 | |
| ≥86% | 82.0±1.0 | 5.8±1.7 | |
| Donor ≥65 yr | 76.2±1.44 | ||
| Recipient 65–69 yr | Donor <65 yr | ||
| 60%–69% | 80.2±1.4 | 7.3±2.0 | |
| 70%–79% | 80.0±1.3 | 7.2±2.0 | |
| 80%–85% | 77.5±1.6 | 4.7±2.1 | |
| ≥86% | 77.2±1.2 | 4.3±1.9 | |
| Donor ≥65 yr | 72.8±1.4 | ||
| Recipient ≥70 yr | Donor <65 yr | ||
| 60%–69% | 74.8±1.9 | 7.3±2.5 | |
| 70%–79% | 74.4±1.8 | 7.0±2.4 | |
| 80%–85%b | 79.1±2.2 | 11.6±2.7 | |
| ≥86% | 69.8±1.4 | 2.4±2.1 | |
| Donor ≥65 yr | 67.4±1.6 |
Values are mean±SEM.
Area calculated from adjusted survival curves generated from Cox multivariate models.
Sixteen recipients remained at risk after 120 months.
Probability of Being Alive and Being Alive with a Function Allograft at 5 Years
The probability of survival 5 years after activation to the waiting list and the probability of being alive with a functioning allograft at this time point are shown for patients with and without diabetes aged 60–64, 65–69, and ≥70 years (Table 6). Overall, the results show a higher or equivalent probability of being alive with a functioning allograft with transplantation from an ECD within the first year after activation to the wait-list compared with transplantation ≥3 years after activation to the wait-list from a non-ECD.
Table 6.
Survival probabilities by patient age and diabetic status
| Patient Age and Diabetic Status | Probability of Being Alive at 5 yr after Wait-Listing | Probability of Being Alive with Functioning Allograft 5 yr after Wait-Listing |
|---|---|---|
| Nondiabetic patients aged 60–64 yr | ||
| ECD after 1 yr | 0.77 (0.75, 0.79) | 0.68 (0.66, 0.71) |
| ECD after 2 yr | 0.76 (0.74, 0.77) | 0.69 (0.67, 0.71) |
| ECD after 3 yr | 0.72 (0.71, 0.73) | 0.66 (0.64, 0.67) |
| ECD after 4 yr | 0.65 (0.64, 0.66) | 0.61 (0.59, 0.62) |
| Non-ECD after 1 yr | 0.82 (0.81, 0.83) | 0.77 (0.76, 0.78) |
| Non-ECD after 2 yr | 0.78 (0.77, 0.79) | 0.75 (0.74, 0.76) |
| Non-ECD after 3 yr | 0.73 (0.72, 0.74) | 0.70 (0.69, 0.71) |
| Non-ECD after 4 yr | 0.66 (0.65, 0.67) | 0.63 (0.62, 0.65) |
| Diabetic patients aged 60–64 yr | ||
| ECD after 1 yr | 0.68 (0.65, 0.70) | 0.60 (0.57, 0.62) |
| ECD after 2 yr | 0.66 (0.64, 0.68) | 0.60 (0.57, 0.62) |
| ECD after 3 yr | 0.60 (0.59, 0.62) | 0.55 (0.53, 0.57) |
| ECD after 4 yr | 0.52 (0.50, 0.53) | 0.48 (0.47, 0.50) |
