Abstract
Background:
Denture hygiene is an important factor in the prevention and treatment of denture stomatitis (DS). This study aimed to evaluate the efficacy of two different mouthwashes (chlorhexidine digluconate 0.12% and hexetidine 0.1%) in eliminating Candida albicans on dentures.
Materials and Methods:
A total of 60 denture wearers (20 men, 40 women; age range 40-80 years) with clinical evidence of DS were randomly divided into 2 test groups and 1 control group. The dentures of each test group were treated by immersion in one of the two mouthwashes while those of the control group were immersed in distilled water. Swab samples from the palatal surfaces of the upper dentures were collected before and after of cleaner use and examined mycologically.
Results:
Reduction in the number of colony-forming units of Candida albicans after immersion of the dentures with chlorhexidine digluconate 0.12% was significantly greater than that of the group using hexetidine 0.1% and those of the control group.
Conclusion:
Hexetidine 0.1% solution tested for the first time as a product of disinfection of the acrylic dentures showed average results after immersion of 8 night hours for 4 days and was less effective than chlorhexidine digluconate 0.12%.
Keywords: Candida albicans, chlorhexidine digluconate, denture stomatitis, hexetidine
Introduction
Denture stomatitis (DS) is a common condition characterized by a generalized inflammation of the palatal mucosa covered by the denture.1 It was reported to affect 15-70% of denture wearers.2 Clinically, DS can vary from petechiae to generalized inflammation with papillary hyperplasia.3
It is a harmless form of oral candidiasis associated with a quantitative increase of yeasts on the mucosa and the denture’s fit surface.4 Usually, a mixture of Candida species such as C. albicans, C. tropicalis, C. krusei, C. guilliermondii, C. glabrata can be isolated from oral candidal lesions.5 In particular, it is known that complete and removable dentures accumulate C. albicans on the porous surface of the acrylic resin.6
Denture hygiene is an important factor in the prevention and treatment of DS. For that, many modalities of oral care and denture-cleansing techniques have been suggested such as mechanical brushing, microwave sterilization and the use of chemical cleansing like soap, effervescent tablets, and mouthwashes.7,8
Chlorhexidine digluconate is an antiseptic and disinfectant agent that is active against various bacteria, viruses, and fungi including C. albicans.9 Its clinical and microbiological efficacy in treating DS has been proven.7,10-12 However, the brown coloration of the acrylic denture soaked in chlorhexidine can be a major obstacle in using it as a daily decontamination product.11
Therefore, other soaking solutions have been suggested such as sodium hypochlorite13 and hexetidine. Jones et al. (1997) described the positive effect of hexetidine in decreasing the adherence of the C. albicans in the epithelial cells of the oral mucosa.14 This adherence is a key factor in the filamentation and therefore in the transition to the virulence of candida generally saprophytes. In addition, hexetidine 0.1% is available at affordable prices in the local market. This will enable the patients, in case this solution was deemed effective, to use it as part of a daily prosthetic hygiene.
However, studies of the hexetidine effect on C. albicans are rare and to our knowledge, its in vivo effect on C. albicans of the acrylic dentures has not been investigated yet.
The aim of this study was to test the efficacy of hexetidine mouth rinse in reducing or eliminating C. albicans associated with DS and to compare its effect to chlorhexidine digluconate.
Materials and Methods
This randomized controlled trial was conducted in accordance with the Helsinki agreement for research on humans, and the study design was approved through the Institutional Review Board and Independent Ethics Committee of the School of Dentistry, Lebanese University, Beirut, Lebanon. Written informed consent was obtained from all participants in the study prior to treatment.
Complete maxillary edentulous denture-wearing patients attending the Department of Oral Pathology and Diagnosis at the Lebanese University during a period of 2 years were examined for clinical evidence of Newton’s type II DS. Newton’s type II is a diffuse erythema involving part or all of the mucosa, which is covered by the denture.15
We included in the study, patients who were: (1) Confirmed having C. albicans in their dentures, (2) aged between 40 and 80 years, (3) healthy, (4) not taking any medication that might affect the oral bacterial flora, and (5) wearing the maxillary full prosthesis for more than 1-year.
