Abstract
Respiratory infection is a major cause of exacerbation in chronic obstructive pulmonary disease (COPD). Infectious contributions to exacerbations remain incompletely described. We therefore analyzed respiratory tract samples by comprehensive real-time polymerase chain reaction (PCR) in combination with conventional methods. We evaluated multiple risk factors for prolonged hospitalization to manage COPD exacerbations, including infectious agents. Over 19 months, we prospectively studied 46 patients with 50 COPD exacerbations, collecting nasopharyngeal swab and sputum samples from each. We carried out real-time PCR designed to detect six bacterial species and eleven viruses, together with conventional procedures, including sputum culture. Infectious etiologies of COPD exacerbations were identified in 44 of 50 exacerbations (88%). Infections were viral in 17 of 50 exacerbations (34%). COPD exacerbations caused by Gram-negative bacilli, including enteric and nonfermenting organisms, were significantly associated with prolonged hospitalization for COPD exacerbations. Our results support the use of a combination of real-time PCR and conventional methods for determining both infectious etiologies and risk of extended hospitalization.
Keywords: COPD, exacerbation, etiology, real-time PCR, prolonged hospitalization, risk factor
Introduction
Acute exacerbations are associated with prolonged hospitalization and increased mortality rates for patients with chronic obstructive pulmonary disease (COPD).1 Most of COPD exacerbations are caused by respiratory infections, particularly involving rhinoviruses (RVs), influenza viruses, Haemophilus influenzae, and Streptococcus pneumoniae.2 Recent reports demonstrated a causal role for viral infection in 29%–44% of COPD exacerbations.3 Conventional methods including sputum culture identify mainly bacteria, which account for 30%–50% of COPD exacerbations.4,5 Furthermore, conventional methods have limited ability to detect not only respiratory viruses but also atypical bacteria. Even common bacteria can go undetected in patients who recently have taken antibiotics. Real-time polymerase chain reaction (PCR) can efficiently detect respiratory viruses as well as typical and atypical bacteria.6,7 However, few studies have evaluated usefulness of real-time PCR in identifying the respiratory virus, atypical bacteria, and polymicrobial infections that can underlie COPD exacerbation.8–10 More extensive examination of infectious etiologies of COPD exacerbations by real-time PCR is of great practical importance.
Advanced age is among the most important risk factors for COPD progression. COPD symptoms tend to worsen during respiratory infection, which tends to prolong COPD-related hospitalization. Extended hospitalization for COPD exacerbation tends to reflect physical deterioration and socioeconomic factors.11 Pseudomonas aeruginosa and other Gram-negative bacilli infect patients with advanced COPD, often prolonging COPD-related hospitalizations. However, a relationship between infection and COPD-related hospitalization has not been clearly demonstrated.
Inflammatory indices may relate to clinical severity of COPD exacerbations.12 COPD exacerbations associated with viral infection recently were linked with high serum C-reactive protein (CRP) concentrations.13 However, various viruses and bacteria might show different inflammatory features during COPD exacerbations, and relationships between specific causative microorganisms and characteristics of inflammation remain incompletely understood.
To identify microbial causes of COPD exacerbations, we carried out real-time PCR with respiratory samples, in combination with conventional methods. We then evaluated risk factors for prolonged hospitalization to manage COPD exacerbations, including types of microorganisms and various biologic markers.
Subjects and methods
Study subjects, definition of COPD exacerbation, and respiratory samples
We prospectively studied patients with COPD diagnosed with COPD exacerbation who were at least 20 years old, presented within 7 days of onset of exacerbation symptoms, and provided written informed consent. Participants had sought treatment in the outpatient clinic or emergency department at The Jikei University Hospital, The Jikei University Daisan Hospital, or Toranomon Hospital between August 2012 and March 2014. COPD exacerbation was defined as any sustained increase beyond patients’ baseline condition in any respiratory symptom that required a change of regular medication or necessitated hospital treatment.14
Our research protocol was approved by the review boards of The Jikei University Hospital, The Jikei University Daisan Hospital, and Toranomon Hospital. After obtaining informed consent from each patient, a nasopharyngeal swab (NPS) and a sputum sample were collected by a physician or nurse.
Data collection
Patient age, sex, smoking history, underlying disease, baseline pulmonary function, laboratory test result, and chest radiographic and computed tomographic findings were ascertained from medical records. We also recorded specific reasons for hospitalization, frequency of COPD exacerbations, mortality, duration of hospitalization, need for intensive care unit admission, and need for ventilatory support.