| Non-ECD after 1 yr | 0.73 (0.72, 0.75) | 0.69 (0.67, 0.70) |
| Non-ECD after 2 yr | 0.70 (0.68, 0.71) | 0.66 (0.65, 0.68) |
| Non-ECD after 3 yr | 0.62 (0.61, 0.63) | 0.60 (0.58, 0.61) |
| Non-ECD after 4 yr | 0.53 (0.52, 0.54) | 0.51 (0.50, 0.52) |
| Nondiabetic patients aged 65–69 yr | ||
| ECD after 1 yr | 0.74 (0.72, 0.76) | 0.67 (0.65, 0.69) |
| ECD after 2 yr | 0.71 (0.70, 0.73) | 0.65 (0.64, 0.67) |
| ECD after 3 yr | 0.66 (0.64, 0.67) | 0.61 (0.60, 0.63) |
| ECD after 4 yr | 0.58 (0.56, 0.59) | 0.55 (0.54, 0.57) |
| Non-ECD after 1 yr | 0.77 (0.76, 0.78) | 0.73 (0.72, 0.75) |
| Non-ECD after 2 yr | 0.74 (0.73, 0.75) | 0.71 (0.69, 0.72) |
| Non-ECD after 3 yr | 0.67 (0.66, 0.68) | 0.64 (0.63, 0.65) |
| Non-ECD after 4 yr | 0.59 (0.58, 0.60) | 0.57 (0.56, 0.58) |
| Diabetic patients aged 65–69 yr | ||
| ECD at 1 yr | 0.63 (0.60, 0.66) | 0.57 (0.54, 0.60) |
| ECD at 2 yr | 0.62 (0.59, 0.64) | 0.56 (0.54, 0.58) |
| ECD at 3 yr | 0.55 (0.54, 0.57) | 0.51 (0.49, 0.53) |
| ECD at 4 yr | 0.46 (0.45, 0.48) | 0.44 (0.42, 0.45) |
| Non-ECD at 1 yr | 0.72 (0.70, 0.73) | 0.69 (0.67, 0.71) |
| Non-ECD at 2 yr | 0.66 (0.65, 0.68) | 0.64 (0.63, 0.65) |
| Non-ECD at 3 yr | 0.58 (0.57, 0.59) | 0.56 (0.55, 0.57) |
| Non-ECD at 4 yr | 0.47 (0.45, 0.48) | 0.47 (0.45, 0.48) |
| Nondiabetic patients≥70 yr | ||
| ECD at 1 yr | 0.67 (0.65, 0.69) | 0.60 (0.58, 0.63) |
| ECD at 2 yr | 0.65 (0.63, 0.67) | 0.59 (0.57, 0.61) |
| ECD at 3 yr | 0.56 (0.55, 0.58) | 0.52 (0.50, 0.54) |
| ECD at 4 yr | 0.47 (0.45, 0.48) | 0.44 (0.43, 0.46) |
| Non-ECD at 1 yr | 0.75 (0.74, 0.77) | 0.72 (0.70, 0.73) |
| Non-ECD at 2 yr | 0.70 (0.68, 0.71) | 0.67 (0.66, 0.69) |
| Non-ECD at 3 yr | 0.61 (0.60, 0.62) | 0.58 (0.57, 0.60) |
| Non-ECD at 4 yr | 0.49 (0.47, 0.50) | 0.47 (0.46, 0.49) |
| Diabetic patients≥70 yr | ||
| ECD at 1 yr | 0.52 (0.47, 0.56) | 0.47 (0.43, 0.51) |
| ECD at 2 yr | 0.51 (0.48, 0.54) | 0.47 (0.44, 0.50) |
| ECD at 3 yr | 0.43 (0.41, 0.45) | 0.40 (0.37, 0.42) |
| ECD at 4 yr | 0.37 (0.35, 0.40) | 0.34 (0.32, 0.36) |
| Non-ECD at 1 yr | 0.62 (0.59, 0.65) | 0.59 (0.56, 0.62) |
| Non-ECD at 2 yr | 0.56 (0.54, 0.59) | 0.54 (0.52, 0.56) |
| Non-ECD at 3 yr | 0.46 (0.44, 0.49) | 0.45 (0.43, 0.47) |
| Non-ECD at 4 yr | 0.39 (0.36, 0.41) | 0.37 (0.35, 0.40) |
Values in parentheses are expressed as 95% confidence intervals.