Patients with systemic diseases such as diabetes, nutritional deficiencies and those wearing their dentures for less than 1-year were excluded.
Sixty patients met the inclusion criteria. They were randomly assigned to one of 3 groups (2 tests and 1 control), of 20 patients each and were asked to avoid cleaning their dentures during the experimental procedure to standardize the study.
A quantitative microbiological measurement was performed the 1st day (D1) from the infected oral mucosa and the fitting side of the dentures.
Patients in the first group had their dentures soaked in a solution of chlorhexidine digluconate 0.12% (Paroex; Sunstar: Butler, USA) during the night for 8 h (from 10 pm to 6 am) for 4 consecutive nights, the ones of the second group in hexetidine 0.1% (Hextril; Godeck Manufacturer for Parke Davis, France) and the third (control) group in distilled water following the same protocol.
A second swab collection destined for a new C. albicans colony count was taken on the day 4 (D4).
One investigator carried out microbiological procedures. The Becton Dickinson (New Jersey, USA) Microbiology System, BBL Culture Swab was used. These systems are sterile devices for collecting and transporting microbiological specimens (Amies, Stuart and Agar Gel).
Swabs were cultured in Sabouraud’s dextrose agar (40 g/l dextrose, 10 g/l peptone and 20 g/l agar) and containing chloramphenicol 0.5 g/l and actidione 0.5 g/l. Candida count was carried out after 48 h incubation at 37°C in aerobic conditions. C. albicans was differentiated from the other species by their production of filaments in 0.5 ml of animal serum.
The primary outcome measure was the relative reduction in C. albicans colony count expressed in colony-forming units (CFU)/ml collected from the denture surface at day 1 (D1) and after the 4 nights of immersion at day 4 (D4). The relative reduction = (a−b)/a, “a” being the number of colonies before immersion and “b” the number of colonies after.
The Kolmogorov–Smirnov test was used to assess the normality of distribution and the data was found to follow a non-normal distribution. Accordingly, the non-parametric Kruskall–Wallis test was applied to test the working hypothesis of difference in the relative reduction of C. albicans between the three groups. If a significant difference was found, a pairwise multiple comparison procedure (Mann–Whitney) was performed to analyze the data.
A confidence level of 0.05 was considered statistically significant. Data were analyzed using the Statistical Package for Social Sciences (IBM, USA), Version 21.0.
Results
Characteristics of the patients in the three groups are summarized in Table 1. When compared at baseline, there was no statistically significant difference in the mean age (P = 0.124), C. albicans count on the palate (P = 0.516) and on the dentures (P = 0.484).
Table 1.
Descriptive statistics of the three study groups.
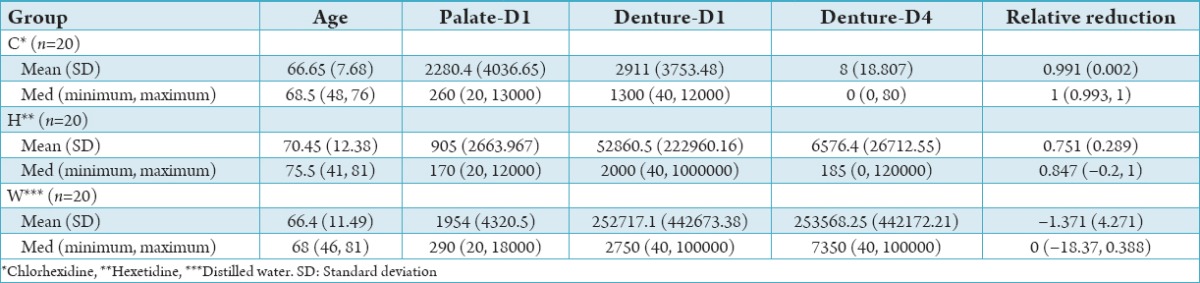
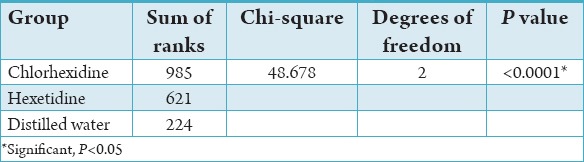
When comparing the relative reduction of C. albicans on the dentures (CFU/ml) between the three groups, a statistically significant difference was found (χ2 = 48.678, P < 0.0001). The chlorhexidine group showed the greatest relative reduction with a sum of ranks of 985, followed by hexetidine (sum of ranks = 621) and distilled water (sum of ranks = 224) (Table 2).
