Abstract
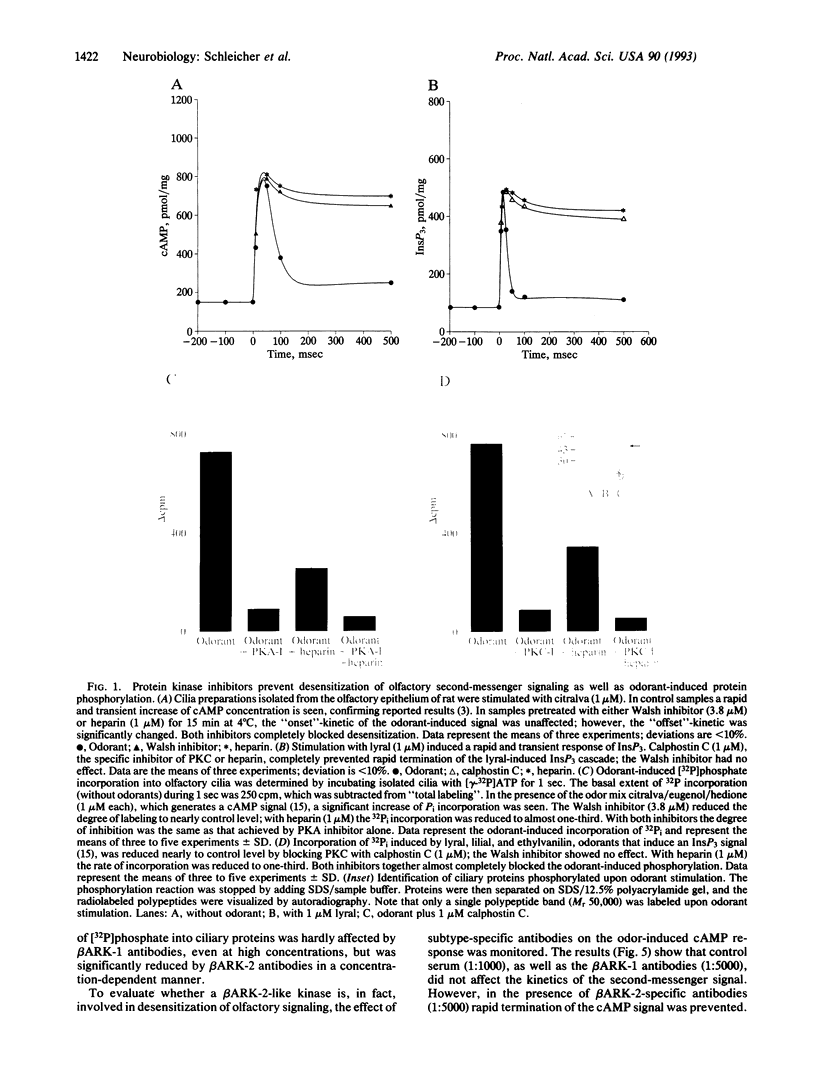
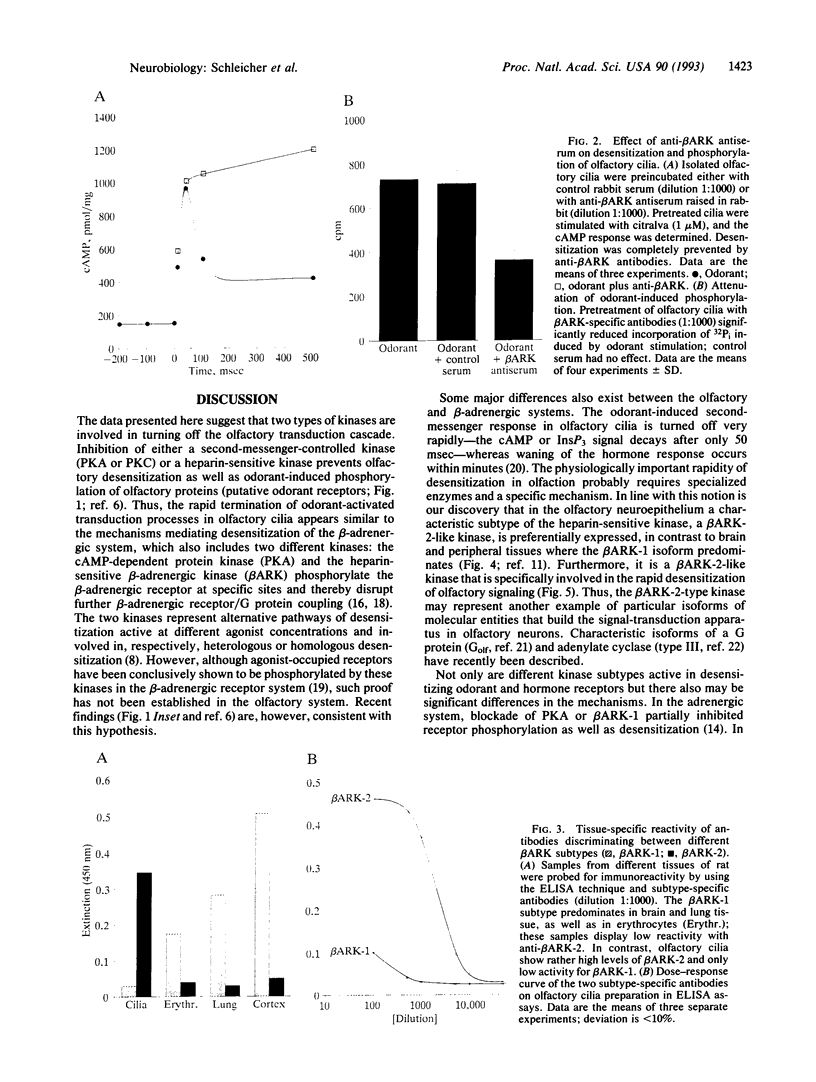
We have previously shown that second-messenger-dependent kinases (cAMP-dependent kinase, protein kinase C) in the olfactory system are essential in terminating second-messenger signaling in response to odorants. We now document that subtype 2 of the beta-adrenergic receptor kinase (beta ARK) is also involved in this process. By using subtype-specific antibodies to beta ARK-1 and beta ARK-2, we show that beta ARK-2 is preferentially expressed in the olfactory epithelium in contrast to findings in most other tissues. Heparin, an inhibitor of beta ARK, as well as anti-beta ARK-2 antibodies, (i) completely prevents the rapid decline of second-messenger signals (desensitization) that follows odorant stimulation and (ii) strongly inhibits odorant-induced phosphorylation of olfactory ciliary proteins. In contrast, beta ARK-1 antibodies are without effect. Inhibitors of protein kinase A and protein kinase C also block odorant-induced desensitization and phosphorylation. These data suggest that a sequential interplay of second-messenger-dependent and receptor-specific kinases is functionally involved in olfactory desensitization.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bakalyar H. A., Reed R. R. Identification of a specialized adenylyl cyclase that may mediate odorant detection. Science. 1990 Dec 7;250(4986):1403–1406. doi: 10.1126/science.2255909. [DOI] [PubMed] [Google Scholar]
- Benovic J. L., DeBlasi A., Stone W. C., Caron M. G., Lefkowitz R. J. Beta-adrenergic receptor kinase: primary structure delineates a multigene family. Science. 1989 Oct 13;246(4927):235–240. doi: 10.1126/science.2552582. [DOI] [PubMed] [Google Scholar]
- Benovic J. L., Onorato J. J., Arriza J. L., Stone W. C., Lohse M., Jenkins N. A., Gilbert D. J., Copeland N. G., Caron M. G., Lefkowitz R. J. Cloning, expression, and chromosomal localization of beta-adrenergic receptor kinase 2. A new member of the receptor kinase family. J Biol Chem. 1991 Aug 15;266(23):14939–14946. [PubMed] [Google Scholar]