Consequences of Continuing To Allow Any Consenting Wait-List Candidate to undergo Transplantation with an ECD Kidney in the United States
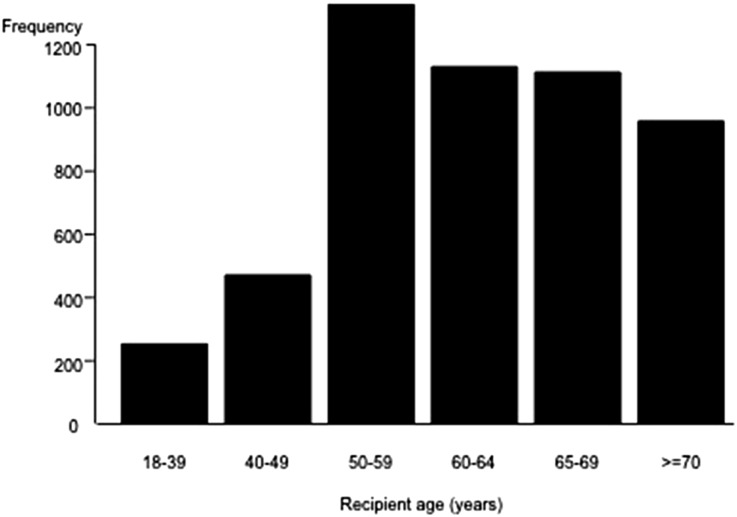
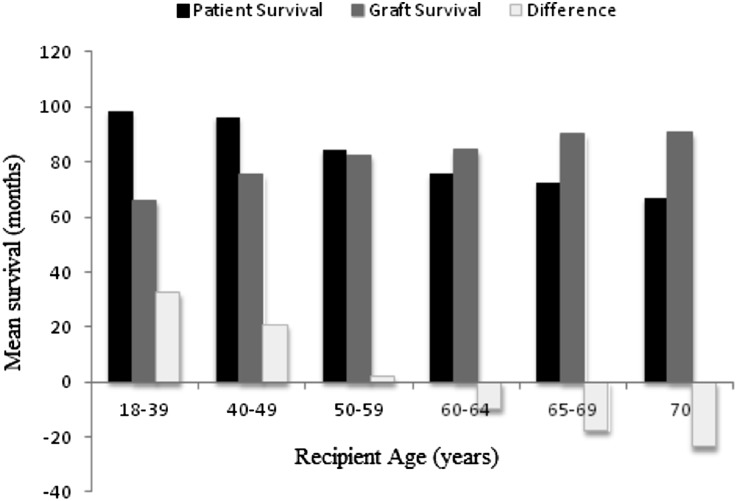
Figure 3 shows the age distribution of 5257 United States kidney transplant recipients from deceased donors aged ≥65 years during the period January 1, 1995–December 31, 2010. Of the recipients, 726 (14%) were <50 years of age. Figure 4 quantifies the difference (in months) between the average patient survival and average death-censored graft survival among different recipient age groups at 10 years after transplantation with a deceased donor ≥65 years. Among patients 18–39 and 40–49 years, the average patient survival exceeded the average death-censored allograft survival. As a result, recipients aged 18–39 years returned to dialysis for an average±SEM of 32±4 months, whereas patients aged 40–49 years returned to dialysis for an average±SEM of 21±3 months in the 10-year time period after transplantation with a deceased donor ≥65 years. In contrast, among recipients aged 50–59 years, the average patient survival (85 months) and graft survival (83 months) over a 10-year period were similar, whereas among recipients aged ≥60 years, patient survival was lower than death-censored allograft survival, indicating that on average transplantation with a deceased donor ≥65 years provided a lifetime of function for patients aged ≥60 years. To understand whether this achievement of a lifetime of allograft function in patients aged ≥60 years in the United States was determined by shorter patient survival after transplantation with a deceased donor kidney aged ≥65 years, we quantified the difference in 10-year patient survival (in months) among recipients aged ≥60 years after transplantation from a deceased donor aged <65 years and ≥65 years (Table 5), after adjustment for differences in patient and transplant characteristics that may effect patient survival. To ensure this information was relevant to the new kidney allocation policy, the results among deceased donors recipients aged <65 years were stratified by the KDPI. The decrement in 10-year mean patients survival with transplantation from a donor aged ≥65 years compared with a donor aged <65 years with KDPI 60%–69% was 8.2, 7.3, and 7.3 months, respectively, in recipients aged 60–64, 65–69, and ≥70 years (Table 5).
