Abstract
Purpose
This study determined (1) how many vessels (i.e. the vessel sampling) are needed to reliably characterize the bulbar conjunctival microvasculature and (2) if characteristic information can be obtained from the distribution histogram of the blood flow velocity and vessel diameter.
Methods
Functional Slit-lamp Biomicroscope was used to image hundreds of venules per subject. The bulbar conjunctiva in 5 healthy human subjects was imaged on 6 different locations in the temporal bulbar conjunctiva. The histograms of the diameter and velocity were plotted to examine whether the distribution was normal. Standard errors were calculated from the standard deviation and vessel sample size. The ratio of the standard error of the mean over the population mean was used to determine the sample size cutoff. The velocity was plotted as a function of the vessel diameter to display the distribution of the diameter and velocity.
Results
The results showed that the sampling size was approximately 15 vessels, which generated a standard error equivalent to 15% of the population mean from the total vessel population. The distributions of the diameter and velocity were unimodal, but also somewhat positively skewed and not normal. The blood flow velocity was related to the vessel diameter (r =0.23, P < 0.05).
Conclusion
This was the first study to determine the sampling size of the vessels and the distribution histogram of the blood flow velocity and vessel diameter, which may lead to a better understanding of the human microvascular system of the bulbar conjunctiva.
Keywords: bulbar conjunctiva, microvasculature, morphology, hemodynamics, functional slit lamp biomicroscopy (FSLB), sample size, distribution histogram
The bulbar conjunctival microvasculature is a terminal blood vessel bed encased in a semi-transparent membrane above the white sclera. This easily observable site allows real time, in-vivo, and non-invasive measurements, which may reveal indicative information about microvascular dysfunction and vasculopathies located in deeply enclosed regions like the brain1,2 and kidney.3 Valuable characterizations have been obtained from this vasculature for ocular, systemic and cerebral diseases such as dry eye syndrome,4 sickle cell anemia,5-7 diabetes,8,9 and Alzheimer's disease.2
The conjunctival microvasculature is an orderly network of branching and anastomosing capillaries, arterioles, and venules. The network is evenly distributed with a ∼1:2 arteriole to venule ratio. The vessel diameters are between 5 and 70 μm.3,10,11 The blood flow velocities measured in venules and arterioles range from 0.52 to 3.26 mm/s.3,10-12 A variety of instruments including devices like slit lamp biomicroscope based systems8,10-12 have been used to image the conjunctival microvascular network.9,10,13-15 In previous studies, the vessel sample size varied largely from 2-12 microvessels per eye12,14-16 to 50 vessels per eye.3 Small vessel sample sizes (e.g. 2-12 vessels) may not represent the structure and blood flow characteristics of the vast number of bulbar conjunctiva blood vessels. Additionally, with small sample sizes, it may not be practical to study the velocity profile and diameter distribution, which depicts subtle morphological and hemodynamic changes. However, large vessel sample sizes may not be necessary especially when the measuring and image processing procedures are often time consuming.
For the bulbar conjunctival microvasculature to be eventually utilized as an image biomarker for detecting early abnormal vascular signs and subtle changes in the aforementioned diseases, the critical knowledge gap regarding vessel sampling and distribution profiles of blood flow velocity and vessel diameter need to be addressed. Although these alterations can be detected in a later disease stage or when more severe conditions appear using relatively small vessel sample sizes,3,7,17 the ability to detect subtle and specific changes in early stage of the disease may provide more information for etiology, progression, and patient care. In the present study, it was hypothesized that there is a sampling cutoff that could reliably represent the characterization of the conjunctival microvasculature. These microvasculature distribution profiles may provide characteristic information for both physiological and pathophysiological conditions. Jiang et al. recently demonstrated that a modified traditional slit-lamp biomicroscope named the Functional Slit-lamp Biomicroscope (FSLB) could be used to image hundreds of vessels on the bulbar conjunctiva of normal subjects.12 The aims of the present study were to (1) determine the sampling size of vessels needed to represent the overall blood vessel diameters and velocities and (2) determine whether the blood velocity distribution with diameter can provide additional characterization for the human microvasculature.