Table 2.
Comparison of the relative reduction in Candida albicans between the three groups.
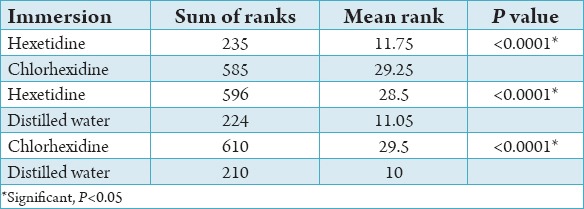
Using the Wilcoxon-Mann–Whitney multiple comparison test, the sum of ranks were compared in pairs and results showed that chlorhexidine was significantly more effective than hexetidine (P < 0.0001), which in turn was more effective than distilled water (P < 0.0001) (Table 3).
Table 3.
Pairwise comparison between the three groups.
Discussion
Among the most important fungal pathogens are yeast species belonging to the genus Candida, which encompasses highly virulent pathogens and responsible for the majority of fungal infections. DS is a common infection associated with C. albicans. A study of Budtz-JØrgensen et al. (1996) detected DS in 72% of denture wearers in an elderly population living in a geriatric institution.16 It has been widely accepted that proper routine cleansing of dentures is required to prevent DS and maintain healthy supporting tissues.3 Kulak-Ozkan et al. (2002) evaluated 70 complete denture wearers clinically and mycologically. They concluded that there exists a statistically significant relationship between DS, presence of yeasts and denture cleanliness.17 It is well accepted that chemical disinfectants have some advantages over mechanical cleaning such as effective disinfection and ease of use.18
This study investigated the effectiveness of two different mouthwashes, chlorhexidine digluconate 0.12% and Hexetidine 0.1%, in eliminating C. albicans on dentures compared to a control solution, distilled water.
Chlorhexidine has been widely considered as the antiseptic of choice for decontaminating the dentures infected by C. albicans. Pavarina et al.10 established a protocol of disinfection in which 10 min of immersion at 4% concentration solution was totally effective in eliminating C. albicans in vivo from complete dentures. However, chlorhexidine 4% solution has shown to negatively affect the hardness and roughness of acrylic resins.19
Pellizzaro et al.3 tested the antimicrobial efficacy of lower concentrations and periods and found that a very short period exposure (90s) to chlorhexidine digluconate 2% was sufficient to completely inactivate C. albicans on an acrylic resin. This effect can be attributed mainly to the chemical mechanism of action of this solution against the fungal cell. MacNeill et al.20 observed that, after the contact with chlorhexidine, C. albicans cells exhibited a severe cytoplasmic degeneration (fragmentation and clumping of the contents, vacuolization, lipid accumulation and condensation) and fragmentation and desquamation of the cell wall, resulting in cell death.
Regardless of the effectiveness, the use of chlorhexidine has been limited by some side effects that could affect the patient compliance such as the brown stains on the acrylic resin.11,12
In our study, over the 20 patients of the first group, dentures soaked in Chlorhexidine digluconate 0.12%, 15 showed a total disappearance of C. albicans (0 CFU/ml) after four nights of immersion (D4), 4 registered a decline in the number of colonies to 20 CFU/ml (result within normal limits) and only one kept an elevated count of 80 CFU/ml. It is worth noting that this last patient had an initial count of 12000 CFU/ml. Therefore, the brown coloration was noted on all first group patients’ prostheses.
For Sixou and Hamel,21 the effect of hexetidine on C. albicans is variable. Jones et al.14 described the positive effect of hexetidine in decreasing the adherence of C. albicans in the epithelial cells of the oral mucosa. A study conducted in vitro by Luc et al.7 shows that hexetidine 0.1% is slightly active on the C. albicans after contact duration of 5 min.