Figure 3.
Age distribution of 5257 kidney transplant recipients from a deceased donor aged ≥65 years in the United States (1995–2010).
Figure 4.
Ten-year mean patient and death-censored allograft survival and difference between curves (in months) after transplantation from a deceased donor aged ≥65 years in the United States. Positive values for the difference (white bars) indicate average patient survival was greater than death-censored allograft survival, whereas negative values indicate death-censored allograft survival was greater then patient survival.
Outcome of Recipients <50 Years with a Failed ECD Transplant
There were 726 (14%) recipients aged <50 years who underwent transplantation from a donor aged ≥65 years in the United States from 1995 to 2010. Of these, 408 (56%) suffered death-censored graft failure after a median time of 37.2 months (quartile 1 [q1]–quartile 3 [q3]: 10.8–74.4 months), and were forced to return to dialysis or undergo repeat transplantation. Among these patients who experienced death-censored allograft failure, 196 (48%) were wait-listed, with a median time of 7.7 months (q1–q3: 2.3–22.2 months), and most of these patients (61%) were sensitized (peak panel reactive antibody >30%). Thirty-six percent of these patients received a second transplant (121 received a deceased donor transplant after a median time of 25.4 months [q1–q3: 8.6–54.0 months] from first allograft failure, whereas 27 received a living donor transplant after a median time of 3.5 months [q1–q3: 0–17.9 months] from first allograft failure).
Discussion
This study was designed to inform the expanded utilization of ECD kidneys in the United States. The international comparison with ESP patients demonstrated that the wider use of ECD kidneys in a restricted patient population aged ≥65 years fell short of providing patients with a lifetime of allograft function by an average of 5.2 months in the ESP. In contrast, the more restricted use of ECDs but unrestricted allocation in the United States provided elderly recipients aged ≥65 years with a lifetime of allograft function but also allocated 46% of these kidneys to recipients aged <60 years and produced far fewer transplants overall. Together these findings suggest wider acceptance but restricted allocation of ECD kidneys in the United States could safely increase transplantation in the elderly while avoiding poor outcomes in younger patients. The international comparison also found the survival of elderly transplant recipients was far lower in the United States. This could be related to differences in patient health status between European and US transplant recipients aged ≥65 years or because of increased transplant-related complications in the United States that are not manifest in the ESP because of differences in clinical transplant practice. Although determination of reasons for these international differences in outcomes was not our primary focus, these findings suggest that international collaboration may be very useful in understanding the key determinants of adverse outcomes after ECD transplantation in elderly patients. Specific issues that could be examined include differences in candidate selection, wait-list management, organ preservation, organ allocation, and early and late post-transplant management that might effect patient survival. For example, it is notable that ESP allocates kidneys from donors aged ≥65 years to recipients aged ≥65 years locally or in a narrow geographic area to minimize cold ischemic time,6 and this resulted in shorter cold times in the ESP in our analysis. The new US allocation policy to forego local prioritization of ECD kidneys3 may paradoxically increase cold ischemic times leading to an increase in adverse outcomes and more resistance to accept ECD offers.
Our estimates of the average difference between patient and allograft survival in ESP and United States recipients provide insight into the extent to which ECD transplantation succeeds in providing elderly patients with a lifetime of allograft function. Our estimates provide another metric confirming the clinical utility of ECD kidneys in elderly patients and support expanded use of ECD in elderly patients among United States recipients ≥60 years of age. Although there is limited literature regarding what transplant outcomes would be acceptable to elderly transplant candidates, in our experience, most elderly patients hope to enjoy transplant function for the remainder of their lives. The approach of calculating the difference in patient versus death-censored allograft failure used in this analysis may therefore be useful in counseling elderly patients regarding the anticipated outcomes after ECD transplantation, especially if coupled with information from previous work from Merion and colleagues demonstrating the benefit of ECD compared with continued wait-listing for a standard criteria donor (SCD) kidney.4 To inform decision making regarding acceptance of an ECD kidney in individual patients, we also determine the probability of survival with a functioning transplant (Table 6). This analysis showed that the probability of being alive with a functioning transplant was higher with rapid ECD transplantation in the first year after wait-listing compared with delayed non-ECD transplantation performed ≥3 years after wait-listing. This information confirms that the benefit of ECD in the elderly is dependent on the timing of transplantation.9 Importantly, these findings may alter decision making in elderly patients previously advised to accept transplantation from an ECD. Under the new kidney allocation system, patients will receive credit for time on dialysis before wait-listing and may now rapidly become eligible for an SCD transplant.3 Our findings suggest that it may not be beneficial for such patients to accept transplantation from an ECD if there is a significant possibility for rapid SCD transplantation.