Methods and Materials
The FSLB imaging system has been described in a previous study.12 Briefly, a digital camera was adapted to a traditional slit-lamp. The camera has a Movie Crop Function (MCF) that uses a center portion of pixels on the camera chip to generate an equivalent of ∼7× magnification for high speed video recording at 60 frames per second (fps). With the built-in optical magnifications of up to 30× (set by a dial on the slit-lamp) the total magnification can reach ∼70× - ∼210×. The magnifications are high enough to capture the movement of red blood cells (RBC)/RBC clusters, which allow the blood flow velocity and vessel diameter to be measured. In the previous study, Jiang et al. demonstrated that red blood cell clusters can be clearly video recorded with a magnification of 70×, a relatively large field of view,12 which is similar to the field of view used by Cheung et al.3 Our system has a high speed of 60 frames per second for video recording when the MCF is on. These special functions are suitable for imaging vessel diameters and velocities of a large sample size of vessels.12 In the present study, MCF with the magnification of 70×, a field of view of 2.84 × 2.10 mm2, and an image size of 640 × 480 pixels was utilized. Six different locations of the temporal bulbar conjunctiva were imaged, which was ∼1 mm away from the limbus, covering a total area of ∼6 × 4 mm2 (Fig. 1).
Fig. 1. Human bulbar conjunctival microvasculature imaged using Functional Slit-lamp Biomicroscope (FSLB).
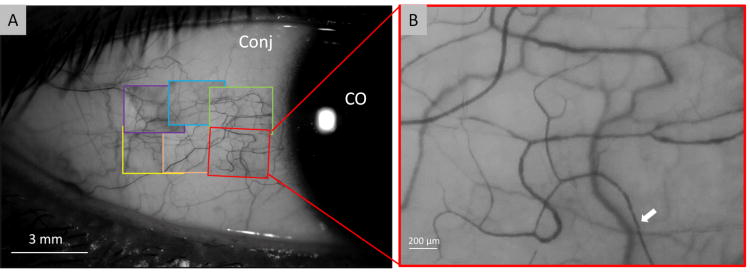
The temporal bulbar conjunctiva, about 1 mm away from the limbus, was imaged by the FSLB (A). Six locations were overlaid with the snapshots from acquired video clips and covered about 6 × 4 mm2 (B). Red blood cell clusters (white arrow) were shown in the enlarged field (B, magnificent 70 ×). Conj: conjunctiva. CO: Cornea.
This study was approved by the institutional review board for human research at the University of Miami. Each subject signed a consent form and was treated in accordance with the Declaration of Helsinki. A total of 5 subjects (3 males and 2 females, age: 36.2 ± 5.4 years old, mean ± standard deviation) were recruited. They were normal healthy subjects (i.e. without systemic or ocular diseases) who have never worn contact lenses.