Real-time PCR to detect viruses and bacteria
The sputum sample was homogenized with an enzymatic sputum lysis reagent (Sputazyme, Kyokuto, Tokyo). NPS samples were suspended in 0.5 mL of pleuropneumonia-like organism broth (Becton, Dickinson and Company, Sparks, MD, USA). NPS and lyzed sputum samples were subjected to DNA and RNA extraction as well as inoculation for bacterial culture. Specifics of DNA/RNA extraction, reverse transcription reaction, and real-time PCR were as described previously.15–17 The Cycleave PCR kit (Takara Bio, Shiga, Japan; catalog number CY216) used for virus in the present study could detect the respiratory syncytial virus subgroups A (RSV A) and B (RSV B); parainfluenza virus types 1 (PIV1), 2 (PIV2), and 3 (PIV3); human metapneumovirus; influenza virus types A (FLU A) and B (FLU B); adenovirus; human bocavirus; and RV.16 Sensitivity of PCR for detection of RSV, PIV, and FLU is reported to be approximately 10 plaque-forming units per well.16
Another Cycleave PCR kit (catalog number CY 214) was used to identify six bacterial pathogens: S. pneumoniae, H. influenzae, Mycoplasma pneumoniae, Chlamydophila pneumoniae, Streptococcus pyogenes, and Legionella pneumophila. Sensitivity of this PCR kit has been described as approximately 10 colony-forming units per well.15
PCR results were considered positive for a pathogen if the pathogen was detected in at least one of the two specimens (NPS and/or sputum). Exceptions were S. pneumoniae, H. influenzae, and S. pyogenes where widespread colonization requires in sputum for PCR diagnosis of infection.
Conventional methods of pathogen identification in COPD exacerbations
Conventional methods used for pathogen identification included sputum culture, serologic tests, urine antigen assays for S. pneumoniae and L. pneumophila, and an FLU antigen assay. Sputum cultures for M. pneumoniae and culture-based species identification were performed by methods described in the Manual of Clinical Microbiology.18 A fourfold increase in M. pneumoniae titres at the convalescent phase compared to the acute phase was considered positive for M. pneumoniae infection. Positive results using urinary antigen assays for S. pneumoniae (Binax, Portland, ME) and L. pneumophila (Binax) were indicative of S. pneumoniae and L. pneumophila infection, respectively. Positive results of an FLU antigen test (Tauns Laboratories, Numazu, Shizuoka, Japan) were accepted as evidence of FLU infection.
Statistical analysis
We used Microsoft Excel 2010 for Statistics (SSRI, Tokyo) and Stat View 5.0 (Tokyo) software programs to perform statistical analysis. Categorical data are presented as frequencies and percentages. As inflammatory biomarkers, we compared white blood cell (WBC) counts and CRP values between five pathogen groups: the S. pneumoniae group, including both pure and mixed S. pneumoniae infection, n=8; the Gram-negative bacilli group, which included enteric and nonfermenting Gram-negative bacilli, n=11; other bacteria, including H. influenzae (3), Staphylococcus aureus (2), and M. pneumonia (2), n=8; mixed viral and bacterial infection, n=6; viruses, n=11; and the “no pathogen detected” group, n=6. Categorical data concerning WBC and CRP, presented as medians with interquartile range, were compared using the Kruskal–Wallis test and then analyzed post hoc by the Bonferroni test. We also analyzed risk factors for extended hospitalizations to treat COPD exacerbations. We divided durations of hospitalization into short-term, less than 14 days, and extended, 14 days or more. Hospitalizations of patients who died were considered extended, and patients not hospitalized were classified as short term. Proportions were compared using the chi-squared test and Fisher’s exact test as appropriate. Age, sex, albumin, and WBC were submitted to univariate analysis. Then, variables associated with a P-value <0.1 were submitted to multivariate analysis. Variables included in the multivariate analysis were age, male sex, albumin, WBC, home oxygen therapy, and enteric or nonfermenting Gram-negative bacilli. Odds ratios were calculated, with 95% intervals and P-values derived from the likelihood ratio.
Results
Baseline characteristics
Between August 2012 and March 2014, we evaluated 50 exacerbations of COPD in 46 patients who met the inclusion criteria. Forty-two patients had a single episode, while four had two episodes during the study period. Characteristics of the 46 patients are summarized in Table 1. Most of the 50 exacerbations occurred in man (43 patients, 86%). Twenty-one patients (45.7%) were receiving home oxygen therapy. Of the 46 patients (60.9%), 28 were diagnosed as stage III or IV of COPD according to the criteria of the Global Initiative for Chronic Obstructive Lung Disease.14
Table 1.