The second part of our study focused on determining the potential downside of continuing to allow any wait-list candidates to receive an ECD kidney in the new US kidney allocation policy. These analyses demonstrated that transplantation of ECD kidneys provides patients <50 years with an insufficient duration of transplant function and that these patients will have to either return to dialysis or undergo repeat transplantation. We found that few of these patients are relisted for transplantation, are frequently sensitized, and are unlikely to receive a repeat transplant. In contrast, among recipients aged 50–59 years, ECD transplants provided nearly equal patient and death-censored allograft survival, whereas in patients ≥60 years death-censored allograft survival exceeded patient survival, indicating that on average ECD kidneys provide patients aged ≥60 years with a lifetime of allograft function. To what extent the survival of patients aged ≥60 years was shortened by transplantation with an ECD kidney is difficult to determine in this observational study. It is however reassuring that we only found a 7–8 month difference in the average 10-year patient survival between recipients of a transplant from a donor aged ≥65 years compared with similar-aged recipients of a deceased donor kidney transplant from a donor aged <65 years with KDPI 60%–69%.
Although some young patients who tolerate dialysis poorly may derive a survival benefit if transplanted rapidly with an ECD kidney,4 our findings challenge the appropriateness of continuing to allow any consenting patient to accept an ECD kidney. In earlier iterations of the kidney allocation policy, a broad (15-year) age matching had been proposed for kidneys with a KDPI>20%.10 However, this proposal was rejected by the US Department of Justice on the grounds that the use of age alone to determine organ allocation was discriminatory.11 Therefore, the default policy to allow any consenting patient to receive an ECD transplant was maintained. Our findings suggest that the European approach to allow elderly patients to opt out of ESP, while prohibiting the transplantation of ECD kidneys in younger candidates who clearly will not derive a survival benefit might have been an important consideration. Although younger patients might derive quality of life benefits from ECD transplantation, it is unknown if this quality of life benefit is durable especially if the transplant fails. Excluding younger patients from ECD would help ensure rapid transplantation that is essential to maximize the benefit of ECD in the elderly (Table 6) and may be justified given that changes to the kidney allocation system are projected to decrease transplantation in candidates aged ≥50 years.3
Readers of our study should consider the inherent limitation of observational studies and that our findings may not be directly applicable to individual patients. In summary, these analyses (1) provide evidence to encourage increased utilization of deceased donors aged ≥65 years in elderly transplant candidates, (2) highlight the need for international collaboration to devise strategies to minimize the risk of death after ECD transplantation, (3) suggest the need to carefully evaluate the effect of regional sharing on cold ischemic time in ECD transplants, (4) demonstrate that the benefits of ECD transplantation in the elderly are dependent on rapid transplantation, and (5) challenge the US policy to allow any consenting patient to proceed with ECD transplantation. These observations may be useful in increasing the safe utilization of ECD kidneys in the United States.
Concise Methods
This study was conducted with the approval of our local hospital research ethics board. The study adheres to the Declaration of Istanbul.