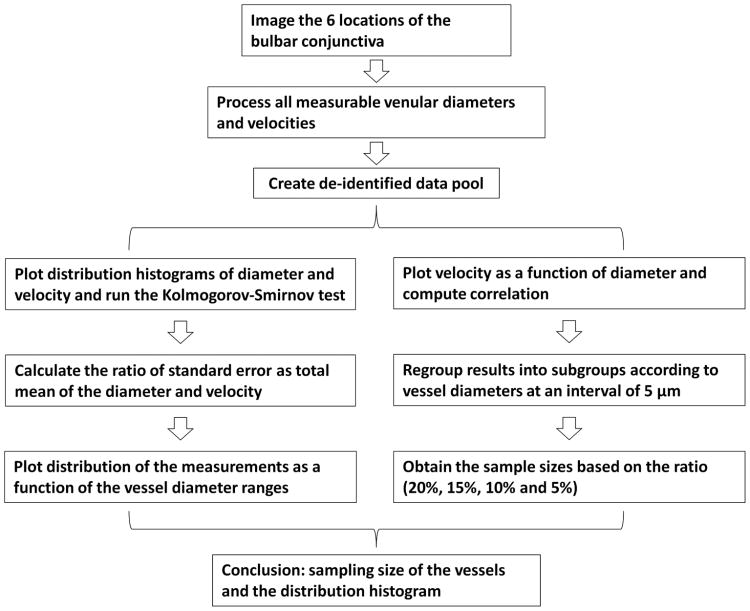
Custom software has been developed and utilized for the quantification of microvascular morphology and hemodynamics as described in a previous publication.12 The software was used to semi-automatically process the video clips to yield vessel diameters, lengths, vessel covering areas, and velocities for all measureable venules. Since blood flow velocity in the conjunctival pre-capillary arterioles can be impacted by the pulse,14 conjunctival post-capillary venules were used in previous studies10-13 and the present study. Venules were identified by observing the direction of blood flow. Typically, the venules are wider in diameter when compared to arterioles, and the blood flow enters a thicker venule from two branching venules. Due to the finite imaging field, some vessels could not be visually determined. In these rare cases, the ambiguous vessels were excluded from the data pool. The length between two bifurcations was defined as the vessel length. The vessel covering area (unit: μm2) was calculated as the vessel length multiplied by the vessel diameter. The measurement of blood flow velocity was based on the space-time image (STI) technique.10,12 Data acquisition, processing and sampling are illustrated in Fig. 2.
Fig. 2. Schematic diagram of data processing and sampling.
The histograms of the diameter and velocity were plotted to examine whether the distribution was normal or not by using the Kolmogorov-Smirnov test.18 The Kolmogorov-Smirnov test was conducted using a commercial program (XLSTAT, Addinsoft USA, New York, NY) to compute the diameter and velocity by using the estimated parameters of the normal distribution. The rejection level alpha was set to 0.05 for rejecting the hypothesis (H0) to show that the sample follows a normal distribution. To depict the profiles of the vessel diameter and blood flow velocity, all vessels were divided into sub-groups. The first group contained all vessels with a diameter < 20 μm and the last group contained any vessels with a diameter ≥ 50 μm. The vessels within diameters of 20 to 50 μm were divided with an arbitrary interval of 5 μm in vessel diameter. The percentage of the averaged vessel counts out of the total vessel count for each sub-group was calculated for the vessel count profile. Averaged blood flow velocities were used to plot the profiles of blood flow velocity as a function of vessel diameter ranges.
All data management and statistical analysis were done in Excel (ver. 2007, Microsoft, Redmond, WA). To determine the relations between the diameter and the blood flow velocity, Pearson's regression was used. Single factor analysis of variance (ANOVA) was used to analyze the differences among individuals and locations of each subject.