Baseline characteristics of patients (n=46)
| n (%) | |
|---|---|
| Male, n (%) | 43 (93.5) |
| Mean age, years ± SD (range) | 76±8.6 (57–92) |
| Mean FEV1 (L), ± SD (range) | 1.41±0.28 (0.5–2.370) |
| Mean %FEV1 (%), ± SD (range) | 54.7±18.1 (26–92.9) |
| Frequencies of COPD exacerbations n/year (range) | 1.5 (1–3) |
| GOLD Stage, n (%)* | |
| I | 2 (4.3) |
| II | 13 (28.3) |
| III | 7 (15.2) |
| IV | 21 (45.7) |
| Home oxygen therapy, n (%) | 21 (45.7) |
| Current smoker, n (%) | 4 (8) |
| Immunization status | |
| Streptocoocus pneumoniae | 23 (50) |
| Influenza virus | 36 (78.3) |
| Prior antibiotics use | 11 (23.9) |
| Macrolides | 6 (13) |
| Fluoroquinolones | 2 (4.3) |
| Cephalosporins | 3 (6.5) |
| Underline disease,** n (%) | |
| Interstitial pneumonia | 2 (4.3) |
| Diabetes mellitus | 4 (8.7) |
| Ischemic heart disease | 4 (8.7) |
| Arrhythmia | 3 (6.5) |
| Cerebrovascular disease | 2 (5.5) |
| Malignant disease | 4 (9) |
| Psychiatric disease | 2 (4.3) |
Notes:
GOLD Stage used, GOLD 2011.14
Some overlap exists.
Abbreviations: COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 second; SD, standard deviation.
Clinical status of patients at COPD exacerbation
Forty-three patients (86%) were admitted to the hospital. The median hospital stay was 16 days (range 5–60 days). Four patients (8%) required ICU admission; subsequently three patients (6%) required mechanical ventilatory support. Radiologic evaluation detected pneumonia in 10 patients (20%). Detected causative agents of pneumonia were S. pneumoniae (four patients), H. influenzae (one patient), infection with both FLU and S. pneumoniae (one patient), mixed bacterial infection with Klebsiella pneumoniae and H. influenzae (one patient), and mixed infection with P. aeruginosa and S. aureus (one patient). Three patients died of severe respiratory failure, attributed to pneumonia caused by S. aureus, S. pneumoniae, or mixed infection with RSV, S. pneumoniae, and H. influenzae, one patient each.
Monthly distribution of frequencies and causative pathogens of COPD exacerbation
Monthly distributions of frequencies and most common causative pathogens for COPD exacerbation are shown in Figure 1. COPD exacerbations followed a clear seasonal pattern, with the highest monthly rates occurring in winter (December through February), when 24 instances of exacerbation were observed (48%). Mean monthly exacerbation rates during the winter (4.0 exacerbations per month) were 1.5 times the mean rate for a typical year (2.5 exacerbations rates per month). Increases in COPD exacerbations coincided with the epidemic period for FLU and S. pneumonia infection.
Figure 1.
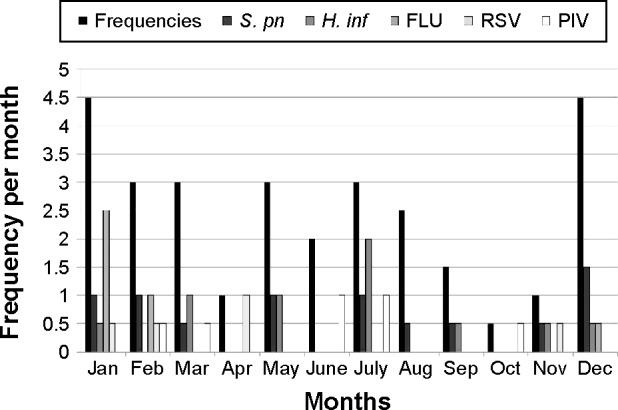
Monthly distribution of frequencies of COPD exacerbations for time of year and for pathogens.
Abbreviations: COPD, chronic obstructive pulmonary disease; S. pn, Streptococcus pneumoniae; H. inf, Haemophilus influenzae; FLU, influenza virus; RSV, respiratory syncytial virus; PIV, parainfluenza virus.
Pathogens identified in COPD exacerbations
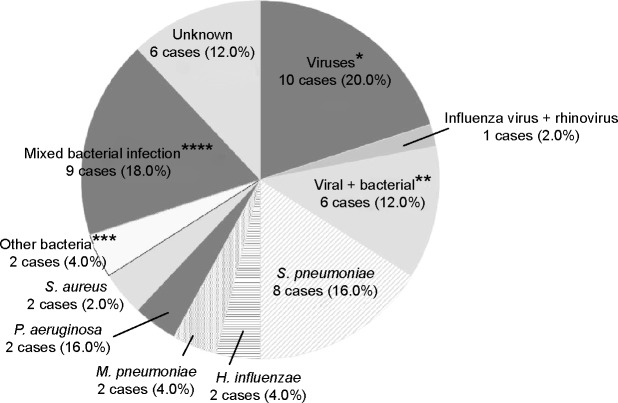
Pathogens identified in respiratory samples by comprehensive real-time PCR and conventional methods are listed in Table 2; percentages of pathogens identified are shown in Figure 2. Conventional methods identified the causative pathogens in only 26 (52%) of the 50 exacerbations, while comprehensive real-time PCR together with conventional methods made the identification in 44 (88%) of the 50 exacerbations. Twenty-eight (56%) were attributed to a single-microbe infection, while 16 (32%) were considered polymicrobial infections. The most commonly identified microorganisms were S. pneumoniae, H. influenzae, and FLU (26%, 18%, and 16% of exacerbations, respectively). Among the 8 of 13 S. pneumoniae-related exacerbations that were culture positive, most strains were covered by pneumococcal polysaccharide vaccine (types 3 (2), 11A (2), 15B (1), 16F (1), 33B (1), and 33F (1)) and were not penicillin resistant. However, most strains detected were macrolide resistant (75%).