Use and Outcomes of ECD Transplantation in the ESP and the United States
Data Sources and Study Population
The study population included recipients of a first, kidney-only transplant from a deceased donor ≥65 years of age captured in the ESP or the US Renal Data System (USRDS). A significant issue limiting international comparisons of transplant outcomes is the lack of validated outcome assessment.12 To overcome this limitation, we restricted the analysis to patients transplanted between January 1, 1999, and December 31, 2003, with follow-up through April 30, 2005, because of the availability of rigorous outcome assessment in this cohort of ESP patients as part of a clinical study.6 Transplant outcomes in the USRDS cohort are routinely validated as previously described.13
Statistical Analyses
Donor and recipient characteristics were described using the median (and quartiles) for continuous variables, or frequencies and proportions for categorical variables; group differences were compared using the Kruskal–Wallis or chi-squared test as appropriate.
Among the subset of recipients aged ≥65 years, we determined the time to allograft failure from any cause, death-censored allograft failure, and death with a functioning graft in ESP and United States patients using the Kaplan–Meier product limit method and compared group differences using the log-rank test. Separate Cox multivariate proportional hazards regression models were used to determine the relative risk of graft loss from any cause, death-censored graft failure, and death with a functioning allograft in ESP patients compared with United States patients after adjustment for differences in donor characteristics (age, sex, history of diabetes, history of hypertension, cause of death [cerebrovascular accident versus other]); recipient characteristics (age, sex, cause of ESRD, body mass index, duration of pretransplant dialysis exposure, peak panel reactive antibody titer); and transplant characteristics (cold ischemic time, use of induction therapy [depleting antibody, nondepleting antibody, none], type of initial maintenance immunosuppressant medication [calcineurin inhibitor (tacrolimus, cyclosporine, neither [including sirolimus]) and use of mycophenolate mofetil or azathioprine]). The proportional hazards assumptions were tested using log-negative-log plots of the within-group survivorship probabilities versus log-time in all models.
Determination of Lifetime of Allograft Function
The area under a population survival curve represents the average (or mean) survival of the population.14 When two populations are compared, the surface area between their corresponding survival curves is the average duration of life gained in the population with the greatest survival relative to the population with the least survival.14 Similar to Meier-Kriesche et al,15,16 we extended this approach to quantify the difference in mean patient survival after transplantation and mean death-censored allograft function. When the area under the population death-censored allograft function curve was greater than or equal to the area under the population patient survival curve, the allograft would have provided a lifetime of allograft function to the recipient. Similarly when the area under the population death-censored allograft survival curve was less than that under the patient survival curve, the allograft would have provided less than a lifetime of allograft function. We quantified the difference between the allograft and patient survival curves at 5 years after transplantation to quantify the mean time the recipient population would require another source of renal replacement therapy (either dialysis or repeat transplantation). The areas under patient and death-censored allograft survival curves (mean survival times) were calculated using the Kaplan–Meier product limit method. The area below the survival curves was provided by SAS 9.4 (Carey, NC), using Irwin’s restricted mean, truncated at 5 years.17 Additional covariates were not included in the models because patient and death-censored allograft survival curves were directly compared within the same populations. In the models, the intercluster correlation between deceased donor kidneys from the same donor (maximum 2 kidneys per donor) was accounted for using the robust sandwich covariance matrix.18,19
Probability of Being Alive and Being Alive with a Functioning Allograft 5 Years after Wait-Listing
We determined the probability of being alive 5 years after wait-listing with a transplant from an ECD or SCD after different durations of waiting time (1, 2, 3, and 4 years) among subgroups of elderly patients (aged 60–64, 65–69, ≥70 years) with and without diabetes as the product of the probability of (surviving and remaining active on the wait-list) × (probability of survival after transplantation irrespective of allograft failure). Similarly, the product of being alive with a functioning allograft was determined as the product of (surviving and remaining active on the wait-list) × (probability of survival without a return to dialysis or repeat transplantation). For these analyses, probabilities were determined using the Kaplan–Meier product limit method, and a non-ECD transplant was defined by a KDPI between 0.20 and 0.85, whereas an ECD kidney was defined by a KDPI≥0.86.
Consequences of Continuing To Allow Any Consenting Wait-List Candidate To Undergo Transplantation with an ECD Kidney in the United States
These analyses included recipients of a first, kidney-only transplant from a deceased donor aged ≥65 years (ECD) captured in the USRDS between January 1, 1995, and December 31, 2010, with follow-up through October 31, 2011.