Results
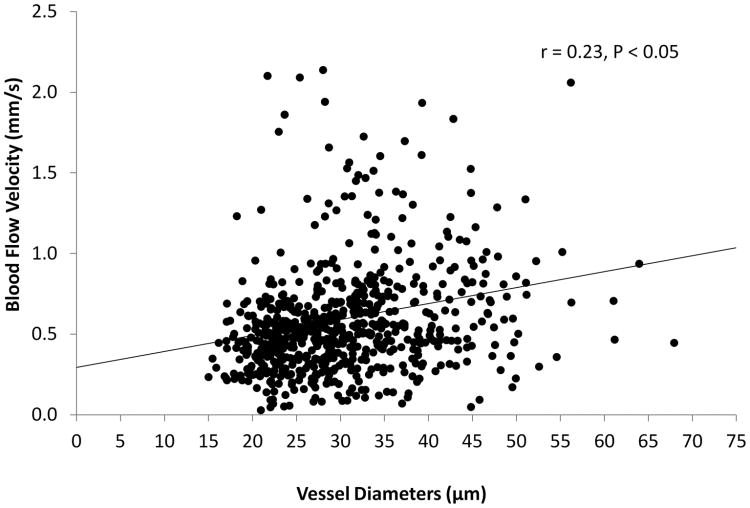
Averaged vessel diameter and velocity, obtained from a total of 767 vessel segments, was 30.4 ± 8.4 μm and 0.59 ± 035 μm, respectively. The analysis of variance indicated that there were significant differences in the blood flow velocity (P < 0.001) and vessel diameter (P < 0.001) among the subjects. Among the imaging locations, there were significant differences of the vessel diameter (P < 0.05) in 4 subjects and significant differences of the blood flow velocity (P < 0.05) in 2 subjects. The blood flow velocity was related to the vessel diameter (r =0.23, P < 0.05, Fig. 3). The ratios of standard error of the mean over the population mean for the diameter and velocity were similar (Fig. 4). The cutoff for the sample sizes of both diameter and velocity was listed in Table 1. The results showed that the sampling size was approximately 15 vessels, which generated a standard error equivalent to 15% of the measurement mean from the total vessel population (Fig. 4). The distributions of the diameter and velocity were unimodal but also somewhat positively skewed and does not follow the normal distribution (P<0.05) (Fig. 5 and 6).
Fig. 3. Relations between the blood flow velocity and vessel diameter.
The blood flow velocity and diameter were measured using FSLB. The blood flow velocity did not correlate with the vessel diameter.
Fig. 4. The ratios of the standard error of the mean over the population mean for measuring the diameter and velocity were plotted as a function of sample sizes.
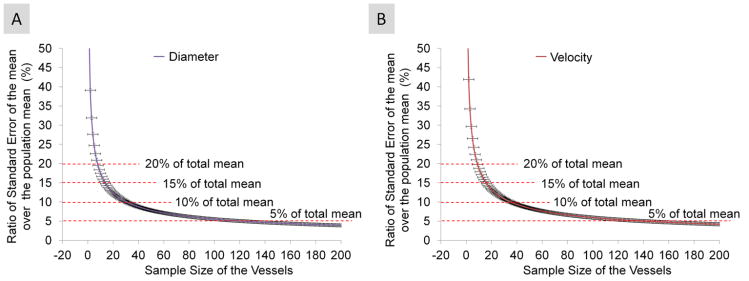
The results showed that the sampling size, approximately 15 vessels, generated a standard error equivalent to 15% of the population mean from the total vessel population for vessel diameter (A) and blood flow velocity (B). Bars = standard error.
Table 1. Sample size of vessels.
| Ratio* | Diameter | Velocity | Mean | SD |
|---|---|---|---|---|
| 20% | 8 | 8 | 8 | 0 |
| 15% | 15 | 14 | 15 | 1 |
| 10% | 32 | 28 | 30 | 3 |
| 5% | 117 | 101 | 109 | 11 |
Calculated as the ratio of standard error of the mean to the population mean.
Fig. 5. The histograms of vessel diameter and blood flow velocity.
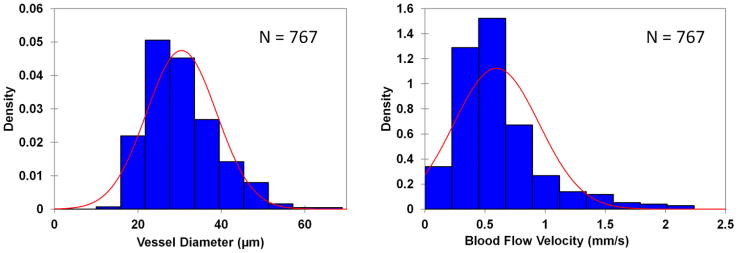
The bell-shaped distributions of vessel diameter (A) and velocity (B) showed a unimodal distribution that was also somewhat positively skewed. None of distributions followed the normal distribution as analyzed using Kolmogorov-Smirnov test, a statistical test for normal distribution.