- Boekhoff I., Breer H. Termination of second messenger signaling in olfaction. Proc Natl Acad Sci U S A. 1992 Jan 15;89(2):471–474. doi: 10.1073/pnas.89.2.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boekhoff I., Schleicher S., Strotmann J., Breer H. Odor-induced phosphorylation of olfactory cilia proteins. Proc Natl Acad Sci U S A. 1992 Dec 15;89(24):11983–11987. doi: 10.1073/pnas.89.24.11983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boekhoff I., Tareilus E., Strotmann J., Breer H. Rapid activation of alternative second messenger pathways in olfactory cilia from rats by different odorants. EMBO J. 1990 Aug;9(8):2453–2458. doi: 10.1002/j.1460-2075.1990.tb07422.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bownds D., Dawes J., Miller J., Stahlman M. Phosphorylation of frog photoreceptor membranes induced by light. Nat New Biol. 1972 May 24;237(73):125–127. doi: 10.1038/newbio237125a0. [DOI] [PubMed] [Google Scholar]
- Breer H., Boekhoff I., Tareilus E. Rapid kinetics of second messenger formation in olfactory transduction. Nature. 1990 May 3;345(6270):65–68. doi: 10.1038/345065a0. [DOI] [PubMed] [Google Scholar]
- Chen Z., Pace U., Heldman J., Shapira A., Lancet D. Isolated frog olfactory cilia: a preparation of dendritic membranes from chemosensory neurons. J Neurosci. 1986 Aug;6(8):2146–2154. doi: 10.1523/JNEUROSCI.06-08-02146.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen P. The role of protein phosphorylation in the hormonal control of enzyme activity. Eur J Biochem. 1985 Sep 16;151(3):439–448. doi: 10.1111/j.1432-1033.1985.tb09121.x. [DOI] [PubMed] [Google Scholar]
- Dohlman H. G., Thorner J., Caron M. G., Lefkowitz R. J. Model systems for the study of seven-transmembrane-segment receptors. Annu Rev Biochem. 1991;60:653–688. doi: 10.1146/annurev.bi.60.070191.003253. [DOI] [PubMed] [Google Scholar]
- Getchell T. V., Shepherd G. M. Adaptive properties of olfactory receptors analysed with odour pulses of varying durations. J Physiol. 1978 Sep;282:541–560. doi: 10.1113/jphysiol.1978.sp012480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hausdorff W. P., Bouvier M., O'Dowd B. F., Irons G. P., Caron M. G., Lefkowitz R. J. Phosphorylation sites on two domains of the beta 2-adrenergic receptor are involved in distinct pathways of receptor desensitization. J Biol Chem. 1989 Jul 25;264(21):12657–12665. [PubMed] [Google Scholar]