We first determined the distribution of ECD transplants as a function of recipient age with recipient age categorized as follows: 18–39, 40–49, 50–59, 60–64, 65–69, and ≥70 years.
In each of the recipient age groups, we calculated the mean patient and death-censored allograft survival (i.e., areas under the unadjusted Kaplan–Meier survival curves) and the difference in these curves at 10 years after transplantation as previously described.
To determine the extent to which the observed differences between patient and death-censored allograft survival among recipients aged ≥60 years was effected by increased death after transplantation with an ECD kidney, we compared the patient survival of recipients aged ≥60 years transplanted with a deceased donor kidney from a deceased donor aged ≥65 years with that of similar-aged recipients who received a kidney from a deceased donor aged <65 years with a KDPI 60%–69%, 70%–79%, 80%–85%, and >85% during the same time period using separate Cox multivariate regression analyses adjusted for recipient factors (sex, race, cause of ESRD, peak panel reactive antibody titer, body mass index, primary insurer, comorbidities [inability to ambulate, chronic obstructive pulmonary disease, congestive heart failure, cerebrovascular disease, peripheral vascular disease, cancer, ischemic heart disease]), transplant factors (HLA mismatch, cold ischemic time), and delayed graft function. To calculate the area below Cox survival curves, we used the trapezoidal rule20: trapezoid heights were estimated using survival probabilities estimated from the Cox model, and trapezoid widths (intervals) were calculated between survival failure times. The analyses were truncated at 120 months (10 years). We quantified the area between the recipient and deceased donor kidney survival curves as either the expected time a recipient would be required to return to dialysis after deceased donor kidney failure (i.e., when recipient survival is longer than deceased donor kidney survival) or the expected loss of potential kidney function after the recipient died (i.e., when deceased donor kidney survival is longer than recipient survival). The area between the curves was calculated as the difference in the areas under the two survival curves. The 95% confidence intervals for the difference in areas were estimated by comparing the 95% confidence bands at event times.
Finally, among patients aged <50 years who received an ECD kidney from a donor aged ≥65 years and suffered death-censored allograft failure, we determined the proportion that were relisted for transplantation, their peak panel reactive antibody titer at the time of repeat wait-listing, and the proportion subsequently retransplanted with a either a deceased or living donor.
Disclosures
None.
Acknowledgments
Jana Noeldeke helped with collection and provision of data from the ESP. The authors also wish to acknowledge the assistance of Axel Rahmel, Medical Director for Deutsche Stiftung Organtransplantation.
Caren Rose is supported by the Kidney Research Scientist Core Education and National
Training Program.
The data reported here have been supplied by the USRDS. The interpretation and reporting of these data are the responsibility of the authors and in no way should be seen as an official government policy or interpretation of the US Government.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
References
- 1.United Network of Organ Sharing : Data acquisition report, 2014. Available at: http://www.unos.org. Accessed December 26, 2014 [Google Scholar]
- 2.Department of Health and Human Services : HRSA: OPTN/SRTR 2012 annual data report. Rockville, MD, Healthcare Systems Bureau, Division of Transplantation, 2014 [Google Scholar]