Fig. 6. The distribution of blood flow velocity as a function of vessel diameter.
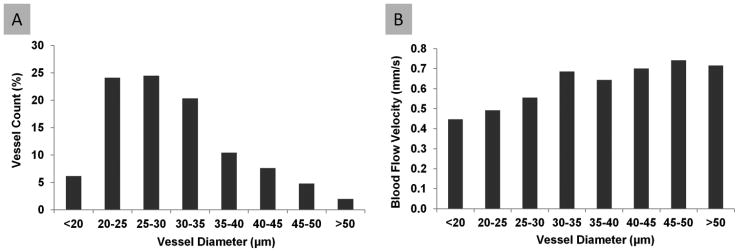
The vessel count (A) showed a unimodal profile as a function of vessel diameter. The blood flow velocity (B) showed a slight increase as the vessel diameter increased.
Discussion
Enclosed in a semi-transparent mucosal tissue and located above the white sclera, the bulbar conjunctival microvasculature stands out starkly from its surroundings. These features allow the region to be studied in-vivo and non-invasively for the characterization of ocular, systemic and cerebral vascular diseases. Some researchers have found differences in conjunctival microvessels using qualitative scales in diseased eyes.1,5,6,8,9,17,19 Cheung et al. used the computer-assisted intravital microscope (CAIM) and developed the Severity Index (SI), which is a qualitative, cumulative severity ranking system for microvascular abnormalities, to categorize the bulbar conjunctival microvascular alterations in type 1 diabetes.8 The SI was found to correlate with the glycated hemoglobin (A1c) levels of T1DM patients but not with the duration of the disease.8,9 Using the similar method, Cheung et al. also characterized conjunctival microvascular abnormalities of patients with sickle cell anemia,6,19 Alzheimer's disease1 and contact lens wear.5
Compared to the qualitative method (e.g. SI), the quantitative measurement of vessel diameter and blood flow velocities may be more sensitive for detecting earlier vasculopathic changes. However, a small vessel sample size may limit the ability to determine early changes of the conjunctival microvasculature, representing the disease of interest, especially when a small group of patients is studied. Although clinically significant differences were found in previous studies, in relation to the present study, it appears that small sample sizes of the vessels may induce sampling biases. If the tolerance of the error for estimating the total mean of the vessel population is 15%, the sample size of the vessel will need to have 15 vessels or more. When a relatively larger number of human subjects is used, the likelihood for differentiating the diseased eye from the normal eye might be possible with a small sample size of measured vessels on each eye.7 To characterize two different subtypes of sickle cell anemia, Wanek et al. used 2 or more blood vessels from each subject (total 42 patients). They divided vessels into large and small vessel categories based on diameter and found a difference of blood flow velocity between patients with different subtypes of the disease.7 In contrast, Cheung et al. used CAIM to measure the blood flow velocity in patients after simultaneous pancreas-kidney transplantation (SPK).3 Multiple video sequences were used and 30-50 vessels were averaged for the measurement. Cheung et al. was able to successfully document the improvement for conjunctival microcirculation in only 12 diabetic patients, 18 months after the SPK surgery, possibly due to the large sample size of measured vessels per eye.3 In the present study, nearly 200 vessels per eye were analyzed. The sampling of more vessels per eye may effectively reduce the study sample size of patients in clinical trials. It may be worth noting that the sampling numbers of vessels may vary for various diseased conditions, depending on the magnitude of the differences in diseased conditions.