Our choice was, despite the above, fixed on hexetidine for the following reasons:
The studies of the action in vivo of hexetidine on C. albicans are rare. Moreover, we did not find any study in the literature review highlighting its action on the C. albicans of the acrylic dentures.
Brown coloration of the denture resins soaked in chlorhexidine has not been described with hexetidine. This color can be a major obstacle in the use of chlorhexidine as a daily decontamination product.
Our results showed that among the 20 patients of the second group, dentures soaked in hexetidine 0.1%, only 2 registered a total disappearance of C. albicans (0 CFU/ml), 16 had reduced number of colonies for more than 80% (3 for more than 60 %), 1 patient went from more than 1000000 CFU/ml before soaking to 120000 CFU/ml at D4 sampling and 1 patient had a higher number of colonies at D4 compared to D1.
The effect of hexetidine on C. albicans is, therefore, variable but is not null. On the other hand, among the 20 dentures soaked in hexetidine, 4 underwent a very light brownish stain (much less than that caused by chlorhexidine). These four prostheses were all old in age. In our opinion, further studies need to be done in order to confirm this point, which may be due to a factor related to the age of the resin.
The third control group, as expected, the results showed absolute ineffectiveness of the distilled water.
Conclusion
Better antifungal effect of chlorhexidine digluconate 0.12% solution is noted, compared to hexetidine 0.1%, as a disinfection product for infected dentures by C. albicans.
However, hexetidine solution presents moderately good results after soaking dentures for 8 h during 4 consecutive nights. In this respect, it deserves subsequently more research, under various operating protocols to enable an objective study of its fungicide effect and explain the resin coloring problem.
Footnotes
Conflicts of Interest: None
Source of Support: Nil
References
- 1.Budtz-Jörgensen E. The significance of Candida albicans in denture stomatitis. Scand J Dent Res. 1974;82:151–90. doi: 10.1111/j.1600-0722.1974.tb00378.x. [DOI] [PubMed] [Google Scholar]
- 2.Gendreau L, Loewy ZG. Epidemiology and etiology of denture stomatitis. J Prosthodont. 2011;20(4):251–60. doi: 10.1111/j.1532-849X.2011.00698.x. [DOI] [PubMed] [Google Scholar]
- 3.Pellizzaro D, Polyzois G, Machado AL, Giampaolo ET, Sanitá PV, Vergani CE. Effectiveness of mechanical brushing with different denture cleansing agents in reducing in vitro Candida albicans biofilm viability. Braz Dent J. 2012;23(5):547–54. doi: 10.1590/s0103-64402012000500013. [DOI] [PubMed] [Google Scholar]
- 4.Budtz-Jörgensen E, Theilade E. Regional variations in viable bacterial and yeast counts of 1-week-old denture plaque in denture-induced stomatitis. Scand J Dent Res. 1983;91(4):288–95. doi: 10.1111/j.1600-0722.1983.tb00818.x. [DOI] [PubMed] [Google Scholar]
- 5.Dangi YS, Soni MS, Namdeo KP. Oral candidiasis: A review. Int J Pharm Sci. 2012;2:36–41. [Google Scholar]
- 6.Pereira-Cenci T, Del Bel Cury AA, Crielaard W, Ten Cate JM. Development of Candida-associated denture stomatitis: New insights. J Appl Oral Sci. 2008;16(2):86–94. doi: 10.1590/S1678-77572008000200002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Luc J, Roques C, Frayret MN, Michel G, Ducani M, Vandermander J. In vitro bacteriocidal activity of 5 oral antiseptics against the principal microorganisms implicated in oral disease. J Parodontol. 1991;10(4):381–7. [PubMed] [Google Scholar]