Table 2.
Identity of pathogens in patients with COPD exacerbation
| Pathogen | Comprehensive real-time PCR
|
Conventional method | Total | ||
|---|---|---|---|---|---|
| NPS | Sputum | Total | |||
| Pathogens detected from samples | 25 (50) | 24 (48) | 32 (64) | 26 (52) | 44 (88) |
| Single pathogen | 21 (42) | 19 (38) | 24 (48) | 21 (42) | 28 (56) |
| Mixed pathogen | 4 (8) | 5 (10) | 8 (16) | 5 (10) | 16 (32) |
| Viral pathogen* | 15 (30) | 9 (18) | 16 (32) | 4 (8) | 17 (34) |
| Influenza virus | 7 (14) | 4 (8) | 7 (14) | 5 (10) | 8 (16) |
| Influenza virus A | 6 (12) | 3 (6) | 6 (14) | 4 (8) | 7 (14) |
| Influenza virus B | 1 (2) | 1 (2) | 1 (2) | 1 (2) | 1 (2) |
| Rhinovirus | 2 (4) | 0 (2) | 2 (4) | ND | 2 (4) |
| Respiratory syncytial virus | 4 (8) | 2 (4) | 4 (8) | ND | 4 (8) |
| Respiratory syncytial virus subgroup A | 2 (4) | 1 (2) | 2 (4) | ND | 2 (4) |
| Respiratory syncytial virus subgroup B | 2 (4) | 1 (2) | 2 (4) | ND | 2 (4) |
| Parainfluenza virus | 3 (6) | 3 (6) | 4 (8) | ND | 4 (8) |
| Parainfluenza virus type 1 | 1 (2) | 1 (2) | 1 (2) | ND | 1 (2) |
| Parainfluenza virus type 3 | 2 (4) | 2 (4) | 3 (6) | ND | 3 (6) |
| Bacterial pathogen* | 13 (26) | 17 (34) | 21 (42) | 23 (46) | 22 (44) |
| Streptococcus pneumoniae | 7 (14) | 11 (22) | 11 (22) | 10 (20) | 13 (26) |
| Haemophilus influenzae | 3 (6) | 8 (16) | 9 (18) | 2 (4) | 9 (18) |
| Mycoplasma pneumoniae | 3 (6) | 1 (2) | 4 (8) | 0 (0) | 4 (8) |
| Chlamydophila pneumoniae | 1 (2) | 0 (0) | 1 (2) | 0 (0) | 1 (2) |
| Staphylococcus aureus | ND | ND | ND | 5 (10) | 5 (10) |
| Enteric/nonfermenting Gram-negative bacilli** | ND | ND | ND | 11 (22) | 11 (22) |
| No pathogen detected | 25 (50) | 26 (52) | 18 (36) | 24 (48) | 6 (12) |
Notes: Data are presented as the number (percentage) of patients.
Some overlap exists.
Enteric or nonfermenting Gram-negative bacilli, Pseudomonas aeruginosa (4), Enterobacter cloacae (3), Klebsiella pneumoniae (1), Serratia marcescens (1), Proteus mirabilis (1), Acinetobactor baumanii (1), Stenotrophomonas maltophilia (1).
Abbreviations: COPD, chronic obstructive pulmonary disease; ND, not determined; NPS, nasopharyngeal swab; PCR, polymerase chain reaction.
Figure 2.
Percentages of pathogens underlying COPD exacerbations.
Notes: *Viruses were as follows: Parainfluenza virus, four cases (8%); influenza virus, three cases (6%); RSV, three cases (6%). **Combined virus and bacterial infections were as follows: influenza virus + Streptococcus pneumoniae, two cases (4%); influenza virus + Mycoplasma pneumoniae, one case (1%); influenza virus + Haemophilus influenzae + M. pneumoniae, one case (1%); RSV + S. pneumoniae, one case (2%); rhinovirus + S. pneumoniae + H. influenzae, one case (2%). ***Mixed bacterial infections were as follows: S. pneumoniae + H. influenzae, one case (2%); Pseudomonas aeruginosa + Citrobactor freundii, one case (2%); P. aeruginosa + Staphylococcus aureus, one case (2%); Enterobacter cloacae + S. aureus, one case (2%); Stenotrophomonas maltophilia + Acinetobacter baumannii, one case (2%); E. cloacae + H. influenzae, one case (2%); S. aureus + H. influenzae, one case (2%); Klebsiella pneumoniae + H. influenzae, one case (2%); E. cloacae + C. pneumoniae, one case (2%). ****Other bacteria were as follows: Proteus mirabilis, one case (2%); Serratia marcesens, one case (2%).