- Hausdorff W. P., Caron M. G., Lefkowitz R. J. Turning off the signal: desensitization of beta-adrenergic receptor function. FASEB J. 1990 Aug;4(11):2881–2889. [PubMed] [Google Scholar]
- Jones D. T., Reed R. R. Molecular cloning of five GTP-binding protein cDNA species from rat olfactory neuroepithelium. J Biol Chem. 1987 Oct 15;262(29):14241–14249. [PubMed] [Google Scholar]
- Kühn H., Dreyer W. J. Light dependent phosphorylation of rhodopsin by ATP. FEBS Lett. 1972 Jan 15;20(1):1–6. doi: 10.1016/0014-5793(72)80002-4. [DOI] [PubMed] [Google Scholar]
- Lefkowitz R. J., Hausdorff W. P., Caron M. G. Role of phosphorylation in desensitization of the beta-adrenoceptor. Trends Pharmacol Sci. 1990 May;11(5):190–194. doi: 10.1016/0165-6147(90)90113-m. [DOI] [PubMed] [Google Scholar]
- Lohse M. J., Benovic J. L., Caron M. G., Lefkowitz R. J. Multiple pathways of rapid beta 2-adrenergic receptor desensitization. Delineation with specific inhibitors. J Biol Chem. 1990 Feb 25;265(6):3202–3211. [PubMed] [Google Scholar]
- OTTOSON D. Analysis of the electrical activity of the olfactory epithelium. Acta Physiol Scand Suppl. 1955;35(122):1–83. [PubMed] [Google Scholar]
- Shear M., Insel P. A., Melmon K. L., Coffino P. Agonist-specific refractoriness induced by isoproterenol. Studies with mutant cells. J Biol Chem. 1976 Dec 10;251(23):7572–7576. [PubMed] [Google Scholar]
- Sibley D. R., Benovic J. L., Caron M. G., Lefkowitz R. J. Regulation of transmembrane signaling by receptor phosphorylation. Cell. 1987 Mar 27;48(6):913–922. doi: 10.1016/0092-8674(87)90700-8. [DOI] [PubMed] [Google Scholar]
- Stryer L. Cyclic GMP cascade of vision. Annu Rev Neurosci. 1986;9:87–119. doi: 10.1146/annurev.ne.09.030186.000511. [DOI] [PubMed] [Google Scholar]
- Wilden U., Kühn H. Light-dependent phosphorylation of rhodopsin: number of phosphorylation sites. Biochemistry. 1982 Jun 8;21(12):3014–3022. doi: 10.1021/bi00541a032. [DOI] [PubMed] [Google Scholar]