Additionally, the bulbar conjunctival vessel network has a unique pattern which makes it possible to relocate the same vessels for repeated measurements and follow-up. When the same vessels are repeatedly measured, the changes may be easily identified even with a small sample size of the vessels. Jiang et al. imaged 5 vessels per subject in 6 human subjects12 to demonstrate the microvascular response to contact lens wear. The vessel diameters and blood flow velocities were increased after wearing contact lenses for 6 hours. Cheung et al. repeatedly measured the same set of vessels (30-50 vessels) in a longitudinal study and demonstrated improved microvasculature in patients after simultaneous pancreas-kidney transplantation.3 However, a registered vessel set might not be possible for the majority of clinical cross-sectional studies. In the present study, it was demonstrated that a random selection of blood vessels may reliably represent the hemodynamic features of the conjunctival microvasculature. The cutoffs found in the present study for reliably measuring the microvasculature are higher than the sampled vessel numbers in most previous studies that ranged from 2-12 blood vessels.7,10-12,15 It could be suggested that more vessels should be imaged to differentiate diseased condition from healthy controls. Analysis of the standard errors from the mean of the total number of vessels as a function of the sampled number of vessels provided the evidence for the sample size selection. The apparent optimized sample size of the vessel was ∼15 vessels for measuring both the vessel diameter and blood flow velocity, when the tolerable sampling error of the total mean was set to 15%. Obviously, a larger sample size of the vessels will be needed if the tolerated standard error is smaller as showed in the present study.
The distributions of the diameter and velocity were unimodal but also somewhat positively skewed and did not follow the normal distribution. Further analysis of the distribution of velocity with vessel diameters could provide additional characteristics about microvascular alternation and vasculopathies beyond what could be obtained from the averaged vessel diameter and blood flow velocity. First, the distribution may enable researchers to identify blood flow velocity changes in specific vessels with a certain vessel diameter range. Second, some diseases may only affect vessels with certain diameters. The mean of the velocity may indicate overall changes while its distribution could further point to these specific vessels. Analyzing the distribution of the vessel diameter, velocity and other microvascular parameters could provide additional information, even if the number of study subject is small.
In summary, the present study was the first study conducted to determine how many blood vessels (i.e. the sample size) was needed to reliably characterize the microvasculature on the bulbar conjunctiva. Additional information can be found from the blood flow velocity distribution with vessel diameter.
Acknowledgments
Grant/financial support: The activity has been supported by Howard Hughes Medical Institute (HHMI) High School Scholars Research Program (LW). The work has been also supported in part by the research grants NIH Center Grant P30 EY014801, a grant from Research to Prevent Blindness (RPB) and Walter G. Ross Chair in Ophthalmic Research (VLP).
Footnotes
Commercial relationship: None
Financial Disclosures: All authors have no proprietary interest in any materials or methods.
References
- 1.Chew SH, Meighan Smith TM, Cheung AT. Alzheimer's disease: more than amyloid. Clin Hemorheol Microcirc. 2010;46:69–73. doi: 10.3233/CH-2010-1356. [DOI] [PubMed] [Google Scholar]
- 2.Smith MM, Chen PC, Li CS, Ramanujam S, Cheung AT. Whole blood viscosity and microvascular abnormalities in Alzheimer's Disease. Clin Hemorheol Microcirc. 2009;41:229–239. doi: 10.3233/CH-2009-1174. [DOI] [PubMed] [Google Scholar]
- 3.Cheung AT, Perez RV, Chen PC. Improvements in diabetic microangiopathy after successful simultaneous pancreas-kidney transplantation: a computer-assisted intravital microscopy study on the conjunctival microcirculation. Transplantation. 1999;68:927–932. doi: 10.1097/00007890-199910150-00005. [DOI] [PubMed] [Google Scholar]