Abbreviations: COPD, chronic obstructive pulmonary disease; RSV, respiratory syncytial virus.
Comparison of WBC, CRP levels among five groups in COPD exacerbation
COPD exacerbations were grouped by causative pathogen (Figure 3). Median WBC and CRP values differed significantly between groups (P<0.01 and P<0.005, respectively). WBC was significantly higher in “the S. pneumoniae group” than “the viral group” (P=0.0005), although WBC in “the S. pneumoniae group” did not differ from findings in any other groups. CRP was significantly higher in “the S. pneumoniae group” and “the mixed viral and bacterial infection group” than “the viral group” (P=0.0012 and P<0.0002, respectively).
Figure 3.
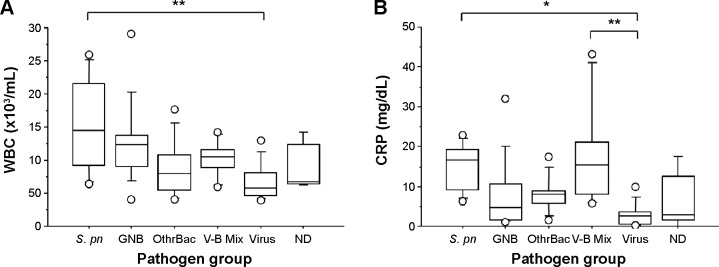
Box-and-whisker plots showing (A) white blood cell (WBC) count and (B) C-reactive protein (CRP) comparing the Streptococcus pneumoniae (S. pn) group, including isolated only S. pneumoniae infection (n=8) with Gram-negative bacilli (GNB) group, including enteric and nonfermenting GNB (n=11); the other bacteria (OthrBac) group, including Haemophilus influenzae, Mycoplasma pneumoniae, and Staphylococcus aureus (n=8); the V-B Mix group, including mixed viral and bacterial infection (n=6); the viral group (n=11); and the not determined (ND) group, in which no pathogen was detected (n=6). Data are presented as median (interquartile range).
Notes: *P<0.005; **P<0.0005.
Risk factors for prolonged hospitalization for COPD exacerbation
We analyzed risk factors for prolonged hospitalization. Univariate and subsequent multivariate analyses were carried out for age, sex, albumin, WBC, and each microorganism (Tables 3 and 4). Respiratory viruses, S. pneumoniae, and H. influenzae were not independent variables related to prolonged hospitalization. However, COPD exacerbation related to Gram-negative bacilli (including enteric and nonfermenting organisms), as well as prior treatment with home oxygen therapy were independent variables related to prolonged hospitalization (OR=5.25, P=0.042; and OR=6.23, P=0.012, respectively).
Table 3.
Risk factors for prolonged hospitalization associated with COPD exacerbation
| Variable | Hospitalization
|
P-value | |
|---|---|---|---|
| ≤14 days* (n=30) | >14 days** (n=20) | ||
| Age >75 years old | 18 (60) | 10 (50) | 0.9621*** |
| Male sex | 30 (100) | 17 (85) | 0.9999**** |
| Albumin <3.5 mg/dL | 13 (43.3) | 8 (40) | 0.8642*** |
| WBC >10,000/µL | 14 (46.7) | 9 (45) | 0.7435*** |
| Home oxygen therapy | 9 (30) | 12 (60) | 0.008*** |
| Streptococcus pneumoniae | 9 (30) | 4 (20) | 0.746**** |
| Haemophilus influenzae | 7 (23.3) | 2 (10) | 0.4589**** |
| Enteric or nonfermentig Gram-negative bacilli | 4 (13.3) | 7 (35) | 0.0406**** |
| Influenza virus | 6 (20) | 2 (10) | 0.6939**** |
Notes: Data are presented as the number (percentage) of patients.
Hospitalization ≤14 days, including outpatients;
hospitalization >14 days, including patients who died;
chi-squared test;
Fisher’s exact test.
Abbreviations: COPD, chronic obstructive pulmonary disease; WBC, white blood cell.
Table 4.