- 8.Sesma N, Rocha AL, Laganá DC, Costa B, Morimoto S. Effectiveness of denture cleanser associated with microwave disinfection and brushing of complete dentures: In vivo study. Braz Dent J. 2013;24(4):357–61. doi: 10.1590/0103-6440201302205. [DOI] [PubMed] [Google Scholar]
- 9.Arikan A, Kulak Y, Kadir T. Comparison of different treatment methods for localized and generalized simple denture stomatitis. J Oral Rehabil. 1995;22(5):365–9. doi: 10.1111/j.1365-2842.1995.tb00786.x. [DOI] [PubMed] [Google Scholar]
- 10.Pavarina AC, Pizzolitto AC, Machado AL, Vergani CE, Giampaolo ET. An infection control protocol: Effectiveness of immersion solutions to reduce the microbial growth on dental prostheses. J Oral Rehabil. 2003;30(5):532–6. doi: 10.1046/j.1365-2842.2003.01093.x. [DOI] [PubMed] [Google Scholar]
- 11.Ernst CP, Prockl K, Willershausen B. The effectiveness and side effects of 0.1% and 0.2% chlorhexidine mouthrinses: A clinical study. Quintessence Int. 1998;29(7):443–8. [PubMed] [Google Scholar]
- 12.Budtz-Jörgensen E, Löe H. Chlorhexidine as a denture disinfectant in the treatment of denture stomatitis. Scand J Dent Res. 1972;80:457–64. doi: 10.1111/j.1600-0722.1972.tb00314.x. [DOI] [PubMed] [Google Scholar]
- 13.Felton D, Cooper L, Duqum I, Minsley G, Guckes A, Haug S, et al. Evidence-based guidelines for the care and maintenance of complete dentures: A publication of the American College of Prosthodontists. J Prosthodont. 2011;20(Suppl 1):S1–S12. doi: 10.1111/j.1532-849X.2010.00683.x. [DOI] [PubMed] [Google Scholar]
- 14.Jones DS, McGovern JG, Woolfson AD, Gorman SP. The effects of hexetidine (Oraldene) on the adherence of Candida albicans to human buccal epithelial cells in vitro and ex vivo and on in vitro morphogenesis. Pharm Res. 1997;14(12):1765–71. doi: 10.1023/a:1012140131757. [DOI] [PubMed] [Google Scholar]
- 15.Newton AV. Denture sore mouth. Br Dent J. 1962;112:357–60. [Google Scholar]
- 16.Budtz-Jlrgensen E, Mojon P, Banon-Clément JM, Baehni P. Oral candidosis in long-term hospital care: Comparison of edentulous and dentate subjects. Oral Dis. 1996;2(4):285–90. doi: 10.1111/j.1601-0825.1996.tb00239.x. [DOI] [PubMed] [Google Scholar]
- 17.Kulak-Ozkan Y, Kazazoglu E, Arikan A. Oral hygiene habits, denture cleanliness, presence of yeasts and stomatitis in elderly people. J Oral Rehabil. 2002;29(3):300–4. doi: 10.1046/j.1365-2842.2002.00816.x. [DOI] [PubMed] [Google Scholar]
- 18.Paranhos Hde F, Panzeri H, Lara EH, Candido RC, Ito IY. Capacity of denture plaque/biofilm removal and antimicrobial action of a new denture paste. Braz Dent J. 2000;11(2):97–104. [PubMed] [Google Scholar]
- 19.Pinto Lde R, Acosta EJ, Távora FF, da Silva PM, Porto VC. Effect of repeated cycles of chemical disinfection on the roughness and hardness of hard reline acrylic resins. Gerodontology. 2010;27(2):147–53. doi: 10.1111/j.1741-2358.2009.00282.x. [DOI] [PubMed] [Google Scholar]
- 20.MacNeill S, Rindler E, Walker A, Brown AR, Cobb CM. Effects of tetracycline hydrochloride and chlorhexidine gluconate on Candida albicans. An in vitro study. J Clin Periodontol. 1997;24(10):753–60. doi: 10.1111/j.1600-051x.1997.tb00193.x. [DOI] [PubMed] [Google Scholar]
- 21.Sixou M, Hamel O. Bacteriological criterias to consider during antiseptic prescriptions as mouth wash in odonto-stomatology. J Parodontol Implant Oral. 2002;21(1):25–41. [Google Scholar]