- 4.Rodriguez JD, Johnston PR, Ousler GW, III, Smith LM, Abelson MB. Automated grading system for evaluation of ocular redness associated with dry eye. Clin Ophthalmol. 2013;7:1197–1204. doi: 10.2147/OPTH.S39703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cheung AT, Hu BS, Wong SA, et al. Microvascular abnormalities in the bulbar conjunctiva of contact lens users. Clin Hemorheol Microcirc. 2012;51:77–86. doi: 10.3233/CH-2011-1513. [DOI] [PubMed] [Google Scholar]
- 6.Cheung AT, Miller JW, Craig SM, et al. Comparison of real-time microvascular abnormalities in pediatric and adult sickle cell anemia patients. Am J Hematol. 2010;85:899–901. doi: 10.1002/ajh.21853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wanek J, Gaynes B, Lim JI, Molokie R, Shahidi M. Human bulbar conjunctival hemodynamics in hemoglobin SS and SC disease. Am J Hematol. 2013;88:661–664. doi: 10.1002/ajh.23475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cheung AT, Tomic MM, Chen PC, et al. Correlation of microvascular abnormalities and endothelial dysfunction in Type-1 Diabetes Mellitus (T1DM): a real-time intravital microscopy study. Clin Hemorheol Microcirc. 2009;42:285–295. doi: 10.3233/CH-2009-1199. [DOI] [PubMed] [Google Scholar]
- 9.Devaraj S, Cheung AT, Jialal I, et al. Evidence of increased inflammation and microcirculatory abnormalities in patients with type 1 diabetes and their role in microvascular complications. Diabetes. 2007;56:2790–2796. doi: 10.2337/db07-0784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shahidi M, Wanek J, Gaynes B, Wu T. Quantitative assessment of conjunctival microvascular circulation of the human eye. Microvasc Res. 2010;79:109–113. doi: 10.1016/j.mvr.2009.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Koutsiaris AG, Tachmitzi SV, Batis N, et al. Volume flow and wall shear stress quantification in the human conjunctival capillaries and post-capillary venules in vivo. Biorheology. 2007;44:375–386. [PubMed] [Google Scholar]
- 12.Jiang H, Zhong J, Debuc DC, et al. Functional slit lamp biomicroscopy for imaging bulbar conjunctival microvasculature in contact lens wearers. Microvasc Res. 2014;92:62–71. doi: 10.1016/j.mvr.2014.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cheung AT, Harmatz P, Wun T, et al. Correlation of abnormal intracranial vessel velocity, measured by transcranial Doppler ultrasonography, with abnormal conjunctival vessel velocity, measured by computer-assisted intravital microscopy, in sickle cell disease. Blood. 2001;97:3401–3404. doi: 10.1182/blood.v97.11.3401. [DOI] [PubMed] [Google Scholar]
- 14.Koutsiaris AG, Tachmitzi SV, Papavasileiou P, et al. Blood velocity pulse quantification in the human conjunctival pre-capillary arterioles. Microvasc Res. 2010;80:202–208. doi: 10.1016/j.mvr.2010.05.001. [DOI] [PubMed] [Google Scholar]
- 15.Koutsiaris AG, Tachmitzi SV, Batis N. Wall shear stress quantification in the human conjunctival pre-capillary arterioles in vivo. Microvasc Res. 2013;85:34–39. doi: 10.1016/j.mvr.2012.11.003. [DOI] [PubMed] [Google Scholar]
- 16.Wanek J, Gaynes B, Lim JI, Molokie R, Shahidi M. Human bulbar conjunctival hemodynamics in hemoglobin SS and SC disease. Am J Hematol. 2013;88:661–664. doi: 10.1002/ajh.23475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cheung AT, Ramanujam S, Greer DA, Kumagai LF, Aoki TT. Microvascular abnormalities in the bulbar conjunctiva of patients with type 2 diabetes mellitus. Endocr Pract. 2001;7:358–363. doi: 10.4158/EP.7.5.358. [DOI] [PubMed] [Google Scholar]
- 18.Marsaglia G, Tsang WW, Wang J. Evaluating Kolmogorov's distribution. Journal of statistical software. 2003;8(18):1–4. [Google Scholar]
- 19.Cheung AT, Miller JW, Miguelino MG, et al. Exchange transfusion therapy and its effects on real-time microcirculation in pediatric sickle cell anemia patients: an intravital microscopy study. J Pediatr Hematol Oncol. 2012;34:169–174. doi: 10.1097/MPH.0b013e31823c27ef. [DOI] [PMC free article] [PubMed] [Google Scholar]