Multivariate analysis of risk factors for prolonged hospitalization associated with COPD exacerbation
| Variable | Logistic multivariate analysis*
|
P-value |
|---|---|---|
| Odds ratio (95% confidence interval) | ||
| Age >75 years old | 1.055 (0.260–4.275) | 0.9404 |
| Male sex | 1.301 (0.029–57.586) | 0.8919 |
| Albumin <3.5 mg/dL | 0.898 (0.216–3.731) | 0.8827 |
| WBC >10,000/µL | 1.548 (0.391–6.132) | 0.5341 |
| Home oxygen therapy | 6.232 (1.506–25.784) | 0.0115 |
| Enteric or nonfermenting Gram-negative bacilli | 5.249 (1.059–26.027) | 0.0424 |
Note:
Logistic multivariate analysis performed using age, male sex, albumin, home oxygen therapy, WBC, and enteric or nonfermenting Gram-negative bacilli.
Abbreviations: COPD, chronic obstructive pulmonary disease; WBC, white blood cell.
Discussion
Our study of comprehensive real-time PCR in patients with COPD exacerbations yielded four major findings. First, combining real-time PCR with conventional methods improved ability to detect infectious etiologies of COPD exacerbations including both bacteria and respiratory viruses. Second, a clear seasonal distribution of COPD exacerbations was observed. Third, COPD exacerbations caused by S. pneumoniae showed higher WBC and CRP values than those caused by viruses. Fourth, COPD exacerbations caused by Gram-negative bacilli were associated significantly with prolonged hospitalization.
A previous report concluded that use of real-time PCR in combination with conventional methods could effectively identify the infectious etiology of COPD exacerbation, particularly improving detection of viruses.9,10 In the present study, we extended application of real-time PCR together with conventional methods to include not only viruses and atypical bacteria but also typical respiratory bacterial pathogens. This increased the infectious etiology detection rate for COPD exacerbation to 88% (44 of 50 exacerbations), while conventional methods could determine the etiology in only 52% (26 of 50 exacerbations). We believe that real-time PCR methods can compensate for shortcomings of conventional methods, such as by extending the range of microorganisms covered, specifically to include respiratory viruses and atypical bacteria. Additionally, in the samples obtained from patients recently taking antibiotics, bacteria still could be detected.19 While cell-culture and serologic methods indicated that viral infections caused approximately 20% of COPD exacerbations,9,20–22 real-time PCR identified respiratory viruses in 16 of the 50 exacerbations that we studied (32%). Viruses identified included FLU, RSV, PIV, and RV, as result consistent with previous studies (Table 2).9 Earlier investigation found M. pneumoniae and C. pneumoniae to explain up to 14% and 5% of COPD exacerbation, respectively.23,24 Although real-time PCR has an advantage in detecting atypical bacteria, we observed relatively low percentage of such atypical pathogens in COPD exacerbations (Table 2). We believe that during the study period only low-level endemic infections prevailed in our locality. Six bacteria, including S. pneumoniae and H. influenza among others, could be detected by real-time PCR in our present study. Among these, H. influenzae was detected more frequently by real-time PCR than by conventional sputum culture, possibly because prior antibiotics use in some patients decreased bacterial numbers. In contrast, S. aureus, as well as P. aeruginosa and other Gram-negative bacilli, were detected frequently by conventional sputum culture but were not covered by available materials for our real-time PCR. Thus, the methods complemented on another. Another advantage of simultaneously using both methods was ability to detect polymicrobial exacerbations, which we could characterize in 16 instances (32%).
Relatively few reports have dealt with seasonal patterns of COPD exacerbations.25 A recent European study demonstrated peak incidence of COPD exacerbations in winter, with a nadir during summer.25 We believe that the present study may be the first to identify seasonal patterns of CODP exacerbations in Japan, which proved similar to the European pattern. The peak of COPD exacerbations in winter were associated with a rise in both bacterial and viral infection, including S. pneumoniae and FLU, suggesting the potential for effective prevention by immunization against these microorganisms.
A recent study found that COPD exacerbations caused by viral infection were significantly associated with high CRP, suggesting a link between infectious etiology of COPD exacerbation and inflammatory biomarkers.12,13 In contrast, our S. pneumoniae group showed significantly higher WBC and CRP than our viral group. In our study, S. pneumoniae infection was in two of three fatal cases. Additional study is needed to determine how closely highly elevated inflammatory biomarkers in COPD exacerbation caused by S. pneumoniae are associated with disease severity and mortality.