- 3.Israni AK, Salkowski N, Gustafson S, Snyder JJ, Friedewald JJ, Formica RN, Wang X, Shteyn E, Cherikh W, Stewart D, Samana CJ, Chung A, Hart A, Kasiske BL: New national allocation policy for deceased donor kidneys in the United States and possible effect on patient outcomes. J Am Soc Nephrol 25: 1842–1848, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Merion RM, Ashby VB, Wolfe RA, Distant DA, Hulbert-Shearon TE, Metzger RA, Ojo AO, Port FK: Deceased-donor characteristics and the survival benefit of kidney transplantation. JAMA 294: 2726–2733, 2005 [DOI] [PubMed] [Google Scholar]
- 5.Grams ME, Womer KL, Ugarte RM, Desai NM, Montgomery RA, Segev DL: Listing for expanded criteria donor kidneys in older adults and those with predicted benefit. Am J Transplant 10: 802–809, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Frei U, Noeldeke J, Machold-Fabrizii V, Arbogast H, Margreiter R, Fricke L, Voiculescu A, Kliem V, Ebel H, Albert U, Lopau K, Schnuelle P, Nonnast-Daniel B, Pietruck F, Offermann R, Persijn G, Bernasconi C: Prospective age-matching in elderly kidney transplant recipients--a 5-year analysis of the Eurotransplant Senior Program. Am J Transplant 8: 50–57, 2008 [DOI] [PubMed] [Google Scholar]
- 7.Cecka JM, Cohen B, Rosendale J, Smith M: Could more effective use of kidneys recovered from older deceased donors result in more kidney transplants for older patients? Transplantation 81: 966–970, 2006 [DOI] [PubMed] [Google Scholar]
- 8.Friedewald JJ, Samana CJ, Kasiske BL, Israni AK, Stewart D, Cherikh W, Formica RN: The kidney allocation system. Surg Clin North Am 93: 1395–1406, 2013 [DOI] [PubMed] [Google Scholar]
- 9.Schold JD, Meier-Kriesche HU: Which renal transplant candidates should accept marginal kidneys in exchange for a shorter waiting time on dialysis? Clin J Am Soc Nephrol 1: 532–538, 2006 [DOI] [PubMed] [Google Scholar]
- 10.Reese PP, Caplan AL: Better off living--the ethics of the new UNOS proposal for allocating kidneys for transplantation. Clin J Am Soc Nephrol 6: 2310–2312, 2011 [DOI] [PubMed] [Google Scholar]
- 11.Graham J: Kidney sharing system may change to better accommodate older patients. Kaiser Health News in Collaboration with The Washington Post, 2013. Available at: http://blog.aarp.org/2013/01/29/kidney-sharing-system-may-change-to-better-accomodate-older-patients/. Accessed June 1, 2014 [Google Scholar]
- 12.Schaefer HM, Kaplan B, Helderman JH: Mortality after kidney transplantation: What lessons can we learn from regional and country variation? Am J Transplant 6: 3–4, 2006 [DOI] [PubMed] [Google Scholar]
- 13.USRDS : Annual data report: Atlas of end stage renal disease in the United States National Institute of Health, National Institute of Diabetes and Digestive and Kidney Disease, Bethesda, MD, 2013 [Google Scholar]
- 14.Lubsen J, Hoes A, Grobbee D: Implications of trial results: the potentially misleading notions of number needed to treat and average duration of life gained. Lancet 356: 1757–1759, 2000 [DOI] [PubMed] [Google Scholar]
- 15.Meier-Kriesche HU, Schold JD, Gaston RS, Wadstrom J, Kaplan B: Kidneys from deceased donors: Maximizing the value of a scarce resource. Am J Transplant 5: 1725–1730, 2005 [DOI] [PubMed] [Google Scholar]
- 16.Meier-Kriesche HU, Scornik JC, Susskind B, Rehman S, Schold JD: A lifetime versus a graft life approach redefines the importance of HLA matching in kidney transplant patients. Transplantation 88: 23–29, 2009 [DOI] [PubMed] [Google Scholar]
- 17.Barker C: The mean, median, and confidence intervals of the Kaplan-Meier survival estimate: Computations and applications. Am Stat 63: 78–80, 2009 [Google Scholar]
- 18.Lee E, Wei L, Amato D: Cox-Type Regression Analysis for Large Numbers of Small Groups of Correlated Failure Time Observations. In: Survival Analysis, State of the Art, edited by Klein JP, Goel PK. Dordrecht, Netherlands, Kluwer Academic Publishers, 1992, pp 237–247 [Google Scholar]
- 19.Lin DY: Cox regression analysis of multivariate failure time data: The marginal approach. Stat Med 13: 2233–2247, 1994 [DOI] [PubMed] [Google Scholar]
- 20.Forbes N, Schachter AD, Yasin A, Sharma AP, Filler G: Limited sampling strategies for sirolimus after pediatric renal transplantation. Pediatr Transplant 13: 1020–1026, 2009 [DOI] [PubMed] [Google Scholar]