Prolonged hospitalization to treat COPD exacerbation often contributes to physical deterioration and medical economic burdens.11 The present study indicated that the COPD exacerbations caused by enteric or nonfermenting Gram-negative bacilli were significantly associated with prolonged hospitalization. The association of Gram-negative bacilli causing COPD exacerbation with prolongation of hospitalization might result from development of selective pressure in patients with COPD from their frequent use of antibiotics, favoring infection by antibiotic-resistant Gram-negative bacilli. Further, airway clearance mechanisms are impaired in COPD, allowing ordinarily nonvirulent bacteria to colonize the lower respiratory mucosa. Such colonization could cause a chronic inflammatory response and progressive deterioration in airway defenses, increasing severity of the exacerbation to prolong hospitalization.26–28
We acknowledge limitations of the present study. First, a relatively small number of samples were evaluated. Second, the limited number of patients precluded meaningful comparison between pneumonic and nonpneumonic exacerbations of COPD. Third, since the study period was relatively short, results were influenced by annual variation in incidence of specific infections. Accordingly, a larger population examined over a longer period should be analyzed.
In conclusion, this study supports the usefulness of combining comprehensive real-time PCR with conventional methods to determine infectious etiologies of COPD exacerbations. We also identified Gram-negative bacilli and requirement of home oxygen therapy as risk factors for prolonged hospitalization to treat COPD exacerbations.
Acknowledgments
We wish to thank all the hospital staff at The Jikei University Hospital, The Jikei University Daisan Hospital, and Toranomon Hospital, Tokyo, Japan. This work was supported financially by Meiji Seika Pharma and Daiichi-Sankyo.
Footnotes
Author contributions
K Shimizu and YY involved in conception and design of research; K Shimizu, YY, NS, MM, NC, and KU performed experiments; K Shimizu, YY, MM, NC, KU, and KN analyzed data; K Shimizu and YY interpreted results of experiments; K Shimizu prepared figure; K Shimizu and YY drafted manuscript; K Shimizu, YY, MM, NC, KU, HU, SH, NS, TK, HW, SI, NT, SM, JK, TN, HH, MK, K Saito, JA, YK, KN, K Kishi, and K Kuwano approved final version of manuscript; K Shimizu and YY drafted manuscript; K Shimizu, YY, MM, NC, KU, HU, SH, NS, TK, HW, SI, NT, SM, JK, TN, HH, MK, K Saito, JA, YK, KN, K Kishi, and K Kuwano edited and revised manuscript. All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Niewoehner DE. The impact of sever exacerbation on quality of life and the clinical course of chronic obstructive pulmonary disease. Am J Med. 2006;119:38–45. doi: 10.1016/j.amjmed.2006.08.006. [DOI] [PubMed] [Google Scholar]
- 2.Sapey E, Stockley RA. COPD exacerbations 2: aetiology. Thorax. 2006;61:250–258. doi: 10.1136/thx.2005.041822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sethi S, Sethi R, Eschberger K, et al. Airway bacterial concentrations and exacerbations of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2007;176:356–361. doi: 10.1164/rccm.200703-417OC. [DOI] [PubMed] [Google Scholar]
- 4.Mohab A, Chandra S, Agawal D, et al. Prevalence of viral infection detected by PCR and RT-PCR in patients with acute exacerbation of COPD: a systematic review. Respirology. 2010;15:536–542. doi: 10.1111/j.1440-1843.2010.01722.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hirschmann JV. Do bacteria cause exacerbations of COPD? Chest. 2000;118:193–203. doi: 10.1378/chest.118.1.193. [DOI] [PubMed] [Google Scholar]
- 6.Templeton KE, Scheltinga SA, van den Eeden WC, et al. Improved diagnosis of the etiology of community-acquired pneumonia in adults. Clin Infect Dis. 2005;41:345–351. doi: 10.1086/431588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lieberman D, Shimoni A, Shemer-Avni Y, et al. Respiratory viruses in adults with community-acquired pneumonia. Chest. 2010;138:811–816. doi: 10.1378/chest.09-2717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.De Serres G, Lampron N, La Forge J, et al. Importance of viral and bacterial infections in chronic obstructive pulmonary disease exacerbations. J Clin Virol. 2009;46:129–133. doi: 10.1016/j.jcv.2009.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hutchinson AF, Ghimire AK, Thompson MA, et al. A community-based, time-matched, case-control study of respiratory viruses and exacerbations of COPD. Resp Med. 2007;101:2472–2481. doi: 10.1016/j.rmed.2007.07.015. [DOI] [PubMed] [Google Scholar]
- 10.Perotin JM, Dury S, Renois F, et al. Detection of multiple viral and bacterial infections in acute exacerbation of chronic obstructive pulmonary disease: a pilot prospective study. J Med Virol. 2013;85:866–873. doi: 10.1002/jmv.23495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Miravitilles M, Murio C, Guerrero T, Gisbert R, DAFNE Study Group Pharmacoeconomic evaluation of acute exacerbations of chronic bronchitis and COPD. Chest. 2002;121:1449–1455. doi: 10.1378/chest.121.5.1449. [DOI] [PubMed] [Google Scholar]
- 12.Papi A, Bellettato CM, Braccioni F, et al. Infections and airway inflammation in chronic obstructive pulmonary disease severe exacerbations. Am J Respir Crit Care Med. 2006;173:1114–1121. doi: 10.1164/rccm.200506-859OC. [DOI] [PubMed] [Google Scholar]
- 13.Clark TW, Medina MJ, Batham S, et al. C-reactive protein level and microbial aetiology in patients hospitalised with acute exacerbation of COPD. Eur Respir J. 2015;45(1):76–86. doi: 10.1183/09031936.00092214. [DOI] [PubMed] [Google Scholar]
- 14.Global Initiative for Chronic Obstructive Lung Disease . Global Strategy for the Diagnosis, Management and Prevention of Chronic Obstructive Lung Disease. Claude Lenfant, MD: National Heart, Lung, and Blood Institute; 2011. (NHLBI/WHO Workshop Report). [PubMed] [Google Scholar]
- 15.Morozumi M, Nakayama E, Iwata S, et al. Simultaneous detection of pathogens in clinical samples from patients with community-acquired pneumonia by real-time PCR with pathogen-specific molecular beacon probes. J Infect Chemother. 2006;44:1440–1446. doi: 10.1128/JCM.44.4.1440-1446.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hamano-Hasegawa K, Morozumi M, Nakayama E, et al. Comprehensive detection of causative pathogens using real-time PCR to diagnose pediatric community-acquired pneumonia. J Infect Chemother. 2008;14:424–432. doi: 10.1007/s10156-008-0648-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Okada T, Morozumi M, Sakata H, et al. A practical approach estimating etiologic agents using real-time PCR in pediatric inpatients with community-acquired pneumonia. J Infect Chemother. 2012;18:832–840. doi: 10.1007/s10156-012-0422-7. [DOI] [PubMed] [Google Scholar]
- 18.Murray PR, Baron EJ, Jorgensen JH, et al. Manual of Clinical Microbiology. 9th ed. Washington, DC: American Society of Microbiology; 2007. [Google Scholar]
- 19.Theerthakarai R, El-Halees W, Ismail M, et al. Nonvalue of the initial microbiological studies in the management of nonsevere community-acquired pneumonia. Chest. 2001;119:181–184. doi: 10.1378/chest.119.1.181. [DOI] [PubMed] [Google Scholar]
- 20.Seemungal TA, Harper-Owen R, Bhowmik A, et al. Respiratory viruses, symptoms and inflammatory markers in acute exacerbations and stable chronic obstructive disease. Am J Respir Crit Care Med. 2001;164:1618–1623. doi: 10.1164/ajrccm.164.9.2105011. [DOI] [PubMed] [Google Scholar]
- 21.Glezen WP, Greenberg SB, Atmar RL, Piedra PA, Couch RB. Impact of respiratory virus infections on persons with chronic underlying conditions. JAMA. 2000;283(4):499–505. doi: 10.1001/jama.283.4.499. [DOI] [PubMed] [Google Scholar]
- 22.Greenberg SB, Cadena A, Lin J, et al. Respiratory viral infections in adults with and without chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2000;162:167–173. doi: 10.1164/ajrccm.162.1.9911019. [DOI] [PubMed] [Google Scholar]
- 23.Lieberman D, Lieberman D, Yaakov BM, et al. Infectious etiologies in acute exacerbations of COPD. Diagn Microbiol Infect Dis. 2001;40:95–102. doi: 10.1016/s0732-8893(01)00255-3. [DOI] [PubMed] [Google Scholar]
- 24.Beaty CD, Grayston JT, Wang SP. Chlamydia pneumoniae, strain TWAR, infections in patients with chronic obstructive pulmonary disease. Am Rev Respir Dis. 1991;16:1408–1410. doi: 10.1164/ajrccm/144.6.1408. [DOI] [PubMed] [Google Scholar]
- 25.Klaus FR, Leonardo MF, Vogelmeier C, et al. Seasonal distribution of COPD exacerbations in the Prevention of Exacerbations with Tiotropium in COPD trial. Chest. 2013;143:711–719. doi: 10.1378/chest.12-1277. [DOI] [PubMed] [Google Scholar]
- 26.Wilson R. A vicious circle hypothesis operating during infective exacerbations of chronic bronchitis. Monaldi Arch Chest Dis. 1994;49:159–164. [PubMed] [Google Scholar]
- 27.Miravitlles M, Calle M, Soler-Cataluña JJ. Clinical phenotypes of COPD: identification, definition and implications for guidelines. Arch Broncopneumol. 2012;48:86–98. doi: 10.1016/j.arbres.2011.10.007. [DOI] [PubMed] [Google Scholar]
- 28.Sethi S, Mallia P, Johnston SL. New paradigms in the pathogenesis of chronic obstructive pulmonary disease II. Proc Am Thorac Soc. 2009;6:532–534. doi: 10.1513/pats.200905-025DS. [DOI] [PubMed] [Google Scholar]